Abstract
Protection of both grey and white matter is important for improvement in stroke outcome. In the present study the ability of a competitive α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) antagonist to protect axons, oligodendrocytes, and neuronal perikarya, was examined in a rodent model of transient focal cerebral ischemia. SPD 502 (8-methyl-5-(4-(N,N-dimethylsulfamoyl)phenyl)-6,7,8,9-tetrahydro-1H-pyrrolo[3,2h]-isoquinoline-2,3-dione-3-o(4-hydroxybutyricacid-2-yl)oxime) was administered as an intravenous bolus (16 mg/kg) 15 minutes before transient (3-hour) middle cerebral artery (MCA) occlusion, followed by an intravenous infusion (16 mg kg−1 hr−1) of the drug for 4 hours. Twenty-one hours after ischemia, axonal damage was reduced by 45% (P = 0.006) in the SPD 502–treated group compared with the vehicle. The anatomic extent of ischemically damaged oligodendrocytes, determined by Tau1 immunoreactivity, was reduced in the cerebral cortex by 53% (P = 0.024) in SPD 502–treated rats compared with vehicle-treated rats, but there was minimal effect in the subcortex. The volume of neuronal perikaryal damage after MCA occlusion was significantly reduced by SPD 502 in the cerebral cortex (by 68%; P = 0.005), but there was minimal change in the subcortex with drug treatment. The AMPA receptor antagonist significantly reduced the anatomic extent of lipid peroxidation (assessed as the volume of 4-hydroxynonenol immunoreactivity), and this may have contributed to its ability to protect multiple cell types in ischemia. The data demonstrate that AMPA blockade protects both grey and white matter from damage induced by transient focal ischemia.
Stroke affects both grey and white matter, and the neurologic consequences of an ischemic insult reflect this. Occlusion of the middle cerebral artery (MCA) is one of the most common causes of stroke, and this artery supplies large regions of the cerebral cortex and provides extensive blood supply to subcortical white matter (Warlow, 1993). In addition, when selected branches of the MCA are occluded and only cortical regions are affected, damage to myelinated fibers within the cortical projection network will also occur. Despite this, assessment of ischemic pathology in experimental models is dominated by histologic staining of neuronal perikarya. When more sensitive and specific methods are used, damage to neuronal axons and oligodendrocytes, as well as neuronal perikarya, is observed (Dewar et al., 1999; Pantoni et al., 1996; Petito, 1986; Petito et al., 1998).
Pharmacological intervention strategies aimed at reducing ischemic brain damage that have been investigated clinically for the treatment of stroke or head injury are largely targeted at pathogenic mediators in neuronal perikarya, for example, activation of N-methyl-D-aspartate (NMDA) receptors. Despite their apparent ability to reduce the extent of neuronal damage in animal models of focal ischemia, NMDA receptor antagonists have not improved functional outcome when given to stroke patients (Lees et al., 2001). The failure to translate the neuroprotection seen in animal models into reductions of functional deficits in patients may be due to the inability of NMDA receptor blockade to reduce axonal damage (Dewar et al., 1999). Experimental support for this is provided by the demonstration that pretreatment with the NMDA antagonist, MK-801, which markedly reduces ischemic damage to neuronal perikarya (Ozyurt et al., 1988), had no protective effect on axons after MCA occlusion (Yam et al., 2000). Similarly, pretreatment with MK-801 did not reduce the pathologic response of oligodendrocytes in the external capsule to focal cerebral ischemia in the rat (Irving et al., 1997). These data are consistent with the lack of NMDA receptors on axons and oligodendrocytes. Despite this, however, a recent study reported that the NMDA antagonist, Aptiganel, reduced pallor of myelin staining measured at a single coronal plane after transient ischemia in the rat (Schabitz et al., 2000). It remains to be established whether this truly represents attenuation of structural damage to axons and oligodendrocytes or whether it reflects a reduction of white matter edema.
To select pharmacological agents that have the potential to protect both grey and white matter from ischemia, it is essential to identify pathogenic mechanisms that occur in neurons, axons, and oligodendrocytes. Following cerebral ischemia, glutamate activates not only NMDA but also α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA)/kainate receptors, and blockade of either receptor reduces damage to neuronal perikarya (Gill, 1994). Evidence for the involvement of AMPA toxicity in white matter damage comes from studies of model systems other than focal cerebral ischemia. For example, glutamate excitotoxicity via AMPA receptors mediates axonal damage after mechanical or ischemic injury of the spinal cord (Agrawal and Fehlings, 1997; Kanellopoulos et al., 2000; Wrathall et al., 1994, 1997). In an in vitro white matter tract preparation, the noncompetitive AMPA antagonist GYKI52466 attenuates the impairment of compound action potentials produced by 60 minutes of anoxia and prevents myelin degradation (Li et al., 1999). AMPA receptors located on the myelin sheath were proposed to be activated by glutamate released from surrounding glia and the axolemma by reversal of the Na+-dependent glutamate transport system (Li et al., 1999; Li and Stys, 2000). AMPA receptors are also located on oligodendrocytes, and AMPA receptor-mediated excitotoxicity has been implicated in oligodendrocyte cell death in vitro and in vivo (Alberdi et al., 2002; Gallo and Russell, 1995; Matute et al., 1997; McDonald et al., 1998; Pantneau et al., 1994; Sanchez-Gomez and Matute, 1999; Tekkok and Goldberg, 2001; Yoshioka et al., 1995). Activation of non-NMDA glutamate receptors induces production of reactive oxygen species in both neurons and glia (Bae et al., 2002; Carriedo et al., 2000; Matsuoka et at, 1999; Mattson et al., 1995). Lipid peroxidation due to free radical attack on cellular membranes results in the formation of 4-hydroxynonenal (4-HNE) (Keller and Mattson, 1998). We have previously reported the presence of this toxic lipid peroxidation by-product in both grey and white matter after transient focal cerebral ischemia in the rat and that 4-HNE is toxic to neuronal perikarya, axons, and oligodendrocytes (McCracken et al., 2000).
SPD 502 is an isatine oxime derivative that has greatly improved physiochemical properties compared with previously used AMPA receptor antagonists such as NBQX, and it reduces neuronal perikaryal damage induced by transient global ischemia (Nielsen et al., 1999). The aim of the present study was to assess whether this novel competitive AMPA receptor antagonist, SPD 502, could protect axons, oligodendrocytes, and neuronal perikarya from transient focal cerebral ischemia in the rat. We also determined if SPD 502 reduced the extent of 4-HNE immunostaining. This study was designed as a proof of the concept that an AMPA receptor antagonist with improved bioavailability had the potential to protect both grey and white matter damage from a transient ischemic insult.
MATERIALS AND METHODS
Surgery
Male Sprague-Dawley rats (280 to 320 g) were deeply anesthetized in a Perspex box using 5% halothane and 30% oxygen/70% nitrous oxide, intubated and mechanically ventilated on 1 to 2% halothane 30% oxygen/70% nitrous oxide for the duration of surgery. Both arterial and venous lines were cannulated to allow blood pressure monitoring and for drug infusion, respectively. Rectal temperature was monitored throughout the period of anesthesia and maintained close to 37°C with heating lamps directed at the lower trunk. Microvascular clips were placed on the left common carotid artery and internal carotid artery (ICA), and the external carotid artery (ECA) was electro-coagulated distal from the bifurcation of the ECA and ICA. A heat-blunted 3.0 monofilament was inserted in the ECA and advanced up through the ICA for approximately 22 mm until resistance was felt, to block the origin of the MCA (Longa et al., 1989). SPD 502 (16 mg/kg; n = 7; Shire Pharmaceuticals) was administered as an intravenous bolus (16 mg/kg) 15 minutes before monofilament insertion and then as a continuous infusion (16 mg kg− hr−1) for 4 hours. Control-treated animals received identical volumes of the vehicle at the same time points (bolus 1 mL/kg of 24 mmol/L NaOH in isotonic saline; infusion 4 mL kg− hr−1 of 6 mmol/L NaOH in isotonic saline). Steady-state drug cerebrospinal fluid levels were estimated to be 300 nmol/L based on the pharmacokinetics and brain entry of the drug (Morrison, unpublished observations). After 3 hours of occlusion the monofilament was removed, animals were allowed to recover and placed in a thermally controlled environment (23°C) for 21 hours. Animals treated with SPD 502 appeared more lethargic than the vehicle-treated animals in the initial postoperative period. In the latter 12 to 24 hours of the recovery period there were no overt behavioral differences between the two treated groups. There was no significant difference between the vehicle-treated and SPD 502–treated groups in the mean arterial blood pressure (92.6 ± 2.4 mm Hg and 86.6 ± 1.8 mm Hg, respectively) or rectal temperature (36.8 ± 0.1°C and 36.9 ± 0.1°C, respectively), before or after occlusion of MCA. Twenty-four hours after the onset of ischemia animals were perfused transcardially with 0.9% saline and 4% paraformaldehyde. Rat brains were processed for paraffin embedding and 6-μm sections cut for histology and immunohistochemistry.
The inclusion criteria for analysis of the tissue were the absence of an intracranial hemorrhage and the presence of neuronal perikaryal damage.
Tissue staining
Sections selected from 8 predetermined coronal planes were stained with hematoxylin and eosin for measurements of ischemic damage to neuronal perikarya (Osborne et al., 1987). Adjacent sections were immunostained with amyloid precursor protein (APP) for measurements of axonal damage, or for tau protein for measurements of oligodendrocyte pathology, as described previously (Valeriani et al., 2000). For descriptive analysis of damage to myelinated fiber tracts, sections were also immunostained for the neurofilament heavy subunit (NF200) as a marker of cytoskeletal structure, or for the myelin proteins myelin basic protein (MBP) and proteolipid protein (PLP). To determine the anatomic extent of the lipid peroxidation product 4-HNE, sections at each of the 8 coronal planes were immunostained using an antibody raised against 4-HNE Michael adducts. For immunostaining, sections were pretreated for antigen retrieval by microwaving in a citric acid buffer (10 mmol/L; pH 6) twice for 5 minutes each time, then incubated in 3% hydrogen peroxide for 30 minutes to block endogenous peroxidase, followed by blocking solution (10% normal horse serum, 0.5% bovine serum albumin, and phosphate-buffered saline) for 1 hour. Sections were exposed to primary antibodies diluted in blocking solution overnight at 4°C. The following antibodies were used: anti-APP (clone 2CC11; 1:500; Chemicon International, Southampton, U.K.); Tau1 (1:750; Chemicon International, Temecula, CA, U.S.A.); anti-NF200 (clone NE14; 1:5,000; Sigma Chemical); anti-MBP (1:10,000; Chemicon); anti-PLP (1:1,000; Oncogene, Cambridge, MA, U.S.A.); and anti–4-HNE (1:2,000; Calbiochem, San Diego, CA, U.S.A.). Sections were then incubated with an appropriate biotinylated secondary antibody, processed with a Vectastain ABC kit, and developed using 3'3' diaminobenzidine tetrahydrochloride as the chromagen (Vector Laboratories, U.K.). In all immunohistochemistry protocols, negative controls were performed by omitting the primary antibody, and this resulted in minimal detected signal in all cases.
Quantification of damage to axons, oligodendrocytes, and neuronal perikarya
Amyloid precursor protein-stained sections at each of the 8 coronal planes were used for quantitation of axonal damage in the following regions: corpus callosum, external capsule, internal capsule, anterior commissure, median forebrain bundle, fornix, optic tract, caudate putamen, and globus pallidus. Large regions were subdivided according to the atlas of Paxinos and Watson (1986) (e.g., corpus callosum was subdivided into forceps minor, genu of corpus callosum, cingulum, splenium of corpus callosum, and forceps major), or simply into dorsal, ventral, medial, and lateral subregions (e.g., corpus putamen). Each region was assessed across the 8 stereotaxic coronal levels used to measure neuronal perikaryal damage (Osborne et al., 1987). This provided a total of 65 individual regions of white matter to be assessed in each animal. Axonal damage in each region was identified as intense APP immunoreactivity in swollen bulbous axons (APP accumulation as a result of disrupted axonal transport) and assigned a score of 1; or, if no APP accumulation was present, a score of 0. The total APP score for the entire hemisphere (65 regions combined) was calculated from the sum of appropriate individual components. Intra-rater and inter-rater reproducibility for the presence or absence of APP are excellent (Imai et al., 2002). The same individual (blinded to treatment) performed all of the analyses in the present study. Data for the vehicle- and SPD 502–treated groups are presented as the median with 95% CL.
The distribution of neuronal perikarya with the characteristic morphology of ischemic damage was delineated onto scaled diagrams of the 8 coronal planes, as previously described (Osborne et al., 1987). The area of ischemic neuronal perikaryal damage was measured in each plane and the volume calculated. The distribution of Tau1-positive oligodendrocytes was mapped onto scaled diagrams (identical to those used in assessment of neuronal perikaryal damage), and the area measured and volume calculated as previously described (Valeriani et al., 2000). Histologic analysis and transcriptions were performed blind to the identity of the treatment. Differences between vehicle-treated and SPD 502–treated animals were determined by two-tailed unpaired Student's t-test. Data are presented as mean ± SD.
Assessment of 4-hydroxynonenal immunoreactivity
The extent of the 4-HNE immunoreactivity in sections at each of the 8 coronal planes was mapped onto line diagrams (identical to those used in assessment of neuronal perikaryal damage). The area of tissue containing 4-HNE immunoreactivity was quantified using an MCID image analyser and the volume calculated. Unpaired Student's t-test was used to assess statistical significance between the control and drug-treated groups. Data are presented as mean ± SD.
RESULTS
Axonal pathology
In the hemisphere contralateral to the occluded MCA, APP immunoreactivity was present at a diffuse, low level. In the hemisphere ipsilateral to the occluded MCA, APP immunoreactivity was intense and present in swollen axons or bulbs (Figs. 1A and 1B). APP is a fast-transported protein, and this indicates that axons in the ipsilateral hemisphere had disrupted protein transport. Axonal damage in the ipsilateral hemisphere was also indicated by the disrupted appearance of neurofilament immunoreactivity, compared with the smooth, regular pattern present in fiber tracts of the contralateral hemisphere (Figs. 1C and 1D), indicating that axons had disrupted cytoskeletal structure. Myelinated fiber tracts in both the ipsilateral and contralateral hemispheres were stained with the antibodies against the myelin proteins, PLP and MBP (Figs. 1F and 1H). The pattern of this staining was markedly disrupted in the ipsilateral hemisphere, confirming that myelinated fiber tracts were damaged (Figs. 1E and 1G).
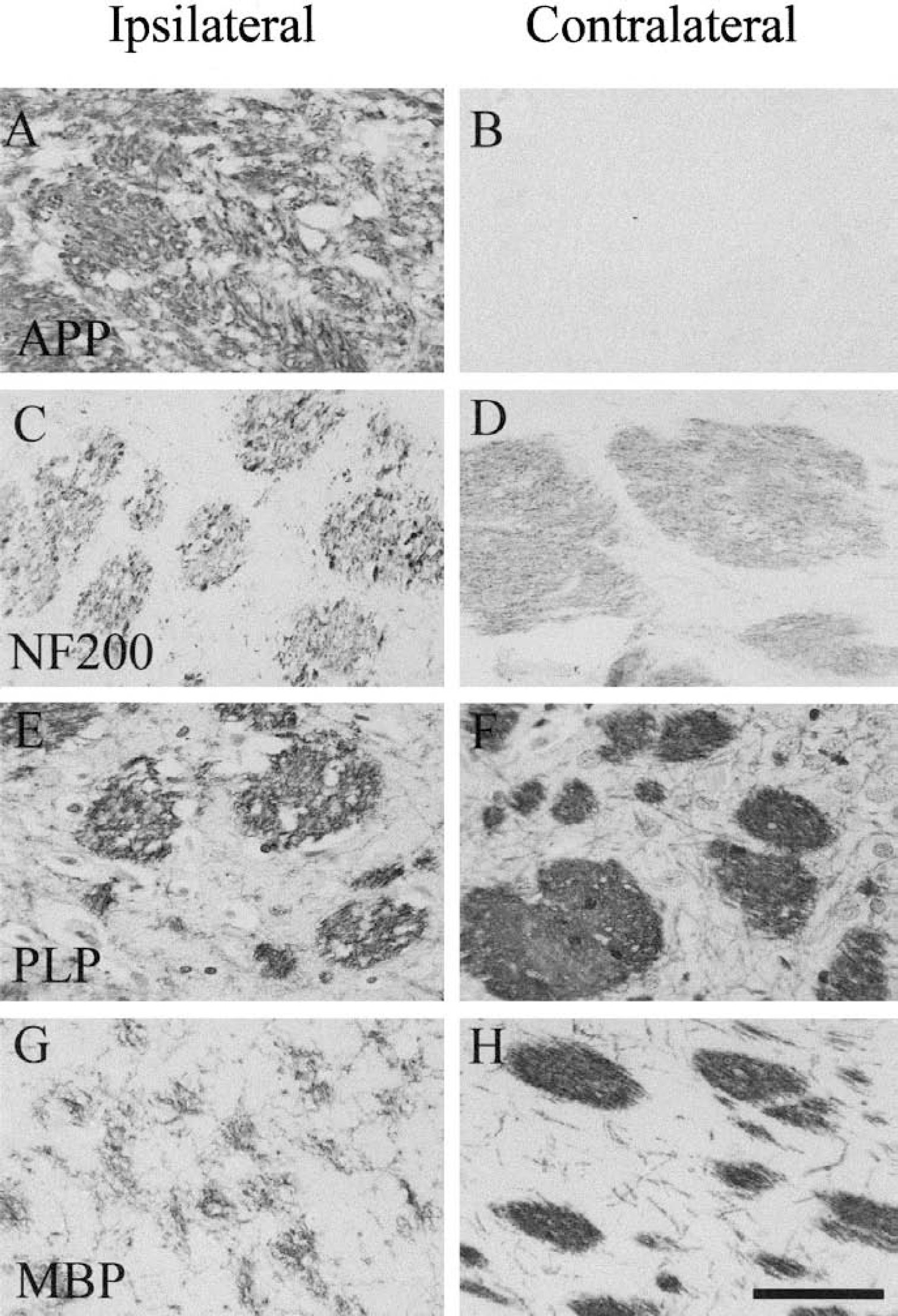
Immunohistochemical staining of myelinated fiber tracts. Representative immunostained sections from vehicle-treated rat showing subcortical myelinated fiber tracts ipsilateral and contralateral to the occluded the middle cerebral artery (MCA). APP immunoreactivity was intense and located in swollen axons or axonal bulbs in the ipsilateral hemisphere but was minimal in the contralateral hemisphere (
Quantitative analysis of axonal damage was performed on the APP-stained sections, as described previously (Imai et al., 2001). APP-immunoreactive axons were present predominantly within the myelinated fiber tracts of the caudate nucleus, internal and external capsules, median forebrain bundle, and fornix. Pretreatment with SPD 502 resulted in less axonal pathology (median 16, 95% CL 8–20) compared with vehicle treatment (median 27, 95% CL 22–30) (P = 0.006) (Fig. 2). Examination of the regional subscores suggested that the reduction in the total APP score in the SPD 502–treated group reflected modest reductions in the majority of myelinated tracts examined rather than a marked reduction in a single region.
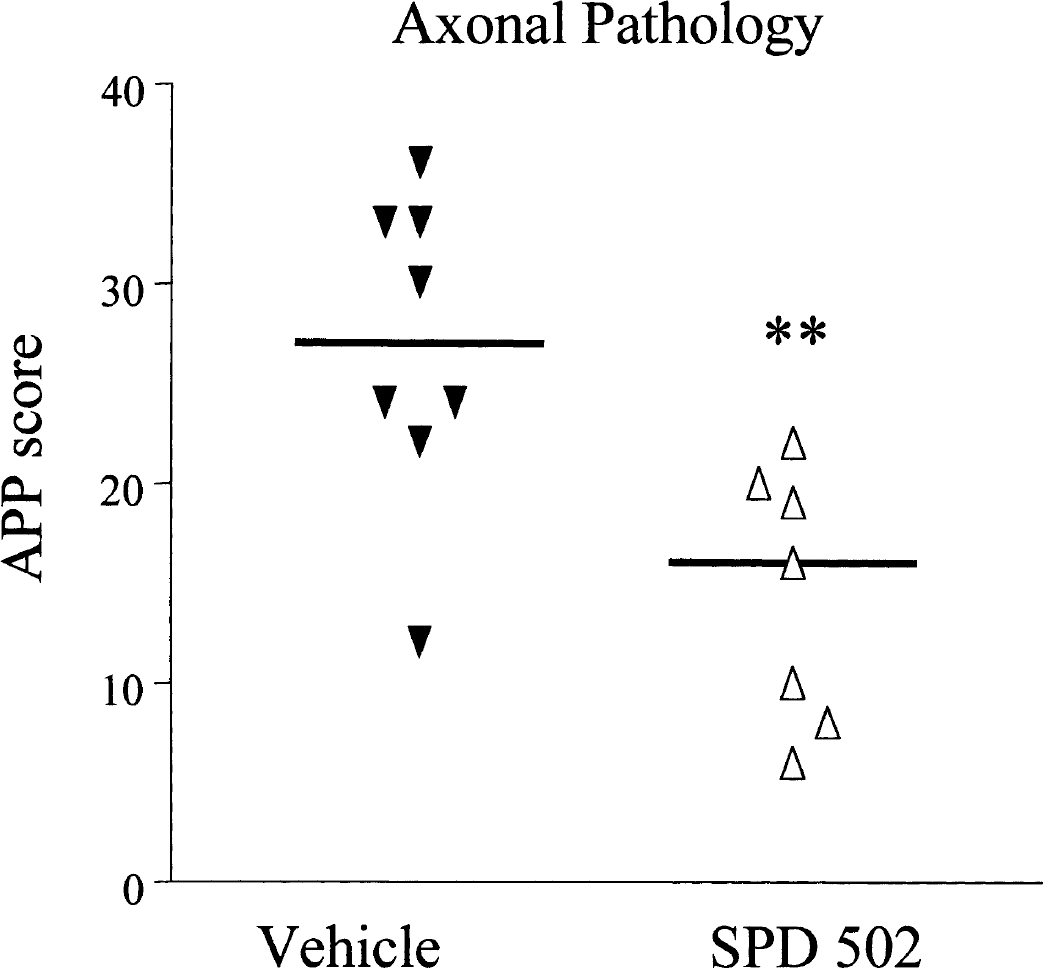
Quantitative assessment of axonal damage: effects of SPD 502. The total APP scores were derived from scoring of presence or absence of APP-positive axons at the 65 predefined neuroanatomic locations in vehicle- and SPD 502–treated groups. SPD 502 significantly reduced axonal damage detected by APP accumulation compared with the vehicle-treated group. Each point represents an individual animal, and the horizontal bar indicates the median. APP, amyloid precursor protein. **P < 0.01, two-tailed Mann-Whitney test.
Oligodendrocyte pathology
Tau1 immunoreactivity in the hemisphere contralateral to the occluded MCA had a diffuse pattern of staining, largely localized to the neuropil or axonal fibers, with minimal staining of cell bodies in either grey or white matter. In the ipsilateral hemisphere, Tau1-positive cells with the characteristic morphology of oligodendrocytes, featuring a thin rim of cytoplasm and small soma, were present in grey and white matter, as described previously (Dewar and Dawson, 1995; Irving et al., 1997; Valeriani et al., 2000).
The anatomic extent of oligodendrocyte pathology in the hemisphere ipsilateral to the occluded MCA was reduced in the SPD 502–treated group (54.5 ± 19 mm3) compared with the vehicle-treated group (79 ± 30 mm3), but this reduction did not reach accepted significance levels (P = 0.09) (Fig. 3). However, the anatomic extent of oligodendrocyte pathology in the cerebral cortex was significantly lower in the SPD 502–treated compared with the vehicle-treated animals. There was no difference between the two groups in the extent of oligodendrocytes pathology in subcortical regions.
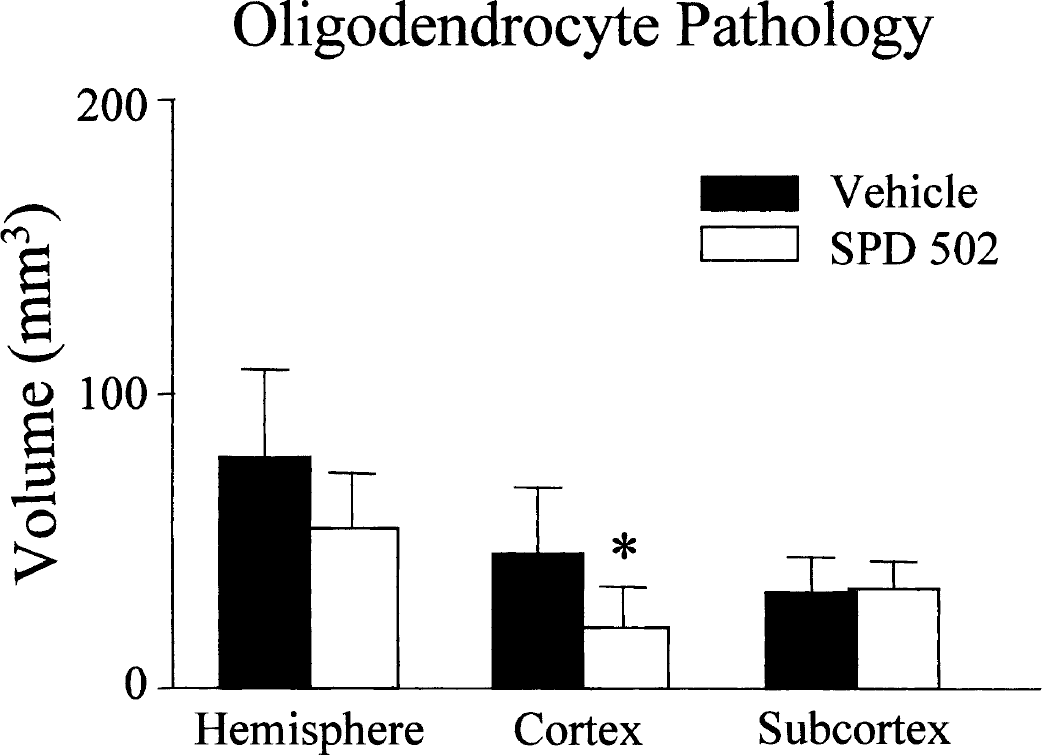
Effects of SPD 502 on the anatomic extent of oligodendrocyte pathology. The anatomic extent of oligodendrocyte ischemic pathology was determined by quantifying the volume of tissue that contained Tau1-positive oligodendrocytes in the hemisphere, cerebral cortex, and subcortex. SPD 502 significantly reduced the volume of oligodendrocyte pathology only in the cerebral cortex. Data are expressed as mean + SD. Vehicle, n = 8; SPD 502, n = 7. *P < 0.05, two-tailed Student's t-test.
Neuronal perikaryal damage
In the cerebral hemisphere ipsilateral to the occluded MCA, the cerebral cortex, caudate nucleus, and anterior hypothalamus clearly exhibited the characteristics of ischemic neuronal necrosis (Valeriani et al., 2000) whereas the contralateral hemisphere showed no evidence of neuronal pathology. The anatomic extent of neuronal damage was significantly reduced in the cerebral hemisphere and cerebral cortex in the SPD 502–treated group compared with the vehicle-treated group (Fig. 4). In contrast, there was no difference between the animals treated with vehicle or SPD 502 in subcortical regions (P = 0.3772) (Fig. 4).
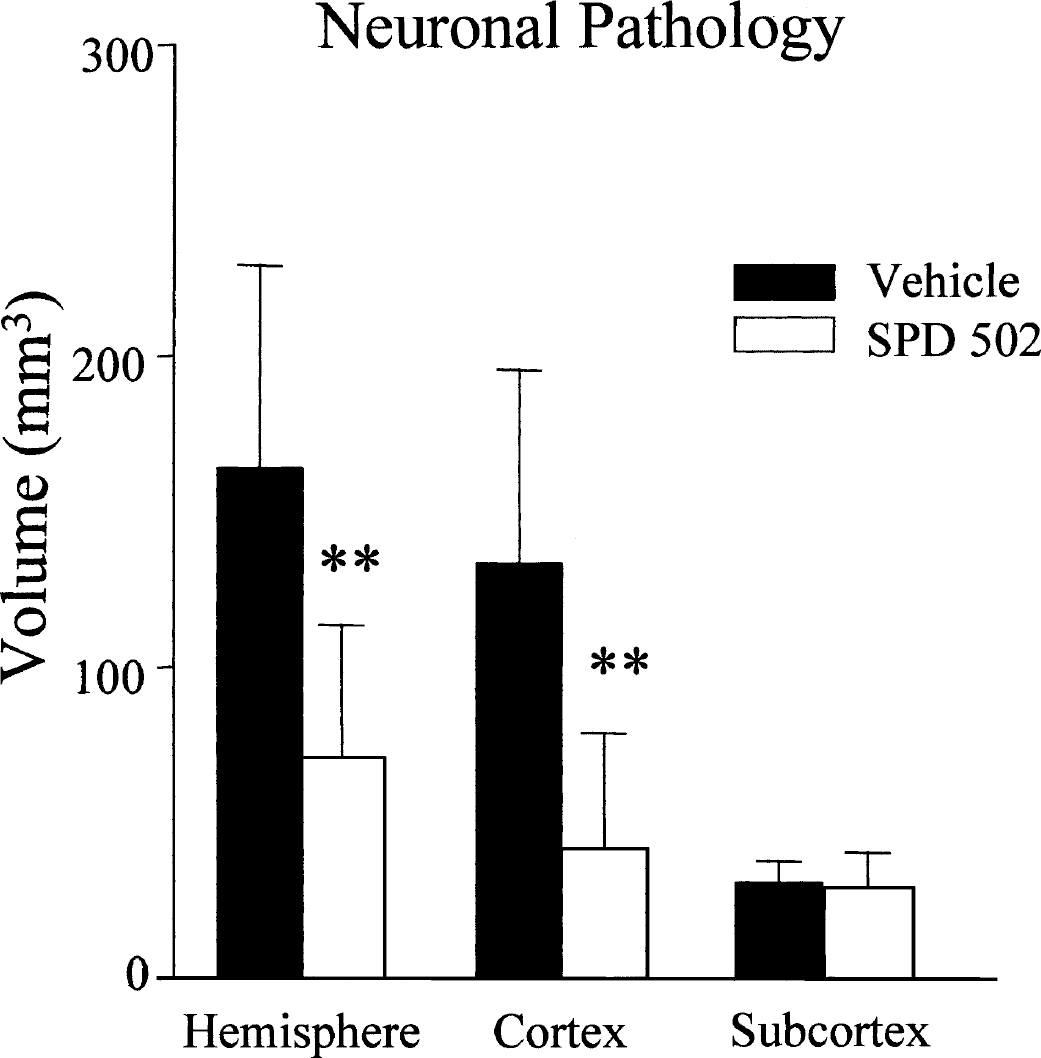
Effects of SPD 502 on the volume of neuronal necrosis. The anatomic extent of neuronal necrosis was determined from the hematoxylin- and eosin-stained sections. SPD 502 significantly reduced the volume of neuronal necrosis in the hemisphere and cerebral cortex but not in the subcortex. Data are expressed as mean + SD. Vehicle, n = 8; SPD 502, n = 7. **P < 0.01, two-tailed Student's t-test.
Extent of 4-hydroxynonenal immunoreactivity
4-Hydroxynonenal immunoreactivity was minimal in the contralateral hemisphere (Fig. 5A). 4-HNE immunoreactivity was increased in neuronal cell bodies, axons, and glial cells throughout the territory of the occluded MCA (Fig. 5B). A boundary of 4-HNE immunoreactivity was clearly demarcated. In the cerebral hemisphere and the cerebral cortex the anatomic extent of 4-HNE immunostaining was significantly less in the SPD 502–treated than in the vehicle-treated group. However, there was no significant difference in the subcortex between the two groups in the volume of 4-HNE immunoreactivity (Fig. 5C).
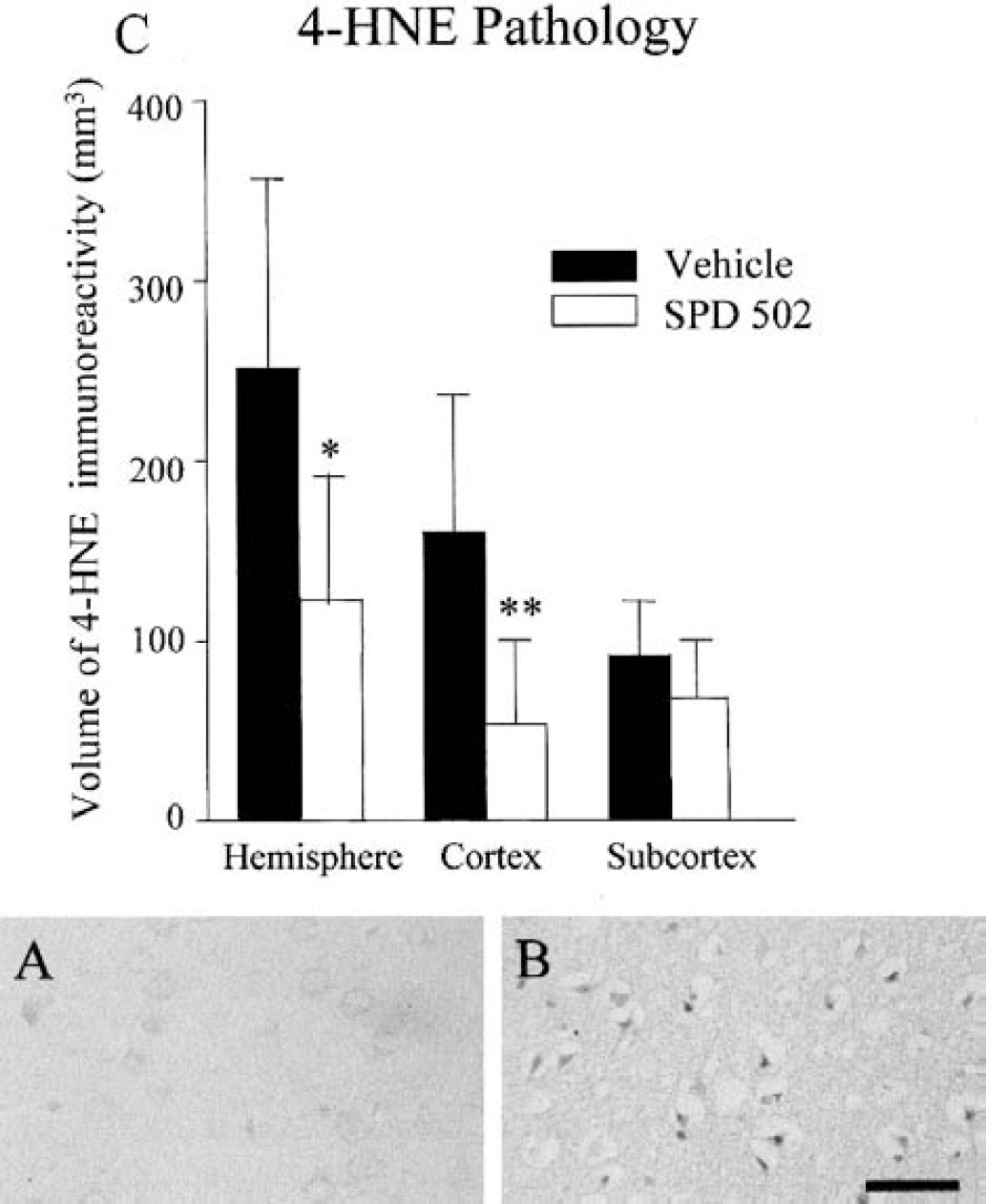
AMPA blockade reduces the extent of oxidative stress after transient cerebral ischemia in the rat. 4-Hydroxynonenal (4-HNE) immunoreactivity was minimal in the hemisphere contralateral to the occluded middle cerebral artery
DISCUSSION
AMPA receptor antagonists have been shown in numerous studies to protect neuronal perikarya from damage in focal cerebral ischemia. The neuroprotective effects of AMPA receptor blockade have been demonstrated with competitive and noncompetitive AMPA antagonists in a variety of models of focal ischemia (Bullock et al., 1994; Gill, 1994; Kawasaki-Yatsugi et al., 2000; Turski et al., 1998). In the present study we demonstrate that the isatine oxime, SPD 502, which has excellent physiochemical properties, reduces neuronal perikarya pathology after transient MCA occlusion. More important, we provide the first evidence that an AMPA receptor blocker protects axons and oligodendrocytes as well as neuronal perikarya in experimental focal cerebral ischemia.
AMPA receptors are present not just on neurones but also on oligodendrocytes. AMPA receptor-mediated excitotoxicity induces not just neuronal damage but has also been implicated in oligodendrocyte cell death (Alberdi et al., 2002; Fowler et al., 2001; Gallo and Russell, 1995; Matute et al., 1997; McDonald et al., 1998; Pantneau et al., 1994; Sanchez-Gomez and Matute, 1999; Tekkok and Goldberg, 2001; Yoshioka et al., 1995). The cytoprotective effects of AMPA receptor blockade in focal ischemia may reflect not just attenuation of ion fluxes at the cellular level, but also other beneficial effects of blocking this receptor.
Ischemic damage is a consequence of the mismatch between metabolic demand and substrate delivery. AMPA receptor blockade reduces glucose use in both grey and white matter (Browne et al., 1994), and any reduction in metabolic demand in ischemic tissues may contribute to the cytoprotective effects of SPD 502. Enhanced substrate delivery (i.e., restoration of blood flow) has been shown to protect axons, oligodendrocytes, and perikarya (Valeriani et al., 2000). It should be noted that, in the present study, even pretreatment with the AMPA antagonist failed to reduce oligodendrocyte and axonal pathology in deep subcortical areas, where the reductions in cerebral blood flow after MCA occlusion are most pronounced (Komjati et al., 2001). Activation of AMPA/kainate receptors stimulates production of oxygen radicals in neurons and glia (Bae et al., 2002; Carriedo et al., 2000; Matsuoka et al., 1999; Mattson et al., 1995). The volume of tissue in which cells were immunopositive for the lipid peroxidation by-product, 4-HNE, was markedly reduced by SPD 502 treatment. 4-HNE is toxic not only to neuronal perikarya but also to oligodendrocytes and axons (McCracken et al., 2000). Reductions of oxidative stress after MCA occlusion with SPD 502 may have contributed to the attenuation of ischemic damage observed in axons, oligodendrocytes, and neuronal perikarya.
The different magnitudes of the volumes of neuronal perikaryal damage, oligodendrocyte pathology, and 4-HNE immunoreactivity in vehicle-treated animals provide evidence of differential sensitivity to ischemia. The greater anatomic extent of 4-HNE immunoreactivity relative to neuronal perikaryal damage is consistent with 4-HNE immunoreactivity reflecting both lethal and sub-lethal lipid peroxidation. The lesser anatomic extent of oligodendrocyte pathology relative to neuronal perikaryal damage may indicate that oligodendrocytes are less sensitive to ischemia than are neuronal perikaryal. In contrast, there is little evidence of differential sensitivity to the AMPA antagonist of the different markers of ischemic pathology. Proportionately similar reductions were observed with AMPA receptor blockade in the volumes of 4-HNE immunoreactivity, neuronal perikaryal damage, and oligodendrocyte pathology.
The present study provides “proof of concept,” that an AMPA receptor antagonist protects not just neuronal perikarya but also axons and oligodendrocytes from transient cerebral ischemia. Drugs that protect gray matter and white matter from ischemic damage should offer significant advantages in the treatment of stroke over drugs that protect only neuronal perikarya (Dewar et al., 1999). Crucial major issues, however, about the pathophysiology of axonal and oligodendrocyte pathology remain to be resolved. The concept of the ischemic penumbra and core is well established for gray matter (Heiss, 2000; Sharp et al., 2000), but has never been extended to white matter. The inability of an AMPA antagonist or antioxidant (Imai et al., 2001), however, to protect axons and oligodendrocytes in areas with the most pronounced reductions in CBF (Komjati et al., 2001) points to the existence of salvageable and nonsalvageable zones in white matter. While neuronal perikaryal pathology is essentially complete 24 hours after MCA occlusion (Jones et al., 1981), the temporal evolution of axonal and oligodendrocyte pathology has been minimally investigated. The continuing development of edema (with the disproportionately greater accumulation of water in white matter) beyond 24 hours could, potentially, lead to further slowly evolving damage in white matter. It will be of interest to determine if the protection afforded to white matter by AMPA receptor blockade at 24 hours is also observed at later times. Unlike gray matter, the window of therapeutic opportunity after the onset of ischemia has never been defined for white matter. The lower levels of CBF that are observed in ischemic white matter, compared with adjacent areas of gray matter (Komjati et al., 2001), complicate the definition of therapeutic window by delaying delivery of the protective agent to ischemic white matter. In the present, proof-of-principle study, pretreatment with the AMPA antagonist was used to avoid such a complication. The superiority of total brain protection (i.e., protection of all cellular elements in the CNS) over neuroprotection (i.e., the protection of only neurons) for stroke will become more apparent once the same fundamental questions are answered in relation to the pathophysiology of white matter ischemia as have been for gray matter.
Footnotes
Acknowledgements
The authors acknowledge the excellent technical assistance provided by staff at the Wellcome Surgical Institute, Glasgow.
