Abstract
Gene therapy is being investigated as a putative treatment option for cardiovascular diseases, including cerebral vasospasm. Because there is presently no information regarding gene transfer to human cerebral arteries, the principal objective of this study was to characterize adenovirus-mediated expression and function of recombinant endothelial nitric oxide synthase (eNOS) gene in human pial arteries. Pial arteries (outer diameter 500 to 1000 μm) were isolated from 30 patients undergoing temporal lobectomy for intractable seizures and were studied using histologic staining, histochemistry, electron microscopy, and isometric force recording. Gene transfer experiments were performed ex vivo using adenoviral vectors encoding genes for bovine eNOS (AdCMVeNOS) and Escherichia coli β-galactosidase (AdCMVLacZ). In transduced arteries, studied 24 hours after exposure to vectors, expression of recombinant β-galactosidase and eNOS was detected by histochemistry, localizing mainly to the adventitia (n = 4). Immunoelectron microscopy localized recombinant eNOS in adventitial fibroblasts. During contractions to U46619, bradykinin-induced relaxations were significantly augmented in AdCMVeNOS-transduced rings compared with control and AdCMVLacZ-transduced rings (P < 0.01; n = 6). The NOS inhibitor L-nitroarginine methylester (L-NAME) caused significantly greater contraction in AdCMVeNOS-transduced rings (P < 0.001; n = 4) and inhibited bradykinin-induced relaxations in control and transduced rings (P < 0.001; n = 6). The current findings suggest that in AdCMVeNOS-transduced human pial arteries, expression of recombinant eNOS occurs mainly in adventitial fibroblasts where it augments relaxations to NO-dependent agonists such as bradykinin. Findings from the current study might be beneficial in future clinical applications of gene therapy for the treatment or prevention of cerebral vasospasm.
Cerebral vasospasm is a prominent delayed pathologic response to the presence of blood in the subarachnoid space. It occurs in approximately one-third of all patients with aneurysmal subarachnoid hemorrhage in which it accounts for more than 20% of overall morbidity and mortality (Dorsch and King, 1994). Despite extensive investigation, the precise mechanism underlying cerebral vasospasm is unknown, although there is evidence implicating defective NO-mediated signaling in this process (Moncada et al., 1991; Cohen, 1995; Onoue et al., 1998). This seems plausible given the pivotal role played by NO in the circulation (Brian et al., 1996; Khurana and Besser, 1997; Hobbs et al., 1999). Recent advances in molecular biology have made it possible to increase NO production in the blood vessel wall through gene transfer, a technique that has considerable therapeutic potential. In agreement with the work of Heistad and colleagues (Ooboshi et al., 1995, 1997; Heistad and Faraci, 1996), the authors have demonstrated expression and function of recombinant endothelial nitric oxide synthase (eNOS) gene in canine cerebral arteries transduced with AdCMVeNOS (Chen et al., 1997, 1998; Tsutsui et al., 1998; Onoue et al., 1999). The authors' animal studies have shown that recombinant eNOS gene can be transferred into the adventitia of cerebral arteries where its expression augments relaxations to NO-dependent agonists. However, the authors have no information regarding gene transfer to human cerebral arteries.
Previously, successful in vitro adenovirus-mediated gene transfer to human vascular tissue has been performedin cultured endothelial and smooth muscle cells (Channon et al., 1996) and in relatively large vessels–including isolated saphenous veins; coronary, internal mammary, and extracranial carotid arteries (Rekhter et al., 1998); and radial artery grafts (Cable et al., 1998). More recently, in vivo cardiovascular gene transfer using adenoviral and plasmid vectors in humans has been reported in studies of patients with critical leg ischemia (Isner et al., 1998; Laitinen et al., 1998) and severe coronary artery disease (Isner and Losordo, 1999). There could be several reasons for the absence of information regarding gene transfer to human cerebral arteries. First, the only nonpostmortem human cerebrovascular model is the pial artery that must be rapidly acquired during appropriate neurosurgical resections to retain its natural advantage over postmortem tissue (Toda et al., 1985). Second, the use of living pial arteries from humans involves challenges generally not found in other studies in which more robust arteries from humans and animals are used. Such challenges include low tissue availability based on unpredictable operating schedules and the requirement for exceedingly delicate handling of pial arteries because of their small diameters (< 1 mm) and thin walls. Third, in the setting of gene transfer these difficulties are compounded by limited access to, and experience with, appropriate vectors and the need for simultaneous, parallel experiments involving stable, functionally matched arterial rings.
Given the lack of information regarding human cerebrovascular gene transfer, occurrence of species variations in vascular reactivity (Hardebo et al., 1983; Ryman et al., 1989) and known unique regulatory aspects of the cerebral circulation (Ryman et al., 1989; Edvinsson et al., 1993; Brian et al., 1996; Thorin et al., 1997; Bevan et al., 1998), the authors believed that a systematic gene transfer study needed to be performed using living, intact human cerebral arteries. In the current work, the authors first established a baseline for their gene transfer experiments by characterizing the morphology and vasomotor reactivity of freshly isolated human pial arteries (including their responses to NO-related compounds). They then used replication-deficient adenovirus encoding LacZ or eNOS cDNA to demonstrate effective expression and function of recombinant transgene in human pial arteries 24 hours posttransduction. Findings from the current study support the possibility of gene therapy as a treatment for cerebrovascular diseases.
MATERIALS AND METHODS
Human tissue
The procedures and handling of human tissue were reviewed and approved by the Institutional Review Board of the Mayo Foundation. Human pial arteries were isolated from temporal lobe specimens in patients undergoing surgery at the Mayo Clinic between June 1998, and February 2000, for intractable temporal lobe epilepsy. During this 20-month period, there was a total of 30 patients (18 male and 12 female) from whom viable tissue could be obtained (ages 8 to 73 years, mean age of 39 years). Arteries were harvested within 5 minutes of removal of the temporal lobe and immediately placed in chilled Krebs-Ringer bicarbonate solution (control solution: 118.3 mmol/L NaCl, 4.7 mmol/L KCl, 2.5 mmol/L CaCl2, 1.2 mmol/L MgSO4, 1.2 mmol/L KH2PO4, 25.0 mmol/L NaHCO3, 0.0026 mmol/L calcium EDTA, and 11.1 mmol/L glucose). On average, 1 to 2 pial arteries (outer diameter 500 to 1000 μm; 1.5 to 2.0 cm in length) were obtained from each specimen. Visualized using an operating microscope, periadventitial pia-arachnoid tissue and intraluminal blood were carefully removed and arteries were sectioned into rings 2 to 3 mm in length. Pial arteries from the first 16 of the 30 patients were used for characterization of morphology and vasomotor reactivity before gene transfer, whereas arteries from the remaining 14 patients were used in gene transfer experiments.
Adenoviral vectors
Recombinant adenovirus containing eNOS cDNA was generated as described in detail elsewhere (Spector and Samaniego, 1995; Chen et al., 1997). Briefly, a plasmid containing cDNA for bovine aortic endothelial cell eNOS (generously provided by Dr. David Harrison, Emory University, Atlanta, GA, U.S.A.) was inserted into the shuttle vector pACCMVpLpA (a gift from Dr. Robert Gerard, University of Texas Southwestern Medical Center, Dallas, TX, U.S.A.) containing cDNA for cytomegalovirus (CMV) promoter. The resulting plasmid was cotransfected with the dl309 variant of adenovirus serotype 5 into human embryonic renal carcinoma (293) cells. Recombinant adenoviral vector (AdCMVeNOS) was generated by homologous recombination. Stocks were prepared from positive viral plaques, and these were used to generate high-titer (1011-1012 plaque forming units (PFU)/mL) preparations as determined by plaque assay (Gerard and Meidell, 1995). Adenovirus containing cDNA for Escherichia coli β-galactosidase (AdCMVLacZ) was used in all gene transfer experiments as a control; it was propagated, isolated, and quantified as described elsewhere (Chen et al., 1997).
Gene transfer
Procedures and handling of tissue exposed to recombinant adenoviral vectors were reviewed and approved by the Institutional Biosafety Committee of the Mayo Foundation. In these experiments, control rings refer to those that were not transduced with adenoviral vectors. Nontransduced arterial rings were incubated in minimum essential medium (MEM) for 40 minutes at 37°C in parallel with, but separate from, rings transduced with AdCMVeNOS or AdCMVLacZ vector (1010 PFU/mL titer). Afterwards, rings were transferred to fresh MEM and incubated for a further 24 hours at 37°C in a CO2 incubator (Forma Scientific, Marjetta, OH, U.S.A.). For gene transfer experiments, the adenoviral titer of 1010 PFU/mL and incubation time of 24 hours were chosen in the context of the authors' previous work using the same vectors in canine cerebral arteries (Chen et al., 1997; Onoue et al., 1999).
Histology and histochemistry
After fixation in 10% buffered formalin, certain arterial segments were embedded in paraffin and cut into serial 5-μm cross-sections. Cut sections were stained with hematoxylin and eosin, Movat pentachrome, or periodic acid Schiff, or processed immunohistochemically using monoclonal mouse antihuman smooth muscle actin antibody (1:150 of stock; Dako, Carpinteria, CA, U.S.A.) and immunoperoxidase methods. For histochemical staining of recombinant β-galactosidase, other arterial segments were fixed for 30 minutes in 2% paraformaldehyde/0.2% glutaraldehyde in phosphate-buffered saline (PBS). They were then rinsed with PBS and placed in X-Gal reagent (Gibco BRL) for 4 hours. Stained vessels were embedded in OCT compound (Sakura Torrance, CA, U.S.A.), and cut into 5-μm cross-sections. The sections were then lightly counterstained with nuclear fast red and dehydrated through graded alcohol washes. For immunohistochemical staining of recombinant eNOS, arterial rings were frozen in OCT and cut into 5-μm cross-sections. These were fixed in acetone (4°C) and then, at room temperature, sequentially incubated for 10 minutes each in 50% methanol/1.5% hydrogen peroxide, 0.25% sodium dodecyl sulfate (SDS; Gibco BRL), and 0.1% sodium azide/0.3% hydrogen peroxide, followed by incubation with 5% normal goat serum/PBS-Tween 20 to block nonspecific binding sites. Mouse monoclonal IgG1 antibody with crossreactivity against bovine eNOS (1:500 of stock; Transduction Laboratories, San Diego, CA, U.S.A.) was applied for 60 minutes at room temperature, followed by incubations with biotinylated rabbit anti-mouse F(ab′)2 (1:200, 20 minutes) secondary antibody and peroxidase-conjugated streptavidin (1:300, 20 minutes; Vector Laboratories, Burlingame, CA, U.S.A.). After a 30 second immersion in 0.1 mol/L sodium acetate buffer (pH 5.2), eNOS immunoreactivity was visualized with 3-amino-9-ethylcarbazole and hematoxylin counterstaining. For control studies, specificity of eNOS immunolabeling was examined by the following three methods: (1) omission of the primary anti-eNOS antibody in the incubation medium, (2) eNOS immunostaining of AdCMVLacZ-transduced vessels, and (3) immunostaining of AdCMVeNOS-transduced vessels with an isotype-matched nonanti-eNOS primary antibody, namely, a mouse IgG1 monoclonal antihuman CD4 antibody (OPD-4, 1:500 dilution; Dako).
Electron microscopy
For electron microscopy, arterial segments were fixed in 4% paraformaldehyde/0.2% glutaraldehyde and embedded in LR White resin. Thin cross-sections of vessels were mounted on 300-mesh nickel grids and stained with uranyl acetate and lead citrate. Examination and photography of stained vascular cells were performed using a CM-10 transmission electron microscope. For localization of recombinant eNOS protein, certain sections were prepared using the immunogold indirect labeling technique (Bendayan, 1989). After mounting as described above, cross-sections were preincubated for 1 hour in PBS-Tween 20 with 2% normal goat serum and 0.15 mol/L glycine and incubated for 2 hours in mouse monoclonal anti-eNOS primary antibody (see above) diluted 1:250 in PBS-Tween 20. They were then rinsed thoroughly in PBS-Tween 20, incubated for 60 minutes in goat anti-mouse IgG-serum, conjugated to 15 nm colloidal gold, and rinsed again in PBS-Tween 20. After drying, these immunogold-labeled grids were stained and photographed as described above. Control specimens were exposed to the same procedure after omission of primary antibody.
Organ chambers
Each ring was connected to an isometric force transducer (Grass FT03; Grass Instruments, West Warwick, RI, U.S.A.) and suspended in an organ chamber filled with 25 mL of control solution (37°C, pH 7.4) aerated with 94% O2:6% CO2. Isometric force was recorded continuously. Suspended arteries were allowed to stabilize in organ chambers for 75 minutes. Each ring was then gradually stretched in increments of 0.2 g to the optimal point of its length-tension curve (approximately 0.4 to 0.6 g) as determined by contractions to thromboxane A2 analog U46619 (10−8 mol/L). To evaluate relaxation responses, rings were contracted with U46619 (10−8 mol/L or 2 × 10−8 mol/L) before addition of relaxing compounds. At the conclusion of each recording and following washout of test compound, sustained functional integrity of arterial rings was determined by contraction to U46619 (10−6 mol/L), or relaxation to bradykinin (10−6 mol/L), or both.
Analysis of vascular reactivity
Concentration-response curves were obtained for all compounds by cumulative addition of the contracting or relaxing substance to the organ chamber solution. The concentration range for each substance tested was determined from experiments performed in animal models. Arterial rings from a minimum of six patients were used to analyze the effect of each substance on vasomotor reactivity. To facilitate accurate comparability, several rings cut from the same artery were studied in parallel, and only rings with stable, nonoscillatory (Gokina et al., 1996) tone matched for contractions to U46619 (10−8 mol/L or 2 × 10−8 mol/L) were used for data analyses. Each ring was exposed to a maximum of two pharmacologic compounds with an intentionally varied order of exposure. For contracting compounds, data are expressed as percentage of maximal contraction induced by the most efficacious vasoconstrictor, endothelin-1 (ET-1, 3 × 10−8 mol/L, 100%). For relaxing compounds, data were expressed as percentage of maximal relaxation induced by papaverine (PPV, 3 × 10−4 mol/L, 100%). In all experiments, the median effective concentration (EC50) was calculated for each compound by linear interpolation between the two concentrations evoking responses just above and below 50% of the maximum in each ring. Relative potencies of compounds were determined by comparison of their respective EC50 values.
Drugs
The following pharmacologic compounds were used: A23187, bradykinin, L-nitroarginine methylester (L-NAME), nimodipine, PPV hydrochloride, prostaglandins F2α and I2, substance P, and UTP (Sigma Chemical, St. Louis, MO, U.S.A.); diethylamine NONOate (DEANONOate) and U46619 (Cayman Chemical, Ann Arbor, MI, U.S.A.); and ET-1 (Phoenix Pharmaceutical, Mountain View, CA, U.S.A.). With the exception of A23187, stock solutions of all drugs were freshly prepared daily using distilled water; A23187 stock solution was prepared using 100% dimethyl sulfoxide (DMSO; Sigma). Dilutions of all drugs were made in distilled water such that volumes of < 0.15 mL were added to the organ chambers. In experiments in which it was used, L-NAME was added 15 minutes before exposure to bradykinin. All drug concentrations are expressed as final molar concentrations in solution.
Data analysis
Results are expressed as mean ± SD. In all experiments, n indicates the number of patients from whom arteries were harvested. Statistical analysis of dose-response curves involved a two-way repeated measures analysis of variance and Bonferroni's post hoc test. Where indicated, differences between EC50 values were calculated through Student's paired t-test. Probability values of P < 0.05 were considered statistically significant.
RESULTS
Baseline morphology and vasomotor reactivity of human pial arteries
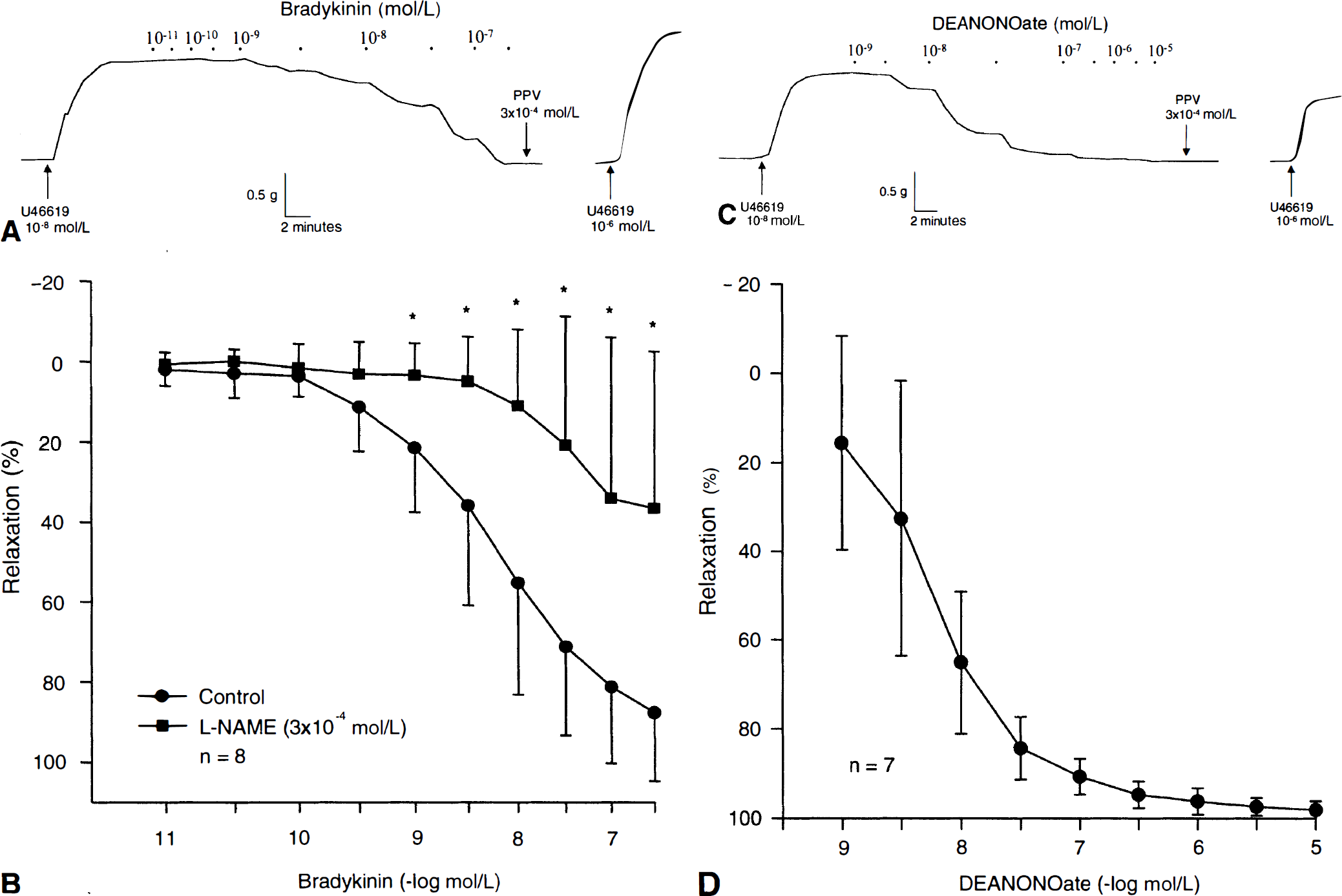
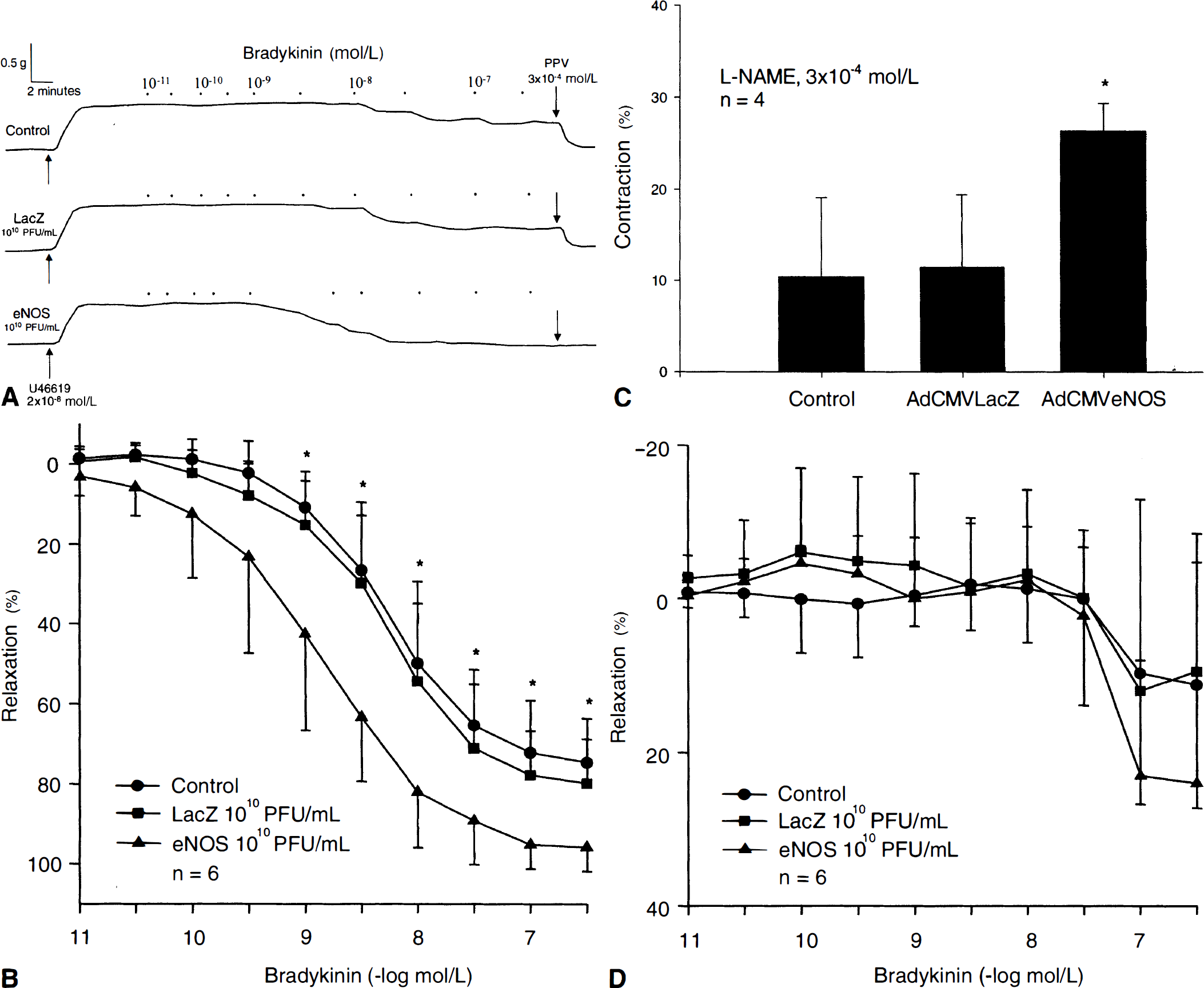
Pial arteries derived from temporal lobectomy patients were found to be morphologically normal after histologic and electron microscopic examination (n = 6;Fig. 1; normal morphology through hematoxylin and eosin and periodic acid Schiff staining not shown). Vasomotor responses to a wide variety of contracting and relaxing compounds could be elicited and readily reproduced in human pial artery rings (Table 1). Endothelin-1 was found to be the most efficacious and potent vasoconstrictor, whereas PPV was found to be the most efficacious vasodilator; nimodipine was the most potent of the non-NO-mediated vasodilators studied (Table 1). The endothelium-dependent peptides bradykinin and substance P and calcium ionophore A23187 caused concentration-dependent relaxations (Table 1). Relaxations to bradykinin (10−11-3 × 10−7 mol/L, −log EC50 8.2 ± 0.5, max relaxation 88 ± 17%, n = 8) were significantly reduced, but not abolished, in the presence of L-NAME (3 × 10−4 mol/L, max relaxation 36 ± 39%;P < 0.05; n = 8;Figs. 2A and 2B). The NO donor DEANONOate also producedconcentration-dependent relaxations (Figs. 2C and 2D). During contractions to U46619 (10−8 mol/L or 2 × 10−8 mol/L), relaxation responses to bradykinin (10−11-3 × 10−7 mol/L) were not significantly different in rings incubated for 24 hours in MEM before isometric force recording compared with nonincubated rings, in which isometric force recording was immediately performed, (P > 0.05; n = 6 and n = 8, respectively;Table 2). Similarly, contractions to ET-1 (10−11-3 × 10−8 mol/L) were also not significantly different in MEM-incubated rings compared with nonincubated rings (P > 0.05; n = 6 and n = 10, respectively;Table 2).
Vasomotor responses to contracting and relaxing compounds in human pial artery
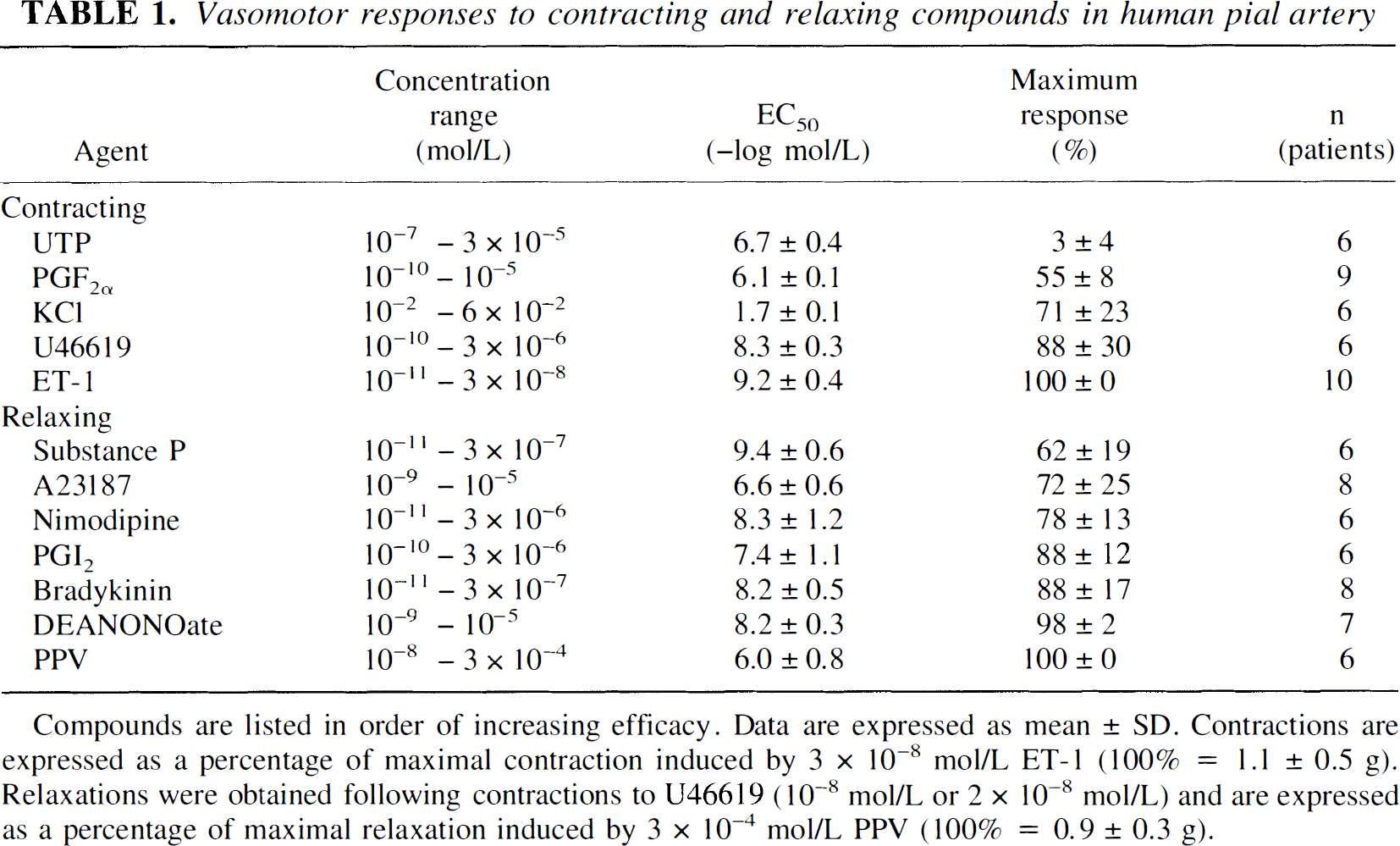
Compounds are listed in order of increasing efficacy. Data are expressed as mean ± SD. Contractions are expressed as a percentage of maximal contraction induced by 3 × 10−8 mol/L ET-1 (100% = 1.1 ± 0.5 g). Relaxations were obtained following contractions to U46619 (10−8 mol/L or 2 × 10−8 mol/L) and are expressed as a percentage of maximal relaxation induced by 3 × 10−4 mol/L PPV (100% = 0.9 ± 0.3 g).
Comparison of vasomotor responses to bradykinin and ET-1 in nonincubated versus 24-hour incubated human pial arteries
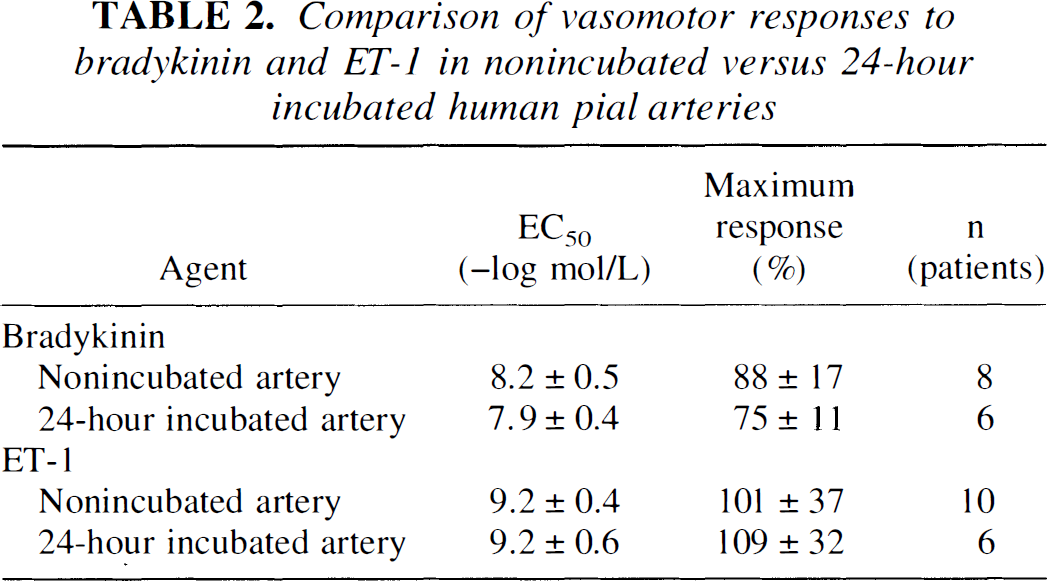
No significant difference (P > 0.05) between arteries in responses to bradykinin (10−11 −3 × 10−7mol/L) or ET-1 (10−11−3 × 10−8mol/L). Data are expressed as mean ± SD. Relaxations were obtained after contractions to U46619 (10−8mol/L or 2 × 10−8mol/L) and are expressed as a percentage of maximal relaxation induced by 3 × 10−4mol/L PPV (100% = 0.9 ± 0.3 g and 0.8 ± 0.3 g for nonincubated and 24-hour incubated arteries, respectively). Contractions are expressed as a percentage of maximal contraction induced by 10−6mol/L U46619 (100% = 1.1 ± 0.5 g and 1.0 ± 0.5 g for nonincubated and 24-hour incubated arteries, respectively).
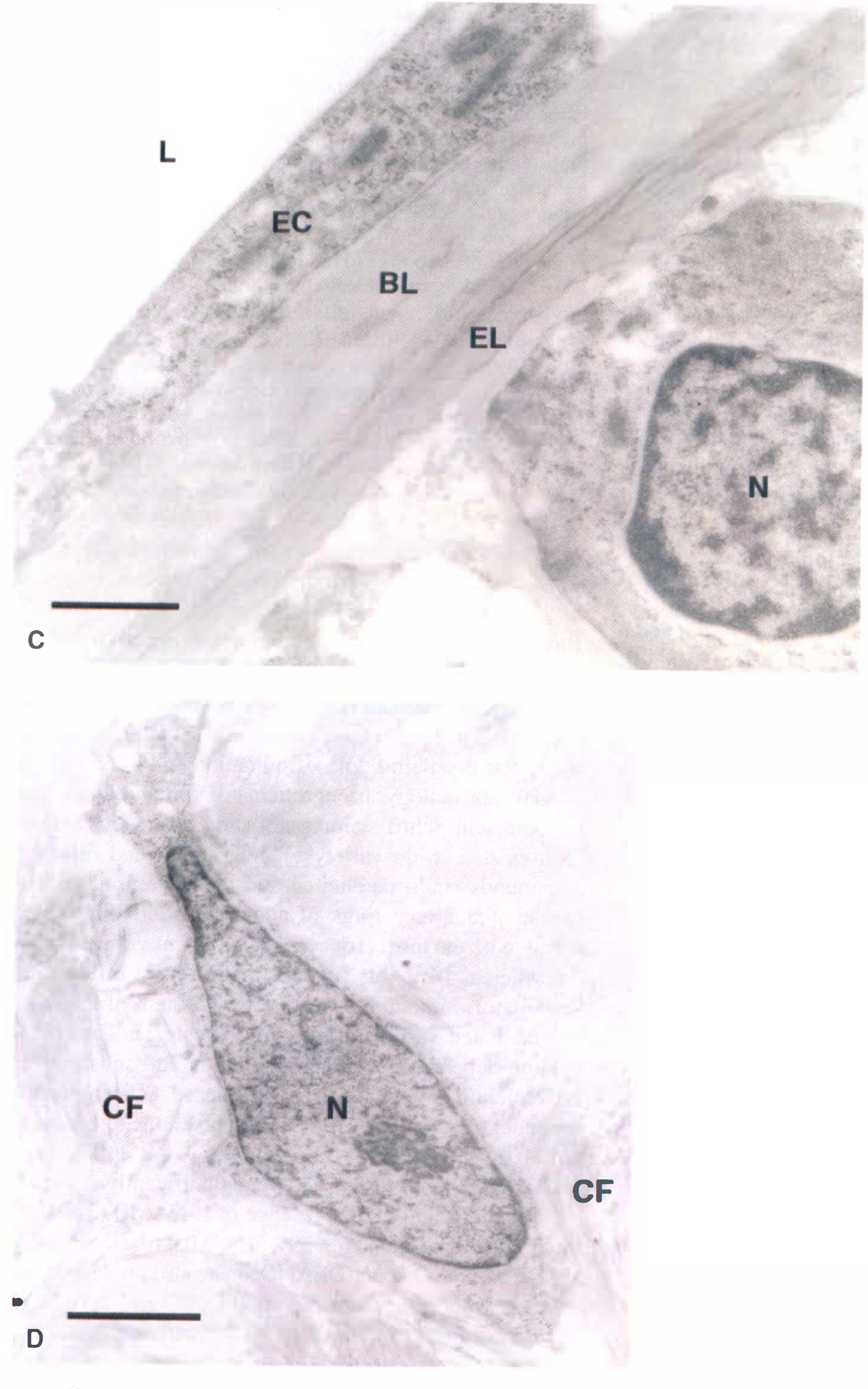
Morphologic studies in human pial artery before gene transfer.
Functional studies in human pial artery before gene transfer.
Recombinant eNOS gene transfer to human pial arteries
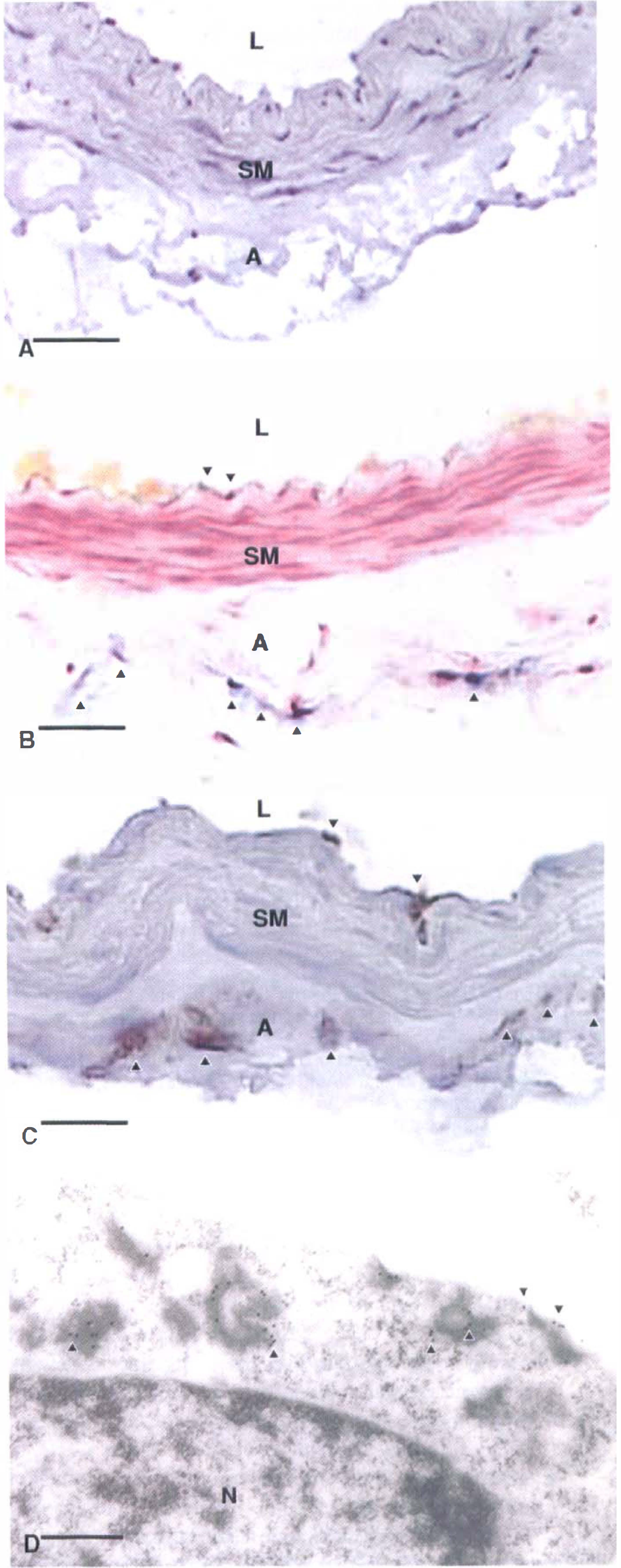
Morphologic studies in human pial artery after gene transfer.
Functional studies in human pial artery after gene transfer.
DISCUSSION
The current study, whose principal objective was to characterize the expression and function of recombinant eNOS transgene in human pial arteries, was performed for several reasons. First and foremost, there is growing interest in the role of recombinant eNOS gene transfer as a potential therapeutic option in vascular diseases including postsubarachnoid hemorrhage cerebral vasospasm. Second, to the authors' knowledge, there is no information regarding gene transfer to human cerebral vessels. Third, undertaking the current study represented a logical continuation of the authors' previous work using adenoviral vectors in animal cerebral arteries. Lastly, the authors believed that investigation of human cerebral arteries was necessary given the known occurrence of species-and vascular territory-related variations in vasomotor reactivity. The principal findings of this study are that effective adenovirus-mediated ex vivo transfer and expression of recombinant genes can be performed in human pial arteries, the main cellular target of adenoviral transduction and synthesis of recombinant protein appears to be adventitial fibroblasts, and expression of recombinant eNOS in pial arteries is associated with augmented relaxations to the NO-dependent agonist bradykinin.
Baseline morphology and vasomotor reactivity of human pial arteries
To establish a structural and functional baseline for their cerebrovascular gene transfer experiments, the authorsfirst studied nontransduced human pial arteries through histology, electron microscopy, and isometric force recording. The current morphologic data, consistent with previous studies in human cerebral arteries (Marinkovic et al., 1979; Dahl, 1986; Kallioinen et al., 1987; Edvinsson et al., 1993; Lee, 1995), suggest that pial arteries derived from temporal lobectomy specimens have normal histologic and cytoarchitectural features, demonstrating no evidence for atherosclerosis, inflammation, traumatic injury, or any other pathology. Furthermore, from a functional perspective, the vasomotor effects of compounds tested in pial arteries before gene transfer were found to be similar to those described in the authors' canine studies (Katusic et al., 1988; Chen et al., 1997; Onoue et al., 1999), with the exceptions of KCl, PGF2α, substance P, and UTP (all of which had poorer efficacy in human pial arteries). Interestingly, UTP was more effective as a vasodilator than a vasoconstrictor in human pial arteries, a finding that was in strong contrast to canine cerebral arteries, but was supported by the human pial artery study of Hardebo et al. (1987) and also consistent with the known occurrence of species variability (Hardebo et al., 1983). With regard to vasomotor responses to NO-related compounds, confirming previous studies in animal and human cerebral arteries (Wahl et al., 1983; Katusic et al., 1988; Petersson et al., 1995), the authors observed concentration-dependent relaxations to the endothelium-dependent peptides bradykinin and substance P, calcium ionophore A23187, and the NO donor DEANONOate. These findings, combined with the observed significant reduction of bradykinin-induced relaxations in the presence of L-NAME, indicate the presence of endogenous NO-mediated signaling in these arteries. This is of obvious importance to the applicability of recombinant eNOS gene transfer experiments in these vessels. However, it should be noted that other mechanisms of relaxation, for example, those mediated by endothelium-derived hyperpolarizing factor (EDHF;Vanhoutte, 1999) might also be present in these arteries, as suggested by the authors' observation of L-NAME-insensitive relaxations to bradykinin. Finally, as a prerequisite for gene transfer studies using adenovirus-transduced human pial arteries (involving 24-hour incubation of rings in MEM), the authors compared the vasomotor reactivity of nonincubated versus 24-hour incubated (nontransduced) human pial artery rings. The authors observed similar vascular reactivity between nonincubated versus incubated arteries. This was indicated by the similarity of the EC50 values and maximum responses to bradykinin and ET-1 in both of these groups. These findings suggest that vasomotor data acquired from 24-hour incubated human pial arteries (which were exclusively used in the authors' gene transfer experiments;Chen et al., 1997) are qualitatively and quantitatively similar to data acquired from nonincubated arteries—that is, there is no significant change in vascular reactivity attributable to incubation.
Recombinant eNOS gene transfer to human pial arteries
As demonstrated by positive histochemical and immunohistochemical staining, respectively, transduction of human pial arteries with adenoviral vectors resulted in expression of β-galactosidase (reporter) and eNOS transgenes in the arterial wall. Expression of recombinant proteins appeared to predominate in the vascular adventitia, a finding consistent with other studies using adenoviral vectors in canine cerebral (Chen et al., 1997; Tsutsui et al., 2000) and human radial (Cable et al., 1998) arteries and attributable to ex vivo transduction. In the current study, the authors used immunogold electron microscopy to identify the cellular target of adenoviral transduction and subcellular localization of recombinant protein. Recombinant eNOS-bound gold particles were detected in adventitial fibroblasts, both in the region of the plasma membrane and in the cytoplasm, but not in the nucleus. This confirmed the current findings in canine basilar arteries transduced ex vivo with AdCMVeNOS (Tsutsui et al., 1998).
In agreement with other vascular gene transfer studies using NOS inhibitors (Ooboshi et al., 1997; Tsutsui et al., 1998), the authors found that L-NAME significantly reduced bradykinin-induced relaxations in control and transduced arteries. Interestingly, the authors observed a significantly greater amount of L-NAME-induced contraction in AdCMVeNOS-transduced arteries compared with control and AdCMVLacZ-transduced pial arteries maintained at resting tension before exposure to L-NAME. One possible explanation for this observation could be that L-NAME inhibits a greater amount of basally active NOS in arteries expressing recombinant eNOS. Finally, reduced contractile responses to UTP, ET-1, KCl, and PGF2α have been reported after transduction with AdCMVeNOS (Chen et al., 1997; Cable et al., 1998; Onoue et al., 1999), findings which provide additional and encouraging implications for the benefit of recombinant eNOS gene transfer in the setting of vasospasm. In freshly isolated, nontransduced human pial arteries, the authors found UTP, KCl, and PGF2α to have poorer contractile efficacy compared with their effects in canine cerebral arteries, and accordingly elected to test contractile responses to ET-1 in control and transduced pial arteries. A significant reduction in ET-1-induced contraction has been reported by Onoue et al. (1999) in canine basilar artery transduced with AdCMVeNOS. In their study, it was proposed that activation of NOS-coupled ETA receptors in genetically modified adventitia overexpressing eNOS led to reversal of the normal vasoconstrictor effect of ET-1. Unlike Onoue et al. (1999), the authors could not demonstrate any significant difference between vasomotor responses of control and transduced human pial arteries exposed to ET-1. This finding could be explained by the absence of ETA receptor expression in human pial artery adventitial fibroblasts.
In summary, the current study demonstrates that recombinant eNOS gene can be functionally expressed in human cerebral arteries after ex vivo adenovirus-mediated transduction. Transgene expression principally occurs in adventitial fibroblasts, resulting in augmented relaxations to the NO-dependent agonist bradykinin. The authors believe that eNOS gene transfer, by restoring the biosynthesis of NO in diseased blood vessels, may be beneficial in the setting of vascular disorders such as cerebral vasospasm in which defective NO production has been implicated.
Footnotes
Acknowledgements
The authors thank Dr. Vijay Shah for his invaluable suggestions regarding the content of this manuscript, Janet Beckman for assistance with its preparation, and Ann Brumm, Belinda Hoebing, and Denise Morgan for their help in coordinating this study.
