Abstract
By means of a novel 18F-fluoro-deoxyglucose PET method designed for cognitive activation imaging in the baboon, the large-scale neural network involved in visual recognition memory in the nonhuman primate was mapped for the first time. In this method, the tracer is injected in the awake, unanesthetized, and unrestrained baboon performing the memory task, and brain imaging is performed later under light anesthesia. Brain maps obtained during a computerized trial-unique delayed matching-to-sample task (lists of meaningless geometrical patterns and delay > 9 seconds) were statistically compared pixel-by-pixel to maps obtained during a specially designed visuomotor control task. When displayed onto the baboon's own anatomic magnetic resonance images, foci of significant activation were distributed along the ventral occipitotemporal pathway, the inferomedial temporal lobe (especially the perirhinal cortex and posterior hippocampal region), and the orbitofrontal cortex, consistent with lesion, single-unit, and autoradiographic studies in monkeys, as well as with activation studies in healthy humans. Additional activated regions included the nucleus basalis of Meynert, the globus pallidus and the putamen. The results also document an unexpected left-sided advantage, suggesting hemispheric functional specialization for recognition of figural material in nonhuman primates.
Although lesion (Mishkin, 1990) and single-unit recording (Brown, 1996) studies have long identified brain structures that are involved in the visual recognition memory in nonhuman primates, such studies cannot reveal the entire activated large-scale network in a single experiment. In normal man, maps of the neural networks involved in specific cognitive functions can be generated with PET or fMRI, complementing the body of knowledge acquired from neuropsychology (Petersen et al., 1988; Dolan and Fletcher, 1999). In disease states, however, impaired performance and heterogeneity in lesion location complicate the interpretation of such studies in terms of neural networks engaged and synaptic reorganization (Becker et al., 1996). Apart from allowing the performance of such studies after controlled brain lesions, cognitive activation mapping in the nonhuman primate would also permit the examination of important issues such as the relationships between large-scale activations and single-unit recordings, and the single-subject cytoarchitectonic correlates of focal activations-none of which is possible in man.
The PET activation paradigm with H215O has been implemented in the conscious monkey (Perlmutter et al., 1991; Takechi et al., 1994; Tsujimoto et al., 1998), but memory investigations have not been reported so far, presumably because of the constraints involved. Indeed, in Perlmutter's method, the supine position would have been unsuitable for cognitive experiments. In the Japanese approach, the monkey is seated within a special PET device tilted 75° from the vertical plane, but potential interference from the anesthesia (performed to prepare the animal), together with the long recovery period in a restrained position before the behavioral session can begin, would be undesirable for cognitive paradigms. fMRI has been recently applied to the awake behaving monkey according to two different paradigms. In the first, the “sphinx” position used to fit the animal into the magnetic bore is a major obstacle to cognitive experimentation involving forelimb responses (Stefanacci et al., 1998; Dubowitz et al., 1998). In the other, the animal is seated within the vertically oriented bore, allowing visual stimulations in the anesthetized state (Logothetis et al., 1999). Applied to the unanesthetized animal, this methodology has been hampered by head motion, while noise caused by fast switching of the gradient coil might induce stress and distraction. However, it is likely that these technical problems should find solutions in the near future. To study visual recognition memory, we have developed a novel PET activation method using 18F-fluorodeoxyglucose (18FDG). This approach is the transposition to PET of the classic Sokoloff's 14C-2-deoxy-D-glucose (14C-2DG) autoradiographic technique (Sokoloff et al., 1977) that has long been successfully applied to cognitive activation studies in monkeys (Macko et al., 1982; Macko and Mishkin, 1985). Unlike the autoradiographic method, however, the PET method allows repeated studies in the living animal. As compared to H215O, one considerable advantage of this approach is that the radiotracer accumulates and fixes in brain tissue for 30 minutes (Phelps et al., 1979), so that imaging can be performed in the anesthetized animal once the behavioral experiment is completed (Chavoix and Baron, 1995; Eberling et al., 1995). Thus, with this method, brain scanning is dissociated from task performance which takes place with an awake, unrestrained animal in its usual testing environment (Fig. 1).
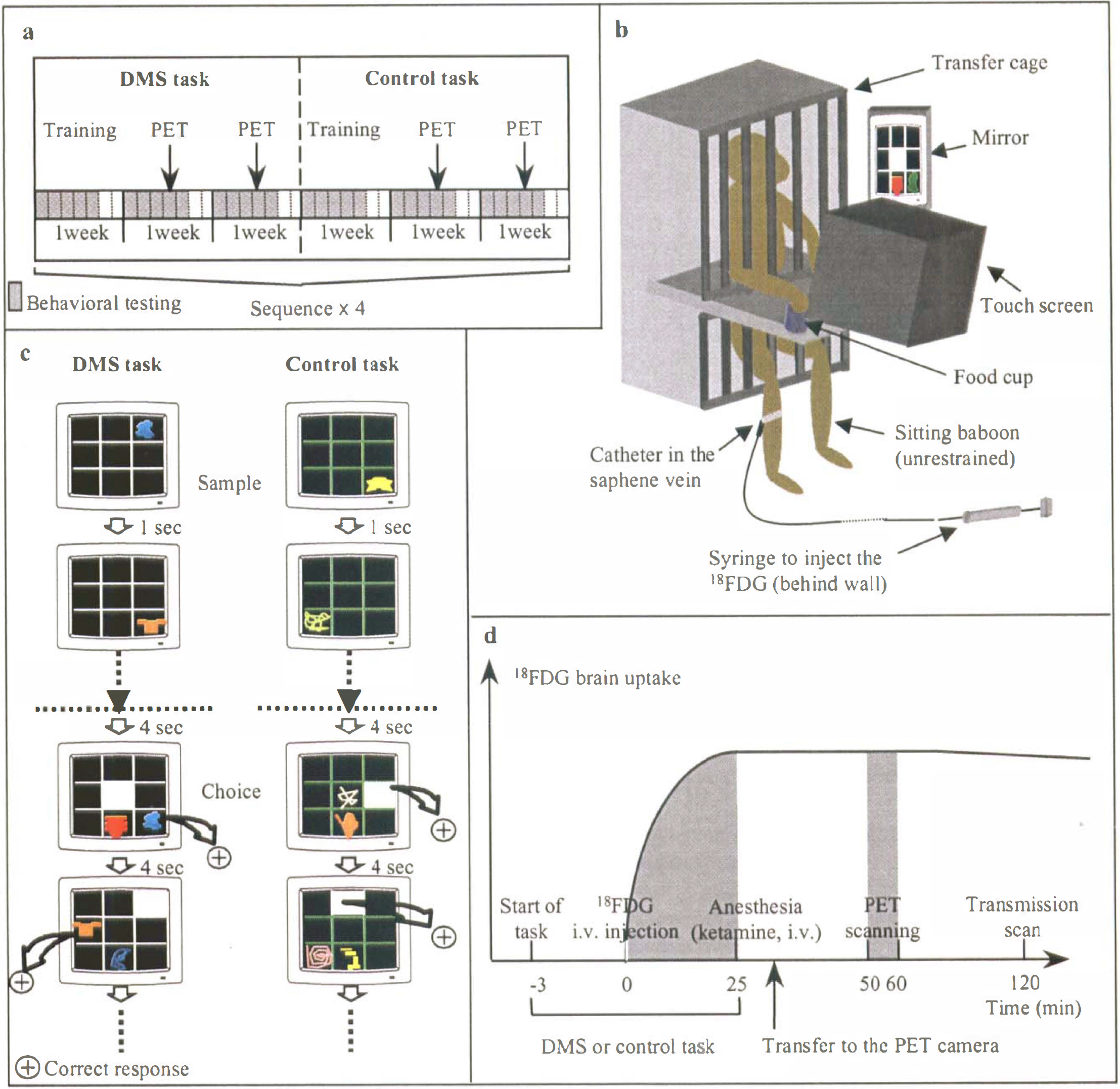
To highlight the neural network involved in long-term visual recognition memory, we compared the brain maps obtained during a computerized visual delayed matching-to-sample (DMS) task with trial unique stimuli, to those obtained during a visuomotor control task (Fig. 1). The DMS, which requires the animal to recognize previously presented item(s) from distractor(s), is a classic task to assess recognition memory in monkeys (Mishkin and Delacour, 1975). The present paradigm was designed so that, relative to the control task, “conscious” encoding (which entails visual discrimination), storage and retrieval, would be engaged only during the DMS task.
MATERIALS AND METHODS
General procedure
The paradigm used for this study was in accordance with institutional guidelines for the animal's care. It is illustrated in Fig. 1. The experimental period extended over 24 weeks during which 8 PET scans per task (i.e., the DMS and the control tasks) were acquired from one young adult male baboon (Papio anubis, 16 kg) (Fig. 1a).
Behavioral paradigm
Testing was conducted in a sound-proof, light-softened room equipped with a computerized test apparatus assembled in-house (Fig. 1b). A white-noise generator masked extra noises, and the monkey's behavior was monitored by a video system. The baboon faced a color monitor fitted with a touch-sensitive screen (27 × 20 cm, Sony CPD-1420E Trinitron) connected to an IBM computer monitor operated from a contiguous control room. The computer was connected to an automatic dispenser (BRS/L VE, Laurel, MD) that, for each correct answer, triggered a brief sound and delivered single 190 mg banana pellets (Noyes, Inc., Lancaster, NH) onto a food cup located near the bottom right of the screen. Visual stimuli displayed onto the screen were meaningless geometric drawings randomly drawn from a pool of 800 different samples. The baboon was first trained on a visual DMS task with trial-unique stimuli which required the baboon to memorize single stimuli. On each trial, the baboon had to touch the drawing appearing pseudorandomly with respect to the 9 locations of a 3 × 3 grid; then, after a 1-second delay, the previous drawing and a new one were simultaneously shown, again pseudorandomly located on the grid, and the baboon was rewarded by touching the previously seen stimulus. After a 4-second intertrial interval, a new trial appeared with a new pair of drawings. For rule learning, the baboon was trained daily on this task, to reach a criterion of 90% correct responses over 90 consecutive trials. Then, the monkey was trained successively on the two following activation tasks: 1) a visual trial-unique DMS task for memory assessment and 2) a visuomotor control task (Fig. 1c). Both tasks were intended to share most cognitive and motor processes, such as visual attention and perception, vigilance and alertness, auditory stimulation, reward processes, motor activity, and feeding behavior. To comply with the constraints of PET, the classic DMS task was adapted as follows: 1) to enhance memory load, we used the list, rather than the delay, condition; each daily session consisted of 35 lists of 5 drawings each; for each list, the 5 drawings were successively shown, followed by the successive presentation of the choices; and 2) to make the 2 tasks as comparable as possible, one neutral stimulus (a white square) was added in the choice phase (Fig. 1c). The animal was successfully trained on both tasks to high performance over 20 days (84.3 ± 3.8% and 98.7 ± 1.6% accuracy for the DMS and the control tasks, respectively). This better performance (P < 0.05) in the control task was inevitable, but implied a proportionally higher number of moves for screen-touching (feeding behavior), auditory stimulation (from the pellet dispenser), and reward processes than in the DMS task. To control for this, the pellet dispenser was deliberately automatically disabled in 15% of correct responses for the control task during the 24-weeks PET experimental phase, a procedure to which the baboon quickly became accustomed. On PET days the baboon performed 25.4 ± 6 and 25.7 ± 5.8 lists during the DMS and the control tasks, respectively (no significant difference). The PET examinations were always performed at the same time of day (around 2 pm) as the training, to limit variability in behavioral and feeding processes.
PET procedure an image analysis
PET data of 18FDG brain uptake were collected according to a 63-cut three-dimensional volume axial acquisition with the high-resolution PET device ECAT HR+ with isotropic resolution of 4.6 × 4.2 × 4.2 mm. MR imaging was also performed according to a T1-weighted three-dimensional volume sagittal acquisition (spoil gradient; time of echo = 9 ms, time of repetition = 48 ms, flip angle = 29°, number of scans: 124, thickness: 0.7 mm), using a GE Signa 1.5 scanner with a 12.7-cm (5-inch) general-purpose receive-only surface coil. This volume was edited using a dedicated software (Vérard et al., 1997). PET data were resliced into 105 coronal cuts (1.45 × 1.45 × 1.45 mm voxels, x, y, z), using the display and processing ECAT software CAPP. Coronal MR images, obtained by interpolation from the sagittal three-dimensional volume acquisition, were then coregistered with the PET images using the AIR software (Woods et al., 1993), and the PET data were masked by the edited MR image. The changes in 18FDG uptake between PET data of the 8 “memory” and 8 “control” conditions were examined voxel by voxel using the SPM96 software (Welcome Dept of Cognitive Neurology, Institute of Neurology, London, U.K.), which uses the general linear model and computes the t-statistic on a voxel basis (Friston et al., 1995). After smoothing with a 6 mm Gaussian filter, the data were normalized for global activity. The comparison (memory minus control) was made using the single subject with replication of conditions analysis of covariance. The voxels exceeding a statistical criterion of P < 0.01 (uncorrected), corresponding to a Z-score of 2.33 or greater, were then superimposed on the corresponding coronal MRI planes of the same baboon (without applying spatial deformation), allowing anatomic identification of the activated areas with the help of Mishkin's stereotaxic atlas of the macaque (unpublished), an atlas of the cortical gyri of the baboon not being available. This statistical cut-off was elected taking into account both the small size of this baboon's brain (n = 56098 voxels) relative to the human brain, and previous knowledge about the brain structures involved in this visual memory processes including 2-DG autoradiographic studies (Macko et al., 1982; Macko and Mishkin, 1985; Friedman and Goldman-Rakic, 1988).
RESULTS
Results are listed in Table 1 and illustrated on coronal MRI planes in Fig. 2. Foci of significant activation were mostly found on the left side, namely in the temporal, prefrontal, parietal, and cingulate cortices as well as in several subcortical areas. Significant bilateral activation were found within the occipitotemporal pathway only.
List of the significantly activated areas with the corresponding z-scores, P values, and location in Fig. 2
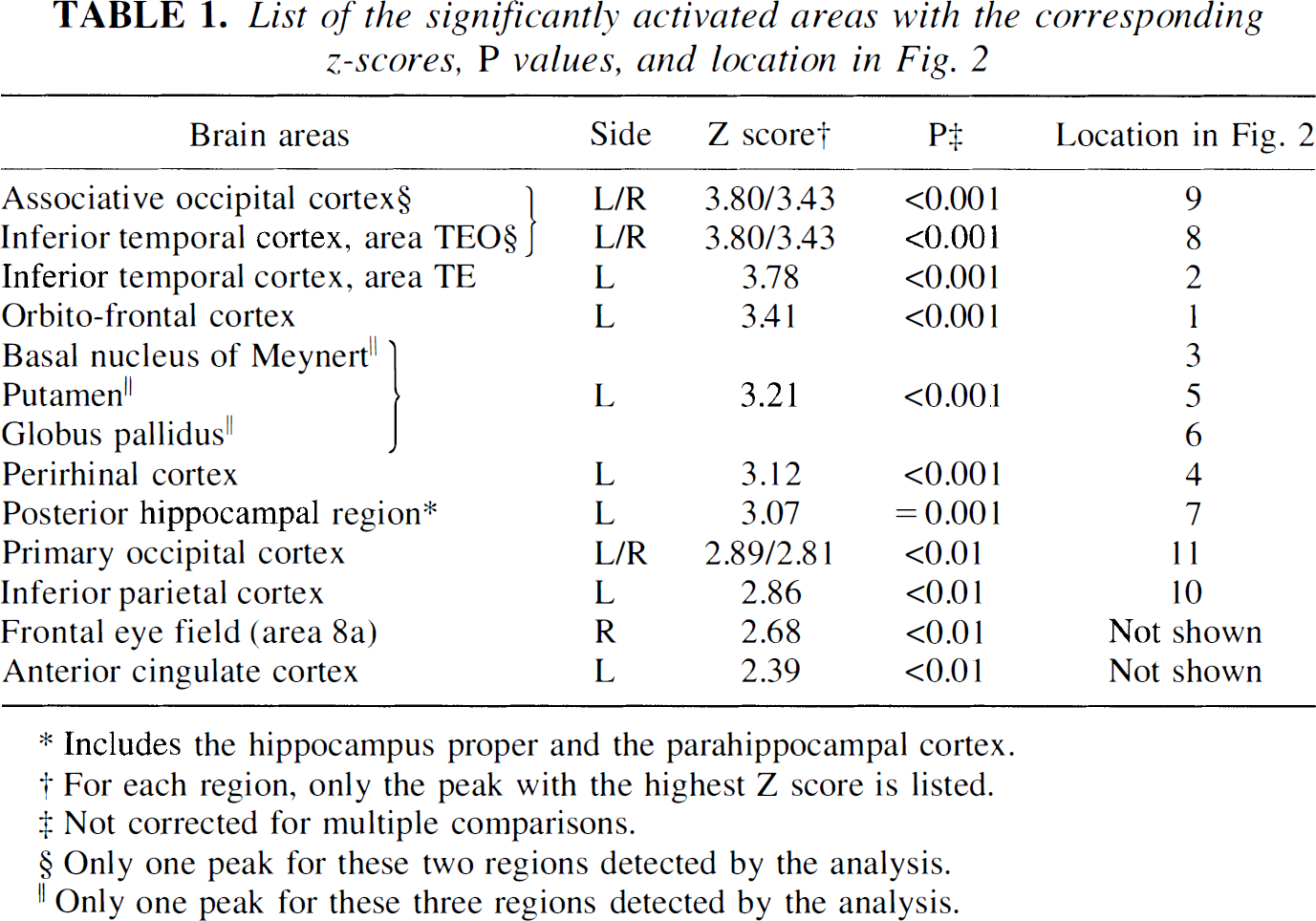
Includes the hippocampus proper and the parahippocampal cortex.
For each region, only the peak with the highest Z score is listed.
Not corrected for multiple comparisons.
Only one peak for these two regions detected by the analysis.
Only one peak for these three regions detected by the analysis.
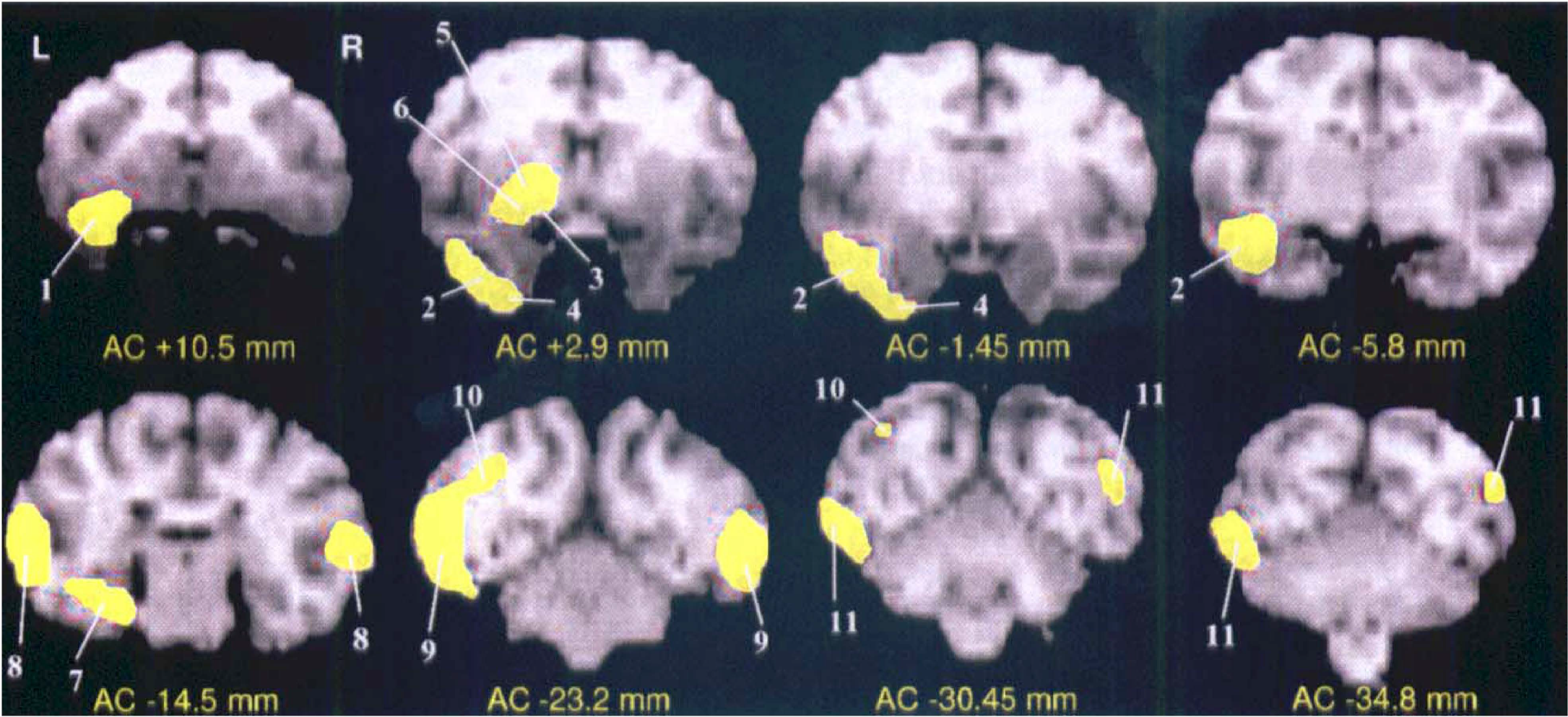
Significant activations as detected by PET and 18F-fluorodeoxyglucose, for the task comparison (delayed matching-to-sample [DMS] minus control). SPM map of the significant voxels superimposed on the coregistered T1-weighted MRI of the same baboon, shown for eight illustrative coronal levels from the anterior commissure (AC). The yellow areas represent the brains regions with significantly activated voxels during visual recognition memory (DMS task) as compared to the visuomotor control task, at the P < 0.01 level, anatomically identified by numbers from 1 to 11 (Table 1). L, left; R, right.
DISCUSSION
In this novel work, we were able to reveal in the intact nonhuman primate and in a single experiment the entire network of brain structures activated by visual recognition memory. With reference to earlier lesion and single-unit recording studies of visual identification (Macko et al., 1982; Macko and Mishkin, 1985) and recognition memory (Mishkin, 1990; Brown, 1996), we found: 1) predictable activations of the ventral (occipitotemporal) visual pathway up to limbic structures and ventromedial prefrontal cortex (i.e., orbitofrontal and anterior cingulate), as well as of the nucleus basalis of Meynert; but 2) more surprisingly, no activation of the thalamus and the entorhinal cortex; and, conversely, 3) significant activations of the putamen and pallidum, and left hemisphere preeminence, notably for the temporal lobe activations.
Our behavioral paradigm was specially designed to allow a categorical comparison of the 18FDG images to highlight visual declarative memory processes (i.e., encoding, storage, and recognition). Thanks to extensive preliminary testing, the DMS and control tasks were successfully equalized for common cognitive and motor processes (Fig. 1), as shown by the lack of significant activation (even at the P < 0.05 level, data not shown) in structures related to auditory stimulation, motor responses and reward, such as the primary auditory and motor cortices and the amygdala, respectively.
Previously, Macko et al. (1982, 1985) used 14C-2DG autoradiography to study two groups of monkeys, one performing a learned visual pattern discrimination task, and the other a “passive” visual task (viewing a rotating drum with a high-contrast geometric pattern). The right hemisphere was visually deafferented in all monkeys, so as to allow comparison of the local cerebral glucose utilization between the activated and the “blind” hemispheres. The results revealed activation of the ventral visual pathway, including the primary and associative visual cortices, the inferior temporal cortex (areas TEO and TE), and the ventral portion of area TG (representing part of the anterior perirhinal cortex), in both tasks. The unexpected lack of clear-cut differences in metabolic maps between the “memory”, as compared to the “passive”, conditions was apparently attributable to differences in several confounding cognitive and motor processes between the two tasks, and to the fact that different animals had to be used in the two tasks. The fact that in our study this same ventral visual pathway was highlighted suggests that it was more engaged in the DMS than in the visuomotor control task, presumably reflecting greater involvement of “visual” processes such as discrimination in the encoding, storage, and recognition of complex visual stimuli as also observed in man (Roland and Gulyas, 1995; Schacter and Wagner, 1999; Kuskowski and Pardo, 1999). Among these areas, the perirhinal cortex was especially markedly activated, consistent with its major involvement in visual recognition memory, as shown by lesion studies in macaques (Meunier et al., 1993) and fMRI in visual encoding in man (Brewer et al., 1998).
Besides the ventral visual pathway, activation also concerned the ventromedial prefrontal cortex, in agreement with its involvement in recognition memory (Bachevalier et al., 1991); Macko et al. (1982, 1985) also reported activation in this area in their visual tasks. We also found significant activation in the posterior hippocampal region, consistent with its implication in nonspatial-and not only spatial-memory, as is also the case for the parahippocampal cortex (Suzuki et al., 1993; Colombo et al., 1998). Previously, 2DG studies in the macaque also revealed activation in the posterior hippocampus during nonspatial working memory tasks (Friedman and Goldman-Rakic, 1988), and hippocampal-parahippocampal activation has been repeatedly documented in man during memory processes (Roland and Gulyas, 1995; Schacter and Wagner, 1999; Tulving and Markowitsch, 1997). Relative to these findings, the lack of significant activation of the entorhinal cortex and the thalamus was less predictable, considering their role in recognition memory (Aggleton and Mishkin, 1983; Leonard et al., 1995; Suzuki et al., 1997). Regarding the entorhinal cortex, this may reflect the following: 1) its input is from multimodal areas (Insausti et al., 1987), as compared to visual areas TE and TEO for the perirhinal cortex (Suzuki, 1996), and thus it may be less specifically involved in visual tasks (Chafee and Goldman-Rakic, 1998); and 2) it is the main, but not the unique, relay between the perirhinal cortex and the hippocampus (Suzuki and Amaral, 1990). Consistent with our findings, no significant activation of the entorhinal cortex during a visual DMS task was observed in a preliminary 2DG autoradiographic study in the monkey (published in abstract) (Davachi et al., 1998). Regarding the thalamus, our results are consistent with the fact that significant activation of this structure has not been reported in non-verbal recognition memory tasks in humans (Desgranges et al., 1998). Finally, these results are also consistent with the fact that visual recognition memory is more severely impaired after perirhinal than entorhinal ablation (Meunier et al., 1993) and after rhinal cortex than thalamic lesion (Parker et al., 1997).
Activation of the nucleus basalis of Meynert as part of a subcortical cluster that also engulfed the ventral putamen and the globus pallidus is not surprising in our task considering its involvement in general attentional processes and its modulatory effects on recognition memory (Voytko, 1996). The other two structures in this cluster are not considered part of the classic visual recognition memory circuit. However, the ventral striatum has been implicated in visual discrimination or short-term memory (Rolls, 1994), and in habit memory which may have been operative in our DMS task. Finally, activation of the inferior parietal cortex and frontal eye field may reflect greater involvement of visuospatial attention and exploration (Chafee and Goldman-Rakic, 1998) in the DMS than the control task.
The striking left hemisphere advantage (present even at P < 0.05, data not shown), especially for the anterior temporal and the subcortical areas, was unexpected since unilateral brain damage induces only mild or no memory impairment. Hemispheric asymmetry has not been reported in previous 2-DG memory studies, though it would have been missed in most because of the procedures used, e.g., the use of a visually deafferented hemisphere as control (Macko et al., 1982; Macko and Mishkin, 1985) or the measurements done in one hemisphere only (Friedman and Goldman-Rakic, 1988). Interpretation for this new observation can only be speculative at this stage, but would be in line with growing evidence for hemispheric specialization in the nonhuman primate (Hamilton and Vermeire, 1988; Fagot and Deruelle, 1997; Gannon et al., 1998). Preferential involvement of the left hemisphere for visual analytic processes has been reported both in man (Kawashima et al., 1998; Vandenberghe et al., 1996; Sergent et al., 1992; Shen et al., 1999) and in the nonhuman primate (Fagot and Deruelle, 1997; Vermeire et al., 1998), which may have relevance to our finding. Indeed, studies in man have shown preferential or exclusive left occipitotemporal activation during object categorization (Sergent et al., 1992) and visual pattern discrimination (Kawashima et al., 1998), respectively. The activation paradigm therefore opens new avenues in the investigation of functional hemispheric specialization in the nonhuman primate.
Footnotes
Abbreviations used
Acknowledgements
The authors thank O. Quinton for help in the use of the software for MRI volume edition, M.C. Petit-Taboué for precious advice and help in the data analysis, A. Parent, A. Rossard, and G. Huguet for their technical assistance during the behavioral and PET experiments, and V. Beaudouin for help in the PET scanning procedure. The authors also thank Dr. M. Mishkin, Prof. R.S.J. Frackowiak, and Dr. M. Meunier for reviewing earlier versions of this manuscript.
