Abstract
Bone marrow-derived cells participate in remodeling processes of many ischemia-associated diseases, which has raised hopes for the use of bone marrow as a source for cell-based therapeutic approaches. To study the participation of bone marrow-derived cells in a stroke model, bone marrow from C57BL/6-TgN(ACTbEGFP)1Osb mice that express green fluorescent protein (GFP) in all cells was transplanted into C57BL/6J mice. The recipient mice underwent permanent occlusion of the middle cerebral artery, and bone marrow-derived cells were tracked by fluorescence. The authors investigated the involvement of bone marrow-derived cells in repair processes 6 weeks and 6 months after infarction. Six weeks after occlusion of the artery, more than 90% of the GFP-positive cells in the infarct border zone were microglial cells. Very few GFP-positive cells expressed endothelial markers in the infarct/infarct border zone, and no bone marrow-derived cells transdifferentiated into astrocytes, neurons, or oligodendroglial cells at all time points investigated. The results indicate the need for additional experimental studies to determine whether therapeutic application of nonselected bone marrow will replenish brain cells beyond an increase in microglial engraftment.
Cells derived from bone marrow (BM) participate in repair mechanisms after many ischemia related diseases, such as myocardial infarction (Orlic et al., 2001a, b ; Rafii et al., 2002), hindlimb ischemia (Asahara et al., 1999; Ikenaga et al., 2001) and stroke (Hess et al., 2002; Priller et al., 2001a). Within the central nervous system, BM-derived cells have been shown to give rise to all three principal cell types: neurons (Brazelton et al., 2000; Mezey et al., 2000; Priller et al., 2001b), astrocytes (Eglitis et al., 1997, 1999) and oligodendrocytes (Zhao et al., 2002), as well as endothelial cells (ECs) of brain vessels (Hess et al., 2002; Zhang et al., 2002). The finding that circulating cells enter the brain at low numbers during normal adult life and differentiate into neurons and glial cells suggests that BM-derived cells participate in the homeostasis of the organ. Recent studies, however, suggest that the potential of BM-derived cells to differentiate into organ specific cells might be less pronounced than anticipated (Castro et al., 2002; Wagers et al., 2002).
After experimental cerebral ischemia, the number of BM-derived cells that enter the brain increases rapidly (Priller et al., 2001a). Although most of these cells are neutrophils, monocytes, and other blood cell types involved in the inflammatory response, recent reports show that blood vessels in close association with the ischemic region contain BM-derived cells expressing EC-specific markers, such as von Willebrand Factor and CD31 (Hess et al., 2002). In these blood vessels, 42% of all endothelial cells were BM-derived 3 d after cerebral infarction. After 7 and 14 d the contribution numbers slightly decreased to 31% and 26%, respectively. Thus, survival of BM-derived ECs might be a transient phenomenon, raising the question whether there is a continuous loss of these cells at later survival times. In addition, although BM-derived cells have been shown to give rise to neurons, astrocytes, and oligodendrocytes in normal adult brains, until now only a limited number of neurons (Hess et al., 2002) and macroglial cells (Eglitis et al., 1999) of BM origin were described under conditions of cerebral ischemia.
The aim of our study was to analyze to what degree BM-derived cells participate in late-stage repair mechanisms and brain tissue regeneration following cerebral infarction. We therefore investigated the distribution and differentiation potential of BM-derived cells 6 weeks and 6 months after experimental stroke in adult mice.
MATERIALS AND METHODS
Irradiation, bone marrow transplantation, and flow cytometry of peripheral blood cells
Eight-week-old C57BL/6J recipient male mice were irradiated by a dose of 11.0 Gy (1,100 rads) to ablate the marrow. Within 6 h, irradiated mice received 1 × 106 donor cells in a volume of 0.1 mL each by tail vein injection. Bone marrow was harvested by flushing tibias and femurs of 8- to 12-week-old female C57BL/6-TgN(ACTbEGFP)1Osb transgenic mice (Jackson Laboratory, Bar Harbor, ME, U.S.A.) with RPMI 1640 (1% fetal calf serum and 1% penicillin/streptomycin). The marrow cores were dissociated by passing through a 21-gauge needle, and the cells were counted after red-cell lysis. In the C57BL/6-TgN(ACTbEGFP)1Osb transgenic mouse line, the “enhanced” GFP complementary DNA is under the control of the chicken β-actin promoter and the cytomegalovirus enhancer thus rendering all tissues, with the exception of erythrocytes and hair, fluorescent green under an ultraviolet light. Six weeks after BM transplantation, examination of peripheral blood samples by flow cytometry (Becton Dickinson, Heidelberg, Germany) revealed that ≥70% of the nucleated cells expressed GFP. All animals survived the transplantation.
Murine cerebral infarction model
All stroke experiments were performed in 14-week-old male mice (C57BL/6J; Charles River, Deisenhofen, Germany) weighing 25 to 32 g. Animals were housed under standard conditions with free access to mice chow and tap water before and after surgery. Irreversible occlusion of the left middle cerebral artery (MCA) was performed as described previously (Beck et al., 2002; Welsh et al., 1987). Briefly, animals were anesthetized by intraperitoneal injection of ketamine (80 mg/kg; Apharmo, Arnhem, Netherlands) and xylazine (10 mg/kg; Bayer, Leverkusen, Germany) and body temperature was maintained at 37°C by means of a rectal probe connected to a heating pad. Using an operating microscope, a U-shape incision was made between the left ear and the left eye. The top and back segments of the temporal muscle were transected, and the skull was exposed by retraction of the temporal muscle. A small opening (1 to 2 mm in diameter) was made in the region over the MCA with a handheld drill. The MCA was occluded by ligation with 10–0 nylon thread (Ethylon, Norderstedt, Germany) and transected distally to the ligation point. Finally, retracted soft tissues were replaced, the wounds were sutured, and the mice were placed back into their cages. Sham-operated control animals were prepared in a similar manner except that the exposed MCA was not occluded. Body temperature was maintained during surgery and until the animals regained consciousness. Thereafter, rectal temperature was checked every 10 to 15 minutes during the following 2 h and, if necessary, was corrected to 37°C by placing a heating pad below the cage. After middle cerebral artery occlusion (MCAO), animals were allowed to survive for 3 d (n = 5), 6 weeks (n = 10 infarcted and n = 6 sham-operated mice), or 6 months (n = 6 infarcted and n = 6 sham operated mice).
Magnetic resonance imaging
To verify successful occlusion of the MCA and to visualize the infarcted territory, we used magnetic resonance imaging (MRI) to analyze the brain of all animals, 24 h after vessel occlusion. Infarct volume was determined by means of a T2-weighted quantitative in vivo MRI as described previously (Plate et al., 1999).
Tissue fixation, embedding, and sectioning: histologic analysis
Three days, 6 weeks, or 6 months after MCAO mice were deeply anesthetized and perfused via the left ventricle of the heart with saline followed by 4% paraformaldehyde. Brain tissues were dissected, stored in 4% paraformaldehyde overnight, and placed in 30% sucrose for 24 h. After cryoprotection with sucrose, tissues were frozen in OCT embedding medium (Sakura Finetec, Torrance, CA, U.S.A.) using dry-ice–cooled isopentane. Tissues were stored at −80°C until further processing. Embedded frozen tissues were sectioned at 10 μm with a Leica cryostat (Leica, Wetzlar, Germany). Frozen sections thawed on silanized (3-aminopropyltriethoxysilane; Fluka, Taufkirchen, Germany) glass slides and dried for at least 2 h at 37°C. We performed serial sectioning of the infarcted area using every 10th section for an antigen-specific antibody staining procedure. To determine the infarct size after 6 weeks' or 6 months' survival, twelve 10-μm-thick frozen sections of each animal were taken at 100-μm intervals. Sections were stained with hematoxylin and eosin, and the infarcted area as well as the area of the ipsilateral hemisphere were determined planimetrically (Optimas 5.1; BioScan Incorporated, Edmondes, WA, U.S.A.). The volume of the evaluated ipsilateral hemisphere and the volume of the evaluated infarcted area itself were calculated by multiplying the hemisphere area/infarct area of each section by the distance (100 μm) between successive sections. The volume of the infarct itself is expressed as percentage of the volume of the evaluated ipsilateral hemisphere.
Quantification of regional engraftment of GFP-positive cells
Engraftment of GFP-positive cells was quantified by counting all GFP-expressing cells in different areas (infarct core, periinfarct region, contralateral cortex, ipsilateral and contralateral choroid plexus, as well as ipsilateral and contralateral lateral ventricular wall of infarcted and sham-operated brains of four representative sections per animal (Table1).
To quantify the number of BM-derived microglial cells, we analyzed eight representative sections per animal that were double-stained using antibodies against the Iba-1 and GFP proteins. In similar fashion, we analyzed endothelial cells using antibodies against vWF and GFP. We then calculated double-stained cells as a percentage of all analyzed microglial or endothelial cells, respectively. This analysis was based on 50 randomly chosen GFP-positive cells as well as 15 vessels in the periinfarct region and in the corresponding parts of the ipsilateral hemisphere. In addition, we analyzed all cells in the ipsilateral and contralateral choroid plexus.
Immunohistochemistry
Immunohistochemistry was carried out as described previously (Beck et al., 2000, 2002). Briefly, cryosections were washed thoroughly in PBS before and after each incubation step. Nonspecific binding sites were blocked by incubation in 20% normal goat serum (NGS; Sigma, Deisenhofen, Germany) and 5% bovine serum albumin/PBS (BSA, Fraction V; Sigma). Sections were microwaved in 0.01-mol/L sodium citrate for 2 to 3 minutes, carefully avoiding boiling. The following reagents were used: mouse anti-glial fibrillary acidic protein (GFAP) (Chemicon, Temecula, CA, U.S.A.), mouse anti-2′,3″-cyclic nucleotide 3″-phophodiesterase (CNPase; Boehringer Mannheim, Mannheim, Germany), mouse anti-neuronal nuclei (NeuN; Chemicon), mouse anti-actin, smooth muscle monoclonal antibody (Chemicon), rabbit polyclonal anti-Iba-1 (kindly provided by Drs. Y. Imai and S. Kohsaka; Imai et al., 1996), mouse anti-HuC/HuD neuronal protein (Molecular Probes, Eugene, OR, U.S.A.), TRITC-labeled lectin from helix pomatia (Sigma), rabbit polyclonal anti-human von Willebrand Factor (DAKO, Glostrup, Denmark), rat anti-mouse F4/80 (Abcam, Oxford, U.K.), anti-green fluorescent protein (anti-GFP), rabbit IgG fraction and anti-green fluorescent protein (anti-GFP488), rabbit IgG fraction, and Alexa Fluor 488 conjugate for double-labeling with other rabbit polyclonal antibodies (both antibodies from Molecular Probes). Omission of the primary antibody served as a control. The following secondary antibodies were used: goat anti-mouse Alexa 568 conjugated IgG (7.5 μg/mL; Molecular Probes), goat anti-rabbit Alexa 568 conjugated IgG (7.5 μg/mL; Molecular Probes), mouse anti-rat Alexa 568 conjugated IgG (7.5 μg/ml; Molecular Probes), goat anti-rabbit Alexa 488 conjugated IgG (7.5 μg/ml; Molecular Probes), or mouse anti-rat Alexa 488 conjugated IgG (7.5 μg/mL; Molecular Probes), respectively. For mouse monoclonal antibodies, we used the M.O.M. immunodetection kit (Vector, Burlingame, CA, U.S.A.) to reduce unspecific background staining. Sections were counterstained with DAPI (4,6 diamidino-2-phenylindole). Evaluation of double-staining was performed using a Leica fluorescence microscope as well as a Zeiss Axioplan 2 Imaging microscope.
RESULTS
Evaluation of infarct size
To verify successful occlusion of the MCA, each brain was scanned by nuclear MRI 24 h after stroke. Permanent focal ischemia resulted in a well-delineated tissue injury in the territory of the occluded middle cerebral artery, including parts of the insular and parietal cortex. The total area of edema was 47 mm3 ±5.4 SEM; n = 16). After the scan, animals were assigned to the 6 weeks (49 mm3 ±8.4 SEM; n = 10) or the 6 months group (45 mm3 ±10.1 SEM; n = 6). A representative NMR image is shown in Figure 1a. The infarcted area was mainly localized to the parietal cortex. Sham-operated mice had no detectable infarction in any part of the brain.
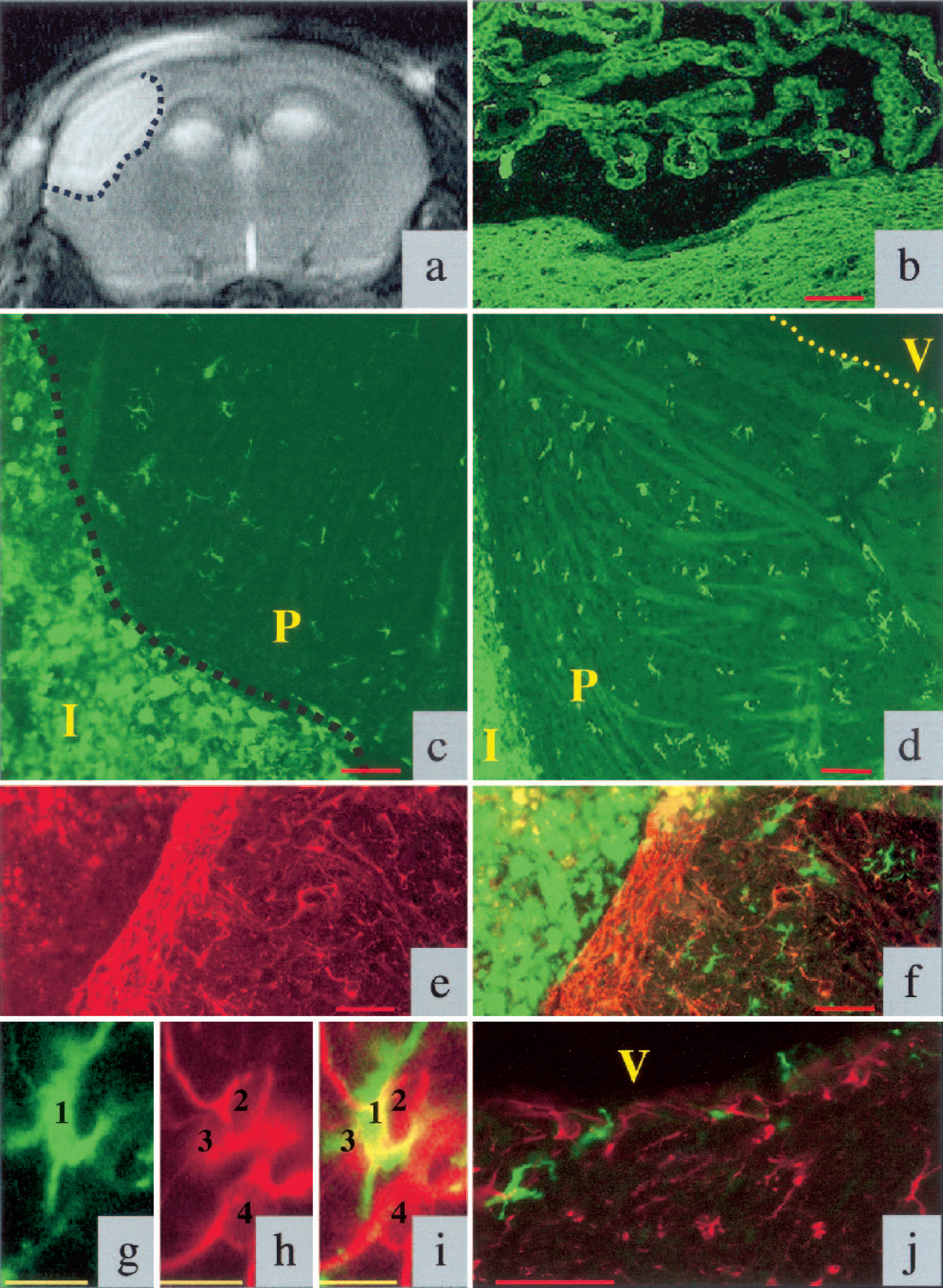
T2-weighted nuclear magnetic resonance image of a mouse brain 24 h after middle cerebral artery occlusion (MCAO). Ischemic infarct is delineated by a dotted line and appears bright on this image (
Six weeks after MCAO, 5.31% of the evaluated ipsilateral hemisphere appeared infarcted. Six months after MCAO, the size of the infarct shrunk to 2.8% of the evaluated ipsilateral hemisphere.
Distribution of GFP-positive cells in infarcted and sham-operated mouse brains
To study the contribution of BM-derived cells in tissue repair and regeneration after stroke, we transplanted BM from C57BL/6-TgN(ACTbEGFP)1Osb mice into C57BL/6J wild-type mice. The recipient mice underwent permanent occlusion of the MCA, and BM-derived cells were tracked by fluorescence. All animals survived the BM transplantation, and only 1 of 34 animals died a few hours after surgery.
We analyzed brain tissue from 21 infarcted and 12 sham-operated mice 3 d (5 infarcted animals), 6 weeks (10 infarcted, 6 sham-operated animals), and 6 months (6 infarcted, 6 sham-operated animals) after occlusion of the MCA. In all sham-operated animals, we observed scattered GFP-positive cells in the choroid plexus, as well as single GFP-positive cells in association with vascular structures, especially with the leptomeningeal blood vessels (data not shown). In addition, we observed few GFP-expressing cells in the olfactory bulb (data not shown).
In comparison with sham-operated animals, infarcted mouse brains showed higher numbers of GFP-positive cells in the ipsilateral hemisphere. Thus, cerebral ischemia is a strong stimulus for the recruitment of BM-derived cells towards the infarcted brain tissue. Six weeks after MCAO, most of the GFP-positive cells surrounded the infarct area (Fig. 1c); many were present within the choroid plexus (Fig. 1b) and in the area between the lateral ventricle and the infarcted zone (Fig. 1d). Thus, GFP-positive cells appeared to have migrated from the ventricular system to the infarcted area. We also detected an accumulation of GFP-positive cells in the wall of the lateral ventricle in the infarcted hemisphere (Fig. 1j).
Six months after MCAO, the number of GFP-positive cells in the periinfarct area was reduced. However, the total number of GFP-positive cells in noninfarcted regions of the brain increased with time primarily owing to the continuous turnover of blood-borne perivascular macrophages. The absolute numbers of GFP-positive cells within the examined brain regions are provided in Table 1.
Bone marrow-derived cells in different parts of the brain following MCAO or sham surgery
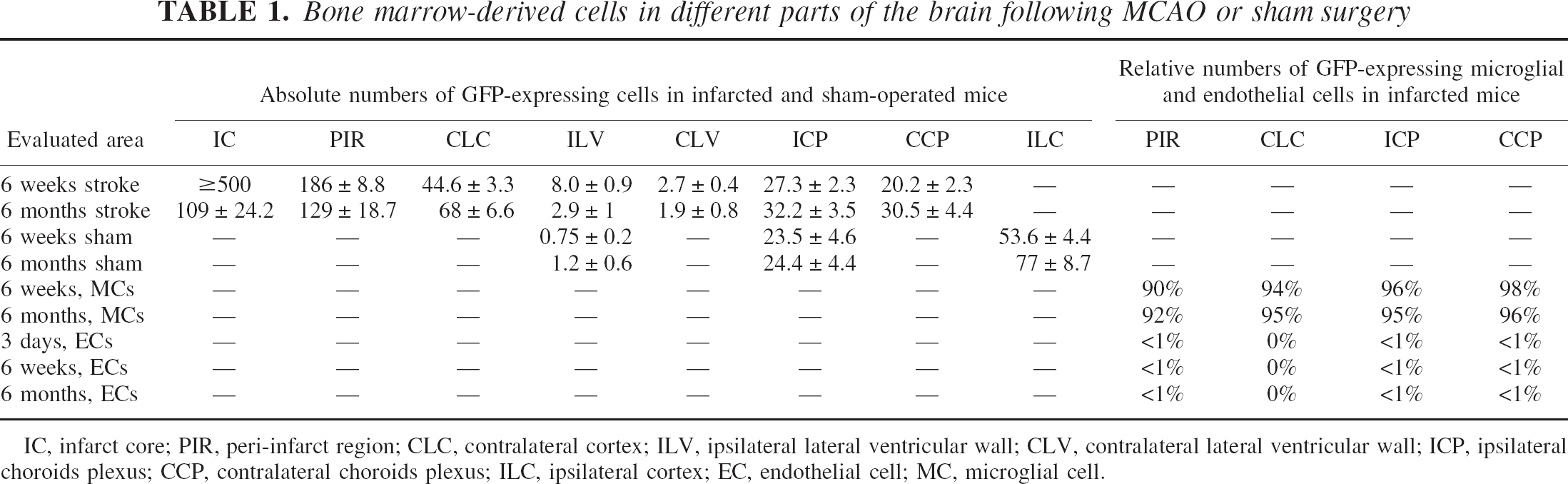
IC, infarct core; PIR, peri-infarct region; CLC, contralateral cortex; ILV, ipsilateral lateral ventricular wall; CLV, contralateral lateral ventricular wall; ICP, ipsilateral choroids plexus; CCP, contralateral choroids plexus; ILC, ipsilateral cortex; EC, endothelial cell; MC, microglial cell.
Characterization of GFP-expressing cell types
We further analyzed the phenotype of the BM-derived cells, marked by GFP expression, with immunofluorescence techniques using antibodies that recognize protein antigens specifically expressed in astrocytes, oligodendrocytes, microglial cells, neurons, and cells of the vascular system.
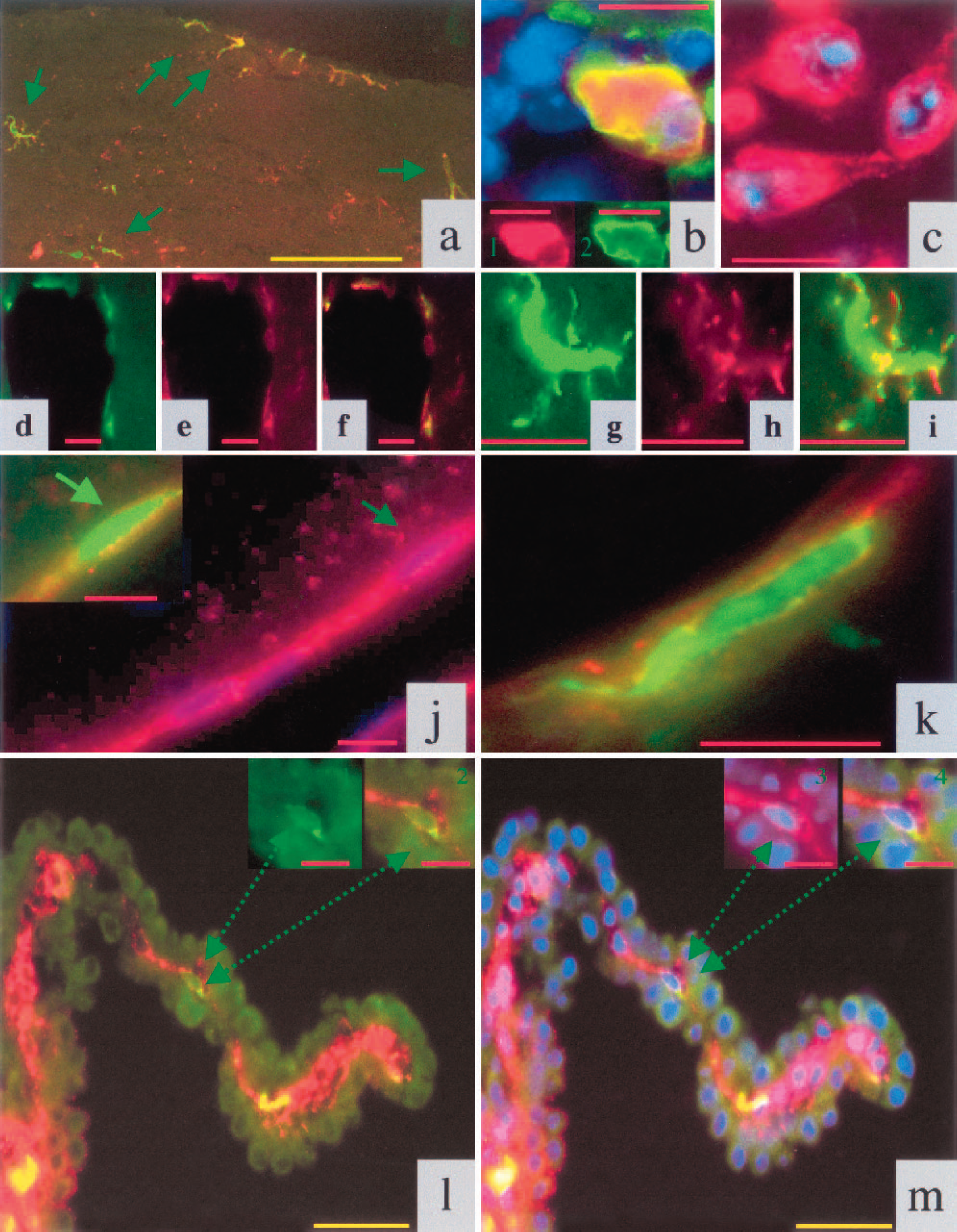
(
DISCUSSION
After experimental stroke, the number of BM-derived cells within the brain increases rapidly (Priller et al.,. 2001a). Our results indicate that GFP-positive, BM-derived cells may enter the brain via the blood stream using two main pathways: (1) by migration through blood vessels in the infarct border zone and (2) through vessels in the choroid plexus. The main function of the choroid plexus is thought to be the continuous production of cerebrospinal fluid. The high constitutive expression of vascular endothelial growth factor in choroid plexus epithelium (Breier et al., 1992) has been postulated to induce vascular permeability and to keep plexus vessels permissive for fluid transmission. Our findings suggest that the choroid plexus acts as an entry port for BM-derived cells that migrate into the brain and participate in repair processes such as microglial response or angiogenesis. Numerous GFP-positive cells were detected in the ventricular walls, in particular on the side of attachment of the choroid plexus, and in the brain parenchyma towards the infarcted area. A possible scenario of how BM-derived cells reach the damaged area would involve arrival via the blood stream in the choroid plexus and subsequent migration along the ventricular wall through the white matter toward the infarct.
Several recent studies showed that BM is a source of endothelial and organ-specific progenitor cells (Asahara et al., 1999; Brazelton et al., 2000; Hess et al., 2002; Mezey et al., 2000; Priller et al., 2001b; Zhang et al., 2002). In the brain, even under normal conditions, BM cells were shown to enter the organ and to differentiate into neurons (Brazelton et al., 2000; Mezey et al., 2000; Priller et al., 2001b) or astrocytes (Eglitis and Mezey, 1997). Other studies however, suggested that under physiologic as well as pathologic conditions, the potential of BM-derived cells to differentiate into organ-specific cells might be less pronounced than anticipated (Castro et al., 2002; Hess et al., 2002; Wagers et al., 2002). In line with these latter findings, our study of late-stage repair mechanisms after cerebral ischemia points out that the differentiation potential of BM-derived brain cells might be limited. Previous studies found a low number of BM-derived neurons (two to four per section) in the ischemic zone (Hess et al., 2002). We detected a similar number of BM-derived neurons in the infarcted area 6 weeks after MCAO. However, these NeuN- or HuC/HuD-positive cells did not show the typical neuronal morphology, such as elaborate dendritic or axonal networks. In addition, the distribution of GFP staining was mainly localized to the cell borders. BM-derived cells expressing neuronal markers were only detectable directly in the ischemia-affected area, but never adjacent to the infarct, or in other parts of the cortex. Thus, it appears unlikely that these cells represent functional neurons. Six months after MCAO we could not detect GFP-expressing neuron in any part of the cortex.
Although it has been proposed that BM-derived cells are able to differentiate into astroglia (Eglitis et al., 1997, 1999) and oligodendroglia (Zhao et al., 2002), in accordance with other studies (Hess et al., 2002; Priller et al., 2001a), we did not detect GFP-positive astrocytes or oligodendrocytes under the conditions of cerebral ischemia. Astroglia are thought to play a pivotal role in the repair mechanisms of the brain. Six weeks after MCAO, astrocytes delineate the infarcted area. No GFP-positive cells were incorporated in this border zone consisting of astrocytes, whereas both GFP-negative astrocytes and GFP-positive microglial cells were located in close proximity in the tissue surrounding the astrocytic wall.
A recent report also showed that BM-derived, GFP-marked endothelial cells incorporate into the vasculature of the brain in periinfarct vessels as soon as 24 h after experimental stroke (Hess et al., 2002). The highest number of GFP-positive endothelial cells was detected 3 d after the infarct (42% of all ECs in GFP-positive periinfarct vessels); this number decreased to 26% of all counted ECs at 14 d.
These data raised the question of whether incorporation of BM-derived ECs after stroke is a transient phenomenon. Therefore we analyzed the participation and differentiation potential of BM-derived GFP-positive cells toward the endothelial cell linage in our mouse stroke model at late repair stages (6 weeks and 6 months after infarction). In contrast to reports at earlier time points (Hess et al., 2002), we detected very few GFP-expressing endothelial cells in periinfarct vessels. We analyzed at least 15 periinfarct vessels per section and 8 sections per animal. The number of GFP-positive cells was less than 1% (6 weeks and 6 months) of all counted ECs. These results suggest either that survival of BM-derived ECs might be limited after cerebral ischemia, or that the differentiation potential of BM-derived cells toward the endothelial cell lineage is low under these experimental conditions. To distinguish between the two possibilities, we analyzed transplanted animals 3 d after stroke and evaluated the number of GFP-expressing endothelial cells. Similar to late stages, we detected only very few GFP-positive endothelial cells 3 d after MCAO. Our results are in accordance to studies that used transgenic mice expressing β-galactosidase in endothelial cells under the transcriptional regulation of the Tie2 promoter as BM donors (Zhang et al., 2002). In this case no β-galactosidase–marked endothelial progenitor cells were detected at the border of the infarct 48 h after stroke. One month after the infarct, a few lacZ-positive endothelial cells (not quantified) were present in the infarct border zone (Zhang et al., 2002).
There are several possible explanations for the discrepancies regarding the differentiation potential of BM-derived cells among the different studies. First, the choice of the stroke model (permanent versus transient cerebral ischemia, embolic cerebral ischemia) might influence the number of transdifferentiated cells. Reperfusion injury in transient models of focal cerebral ischemia might lead to a different gene-induction pattern. Second, methods with various degrees of sensitivity were used to track BM-derived cells, including nuclear β-galactosidase and Y-chromosome tagging. We believe that this possibility could not explain our results because fluorescently labeled cells with EGFP under the influence of the chicken β-actin promoter and the cytomegalovirus enhancer-driven EGFP is the most powerful tool for tracing engrafted cells (Fukada et al., 2002; Okabe et al., 1997). EGFP is easily detectable and allows detailed analysis of cell types by double-immunofluorescence labeling studies. Third, the endothelium of the choroid plexus and the cerebral vessels, especially those surrounding the infarct area, appear to be the two principal pathways for BM-derived cell entry in the brain. Therefore, it may be difficult to distinguish between ECs and cells that are just passing through the vascular wall (Figs. 2d–2f) or macrophages/microglial cells that adhere to the endothelium of the choroid plexus. In this case the number of BM-derived ECs can be overestimated. Fourth, the potential of BM cells to differentiate into all three principal cell types of the CNS might be limited in cerebral ischemia. Although astrocytes, as well as neurons, are derived from the same progenitor cells during embryogenesis, and adult neural stem cells are defined by their ability to produce both cell types (Weiss et al., 1996), we could not identify BM-derived normal neuronal cells or astrocytes (Hess et al., 2002; Priller et al., 2001a). Possibly, cerebral ischemia and consecutive astrogliosis disturb the potential of BM cells to differentiate into astrocytes or oligodendrocytes.
Taken together, our results suggest that therapeutic application of BM in stroke patients might not be an effective treatment to replace lost neurons. However, the therapeutic stimulation of postischemic angiogenesis might be of significant clinical interest. The induction of angiogenesis after cerebral ischemia could be interpreted as a natural defense mechanism that restores oxygen and nutrient supplies (Beck et al., 2000). Recent findings suggest that neovascularization after stroke depends not only on sprouting of preexisting endothelial cells, but also involves a contribution of BM-derived progenitor cells (Hess et al., 2002; Zhang et al., 2002). Although we detected only few GFP-positive ECs at 3 d, 6 weeks, and 6 months after MCAO, it is possible that an integration of these progenitor cells in preexisting or proliferating blood vessels of the infarct border zone is a powerful stimulus that promotes neovascularization in the infarcted brain.
Our data show that almost all recruited cells are positive for microglial markers after brain infarct. Therefore it is conceivable that application of unselected BM after stroke might primarily increase microglial engraftment. Activated microglial cells have been shown to produce neurotoxins, reactive oxygen species, toxic prostanoids, and to contribute to excitotoxicity (Cinelli et al., 2001; Dirnagl et al., 1999; Kim et al., 2000; Tikka et al., 2001). In addition, tempering the microglial response was shown to be beneficial in an experimental model of stroke (Hughes et al., 2002). Thus, an enhanced microglial response could lead to serious undesired effects in patients. However, studies using selected BM populations for cell therapy after stroke showed promising beneficial effects in the functional recovery of the animals (Li et al., 2000, 2002). It will be interesting to test the therapeutic use of selected BM populations, such as the side population (Jackson et al., 2001) or multipotent adult progenitor cells (Jiang et al., 2002), to evaluate whether these strategies might provide an alternative way to induce neovascularization and neuronal tissue repair without stimulating microglial engraftment.
Footnotes
Acknowledgments:
The authors thank Drs. Yoshinori Imai and Shinichi Kohsaka (Tokyo, Japan), who kindly provided the Iba-1 antibody used in this study. They also thank Sonja Thom and Albert Geishauser for their excellent technical assistance.
