Abstract
Quantification of dopamine transporter (DAT) availability with [18F]FE-PE2I PET enables the detection of presynaptic dopamine deficiency and provides a potential progression marker for Parkinson`s disease (PD). Simplified quantification is feasible, but the time window of short acquisition protocols may have a substantial impact on the reliability of striatal binding estimates. Dynamic [18F]FE-PE2I PET data of cross-sectional (33 PD patients, 24 controls), test–retest (9 patients), and longitudinal (12 patients) cohorts were used to assess the variability and reliability of specific binding ratios (SBR) measured during early peak and late pseudo-equilibrium. Receiver operating characteristics area under the curve (PD vs. controls) was high for early (0.996) and late (0.991) SBR. Early SBR provided more favourable effect size, absolute variability, and standard error of measurement than late SBR (caudate: 1.29 vs. 1.23; 6.9% vs. 9.8%; 0.09 vs. 0.20; putamen: 1.75 vs. 1.67; 7.7% vs. 14.0%; 0.08 vs. 0.17). The annual percentage change was comparable for both time windows (−7.2%–8.5%), but decline was significant only for early SBR. Whereas early and late [18F]FE-PE2I PET acquisitions have similar discriminative power to separate PD patients and controls, the early peak equilibrium acquisition can be recommended if [18F]FE-PE2I is used to measure longitudinal changes of DAT availability.
Introduction
The pathophysiology of Parkinson`s disease (PD) is characterized by the loss of dopaminergic cell bodies in the substantia nigra1,2 and the degeneration of nigrostriatal projections. 3 The impairment of dopamine-related neurotransmission in the striatum is linked to most of the classical motor features (i.e. tremor, rigidity, and bradykinesia) of early PD. 4 A key molecule for dopaminergic function is the dopamine transporter (DAT), which is localized on the plasma membrane of presynaptic cell bodies, axons and nerve terminals and removes free dopamine from the synaptic cleft. 5
Molecular imaging of the DAT enables the quantification of the presynaptic neuronal integrity and thereby allows to assess the dopaminergic depletion in patients with regard to their clinical presentation. 6 At present, the main indication for DAT imaging in clinical use is to support differential diagnosis in patients with suspected neurodegenerative parkinsonism by distinguishing PD from essential tremor.7,8 In the last years, DAT quantification got increasingly attention for the use as biomarker in the evaluation of potential disease-modifying treatments and neuroprotective agents in clinical trials. 9 In this context, DAT imaging serves as a secondary outcome measure for treatment effects10,11 and has recently qualified as enrichment biomarker. 12
Several radioligands targeting the DAT have been developed over the last decades, but only few SPECT ligands have reached a broad clinical application.
13
To take advantage of the higher resolution and sensitivity of modern PET systems, the novel radioligand [18F]-(
However, these quantification methods rely on dynamic PET acquisitions over 90 min, which is not always compatible with a clinical setting, where scan time might be limited or patients cannot endure a long scan duration. Previous studies examined to what extent the calculation of the specific binding ratio (SBR) is feasible for [18F]-FE-PE2I PET.19–21 SBR is a simplified quantification method based on static images of a reduced scan time. Besides scan duration, the optimal time window for SBR estimation according to the [18F]-FE-PE2I time activity curves is of particular interest: whereas first results proposed a static acquisition during the radioligands early peak equilibrium, 19 also image data of the late pseudo-equilibrium have been recently used 20 and recommended as more favourable for SBR estimation. 21
This study aims to expand on earlier SBR findings with a larger cohort of PD patients and control subjects and with two subgroups of PD patients, who underwent two [18F]-FE-PE2I PET examinations to assess either test–retest reliability or longitudinal changes after a two-year follow-up. The purpose was to understand whether simplified quantification of DAT is more reliable during the early peak or the late pseudo-equilibrium, and if a reduction of scan time to 18 min is feasible during early and late equilibrium. The time window that provides more reliable quantitative estimates should be preferably used for two potential clinical indications of [18F]-FE-PE2I PET: differential diagnosis of parkinsonism and measurement of disease progression in PD.
Material and methods
Subjects
The participants included in this study were part of three studies approved by the Ethics Committee of the Stockholm Region, by the Swedish Ethical Review Authority, by the Radiation Safety Committee of the Karolinska University Hospital, Stockholm, Sweden, and by the Swedish Medicinal Product Agency. The studies were registered as Clinical Trials in the EudraCT database (2011-002005-30, 2017-001585-19, and 2017-003327-29). The studies were conducted according to the ethical standards of the Ethics Committee of the Stockholm Region and the Swedish Ethical Review Authority, and were in line with the Helsinki Declaration of 1975 (and as revised in 1983). Written, informed consent was obtained from all subjects prior to participation. Healthy controls were recruited by an advertisement in a local newspaper. PD patients were contacted at the Movement Disorder Clinic of the Karolinska University Hospital, Stockholm, Sweden, the Academic Specialist Center, Stockholm, Sweden, and through the Swedish Parkinson patient`s association in Stockholm. All participants underwent the same screening procedure, i.e. exclusion of clinically relevant comorbidities, psychiatric conditions, illicit drug abuse or alcoholism, as assessed by structured interview, physical examination, blood tests, electrocardiogram, and brain MRI. Mini-Mental State Examination was performed to exclude cognitive decline. PD patients fulfilled the clinical diagnosis of PD according to the UK Parkinson Disease Brain Bank criteria. 22 In total, 24 healthy subjects (62 ± 8 years) and 33 PD patients (cross-sectional cohort, 63 ± 9 years) were included. Demographic and clinical data are presented in Table 1 and in the Supplemental Data.
Demographic and clinical data of controls and PD patient cohorts.
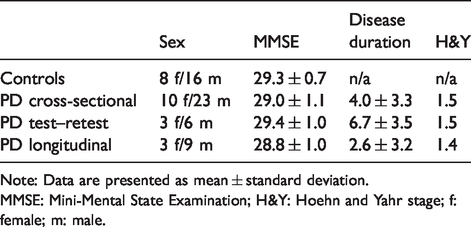
Note: Data are presented as mean ± standard deviation.
MMSE: Mini-Mental State Examination; H&Y: Hoehn and Yahr stage; f: female; m: male.
Imaging procedures
All participants underwent brain MRI scans on a 3 Tesla system (Discovery MR750; GE Healthcare) prior to PET examination as part of the initial evaluation and to delineate anatomic brain volumes of interests (VOI). Two subgroups of patients were invited to perform a second [18F]-FE-PE2I PET examination with the purpose to investigate test–retest reliability (10 PD patients) or longitudinal DAT binding changes (20 PD patients). The final numbers of patients, who had two [18F]-FE-PE2I PET examinations of sufficient quality for data analysis were reduced due to various reasons, such as technical failure during the scan, loss of contact with the patient, or inability/unwillingness to participate in a second PET examination at follow-up. They underwent PET scans either within 12 ± 8 days (test–retest cohort: 9 PD patients, 65 ± 7 years) or after 2 ± 0 years (longitudinal cohort: 12 PD patients, 62 ± 8 years). All patients performed PET measurements after suspension of dopaminergic replacement therapies for at least 12 h. [18F]-FE-PE2I was prepared via nucleophilic radiofluorination of its tosylate precursor as previously described. 23 Details of molar activity, injected radioactivity and injected mass are provided in the Supplemental Material.
Dynamic PET measurements were obtained using a high-resolution research tomograph (HRRT) system (Siemens Medical Solutions). A 6-min transmission scan with a 137Cs source was performed for attenuation correction. [18F]-FE-PE2I was injected as i.v. bolus over 10 s, and the catheter was flushed with 10 mL NaCl. Emission data were acquired in list mode over 93 min. PET data were reconstructed in 37 frames of increasing duration (8 × 10 s, 5 × 20 s, 4 × 30 s, 4 × 60 s, 4 × 180 s, 12 × 360 s) using three-dimensional ordinary Poisson ordered subset expectation maximization with modelling of the system`s point spread function. Frame-to-frame motion correction of reconstructed images was applied as previously described. 24
Image analysis and DAT quantification
Image processing and analysis were performed using an in-house pipeline named Solena written in MATLAB (MATLAB r2014b, The MathWorks, Inc.). Within Solena, T1-weighted MP-RAGE sequences of each individual were segmented with FreeSurfer (FreeSurfer v6.0.0, http://surfer.nmr.mgh.harvard.edu/)
25
and the generated segmentation masks were used to define VOIs of the caudate nucleus and the putamen, and one reference region containing the cerebellum. Subsequently, MRI and dynamic PET images were co-registered. Different outcome measures were used to quantify DAT density in each VOI: dynamic PET data were analysed with simplified reference tissue model (SRTM)
26
to estimate binding potential (
Statistical analysis
All analyses were performed using the statistics software R (R v3.6.1, http://www.R-project.org/).
Cross-sectional cohort and control subjects
Linear regression analysis and 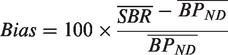
The coefficient of variation (COV) was used as a measure of variability and obtained by dividing the standard deviation (σ) by the mean (µ) of each outcome measure
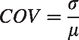
Furthermore, Cohen`s effect size 
Lastly, we evaluated group differences with a two-sample
Test–retest cohort
Test–retest data were used to evaluate the agreement and reliability of outcome measures.
27
The absolute variability (AbsVar) refers to the agreement between the two measurements and was calculated for 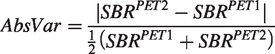
Intraclass correlation coefficient (ICC) was assessed for all binding estimates as a measure of reliability and differentiability and was calculated according to a one-way random model.
28
We also calculated the standard error of measurement (SEM), which indicates the precision of the individual’s binding estimate and is expressed by the standard error of each measurement around the estimated binding value.
29
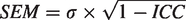
Longitudinal cohort
Group differences of binding estimates between baseline and two-year follow-up PET measurements were evaluated by using the paired 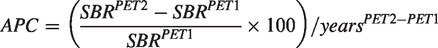
Results
Linear regression analysis and dispersion metrics
Regression analysis showed that early (
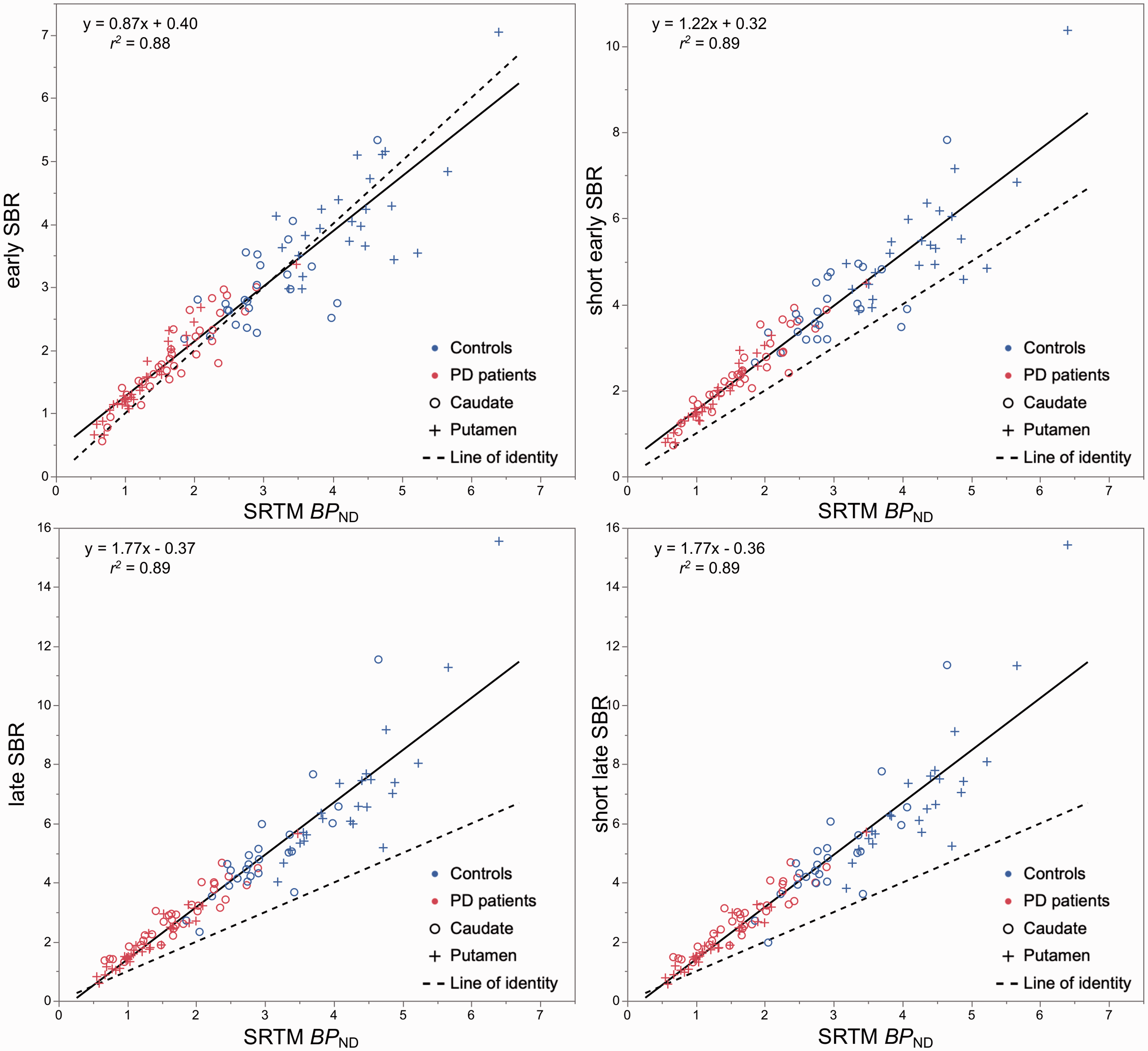
Scatter plots and linear regression analysis between specific binding ratio (SBR) during early peak (left) and late pseudo-equilibrium (right), and binding potential (
Binding estimates and bias in controls and PD patients.
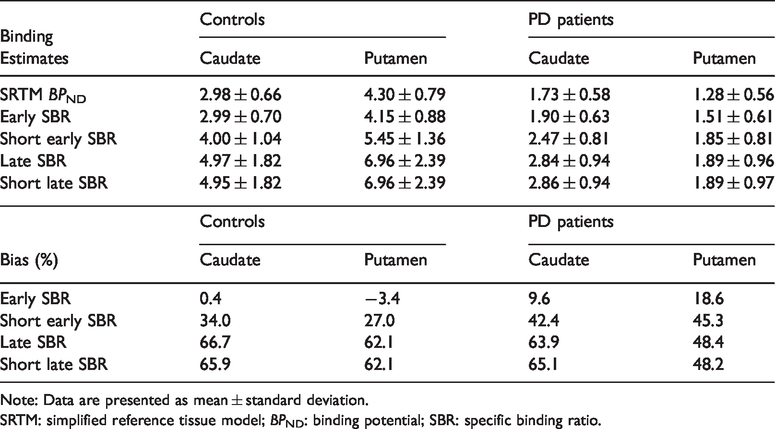
Note: Data are presented as mean ± standard deviation.
SRTM: simplified reference tissue model;
Effect size, variability, test–retest metrics and longitudinal change of DAT binding measures.
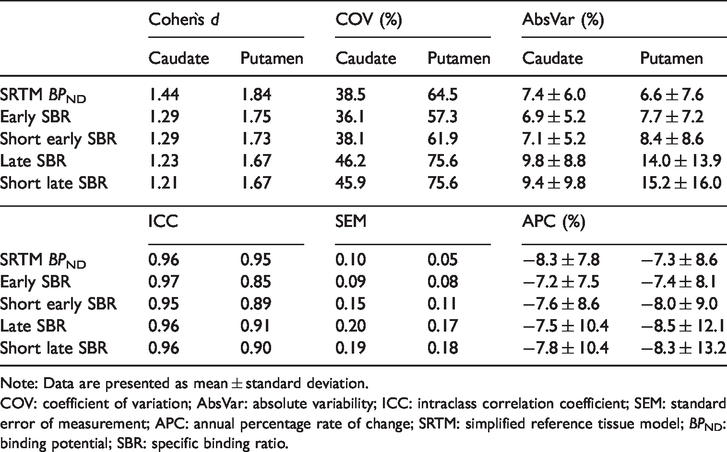
Note: Data are presented as mean ± standard deviation.
COV: coefficient of variation; AbsVar: absolute variability; ICC: intraclass correlation coefficient; SEM: standard error of measurement; APC: annual percentage rate of change; SRTM: simplified reference tissue model;
Discriminative analysis between PD patients and controls
Similar effect sizes in the caudate and the putamen were observed for all five DAT binding measures. Highest Cohen`s
Test–retest metrics
Test–retest agreement and reliability of binding estimates were overall slightly better for early SBR measures as compared to late SBR measures (Table 3 and Figure 2). The average AbsVar was similar for
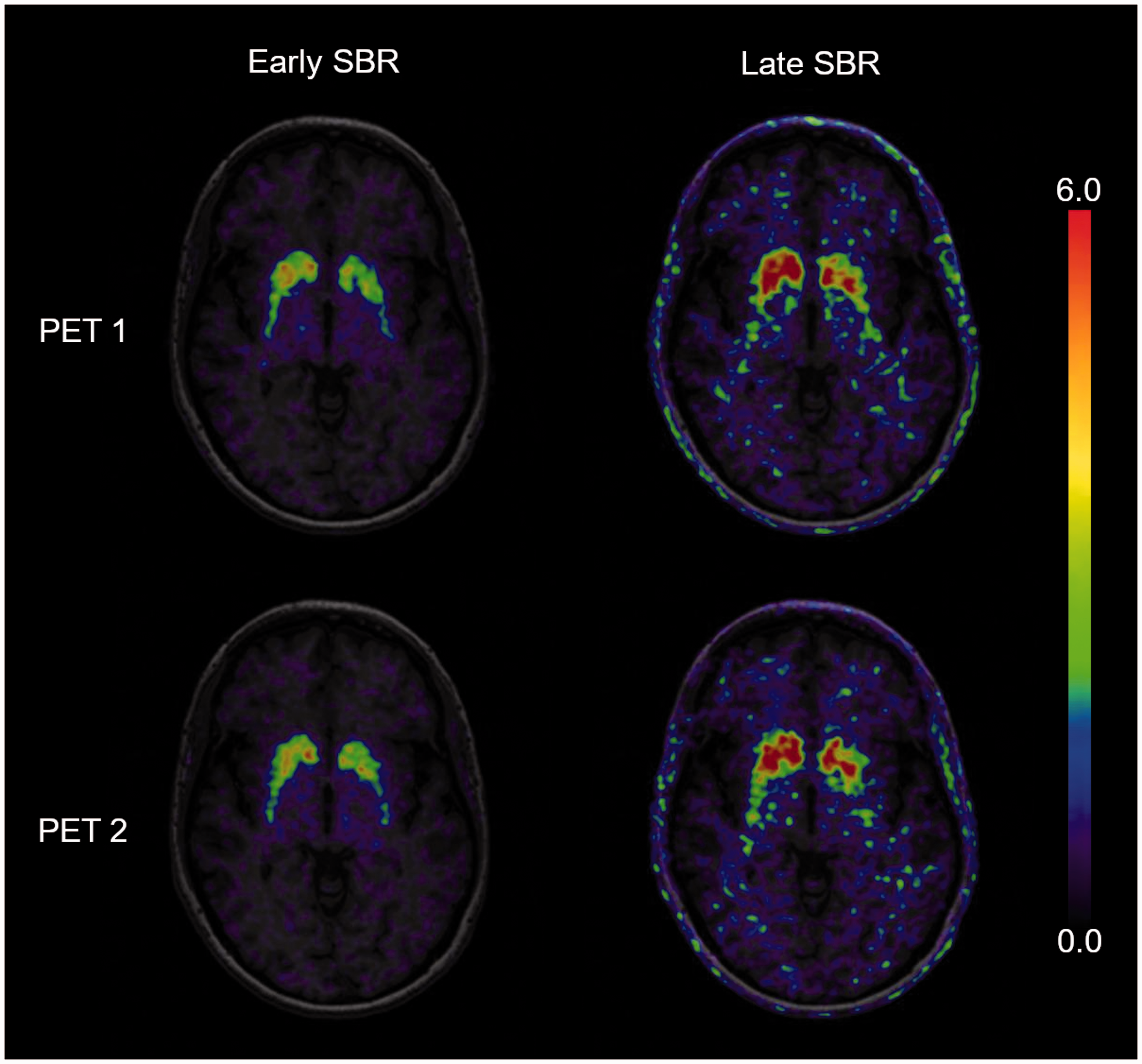
Specific binding ratios (SBR) during the early peak (left) and the late pseudo-equilibrium (right) of one patient, performing two [18F]-FEPE2I PET scans within seven days. The cerebral uptake is scaled to the cerebellum with subtraction of the unspecific binding in the reference region.
Longitudinal DAT binding changes
The average APC was in a narrow range (−7.2% – 8.5%) and almost equal for all binding measures in both striatal regions (Table 3). However, when comparing the DAT binding after two-year follow-up with baseline values, significant within-group differences were observed for
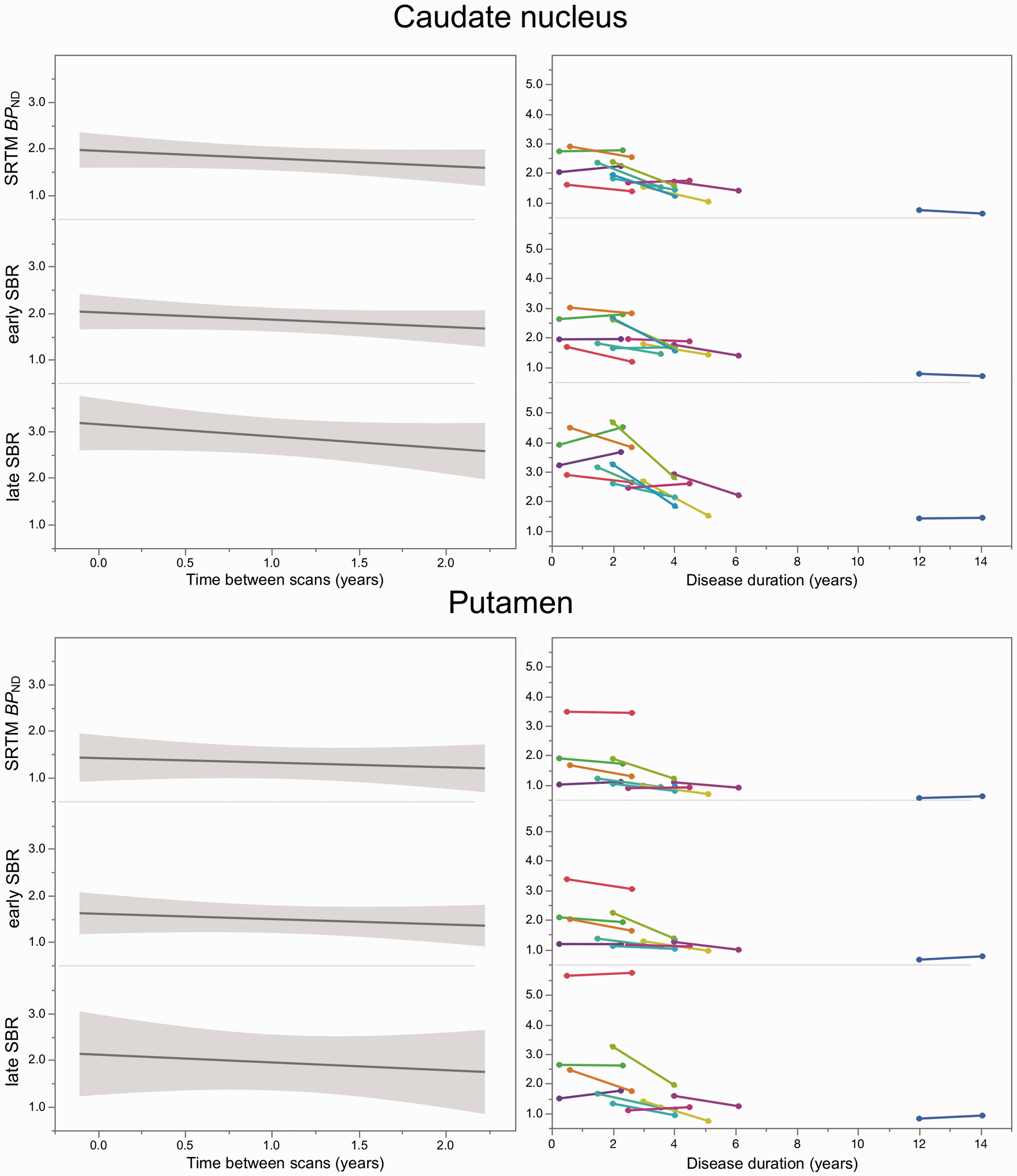
Slope of dopamine transporter (DAT) binding estimates between baseline and two-year follow-up (left panels; with grey shaded 95% confidence intervals). Right panels show the longitudinal changes of the individuals’ DAT binding estimates plotted over disease duration. SRTM: simplified reference tissue model;
Discussion
The aim of this study was to directly compare the performance of the simplified quantification of [18F]-FE-PE2I PET during early peak and during late pseudo-equilibrium in the context of potential future indications in clinical routine. There are two main findings of this study: first, the SBRs of both equilibrium states showed similar and high discriminative values to differentiate PD patients from controls; second, SBR during the early equilibrium provided less variability and a more favourable reliability than late SBR, suggesting that early SBR should be chosen for simplified DAT quantification.
Correlation, bias, effect size and discriminative power
Correlational analyses with the
Slight differences were found in comparison to previous results. One recent study showed an AUC of only 0.89 for the SBR of [18F]-FE-PE2I during the late equilibrium.
20
The authors attributed this to the fact that the clinical diagnosis of PD was not confirmed at follow-up in four of the patients enrolled as PD, causing normal DAT binding values in the PD cohort. Patients that fulfil the clinical criteria of PD, but show normal DAT binding in the putamen are often referred to as subjects with a scan without evidence of dopaminergic deficit (SWEDD) and are unlikely to maintain the diagnosis of PD after long-term follow-up.
30
In our study, only one patient (who also participated in the longitudinal study) belongs to this group. The diagnostic accuracy may, therefore, differ between study cohorts. However, all evaluated SBRs agreed with the reference
Test–retest metrics and longitudinal analysis
The precise and reliable quantification of DAT binding is of utmost importance for within-subject comparison. In this study, although the ICC was similar for
In view of the need to accelerate drug discovery and development for the treatment of alpha-synucleinopathies,37,38 the reduction of sample size and study duration is of great relevance to reduce costs and resources. Methodological advancements support this aim, but do not overcome general shortcomings of DAT imaging to measure PD progression and severity, e.g. the flooring effect of DAT availability at advanced disease stages, or the involvement of several brain regions and neurotransmitter systems to PD pathology. 39 Likewise, possible subtle effects of dopaminergic treatment on DAT binding cannot be excluded,40–42 particularly in longitudinal evaluations as in our cohort, in which the levodopa equivalent daily dose increased from 238 ± 204 at baseline to 596 ± 391 at follow-up. To best possible control for such effects in this study, all patients suspended antiparkinsonian medication before PET examinations.
Choice of short imaging protocol
According to the overall results, if a dynamic acquisition >60 min is not feasible, the imaging protocol of [18F]-FE-PE2I PET should follow the purpose. The use of late SBR measures suffices for clinical applications in patients with parkinsonism or when DAT-PET is used as enrichment biomarker in clinical trials. On the other hand, an acquisition protocol covering the early equilibrium should be the preferred method if DAT imaging is used as quantitative marker. This might be the case if DAT-PET with [18F]-FE-PE2I is used in clinical trials or as research tool for correlation with clinical parameters (e.g. motor scores, neuropsychological measures, cognition and behaviour). For both equilibrium states, a better count statistic due to an acquisition over 30 min provides less variability and a better reliability as compared to a scan duration of 18 min, and therefore should be considered if possible.
Limitations
This study has some limitations. Although the aim is to support the translation of [18F]-FE-PE2I PET into clinical practice, the data were acquired with a research PET system. To simulate a clinical setting, the HRRT image data were smoothed to a resolution comparable to data derived from a clinical PET system. The analysis of these simulated data (see Supplemental material) confirmed our main findings, though additional studies are needed to corroborate the results obtained with the high-resolution system. In particular, the process of smoothing the data to lower the resolution of the reconstructed images produced a decrease in the SBR values. The overestimation of SBR in comparison with the gold standard
Conclusion
Simplified quantification of [18F]-FE-PE2I PET during either early peak or late pseudo-equilibrium and scan time reduction to 18 min retains discriminative power to separate PD patients and healthy controls. Since early SBR shows better reliability, the acquisition during early peak equilibrium is preferable if [18F]-FE-PE2I PET is used as quantitative estimate for disease severity and progression.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20958755 - Supplemental material for Simplified quantification of [18F]FE-PE2I PET in Parkinson’s disease: Discriminative power, test–retest reliability and longitudinal validity during early peak and late pseudo-equilibrium
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20958755 for Simplified quantification of [18F]FE-PE2I PET in Parkinson’s disease: Discriminative power, test–retest reliability and longitudinal validity during early peak and late pseudo-equilibrium by Joachim Brumberg, Vera Kerstens, Zsolt Cselényi, Per Svenningsson, Mathias Sundgren, Patrik Fazio and Andrea Varrone in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work has been supported by a grant from the Swedish Foundation for Strategic Research and by grants to Andrea Varrone from the Swedish Science Council, from a private donation, the Åhlen Foundation, and from H. Lundbeck A/S. Joachim Brumberg received a scholarship from the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG, grant BR 6121/1-1).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Joachim Brumberg, Vera Kerstens, Zsolt Cselényi, Per Svenningsson, Mathias Sundgren. Patrik Fazio, and Andrea Varrone, report no conflict of interest.
Authors’ contributions
JB and AV designed and conceptualized the study, analysed and interpreted the data and drafted the manuscript. VK and PF acquired the data, analysed and interpreted the data and revised the manuscript. ZC analysed and interpreted the data and revised the manuscript. PS, and MS interpreted the data and revised the manuscript. All authors read and approved the final manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
