Abstract
Nuclear factor-κB (NF-κB) plays a key role in inflammation, which is involved in the development of cerebral vasospasm after subarachnoid hemorrhage (SAH). In the present study, we assessed the potential role of NF-κB in regulation of cerebral vasospasm. Nuclear factor-κB DNA-binding activity was measured in cultured vascular smooth muscle cells (VSMCs) treated with hemolysate and pyrrolidine dithiocarbamate (PDTC, 80 μmol/L), an inhibitor of NF-κB. Forty-two rabbits were divided into three groups: control, SAH, and PDTC groups (n = 14 for each group). The caliber of the basilar artery was evaluated. Nuclear factor-κB DNA-binding activity and the gene expression levels of cytokines and adhesion molecules in the basilar artery were measured. Immunohistochemical study was performed to assess the expression and localization of tumor necrosis factor (TNF)-α, intercellular adhesion molecule (ICAM)-1, and myeloperoxidase (MPO). It was observed that NF-κB DNA-binding activity was significantly increased by treatment with hemolysate in cultured VSCMs, but this increase was suppressed by pretreatment with PDTC. Severe vasospasm was observed in the SAH group, which was attenuated in the PDTC group. Subarachnoid hemorrhage could induce increases of NF-κB DNA-binding activity and the gene expression levels of TNF-α, interleukin (IL)-1β, ICAM-1, and vascular cell adhesion molecule (VCAM)-1, which were reduced in the PDTC group. Immunohistochemical study demonstrated that the expression levels of TNF-α, ICAM-1, and MPO were all increased in the SAH group, but these increases were attenuated in the PDTC group. Our results suggest that NF-κB is activated in the arterial wall after SAH, which potentially leads to vasospasm development through induction of inflammatory response.
Introduction
Cerebral vasospasm is the most common cause of mortality and morbidity in patients with subarachnoid hemorrhage (SAH) (Treggiari-Venzi et al, 2001). Prevention of cerebral vasospasm is considered as a major goal in the management of patients surviving SAH. However, the exact pathogenesis of cerebral vasospasm remains obscure. It has hindered the development of effective and specific treatment paradigms for cerebral vasospasm.
There is increasing evidence that inflammation is critical in the development of cerebral vasospasm (Dumont et al, 2003). Clinical and experimental studies have shown the increased levels of the cytokines and adhesion molecules in the cerebrospinal fluid and serum after SAH (Dumont et al, 2003). However, concentrations in the cerebrospinal fluid and serum may not reflect their actual levels in the tissue and cannot provide virtual and firsthand information to understand the pathogenesis of cerebral vasospasm.
The inducible transcription factor nuclear factor-κB (NF-κB) represents various dimeric complexes of members of the Rel protein family, which includes Rel (c-Rel), RelA (p65), RelB, NF-κB1 (p50 and its precursor p105), NF-κB2 (p52 and its precursor p100) (Ghosh et al, 1998). Nuclear factor-κB is sequestered in the cytoplasm bound to the inhibitory IκB protein in most unstimulated cells and translocates to the nucleus via degradation of IκB once activated. The translocated NF-κB can bind to the specific DNA sequence in the chromatin with subsequent activation of specific subsets of genes involved in the inflammatory response. Nuclear factor-κB could activate the genes coding cytokines, chemokines, and adhesion molecules (Zingarelli, 2005). Therefore, the inflammatory responses in many diseases are mediated by the NF-κB pathway. Some studies have reported that the NF-κB pathway could be activated in cerebral ischemia and traumatic brain injury (Hang et al, 2004; Huang et al, 2001; Kaltschmidt et al, 2005; Nurmi et al, 2004a; Nonaka et al, 1999; Song et al, 2005; Stephenson et al, 2000; Zhang et al, 2005). Moreover, the role of NF-κB in smooth muscle contraction was also confirmed (Shi et al, 2003). Hence, we hypothesized that NF-κB is important in the development of cerebral vasospasm and might be a therapeutic target for cerebral vasospasm.
The antioxidant pyrrolidine dithiocarbamate (PDTC) is an established NF-κB inhibitor (Schreck et al, 1992) and has been widely used in the experimental study on the treatment of diseases related to the inflammation through inhibiting NF-κB activation. It was reported that PDTC could protect against brain ischemia (Nurmi et al, 2004b) and spinal cord trauma (La Rosa et al, 2004). However, to date, no study has been conducted to investigate the influence of PDTC on cerebral vasospasm.
Thus, the present study was aimed at evaluating the change of basilar arterial NF-κB DNA-binding activity after SAH and determining the potential role of NF-κB in the development of cerebral vasospasm. In addition, the influence of PDTC on cerebral vasospasm was also investigated.
Materials and methods
Animal Preparation
The experimental protocol using animals was approved by the Animal Care and Use Committee of Nanjing University and conformed to the Guide for the Care and Use of Laboratory Animals from the National Institutes of Health (NIH Publications No. 80-23).
All New Zealand white rabbits used in the present study were purchased from the Animal Center of the Chinese Academy of Sciences (Shanghai, China). They were acclimated in a humidified room and maintained on the standard pellet diet at the Animal Center of Jinling Hospital for 10 days before the experiment. The temperature in both the feeding room and the operation room was maintained at about 25°C.
Hemolysate Preparation
The hemolysate was prepared as described previously (Zubkov et al, 1999). Briefly, arterial blood collected from rabbits was centrifuged at 2500g for 15 mins, and the supernatant was discarded. The erythrocyte fraction was washed three times with saline (saline:erythrocyte fraction, 1:3, v/v). After the erythrocytes were disintegrated by ultrasonic waves and the particulate material was centrifuged at 15,000g for 90 min. The supernatant (hemolysate) was collected and stored at −80°C until further analysis. The oxyhemoglobin concentration was 0.85 ± 0.14 mmol/L, which was determined spectrophotometrically according to the reference (Kahn et al, 1981).
Vascular Smooth Muscle Cells Culture
Isolated vascular smooth muscle cells (VSMCs) were prepared from rabbit basilar artery as described previously (Kawanabe et al, 2002). Briefly, male New Zealand white rabbits, weighting from 1.8 to 2.2 kg, were anesthetized with an intramuscular injection of a mixture of ketamine (40 mg/kg) and droperidol (2.5 mg/kg) and killed by exsanguination. The basilar artery was removed, cleaned from surrounding tissues, dissected into small strips, and transferred to 100-mm culture flasks. They were then cultured at 37°C in a humidified atmosphere of 5% CO2/95% air. The growth medium was Dulbecco's modified Eagle medium (Invitrogen Corp., Carlsbad, CA, USA) supplemented with 15% fetal bovine serum (also from Invitrogen Corp.), 100 U/mL of penicillin, and 100 μg/mL of streptomycin. The medium was changed every 2 days. The confluent VSMCs were routinely subcultured at a split ratio of 1:3. Cells from passages 2 to 4 were used, which were characterized as smooth muscle cells by the presence of positive immunohistochemical staining for α-actin and by a characteristic hills-and-valleys appearance (Steele et al, 1991).
The cells were treated with hemolysate at a concentration of 10%, and preincubated with PDTC (80 μmol/L) for 1 h before adding hemolysate. After 48 h, the cells were collected for electrophoretic mobility shift assay (EMSA).
Nuclear Protein Extraction From Cultured Cells
Nuclear protein of the cultured cells was extracted and quantified as described (Andrews and Faller, 1991). Cells were washed twice with 4 mL ice-cold phosphate-buffered saline (PBS), then scraped in 1.5 mL of cold PBS, and pelleted for 10 secs and resuspended in 500 μL of ice-cold Buffer A (10 mmol/L N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (pH 7.9), 10 mmol/L KCl, 2 mmol/L MgCl2, 0.1 mmol/L ethylenediaminetetraacetic acid, 1 mmol/L dithiothreitol, and 0.5 mmol/L phenylmethylsulfonyl fluoride) (all from Sigma-Aldrich Inc., St Louis, MO, USA). Then the mixture was incubated on ice for 20 mins, and vortexed for 30 secs after addition of 50 μl NP-40 (Sigma-Aldrich Inc., St Louis, MO, USA). The mixture was centrifuged for 10 mins (5000g, 4°C). The pellet was then suspended in 50 μl ice-cold buffer B (50 mmol/L N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (pH 7.9), 50 mmol/L KCl, 300 mmol/L NaCl, 0.1 mmol/L ethylenediaminetetraacetic acid, 1 mmol/L dithiothreitol, 0.5 mmol/L phenylmethylsulfonyl fluoride, with 10% (v/v) glycerol) and incubated on ice for 30 mins with frequent mixing. After centrifugation (12,000g, 4°C) for 15 mins, the supernatants were collected as nuclear extracts and stored at −80°C until use.
Animal Experimental Design
Forty-two adult male New Zealand white rabbits weighing from 2.4 to 2.8 kg were assigned randomly to three groups: (1) the control group (n = 14) with intracisternal saline injection; (2) the SAH group (n = 14) with SAH and killed on day 5; (3) the PDTC group (n = 14) with SAH, intracisternal administration of PDTC (3 mg/kg) from day 0 to day 5, and then killed on day 5.
Six rabbits in each group were killed with the fixation-perfusion method. The basilar arteries were taken for hematoxylin and eosin, as well as immunohistochemical staining. The other rabbits in each group were exsanguinated and decollated. The basilar arteries were removed and frozen in liquid nitrogen immediately for EMSA and semiquantitative reverse transcriptase-polymerase chain reaction (RT-PCR) analysis.
Two-Hemorrhage Rabbit Model
Experimental SAH was produced according to the two-hemorrhage method (Varsos et al, 1983). The rabbits were anesthetized with an intramuscular injection of a mixture of ketamine (25 mg/kg) and droperidol (1.0 mg/kg) on day 0. Under spontaneous breathing, a 23-gauge butterfly needle was inserted percutaneously into the cisterna magna. After withdrawal of 1.5 mL cerebrospinal fluid, the same amount of nonheparinized fresh autologous auricular arterial blood was slowly injected into the cisterna magna for 1 min under aseptic technique. Then animals were kept in a 30° head-down position for 30 mins. After recovery from anesthesia, they were returned to the feeding room. Forty-eight hours after the first SAH, the second injection was performed in the same manner as the first. In control animals, the same technique was applied with injection of sterile saline instead of blood. In the PDTC group, PDTC (3 mg/kg) was injected into cisterna magna as the blood injection manner every 24 h from 1 min before first blood injection to the last day.
Perfusion-Fixation
On day 5, seven rabbits in each group were anesthetized with an intramuscular injection of a mixture of ketamine (40 mg/kg) and droperidol (2.5 mg/kg). The animals were then intubated endotracheally with a 3.5 mm diameter tracheal tube and mechanically ventilated with a rodent ventilator (SGC, Xi'an, China). Perfusion-fixation was then performed. After thorax was opened, a cannula was placed in the left ventricle. Subsequently, the descending thoracic aorta was clamped, and the right atrium was opened. Perfusion was begun with 500 mL of physiologic PBS (pH 7.4) at 37°C, followed by 500 mL of 10% buffered formaldehyde under a perfusion pressure of 120 cm H2O. After perfusion-fixation, the whole brain with the basilar artery was removed and immersed in the same fixative solution.
Measurement of Blood Vessel Cross-Sectional Area
The degree of cerebral vasospasm was evaluated by the measurement of basilar artery lumen's cross-sectional areas. The formalin-fixed and paraffin-embedded basilar artery sections (4 μm in thickness) were deparaffinized, hydrated, washed, and stained with hematoxylin and eosin. Then micrographs of the basilar arteries were put into the computer. Cross-sectional areas of blood vessels were determined by an investigator without knowing the group setting, using the High Definition Medical Image Analysis Program (HMIAP-2000, developed by Tongji Medical University, Wuhan, China). The areas were calculated by measuring the perimeter of the actual vessel lumen and then calculating the area of an equivalent circle (area = πr2, where r is the radius) based on the calculated equivalent r value from the perimeter measurement (r = perimeter/2π), thus correcting for vessel deformation and off-transverse sections. For each vessel, three sequential sections (midpoint of the proximal, the middle and the distal) were taken, measured, and averaged.
Immunohistochemical Study
Immunohistochemistory on formlin-fixed paraffin-embedded sections was performed to determine the immunoreactivity of tumor necrosis factor (TNF)-α, intercellular adhesion molecule (ICAM)-1, and myeloperoxidase (MPO). Sections were deparaffinized and rehydrated in graded concentrations of ethanol to distilled water. Endogenous peroxidase activity was blocked with 3% H2O2 for 5 mins, followed by a brief rinse in distilled water and a 15-mins wash in PBS. Sections were placed in 10 mmol/L citrate buffer (pH 6.0), and heated in a microwave oven at 95°C for 30 mins, then cooled at room temperature for 20 mins and rinsed in PBS. Nonspecific protein binding was blocked by 40-mins incubation in 5% horse serum. Sections were incubated with primary antibodies (anti-TNF-α, anti-ICAM-1 and anti-MPO, diluted 1:100, 1:150, and 1:200, respectively, all from Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) for 1 h at room temperature, followed by a 15-mins wash in PBS. Sections were incubated with HRP-conjugated goat anti-rabbit IgG (1:500 dilution) for 60 mins at room temperature. Diaminobenzidine was used as chromogen and counterstaining was performed with hematoxylin.
Nuclear Protein Extracts From Basilar Arteries
Nuclear protein of the vessels was extracted and quantified as described (Hang et al, 2005). Briefly, frozen vascular tissue was homogenized in 0.8 mL ice-cold Buffer A. The homogenates were then incubated on ice for 20 mins, and vortexed for 30 secs after addition of 50 μl NP-40. The mixture was then centrifuged for 10 mins (5000g, 4°C). The pellet was resuspended in 50 μl ice-cold buffer B and incubated on ice for 30 mins with frequent mixing. After centrifugation (12,000g, 4°C) for 15 mins, the supernatants were collected as nuclear extracts and stored at −80°C until use.
Electrophoretic Mobility Shift Assay
Electrophoretic mobility shift assay was performed using a commercial kit (Gel Shift Assay System; Promega, Madison, WI, USA). Consensus oligonucleotide probe (5′-AGT TGA GGG GAC TTT CCC AGG C-3′) was end-labeled with T4-polynucleotide kinase. Nuclear protein (30 μg) was preincubated in a total volume of 9 μl in a binding buffer, consisting of 10 mmol/L Tris-HCl (pH 7.5), 4% glycerol, 1 mmol/L MgCl2, 0.5 mmol/L ethylenediaminetetraacetic acid, 0.5 mmol/L dithiothreitol, 0.5 mmol/L NaCl, and 0.05 mg/mL poly(di-dc), for 10 mins at room temperature. After addition of the 32P-labled oligonucleotide probe, the incubation was continued for another 20 mins at room temperature. The reaction was stopped by adding 1 μl of gel loading buffer and the mixture was resolved on a native 4% polyacrylamide gel in 0.5 × TBE (Tris-borate-ethylenediaminetetraacetic acid) buffer. After electrophoresis was conducted at 390 V for 1 h, the gel was vacuum-dried and detected by autoradiography using X-ray film (Fuji Hyperfilm) at −70°C with an intensifying screen.
RNA Extraction and Reverse Transcriptase-Polymerase Chain Reaction
The abundance of TNF-α, interleukin (IL)-1β, ICAM-1, and vascular cell adhesion molecule (VCAM)-1 mRNA in each treatment group was determined by RT-PCR. Total RNA from tissue was extracted using TriPure Reagent (Roche Diagnostics Corp., Indianapolis, IN, USA) in accordance to the manufacturer's instructions. RNAs were reverse transcribed in a final volume of 20 μl solution containing 4 μg of total RNA, 20 U RNase inhibitor, 0.04 μmol of each dNTP, 0.5 μg oligo(dT)15, and 15 U AMV reverse transcriptase (all from Promega, Madison, WI, USA) for 1 h at 42°C. The reaction was terminated by an initial denaturation step at 95°C for 5 mins. The cDNA was stored at −20°C or used for PCR immediately. Polymerase chain reaction amplification was performed in a total volume of 25 μl solution containing 4 μl cDNA, 0.05 μmol MgCl2, 2.5 U Taq polymerase, 0.02 μmol of each dNTP, specific oligonucleotide primers (Table 1) for TNF-α, IL-1β, ICAM-1, VCAM-1, or GAPDH genes, and 2.5 μl 10 × Taq polymerase reaction buffer. Each PCR cycle included a denaturation step at 94°C for 30 secs, a primer annealing step for 30 secs (the temperatures are listed in Table 1), an extension step at 72°C for 50 secs, and a final extension step at 72°C for 7 mins. The number of cycles is shown in Table 1. The amplified fragments were then detected by agarose gel electrophoresis and visualized by ethidium bromide staining. The gel was captured as a digital image and analyzed using Glyko Bandscan software (Glyko, Novato, CA, USA). Values for each sample were normalized against respective GAPDH level.
Sequences of PCR primers
Statistical Analysis
All data were presented as mean ± s.e.m. of at least three independent experiments, as detailed in figure legends. All data were subjected to one-way analysis of variance (ANOVA) combined with Tukey's multiple comparison test. Statistical significance was defined as P < 0.05.
Results
Effects of Hemolysate and Pyrrolidine Dithiocarbamate on the Activity of Nuclear Factor-κB in Cultured Vascular Smooth Muscle Cells
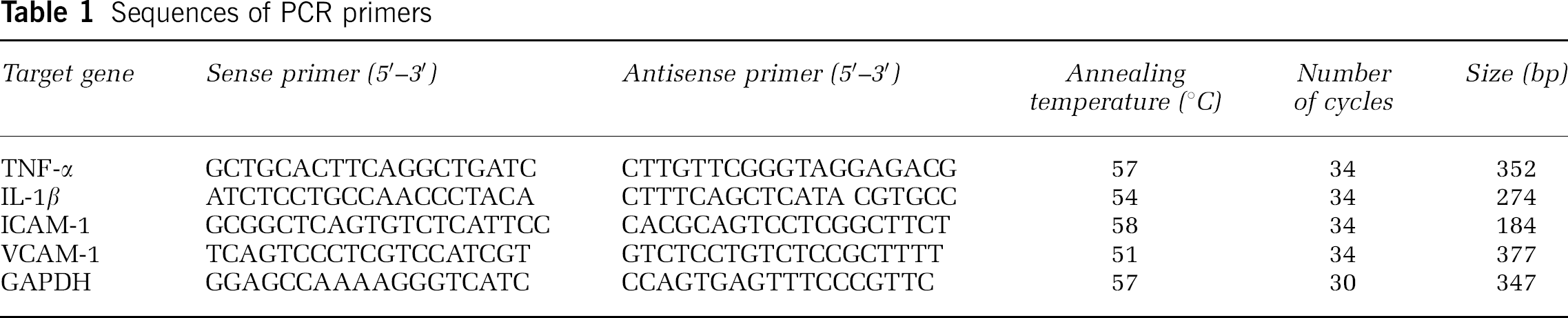
There was a significant difference in the DNA-binding activity of NF-κB among the control, hemolysate, and hemolysate/PDTC groups (P < 0.001, ANOVA; Figures 1A and 1B). A significant difference was found between the control group and the hemolysate group (P < 0.01, Tukey's multiple comparison test). The hemolysate group and the hemolysate/PDTC group also exhibited a significant difference (P < 0.05, Tukey's multiple comparison test). In other words, the DNA-binding activity of NF-κB was significantly increased by treatment with hemolysate, and the increased activity was markedly suppressed by the preincubation of PDTC.
Effects of PDTC on the NF-κB DNA-binding activity in the hemolysate-treated VSMCs. (
Rabbit Subarachnoid Hemorrhage Model
General observation: No SAH-related death was observed in all experimental groups. The body weights did not exhibit a significant difference among all groups (P > 0.05, ANOVA, data not shown). The residual subarachnoid clots appeared around the basilar arteries of the rabbits in the SAH and PDTC groups.
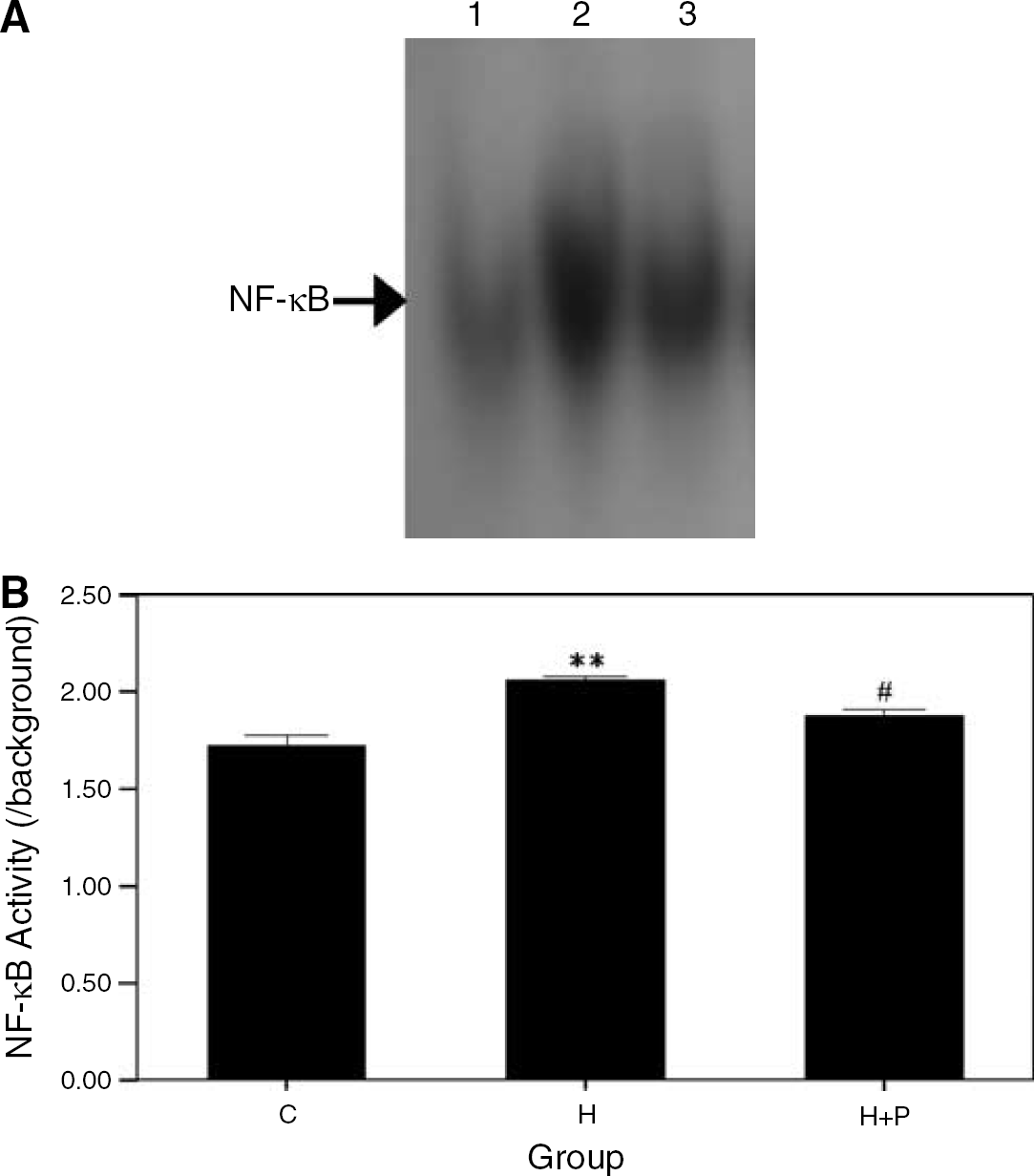
Histologic examination and the basilar artery cross-sectional area: Profound vasospasm could be observed in the basilar arteries of the rabbits in the SAH group, and the vasospasm was attenuated in the PDTC group (Figures 1A–1C). The internal elastic lamina corrugation and thickened media were severe in the SAH group and were also attenuated in the PDTC group. In contrast, we found no corrugation of the internal elastic lamina and no thickening of the media in the control group.
There was a significant difference in the cross-sectional area of basilar artery among all the groups (P < 0.01, ANOVA; Figures 2A–2D). A significant difference was detected between the SAH (252752.34 ± 19847.31 μm2) and the control (826620.56 + 19266.43 μm2) groups (P < 0.01, Tukey's multiple comparison test). There was also a significant difference in the basilar arterial cross-sectional area between the PDTC (483371.71 ± 33851.85 μm2) and SAH groups (P < 0.01, Tukey's multiple comparison test).
Changes in the cross-sectional area of basilar arteries in the experimental SAH model. (
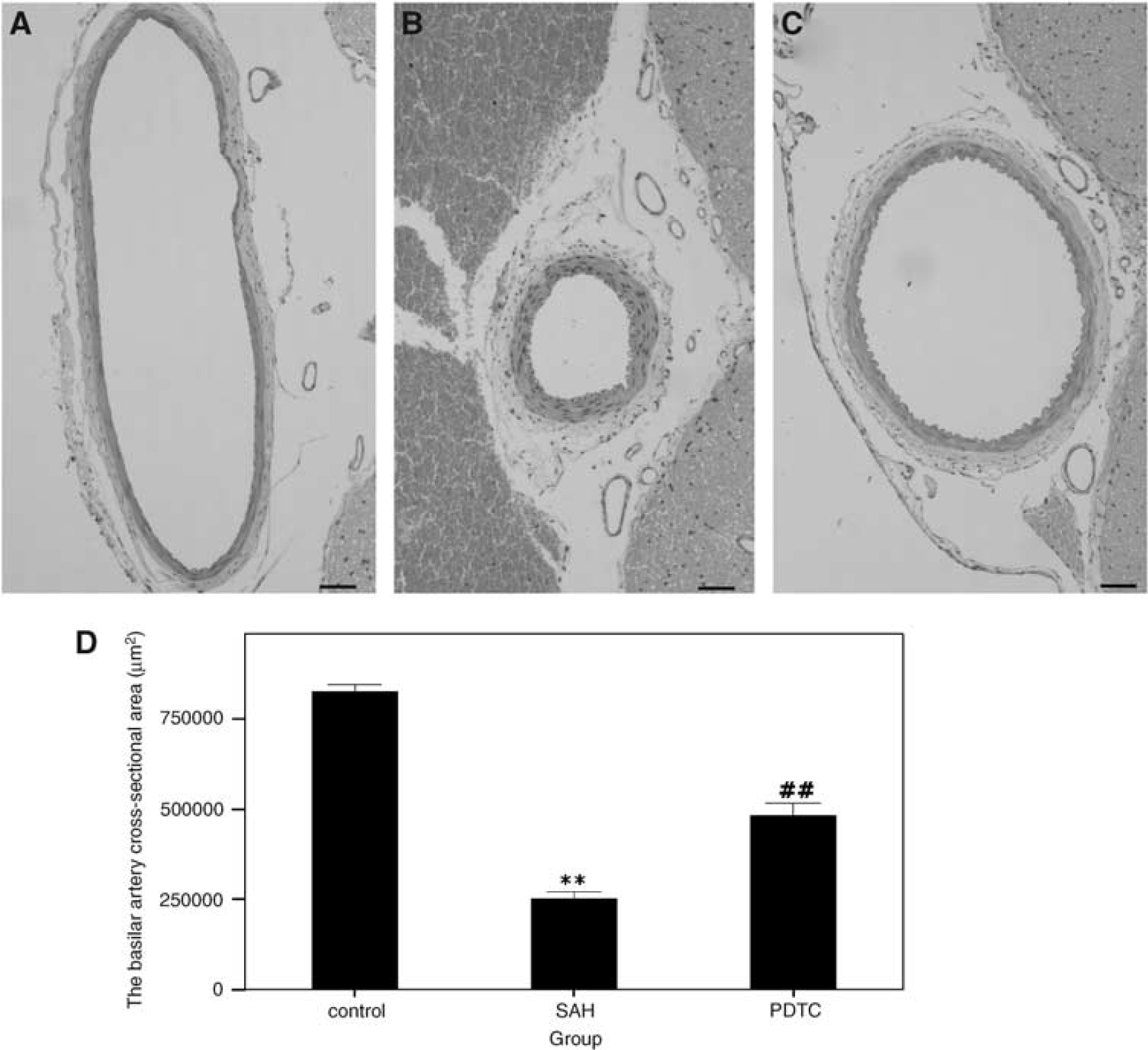
Immunohistochemical study for tumor necrosis factor-a, intercellular adhesion molecule-1, and myeloperoxidase: The immunohistochemical study showed that both TNF-α and ICAM-1 were expressed at lower levels in the control group, while they were expressed at relatively high levels in the SAH group. In the PDTC group, the expressions of TNF-α and ICAM-1 were reduced compared with the SAH group. Tumor necrosis factor-α immunoreactivity was located in the intimal, medial, and adventitial cells (Figures 3A–3C). However, ICAM-1 immunoreactivity was mainly expressed in the intimal and adventitial cells (Figures 3D–3F). The expression of the MPO showed the same tendency as TNF-α and ICAM-1, which was detected intensively in adventitia and moderately in the intima (Figures 3G–3I).
Immunohistochemical study of TNF-α (
EMSA for nuclear factor-κB: The NF-κB DNA-binding activity in the basilar artery was assessed using the EMSA. A significant difference was found in the NF-κB DNA-binding activity between the control and SAH groups (P < 0.01, Tukey's multiple comparison test; Figure 4B). The elevated NF-κB DNA-binding activity in the basilar artery was significantly reduced in the PDTC group compared with the SAH group (P < 0.01, Tukey's multiple comparison test).
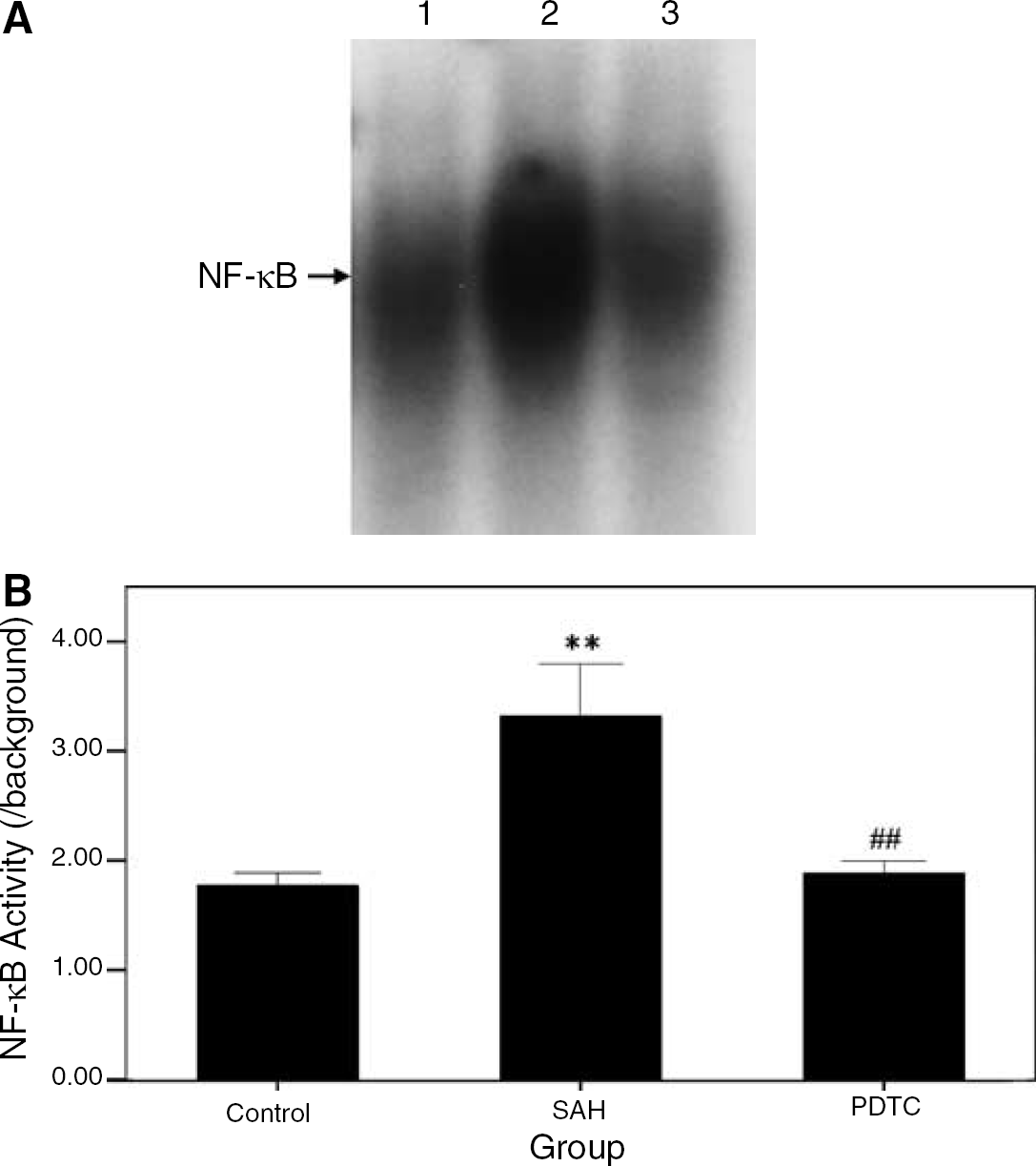
The NF-κB DNA-binding activity detected by EMSA. (
Reverse transcriptase-polymerase chain reaction for the gene expressions levels of tumor necrosis factor-α, interleukin-1β, intercellular adhesion molecule-1, and vascular cell adhesion molecule-1: There were significant differences in the gene expression levels of TNF-α, IL-1β, ICAM-1, and VCAM-1 among the control, SAH, and PDTC groups (P < 0.01, ANOVA; Figures 5A and 5B). The average expression levels of TNF-α, IL-1β, ICAM-1, and VCAM-1 in the SAH group were all higher than those in the control group (all P < 0.01, Tukey's multiple comparison test), which were attenuated by treatment with PDTC (all P < 0.05, Tukey's multiple comparison test).
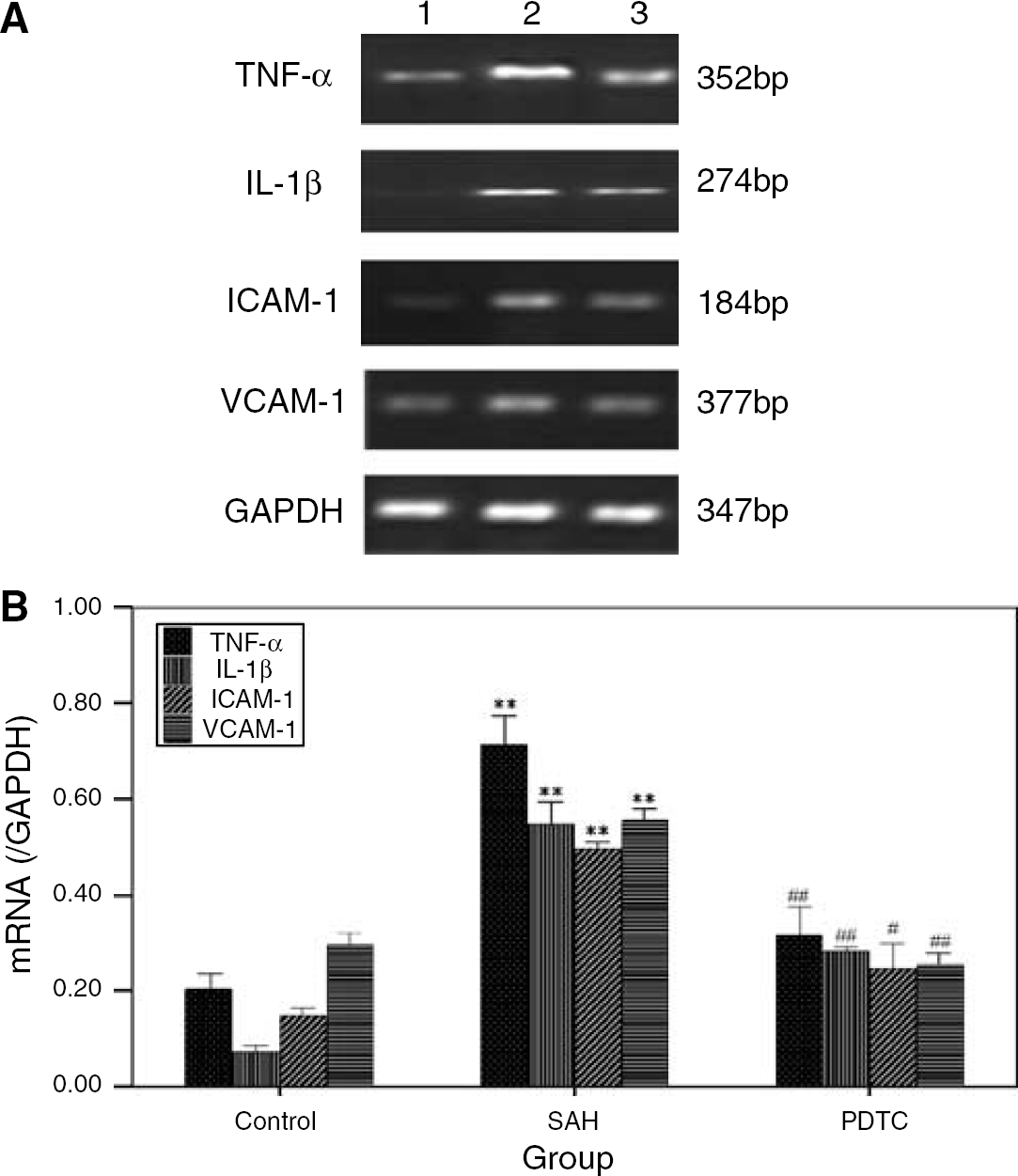
The gene expression levels of TNF-α, IL-1β, ICAM-1, and VCAM-1 in the basilar artery. (
Discussion
The main findings of this study are as follows: (1) NF-κB DNA-binding activity in the cultured VSMCs was increased by the treatment with hemolysate and this increase was reduced by the pretreatment with PDTC; (2) SAH-induced increases of NF-κB DNA-binding activity and the expressions of cytokines and adhesion molecules could be suppressed by intracisternal administration of PDTC in the SAH model; (3) the inhibitor of NF-κB, PDTC, could attenuate cerebral vasospasm. These findings suggest that activation of NF-κB pathway after SAH may facilitate the development of cerebral vasospasm in the rabbit SAH model. Moreover, PDTC could be a potential therapeutic agent for cerebral vasospasm after SAH.
Inflammation and Cerebral Vasospasm
Inflammation is a complicated and multifaceted response aiming to defend against foreign antigens. In the circumstance of SAH, a complex series of cellular and molecular events is elicited by the presence of a blood clot in the subarachnoid space, culminating in a robust inflammatory response. Following cellular and molecular events were involved: (1) expressions of adhesion molecules that evoke leukocytes adhesion to the endothelium; (2) cytokines production; and (3) immunoglobulin and complement activation. Cerebral vasospasm might result from the interaction among those events (Dumont et al, 2003).
In the present study, we analyzed the degree of leukocyte infiltration in the vessel wall through the assessment of MPO expression and evaluated the production of the cytokines and adhesion molecules. It was well proven that inflammatory response was triggered in the basilar artery after experimental SAH.
Myeloperoxidase, the abundant heme enzyme both in neutrophils (5% of dry weight) and monocytes (1% of dry weight), is considered to be an index of leukocyte infiltration. Excessive MPO released by neutrophils is linked to the pathologic process in inflammatory disorders (Gaut et al, 2001). The close interactions between MPO and the vessel wall appear to be important for the pathophysiology of some vascular diseases. Myeloperoxidase seems not only injure the vessel wall by its oxidative armament, but also elicit subtle changes in the local vascular redox state, and subsequently cause prominent changes in cellular signaling cascades, which in turn is a prerequisite for the phenotype of vascular inflammatory diseases. The important role of MPO in the atherosclerotic disease has been studied intensively (Lau and Baldus, 2006). The results of the present study demonstrated that MPO activity was increased in the SAH group. The MPO-positive cells were detected intensively in the adventitia and moderately in the intima, which implied the leukocyte infiltration occurred and the inflammatory response was elicited in the vessel wall of the basilar artery after experimental SAH.
Intercellular adhesion molecules and vascular cell adhesion molecules, members of the immunoglobulin super-family in adhesion molecules, are important in leukocyte recruitment during the inflammatory process. These include ICAM-1 and VCAM-1, which could be profoundly upregulated after cytokine challenge. The present study demonstrated that the abundance of ICAM-1, VCAM-1 mRNA, as well as ICAM-1 protein level was increased in the basilar artery in the SAH group. The immunohistochemical study showed ICAM-1 was mainly distributed at the intima and adventitia of the basilar artery. These findings were consistent with the previous study (Handa et al, 1995).
Cytokines are recognized as small and nonstructural proteins, which are pleiotropic and have multiple diverse biologic activities (Sercombe et al, 2002). Some cytokines, such as TNF-α, IL-1α, IL-1β, IL-6, and IL-8, have found to be upregulated in experimental and/or clinical cerebral vasospasm after SAH (Aihara et al, 2001; Fassbender et al, 2001). Our studies showed the expression levels of TNF-α and IL-1β genes were increased in the basilar arterial wall of SAH rabbbits. In addition, the immunohistochemical study indicated TNF-α immunoreactivity was located in the intimal, medial, and adventitial cells. These results and the previous findings suggest that the expression and secretion of cytokines should be an important step in the inflammatory response in the vessel wall after SAH.
Nuclear Factor-κB and Cerebral Vasospasm
Owing to the central role of NF-κB in immunity and inflammation, it has drawn extensive attentions in the research on the diseases including stroke and traumatic brain injury (Hang et al, 2004; Zhang et al, 2005). In addition, the important role of NF-κB in vascular immunity and inflammation has also been well characterized according to the previous study in vivo and vitro (Bellas et al, 1995; Monaco and Paleolog, 2004; Shi et al, 2003). Some atherosclerosis-related studies have demonstrated that activation of NF-κB in the endothelium after stimulation could increase the expressions of cytokines and adhesion molecules. Some of these inflammatory mediators could also activate NF-κB, hence creating a positive feedback that amplifies the inflammatory response (Monaco and Paleolog, 2004). It has also been reported that NF-κB could be activated and subsequently regulated the expressions of proinflammatory cytokines in the smooth muscle, which appeared to be essential for smooth muscle proliferation (Bellas et al, 1995; Zeiffer et al, 2004). Since inflammation in response to subarachnoid blood is a plausible candidate pathway leading to cerebral vasospasm, it is reasonable to speculate that NF-κB could play a potential role in the development of cerebral vasospasm.
Our present study demonstrated that the NF-κB DNA-binding activity was increased in the cultured VSMCs after hemolysate stimulation. In the animal model, NF-κB activation was upregulated in the SAH group compared with the control group. However, it should be indicated that inflammatory cells infiltrating to the arterial wall and the endothelial cells also contribute to the effect of SAH on NF-κB activity in vivo besides the smooth muscle cells. Based on our results, it could be speculated that the hemolysate of the subarachnoid blood clot after SAH stimulated the NF-κB activation and then upregulated the expressions of cytokines and adhesion molecules, the inflammatory response in the vessel wall was then triggered and cerebral vasospasm was induced ultimately.
In our previous study we have observed that the basilar artery showed the most severe vasospasm on day 3 and day 5 in the rabbit two-hemorrhage SAH model (Zhou et al, 2007). Cerebral vasospasm on day 5 might be regarded as chronic vasospasm, which is similar to the chronic cerebral vasospasm in humans. Hence, day 5 is an appropriate time-point to detect the NF-κB DNA-binding activity and to estimate the effects of PDTC on the cerebral vasospasm. However, we could not exclude the possibility that NF-κB DNA-binding activity is increased before day 5. The former activation of NF-κB could induce the inflammatory response in arterial wall and may contribute to cerebral vasospasm eventually. Further studies, such as the studies on the time course of DNA-binding activity after SAH, are essential to understand the importance of NF-κB in the pathogenesis of cerebral vasospasm.
Pyrrolidine Dithiocarbamate and Cerebral Vasospasm
Pyrrolidine dithiocarbamate has been well established as a relatively selective inhibitor of NF-κB by preventing degradation of IκB (Liu et al, 1999). In the present study, cerebral vasospasm after SAH could be attenuated by intracisternal injection of PDTC. Electrophoretic mobility shift assay data showed that PDTC inhibits elevated NF-κB DNA-binding activity in the SAH model. These results suggest that the activation of NF-κB was involved in the development of cerebral vasospasm and the therapy effects of PDTC were possibly mediated via inhibition of NF-κB activation. However, because PDTC has a variety of other biochemical activities such as heavy metal chelation, thiol modification, and oxygen radical scavenging properties (El Eter et al, 2005; Iseki et al, 2000; Li et al, 1999; Mihm et al, 1991), The possibility that PDTC attenuates cerebral vasospasm in part through these actions directly was not excluded by the present study. Furthermore, one previous study even showed that PDTC at high concentrations could induce glutathione oxidation and apoptotic cell death in cultured endothelial cells (Moellering et al, 1999). Therefore, further studies are warranted to discern the exact mechanisms of the inhibitory effect of PDTC on cerebral vasospasm.
Several investigators have reported that NF-κB is activated in the CNS such as in neurons and glial cells after cerebral ischemia and traumatic brain injury (Hang et al, 2004; Huang et al, 2001; Kaltschmidt et al, 2005; Nurmi et al, 2004a; Nonaka et al, 1999; Song et al, 2005; Stephenson et al, 2000; Zhang et al, 2005). Moreover, PDTC, the inhibitor of NF-κB, could be expected to act as a new agent for neuroprotection (Nurmi et al, 2004b). In addition to inhibiting the abnormal reactions of the major arteries, PDTC may be able to protect neuronal activity and prevent the neuronal apoptosis, and improve microcirculation after SAH. It is assured that further ingenious studies are needed and will be conducted in our laboratory.
In summary, we have shown that the inflammatory response mediated by NF-κB has occurred after SAH. And the NF-κB pathway plays a potential role in the pathogenesis of cerebral vasospasm. Pyrrolidine dithiocarbamate, an inhibitor of NF-κB, could attenuate the degree of cerebral vasospasm, so it may be a potential therapeutic agent for cerebral vasospasm.
Footnotes
Acknowledgements
The authors thank Dr Wu Bo for his generous assistance in pathologic examination and Dr Yi-fang Wang for her critical review and editing of the manuscript. This work was supported by grants from the Jinling Hospital of Nanjing, Jiangsu province, China.
