Abstract
The ability of the NMDA receptor antagonist, MK-801, to protect myelinated axons after focal cerebral ischemia has been examined. Amyloid precursor protein (APP) immunocytochemistry was used to assess the anatomic extent of axonal injury, and conventional histopathology was used to assess the volume of ischemic damage to neuronal perikarya. The middle cerebral artery was permanently occluded in 16 cats. The cats were treated with either vehicle or MK-801 as a 0.5-mg/kg bolus at 15 minutes before middle cerebral artery occlusion, followed by an infusion of 0.14 mg/kg per hour. After 6 hours, the animals were killed and the brains processed for histology and immunocytochemistry. The volume of neuronal necrosis was determined from 16 preselected coronal levels of the brain. The circumscribed zones of APP accumulation in axons were mapped onto images at the same 16 coronal levels, and quantitative analysis was performed using a transparent counting grid, randomly placed over each image. The histologic appearance and anatomic location of axons with increased APP immunoreactivity was similar in animals treated with vehicle and MK-801. MK-801 failed to reduce the hemispheric APP score significantly. In vehicle-treated animals, there was a significant association between the volume of neuronal necrosis and the amount of APP immunoreactivity. MK-801 significantly reduced the slope of the association between the volume of neuronal necrosis and the amount of APP immunoreactivity compared with that observed in vehicle-treated animals. As a result, the ratio of hemispheric APP score and volume of neuronal necrosis was significantly increased with MK-801 treatment. The inability of NMDA receptor antagonists to protect axons may limit their functional efficacy in improving functional outcome after stroke.
Myelinated axons are essential for the normal functioning of the brain. The function and ultimately the structural integrity of white matter are, like gray matter, dependent on a continuous supply of energy. White matter has long been considered to be less vulnerable than gray matter to ischemia (Marcoux et al., 1982). However, recent evidence using a variety of markers targeting different structures within axons has demonstrated the acute vulnerability of axons to cerebral ischemia in vivo (Dewar and Dawson, 1997; Yam et al., 1998a), and the mechanisms of axonal injury and strategies for its protection appear to differ from those in neuronal cell bodies in gray matter. Waxman et al., using an in vitro model of isolated optic nerves, have shown that myelinated axons are susceptible to anoxia in the absence of their perikarya and that functional and structural impairment occurs within the axon itself, and not only as a consequence of impaired perikaryal function (Waxman et al., 1991, Waxman et al., 1992). Although Stys 1996, Stys 1998) has proposed that at least qualitatively the mechanisms described using the in vitro optic nerve model are pertinent to in vivo conditions, axonal injury in vivo after focal cerebral ischemia has been the subject of limited investigation.
Quantitative immunocytochemical methods have been developed to define axonal pathology after focal cerebral ischemia (Yam et al., 1998b). Amyloid precursor protein (APP) is transported by fast anterograde axonal transport (Koo et al., 1990). After a variety of cerebral insults, APP accumulates in bulblike swellings at the point of transport failure, and these are thought to be a precursor of axotomy (Kawarabayashi et al., 1991; Otsuka et al., 1991; Nakamura et al., 1992; Povlishock, 1992; Stephenson et al., 1992; Kalaria et al., 1993). APP immunocytochemistry has been used widely to detect axonal injury after head injury and ischemic insults in humans and animals because of its high sensitivity (Stephenson et al., 1992; Kalaria et al., 1993; Blumbergs et al., 1994; Sherriff et al., 1994a; Suenaga et al., 1994; Gentleman et al., 1995; McKenzie et al., 1996). A topographic relation and a statistical association have been demonstrated in the rat between axonal APP accumulation and neuronal necrosis after middle cerebral artery (MCA) occlusion in the absence of pharmacologic intervention (Yam et al., 1997).
Many drugs targeted at N-methyl-D-aspartate (NMDA) receptors have advanced to clinical trials in stroke and head injury based on their proven ability to reduce gray matter damage in animal models of focal cerebral ischemia (McCulloch, 1992). The outcome to date of the clinical trials of NMDA receptors has been disappointing, with no proof of improved functional outcome in patients (Dyker and Lees, 1998). Although the failure to translate preclinical insight into therapy is multifactorial, we have proposed that the inability of NMDA receptor antagonists to improve functional outcome in humans may relate to their failure to protect oligodendrocytes and axons in white matter (Dewar et al., 1999b). White matter contains negligible levels of NMDA receptors (Jones and Baughman, 1991), and NMDA antagonists are not protective in the rat optic nerve model (Stys, 1998).
In the present study, we sought first to establish in the cat whether the anatomic extent of axonal injury in white matter (assessed by APP accumulation) was related to the volume of neuronal necrosis in gray matter (assessed by conventional histopathology). Second, we examined whether pretreatment with the NMDA antagonist, MK-801, altered the relative amounts of white matter and gray matter injury, assessed using the same techniques. The investigations were conducted in the cat because of its similar proportion of white matter to humans and because of the compelling evidence that MK-801 reduces ischemic damage to neuronal perikarya in gray matter in this species (Ozyurt et al., 1988; Park et al., 1988a; Uematsu et al., 1991; Dezsie et al., 1992).
METHODS
Sixteen adult cats were anesthetized and MCA occlusion was performed. The cats were anesthetized initially with alphaxalone/alphadolone (9 mg/kg given intravenously), intubated, and connected to a positive-pressure ventilator delivering nitrous oxide and oxygen in an open circuit. Polyethylene catheters were inserted into both femoral veins and one femoral artery for the administration of drugs and the continuous monitoring of arterial blood pressure and blood gases. Anesthesia was maintained throughout the course of the investigation with intravenous chloralose (60 mg/kg), supplemented as necessary to prevent the return of the corneal reflex. Throughout the experimental period, the animals were maintained normotensive (mean arterial blood pressure greater than 80 mm Hg) and normocapnic (PaCO2 close to 32 mm Hg) by adjusting the stroke volume of the respirator. Metabolic acidosis was corrected by administration of 8.4% sodium bicarbonate. Samples of arterial blood were taken frequently to determine the respiratory status using a direct-reading electrode system. The cats were maintained normothermic by means of a heating blanket with feedback control using a rectal thermometer.
The left MCA was occluded using a transorbital approach. The head of the cat was placed in a stereotactic frame. With microsurgical techniques, the left orbit was exenterated and the optic foramen and optic fissure were enlarged with a dental drill to expose the dura mater overlying the MCA close to its origin. With the aid of an operating microscope, the dura was incised and the MCA exposed. The trunk of the MCA and all visible branches of the lenticulostriate arteries were coagulated with bipolar diathermy and divided with microscissors. After MCA occlusion, anesthesia was maintained for six hours, and physiologic parameters were monitored (Table 1).
Physiologic variables before and after MCA occlusion in the cat
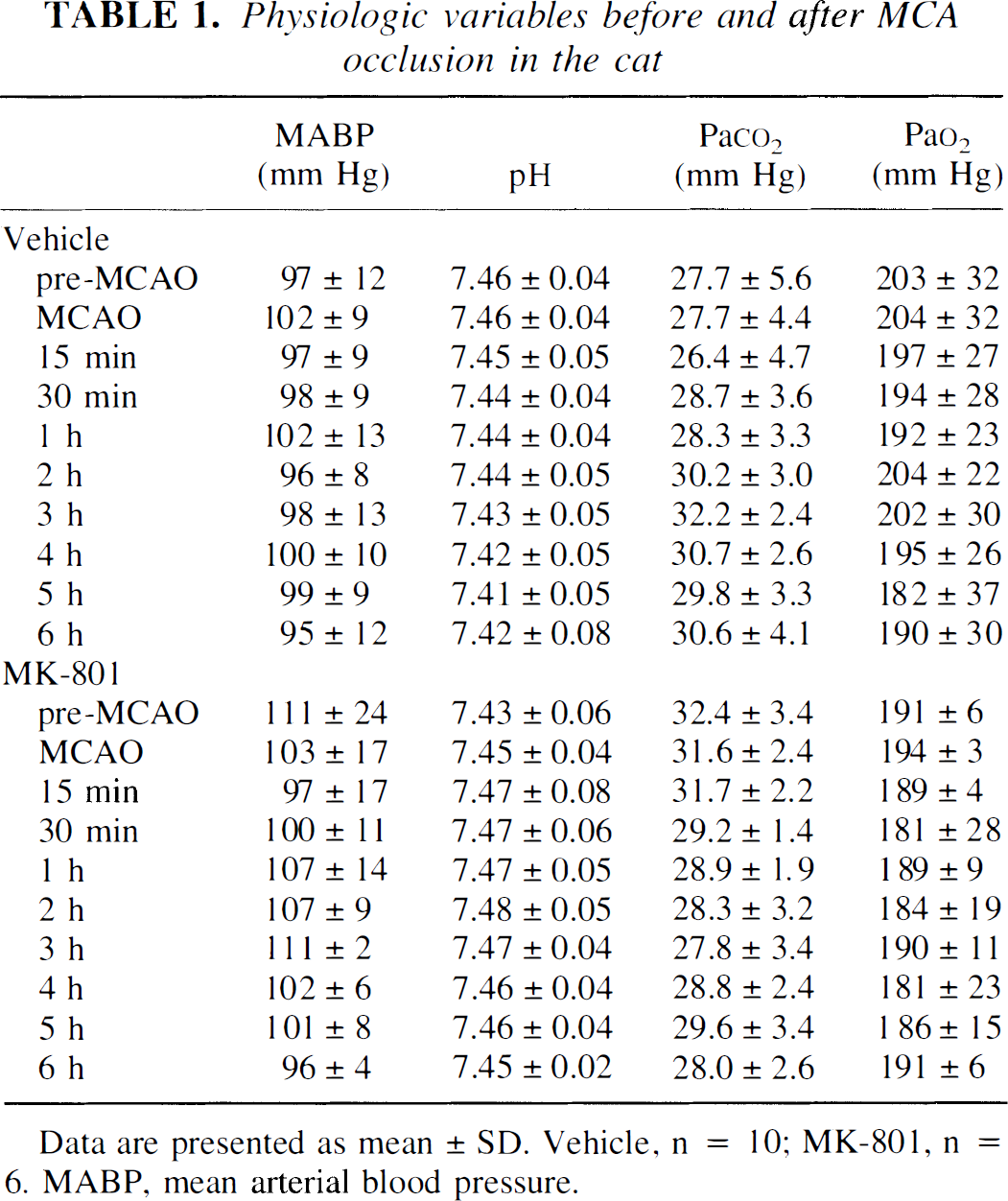
Data are presented as mean ± SD. Vehicle, n = 10; MK-801, n = 6. MABP, mean arterial blood pressure.
The cats were randomly divided into a vehicle-treated control group (n = 10) and a drug-treated group (n = 6). The drug-treated group received an intravenous bolus of MK-801 (0.5 mg/kg) by intravenous injection 15 minutes before MCA occlusion; then, after completion of the bolus injection, MK-801 was infused at 0.14 mg/kg per hour (0.15 mL/min) for the duration of the experiment. The bolus and infusion concentrations were selected to achieve a steady-state target plasma concentration of 300 nmol/L, a dosing regimen based on the plasma half life of MK-801 in this species (Ozyurt et al., 1988 and their unpublished observations), knowledge of loading dose requirements (Mayer al., 1975), and definitions of the plasma concentrations of MK-801 required for neuroprotection (Gill et al., 1991). The vehicle-treated control group of 10 cats received a 1-mL intravenous injection of saline 15 minutes before MCA occlusion, followed by an intravenous saline infusion at a rate of 0.15 mL/min for the duration of the experiment.
Six hours after MCA occlusion, the cats were killed by transcardiac perfusion fixation with 40% formaldehyde, glacial acetic acid, and methanol (FAM) in a ratio of 1:1:8. Briefly, the cat was placed in a supine position and given heparin. A thoracotomy was performed and a cannula introduced into the ascending aorta through the left ventricle. Physiologic saline was infused at a pressure equivalent to mean arterial pressure until blood was removed from the body. This was followed immediately by 1.5 L of FAM fixative at the same pressure. After perfusion, the cats were decapitated and the head was stored in fixative at 4°C for at least 12 hours; the brain was then removed and immersed in FAM for a further 24 hours before transfer to 70% methanol. After marking the left side of the forebrain with India ink, the hindbrain was detached by a cut through the midbrain and the cerebral hemispheres were cut into three equally spaced coronal blocks. These blocks were embedded in paraffin wax and 6 sections 10 μm thick were cut at 200-μm intervals throughout the specimens.
Sections were stained by hematoxylin and eosin and a method that combined Luxol fast blue and cresyl violet. The sections that corresponded most closely to 16 stereotactically predetermined coronal planes of cat brain from the atlas of Reinoso-Suarez (1961) were examined by conventional light microscopy without prior knowledge of the animal's history. Morphologic characteristics of early neuronal necrosis (microvacuolation, shrinkage, triangulation, and hyperchromasia of the cell body and nucleus) (Brierley and Graham, 1984) were charted on the diagrams drawn to scale and formed the basis of quantitative assessment of ischemic brain damage in a manner analogous to that previously described for the rat (Osborne et al., 1987). The area of ischemic necrosis in the cerebral hemisphere, cerebral cortex, and caudate nucleus was determined from the diagrams at each of the 16 coronal planes. The volumes of ischemic neuronal necrosis were calculated from the areas of damage at the different coronal planes and their anterior-posterior coordinates.
Sections adjacent to those used for mapping areas of neuronal necrosis were prepared for immunocytochemistry so that the amount of APP immunoreactivity could be quantified at the same coronal levels. Sections were mounted on slides coated with poly-L-lysine and dried at 37°C overnight. To remove the wax, the sections were placed in Histoclear (Natural Diagnostics, Atlanta, GA, U.S.A.). The sections were dehydrated by placing them in absolute alcohol for 2 minutes, and they were then rinsed in methanol. They were left in methanol and hydrogen peroxide solution (0.5%) for 30 minutes. To enhance APP immunoreactivity, the sections were microwaved (Sherriff et al., 1994b) in citrate buffer (pH 6) for 5 minutes, reexamined to ensure they were still covered with buffer, microwaved for a further 5 minutes, and then allowed to cool for 20 minutes. The sections were rinsed in tap water and phosphate-buffered saline (PBS) for 2 minutes. Bovine serum albumin (0.05%) and normal horse serum (10%) in PBS was applied for 1 hour to block nonspecific binding sites. The excess blocker was wiped off, and optimally diluted primary antibody at a concentration of 1:300, which had been made up in the bovine serum albumin and normal horse serum in PBS, was applied and left overnight at 4°C. As controls, the primary antibody was eliminated. No positive staining was seen in any of the control sections. The sections were washed in PBS for 3 × 10 minutes. Diluted biotinylated antibody solution was applied for 1 hour, and sections were washed again in PBS for 3 × 10 minutes. The avidin/biotinylated horseradish peroxidase solution (Vectastain Elite ABC Kit, Vector Laboratories, Peterborough, U.K.), which had been prepared 30 minutes before use, was applied for 1 hour. The washing procedure with PBS was repeated, and then the sections were allowed to develop in 3,3′-diaminobenzidine solution (Sigma, Poole, U.K.) for 8 to 10 minutes. Finally, the sections were rinsed with water, dehydrated, cleared, and mounted in DPX mountant (Lamb, London, U.K.).
Because diaminobenzidine was used as the chromagen in the immunocytochemistry, the increase in APP immunoreactivity in the white matter viewed microscopically stained intensely brown (Fig. 1). Slides stained for APP from 16 coronal sections from each animal, at the defined coronal planes, were placed on a light box, and the image was captured using an 8-bit standard CCD video camera and transferred to an MCID-M4 image analyzer. The complete coronal section could then be viewed on a monitor and digitized, and the image was printed using a laser printer. The increased APP immunoreactivity was visible on the images (see Fig. 1A). The sections were also viewed using a conventional light microscope at ×2.5 magnification. Any increased APP immunoreactivity visible on the slides but not visible on the scanned images was transcribed onto the image. This procedure was repeated for each animal at each coronal level so that the region of increased APP immunoreactivity could be established at each coronal level for each animal in both groups.
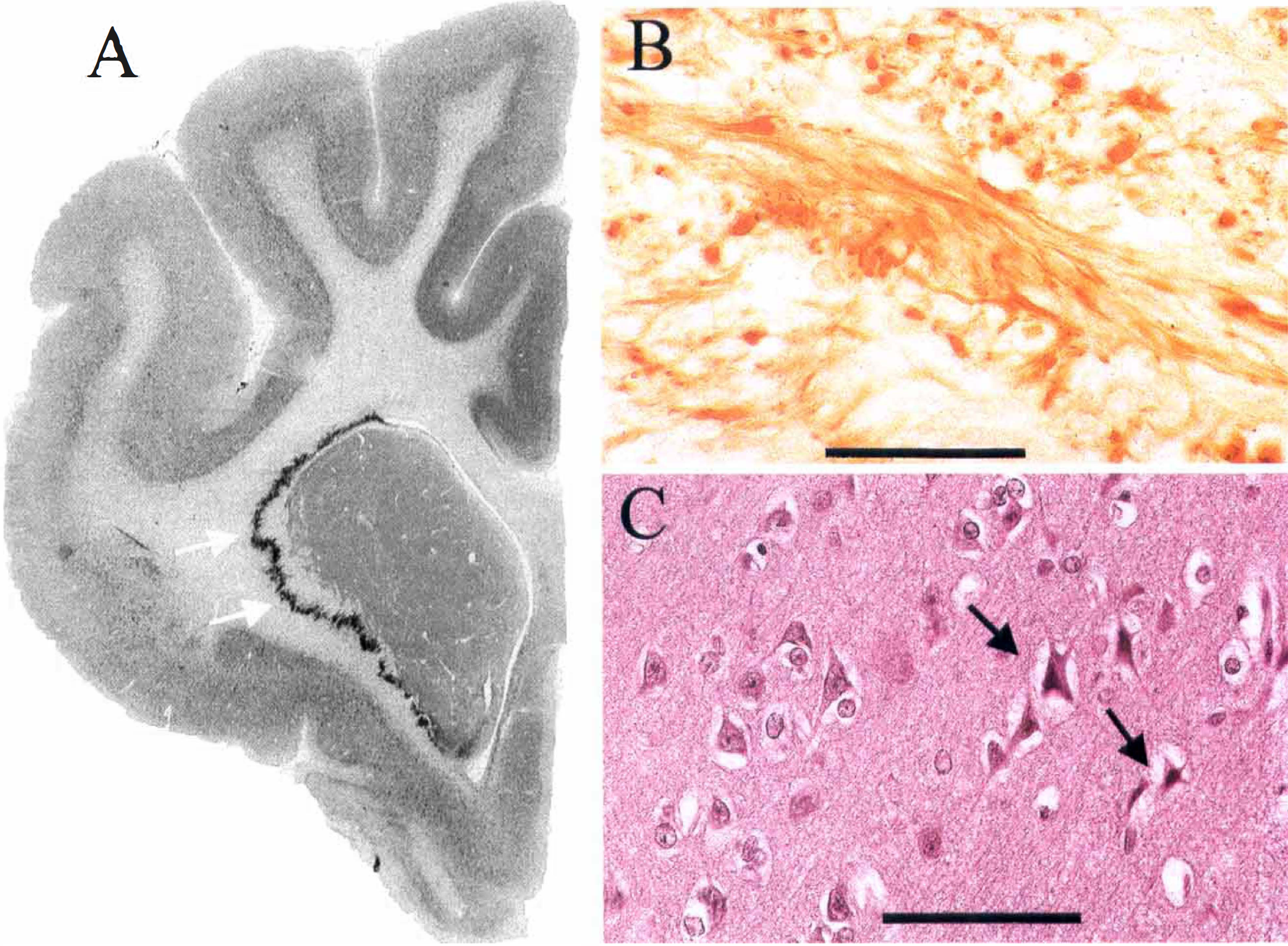
Amyloid precursor protein (APP) accumulation and neuronal necrosis six hours after middle cerebral artery occlusion in the cat. (
For quantitative analysis of the APP immunoreactivity, we analyzed the images from 16 coronal sections, for each animal in both groups, showing the increased APP immunoreactivity. A transparent counting grid, arranged in a regular square array, was randomly placed over each image, and the number of points overlying the area of increased APP immunoreactivity was counted. The total APP score for each animal was the sum of the points on target for all 16 coronal levels.
The intrarater variability and interrater reliability were assessed on multiple randomly selected samples of images from the two cat populations and comparing the two sets of results. Using the counting grid for the quantitative method of APP analysis, the intrarater correlation (r = 0.99) and the interrater correlation (r = 0.96) was excellent.
Our hypothesis was that MK-801 would fail to reduce the hemispheric APP score but would alter the relation with the volume of neuronal necrosis. To test this hypothesis, the hemispheric APP scores in both groups were compared using Student's t-test. The relation between the hemispheric APP score and the volume of neuronal necrosis was described by linear regression analysis (Pearson's). The slopes of the regression analysis for the vehicle and treatment groups were compared, as was the ratio of the hemispheric APP score and the volume of neuronal necrosis in individual animals between the two treatment groups using Student's t-test. To avoid the statistical issue of dimensionality, no other analyses were performed on the subcomponents of these data sets (e.g., APP scores in individual coronal planes, areas of neuronal necrosis, volumes of neuronal necrosis in different regions), which are reported for descriptive purposes. Data are presented as mean ± standard deviation.
RESULTS
Histopathology and APP immunocytochemistry
All brains were judged to be well perfusion-fixed, as evidenced by good neuronal morphology, the absence of intravascular blood, and the lack of cytoplasmic artifacts (Brown and Brierley, 1968). Ischemic damage was observed only within the territory of the occluded MCA (i.e., the dorsolateral cortex and the neostriatum). These areas showed the morphologic characteristics of early neuronal necrosis (Brierley and Graham, 1984): neuronal perikarya with shrinkage and triangulation of the nucleus and cytoplasm and increased basophilia of cytoplasm (see Fig. 1C). In the hemisphere ipsilateral to the occluded MCA, circumscribed zones of intense APP immunoreactivity were observed in swollen or bulbous axons in subcortical white matter (see Fig. 1B). These zones of axons with increased APP immunoreactivity were particularly prominent in the ventral and lateral aspects of white matter and in white matter immediately adjacent to the caudate nucleus (see Fig. 1A). The histologic appearance and anatomic location of the axons with increased APP immunoreactivity were similar in animals treated with vehicle and MK-801.
MK-801 and axonal injury
MK-801 failed to reduce significantly the total APP score summated from the 16 coronal levels examined (Fig. 2). The total APP score (summated from individual scores in 16 planes) was 253 ± 65 for the vehicle-treated group and 322 ± 52 for the MK-801 group (see Fig. 2B). The APP scores for each of the 16 planes were similar in vehicle-treated animals (Fig. 3). Although MK-801 did not significantly alter the index of axonal injury (i.e., the APP score) MK-801 did alter significantly the relation between axonal injury and the volume of neuronal necrosis (Fig. 4). In vehicle-treated animals, there was a significant association (r = 0.69, P < 0.03) between the hemispheric APP score and the volume of neuronal necrosis. MK-801 treatment altered significantly (P < 0.02) the slope of the association from 43 ± 16 × 10−3 in vehicle-treated animals to 6 ± 15 × 10−3 in MK-801 animals (see Fig. 4). The alteration in the relation between axonal injury and necrosis of neuronal perikarya was also reflected by an increase in the ratio of hemispheric APP score and volume of neuronal necrosis from 0.17 ± 0.07 in the vehicle group to 0.32 ± 0.16 in the MK-801 group (P < 0.02) (see Fig. 2C).
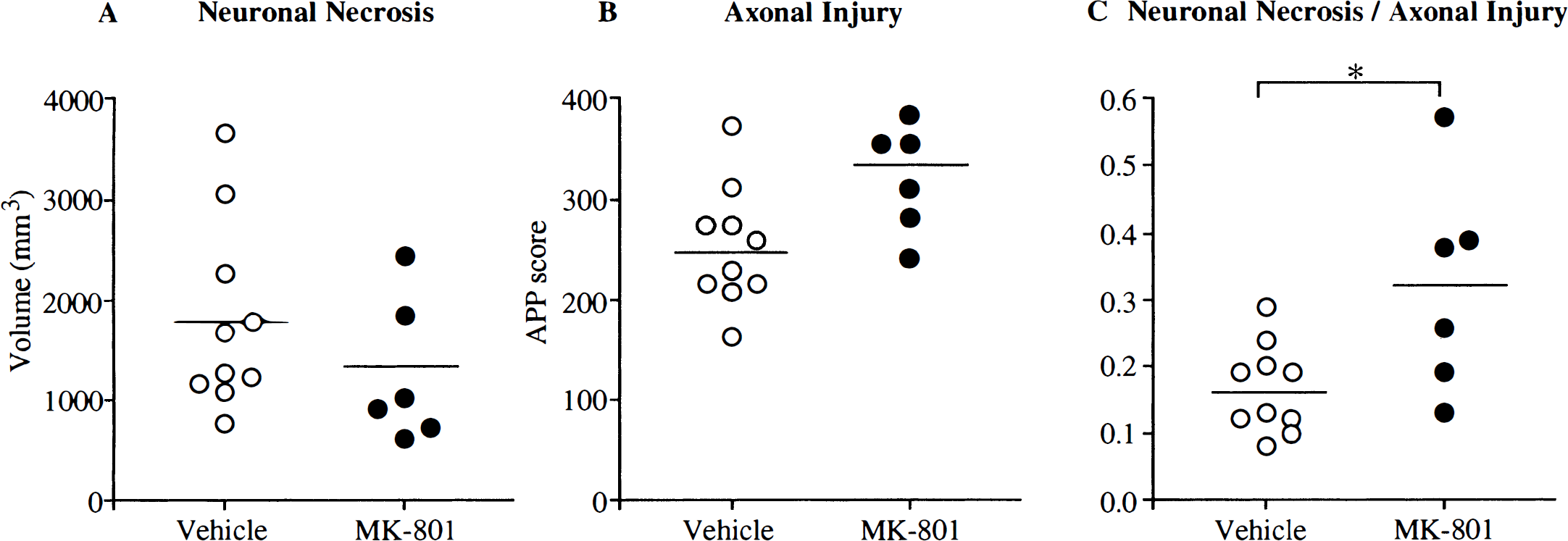
The effects of MK-801 on total hemispheric neuronal necrosis and axonal injury and their ratio in individual animals. (
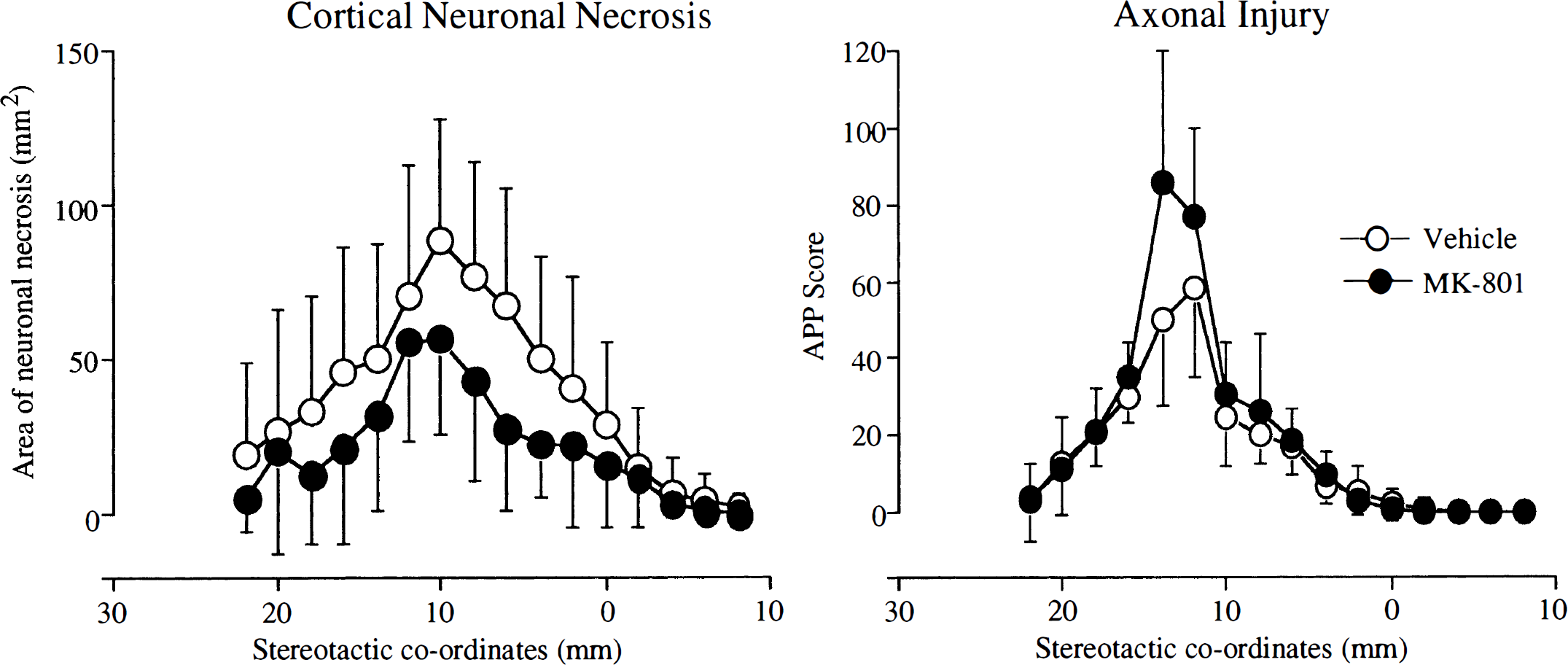
The effect of MK-801 on the amount of cortical neuronal necrosis and axonal injury at the 16 defined coronal planes. Although MK-801 animals had a smaller area of cortical neuronal necrosis at each coronal plane, there was no reduction in the amyloid precursor protein score compared with the vehicle-treated animals. Data points are the mean at each coronal plane (stereotactic coordinates), bars represent standard deviation. Vehicle, n = 10; MK-801, n = 6.
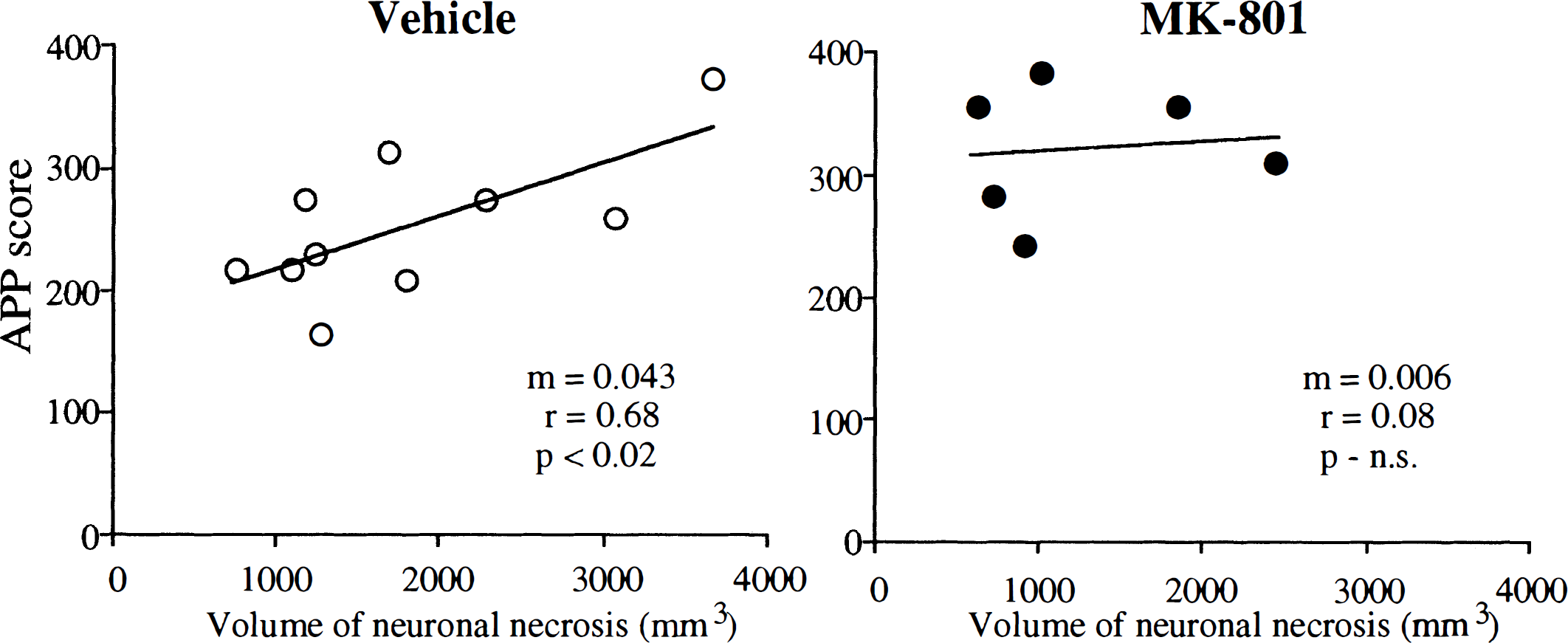
Relations between the volume of neuronal necrosis and amyloid precursor protein (APP) scores in vehicle-treated ○ and MK-801 • animals. The linear regression line is shown for each group. In vehicle-treated animals there was a significant association between the hemispheric APP score and the volume of neuronal necrosis. The slope of the regression line (m) in the MK-801 group was significantly less than in the vehicle-treated group (P < 0.02), indicating that the relation between neuronal necrosis and axonal injury was altered by MK-801 treatment. N.S., not significant.
There were no significant differences in any of the key physiologic variables between the vehicle-treated and MK-801 groups (see Table 1).
DISCUSSION
Quantitative accumulation of APP has been used previously to assess axonal injury in the rat after focal ischemia (Yam et al., 1998b). The present approach in the cat has several major advantages for assessing the ability of drugs to alter ischemic damage to myelinated axons. The use of a species (cat rather than rat) with a large gyrencephalic brain more closely reflects the cerebral anatomy of humans. In the cat, the proportionately larger volume of white matter compared with the rat allows easier assessment of protective effects in white matter after therapeutic intervention. In addition, the use of a counting grid in the present study rather than a semiquantitative score (Yam et al., 1998b) improved both the sensitivity and objectivity of the assessment method.
Our investigations provide insight into three aspects of axonal injury in white matter after focal cerebral ischemia. Increased APP immunoreactivity was consistently observed in white matter tracts after focal cerebral ischemia in the cat, emphasizing that axonal pathology is a consistent consequence of permanent MCA occlusion in this species. Second, the larger the volume of the ischemic lesion in gray matter, the greater the extent of increased APP immunoreactivity. Finally, the results demonstrate that MK-801 fails to reduce the amount of myelinated fiber tract damage, as assessed by accumulation of APP in animals, whereas the agent predictably attenuated the amount of gray matter damage.
The mechanisms of cellular injury in white matter are less well understood than those in gray matter. However, myelinated fiber tracts, both in vitro and in vivo, are vulnerable to anoxic or ischemic injury, independent of perikaryal damage. Isolated optic nerves display structural damage in response to relatively short periods of anoxia (Stys, 1998; Waxman et al., 1992), as do myelinated tracts in vivo in response to short periods of focal ischemia (Dewar and Dawson, 1997; Yam et al., 1998a). In both experimental situations, axonal damage is associated with derangements of cytoskeletal structure involving both microtubule and neurofilament proteins. Cytoskeletal breakdown at the point of ischemic injury is presumed to precede the accumulation of transported proteins such as APP (Stephenson et al., 1992; Yam et al., 1997, Yam et al., 1998a; Dietrich et al., 1998). This sequence of events implies that axonal transport of proteins continues in portions of the axon distal to the point of injury. Therefore, APP accumulation may not reveal all ischemically damaged axons, because those in which there is no axonal transport will not be detected. Staining of the tissue with a marker that directly detects cytoskeletal breakdown, such as neurofilament antibodies, may reveal the full anatomic extent of axonal damage. However, in terms of a potential therapeutic approach, axons whose terminals and perikarya are also damaged by the ischemic insult may not constitute a salvageable target. Thus, although APP accumulation may have underestimated the full extent of axonal damage in the tissue, it provides an assessment of the axons that may benefit from therapeutic intervention such as early reperfusion (Valeriani et al., 2000).
The temporal evolution of ischemic damage to neuronal perikarya, and its reversibility, has been the subject of intensive investigation (Jones et al., 1981). It remains to be established whether the temporal evolution of axonal injury parallels that of neuronal perikarya in relation to both the duration and severity of the ischemic insult. It is interesting that the APP scores in the MK-801 group were modestly increased compared with the controls; this may be due to rescue by the drug of neuronal perikarya that could maintain protein transport to the point of damage in their axons.
Although both components of the functional unit comprising the axon and its oligodendrocytes (Petito, 1986; Dewar and Dawson, 1995; Pantoni et al., 1996; Irving et al., 1997; Dewar et al., 1999a) are susceptible to ischemic damage, the major mechanistic advances in the therapy of stroke during the past decade have been targeted toward the protection of the neuronal cell body, with protection of white matter being largely neglected. After ischemia, the extracellular concentration of the excitatory neurotransmitter glutamate has been show to increase to levels that are toxic to neurons. Consequently, drug development has been targeted at the blockade of glutamate release or the prevention of its action at the postsynaptic receptors (Wahlgren, 1997). Because the predominant pathway for calcium ion entry into neuronal perikarya in ischemic tissue of the central nervous system is via the glutamate-activated NMDA receptor channel complex, much effort has been focused on the discovery and development of molecules that can inhibit the ion fluxes induced by activation of this receptor-channel complex. NMDA antagonists, including MK-801, have been shown in the experimental setting to reduce the volume of neuronal necrosis after an ischemic insult (Ozyurt et al., 1988; Park et al., 1988a, Park et al., 1988b). In contrast to these results, we demonstrate here that MK-801 fails to reduce the extent of axonal damage. Exposure to glutamate in vitro did not induce cytoskeletal breakdown within axons of the spinal cord, although glutamate exposure did induce myelin degradation and cytoskeletal damage in oligodendrocytes by means of AMPA receptors (Li and Stys, 2000). Oligodendrocytes in vitro and in vivo are insensitive to NMDA (McDonald et al., 1998; Sánchez-Gómez and Matute, 1999. MK-801 failed to reduce the extent of ischemic pathology in oligodendrocytes after MCA occlusion in the rat (Irving et al., 1997).
The reported success of NMDA antagonists in the protection of gray matter has permitted the progression of drugs to clinical trials in humans (Small and Buchan, 1997; Bullock, 1998). The results of the phase III trials have generally been disappointing. Some of the trials have been stopped prematurely when interim analysis has raised concerns about the risk-benefit ratio, and others have been stopped when no difference was found between active treatment and placebo (Muir and Lees, 1995). Even in those trials that have been completed, the results have not necessarily proven the clinical efficacy of the drugs concerned. The reasons for these drug failures are almost certainly multifactorial. However, we cannot ignore that NMDA antagonists are targeted toward the salvation of gray matter and therefore may not be effective in cell components of white matter that are devoid of NMDA receptors (Jones and Baughman, 1991).
The functional integrity of the central nervous system depends not only on the normal operation of neuronal perikarya but also on their axonal interconnections (Filley, 1998). The functional importance of axonal damage in humans is highlighted by clinicopathologic studies of traumatic brain injury, where the extent of diffuse axonal injury is a major determinant of patient morbidity (Gennarelli, 1996). Diffuse axonal injury is an important cause of the vegetative state and severe disability after a head injury (Adams et al., 1989). However, the protection of myelinated fiber tracts has hitherto been largely neglected in preclinical investigations of drug action. The use of agents targeted toward the protection of gray matter alone, such as MK-801, may be irrelevant to the preservation of brain function after a stroke or head injury. The success of future strategies will depend on the development of either single agents or combination therapy targeted at the protection of both gray and white matter. Brain protection, rather than neuroprotection, may be the key to future success and should be the goal of antiischemic drug development.
Footnotes
Acknowledgments
The authors thank the technical and secretarial staff at the Wellcome Surgical Institute.
