Abstract
Bone marrow stromal cells (MSCs) are an excellent source of cells for treating a variety of central nervous system diseases. In this study, we report the efficient induction of committed neural progenitor cells from rat and human MSCs (NS-MSCs) by introduction of cells with the intracellular domain of Notch-1 followed by growth in the free-floating culture system. NS-MSCs successfully formed spheres, in which cells highly expressed the neural precursor cell markers. The commitment of spheres to neural lineage cells was confirmed by their successful differentiation into neuronal cells when exposed to a differentiation medium. To determine the therapeutic potential of NS-MSCs, cells were transplanted into the cortex and striatum in a rat model of focal cerebral ischemia. The survival, distribution, and integration of NS-MSCs in the host brain were very high, and at day 100, grafted NS-MSCs were positive for dopaminergic, glutamatergic, and γ-amino butyric acid (GABA)ergic neuronal markers. They extended long neurites for nearly 6.3 mm and many of these expressed synaptophysin. Significant behavioral recovery was also observed in limb-placing and water-maze tests. These suggest a high potential for this MSC approach in the replenishment of neural cells for stroke and for a wide range of neurodegenerative conditions that require various types of neural cells.
Keywords
Introduction
Stroke is a major cause of death, followed closely by cancer and myocardial infarction, and is characterized by progressive neurologic deficits. Effective treatments for restoring lost neurologic function are currently lacking (Bliss et al, 2007; Lindvall and Kokaia, 2006). Cell transplantation is one potential strategy for the treatment of stroke and other progressive neurodegenerative diseases, such as Parkinson's disease, Alzheimer's disease, and others. Embryonic stem cells (ESCs) and neural stem cells (NSCs) have prompted great interest because of their potential to replenish lost neuronal cells (Reubinoff et al, 2001). Stem and/or neural progenitor cells derived from the umbilical cord blood, amnion/placenta, and adipose tissues are also candidates for cell transplantation therapy (Borlongan et al, 2004; Igura et al, 2004; In ‘t Anker et al, 2003; Lin et al, 2006).
Bone marrow contains mesenchymal stem cells, also known as bone marrow stromal cells (MSCs) that possess the potential to differentiate into other cell types (Prockop, 1997; Tang et al, 2007). MSCs are promising candidates for clinical application because they are easily isolated from patient bone marrow aspirates and are readily expanded in vitro for auto-transplantation without posing major ethical problems (Dezawa et al, 2005). Naive MSCs have been reported by several groups to enhance functional recovery after stroke, with the mechanism of action postulated to be mediated by the trophic effects of MSCs rather than by the spontaneous neuronal differentiation of MSCs in the host brain (Chen et al, 2001a, b ; Chen and Chopp, 2006; Li et al, 2000, 2002; Shen et al, 2007).
In the acute stage of brain injury, neuroprotective treatment may effectively prevent the progressive loss of damaged neuronal cells, but in advanced degenerative stages, neuronal replacement is deemed necessary for cell therapy to exert its benefits. We reported earlier that functional dopaminergic neurons can be specifically induced from human MSCs with a very high efficiency without glial development by the Notch intracellular domain (NICD) transfection followed by the administration of a certain combination of trophic factors, including glial cell line-derived neurotrophic factor (CDNF) (PeproTech, Inc., Rocky Hill, NJ, USA) (Dezawa et al, 2004; Mimura et al, 2005).
In this study, we newly found that rat and human MSCs shift to a neural progenitor-like state, alternatively, committed neural precursor cells, after receiving Notch signal followed by the culture in the free-floating system. Notch intracellular domain-transfected MSCs (NICD-MSCs) successfully formed spheres (NS-MSCs) that highly expressed neural precursor cell markers. The commitment of spheres to neural lineage cells was further evaluated in vitro, which showed that sphere-derived cells successfully differentiated into neuronal cells when exposed to a differentiation medium, and expressed neurotransmitters or related markers, as well as released neurotransmitters. However, in contrast to neurospheres grown from the brain, the bone marrow-derived NS-MSCs showed a low capacity for differentiating into astrocytes. When rat MSCs-derived neural progenitor-like cells were transplanted into a rat focal ischemia model, they showed a much higher efficiency survival, distribution, and integration in the host brain compared with using naive MSCs. They became postmitotic and adapted well to the host microenvironment to differentiate into cells that were positive for dopaminergic, glutamatergic, and γ-amino butyric acid(GABA)ergic neuronal cell markers. Significant behavioral recovery was recognized in the limb-placing and water-maze tests. In addition, tumor formation was not observed up to 100 days after transplantation as judged by histologic analysis.
As MSCs are useful cells in that they can be obtained easily from patients or a bone marrow bank and can be expanded in culture with fewer ethical problems, our finding offers a new efficient in vitro neural progenitor differentiation method to provide a realistic source of neuronal cells for cell therapy.
Materials and methods
Preparation of Marrow Stromal Cells and Neural Induction
The usage of human MSCs for the experiment was approved by the Kyoto University Graduate School and Faculty of Medicine, Ethics Committee. All animal experiments were approved by the Animal Care and Experimentation Committee of the Kyoto University Graduate School of Medicine. Human MSCs were purchased from Cambrex (East Rutherford, NJ, USA). Rat MSCs were harvested according to methods published earlier (Dezawa et al, 2001). Both rat and human MSCs were maintained in α-MEM (α-minimum essential medium) (Sigma, St Louis, MO, USA) containing 10% fetal calf serum (FCS) and kanamycin at 37°C with 5% carbon dioxide (CO2). Rat and human MSCs were then transfected with a vector (pCI neo-NICD) containing the mouse NICD (the NICD cDNA coded for a transmembrane region that included a small fragment of the extracellular domain, followed by a sequence encoding the entire intracellular domain of mouse Notch, initiating at amino acid 1,703 and terminating at the 3'-untranslated sequence). This fragment was subcloned into a pCI-neo vector (Promega Corp., Madison, WI, USA) and was transfected with MSCs using Lipofectamine 2000 (Invitrogen Corp., Carlsbad, CA, USA) and selected by G418 (Invitrogen Corp.) for 7 days, according to the manufacturer's instructions.
Sphere Formation
After G418 selection, both rat and human NICD-transfected cells (referred to as NICD-MSCs) were washed and cultured in α-MEM containing 10% fetal calf serum for 2 days for recovery. After recovery for 2 days, cells were washed and subjected to the free-floating culture for 8 days to generate spheres (referred to as NS-MSCs); i.e., neurobasal medium (Invitrogen Corp.) supplemented with B27 supplement (Invitrogen Corp.), 20 ng/mL bFGF (basic fibroblast growth factor) (R&D Systems Inc., Minneapolis, MN, USA) and 20 ng/mL epidermal growth factor (EGF) (R&D Systems Inc.) at a cell density of 1 × 105 cells per mL on low cell-binding dishes (Nalgene Nunc, Rochester, NY, USA) (Takagi et al, 2005). For transplantation, rat spheres, NS-MSCs, were collected and transplanted into the rat (MCAo) middle cerebral artery occlusion model 8 days after the free-floating culture system.
For the differentiation of rat and human NS-MSCs into neuron-like cells in vitro, spheres were isolated manually and plated onto laminin (BD Sciences, Bedford, MA, USA) coated slides in a neurobasal medium containing the B27 supplement, 1% fetal bovine serum (FBS) by withdrawal of EGF and by the addition of 10 μmol/L Forskolin (Calbiochem, La Jolla, CA, USA), 20 ng/mL CNTF (ciliary neurotrophic factor) (R&D Systems Inc.), and 20 ng/mL bFGF (Dezawa et al, 2004). After 1 week of culture, cells were fixed and evaluated by immunocytochemistry. To examine the ability to form secondary to fourth spheres, rat and human NS-MSCs were dissociated into single cells and cultured in the free-floating culture system for 8 days in each step.
Induction of Permanent Focal Cerebral Ischemia
Permanent focal cerebral ischemia was induced in 10-week-old male Wister rats using the intraluminal filament technique according to the modified procedure of Koizumi et al (1986) and Ohta et al (2006). Details of the procedure are available in Supplementary Information.
Transplantation
Three days after MCAo, under inhalational anesthesia with 2% halothane, the rats were fixed in a stereotaxic frame (Narishige, Tokyo, Japan). During the expansion and recovery of rat NICD-MSCs after G418 selection, and green fluorescent protein (GFP) lentivirus was added to the culture medium for 2 days for the labeling of rat NICD-MSCs as described earlier (Nguyen et al, 2005; Shimizu et al, 2007). After GFP-lentivirus infection, they were subjected to the free-floating culture system to generate rat NS-MSCs, and were injected as spheres into the following places using an electric injector (Muromachi Kikai Co., Tokyo, Japan): 3.5 × 104 cells per 7 μL of rat NS-MSCs into the striatum (from the bregma: anterior (A) 0.0 mm, right (R) + 2.0mm, ventral (V) −4.5mm, incisor bar −3.3 mm) and 1.5 × 104 cells per 3 μL of those (from the bregma: A 0.0mm, R + 2.0mm, V −2.0 mm, incisor bar −3.3 mm) into the cortex along the same tract using a Hamilton microsyringe (Hamilton Company, Reno, NV, USA) fitted with a 26-gauge blunt needle. For the control experiment, naive rat MSCs transplantation (cells which were dissociated into a single cell with trypsin were transplanted in the same manner as described above) and vehicle injection (7 and 3 pL of α-MEM into the striatum and cortex, respectively) were performed. The distribution of animals was NS-MSCs (n = 13), MSCs (n = 9), vehicle (n = 11).
Measurement of Lesion Volume
At 100 days after transplantation, animals were perfused through the heart with cold saline and 4% paraformaldehyde in 0.1 mol/L PBS (phosphate-buffered saline) under deep anesthesia (overdose of pentobarbiturate). The brains were cut coronally at 2-mm intervals. Detailed methods of calculating the lesion volume are described in Supplementary Information.
Retrograde Tracer Labeling
Retrograde tracer labeling with Fluorogold (Molecular Probes, Carlsbad, CA, USA) was performed as described earlier (Hayashi et al, 2006). Briefly, at 7 days before transcardiac perfusion, the rats received an injection of 0.1 μL of hydroxyl stilbamidine (equivalent to Fluorogold) into the substantia nigra (from the bregma: A −5.0 mm, R + 2.3 mm, V −8.25 mm incisor bar −3.3 mm) on the grafted side through a 26-gauge Hamilton microsyringe under general anesthesia.
Antibodies
Primary antibodies used in immunocytochemistry and immunohistochemistry are listed in Table 1. Secondary antibodies used were antirabbit IgG (immunoglobulin G) conjugated either to Alexa Fluor 488 or 568, and antimouse IgG conjugated either to Alexa Fluor 488 or 568 (1:500; Molecular Probes, Carlsbad, CA, USA).
List of primary antibodies
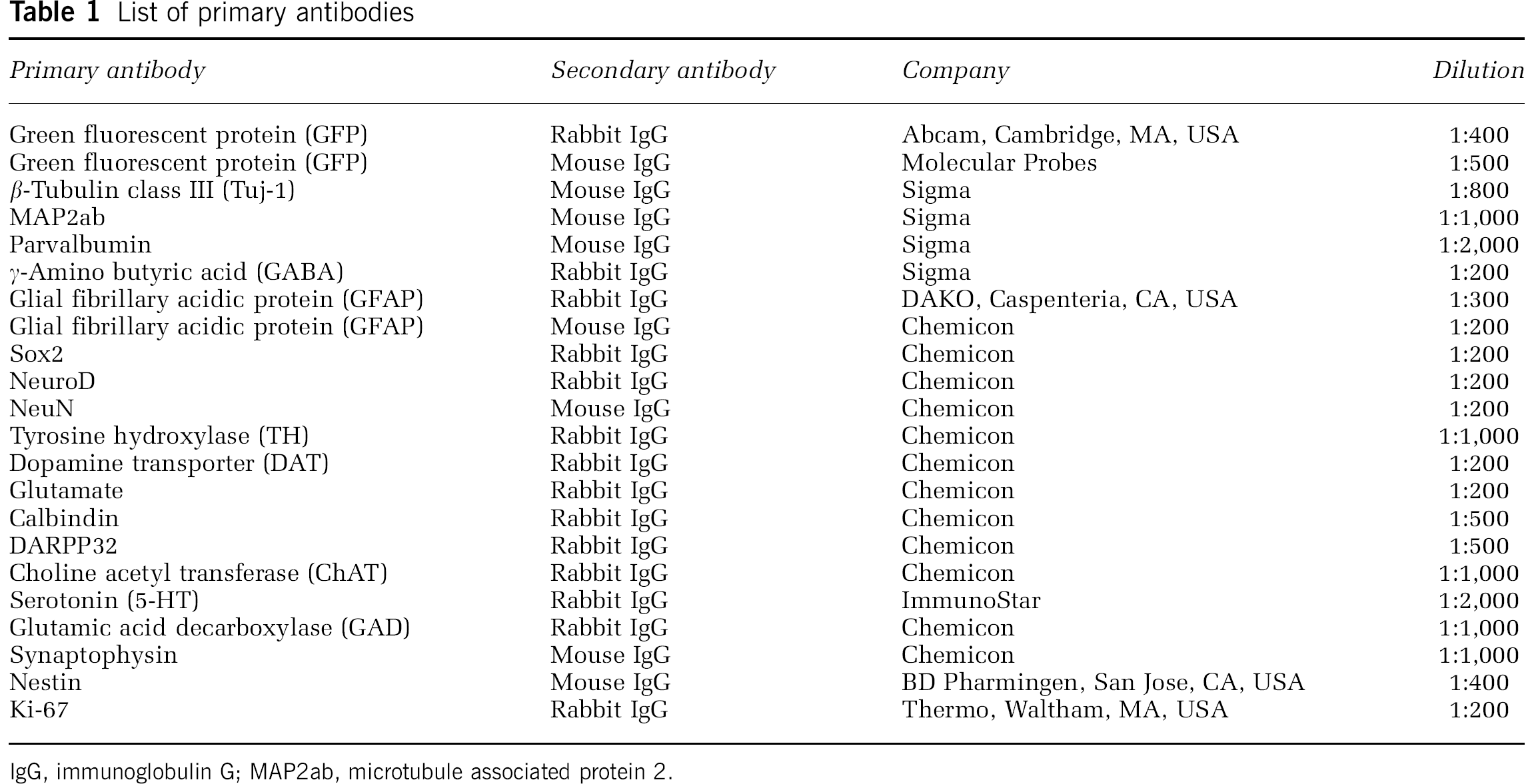
IgG, immunoglobulin G; MAP2ab, microtubule associated protein 2.
Immunocytochemistry
Differentiated cells from rat and human NS-MSCs were fixed with 4% paraformaldehyde in 0.1 mol/L PBS. Spheres derived from rat and human MSCs were fixed with 4% paraformaldehyde, cryoprotected in a series of sucrose solutions (15, 20, and 25% sucrose in 0.02 mol/L PBS) at 4°C for 2 days, and then cut into 10-μm-thick sections using cryostat (Leica CM 1850, Wetzlar, Germany). A detailed procedure of immunocytochemistry is described in Supplementary Information.
The total number of cells was evaluated by counting of 4', 6-diamidino-2-phenylindole- (DAPI)-positive nuclei, and the percentage of immunoreactive cells was evaluated using the Nikon confocal microscope system C1si (Nikon Corporation, Tokyo, Japan). The cells in five fields, which include 100 to 500 cells, were counted for three independent cultures.
Western Blot
pCI-neo-NICD-GFP vector was constructed by the insertion of EGFP gene fragment (Promega Corp.) into the pCI-neo-NICD vector. Six, 12, 24, 36, 48 h and 3 and 5 days after the introduction of pCI-neo-NICD-GFP plasmid by Lipofectamine 2000, cells were harvested and subjected to western blot analysis as described in Supplementary Information. G418 was administrated from 48 h to 5 days after lipofection for the selection of the transfected cells.
Reverse Transcription-PCR
To analyze the relative expression of different mRNAs, the amount of cDNA was normalized on the basis of the signal from ubiquitously expressed β-actin. Primer sequences and precise conditions are β-actin: 5'-AACTGGGACGATATG GAGAA-3' (forward) and 5'-GTAACCCTCATAGATGGG CA-3'(reverse) (TM, melting temperature; 66°C, 25 cycles); neuron-specific enolase (NSE): 5'-GGTGAAGGAAGCCA TTGACAA-3' (forward) and 5'-ATGCCGACATTGGCTGT GA-3' (reverse) (TM, melting temperature; 66°C, 27 cycles); and choline acetyltransferase (ChAT): 5'-GGAGCTAT TCCTCTTTCGGGATT-3' (forward) and 5'-GTCAGTCATG GCTTGCACAAA-3' (reverse) (TM; 66°C, 40 cycles). Total RNA of rat adult brain was used in the reaction of the positive control for each factor.
HPLC (High Performance Liquid Chromatograph)
NS-MSCs cultured in the free-floating culture system for 7 days were replated and cultured in α-MEM with 1% serum, 10 μmol/L Forskolin, 20 ng/mL CNTF, and 20 ng/mL bFGF, followed by the administration of glial cell line-derived neurotrophic factor (50 ng/mL) for 5 days. The concentration of dopamine release was determined by HPLC using a reverse-phase column and an electrochemical detector system (Eicom, Kyoto, Japan) as described earlier (Dezawa et al, 2004). Details of the procedure are available in Supplementary Information.
Behavioral Analysis
The limb-placing test was a modified version of a test described by De Ryck et al (1989) and Jeong et al (2003). The test was performed the day before ischemia induction and after transplantation on days 4, 10, 14, 21, 28, 56 and 84. The total score ranged from 0 to 7.
The Morris water-maze test was performed as described earlier (Mimura et al, 2005; Morris, 1984). This test was performed from day 96 to day 100 after transplantation. Each detailed procedure is described in Supplementary Information. After behavioral follow-up, at 100 days after transplantation, each animal underwent perfusion fixation under anesthesia. In all, 10-μm-thick brain cryosections were prepared by cryostat. As for the positive control, normal rats (n = 8) were used.
Immunohistochemistry
In 14 and 100 days after transplantation, each animal was perfused through the heart with cold saline and 4% paraformaldehyde in 0.1 mol/L PBS under deep anesthesia (overdose of pentobarbiturate). The brains were cut coronally at 2-mm intervals, and 6 brain slices were prepared totally. Each brain slice was cut into 10-μm-thick coronal sections using a cryostat, and one cryosection was chosen from each slice. Transplanted cells of 10 fields in each cryosection were counted using the Nikon confocal microscope system C1si for three animals, and the cell number of positive cells in serial sections was calculated. Cells of thickness larger than 10-μm could have been counted multiple times. For this, the size and thickness of cells were corrected using the Abercrombie method for controlling the bias in counting (Abercrombie, 1946).
Details of the immunostaining procedure are described in Supplementary Information. The samples were inspected under Nikon confocal microscope system C1si.
The numbers of animals subjected to experiments were NS-MSCs = 13, MSCs = 9, vehicle = 11 (for 100 days after transplantation), and NS-MSCs = 8, MSCs = 8 (for 14 days after transplantation).
Real-Time PCR
Total RNA was collected using the RNeasy Mini Kit (Qiagen GmbH, Hilden, Germany), and cDNA was synthesized using High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). Primers and probes for NSE (γ, γ-enolase), sodium channel voltage-gated type III Scn3a, and β-actin were purchased from TaqMan Gene Expression Assay (Applied Biosystems) and subjected to real-time PCR according to the manufacturer's instructions.
To estimate the number of transplanted cells that survived and integrated in the host tissue 100 days after grafting, real-time PCR was performed using a 7300 sequence detection system (Applied Biosystems). Realtime PCR primers and the probe for GFP used in this study were 5'-AGTCCGCCCTGAGCAAAGA-3' and 5'-TCCAGC AGGACCATGTGATC-3', and 5'-FAM-CCCAACGAGAAG CG-MGB-3', respectively. Standard curves were generated by serially diluting cDNA derived from cultured MSCs. Values were normalized by assays for β-actin using TaqMan β-actin Detection Reagents (Applied Biosystems).
Statistical Analysis
Data are expressed as mean ± s.e.m. Data were compared using ANOVA (analysis of variance) with pairwise comparisons by the Bonferroni method. As the score of the limb-placing test is ordinal (i.e., 8 levels (0 to 7)), the Mann—Whitney U-test was chosen for the nonparametric statistical analysis of the limb-placing test.
Results
Generation of Spheres from Marrow Stromal Cells
After NICD transfection into MSCs (NICD-MSCs), morphologic changes were observed, i.e., the cells became smaller (Figures 1A and 1B). These NICD-MSCs were subjected to the free-floating culture system (neurobasal medium supplemented with B27, bFGF, and EGF for 8 days) to form spheres (NS-MSCs, Figure 1C). Naive MSCs formed spheres but the efficiency was very low compared with that of both rat and human NICD-MSCs; in rats, NICD-MSCs produced seven times more spheres (~35 spheres per 10,000 cells) than did naive MSCs (~5 spheres per 10,000 cells), and in humans, NICD-MSCs produced ~50 spheres per 10,000 cells in all clones examined, whereas naive MSCs produced 0 to 4 spheres per 10,000 cells. The diameter of the NS-MSCs spheres was ~60 to 150 μm, whereas that of MSCs was mostly under 60 μm. Rat and human NS-MSCs were dissociated and subjected to secondary, third, and fourth sphere formation. Eight days after replating, secondary spheres could be recognized but few third and no fourth generation were observed (data not shown). In this regard, the sphere formation of NS-MSCs is limited.
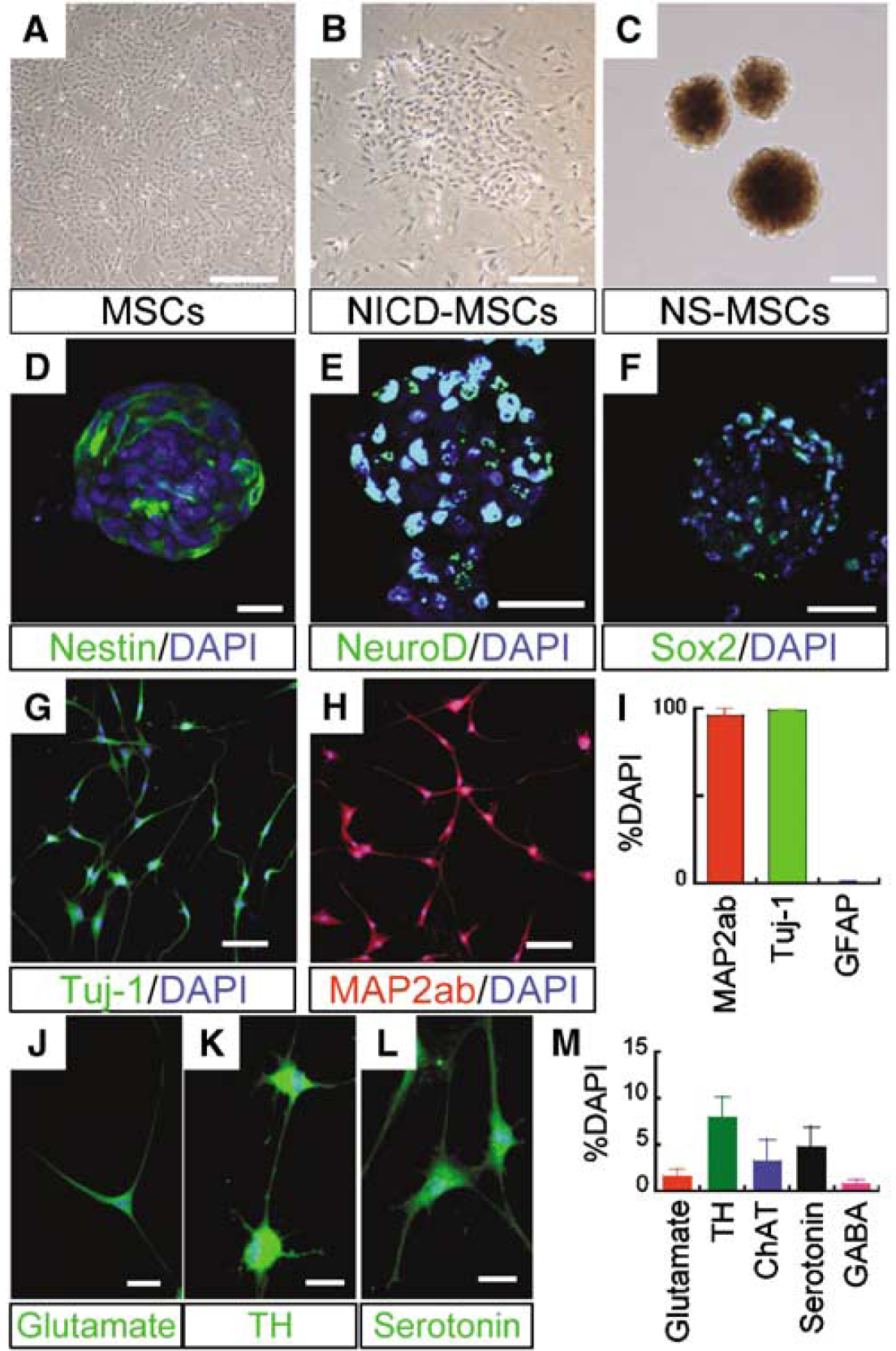
Neuronal induction from marrow stromal cells (MSCs). (
To determine whether the spheres contained cells with neural progenitor properties, we examined the expression of Sox2, nestin, and NeuroD. In naive rat MSCs, a few cells were positive for Sox2 (2.0 ± 1.4%), nestin (2.3 ± 0.9%), and NeuroD (9.4 ± 2.2%). In contrast, induced spheres contained much higher percentages of cells positive for nestin (61 ± 4.8%), NeuroD (96.8 ± 0.9%), and Sox2 (93.9 ± 2.4%) (Figures 1D–1F). Almost identical data were obtained from human MSCs.
To examine whether cells in the spheres were committed to a neural lineage cell, spheres were dissociated, replated to adherent dishes, and then differentiated in vitro by low serum and withdrawal of EGF. Adhered rat and human cells had neurite-like processes with abundant varicosities and were immunoreactive to markers of neuronal cells, β-tubulin isotype III (Tuj-1) (98.5 ± 0.5%) and microtubule associated protein 2 (MAP-2) (95.7 ± 3.7%) (Figures 1G–1I). A very small number of cells were immunopositive for the astrocyte marker, glial fibrillary acidic protein (0.7 ± 0.3%). The expression of transmitter-related markers in rat cells was 1.6 ± 0.7% (glutamate), 7.9 ± 2.2% (TH (tyrosine hydroxylase)), 3.2 ± 2.3% (ChAT), 4.7 ± 2.3% (serotonin), and 0.7 ± 0.4% (GABA) (Figures 1J–1M), and that in human cells was 8.2 ± 5.7% (glutamate), 8.9 ± 4.1% (TH), 4.7 ± 2.4% (ChAT), 3.8 ± 2.9% (serotonin), and 3.8 ± 3.7% (GABA), suggesting their mature neuronal subtypes. We also confirmed their expression of neuronal markers in RT-PCR and real time-PCR. The clear upregulation of NSE after neuronal induction from NS-MSCs was recognized in RT-PCR (Supplementary Figure 1). They expressed not only NSE but also ChAT, one of the markers for functional neurons. In real-time PCR, both NSE and voltage-gated sodium channel type III Scn3a were shown to be substantially upregulated or expressed in neuronal cells induced from NS-MSCs but not in naive MSCs (Supplementary Figure 2). In the HPLC measurement, neuronal cells induced from human and rat NS-MSCs showed 1.0 and 0.5 pmol/L per 106 cells of dopamine release into the culture media in response to high potassium depolarizing stimuli, respectively, whereas dopamine release from naive human and rat MSCs was under the detection level.
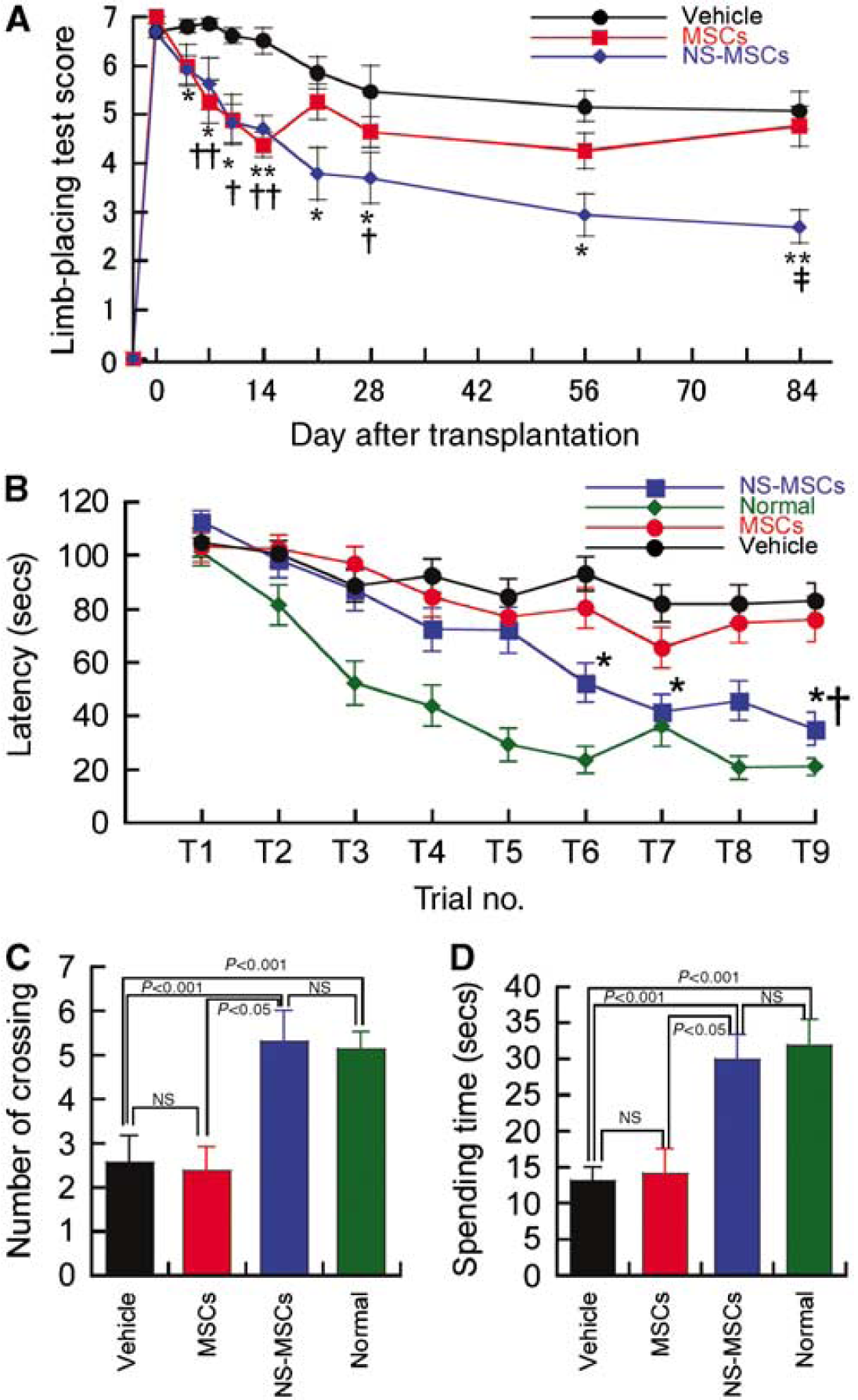
Behavioral analysis. (
As the introduction of an exogenous gene is an extra hurdle for the safety aspect in the cell transplantation therapy, we tracked the expression of the exogenous NICD gene after transfection with pCI-neo-NICD-GFP genes at protein level by western blot in both rat and human MSCs. The NICD-GFP expression was detectable from 12 h after introduction and reached to the maximum expression from 24 to 48 h, and returned to the basal level by 5 days (data not shown), suggesting that the introduced exogenous NICD does not remain within rat and human MSCs for a longer period.
Transplantation of NS-MSCs into Focal Cerebral Ischemic Lesions
To evaluate whether NS-MSCs were able to integrate into the host brain and contribute to the amelioration of the neurodegeneration, we transplanted the rat NS-MSCs into a rat model of permanent focal cerebral ischemia. All rats exposed to focal cerebral ischemia had areas of severe corticostriatal infarction. Each animal received 50,000 cells, which were transplanted into the striatum and cortex after 3 days of MCAo. For control groups, the same number of naive MSCs (MSCs) or vehicle was injected.
The tissue defect observed in each experimental group was defined as the lesion. The mean lesion volumes in the NS-MSCs, MSCs, and vehicle groups on day 100 were 27.1 ± 4.3%, 23.0 ± 2.3%, and 31.4 ± 5.0%, respectively. There was no statistical difference among groups.
Limb-Placing Test
Rats with focal cerebral ischemia initially showed severe impairment in the limb-placing test (Figure 2A). From days 4 to 84 after transplantation, the mean score of the NS-MSCs group was significantly improved compared with that of the vehicle group, indicating that the NS-MSCs group significantly induced long-term behavioral recovery. The mean score of the NS-MSCs group was also significantly improved compared with that of the MSCs group at day 84. In contrast, the MSCs group showed significantly improved performance compared with the vehicle group only on days 10 to 14 and 28. At days 21 and 56 to 84, there was no significant difference between the MSCs and vehicle groups.
Water-Maze Test
The mean latency time recorded for NS-MSCs, MSCs, vehicle, and normal groups is presented in Figure 2B. The NS-MSCs group had a significantly shorter latency time to reach the escape platform than the MSCs and vehicle groups. In the spatial probe test, the number of platform location crossings and the amount of time spent in the target quadrant were significantly longer in the NS-MSCs group compared with that in the MSCs and vehicle groups (Figures 2C and 2D) again suggesting a functional effect of the NS-MSC grafted cells. There was no statistical difference between NS-MSCs and normal rats.
Histologic Analysis
Cells were labeled with GFP lentivirus before transplantation. Tumor formation, a massive occupying effect in the brain tissue as in the case of embryonic stem cell transplantation (Takagi et al, 2005), could not be observed in the grafted brain during the 100-day follow-up in all groups, including NS-MSC grafted rats. Green fluorescent protein-positive NS-MSCs were widely observed at the lesion boundary, ipsilateral cortex, and striatum but most intensively around the lesion site (Figure 3A). Their migration was confined to the ipsilateral site, and GFP-positive NS-MSCs were not detected in the contralateral side. In the NS-MSCs group at day 100, 21 ± 1.3 × 104 GFP-labeled cells survived. On the basis of the initial number of transplanted cells (5 × 104), the NS-MSCs appeared to have proliferated after transplantation. Among the GFP-labeled cells, 4.7 ± 0.02% were immunoreactive for Ki67 at day 14, but Ki67-positive cells were not observed at 100 days, suggesting that the cells did not continue to divide. In the MSCs group, in contrast, there were 1.3 ± 0.46 × 104 GFP-positive cells at day 100 and these were mostly detected around the injected area, and rarely observed to be migrating through the parenchyma in the cortex or the striatum (Figure 3A). Furthermore, a successful neuronal differentiation was not observed in the MSCs group. To further confirm the difference in GFP-positive cell numbers between the NS-MSCs and MSCs groups, we performed real-time PCR for the GFP gene. At day 100, the NS-MSCs-transplanted brain showed a 19.6 ± 6.9 times greater GFP gene content compared with the MSCs-transplanted brain.
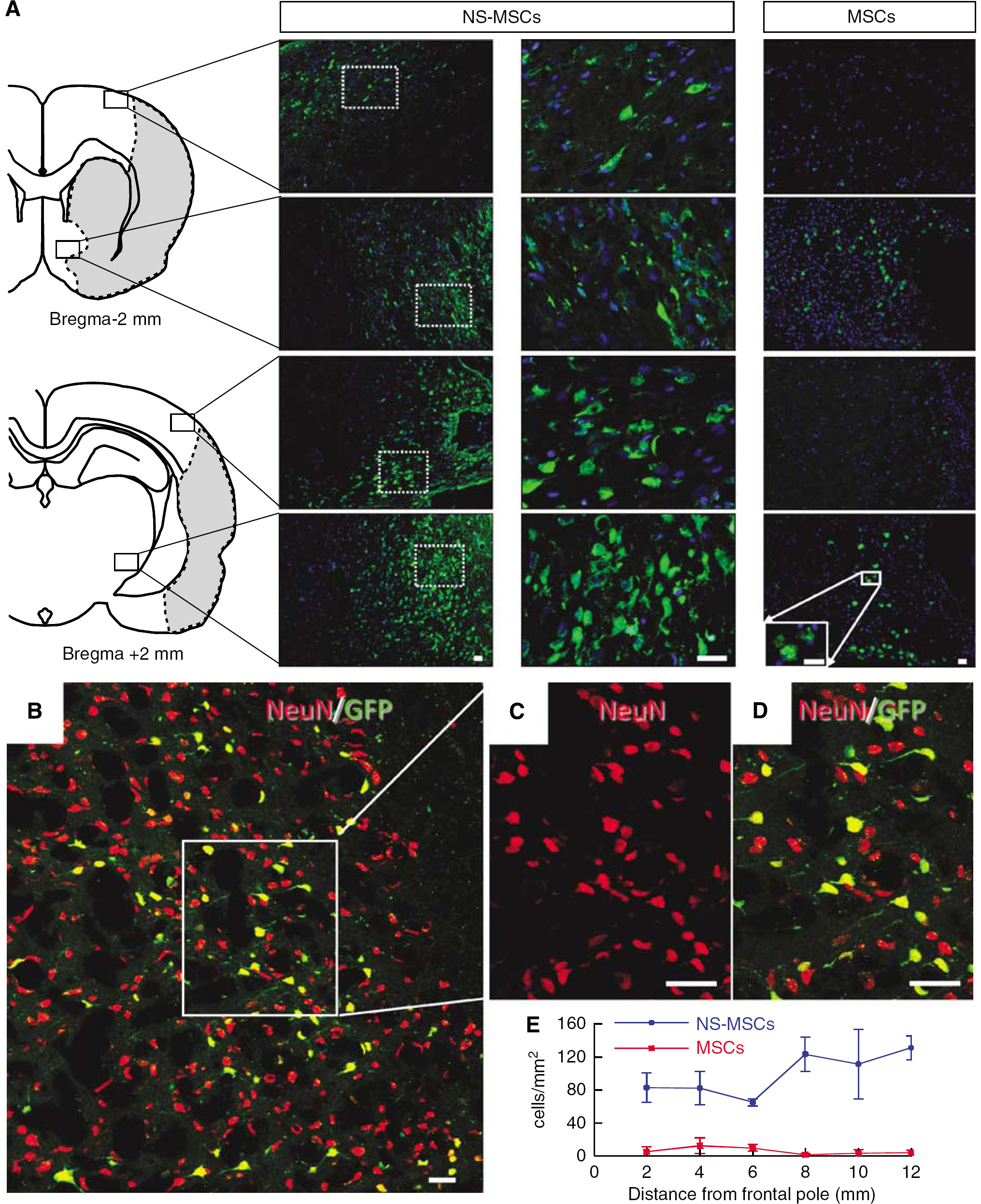
Transplanted cells in the brain lesion (day 100). (
To determine whether transplanted cells expressed neural markers, sections were immunostained with NeuN and glial fibrillary acidic protein antibodies. In the NS-MSCs group, the percentages of GFP-positive cells that were NeuN- and glial fibrillary acidic protein-positive were 79.5 ± 0.1% (Figures 3B–3D) and 1.9 ± 0.03% (data not shown), respectively. In the MSCs group, neither NeuN- nor glial fibrillary acidic protein-positive cells were detected (data not shown). The number of GFP-positive cells in the NS-MSCs and MSCs groups in each section was counted from the anterior to the posterior of the brain. Remarkably, NS-MSCs were observed widely in the antero-posterior axis throughout the brain, in addition to the transplanted site, whereas only a small number of transplanted MSCs were identified, and these were located only around the lesion.
The expression of differentiation markers and neurotransmitter-related markers in NS-MSCs was then examined. The GFP-positive cells were also immunoreactive to dopamine transporter (32.8 ± 11.4%), TH (41.4 ± 17.6%), glutamic acid decarboxylase (10.2 ± 5.7%), glutamate (4.1 ± 0.4%), calbindin (7.7 ± 2.5%), DARPP32 (13.1 ± 12.5%), and parvalbumin (18.9 ± 9.9%) (Figure 4).
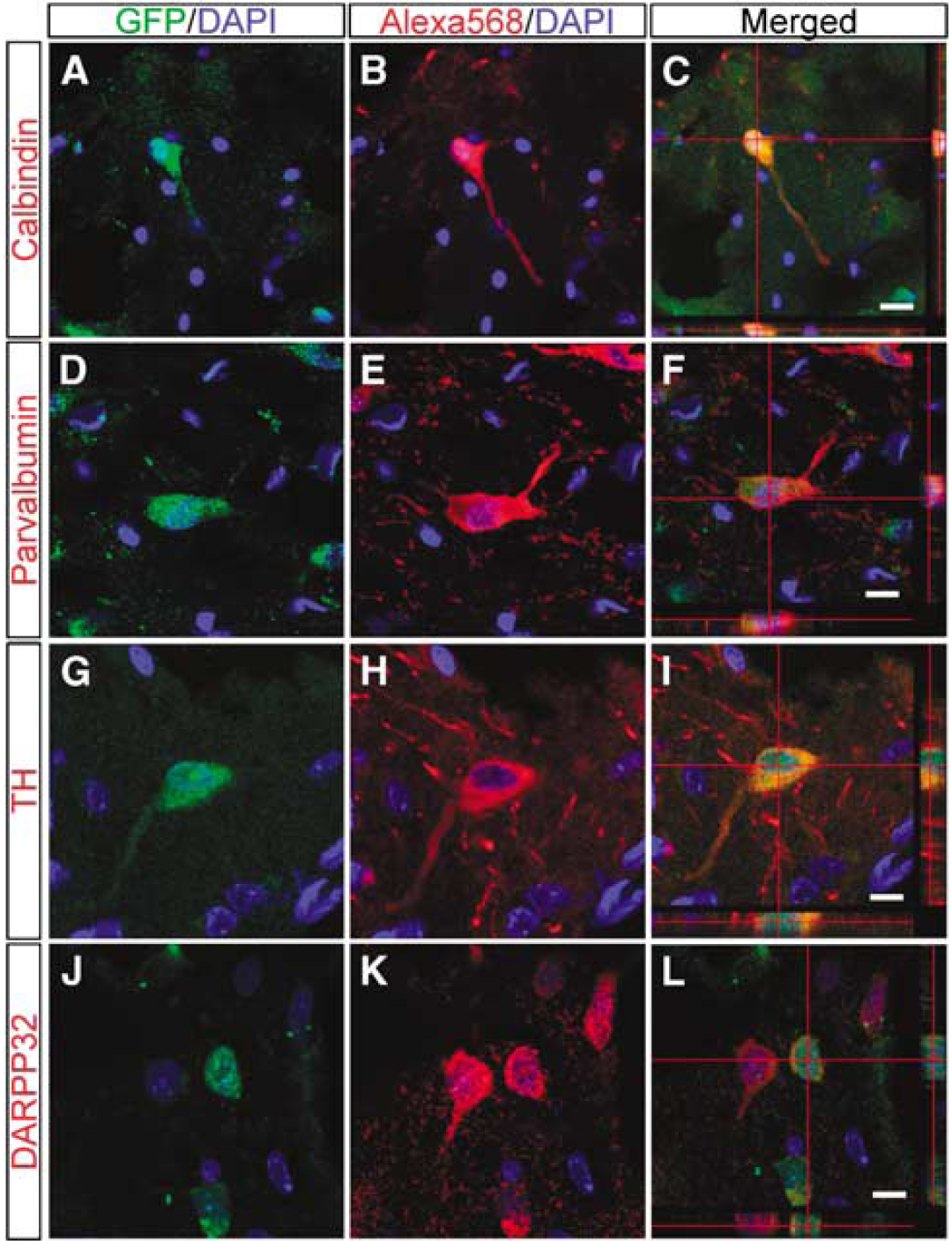
Immunohistochemistry of green fluorescent protein (GFP)-positive transplanted NS-MSCs (day 100). (
Fluorogold Tracing and Synaptophysin Staining
To assess the possibility that the transplanted NS-MSCs extended neurites with active transport capacity in the host brain, we injected a retrograde tracer at 93 days after transplantation. Fluorogold was injected into the substantia nigra, and then the grafted area was subjected to Fluorogold detection at 100 days. Of the GFP-positive cells, 35.2 ± 7.3% of the cells were Fluorogold-positive and had a neuron-like morphology (Figures 5A–5F). We also investigated the expression of synaptophysin, and 15.78 ± 2.94% of GFP-positive cells showed synaptophysin positivity in the striatum (Figures 5G–5J). These data suggest that grafted cells extended long processes from the striatum to the substantia nigra and some of these expressed the synaptic marker synaptophysin.
Discussion
In this study, we report that committed neural progenitor cells, NS-MSCs, can be efficiently differentiated from rat and human MSCs by NICD introduction followed by the free-floating culture system that promotes the formation of spheres. Cells in these spheres expressed markers related to neural progenitor cells, and were shown to be committed to neural lineage cells by immunocytochemistry and grafting into the brain. NS-MSCs also differentiated into neuronal cells after transplantation into a rat focal cerebral ischemia model, and were shown to be immunoreactive to various neurotransmitter-related markers within the host tissue, suggesting their potential to differentiate into various types of neuronal cells. Their contribution to functional recovery was also observed for up to 100 days after transplantation. Importantly, no tumors were detected. On the basis of these results, NS-MSCs may be a good source of neuronal lineage cells for cell transplantation therapy in stroke.
Accumulating evidence shows that naive MSCs transplantation into the ischemic brain leads to improved behavior (Chen and Chopp, 2006; Li et al, 2002). According to these reports, the survival rate of MSCs in the host brain was under 10%, and the proportion showing neuronal differentiation was 1% to 2%. The functional recovery induced by these naive MSCs transplantation may partly be mediated by the trophic effects of MSCs or by the intrinsic parenchymal cells stimulated by MSCs (Chen and Chopp, 2006; Li et al, 2002). However, in this study, a large number of GFP-labeled NS-MSCs was measured in the host brain even after 100 days of transplantation, and most of the cells expressed neuronal markers and neurotransmitter-related markers. In addition, Fluorogold tracing showed that the transplanted cells extended neurites for some distance in the host brain and might have formed synapses with host neurons. The true contribution of the neurite extension of transplanted NS-MSCs remains unclear; although our results suggest that the differentiation or orientation of MSCs into cells with neural properties before transplantation is effective for the survival and integration of MSCs.
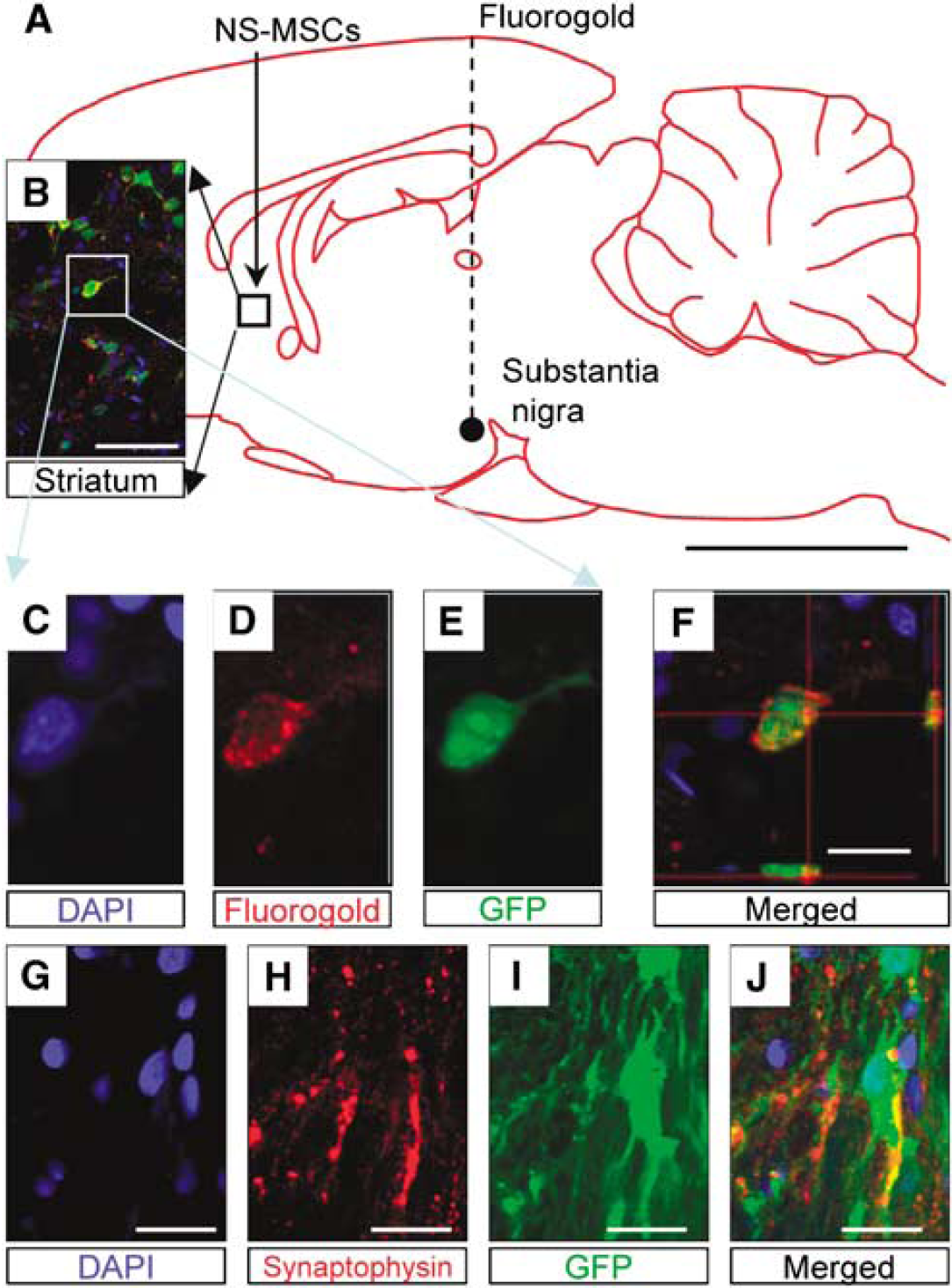
The expression of retrograde tracer Fluorogold injected in the targeted area of the substantia nigra and synaptophysin expression in the graft (day 100). (
In our result, recovery occurs long before the cells can have integrated into the host brain. In the limb-placing test, the MSC group showed slight recovery in the earlier period after operation, and the mean lesion volumes between the NS-MSCs and MSCs groups showed no statistical difference. Perhaps, the recovery in the earlier period (before 14 days) resulted from the trophic effect of transplanted cells rather than from the replenishment of lost cells. As mentioned above, naive MSCs are known to have trophic effects and therefore, it is no wonder that NS-MSCs also procure the same effect. However, in the later period, MSCs do not survive, whereas NS-MSCs do, which are migrated and differentiated in the host tissue. Although the cell replacement by transplanted NS-MSCs cannot be the entire contribution for the functional recovery, this difference may explain the functional recovery shown in NS-MSCs groups at a later period of 100 days.
Under stressful conditions, MSCs are shown to exhibit neuron-like morphology and to express neuronal markers, but without ever having undergone protein synthesis, indicating that the neuron-like morphologic and immunocytochemical changes are artifacts (Lu et al, 2004). Indeed, in their report, NeuN and NSE were positive to these cells in immunocytochemistry, whereas upregulation of these markers could not be recognized in RT-PCR. In contrast, we confirmed the clear upregulation of NSE after neuronal induction from NS-MSCs, and expression of ChAT, one of the markers for functional neurons, in RT-PCR (Supplementary Figure 1). In real-time PCR, both NSE and voltage-gated sodium channel type III Scn3a were shown to be expressed substantially in neuronal cells induced from NS-MSCs but not in naive MSCs (Supplementary Figure 2). High performance liquid chromatograph also showed the effective production of dopamine from neuronal cells induced from NS-MSCs. These results collectively suggest that, different from stress-induced neuron-like cells, neuronal cells differentiated from NS-MSCs are functional as the upregulation of markers related to functional neurons are shown as described above.
Compared with fully differentiated postmitotic neuronal cells, NS-MSCs have several advantages. First, because of the premature state of NS-MSCs, they have a greater survival rate, distribution, and integration in the host brain. For differentiated dopaminergic neurons induced from MSCs (Dezawa et al, 2004; Mimura et al, 2005), the survival ratio was only 20% to 30% after transplantation into the Parkinson's disease model rat and their distribution was mostly confined to the striatum, suggesting their limited ability for migration and integration in the host tissue. However, NS-MSCs were widely observed in the lesion boundary, ipsilateral cortex, and striatum in the stroke brain and 21 ± 1.3 × 104 cells were counted after the transplantation of 50,000 cells. As 4.7% of GFP-positive NS-MSCs were positive for Ki67 at 14 days, but GFP-positive cells were negative for Ki67 at 100 days, a subpopulation of NS-MSCs might have proliferated at an earlier period, but most of them became postmitotic by day 100. These observations might partly explain the following: (1) the ability of NS-MSCs to proliferate and migrate because of their premature state and (2) although NS-MSCs show neural lineage commitment in vitro, their differentiation into functional neuronal subtypes in the host tissue, such as dopaminergic, glutamatergic, and GABAergic marker-positive cells might subsequently be regulated by the host microenvironment.
Second, NS-MSCs differentiated into various kinds of transmitter-related marker positive cells within the host brain. They also extended neurites for a long distance for ~6.3 mm. This suggests that premature NS-MSCs have higher flexibility to adapt to the host microenvironment and to differentiate into various cell types rather than fully differentiated neuronal cells. Such properties would be beneficial when replenishing neural cells are required to cover a large degenerated region and various types of neural cells are required, such as in the case of stroke. Conversely, in Parkinson's disease, as dopamine-producing cells are the main target, induction and differentiation into dopaminergic neurons before the transplantation would be preferable for functional recovery (Dezawa et al, 2004; Mimura et al, 2005).
Third, NS-MSCs are more easily prepared than fully differentiated neuronal cells, which might be beneficial from a practical aspect. Precise control and maintenance are required to achieve the differentiation of neuronal cells from human MSCs, appropriate density in the case of seeding and pertinent harvesting method. As for the NS-MSCs method, this free-floating culture method is not highly dependent on cell density and does not require harvesting. Moreover, in general, mature neurons are vulnerable compared with neural progenitors for their maintenance. Considering these three aspects, the NS-MSCs method will clinically be feasible for treating acute injuries like stroke.
Earlier reports have described the induction of neurosphere-like cells from MSCs using trophic factors and/or the medium used for neural stem cells (Hermann et al, 2004; Lee et al, 2003). Notably, in these reports, the ratio of glial cell differentiation is higher than that of neuronal cells; induction rates of mature neurons versus astrocytes derived from neurosphere-like cells are 6 ± 2%:13 ± 4% (Hermann et al, 2004) or 6.5%:32.9% (Lee et al, 2003). In our system, almost all of the NS-MSCs differentiated into neuronal cells and a very small number of glial cells were produced, both in vitro and in vivo. This finding suggests that NICD introduction and sphere formation strongly shifted the MSCs to a neuronal potential.
The number of GFP-labeled transplanted cells at 100 days after transplantation was four times higher than that at 14 days. Therefore, we conducted the study on Ki67 staining, a marker for cell proliferation related to tumor genesis, which showed that Ki67-positive cells were detected in 14 but not in 100 days. Furthermore, the percentage of GFP-positive cells that expressed NeuN at 100 days was very high (79.5 ± 0.1%). Some of them might get disposed from the organism after transplantation, but overall, this result suggests that large number of Ki67-positive cells at 14 days did not continue to divide and differentiated into neuronal phenotype by 100 days. Consistently, the ability to generate spheres was limited in NS-MSCs, i.e., NS-MSCs could generate second and third spheres but were unable to show fourth sphere formation. In addition, tumor formation was not detected up to 100 days. Many reports suggested that MSCs are less tumorigenic than fetus-derived stem cells or embryonic stem cells, and few reports with regard to MSC transplantation into stroke model animals reported ectopic tissue formation or carcinogenesis (Chen et al, 2001a, b ). Indeed, these earlier reports do not completely cast aside the tumorigenic possibility of MSC or MSC-derived cells. The most reliable safety evaluation will not be performed in the rat experiment but in the higher mammals like monkeys; therefore, we recognize that evaluation in the monkey experiment is necessary in the future.
Notch signaling inhibits neuronal differentiation and promotes glial differentiation during development (Lundkvist and Lendahl, 2001). Although our results seem inconsistent with the well-known action of Notch signaling, it is presumed that cell susceptibility to Notch signaling in MSCs is different from cells in the process of neuronal development. Our results suggest distinct cellular responses to Notch signals; e.g., the protein repertoire and active factors may be quite different between conventional neural progenitor cells and MSCs. We recognized in this experiment that NICD transfection leads to an upregulation of NeuroD in the luciferase assay as described earlier (Dezawa et al, 2004), which suggests that, at least in MSCs, cells are orientated to a neural differentiation by NICD introduction. Furthermore, treatment with the free-floating culture system might have selected cells with a high potential for neural differentiation. The precise mechanism underlying the MSCs acquisition of neural progenitor cell properties in this system needs to be clarified.
Several reports indicated that remote degeneration of neurons together with the change in Bcl-2 and tumor necrosis factor-α expression level occurs in the substantia nigra after focal ischemia (Arango-Davila et al, 2004; Loos et al, 2003). These observations suggested that the nigrostriatal pathway was damaged after focal ischemia. Earlier reports showed the improvement of behavioral dysfunction after striatal transplantation of embryonic stem cells, which was assessed by methamphetamine injection, the test known to estimate the nigrostriatal pathway (Yanagisawa et al, 2006). The result indicated that striatal transplantation could repair the damage of the nigrostriatal pathway. Our result also showed the same effect of striatal transplantation.
Furthermore, several earlier studies indicated that hemispheric damage influenced the result of water-maze test (Puurunen et al, 2001; Veizovic et al, 2001). In addition, Yonemori et al (Yonemori et al, 1999) reported that focal cerebral ischemia caused spatial memory disturbance in rats. Therefore, in our experiment, the improvement of this test might have been brought about by the improvement of both cognitive and motor functions.
Human MSCs have a high proliferation ability; 20 to 100 mL of bone marrow aspirate provides 1 × 107 MSCs within several weeks. Considering the sphere formation efficiency described above, as many as 5 × 104 spheres may be obtained from a patient's bone marrow within a reasonable time period. We also confirmed that cryopreserved NS-MSCs could proliferate and repeatedly form spheres while maintaining their neural progenitor-like characteristics (data not shown). Therefore, not only for autologous transplantation, but also for a cell-providing system using the same human leukocyte antigen subtype, MSCs from a healthy donor might be a realistic approach for cell therapy. This MSC cell therapy approach may be applicable for stroke victims and for those of other neurodegenerative diseases.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
