Abstract
Recent reports have shown that bone marrow-derived cells (BMDCs) contribute to the formation of vasculature after stroke. However, the mechanism by which mural cells are formed from BMDC remains elusive. Here, we provide direct evidence that the cell fusion process contributes to the formation of pericytes after stroke. We generated mouse bone marrow chimeras using a cre/lox system that allows the detection of fusion events by X-gal staining. In these mice, we detected X-gal-positive cells that expressed vimentin and desmin, specific markers of mature murine pericytes. Electron microscopy confirmed that fused cells possessed basal lamina and characteristics of pericytes. Furthermore, induction of stroke increased significantly the presence of fused cells in the ischemic area. These cells expressed markers of developing pericytes such as NG2. We conclude that cell fusion participates actively in the generation of vascular tissue through pericyte formation under normal as well as pathologic conditions.
Introduction
Adult bone marrow-derived stem cells (BMDCs) represent a promising therapeutic agent for the treatment of damaged ischemic tissue (Tang et al, 2007). Numerous studies have reported significant functional improvements of stroke symptoms after BMDC transplant (Tang et al, 2007). In the past years, the exact mechanism by which these cells elicit recovery (e.g., secretion of trophic/growth factors, cell fusion, or transdifferentiation) has been extensively investigated. Thus, it has been reported that BMDC secrete cytokines and growth factors that promote angiogenic and neurorestorative processes (Chen and Chopp, 2006). Moreover, several studies have shown the contribution of BMDC or hematopoietic stem cells to the formation of vasculature after stroke, including the generation of endothelial cells (ECs; Bailey et al, 2006) and mural cells (Kokovay et al, 2006; Rajantie et al, 2004). Some of them have shown definitively that EC are generated by transdifferentiation and not by cell fusion (Bailey et al, 2006). However, the mechanism by which mural cells are formed remains elusive. Therefore, we postulated that cell fusion might be implicated in the generation of perivascular cells under normal conditions and after stroke. To test this hypothesis, we performed BMDC transplants with cells constitutively expressing Cre from a ubiquitous actin-driven promoter into neonatal recipient mice of the reporter strain R26R, in which LacZ is only expressed after excision of a loxP-flanked stop cassette by Cre-mediated recombination. This system allows us to detect fusion events (Alvarez-Dolado et al, 2003) before and after stroke induction.
Materials and methods
Animals and Bone Marrow Transplants
Animal procedures were performed in accordance with the Spanish legislation (RD 1201/05) and under the guidelines of the CIPF animal care committee. Neonatal chimeras were generated as described previously (Yoder et al, 1996). Briefly, busulfan (20 mg/mL in PBS containing 1% dimethyl sulfoxide; Sigma, Madrid, Spain, B2635) was injected i.p. at a dose of 15.5 mg/kg to pregnant R26R dams at 17.5 and 18.5 gestational days, for sublethal conditioning of the future pups. The newborn busulfan-conditioned R26R mice were transplanted, via maxillofacial vein, with 50 μL of a BMDC suspension (2 × 105 cells/μL) from 8- to 10-week-old mice expressing Cre recombinase under the control of the hybrid regulatory element cytomegalovirus enhancer/β-actin promoter. For further explanation of the cell fusion detection system by X-gal using these mouse lines see Supplementary information.
Unilateral Cerebral Hypoxia—Ischemia
At 2 months after BMDC transplant, we induced a hypoxia—ischemia episode by occlusion and posterior reperfusion of carotid artery (Qi et al, 2004). For detailed explanation of the procedure see the Supplementary Information.
Histology, Electron Microscopy, and Quantifications
Bone marrow-derived cell-transplanted mice (2 months old), and ischemic BMDC-transplanted mice (2.5 months old, 2 weeks after stroke) with the correspondent sham-operated controls (2.5 months old) were anesthetized and transcardially perfused with 40 mL of 2% paraformaldehyde plus 0.25% glutaraldehyde. Brains were dissected and serially cut in 12 series of 50-μm vibrotome sections. For detailed description of X-gal staining, electron microscopy, and immunohistochemistry see the Supplementary information. Cell counting was performed in 10 brain sections, serially separated each one 600 μm, what corresponds to approximately one-twelfth of the whole forebrain. The samples were analyzed with a DM6000B microscope (Leica), using 10, 20, and 63X Plan-Neofluar objectives. Digitized images were obtained with a DFC480 digital camera and IM500/FW4000 image manager software (Leica).
Results
To study the possible contribution of the cell fusion process to vasculogenesis, we analyzed the brains of chimeric R26R mice reconstituted with BMDC-expressing Cre under the control of β-actin promoter. This transplant strategy allows the detection of fused cells by X-gal staining (see Materials and methods section) (Alvarez-Dolado et al, 2003). At 2 months after the BMDC transplant, we observed X-gal-positive cells associated with the vasculature in the brains of chimeric mice (Figure 1A). Their frequency was 251±76 cells/brain (n=5), distributed uniformly throughout the areas of the brain. Interestingly, 13.3±8.4% of these cells contained two nuclei (Figures 1C and 1D), further supporting their formation by cell fusion. The presence of single nucleated X-gal-positive cells can be explained by the later formation of synkarions, or the generation of new progeny after reductive mitosis (Alvarez-Dolado, 2007).
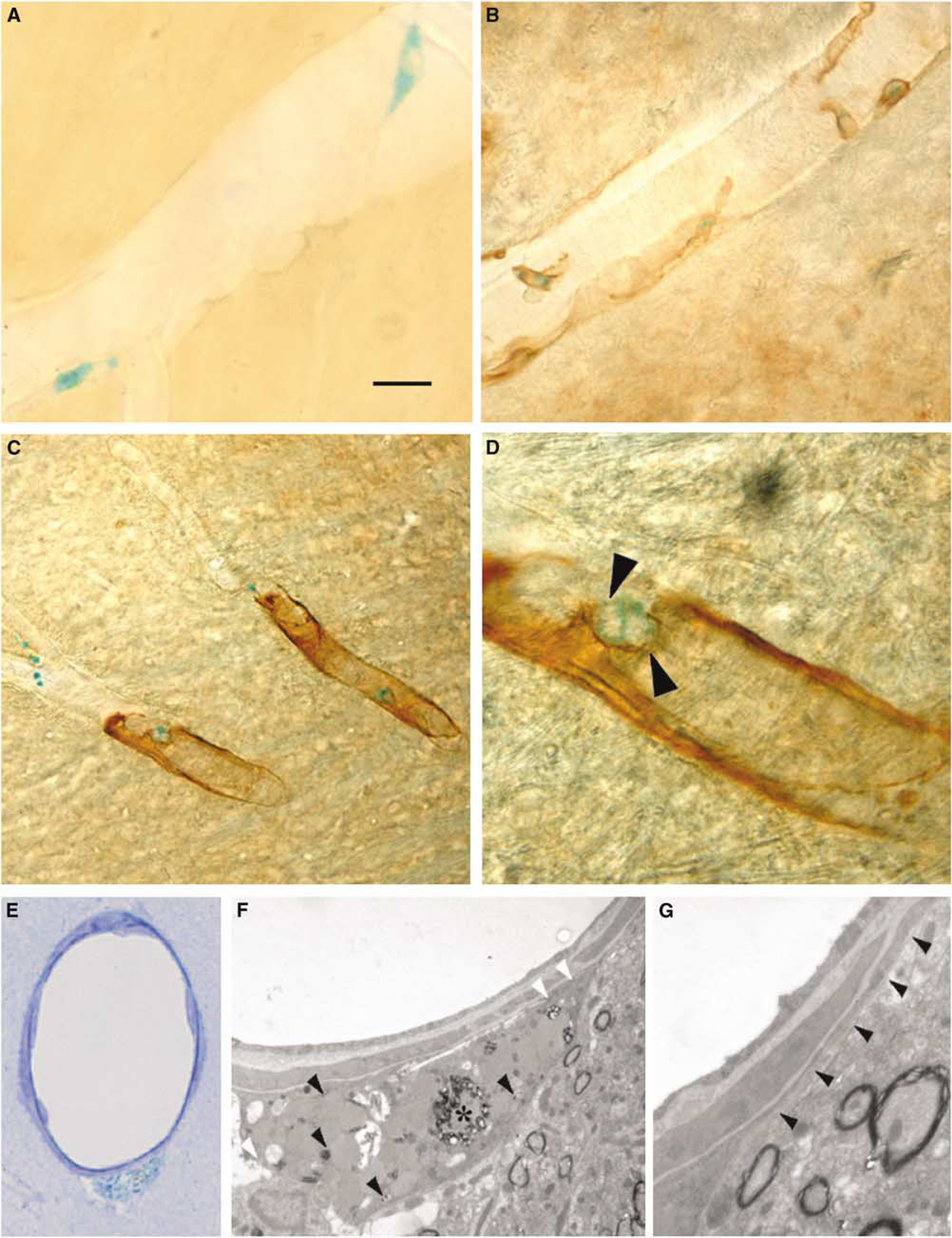
Identification of fused cells as pericytes in the brain vasculature. Analysis of mouse R262/Cre bone marrow chimeras by X-gal staining detects positive cell in the brain vasculature (
The morphology and location of the fused cells, always adjacent to endothelium and away from the lumen, resembled those of perivascular macrophages or pericytes. To further define the nature of these cells, we performed immunohistochemical analysis. The majority of the X-gal-positive cells coexpressed markers for mature, differentiated rodent pericytes (vimentin: 83.4±7.1%, Figure 1B; desmin: 78.6±9.9%, Figures 1C and 1D; smooth muscle α-actin: 74.8±6.6%), or CD45 (32.6±6.3%), a marker of perivascular macrophages. These data suggested the existence of a population of fused cells that coexpress CD45 and markers of mature pericytes. To confirm this possibility, we performed a triple desmin/CD45/β-galactosidase immunofluorescent colocalization. After analysis, we observed that 12.7±2.6%, of the β-galactosidase-positive cells coexpressed CD45 and desmin. This suggests that a small population of fused pericytes may be keeping the expression of markers from both fusion partners, what indicates that one of them should be of hematopoietic lineage. In contrast, most of the fused cells were negative for the developing pericyte marker NG-2 (0.6±0.4%). In addition, we never observed cells that were positive for both X-gal and CD-31, or von Willebrand factor (data not shown), excluding the possibility that fused cells were endothelial. This is consistent with previous reports showing that EC are generated by transdifferentiation of hematopoietic stem cells and not by cell fusion (Bailey et al, 2006).
The immunohistochemical data strongly suggested that, under normal conditions, cell fusion contributes to the generation of mature pericytes and perivascular macrophages. However, none of the markers we used are absolutely specific for pericytes, neither do they recognize all pericyte populations (Ozerdem and Stallcup, 2003; Tigges et al, 2008). Furthermore, after cell fusion, genes from both fusion partners are expressed in the resulting heterokaryon (Weimann et al, 2003), complicating the use of a single immunologic marker for its characterization. Therefore, we performed electron microscopy analysis to unequivocally identify the fused cells (Figures 1E–1G). X-gal staining is easily identifiable under electron microscopy by a precipitate in the endoplasmatic reticulum. X-gal-positive cells presented basal lamina and extensions that almost surrounded the whole capillary (Figure 1G). They contained abundant plasmalemmal vesicles, few mitochondria, and heterochromatin in the periphery of their nuclei. We also observed abundant lysosomes and, occasionally, lipid droplets (Figure 1F), all of which are typical characteristics of central nervous system pericytes (Tilton, 1991). These observations further established the identity of X-gal-positive cells as pericytes, and confirmed that a small fraction of this cell type is generated by cell fusion under normal conditions.
Several groups have recently shown the contribution of BMDC and hematopoietic stem cells to the formation of pericytes after stroke (Kokovay et al, 2006; Rajantie et al, 2004). To study the implication of cell fusion during poststroke angiogenesis, a cerebral ischemia/hypoxia episode was induced in 2 months old BMDC chimeric mice. Their brains were analyzed 2 weeks after stroke and were compared to those from sham-operated controls (2.5 months old). We observed a threefold increase of X-gal-positive cells in the poststroke vasculature (786±183 cells/brain, n=5) compared with their controls (258±82 cells/brain, n=5), mostly concentrated in the periphery of the ischemic area (Figures 2A–2C). The number of X-gal/vimentin and X-gal/desmin double-positive cells was slightly higher than under normal conditions, although not statistically significant (Figure 2I). In sharp contrast, we observed a 100-fold increase of X-gal/NG2-positive cells in the area of penumbra (489±102 cells/brain after stroke versus 5±3 cells/brain in controls, representing 62.3±12.9% of the X-gal-positive cells after stroke, Figures 2D and 2I). Interestingly, we also observed a sevenfold increase in X-gal/CD45-positive cells (609±143 cells/brain after stroke versus 83±16 cell/brain in controls, representing 77.4±18.3% of the X-gal-positive cells after stroke, Figure 2I). These data indicated the presence of fused cells double positive for CD45 and NG2. Triple immunohistochemical analysis confirmed that the majority, if not all, of the NG2/X-gal-positive cells also coexpressed CD45 (Figures 2E–2H). These results strongly suggest that stroke promotes the formation of new vasculature by generating pericytes through cell fusion.
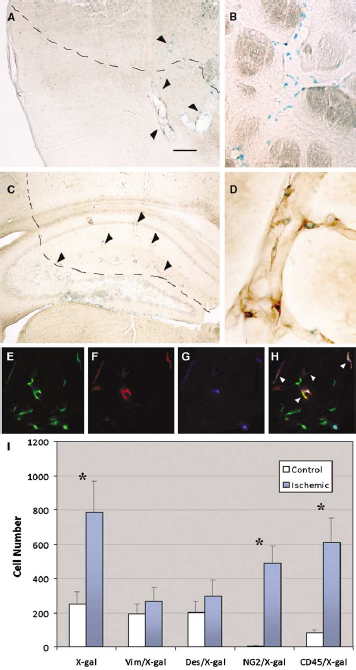
Stroke induces pericyte formation by cell fusion. (
Discussion
Pericytes are consider a heterogeneous population of cells, which are difficult to define and lack a clear ontogeny; additionally, they also possess the capacity to differentiate to other mesenchymal cell types, including smooth muscle and fibroblasts (Gerhardt and Betsholtz, 2003). Here we provide direct evidence of pericyte fusion under normal as well as pathologic conditions. This finding may explain the difficulty to detect a defined population of pericytes, its ambiguous origin, the lack of consensus in the literature for adequate specific markers, and may introduce a more dynamic concept of vascular cells, as they may change from one cell type to another as facilitated by fusion and/or transdifferentiation processes.
In the past years, several groups have extensively shown the contribution of BMDC or hematopoietic stem cells to the formation of vasculature after stroke (Bailey et al, 2006; Kokovay et al, 2006; Rajantie et al, 2004). However, the mechanism of mural cells formation from BMDC remained elusive. Our findings, together with those from other groups, strongly suggest that cell fusion and transdifferentiation processes work in concert during vasculogenesis. EC would be generated mainly by transdifferentiation of hematopoietic precursors (Bailey et al, 2006), whereas at least a subpopulation of pericytes would be formed by cell fusion. Nevertheless, our data do not exclude the possibility that BMDC also contribute to the formation of pericytes through a transdifferentiation process.
Our observations reveal an important increment in cell fusion events after stroke. Recently, inflammation has been shown as a potent modulator of cell fusion (Johansson et al, 2008). Stroke is usually associated with inflammation, which may explain the increased fusion observed in ischemic brains, particularly, in the penumbra area. This increment in fusion may also be facilitated by the invasion of macrophages in the ischemic area. These cells fuse in response to infections and injuries (Vignery, 2008). Supporting this idea is our observation of a sevenfold increment in CD45/X-gal-positive cells, and that all the new fused pericytes (X-gal/NG2-positive cells) coexpress CD45, suggesting a recent fusion event with macrophages. An alternative explanation for the increment in the number of fused cells may be the proliferation of previous fused cells that generate new progeny in response to stroke.
The induction of cell fusion has important therapeutic implications. It promotes the recovery of degenerative diseases and the rescue of recessive mutations (Alvarez-Dolado, 2007). Given the important role of pericytes in the initiation of vasculogenesis (Ozerdem and Stallcup, 2003; Tigges et al, 2008) and their regulation of EC proliferation and differentiation (Gerhardt and Betsholtz, 2003), our findings implicate an active, physiologic role for cell fusion during angiogenesis, and may open new therapeutic perspectives in the application of BMDC and cell fusion for stroke treatment. Future efforts will be devoted to the identification of fusion partners and to the study of how this process is induced by factors related with poststroke inflammation.
Footnotes
Acknowledgements
We thank Deborah Burks for her criticism and Mario Soriano for EM technical expertise. This work was supported by grants from Spanish Ministry of Health (FIS 04/2744) and Regenerative Medicine Programme from the CIPF. M.P-G and IZ were recipients of PhD fellowships from CIPF and Generalitat Valenciana, respectively.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
