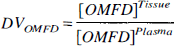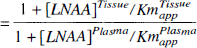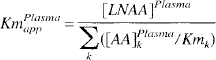Abstract
The distribution volume (DY) of 6-[F-18]fluoro-L-DOPA (FDOPA) in the cerebellum recently has been linked using positron emission tomography (PET) to plasma large neutral amino acid (LNAA) concentrations in monkeys. In this article the authors provide additional experimental support for this relation by directly measuring the DY as the steady-state tissue to plasma radioactivity ratio in rats using a labeled LNAA analog 3-O-methyl-6-[F-18]FDOPA (OMFD), a compound that has no known specific enzyme or receptor interactions in brain tissue. The measured DY for OMFD (tissue OMFD concentration/plasma OMFD concentration) was found to be inversely related to plasma LNAA concentrations. The relation (DY = 1.5−0.00094* [LNAA], R^2 = 0.79) resulted in an 8% DY decrease per 100 nmol/mL plasma LNAA increase within the observed range of 330 to 510 nmol/mL. This was similar to recent noninvasive observations with FDOPA PET in vervet monkeys and with 6-[F-18]Fluoro-m-tyrosine PET in squirrel monkeys. The OMFD striatum to cerebellum (Str/Cb) ratio was greater than 1.0 for all measurements, averaging 1.09 ± 0.04, and was approximately equal to the Str/Cb LNAA ratio of 1.12 ± 0.05. This current study verifies the variation of DV of OMFD or FDOPA as a function of plasma LNAA concentrations and suggests the possibility of using OMFD for measuring cerebral LNAA noninvasively with PET.
Recent observations with positron emission tomography (PET) in vervet monkey demonstrated that the distribution volume (DV) of 6-[F-18]fluoro-L-DOPA (FDOPA) was inversely related to the plasma large neutral amino acid (LNAA) concentrations (Stout et al., 1998). This finding has also been observed in squirrel monkey PET studies using FDOPA and for an amino acid analog, 6-[F-18]Fluoro-m-tyrosine (Stout et al, 2000). A simple explanation has been offered to account for this effect (Huang et al., 1998): the transport of LNAA between plasma and tissue is limited by saturation of the transporter system (Smith et al., 1987) and the DV of a labeled LNAA is a function of LNAA levels that are dependent on the apparent half-saturation con centration of the transporter (Kmapp). This effect provides a simple way in which the brain LNAA concentrations can be maintained at a fairly constant level. This mechanism is crucial, because plasma LNAA concentrations are known to vary significantly with diet (Fernstrom et al., 1979; Voog and Ericksson, 1992a), over the course of the day (Wurtman et al., 1968; Voog and Eriksson, 1992b) and among subjects (Scriver et al., 1985).
The current study measures directly the DV in rat brain of a labeled LNAA as a function of plasma LNAA concentrations using the amino acid analogue 3-O-methyl-6-[F-18]Fluoro-L-DOPA (OMFD). Other investigations have shown that OMFD, a major peripheral metabolite of FDOPA, uses the same LNAA transport system and has no apparent specific enzymatic or receptor interactions within the brain (Doudet et al., 1991; Wahl et al., 1994). Therefore, tissue DV of OMFD can be directly determined in both striatum and cerebellum when both tissue and plasma concentrations approach constant levels. These measurements can be used to further support the proposed blood—brain barrier (BBB) saturation mechanism that explains the variation of DV of a labeled inert LNAA as a function of the plasma LNAA level (Huang et al., 1998).
In addition, the combination of multiple pairs of tissue and plasma LNAA concentrations, together with the OMFD DV, allowed estimation of LNAA transport constants (Kmapp) for plasma and tissue. The Kmapp values obtained were then compared with those obtained by other means (Huang et al., 1998). The result shows that a consistent Kmapp value can potentially be used in combination with OMFD DV measurements from PET data and plasma LNAA concentrations to estimate tissue LNAA concentrations in vivo. These findings and their implications are also discussed.
MATERIALS AND METHODS
Fifteen male Sprague—Dawley rats were divided into 5 groups (n = 3) and allowed free access to food for 1 week before experiments. Approximately 16 hours before the experiments, animals were given free access overnight to diets consisting of either 0% (fasted), 16%, 24%, 35%, or 45% protein diets. The 16% and 24% protein diets were commercially available (Teklad, Madison, WI, U.S.A.). The 35% and 45% diets were produced by adding casein (Sigma, St. Louis, MO, U.S.A.) to crushed 24% protein pellets, mixed with a small amount of water and dried into pellets.
Synthesis of OMFD followed a previously published method (Adam et al., 1994) with minor modifications (radiochemical purity >99%). Rats were consciously injected with OMFD through tail vein at 10 to 11 in the morning. Blood samples were withdrawn and plasma radioactivity determined in a gamma well counter at 60 minutes postinjection, when plasma OMFD values approached a constant level (see below). Plasma samples were deproteinated with an equal volume of 0.8 mol/L perchloric acid, filtered, and stored at −4°C for later LNAA analysis. After blood sampling, the animals were immediately decapitated and the striatum and cerebellum tissues were removed and immediately frozen in liquid nitrogen. After weighing and well counting, tissues were homogenized in an equal volume of 0.8 mol/L perchloric acid by glass rod and sonicated for 30 seconds. The slurry was filtered (0.22 micron) and stored at −4°C for subsequent LNAA analysis.
The percentage of plasma radioactivity due to unmetabolized OMFD at 60 minutes was determined in 4 rats by HPLC (C-18 column, 1 mL/min, 80% 0.1 mol/L NaH2P04, 2.6 mmol/L octane sulfonic acid sodium salt, 0.1 mmol/L EDTA, pH 3.1, 20% methanol). In 1 rat, plasma samples were drawn at 30-minute intervals for 2 hours to characterize the rate of metabolite formation and to support the notion that plasma OMFD had reached a stable value at 60-minute postinjection. The effect of L-(−)-alpha-hydrazino-3,4-dihydroxy-alpha-methylhydrocinnamic acid monohydrate (carbidopa) administration on metabolite formation was examined in three additional rats. Carbidopa was administered (10 mg/kg, IP) 30 minutes before OMFD injection and metabolite formation was determined at 15-minute intervals for 2 hours in one rat and at 60 and 120 minutes in all 3 rats.
Quantification of LNAA concentrations in tissue and plasma was determined based on previously published methods (Keen et al., 1989; Stout et al., 1998). In brief, samples were partially neutralized with an equal volume of 0.3 mol/L potassium hydroxide, chilled for 5 minutes, and filtered. Equal volumes of sample and o-phthaldialdehyde (1 mg/mL, Sigma) were mixed, incubated for 2 minutes, and injected onto a C-18 HPLC column (Beckman Instruments, Fullerton, CA, U.S.A.) at 0.8 mL/min. The eluent was ramped from 70:30 0.1 mol/L sodium phosphate:methanol to 35:65 at 30 minutes and the output was measured by fluorescence detection. Peak areas were converted to micromolar values by comparison with known dilutions of LNAA standards. The total LNAA concentration in plasma was calculated as the sum of tyrosine, phenylalanine, methionine, leucine, isoleucine, valine, and total tryptophan concentrations (see Discussion).
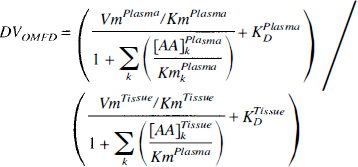
Distribution volumes were calculated for LNAA and OMFD (Eq. 1) in both striatum and cerebellum, where [OMFD] was the radioactivity concentration measured in either plasma or tissue at 60 minutes postinjection, when the OMFD concentrations had reached an approximate steady-state. The LNAA DV was determined by the tissue to plasma LNAA concentration ratio. With OMFD present as an inert LNAA in only tracer quantities, the DV on OMFD can be described as a function of the LNAA concentrations to Km ratios (Huang et al., 1998) (Eq. 2). The KmPlasmaapp and KmTissueapp for striatum and cerebellum were estimated from Eq. 2 using DVOMFD estimates from Eq. 1, where [LNAA] was the sum of seven large neutral amino acids in plasma or tissue. The KmTissueapp was also estimated (Eq. 2) by constraining the KmPlasmaapp to the value determined using Eq. 3.
Estimates of KmPlasmaapp were also calculated from individual LNAA concentrations [AA]k (Huang et al., 1998) using previously published individual Kmk values for conscious rats (Miller et al., 1985) as
The estimated KmPlasmaapp value from Eq. 3 and DV
The contribution of OMFD diffusion across the BBB could potentially contribute a significant fraction of the BBB transport. Therefore, the authors estimated the diffusion fraction for OMFD transport using a modification of Eq. 2, in which a nonsaturable diffusion term was added to the forward and reverse transport of OMFD. With the addition of the diffusion term, the distribution volume of OMFD would be
where Vm and Km are the maximal rate and half saturation constants for carrier-mediated transport across the BBB, and Kd is the diffusion rate. Superscripts indicate whether the value is for tissue or plasma. Using Eq. 3, and assuming that (VpmKTm)/(VTmKpm) ≃ 1, the above equation can be rearranged to
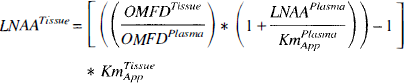
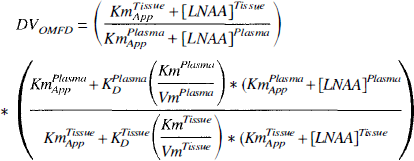
In this form, the diffusion component, Kd*(Km/Vm), in the equation represents the fraction of OMFD transported through diffusion when there is zero competition (that is, [LNAA] = 0). The equation was further constrained by assuming this diffusion fraction was equal for forward and reverse transport and by fixing KmPlasmaapp to the estimated value of 84.3 nmol/mL. Estimates of KmTissueapp and the diffusion fraction were then obtained in striatum by fitting the directly measured DV
RESULTS
OMFD metabolism
Plasma OMFD concentration values were relatively constant (±3.5%) over the range of 30 to 120 minutes after injection. A metabolite was evident in the plasma HPLC data that was formed at a nearly linear rate of 17.3% ± 3.2% per hour (R^2 = 0.92; Fig. 1). Similar metabolite formation was observed with carbidopa (10.0% ± 5.3% per hour), indicating that the OMFD metabolism was not significantly affected (P > 0.1) by inhibition of peripheral aromatic amino acid decarboxylase (AAAD). The single metabolite was consistent with sulfate conjugates (Melega et al., 1991), which because of its charged nature is not known to cross the BBB (Doudet et al., 1991; Wahl et al., 1994). Because the hydrophilic metabolite remains in the bloodstream and does not cross the BBB through the LNAA transporter, the metabolite fraction was subtracted from the plasma radioactivity for determination of the plasma input function used in subsequent data analysis.
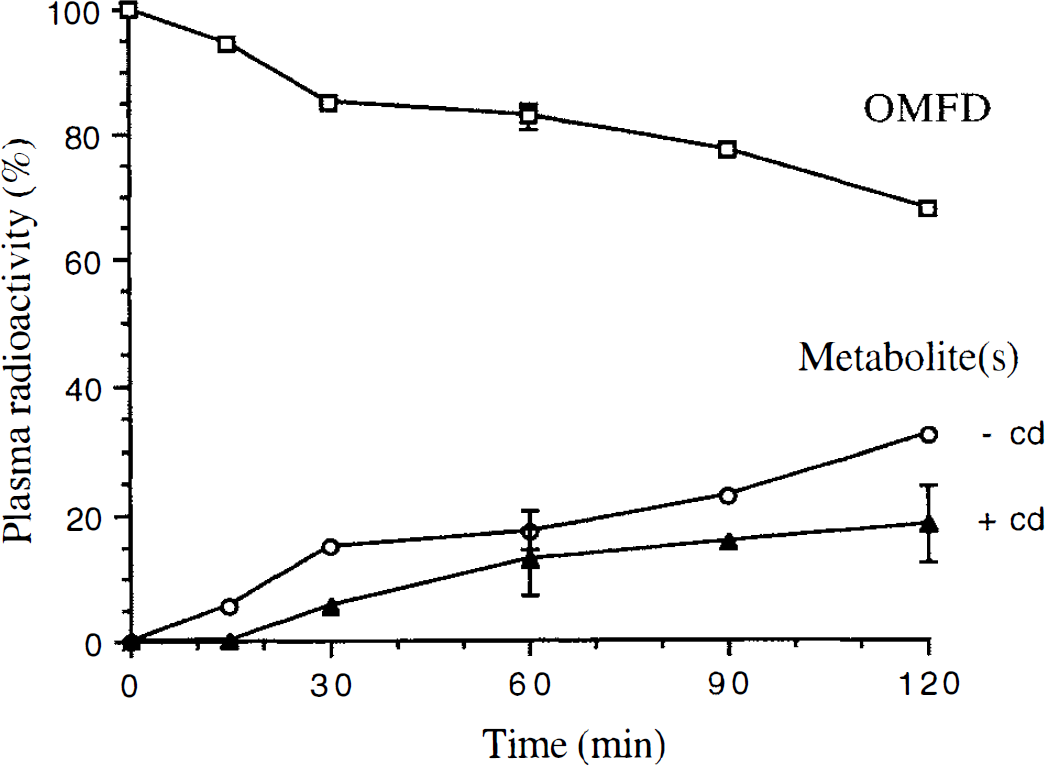
Plasma radioactivity profile indicating OMFD metabolism in rat. OMFD (open box) and metabolites (open circle) without carbidopa. Single experiment except at 60 minutes where n = 4. Triangle represents single carbidopa experiment except at 60 and 120 minutes where n = 3.
In tissue, only a single peak corresponding to OMFD and no metabolite fraction was observed. The authors' detection system was sufficiently sensitive to pick up the metabolite fraction if it were present at levels similar to that in plasma (10% to 17%). Trace quantities of the metabolite may have been present but below the level of detection.
Striatum and cerebellum distribution volume data
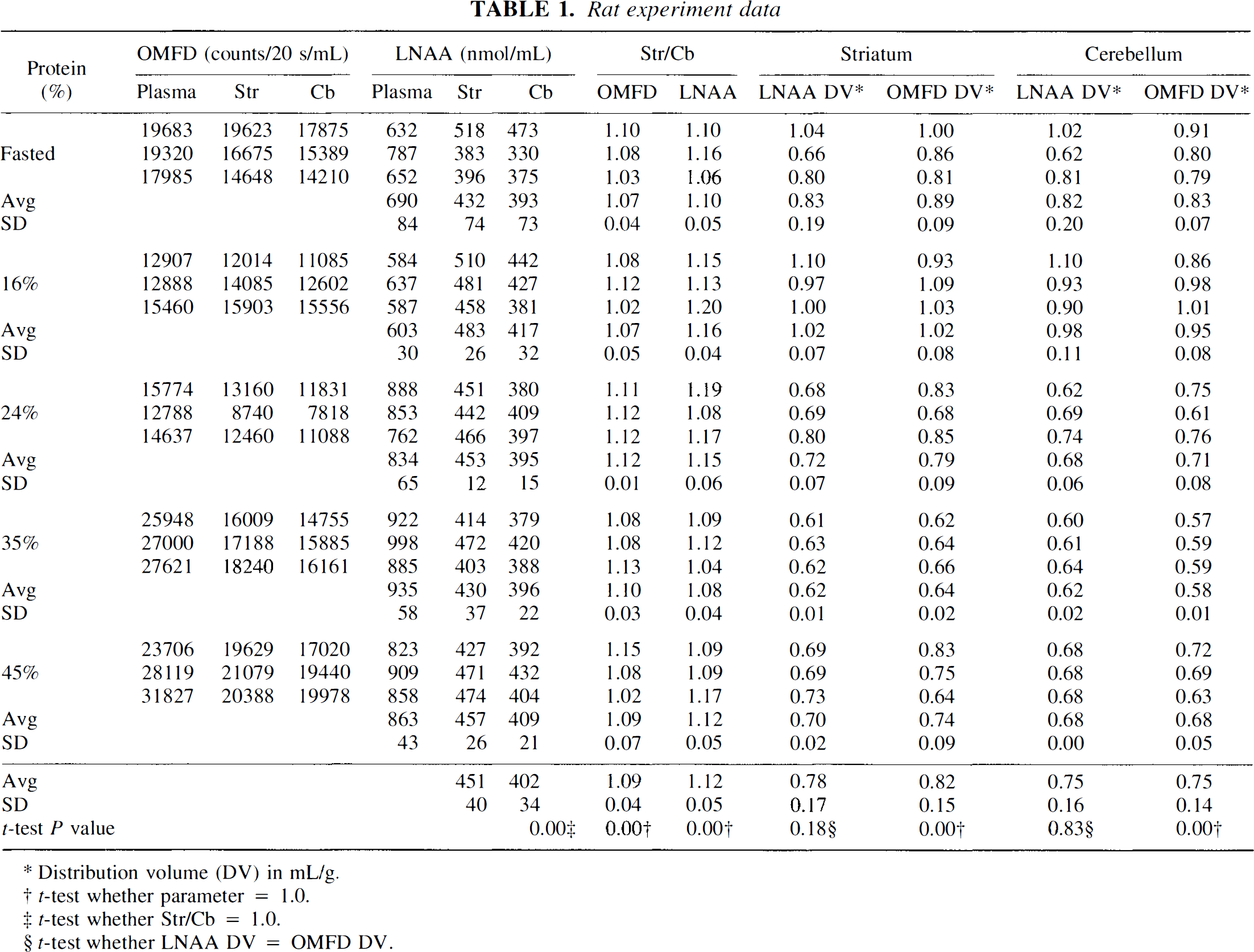
Striatum and cerebellum tissue LNAA concentrations remained unchanged over the measured range of plasma LNAA concentrations (Fig. 2). The average striatum LNAA concentration (457 ± 40 nmol/mL; Table 1) was significantly higher (12%, t-test, P < 0.01) than cerebellum concentrations (average 402 ± 34 nmol/mL). Striatum to cerebellum ratios for all experiments were greater than 1.0 (Fig. 3A) and were consistent with having a normal distribution (W-test, α = 0.1, Wadsworth, 1990). No significant difference between OMFD (1.09 ± 0.04) and LNAA (1.12 ± 0.05) Str/Cb ratios (t-test, P > 0.1) was found, and the two measurements were closely correlated (DV
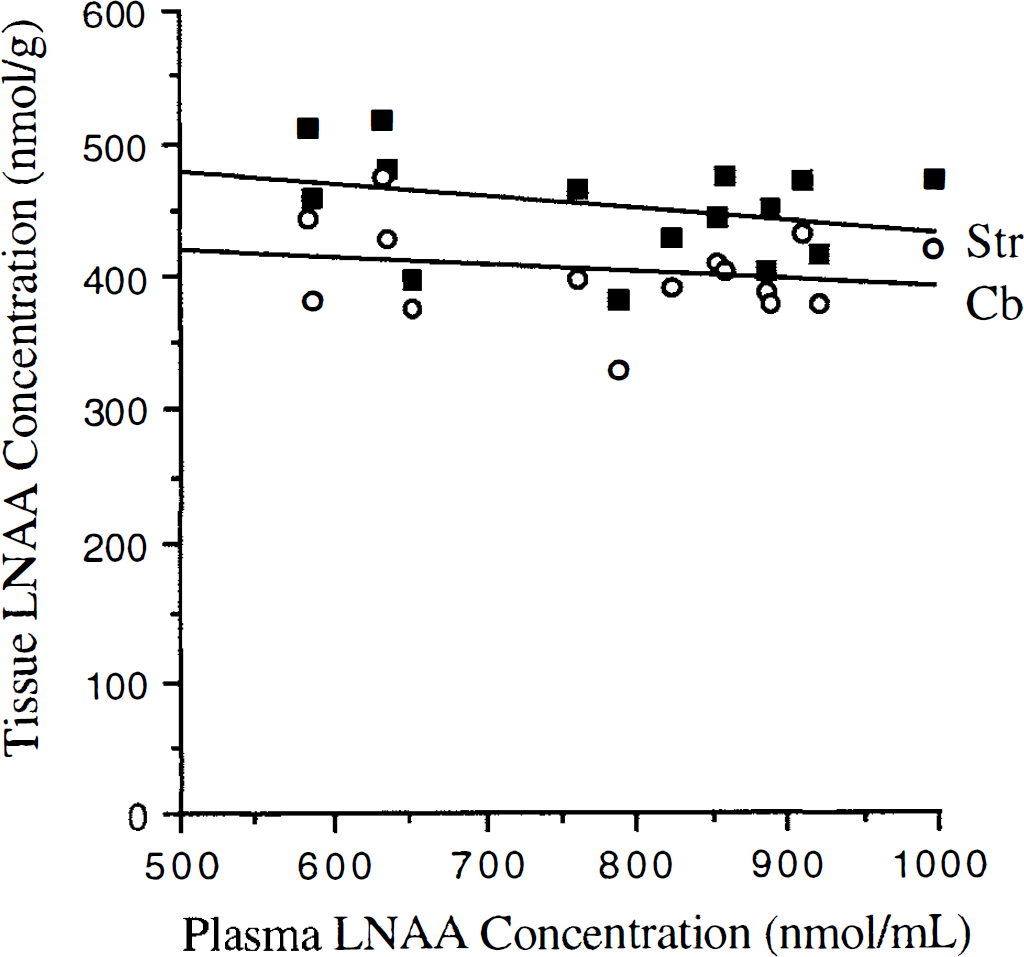
Striatum (Str. closed boxes) and cerebellum (Cb, open circles) large neutral amino acid (LNAA) concentrations versus plasma LNAA concentrations.
Rat experiment data
Distribution volume (DV) in mL/g.
t-test whether parameter = 1.0.
t-test whether Str/Cb = 1.0.
t-test whether LNAA DY = OMFD DV.
Using individual LNAA Km values for conscious rats (Miller et al., 1985) and measured plasma LNAA concentrations, a KmPlasmaapp value of 84.3 ± 2.5 nmol/mL was estimated based on Eq. 3. Fitting results for KmTissueapp from Eq. 2 using KmPlasmaapp fixed to 84.3 and summed LNAA concentrations resulted in a striatum KmTissueapp value of 65.9 ± 1.9 nmol/mL (R^2 = 0.71), which was 7% higher than the cerebellum value of 61.6 ± 1.6 nmol/mL. Estimates of OMFD KmPlasmaapp and KmTissueapp using Eq. 2 without constraining the KmPlasmaapp resulted in higher and more variable estimates (striatum: KmPlasmaapp 146 ± 157 nmol/mL, KmTissueapp 116 ± 129; cerebellum: KmPlasmaapp 70 ± 114, KmTissueapp 51 ± 85). The relatively narrow range of LNAA concentrations and small number of animals probably contributed to the large variability in the unconstrained estimates.
Estimation of LNAA tissue concentrations
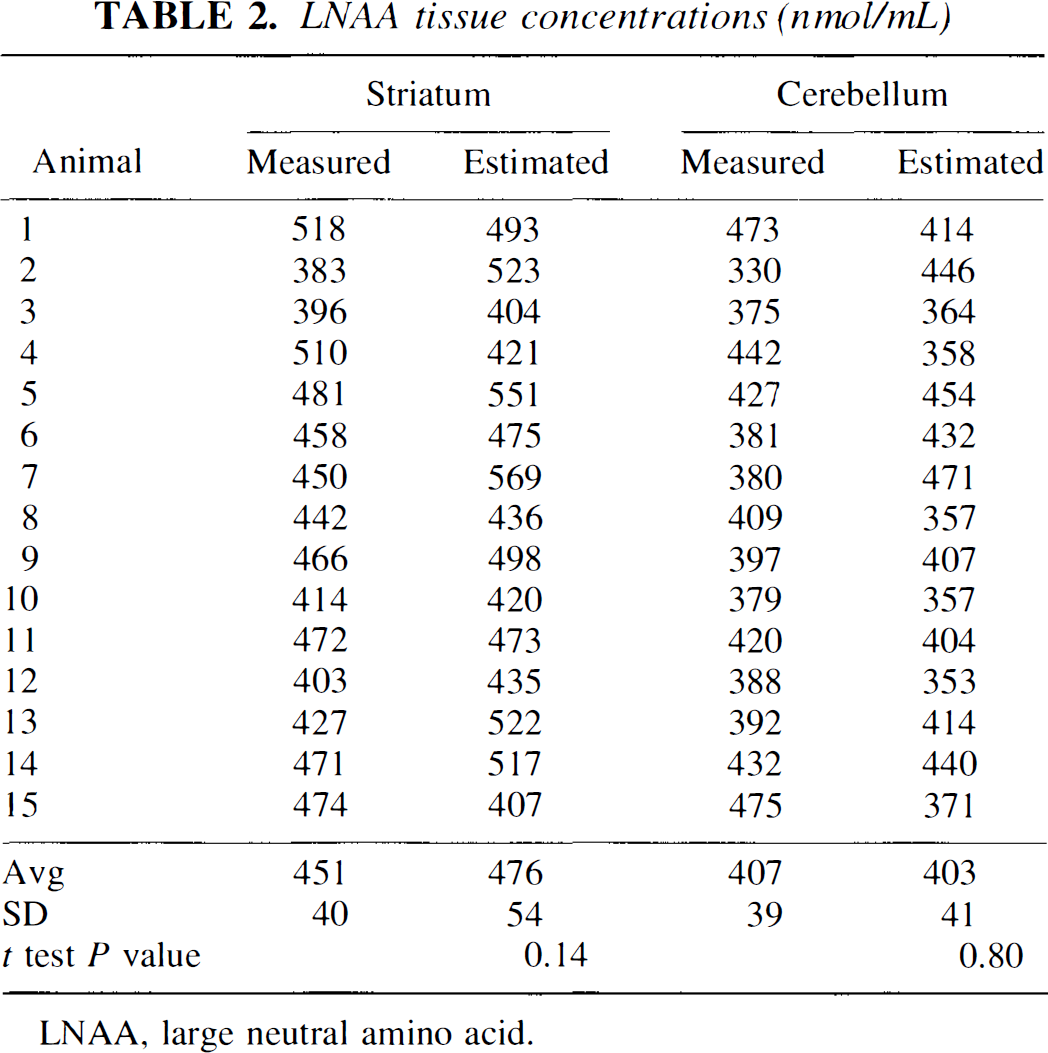
Tissue LNAA concentrations were estimated from Eq. 4 using OMFD concentrations in plasma and tissues and plasma LNAA concentrations, KmPlasmaapp value of 84.3 nmol/mL and KmTissueapp of either 65.9 nmol/mL for striatum or 61.6 nmol/mL for cerebellum. Estimates of tissue LNAA concentrations (Table 2) did not significantly differ from the measured LNAA concentrations (t-test, P > 0.1) in either striatum or cerebellum; however, the individual estimates were poorly corelated with their respective measured values (r = 0.01). The average absolute difference between the estimated and measured measurements was 10% ± 9% for striatum and 12% ± 9% for cerebellum.
LNAA tissue concentrations (nmol/mL)
LNAA, large neutral amino acid.
Diffusion contribution
The addition of a diffusion component to account for inward and outward movement of OMFD across the BBB resulted in only minor changes in the estimated KmTissueapp (Table 3). The diffusion component term (Kd Fraction) was estimated as 0.0053 ± 0.0186, which is not significantly different from zero (F-test). Similar results were obtained for data in the cerebellum (Table 3). Examination of the residual sum of squares by F-test showed that the addition of the diffusion component did not significantly change the fitting results for striatum or cerebellum data, indicating that the present data are consistent with the notion of an insignificant amount of nonsaturable OMFD diffusion across the BBB at normal plasma LNAA conditions.
Diffusion component fitting results
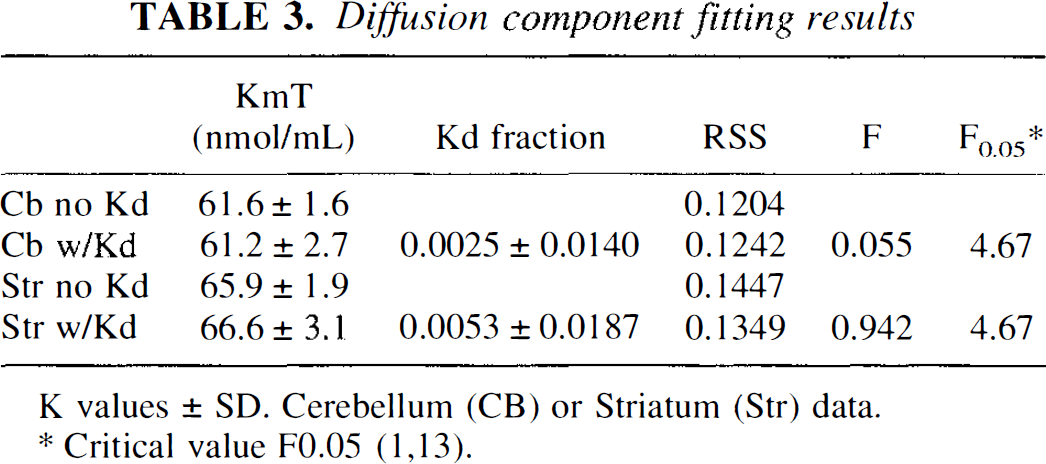
K values ± SD. Cerebellum (CB) or Striatum (Str) data.
Critical value F0.05 (1,13).
DISCUSSION
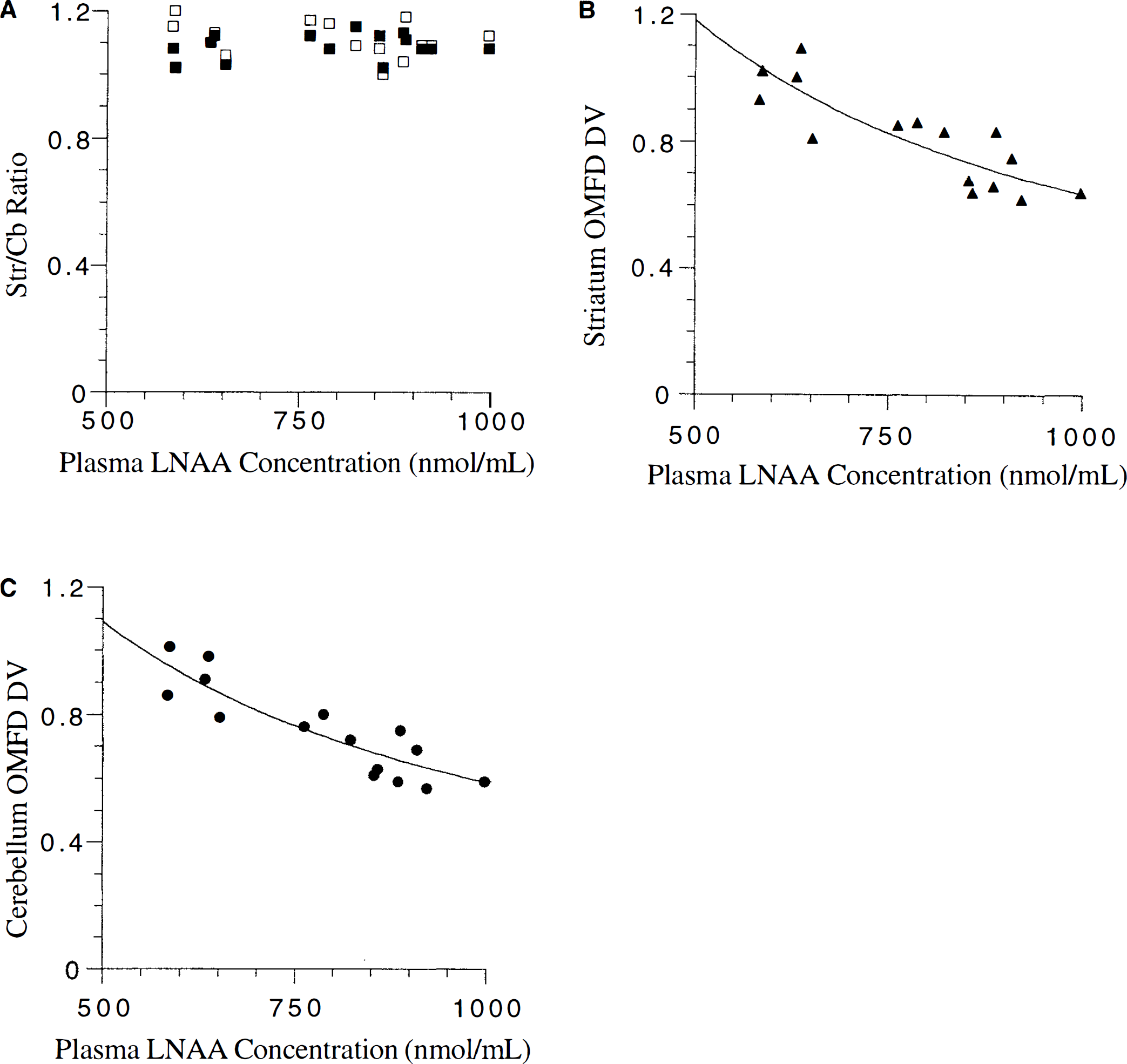
The agreement of OMFD DV data with previous results, using a variety of methods and amino acids (Fig. 4), strengthens the validity of DV variability of labeled LNAA in the brain. In addition, the inert nonspecific uptake of OMFD allowed DV measurements in the striatum, which were not possible in FDOPA experiments because of accumulation of its AAAD-dependent product, 6-[F-18]fluorodopamine.
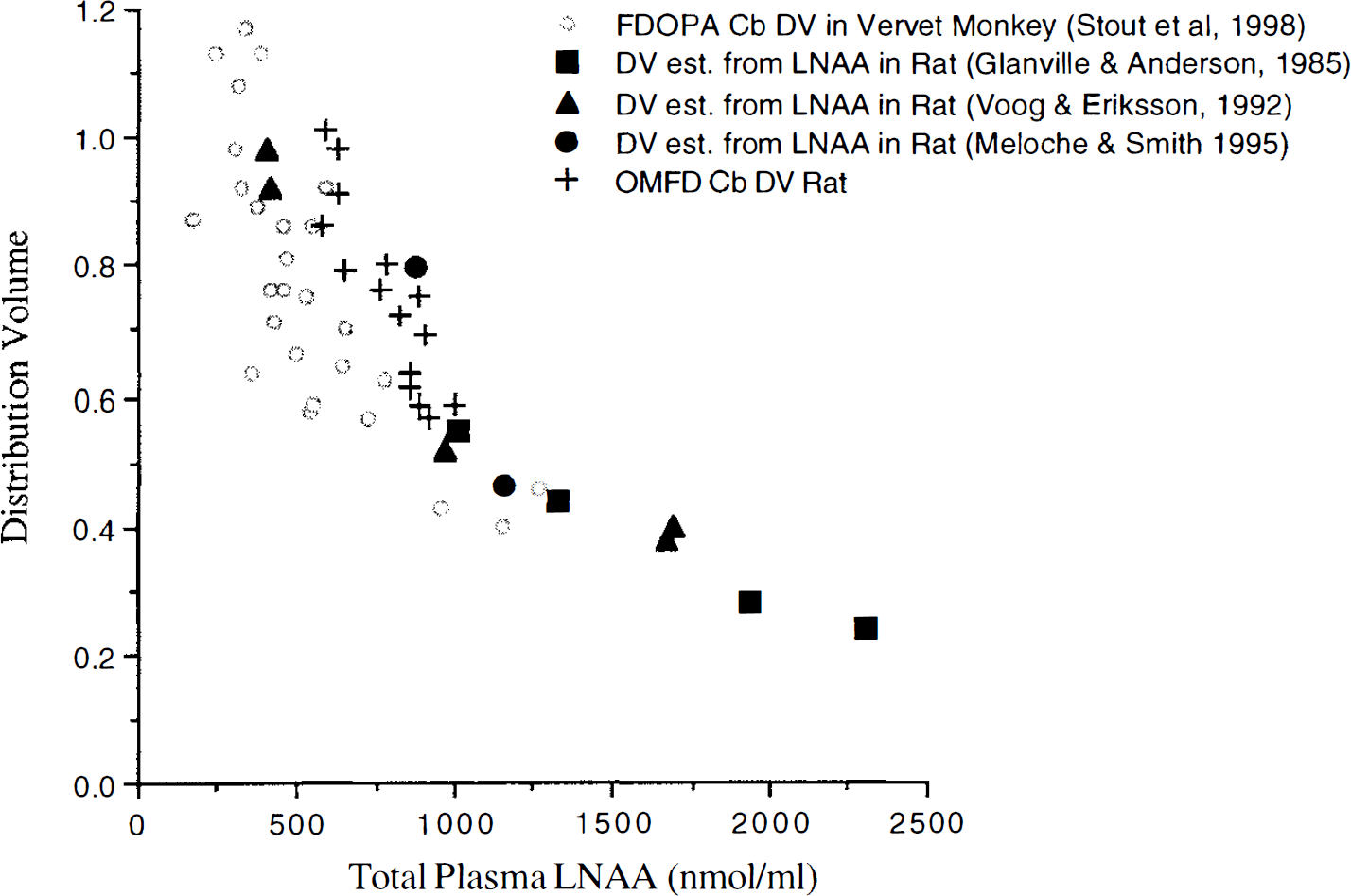
Distribution volume (DV) measurements of FDOPA in vervet monkey cerebellum (Cb), large neutral amino acid (LNAA) in whole rat brain, and OMFD in rat cerebellum versus plasma LNAA concentrations.
Estimates of the diffusion fraction showed that the addition of the diffusion component did not significantly alter the estimates of KmTissueapp. Previous work (Miller et al., 1985) suggested that diffusion could play a significant role with LNAA concentrations over 100 μmol/L, whereas others have indicated diffusion becomes significant only at concentrations over 10 mmol/L (Smith et al., 1987). The current results (Fig. 4) were consistent with those reported by Smith et al. (1987), in which the diffusion component was determined to be insignificant at normal physiologic LNAA concentrations. The current data regarding DV
Tryptophan is the only LNAA that binds to albumin, and some controversy exists over whether the total or unbound fraction should be used to determine the concentration available for competition with other LNAA (Salter et al., 1989; Pardridge, 1977). The authors chose to use the total plasma tryptophan concentration because it has been shown that the albumin-bound fraction will readily dissociate in brain capillaries and become available for transport across the BBB (Pardridge, 1990; Pardridge and Fierer, 1990). In the authors' experiments, tryptophan levels were very consistent, averaging 12%±1% of the total LNAA concentration. Also, because the authors do not expect large changes in the free:bound ratio among the animals resulting from the dietary conditions used, the current data are unlikely to be affected by the exclusion of a bound tryptophan component.
Tissue LNAA concentrations remained fairly constant over a range of plasma LNAA concentrations, which was similar to previous investigations in rats (Glanville and Anderson, 1985; Meloche and Smith, 1995). With LNAA levels affected little by plasma LNAA concentrations, an inverse correlation was observed between plasma LNAA concentrations and DV of an inert labeled LNAA (according to Eq. 2; Fig. 4). The OMFD DV so determined matched well with previous data in rats and monkeys (Huang et al., 1998; Stout et al., 1998) and closely followed Michaelis—Menten transporter kinetics. The constant LNAA tissue levels also resulted in little change in Str/Cb ratios of both OMFD and LNAA over the observed range of plasma LNAA concentrations (Table 1) because both striatum and cerebellum OMFD levels are affected in the same way by plasma LNAA variations.
The use of a bolus injection raises the issue of whether a steady-state or constant OMFD level was achieved and present at the current 60-minute sampling time. The authors examined this issue by creating a simulated OMFD input function and estimating DV
Variations in plasma LNAA levels could potentially affect the clearance of plasma OMFD and affect the constancy of plasma OMFD at 60 minutes, however this is unlikely given the slow clearance and metabolism of OMFD in the body. Furthermore, the majority of clearance occurs during the first several minutes, where the OMFD is distributed into the body. Because this initial distribution occurs predominantly at early times and plasma OMFD levels were stable at 30 to 120 minutes.
The KmPlasmaapp estimate calculated from the plasma LNAA data (84.3 nmol/mL) was quite close to the average KmPlasmaapp reported previously (87.4 nmol/mL; Huang et al., 1998). Estimates of KmPlasmaapp and KmTissueapp without fixing KmPlasmaapp to the plasma-derived value (Eq. 2) resulted in higher values with substantial variability. However, the ratio of KmPlasmaapp to KmTissueapp was the same as that obtained by fixing KmPlasmaapp to the plasma-derived value. This suggests that, whereas the accuracy of the individual estimates might be somewhat uncertain, the ratio of plasma to tissue Kmapp is robust. Interestingly, the tissue and plasma Kmapp values are not equal; there is a higher affinity (that is, lower KmTissueapp) for brain-to-blood transfer. With KmPlasmaapp relatively small compared with normal plasma LNAA concentrations, and with both KmTissueapp and KmPlasmaapp having similar values, the DV of OMFD is approximately equal to the tissue-to-plasma ratio of LNAA (Eq. 2).
A potentially useful application of the Kmapp estimates is that, together with plasma LNAA concentrations, the tissue LNAA concentrations can be estimated using only PET and plasma LNAA data (Eq. 4). This technique may enable tissue LNAA concentrations in brain to be estimated in vivo using noninvasive techniques. The KmPlasmaapp was first derived from plasma data, then subsequently used to obtain the KmTissueapp for striatum and cerebellum. These Kmapp values were used to estimate tissue LNAA concentrations from the PET and blood data. The resulting estimates were not significantly different from the measured values, though the variability was rather high. Although the Kmapp estimates were derived and used on the same set of data, they agreed well with those obtained in rats by others (Huang et al., 1998). This approach may have some use in the diagnosis of diseases that dramatically change the tissue LNAA concentrations. Individual tissue estimates using the current data, which has a standard deviation of 10% to 12% ± 9%, indicate that only large changes in LNAA concentrations (≳20%) would be observable using this method. Further investigation is needed to refine the estimations to provide more sensitive estimates of tissue LNAA concentrations.
The concentrations of LNAA and OMFD were approximately 10% higher in striatum compared with cerebellum. This suggests that the cerebellum may slightly underestimate nonspecific uptake when used as a reference region for FDOPA kinetic modeling. Experiments in monkeys (Doudet et al., 1991) and humans (Wahl et al., 1994; Dhawan et al., 1996) suggested a similar finding, with OMFD having a slightly higher DV in the striatum. However, these groups were not able to establish a significant difference using in vivo imaging techniques, perhaps because of camera resolution and statistical noise limitations.
Slow peripheral OMFD metabolism was observed in rats (~17% per hour), with the formation of a hydrophilic metabolite. This was consistent with other work in monkeys (Doudet et al., 1991), where OMFD metabolites accounted for ~13% of plasma radioactivity after 30 minutes. In humans, several investigations have shown little (5%; Wahl et al., 1994) or no appreciable metabolite formation (Dhawan et al., 1996). Because the OMFD metabolite is highly polar and unlikely to cross the blood—brain barrier, or act as a substrate for the LNAA transporter, the activity associated with the metabolite was subtracted from the plasma in calculations of OMFD distribution volumes. With carbidopa, the amount of plasma metabolite was reduced, but not equal to zero, indicating that: 1) AAAD does not play an important role in metabolite formation, suggesting that OMFD is not decarboxylated into an amine form, which might be trapped in the brain; or 2) peripheral AAAD activity is not completely blocked with the dose of carbidopa administered, even though this dose has been shown to block AAAD activity in FDOPA rat experiments (Melega et al., 1991). The lack of significant effect of carbidopa on metabolite formation also suggests that this plasma metabolite may also be present in FDOPA experiments, where significant amounts of OMFD are peripherally formed and present in the bloodstream. Plasma LNAA concentrations could potentially affect peripheral OMFD metabolism, however a substantial effect on the distribution volume results is unlikely given the slow OMFD metabolism and constant OMFD concentration at 60 minutes. In addition, the concentration of the metabolite is not a concern for the current measurements because it does not cross the BBB due to its polar nature.
Because of the apparent saturation of the LNAA BBB transporter, the distribution volume of OMFD was inversely related to plasma LNAA concentrations. The wide range of normal plasma LNAA concentrations and the associated distribution volume variability suggest that one cannot assume that the DV is constant for labeled LNAA or their analogues. In addition, the distribution volume of OMFD in rat cerebellum appears to be slightly lower than in the striatum, and therefore may underestimate the nonspecific uptake when used as a reference region in FDOPA kinetic modeling. Estimates of LNAA transport constants for plasma, striatum, and cerebellum were similar to those from other experiments. This raises the possibility that tissue LNAA concentrations may be estimated using only blood and PET data of an OMFD determination.
Footnotes
Acknowledgments
The authors thank Ron Sumida, Zarina Kiziloglu, Der-Jen Liu, and Waldemar Ladno for help with the experiments, and the cyclotron staff for isotope production.