Abstract
Reversible focal ischemia may lead to delayed tissue injury despite primary restoration of blood flow and metabolism. The authors investigated whether such delayed changes also occur after thrombolytic treatment of thromboembolic stroke. Clot embolism of the middle cerebral artery (MCA) was produced in C57/B16J mice by intracarotid injection of heterologous clots. One hour after embolism, one group was treated with intracarotid infusion of rt-PA (10 mg/kg). The untreated control group received an equal amount of vehicle. Just before onset of treatment and after 1, 3, 6, and 24 hours, animals were frozen in situ and cerebral blood flow (CBF), cerebral protein synthesis (CPS), ATP content, and DNA fragmentations (TUNEL) were imaged on cryostat sections using double tracer autoradiography, bioluminescence, and immunohistochemical techniques, respectively. In untreated animals (n = 20), CPS was suppressed in approximately 68% of hemispheric transsection at 1 hour after embolization. The ATP depleted area was smaller (approximately 58%), but between 6 and 24 hours it merged with that of CPS suppression. TUNEL-positive neurons became visible between 6 and 24 hours exclusively in regions with ATP depletion. rt-PA-induced thrombolysis (n = 20) led to the gradual improvement of blood flow. At 24 hours, ATP depletion was fully reversed and the CPS suppression area declined to approximately 16% of hemispheric transsection. Despite progressive metabolic recovery, large numbers of neurons became TUNEL-positive and animals died between 24 and 48 hours. Thrombolysis after clot embolism restores metabolic activity including protein synthesis, but the therapeutic benefit is limited by secondary injury that requires additional treatment to improve final outcome.
Experimental and clinical studies have established that thrombolytic treatment of stroke improves neurologic outcome (Hacke et al., 1999), but the degree of improvement is still unsatisfactory. Under clinical conditions this may be explained by the long treatment delay, but the experimental data demonstrate that initiation of therapy after as early as 15 minutes after clot embolism reverses ischemic brain damage only partially (Busch et al., 1998; Kilic et al., 2000). Two different pathophysiologic events may account for this failure. The first event is the noninstantaneous restoration of blood flow that according to earlier studies of transient global ischemia may aggravate ischemic damage because of the development of massive postischemic brain edema (Hossmann, 1997). The second pathophysiologically important event is the molecular evolution of delayed postischemic damage (Lipton, 1999; Siesjöet al., 1999). Over the past years, evidence has accumulated that transient occlusion of the MCA brought about by reversible insertion of an intraluminal nylon thread produces delayed infarction, the volume of which may approximate that which is observed after permanent vascular occlusion (Du et al., 1996; Kuroda and Siesjö, 1997). In a recent study of reversible thread occlusion in mice, the authors investigated the temporal and regional evolution of delayed tissue injury using a battery of pictorial methods that allowed the simultaneous imaging of various metabolic and genomic variables (Hata et al., 2000a). This study revealed that reperfusion after 1 hour MCA occlusion led to the full recovery of energy metabolism, but not of protein biosynthesis, and that delayed injury was initiated by secondary energy failure in the region of persisting inhibition of protein synthesis. Obviously, the understanding of the mechanisms of this secondary injury is of considerable interest for thrombolytic treatment because the combination of hemodynamic with molecular interventions may improve the final outcome of stroke (Steiner and Hacke, 1998).
However, before proceeding to the detailed analysis of these delayed injury mechanisms, it should be clarified whether metabolic recovery after thrombolysis-induced reperfusion is replicated by reversible thread occlusion. There are, in fact, serious concerns about the validity of this assumption. During thrombolysis, cerebral blood flow is resumed much more slowly which could be deleterious if this delay enhances the formation of ischemic brain edema (Hossmann, 1997). There are also concerns about the pharmacologic side effects of thrombolytic agents, such as the previously documented aggravation by tissue plasminogen activator of ischemic and excitotoxic injury (Tsirka et al., 1995; Wang et al., 1998; Nagai et al., 1999). Therefore, in the current investigation, the authors used the same multiparametric imaging approach as in their previous thread occlusion study (Hata et al., 2000a) to investigate the evolution of brain infarction after middle cerebral clot embolism treated by thrombolysis with recombinant tissue plasminogen activator. The authors' observations suggest that there are, in fact, major differences between the two models of reperfusion which must be taken into account when novel molecular treatment strategies are developed to improve the outcome of thrombolytic treatment of stroke.
MATERIALS AND METHODS
Experiments were performed in accordance with the National Institutes of Health guidelines for the care and use of laboratory animals, and approved by the local governmental authorities (Regierungspräsident Köln, registration number 7/98). Animals were housed under diurnal lighting conditions and allowed access to food and water ad libitum until the day of the experiment. Anesthesia was induced by 1.5% halothane and maintained with 1% halothane in 70% N2O and 30% O2.
Animal groups
Sixty-three adult male C57Black/6J mice weighing 20 to 30 g were submitted to middle cerebral artery (MCA) clot embolism (Kilic et al., 1998) followed by treatment with or without intraarterial rt-PA at 1 hour after embolization. Blood flow in the MCA territory was monitored by transcranial laser–Doppler flowmetry. Based on these recordings, three animals were excluded because of spontaneous reperfusion, and 5 rt-PA treated animals were excluded because of unsuccessful recanalization. From the remaining 55 animals, 25 were treated with rt-PA at 1 hour after embolization (treated group), 25 received vehicle infusion without rt-PA at 1 hour after embolization (untreated group), and 5 were killed 1 hour after embolization. Survival times after rt-PA or vehicle infusion were 1, 3, 6, and 24 hours, respectively (n = 5 per time point). Five treated and 5 untreated animals selected for 3 days survival died before the termination of the experiment.
Clot preparation
First, 0.4 mL of fresh heterologous arterial blood was mixed with 0.08 mL of thrombin (1 mg/mL; Sigma, St. Louis, MO, U.S.A.) and immediately injected into a polyethylene PE10 catheter, in which the blood was allowed to clot at ambient pressure for 2 hours at 37°C, followed by 22 hours clot stabilization at 4 °C. The clots were then taken from the catheter, rinsed in saline to remove red blood cells, and inspected under the microscope for selection of fibrin rich segments. These segments were cut into cylindrical emboli of 150 μm in diameter and 4 mm in length, suspended into phosphate-buffered saline (PBS), and drawn into a PE10 catheter for subsequent embolization.
Surgical procedures
Rectal temperature was maintained between 36.5°C and 37°C using a feedback-controlled heating system. Laser–Doppler flow (LDF) was recorded with a flexible probe attached to the skull above the territory of the MCA, 2 mm caudal to bregma and 6 mm lateral to midline. After midline neck incision, the right common carotid artery was isolated and the external carotid and thyroid and occipital arteries were ligated and cut. After ligation of the pterygopalatine artery, two microvascular clips (FE691; Aesculap, Tuttlingen, Germany) were temporarily placed on the common and internal carotid arteries. A small arteriotomy was made on the external carotid artery and the PE10 catheter loaded with 4 clots of 150 μm in diameter and 4 mm in length was inserted into the external carotid artery. The tip of the catheter was placed close to the carotid bifurcation. After removal of the clip from the internal carotid artery, the clots were flushed into the internal carotid artery over a period of 5 seconds.
rt-PA treatment
Thrombolysis was performed by intracarotid infusion of 10 mg/kg human single-chain rt-PA, Actilyse (INN: Alteplas, Boehringer Ingelheim, Germany) dissolved in 0.1 mL distilled water (vehicle). This dose was chosen to account for the high resistance of murine clots to human rt-PA (Lijnen et al., 1994). Infusion of rt-PA started 1 hour after clot embolism and continued for 30 minutes. Untreated animals received an infusion of the vehicle without rt-PA.
Forty-five minutes before death, L-(4,5-3H)-leucine (300 μCi/animal, specific activity 151 Ci/mmol; Amersham, Braunschweig, Germany) was administered intraperitoneally for the evaluation of the cerebral protein synthesis (CPS) rate (Mies et al., 1997). Two minutes before sacrifice, 4-iodo-N-methyl-[14C]antipyrine (IAP; 20 μCi/animal, specific activity 40 to 60 mCi/mmol; Biotrend Chemicals, Cologne, Germany) dissolved in 0.2 mL saline was injected intraperitoneally for measurement of cerebral blood flow (CBF) (Maeda et al., 2000). The experiments were terminated by in situ freezing. Brains were removed in a cold temperature cabinet (−20°C) and cut into 20 μm coronal cryostat sections at −20°C. Sections were mounted on coverslips for ATP-bioluminescence, on object holders for CPS and CBF autoradiography, and on poly-L-lysine coated slides for terminal transferase biotinylated-UTP nick end labeling (TUNEL).
Regional measurement of ATP, blood flow, and protein synthesis
Pictorial measurements of ATP were performed using ATP specific bioluminescence (Kogure and Alonso, 1978). For measurement of CBF, brain slices were dried on a hot plate and exposed for 10 days to X-ray film (Hyperfilm β-max; Amersham) with calibrated [14C] standards (Maeda et al., 2000). Then brain slices were incubated in 10% trichloroacetic acid (TCA) to remove [14C]iodoantipyrine and [3H]-labeled free leucine and metabolites other than proteins for the measurement of CPS. Subsequently, slices were exposed for 14 days with 3H standards to tritium-sensitive X-ray film (Hyperfilm 3H; Amersham) for autoradiography of 3H-labeled proteins (Mies et al., 1997).
Morphometric analysis of ischemia-induced metabolic disturbance and flow changes
Bioluminescence, autoradiographic images, and silver impregnated sections were digitized with a CCD camera system and analyzed using the National Institutes of Health image software. Impairment of energy metabolism was defined as the decline of ATP to less than 30% of the mean value of the contralateral side. The threshold for CPS inhibition was set to the lowest CPS value of the nonischemic hemisphere excluding fiber tracts. Local CBF was calculated according to Sakurada et al. (1978). The integrated tracer input radioactivity was estimated from final heart blood sampling, assuming linearity of [14C] IAP radioactivity between tracer injection and termination of the experiment (Maeda et al., 2000). The threshold for the critical reduction of CBF was set at 20% of the opposite hemisphere.
The areas of critical CBF reduction, ATP depletion, CPS inhibition, and tissue injury (loss of silver impregnation) were outlined and expressed as percent of the edema-corrected hemispheric transsection (Swanson et al., 1990). The outlines of ATP and CPS suppression were superimposed to demarcate the penumbra, that is, the metabolically dissociated area in which protein synthesis is suppressed but ATP is preserved, from the infarct core and the metabolically intact periinfarct surrounding.
Terminal transferase biotinylated-UTP nick end labeling
TUNEL was performed as described previously (Hata et al., 2000b). Briefly, coronal brain sections were fixed for 15 minutes in ice-cold 4% paraformaldehyde phosphate-buffered saline (PBS), pH 7.4. Subsequently, the sections were washed twice in 70% ethanol (1 minute), once in PBS (3 minutes), once in 0.3% hydrogen peroxide PBS (5 minutes), and then again in PBS (5 minutes). After equilibration for 15 minutes in TDT buffer (100 mmol/L potassium cacodylate, 2 mmol/L cobalt chloride, 0.2 mmol/L dithiothreitol), the buffer was quantitatively removed, sections were incubated in 50 μL reaction mix (10 pmol/L biotin-16-dUTP (Boehringer, Mannheim, Germany) and 150 U/mL terminal deoxynucleotidyl transferase (Life Technologies, Eggenstein, Germany)) in TDT buffer, and covered with a coverslip. After incubation for 60 minutes at 37°C, the reaction was terminated by washing the sections for 15 minutes in TB buffer (300 mmol/L sodium chloride, 30 mmol/L sodium citrate). Incorporated biotin was visualized using the avidin-biotin-peroxidase complex method (Vector Laboratories, Burlingame, CA, U.S.A.) as recommended by the supplier. Finally, the sections were dehydrated and embedded in Eukitt (Kindler GmbH, Freiburg, Germany).
Histology
Histologic evaluation was performed using silver impregnation (Vogel et al., 1999). After the development of autoradiographic images (CBF and CPS), sections were exposed to the silver impregnation solution for 2 minutes. The slices then were rinsed in distilled water 6 times for 1 minute and transferred to a developer solution for 3 minutes. After further rinsing in distilled water, 3 times for 1 minute, they were air-dried and scanned with a CCD camera.
Incidence maps of regional alterations
To evaluate the regional reproducibility of TUNEL, as compared with the reduction of CBF, CPS, and ATP, regional incidence maps were constructed. The areas of biochemical disturbances were outlined on representative brain sections of each individual experiment as described above and superimposed at the level of caudate-putamen. Using the image analysis software, the incidence of flow and metabolic alterations was calculated for each pixel and expressed in percent of the number of animals in each group.
Statistics
All values are given as means ± SD. Differences were evaluated by one-way analysis of variance followed by Bonferroni's multiple comparison test. P < 0.05 was considered to indicate statistical significance.
RESULTS
General physiologic observations and mortality
During the initial 2 hours of vascular occlusion, no significant changes in mean arterial blood pressure occurred. There were also no significant group differences in arterial pO2, arterial pCO2, or pH values between rt-PA treated and vehicle treated animals (Table 1). Up to 24 hours after embolism, mortality of animals was under 20% in both experimental groups. However, with longer survival times mortality increased, and all animals selected for more than 24 hours observation (n = 5 per group) consistently died between the first and third day. Brain autopsy performed in 5 of these animals (3 treated and 2 untreated mice) consistently revealed transtentorial herniation caused by severe brain edema. One treated mouse in addition exhibited massive intracerebral hemorrhage.
Physiologic variables after clot embolism of middle cerebral artery in mice
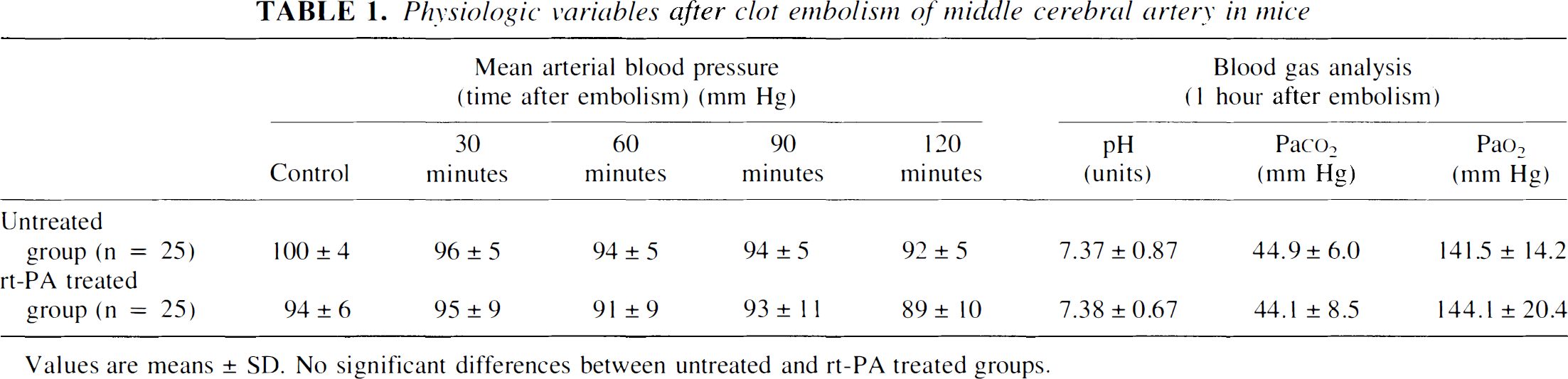
Values are means ± SD. No significant differences between untreated and rt-PA treated groups.
Laser–Doppler flowmetry
After the injection of clots LDF decreased sharply to below 20% of preischemic control in all animals (Fig. 1). In 25 out of 28 untreated animals, blood flow remained at this level until the end of the observation time (2 hours after clot embolism); the other 3 animals exhibited spontaneous reperfusion and were excluded. Infusion of rt-PA starting 1 hour after embolism led to gradually improving blood flow within 60 minutes of treatment in 25 out of 30 animals; the other 5 animals did not respond to the treatment and were also excluded. In the successfully treated animals, LDF returned to approximately 80% of control at the end of the observation time (Fig. 1).
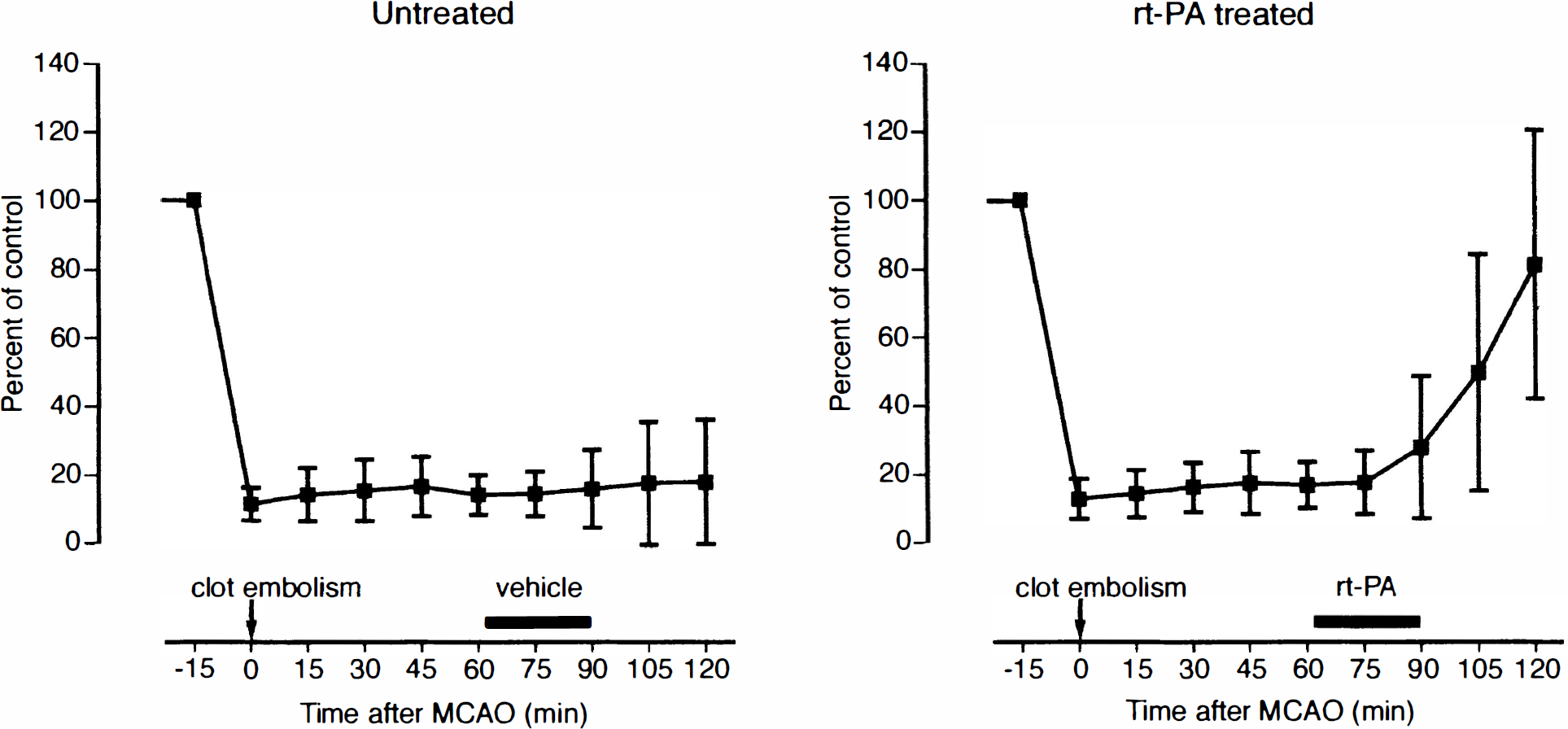
Laser–Doppler recording of cortical blood flow after clot embolism of middle cerebral artery. One hour after embolism, animals received either an intracarotid infusion of rt-PA (treated group, right) or of vehicle (untreated group, left). Note gradually progressing restoration of blood flow after rt-PA treatment. MCAO, middle cerebral artery occlusion. Values are means ± SD; n = 25 per group.
Regional blood flow and metabolism
After clot embolism, regional CBF and metabolism was measured at the following time points: 1 hour after clot embolism and 1, 3, 6, and 24 hours after infusion of vehicle (untreated group) or rt-PA (treated group). Animals selected for 3 days recovery did not survive in either group.
One hour after embolism, the area of critically reduced CBF (that is, CBF decline < 20% of control) was 57% of the ipsilateral hemisphere (Figs. 2 and 3). This area corresponded to that of ATP depletion (58.3 ± 10.1%) but was smaller than that of CPS suppression (68.0 ± 6.7%), which agrees with the previously established higher threshold of CPS (Mies et al., 1991). The flow rate in the center of the ATP-depleted area was 6.2 ± 3% of that in the homotopic region of the contralateral hemisphere.
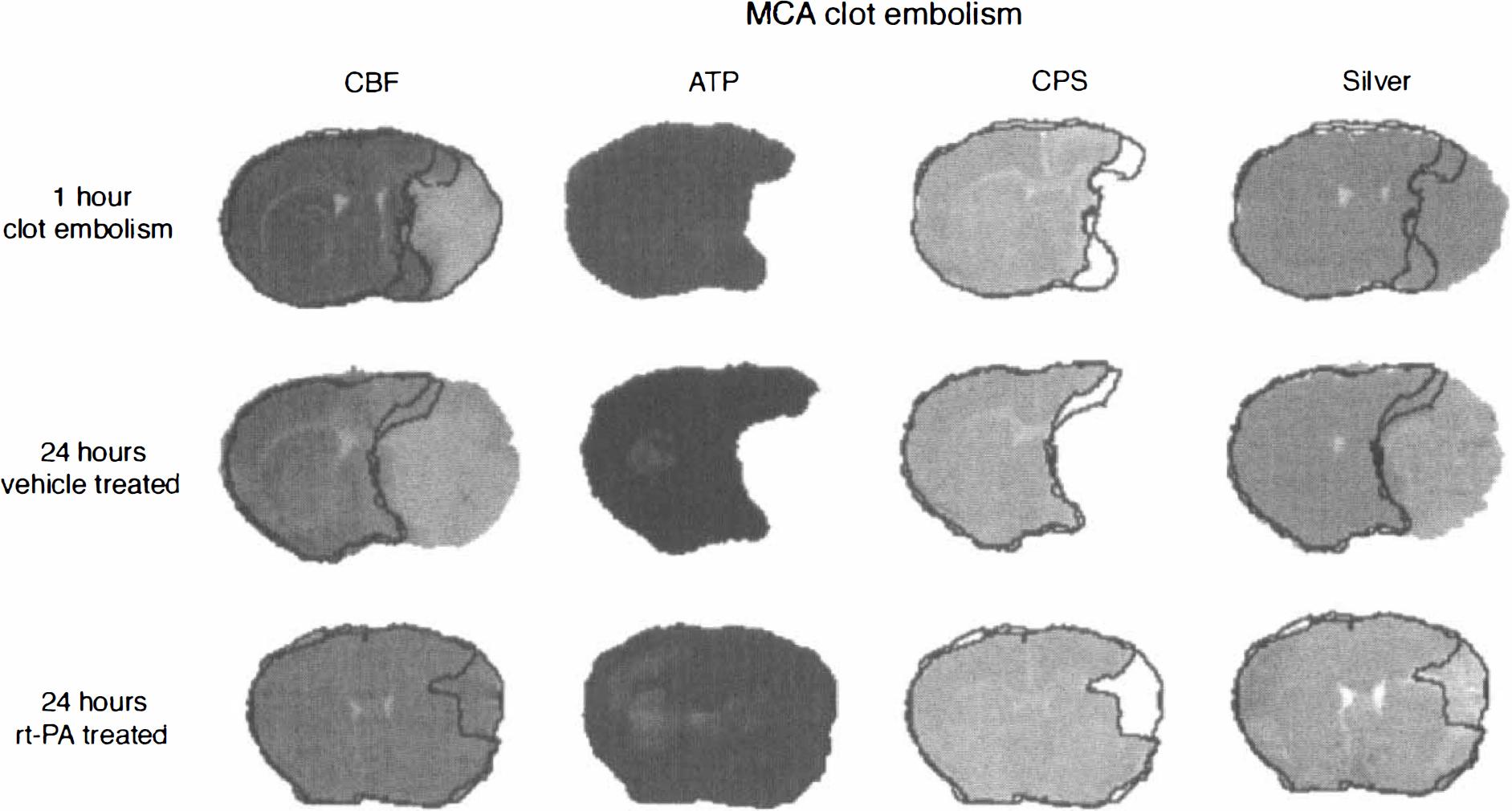
Imaging of cerebral blood flow (CBF), cerebral protein synthesis (CPS), ATP content, and morphologic changes (silver impregnation) at 1 hour after clot embolism and at 24 hours after either vehicle or rt-PA treatment. The outlines of ATP and CPS are superimposed to demarcate the metabolically impaired areas from the normal brain tissue. MCAO, middle cerebral artery occlusion.
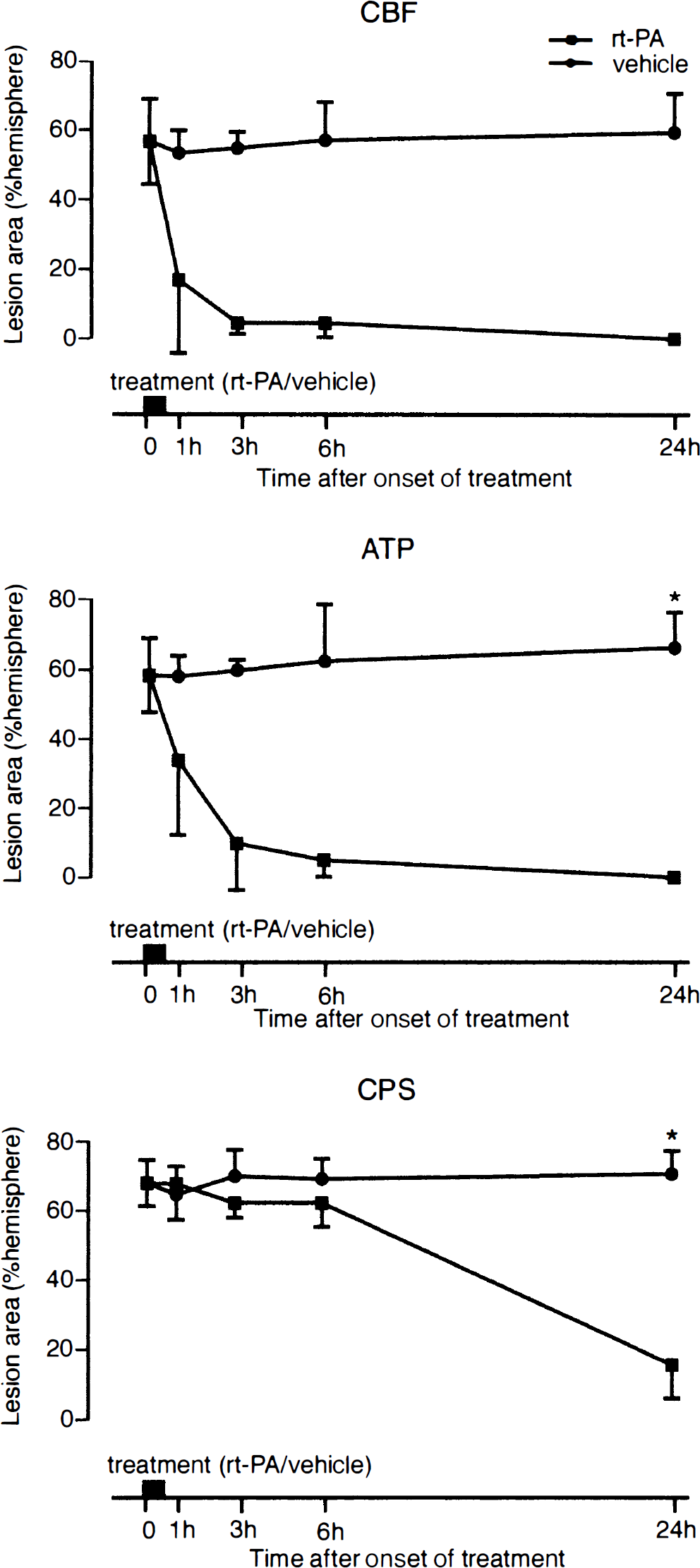
Effect of rt-PA-induced thrombolysis on the evolution of ischemic injury after clot embolism of middle cerebral artery. Hemispheric areas of reduced cerebral blood flow (CBF) and suppressed ATP and cerebral protein synthsis (CPS) were measured at the level of caudate-putamen (means ± SD). Thrombolysis induces gradual improvement of blood flow, followed first by recovery of ATP and, after longer delays, of CPS. * P < 0.05.
In the untreated group, 3 out of the 20 surviving animals exhibited spontaneous reperfusion with partial recovery of ATP. When these animals were rejected, the area of CBF and CPS suppression did not change, but the area of the ATP-depleted region gradually increased and between 6 and 24 hours merged with that of CPS suppression (66.1 ± 0.2% and 67.7 ± 8.9% for ATP and CPS suppression at 24 hours, respectively; Figs. 2 and 3).
In rt-PA treated animals, areas with critically reduced CBF disappeared within 24 hours, and in the center of the MCA territory mean flow rate increased to 87.4 ± 11.2% of the opposite hemisphere. The improvement of blood flow was associated with the gradual disappearance of the ATP and the CPS suppressed areas (0% and 15.5 ± 9.6% for ATP and CPS suppression at 24 hours, respectively; Figs. 2 and 3). Interestingly, ATP recovery clearly preceded the restoration of CPS, which remained severely suppressed for at least 6 hours after the initiation of treatment.
DNA fragmentations
Fragmentation of nuclear DNA was visualized by TUNEL. Labeling was confined to neurons and appeared much later than the changes in ATP and CPS. In untreated animals, TUNEL-positive neurons were detected between 6 and 24 hours after vehicle infusion. Twenty-four hours after the onset of treatment, the regional distribution of TUNEL corresponded clearly to that of ATP and CPS inhibition which, in turn, colocalized with the region of critically reduced CBF (Fig. 4).
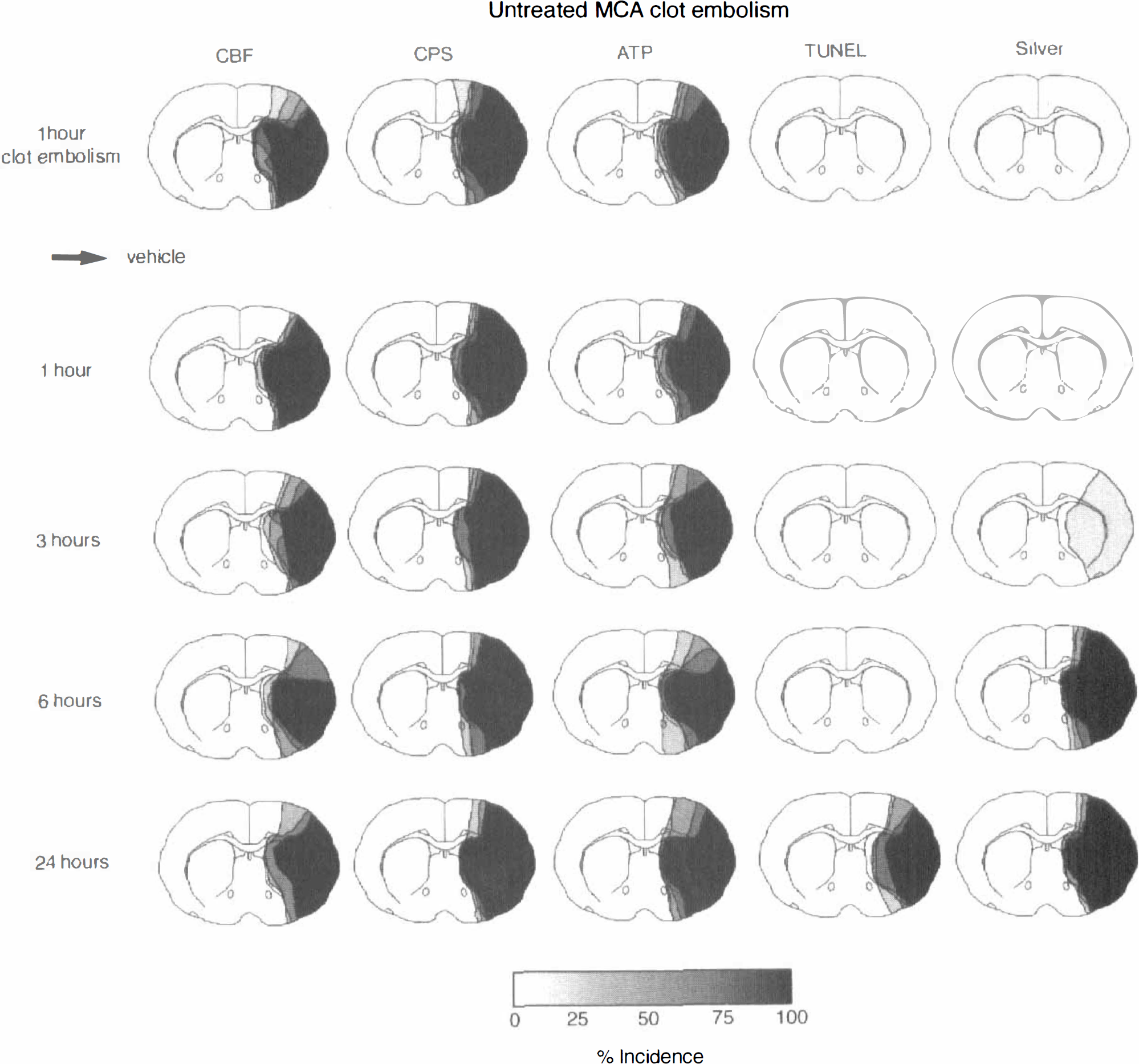
Evolution of ischemic injury after clot embolism without (vehicle) treatment. Incidence maps of reduced cerebral blood flow (CBF), suppressed cerebral protein synthesis (CPS), ATP depletion, double strand DNA fragmentations (TUNEL), and morphologic injury (loss of silver impregnation) at various times after middle cerebral artery (MCA) clot embolism. Note late manifestation of DNA fragmentation as compared with biochemical and morphologic alterations.
In rt-PA treated animals, the number and regional extension of TUNEL-positive neurons was distinctly reduced as compared with the untreated animals and corresponded approximately to that of persisting inhibition of CPS. Interestingly, TUNEL colocalized with the areas of persisting ATP depletion after 6 hours but there was a distinct dissociation between the recovery of ATP and the progression of TUNEL at 24 hours (Fig. 5).
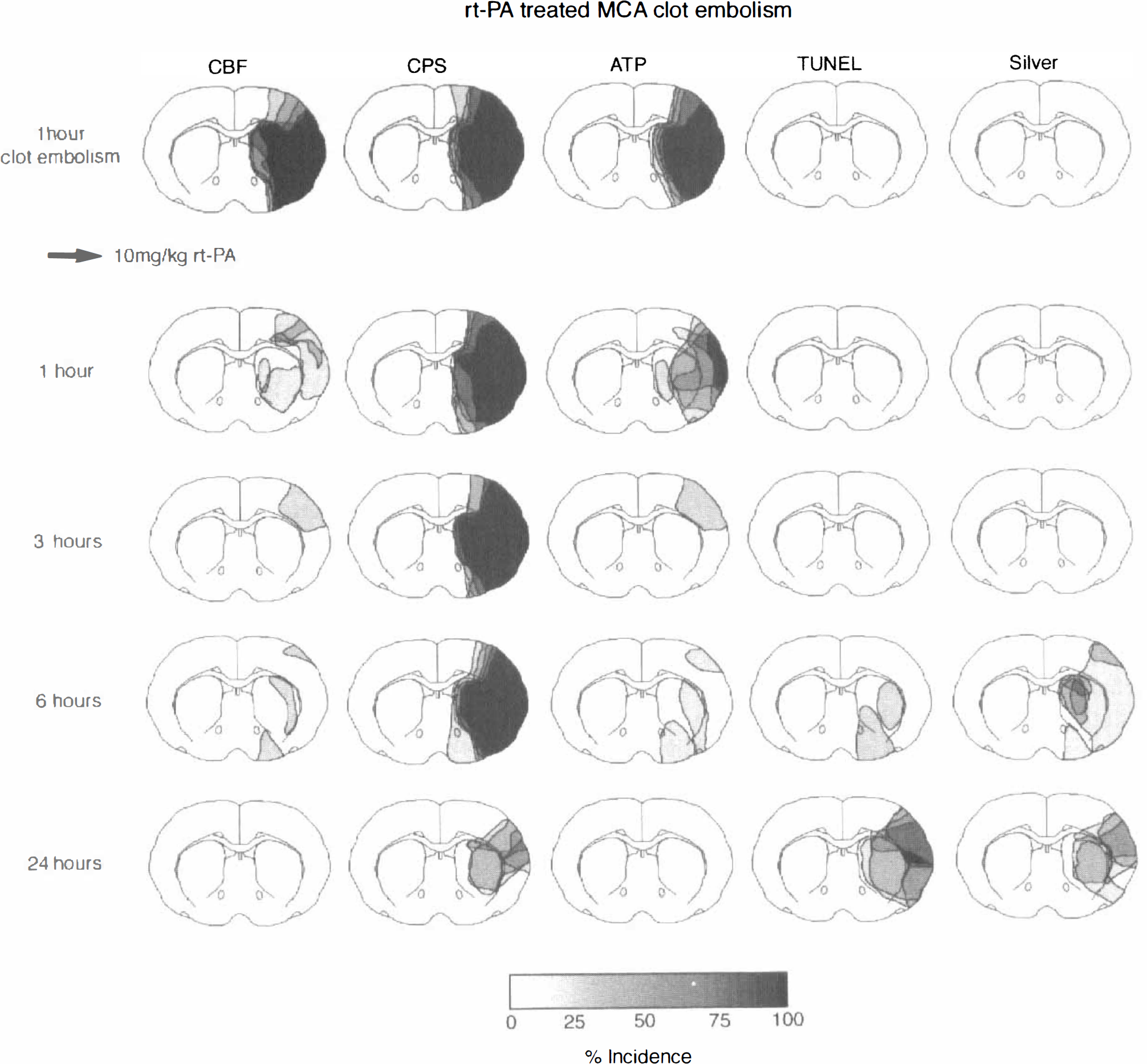
Recovery of ischemic injury after thrombolytic treatment of clot embolism. Incidence maps of reduced cerebral blood flow (CBF), suppressed cerebral protein synthesis (CPS), ATP depletion, double strand DNA fragmentations (TUNEL), and morphologic injury (loss of silver impregnation) before and at various times after rt-PA-induced thrombolysis. Note gradual return of blood flow and energy metabolism, followed by partial restoration of protein synthesis. Hemodynamic and metabolic recovery does not prevent DNA fragmentations and morphologic injury.
Morphologic changes
The evolution of morphologic injury was followed by silver impregnation and histologic staining with hematoxylin and cresyl violet. In the untreated animals, a sharply demarcated loss of staining intensity was observed from 6 hours on in the region of suppressed metabolic activity (Figs. 2 and 4).
In rt-PA treated animals, changes were much less pronounced, particularly in the hematoxylin and eosin and cresyl violet stained sections. The silver impregnation showed loss of staining intensity from 6 hours on in some, but not all animals, and again primarily in the region of persisting inhibition of protein synthesis (Figs. 2 and 5). However, the reproducibility of these changes and the sharpness of the morphologic demarcation was less consistent than in the untreated animals.
DISCUSSION
In untreated animals, MCA clot embolism produced permanent vascular occlusion in approximately 80% of experiments. In 20% of experiments, various degrees of spontaneous thrombolysis occurred that, however, could be readily recognized by LDF and iodoantipyrine autoradiography, allowing exclusion from further analysis. In the successfully embolized animals, large infarcts evolved, the size of which (68% of ipsilateral hemisphere at the level of caudate–putamen) was similar to that produced by the more commonly used intraluminal thread occlusion model (59% of hemisphere;Hata et al., 2000b). At 1 hour after clot embolism, the volume of ATP-depleted tissue was smaller than that in which CPS was suppressed, but between 6 and 24 hours the metabolically disturbed regions became congruent, which confirms the authors' previous observation that the suppression of CPS heralds the final size of the infarct (Hata et al., 2000b). Clot embolism also produced TUNEL-visible DNA fragmentations that, however, appeared late in the center of the energy-depleted tissue, but not in the penumbra, excluding classical apoptosis as a major factor of infarct expansion. Again this agrees with the observations made after permanent thread occlusion (Hata et al., 2000b).
Thrombolysis of clots initiated at 1 hour after embolism by intracarotid infusion of 10 mg/kg rt-PA partially reversed these changes. Twenty-four hours after the start of treatment, energy metabolism had recovered in all animals, and the volume of CPS suppressed tissue was distinctly smaller. Despite this reduction of ischemic injury, animals did not survive for more than a few days, indicating that some serious disturbances persisted that led to the fatal outcome.
One of these disturbances could be postischemic brain edema. Edema is a common complication of stroke, which in severe cases may provoke intracranial hypertension and transtentorial herniation, and which accounts for up to approximately 80% of stroke fatalities during the first week after ischemia under clinical conditions (Silver et al., 1984). After thrombolysis of clot embolism, the risk of edema is particularly high because microemboli originating from fragmented clots produce almost instantaneous breakdown of the blood–brain barrier (Schuier et al., 1978; Busch et al., 1997; Rosenberg, 1999). Thrombolysis is also associated with activation of neutral proteases such as matrix-degrading metalloproteinases that affect microvascular integrity and lead to blood–brain barrier dysfunction (Rosenberg et al., 1996; Pfefferkorn et al., 2000).
Another ischemia-associated pathologic alteration that could not be prevented by thrombolysis was the evolution of DNA fragmentations. In most treated animals neurons became TUNEL-positive in the MCA territory despite recovery of energy metabolism, which is basically different from permanent and transient MCA thread occlusion in which TUNEL colocalizes with areas of ATP depletion (Hata et al., 2000a, 2000b). Among the various factors contributing to DNA damage, a pharmacologic side effect of rt-PA has to be considered. Tsirka et al. (1995) reported that exitotoxin-induced neuronal degeneration was mediated by t-PA, and Flavin et al. (2000) observed induction of neuronal apoptosis by microglial t-PA. Wang et al. (1998) described reduction of infarct size in t-PA deficient mice and an increase of injury after rt-PA application in t-PA deficient or wild type animals, which would be in support of t-PA neurotoxicity. t-PA dependency of ischemic injury was also reported by Nagai et al. (1999) who reduced or enhanced t-PA activity by transgenic mutations. However, Meng et al. (1999) were not able to replicate these findings. In the authors' laboratory, application of a high dose of t-PA in a reversible thread occlusion model even led to a substantial reduction of infarct volume and of the associated brain edema, presumably as the result of a more efficient recirculation rate (Kilic et al., 1999).
Currently, it is not possible to decide whether molecular disturbances or the edema associated alterations are responsible for the low survival rate after rt-PA treatment, but it is obvious that additional interventions have to be performed to improve outcome (Steiner and Hacke, 1998). It is widely assumed that insights into the mechanisms of secondary ischemic injury obtained after reversible MCA thread occlusion may provide important hints for such a combination therapy. However, the comparison with the current thrombolysis model clearly demonstrates that such an analogy is not supported by the data. After 1 hour, reversible thread occlusion ATP recovers much faster than in the current study, but CPS inhibition is never reversed, and between 6 and 12 hours of recirculation, ATP begins again to decline to merge with the area of suppressed CPS shortly later (Hata et al., 2000a). This secondary energy depletion has been related to a pathobiochemical cascade of severe cellular stress that involves, among others, free radical production, peroxynitrate formation, endoplasmic reticulum calcium depletion, mitochondrial permeability transition, cytochrome c release, and the activation of apoptosis-promoting enzymatic reactions (Siesjöet al., 1999; Lipton, 1999; Paschen and Doutheil, 1999). In rt-PA-induced reperfusion of clot embolism, the pathobiochemical cascade is probably different because the inhibition of protein synthesis—a hallmark of ongoing cellular stress—is reversed, and secondary energy failure does not occur within the observation period of 24 hours. Fragmentation of DNA occurs in the presence, and not the absence, of energy and protein metabolism as after thread occlusion and, consequently, could have basically different biochemical consequences. Therefore, the factors responsible for the secondary complication must be studied in the thrombolysis and not in a reversible thread occlusion model to avoid misleading conclusions.
In summary, the current investigation into the dynamics of metabolic disturbances after rt-PA treated clot embolism of the MCA in mice reveals that energy metabolism and protein synthesis are remarkably reversed within 24 hours. Despite this improvement, TUNEL-positive cells appear from 6 hours on in the recirculated tissue and treated and untreated animals die between the first and the third day after clot embolism. The dynamics of metabolic recovery after thrombolysis differ sharply from that after reversible thread occlusion, suggesting a different pathophysiology of delayed ischemic injury. The elucidation of this difference will be crucial for the development of combination strategies intended to improve the therapeutic outcome of thrombolysis.
Footnotes
Acknowledgements
The authors acknowledge the technical assistance of Mrs. U. Beckmann, Mr. Janz, and Mr. Janus and the secretarial help of Mrs. D. Schewetzky. The authors also thank Mr. B. Huth for the preparation of the artwork.
