Abstract
The authors transplanted adult bone marrow nonhematopoietic cells into the striatum after embolic middle cerebral artery occlusion (MCAO). Mice (n = 23; C57BL/6J) were divided into four groups: (1) mice (n = 5) were subjected to MCAO and transplanted with bone marrow nonhematopoietic cells (prelabeled by bromodeoxyuridine, BrdU) into the ischemic striatum, (2) MCAO alone (n = 8), (3) MCAO with injection of phosphate buffered saline (n = 5), and (4) bone marrow nonhematopoietic cells injected into the normal striatum (n = 5). Mice were killed at 28 days after stroke. BrdU reactive cells survived and migrated a distance of approximately 2.2 mm from the grafting areas toward the ischemic areas. BrdU reactive cells expressed the neuronal specific protein NeuN in 1% of BrdU stained cells and the astrocytic specific protein glial fibrillary acidic protein (GFAP) in 8% of the BrdU stained cells. Functional recovery from a rotarod test (P < 0.05) and modified neurologic severity score tests (including motor, sensory, and reflex;P < 0.05) were significantly improved in the mice receiving bone marrow nonhematopoietic cells compared with MCAO alone. The current findings suggest that the intrastriatal transplanted bone marrow nonhematopoietic cells survived in the ischemic brain and improved functional recovery of adult mice even though infarct volumes did not change significantly. Bone marrow nonhematopoietic cells may provide a new avenue to promote recovery of injured brain.
Fetal neural stem cells can form new neurons (Gage, 1998; Snyder et. al., 1997) and reduce behavioral deficits in damaged and compromised animal and human brain (Bjorklund and Lindvall, 1999; Gage, 2000). A series of patients with Parkinson's disease have been treated with mesencephalic cells from human fetuses obtained from 6-to 9-week-old abortuses (Spencer et al., 1992; Freed et al., 1995; Kordower et al., 1995; Defer et al., 1996; Lopez-Lozano et al., 1997). Some patients exhibited significant improvement of clinical symptoms and synthesis of dopamine as assayed by fluorodopa uptake by positron emission tomography (Spencer et al., 1992; Freed et al., 1995; Kordower et al., 1995; Defer et al., 1996), even 10 years after transplantation (Bjorklund and Lindvall, 1999). Fetal stem cells implanted into the striatum have also been used to reconstruct damaged basal ganglia circuits and to ameliorate behavioral deficits in ischemic rats (Onizuka et al., 1996; Goto et al., 1997). However, there are critical ethical and logistical problems surrounding the use of embryonic neural stem cells for the treatment of central nervous system diseases. For these reasons, attempts have been made to develop alternative cells for neurotransplantation, such as encapsulated cells, genetically engineered cells, and a variety of transformed cell lines (Kang et al., 1993; Bankiewicz et al., 1993; Gage, 1998; Borlongan et al., 1998). However, in all of these cases immunorejection is inevitable. Bone marrow is a source of stem cells and may be a better choice for transplantation because issues of rejection have been overcome (Azizi et al., 1998).
Hematopoietic stem and progenitor cells (HSCs) resident in bone marrow give rise to blood elements and have been genetically altered ex vivo and reimplanted in patients (Karlsson, 1991; Walsh et al., 1994). In addition to HSCs, bone marrow also contains a subset of heterogeneous nonhematopoietic cells, isolated by their adherence to plastic, which include marrow stromal cells (MSCs, also referred to as mesenchymal stem and progenitor cells) (Prockop, 1997; Kopen et al., 1999, Colter et al., 2000). The nonhematopoietic cells have attracted interest because of their ability to function as a feeder layer for the growth of hematopoietic stem cells, their multipotentiality for differentiation, and their possible use for cell and gene therapy (Pereira et al., 1995, 1998; Mason et al., 1998; Riew et al., 1998; Prockop, 1998; Bruder et al., 1998; Horwitz et al., 1999; Colter et al., 2000). Culture-expanded MSCs have also been infused into adult cancer patients, opening the door to MSC therapy for a number of diseases (Lazarus et al., 1995). Studies indicate that cells derived from bone marrow survive, proliferate, migrate, and differentiate into glial and neuronal phenotypes (Azizi et al., 1998; Eglitis et al., 1997, 1999; Prockop, 1997, 1998). These data indicate that MSCs are: (1) easy to isolate from the small aspirates of bone marrow that can be obtained under local anesthesia, (2) capable of rapid proliferation in culture, (3) amenable to survival and integration in the host brain, and (4) immunologically inert. Bone marrow nonhematopoietic cells contain stem cells that may serve to replenish lost cells; moreover, they contain a variety of cells that secrete substances that may lead to recovery. Plastic-adherent cells from bone marrow normally support hematopoietic progenitors and secrete a number of hematopoietic cytokines and trophic factors—for example, interleukins, stem cell factor, and colony-stimulating factor-1 (Eaves et al., 1991; Majumdar et al., 1998; Colter et al., 2000).
Experimental occlusion of the middle cerebral artery (MCA) in animals produces neurobehavioral deficits parallel to those seen in ischemic stroke in human patients (Borlongan et al., 1997). In the current study, the authors tested the hypothesis that intracranial transplantation of adult bone marrow nonhematopoietic cells into the adult mouse after stroke survive, migrate, and differentiate to parenchymal phenotypes. Behavioral tests for functional restoration of ischemic damaged tissue were also scored after MCAO and transplantation, using a rotarod test and modified neurologic severity score (NSS).
MATERIALS AND METHODS
Preparation of donor bone marrow nonhematopoietic cells
Primary cultured bone marrow cells were obtained from adult mice (C57BL/6J, Jackson Laboratories, Bar Harbor, ME, U.S.A.) receiving 5-fluorouracil (5-FU, 150 mg/kg) intraperitoneally 2 days before harvesting (Randall and Weissman, 1997). Using a 21-gauge needle connected to a 1 mL syringe with phosphate-buffered saline (PBS, 0.5 mL), fresh complete bone marrow was harvested aseptically from tibias and femurs. Bone marrow was then mechanically dissociated until a milky homogenous single-cell suspension was achieved. Red blood cells were removed from bone marrow using 0.84% NH4Cl. Numbers of nucleated marrow cells were measured by a cytometer, and 2 × 106 nucleated cells were seeded into each tissue culture flask in Iscove's Modified Dulbecco's medium supplemented with fetal bovine serum (10%). After 3 days of incubation, cells tightly adhered to plastic and were resuspended to fresh Iscove's Modified Dulbecco's medium in new flasks and were grown for three passages. To identify cells derived from bone marrow, bromodeoxyuridine (BrdU), which labels DNA during the S phase, was added into the medium at 72 hours before transplantation. For immunostaining, the adherent cells were subcultured in chambered slides.
Embolic MCAO and transplantation
All experimental procedures have been approved by the Care of Experimental Animals Committee of Henry Ford Hospital. Experimental adult mice (C57BL/6J, weighing 27 to 35 g) were subjected to MCAO and transplanted with bone marrow nonhematopoietic cells (n = 5). Control mice were subjected to MCAO alone (n = 8), injection of PBS into the ischemic striatum (n = 5), and transplantation of bone marrow nonhematopoietic cells into the normal striatum (n = 5). Middle cerebral artery occlusion was induced using an embolic model developed in the authors' laboratory (Zhang et al., 1997). Briefly, using a facemask, mice were anesthetized with 3.5% halothane and anesthesia was maintained with 1.0% halothane in a 2:1 mixture of N2O:O2. A single intact fibrin-rich 24-hour-old homologous clot (8 mm × 0.000625 mm2, 0.18 μL) was placed at the origin of the MCA through a modified PE-50 catheter. Surgical and physiologic monitoring procedures were identical to those previously published (Zhang et al., 1997). Transplantation of donors into the striatum was carried out according to the method described by Goto et al. (1997). Briefly, at 4 days after MCAO (n = 18), mice were mounted on a stereotaxic frame (Stoelting, Wood Dale, IL, U.S.A.). Using aseptic technique, a burr hole (1 mm) was made on the right side of the skull to expose the dura overlying the right cortex. Semisuspended cells (1 × 105 in 3 μL PBS) were slowly injected over a 10-minute period into the right striatum (antero/posterior = 0 mm, lateral to midline = 2.0 mm, and vertical to dura = 3.5 mm from the bregma) (Franklin and Paxinos, 1997). This position approximates the ischemic boundary zone in the striatum. The needle was retained in the striatum for an additional 5-minute interval to avoid donor reflux. Immunosuppressants were not used in any animal.
Behavioral tests
Each mouse was subjected to a series of behavioral tests to evaluate various aspects of neurologic function by an investigator who was blinded to the experimental groups. Measurements were performed before stroke and at 28 days after stroke. These deficits were various in animals, but primarily consisted of flexion of forelimbs, head moving more than 10° to the vertical axis, and placing test.
Neurologic severity score for mouse behavioral tests
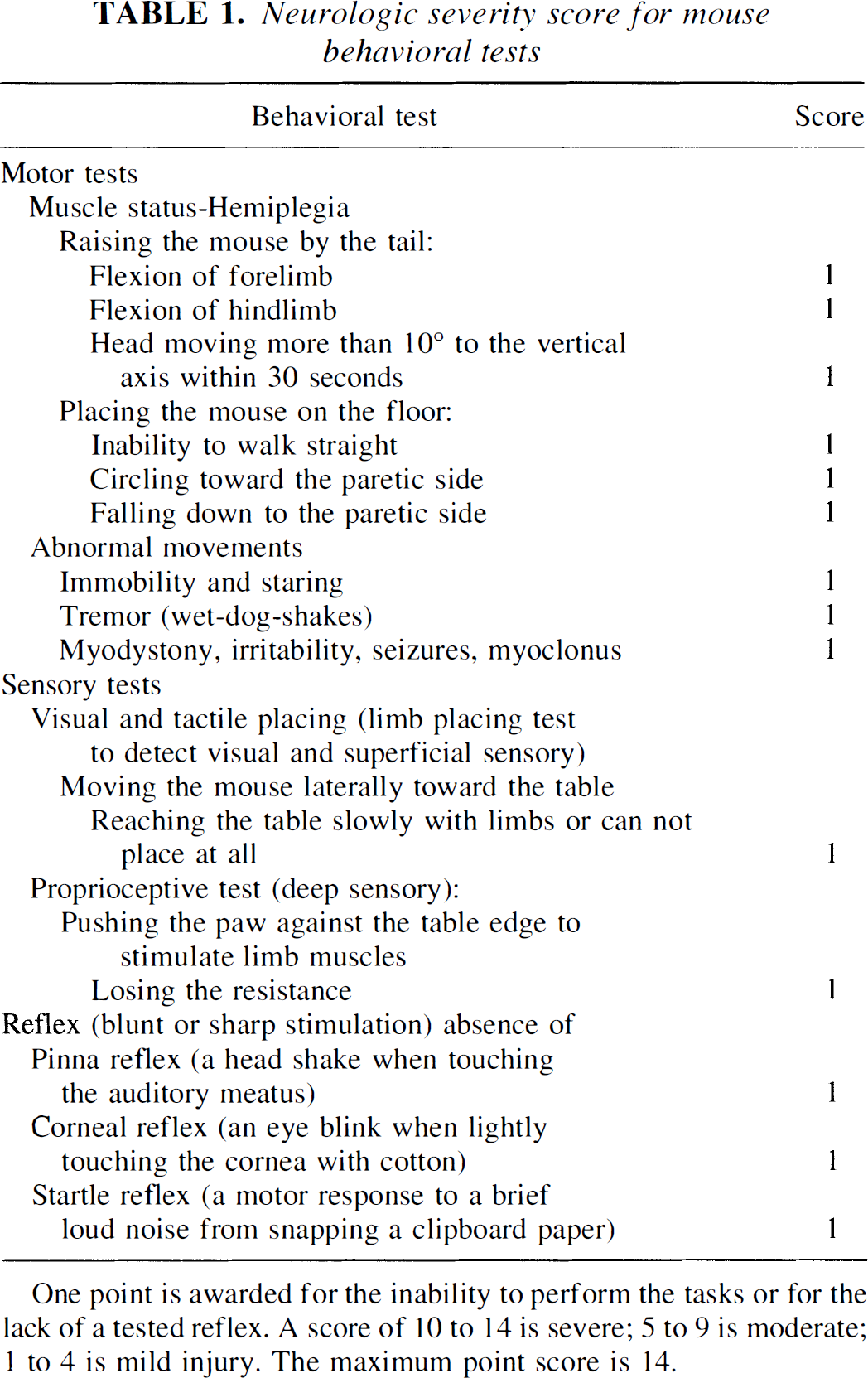
One point is awarded for the inability to perform the tasks or for the lack of a tested reflex. A score of 10 to 14 is severe; 5 to 9 is moderate; 1 to 4 is mild injury. The maximum point score is 14.
Histologic and immunohistochemical assessment
Mice were allowed to survive for 28 days after MCAO when the animals were then reanesthetized with ketamine (44 mg/kg) and xylazine (13 mg/kg). The mouse brains were fixed by transcardial perfusion with saline, followed by perfusion and immersion in 4% paraformadehyde. The forebrain was sectioned into seven equally spaced (1 mm) coronal blocks and then the brain blocks were embedded in paraffin. Sections (6-μm thick) were cut from each block in the coronal plane and stained with hematoxylin and eosin (H&E). Sections were traced using the Global Lab Image analysis system (Data Translation, Malboro, MA, U.S.A.). The indirect lesion area, in which the intact area of the ipsilateral hemisphere was subtracted from the area of the contralateral hemisphere, was calculated (Swanson et al., 1990). The lesion volume is presented as a volume percentage of the lesion compared with the contralateral hemisphere.
Immunohistochemical staining (Li et al., 1998b; Li and Chopp, 1999) was used to detect cells derived from bone marrow nonhematopoietic cells. Briefly, a series of adjacent 6-μm-thick sections (25-μm interval) from each block were analyzed using light and fluorescent microscopy (Olympus, BH-2, Lake Succcess, NY, U.S.A.) and a Bio-Rad MRC 1024 (argon and krypton) laser-scanning confocal imaging system mounted on a Zeiss fluorescent microscope (Bio-Rad, Cambridge, MA, U.S.A.). Cells in chambered slides were fixed with acetone for 5 minutes. To expose the epitope, slides were treated with 50% formamide solution for 30 minutes followed with 2 N HCl for 10 minutes. After blocking in normal serum, sections and cells were treated with the monoclonal antibody against BrdU (dilution 1:100; Calbiochem, San Diego, CA, U.S.A.). After sequential incubation with peroxidase conjugated rabbit anti-mouse IgG (dilution 1:100; Dakopatts, Carpinteria, CA, U.S.A.), the secondary antibody was bound to the first antibody against BrdU. Diaminobenzidine was then used as a chromogen. Counterstaining of sections by hematoxylin was also performed. Cells derived from bone marrow nonhematopoietic cells were identified using morphologic criteria (Pollak and Friede, 1977; Kelemen et al., 1979; Bonner et al., 1999) and by BrdU reactivity (the tracer), present only in the donor cells.
To determine the fate of the BrdU positive cells, the authors performed immunoperoxydase and immunofluorescence double labeling for BrdU and for astrocytes specific antibody, glial fibrillary acidic protein (GFAP, dilution 1:1000; Dakopatts, Carpinteria, CA, U.S.A.). Each coronal section was initially treated by the primary antibody against BrdU, as described above. Sections were then treated with GFAP as the second primary antibody. Fluorescein isothiocyanate conjugated secondary antibody (dilution 1:20; Calbiochem) was added for double-label immunoreactivity identification. Immunofluorescence double-labeling was used using BrdU-Cy5 (red, Accurate, Westbury, NY, U.S.A.) and NeuN (dilution 1:200; Chemicon, Temecula, CA, U.S.A.)-fluorescein isothiocyanate (green). Negative control sections from each animal received identical preparations for immunohistochemical staining, except that primary antibodies were omitted. Fluorochromes on the section were excited by the laser beam at 488 nm (Cy5) and 647 nm (fluorescein isothiocyanate), and emissions were acquired sequentially with a photomultiplier tube through 522 nm and 670 nm emission filters, respectively.
In each animal the authors measured the total number of BrdU reactive cells on 10 sequential slides (25-μm interval) for each block from all 7 blocks (A to G). These sections encompassed the ischemic center and the boundary zone of the damaged tissue and the transplanted area. The light and fluorescein microscopic features on immunoperoxydase preparation used to identify BrdU positive cells include small and round or oval nucleus with intensely stained chromatin (×132 magnification of an Olympus BH2). A conservative estimate of the diameter of the bone marrow-derived cells was 12 μm; thus, the total number of BrdU reactive cells estimated to be within a 1-mm-thick block was equal to the average number measured within 10 sections multiplied by the number of 12-μm-thick sections within 1 mm. The total number of BrdU reactive cells in the whole forebrain was then calculated by adding the numbers of BrdU reactive cells from all 7 blocks. In addition, 6-μm-thick sections from paraffin D block, corresponding to coronal coordinates for bregma −0.5∼0.5 mm (Franklin and Paxinos, 1997), was used using double staining. A total of 500 BrdU reactive cells per animal were counted to obtain the percentage of each cell type specific marker (NeuN, GFAP) colocalized with BrdU in cells. The expression of BrdU reactive cells with cell type specific immunoreactivity was quantified by counting at least 10 fields in each slide at a magnification of ×132.
Statistical analysis
The lesion volume, the number of cells derived from bone marrow, and neurologic deficits (the rotarod test and NSS) of mice were subjected to paired t-tests to evaluate differences between control (MCAO alone) and each treatment. Data are presented as mean values ± SD. The level of significance was set ≤ 0.05.
RESULTS
Donor cells
Cells tightly adhered to the plastic of culture flasks and most were spindle-shaped, but some were round or flat in appearance. These cells replicated for three passages as clonal rosettes without changing their morphology (Fig. 1A). BrdU reactive cells (> 90% of all cells) immediately before transplantation are shown in Fig. 1B.
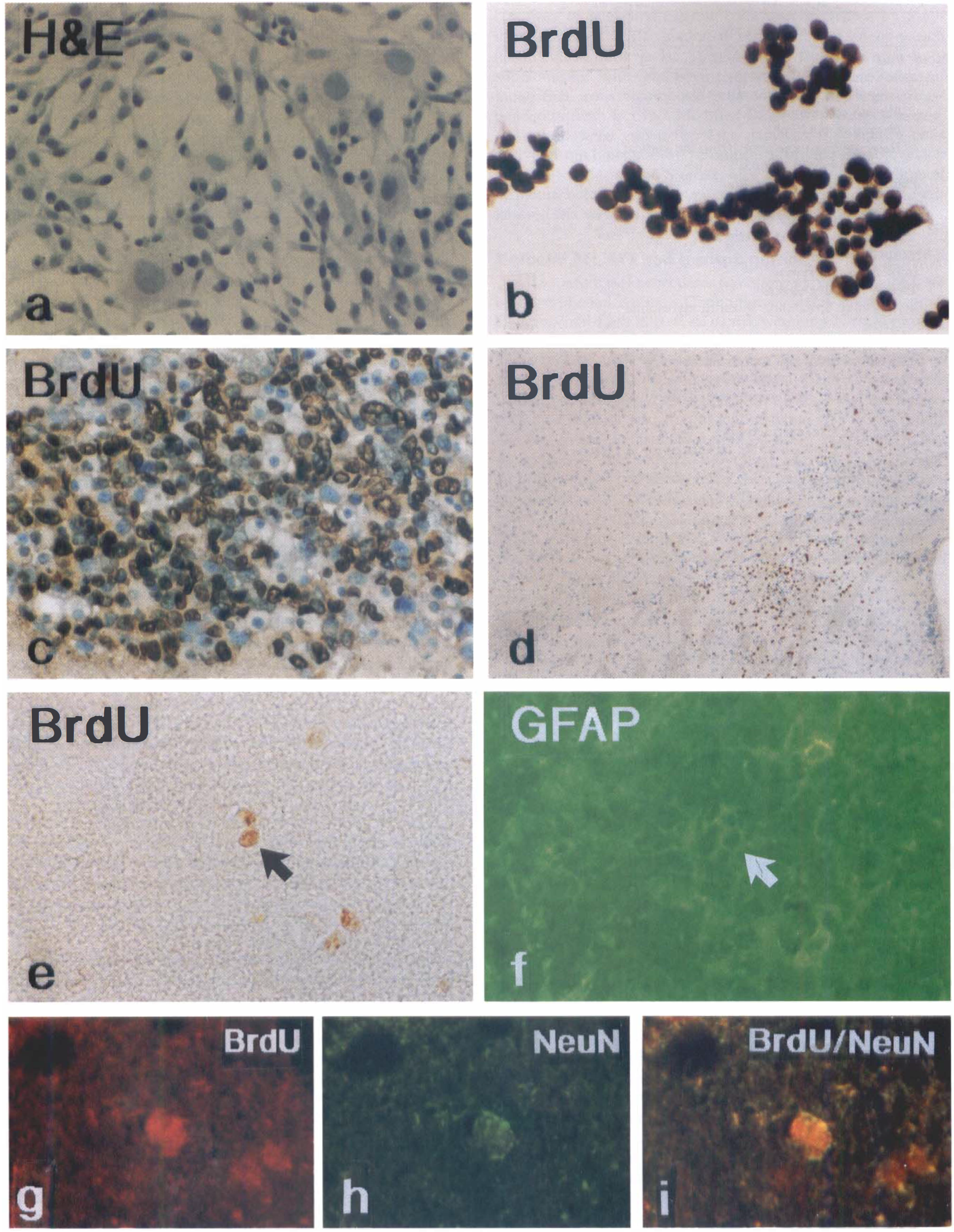
Photomicrographs show the morphologic characteristics of bone marrow nonhematopoietic cells.
Ischemic tissue
The blood gasses were within normal ranges for all animals and did not differ among groups (data not shown). Within 6-μm-thick coronal sections, H&E staining indicated that there was no significant gliosis or infiltration of leukocytes around the bone marrow nonhematopoietic cell transplantation site in all mice. Compared with control mice (17.5 ± 10.2 mm3) subjected to MCAO alone, transplantation of bone marrow nonhematopoietic cells (16.7 ± 19.4 mm3) did not significantly alter the volume of ischemic damage at 28 days after MCAO. No significant change of the ischemic volume was observed in the mice subjected to MCAO with PBS injection (19.4 ± 18.4 mm3).
Survival, migration, and differentiation of bone marrow-derived stromal cells after intrastriatal transplantation
Within the brain tissue, cells derived from bone marrow were characterized by small nuclei, round-to-oval dark purple by H&E staining, and dark brown nuclei by BrdU staining (Fig. 1C) with irregularly shaped and thin cytoplasm. At 28 days after MCAO (24 days after bone marrow cell transplantation), 540 BrdU-labeled cells were detected in the brain of 1 of 5 mice subjected to bone marrow nonhematopoietic cells transplantation into the normal striatum without MCAO. Scattered BrdU reactive cells were only observed along the needle track in the nonischemic brains of the remaining four mice. The numbers of BrdU reactive cells (6,660 ± 2,820) was significantly higher (P < 0.05) in the ischemic mouse brain subjected to MCAO with bone marrow nonhematopoietic cell transplantation than in the nonischemic mouse brain with MSC transplantation. Approximately 0.5% and 6% BrdU reactive cells were readily detected at one month after transplantation of 1 × 105 BrdU labeled cells transplanted into the nonischemic striatum and into the ischemic mouse striatum, respectively.
Cells derived from bone marrow were primarily concentrated within or near the injected site in the striatum (Fig. 1C). BrdU reactive cells derived from bone marrow migrated into multiple areas of the brain including the corpus callosum and cortex, at a distance of at least 2.2 mm (Fig. 1D). Double staining demonstrated that approximately 8% (Fig. 1E to 1F) and 1% (Fig. 1G to 1I) of BrdU reactive cells expressed GFAP and NeuN phenotypes, respectively.
Behavioral tests
No significant differences of the rotarod test were detected among groups before surgery. The rotarod test and NSS demonstrated neurologic and functional deficits resulting from the MCAO ischemic insult, and no significant differences were noted among groups before transplantation. Within each group, the behavioral deficits evaluated by the modified rotarod test and NSS after MCAO showed a progressive recovery over time from 1 day to 28 days after MCAO. At 28 days after MCAO, mice transplanted with bone marrow nonhematopoietic cells showed a significant improvement in the rotarod test (P < 0.05; Fig. 2A) and in NSS (P < 0.05; Fig. 2B), compared with controls subjected to MCAO alone. Nonischemic mice subjected to transplantation of bone marrow nonhematopoietic cells into the striatum performed all tasks, thus obtaining an NSS of 0.
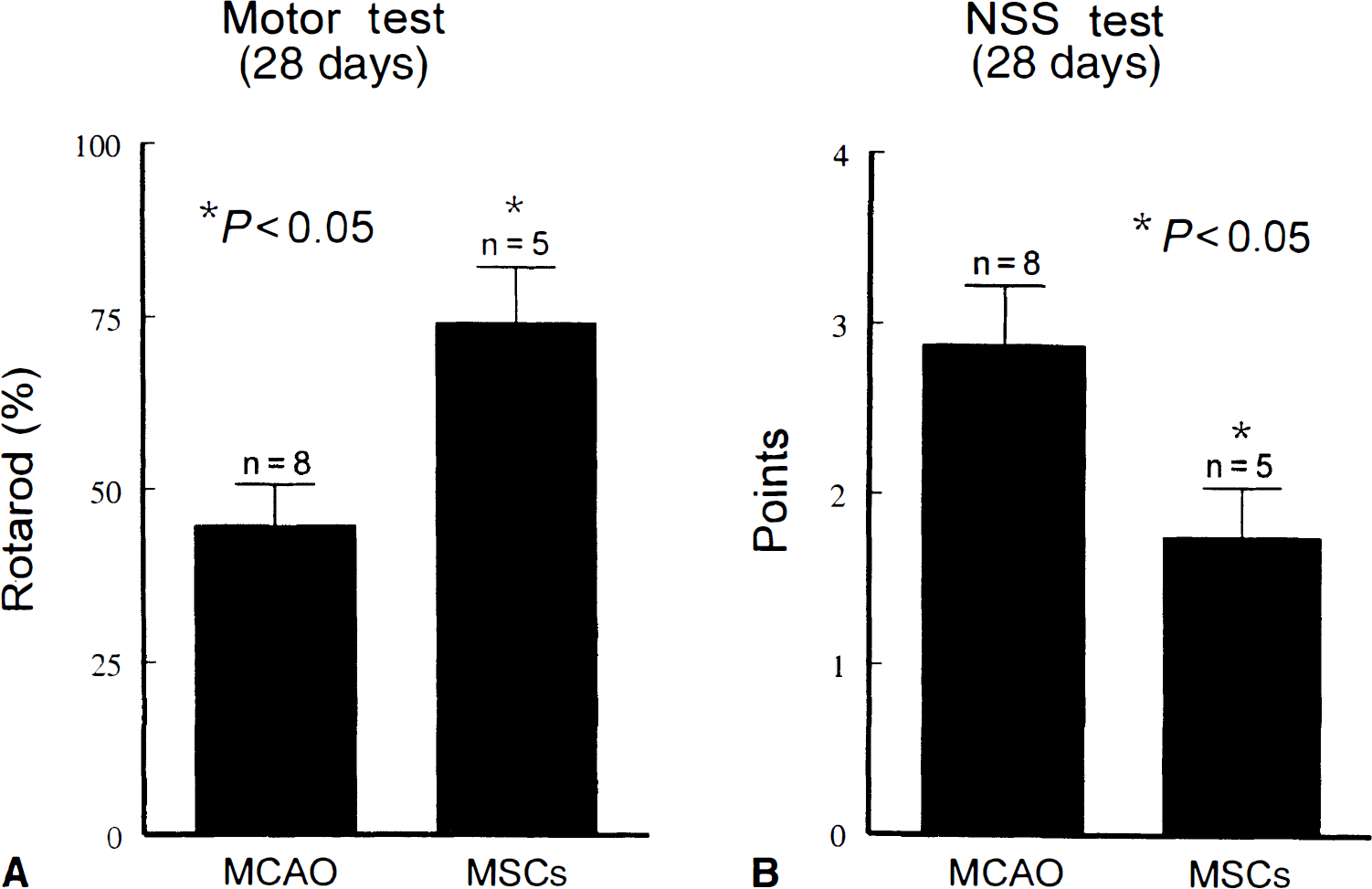
At 28 days after middle cerebral artery occlusion (MCAO), neurologic functional tests indicate partial recovery in all mice with or without bone marrow nonhematopoietic cell transplantation. Neurologic severity score (NSS) test score points are low. Various deficits were present in animals, however, most of the functional deficits reside in flexion of forelimbs, head movement more than 10° to the vertical axis, and the paw placing test. Mice transplanted with marrow stromal cells (MSCs) showed a significant improvement in the rotarod test (
DISCUSSION
The current data demonstrate that adult bone marrow nonhematopoietic cells survive, migrate, and differentiate into phenotypes of parenchymal cells in the adult mouse brains after ischemia. Intrastriatal transplantation of bone marrow nonhematopoietic cells was effective in reducing ischemia-induced behavioral deficits in mice.
Stem cells are multipotent, that is, they can form different cell types. Stem cells have been isolated from various tissues in animals and humans, including embryonic (embryo), hematopoietic (bone marrow), mesenchymal (bone marrow), and even neuronal (adult brain) tissues. Two kinds of stem cells (HSCs and MSCs) localize in normal adult bone marrow. Hematopoietic stem cells provide a continuous source of progenitors for red blood cells, platelets, monocytes, granulocytes, and lymphocytes. Marrow stromal cells readily differentiate into osteoblasts, chondroblasts, adipocytes, and even myoblasts and brain glial cells (Vogel, 2000; Prockop, 1997). Marrow stromal cells have several advantages over HSCs (unpublished data, 2000) for use in cell therapy after MCAO. The fraction of HSCs of fresh bone marrow cells placed into culture was relatively high. Most of the HSCs were lost as the bone marrow cells were maintained as primary cultures for 2 or 3 weeks, and HSCs were difficult to expand in culture. Hematopoietic stem cells did not express neural phenotypes in ischemic mouse brains at 28 days after transplantation. Bone marrow nonhematopoietic cells are easily isolated in the medium by their tendency to adhere to plastic and readily proliferate in culture.
Gage et al. (1995) indicate that stem cells from one region will, when transplanted to ectopic central nervous system sites, differentiate with a terminal phenotype appropriate for that ectopic site. Importantly, a lesioned central nervous system is a different environment than an intact central nervous system and markedly alters the terminal differentiated phenotype of the transplanted cells (Gage et al., 1995). In their studies, neuronal differentiation was restricted to the lesioned area. In the current model, the authors found that MSCs retained the capacity to respond to local epigenetic signals in the nonischemic and ischemic brain, as MSCs can survive and differentiate into neuronal (NeuN) and glial cell (GFAP) phenotypes. In all ischemic mice, the sum fraction of double-labeled cells was ∼1% BrdU-NeuN and ∼8% BrdU-GFAP.
BrdU reactive cells survive and differentiate not only in the ischemic brain, but they are also present in the brain without ischemia. The current data indicate that ∼0.5% of BrdU reactive cells survive at 24 days after injection into the normal striatum of a mouse. The survival rate, however, is less than the ∼20% of adherent heterogeneous bone marrow cells reported to survive at 30 days after striatal implantation in normal rats (Azizi et al., 1998). The small number (∼0.5%) of BrdU reactive cells that survive in the normal mouse brain might be because of the possibility that infused cells divide rapidly and consequently dilute the label below levels of detection of BrdU, or because of the different animal strains used. However, the small number of BrdU-labeled cells is more likely attributed to the fact that the transplanted bone marrow nonhematopoietic cells are a heterogeneous population containing few MSCs. Phinney et al. (1999) and Kopen et al. (1999) recently reported a method to eliminate unwanted adherent cells from the cultures through immunodepletion. After immunodepletion, MSCs appear as a uniform population of fibroblastoid-shaped cells that are devoid of CD11b or CD45 expression (cell surface receptors of myelopoietic cells). They estimate that MSCs represent only 10% to 20% of the total cell population in plastic adherent marrow cell cultures. Despite the heterogeneous nature of these cultures, the ischemic microenvironment appears to influence and facilitate bone marrow derived stromal cell survival and proliferation. The ischemic boundary zone is threatened tissue. This ischemic boundary zone expresses developmental genes and proteins (Li and Chopp, 1999). The current data demonstrated that the BrdU reactive cells were highly concentrated in the ischemic boundary zone after MCAO. Molecules expressed in the ischemic boundary zone might play a role in the subsequent survival and differentiation, or migration, or both, of transplanted bone marrow nonhematopoietic cells.
The current data indicate that intrastriatal transplantation of bone marrow nonhematopoietic cells improves behavioral restoration after ischemia in mice. These data were consistent with earlier reports showing that grafts of fetal striatal cells implanted into the caudatoputamen partially restore function after stroke (Aihara et al., 1994; Nishino et al., 1993). The authors used two tests (the rotarod test and NSS) to detect the neurologic behavioral responses to experimental ischemia in the mice. The rotarod test is a well-established procedure for testing limb motor coordination and balance aspects of motor performance in mice. In the current study, the authors confirmed that the accelerating rotarod task is a sensitive index to detect mild motor deficits at one month after the onset of ischemia with bone marrow nonhematopoietic cell transplantation. Similar to the rotarod test, the NSS can be used to assess mild motor, sensory, and reflex deficits after stroke. The NSS is an economical, simple, and rapid test for animals in characterizing the asymmetric behavior in unilateral damage.
Bone marrow nonhematopoietic cells contain stem cells that may serve to replenish lost cells in the ischemic damaged brain, and they contain a variety of cells that secrete substances that may lead to recovery. However, in the current study, the authors could not directly relate numbers of BrdU labeled cells to functional improvement. At this point, observations are limited to survival, migration, and differentiation of BrdU labeled cells, and the improvement of functional outcome with bone marrow nonhematopoietic cell transplantation. However, it is unlikely that the approximate 6,660 cells derived from bone marrow that survive and ∼1% and ∼8% of BrdU reactive cells with neural phenotypes of NeuN and GFAP were capable of providing behavioral improvement in mice treated with MSCs after MCAO. The study by Horwitz et al. (1999) provides a first glimpse of the therapeutic potential of a small fraction of MSCs in patients with osteogenesis imperfecta. To explain how the presence of only 1.5% to 2.0% donor mesenchymal cells in their study could lead to improvement in behavioral recovery, they indicate that perhaps the recipients are gradually replacing the abnormal matrix with normal collagen produced from the rare donor cells. Interaction of transplanted tissue with the host brain may also lead to production of trophic factors (Unsicker, 1993, 1994). This suggests that bone marrow nonhematopoietic cells may secrete neurotrophins in the ischemic brain. Bone marrow nonhematopoietic cells normally support hematopoietic cells and bone cells and secrete a number of hematopoietic cytokines and factors, for example, bone marrow nonhematopoietic cells express bone morphogenetic proteins (BMPs) (Bi et al., 1999). Recent studies demonstrate that BMPs trigger neuronal differentiation of neocortical precursors within the ventricular zone (Li et al., 1998a). The bone marrow nonhematopoietic cells secrete colony-stimulating factor-1, interleukins, stem cell factor, and other hematopoietic regulatory molecules (Maysinger et al., 1996; Eaves et al., 1991). The hematopoietic cytokine colony-stimulating factor-1 is a growth factor in the central nervous system (Maysinger et al., 1996). In summary, the current data indicate that bone marrow nonhematopoietic cells transplanted into brain generate neural-like cells and promote the recovery of neurologic deficit.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Cecylia Powers, Cynthia Roberts, and Xiuli Zhang for their technical assistance, and Denice Bliesath and Rita Tobey for their secretarial support.
