Abstract
Assessment of serotonin release in the living brain with positron emission tomography (PET) may have been hampered by the lack of suitable radioligands. We previously reported that fenfluramine caused a dose-dependent reduction in specific binding in monkeys using a classical displacement paradigm with bolus administration of [11C]AZ10419369. The aim of this study was to confirm our previous findings using an equilibrium approach in monkey. A total of 24 PET measurements were conducted using a bolus infusion protocol of [11C]AZ10419369 in three cynomolgus monkeys. Initial PET measurements were performed to assess suitable
Introduction
Positron emission tomography (PET) can be used to assess alterations in neurotransmitter levels
Positron emission tomography imaging of endogenous serotonin (5-HT) release has been attempted, but may have been hampered by the lack of suitable radioligands (Paterson et al, 2010). The so far most promising radioligands for imaging of serotonin release are targeting the 5-HT1A receptor, and though initial animal studies were encouraging for the antagonist [18F]MPPF, the results have not been consistent across brain regions and species (Udo de Haes et al, 2002, 2006; Zimmer et al, 2002). The 5-HT1A receptor agonist [11C]CUMI-101 has recently been shown suitable for the study of serotonin release in nonhuman primates (Milak et al, 2010). However, due to the lack of a reference region, the quantification is relatively elaborate and may limit its applicability in the clinical setting.
In a recent radioligand discovery program, we developed the selective 5-HT1B receptor antagonist radioligand [11C]AZ10419369 (Andersson et al, 2010; Pierson et al, 2008). Previously, we reported that [11C]AZ10419369 is suitable for
In an initial study using the potent serotonin releaser fenfluramine, we showed a reduction in specific binding of [11C]AZ10419369 in monkey (Finnema et al, 2010). Fenfluramine decreased [11C]AZ10419369 binding in a dose-dependent manner with a regional average of 27% after 1.0 mg/kg and 50% after 5.0 mg/kg. This study showed for the first time a major decrease in radioligand receptor binding measured with PET in the primate brain after administration of a strong serotonin releasing agent. Importantly, our observations have recently been confirmed by two other PET centers using the 5-HT1B receptor antagonist [11C]P943 (Cosgrove et al, 2011; Ridler et al, 2011). These three PET studies indicate that 5-HT1B receptor radioligands are promising tools for measurement of endogenous serotonin release and provide good reason for further development of this methodology.
In our initial work (Finnema et al, 2010), we used a fenfluramine displacement paradigm with bolus injections of [11C]AZ10419369. A displacement paradigm is attractive for quantification of neurotransmitter release because it allows for direct observation of changes in neuronal activity during pharmacological or physiological stimulation, which are reflected in modified radioligand binding. However, one possible limitation of our initial fenfluramine study was the quantification of [11C]AZ10419369 binding by the late time method approach (Ito et al, 1998). A methodology considered more suitable for quantification of neurotransmitter release, when using a displacement paradigm, is the equilibrium approach, which uses the administration of radioactivity by a bolus and constant infusion (BI protocol; Carson et al, 1993; Lassen, 1992).
The aim of the present PET study was to confirm the effect of fenfluramine on [11C]AZ10419369 5-HT1B receptor binding using an equilibrium approach in monkey.
Materials and methods
PET Measurements in Nonhuman Primates
Three female cynomolgus monkeys (
Radiochemistry
[11C]AZ10419369 was prepared by
PET System
Positron emission tomography measurements were conducted using the High Resolution Research Tomograph (Siemens Molecular Imaging, Knoxville, TN, USA). List-mode data were reconstructed using the ordinary Poisson-3D-ordered subset expectation maximization algorithm, with 10 iterations and 16 subsets including modeling of the point spread function. The corresponding in-plane resolution with ordinary Poisson-3D-ordered subset expectation maximization point spread function was 1.5 mm in the center of the field of view and 2.4 mm at 10-cm off-center directions (Varrone et al, 2009).
PET Experimental Procedure
Anesthesia was initiated by intramuscular injection of ketamine hydrochloride (~10 mg/kg, Ketaminol, Intervet AB, Boxmeer, The Netherlands) and maintained by a mixture of sevoflurane (1.5% to 8%; Abbott Scandinavia AB, Solna, Sweden), oxygen (~30%), and medical air after endotracheal intubation. In all PET measurements the head was immobilized with a fixation device (Karlsson et al, 1993). Body temperature was maintained by Bair Hugger Model 505 (Arizant Healthcare, Eden Prairie, MN, USA) and continuously monitored by an oral thermometer. Electrocardiogram, heart rate, and respiratory rate were continuously monitored throughout the experiments, and blood pressure was monitored at least every 15 minutes.
On a typical experimental day, the monkey was intubated ~ 1 hour before the first PET measurement. A saline infusion (10 mL/h) was also started at the same time to ensure a continuous fluid administration during the day, but was stopped during the BI protocol of the radioligand. The radioligand was intravenously administrated in a sural vein of the monkey using a BI protocol, consisting of a bolus administration of [11C]AZ10419369 (in 4 mL saline) over ~ 5 seconds, and a continuous infusion of radioligand with a pump at a flow rate of ~10 mL/h initialized at the same time as the bolus injection. Attenuation correction data were acquired with a 6-minute transmission measurement using a single 137Cs source. List-mode data were acquired continuously for 125 or 155 minutes starting at the injection of [11C]AZ10419369. Positron emission tomography images were then reconstructed with a series of 26 or 31 frames of increasing duration (60 seconds × 3, 180 seconds × 6, 360 seconds × 17, or 360 seconds × 22).
Study Design
To obtain the most suitable BI protocol, six preparatory PET experiments were conducted in one monkey, which included
In the second part of the study, we examined the three monkeys each two times with [11C]AZ10419369 on two experimental days. On each day, a baseline measurement was followed by a displacement measurement with fenfluramine 1.0 or 5.0 mg/kg. Positron emission tomography measurements were conducted for 153 minutes and were initiated three and a half hours apart. A sterile solution of (±)-fenfluramine. HCl in saline was intravenously infused over 5 minutes, starting 80 minutes after the start of the BI protocol of [11C]AZ10419369. The doses of fenfluramine (1.0 and 5.0 mg/kg) are expressed as relative to the free base.
In the third part of the study, we examined the three monkeys each two times on one experimental day. A baseline measurement was followed by a pretreatment measurement with fenfluramine 5.0 mg/kg. PET measurements were conducted for 123 minutes and were initiated three and a half hours apart (Figure 1). A sterile solution of fenfluramine in saline was intravenously infused over 5 minutes, starting 30 minutes before the start of the BI protocol of [11C]AZ10419369. To minimize confounding carryover effects between experiments a minimum of 1 month elapsed between all experimental days.
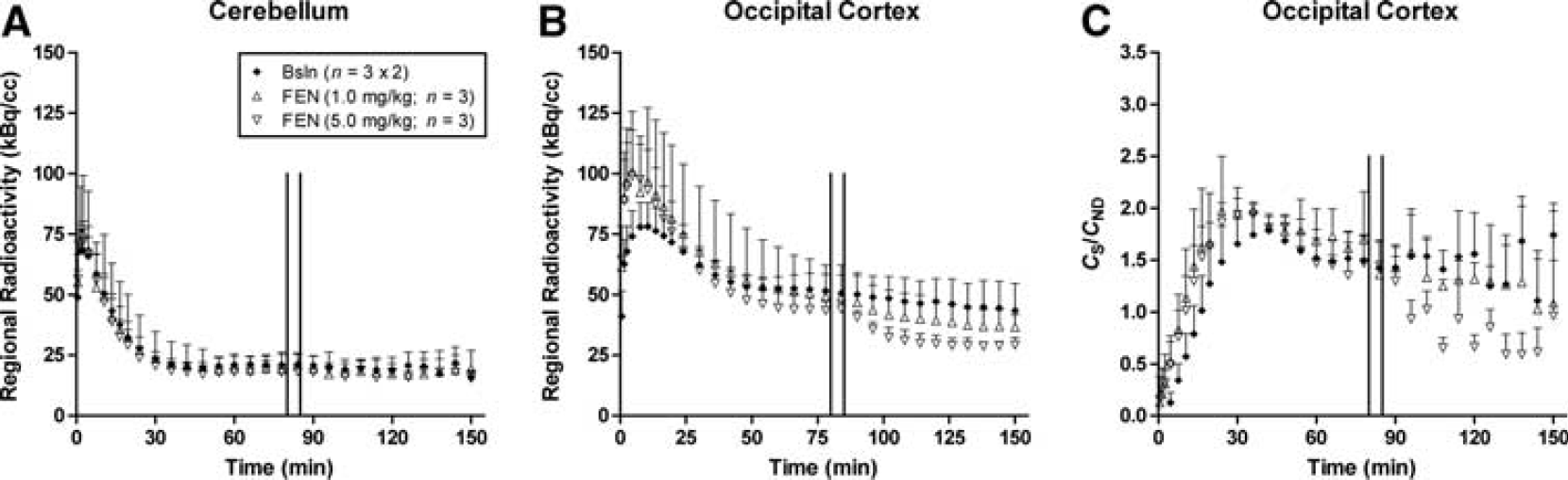
Mean time activity courses of radioactivity in cerebellum (
Regions of interest Definition
Brain magnetic resonance imaging was performed in a 1.5-T GE Signa system (General Electric, Milwaukee, WI, USA). AT1-weighted image was obtained for coregistration with PET and delineation of anatomic brain regions. The T1 sequence was a 3D spoiled gradient recalled protocol with the following settings: repetition time 21ms, flip angle 35°; field of view 12.8; matrix 256 × 256 × 128; 128 × 1.0 mm slices; and 2 number of excitations (NEX). The sequence was optimized for trade-off between a minimum of scanning time and a maximum of spatial resolution and contrast between gray and white matter.
Before delineation of regions of interests, the orientation of the brain was spatially normalized by having the high-resolution T1-weighted magnetic resonance images reoriented according to the line defined by the anterior and posterior commissures being parallel to the horizontal plane and the interhemispheric plane being parallel to the sagittal plane. The T1-weighted magnetic resonance images were then resliced to the resolution of the High Resolution Research Tomograph PET system, 1.219 × 1.219 × 1.219mm. The standardized T1-weighted magnetic resonance images were used as an individual anatomical template for each monkey. Regions of interests were defined manually on the reoriented magnetic resonance images. Regions of interests were drawn in the horizontal projection for the caudate nucleus (0.49 ± 0.05 cm3), cerebellum (CB, 0.91 ± 0.09 cm3), dorsal lateral prefrontal cortex (0.88 ± 0.12 cm3), globus pallidus (0.24 ± 0.02 cm3), midbrain (0.52 ± 0.08 cm3), occipital cortex (OC, 6.79 ± 1.09 cm3), putamen (0.70 ± 0.11 cm3), thalamus (0.69 ± 0.08 cm3), whole brain (62.77 ± 3.46 cm3), and in the coronal projection for the hippocampus (0.28 ± 0.07 cm3).
Mean PET images representing mean radioactivity between 0 to 57 minutes of the baseline PET measurement were coregistered to the magnetic resonance images using the normalized mutual information algorithm in PMOD (PMOD Technologies, Zurich, Switzerland). The generated transformation matrices were then applied to the dynamic emission data set of both PET measurements obtained on the same day.
Positron Emission Tomography Quantification
All calculations were based on the assumption that radioactivity in brain represents unchanged radioligand. Regional radioactivity concentrations were decay-corrected and expressed in kBq/mL. The CB is a region with negligible density of 5-HT1B receptors (Varnäs et al, 2001, 2004) and was used as an approximate for the sum of free (
Data for a time interval when
For illustrative purposes,
Statistical Analysis
The measurements performed with the displacement paradigm allow for estimation of the reproducibility of regional
The effect of fenfluramine on the outcome measure (
Results
Selection of Kbol, Equilibrium Conditions, and Test-Retest Reproducibility
Initially, six different
The time course for mean radioactivity in the OC and CB obtained during constant infusion of [11C]AZ10419369 are shown in Figures 1A and 1B. The time courses for the mean ratio of the specific binding (
Integrated total radioactivity concentration (
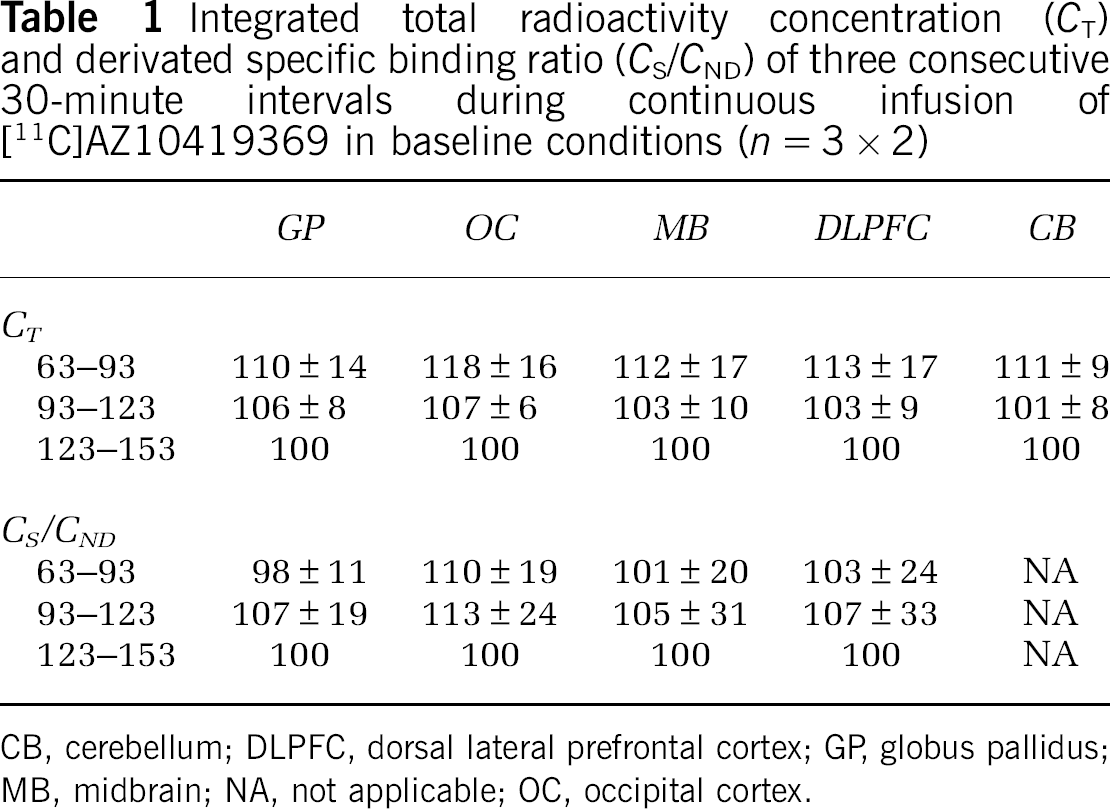
CB, cerebellum; DLPFC, dorsal lateral prefrontal cortex; GP, globus pallidus; MB, midbrain; NA, not applicable; OC, occipital cortex.
The displacement studies with fenfluramine allowed for evaluation of test-retest reproducibility of
Test-retest reproducibility of [11C]AZ10419369
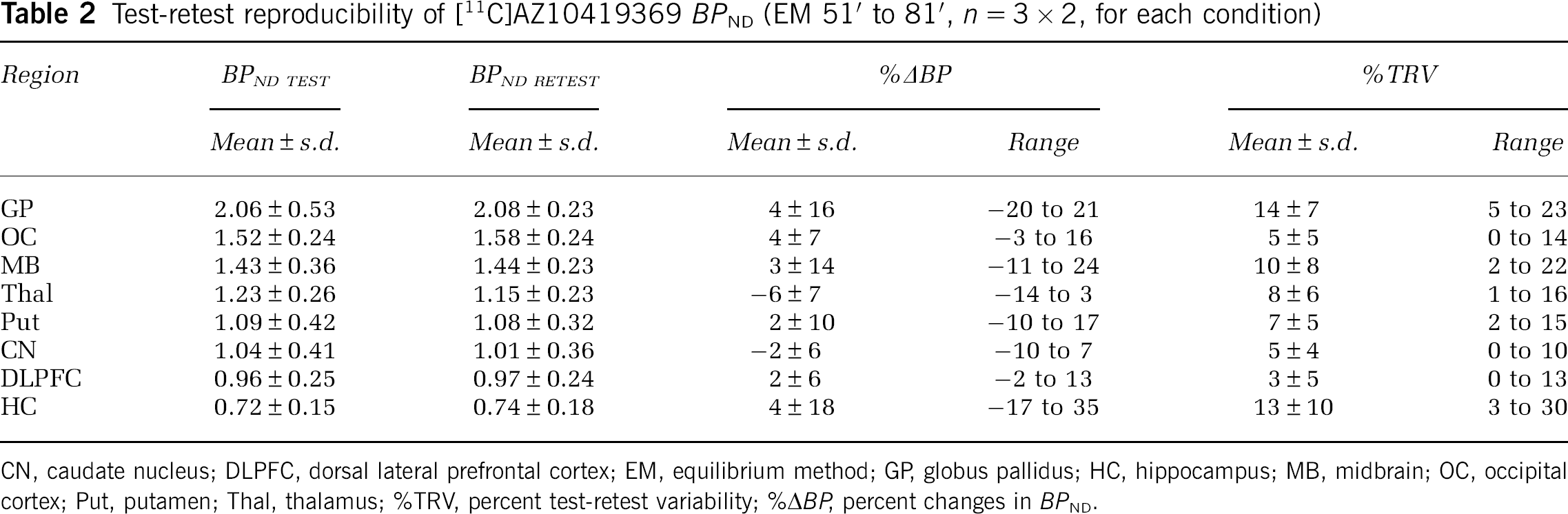
CN, caudate nucleus; DLPFC, dorsal lateral prefrontal cortex; EM, equilibrium method; GP, globus pallidus; HC, hippocampus; MB, midbrain; OC, occipital cortex; Put, putamen; Thal, thalamus; %TRV, percent test-retest variability; %Δ
Fenfluramine Effect
In both the displacement and pretreatment studies, fenfluramine caused an evident, dose-dependent, decrease in [11C]AZ10419369 binding in all 5-HT1B receptor containing regions (Figures 1–4, Table 3).
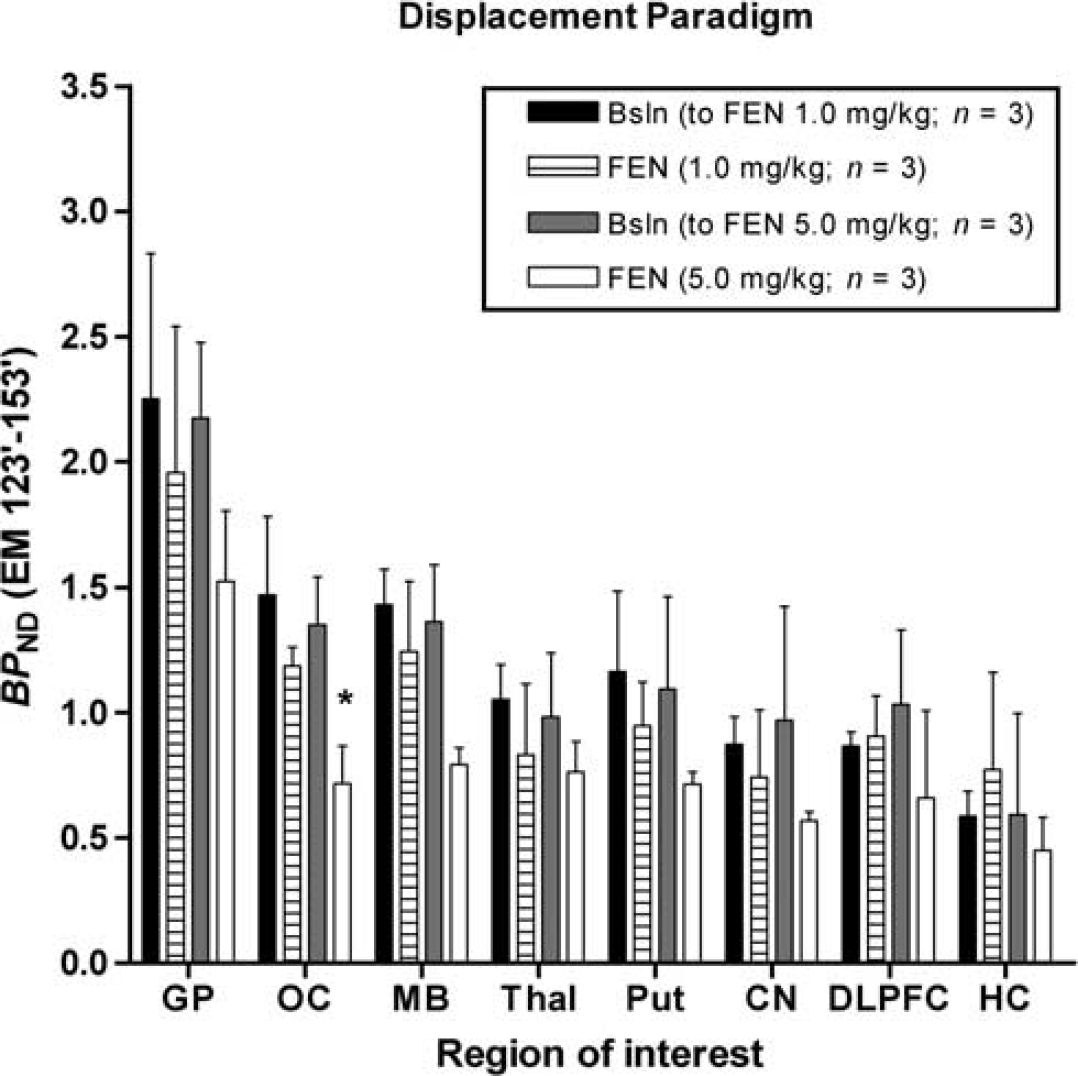
Mean binding potential (
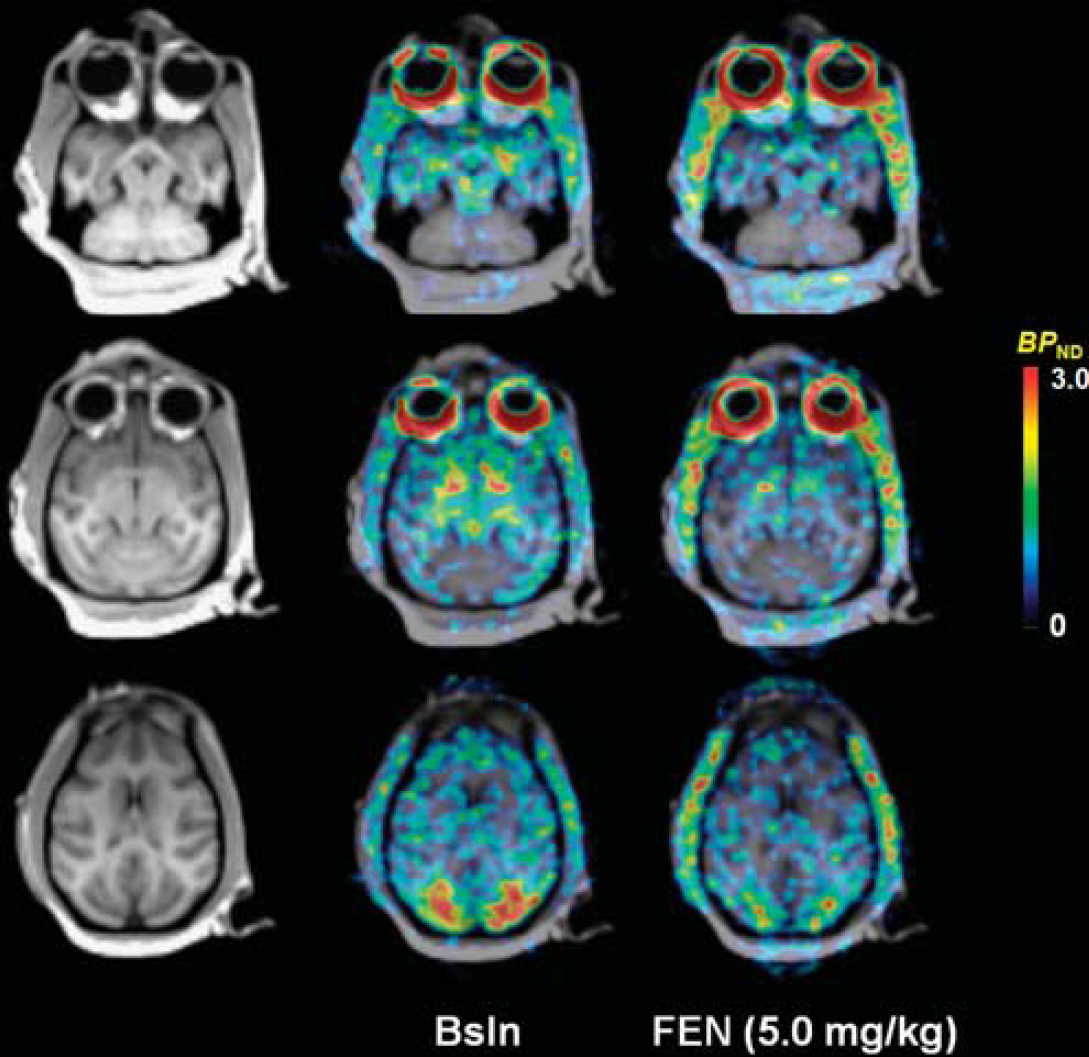
Magnetic resonance images and corresponding overlaid
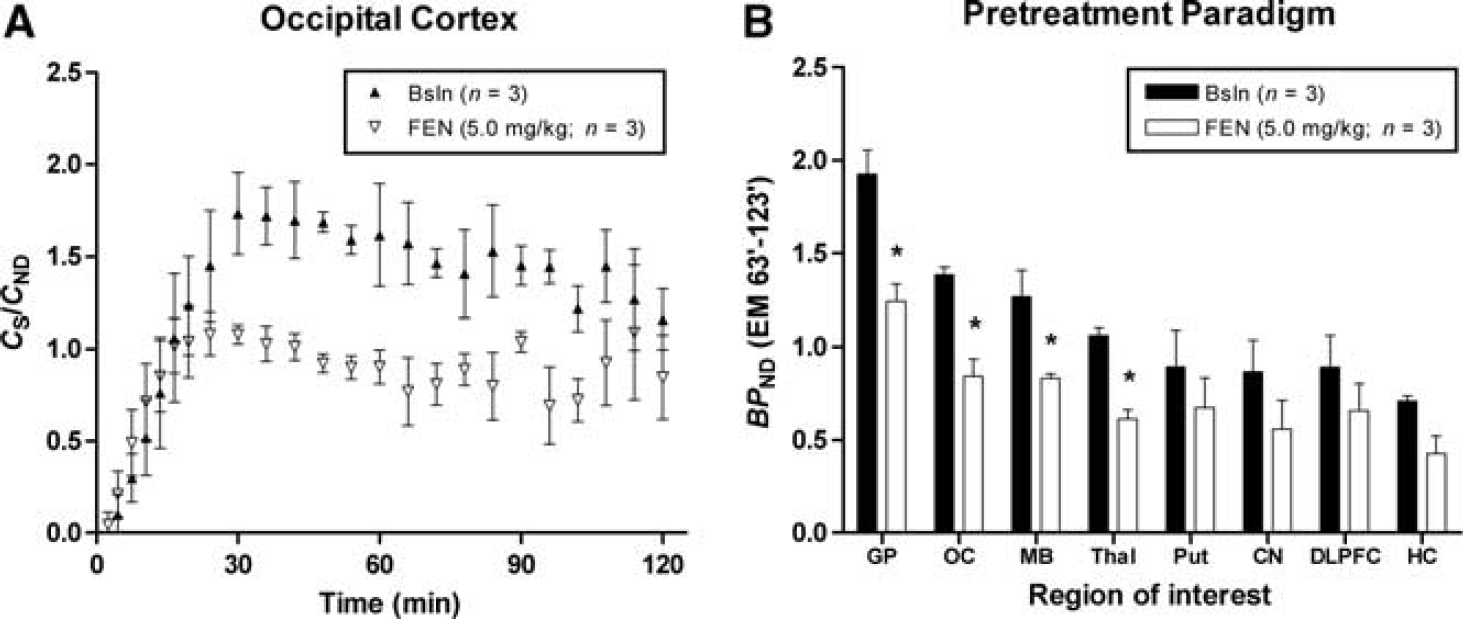
Time activity courses of mean
Comparison of fenfluramine (1.0 and 5.0 mg/kg) effect on [11C]AZ10419369 binding potential (BPND) measured with a displacement and pretreatment paradigm in three cynomolgus monkeys (M1 to M3)
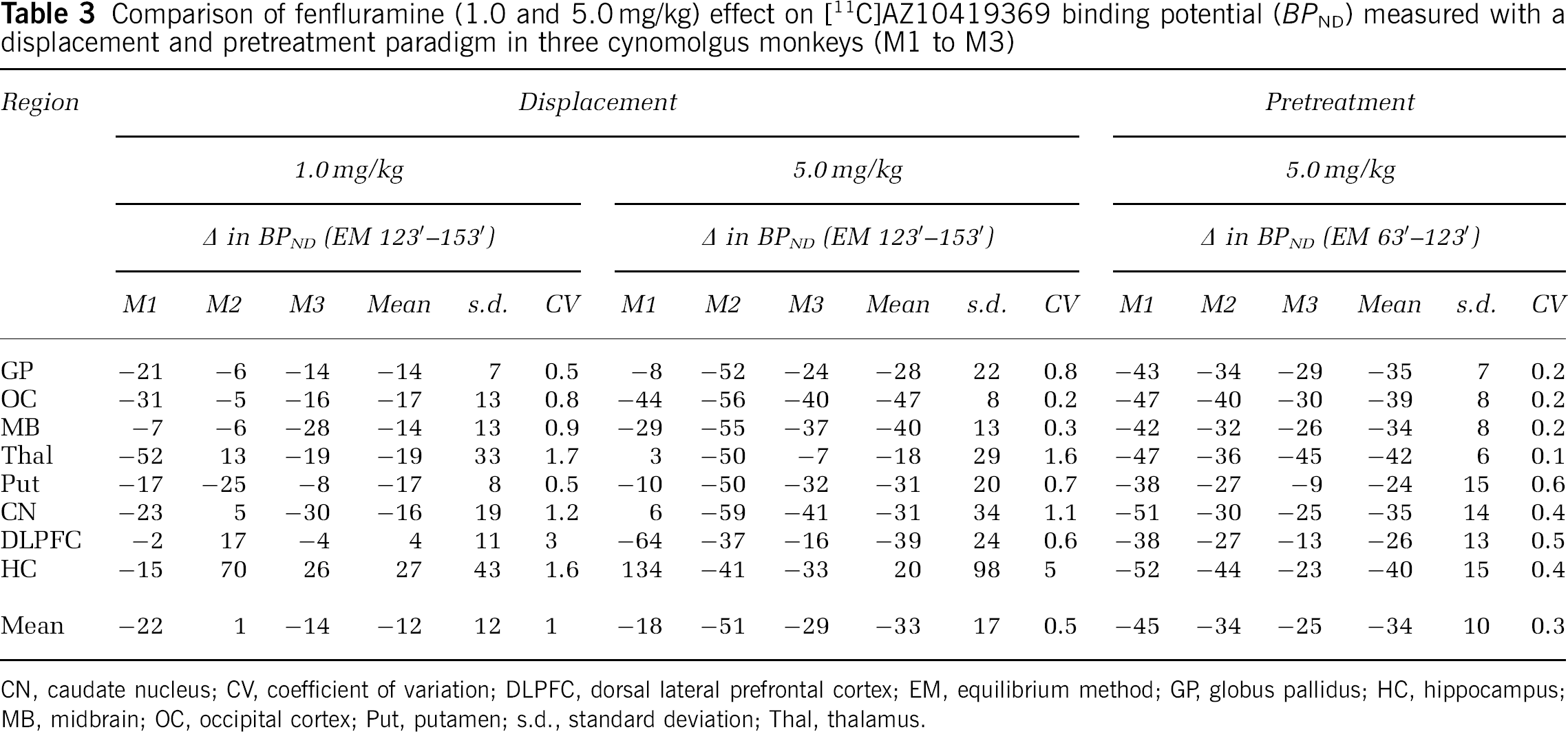
CN, caudate nucleus; CV, coefficient of variation; DLPFC, dorsal lateral prefrontal cortex; EM, equilibrium method; GP, globus pallidus; HC, hippocampus; MB, midbrain; OC, occipital cortex; Put, putamen; s.d., standard deviation; Thal, thalamus.
In the displacement paradigm, fenfluramine (1.0 mg/kg) did not significantly reduce [11C]AZ10419369
In the displacement paradigm, fenfluramine (5.0 mg/kg) did not significantly reduce [11C]AZ10419369
In the pretreatment paradigm, fenfluramine (5.0 mg/kg) significantly reduced [11C]AZ10419369
Discussion
We previously showed that administration of the potent serotonin releasing agent fenfluramine decreased [11C]AZ10419369 receptor binding in nonhuman primates using PET (Finnema et al, 2010). The aim of the present PET study was to confirm the effect of fenfluramine on [11C]AZ10419369 5-HT1B receptor binding during equilibrium conditions in monkey. First, a BI protocol for administration of [11C]AZ10419369 was developed for
Development of a BI protocol is facilitated by
During four of the initial
When applying the BI approach, an important criterion for proper quantification is to achieve a true equilibrium for [11C]AZ10419369 in brain regions. One approach to examine the achievement of a true binding equilibrium for [11C]AZ10419369 is to compare
To further validate the methodology for measurement of drug-induced changes in radioligand binding, we also made a small test-retest analysis in the current displacement study. Comparison of
The test compound fenfluramine is generally considered a substrate for the serotonin transporter protein (SERT) and may increase extracellular serotonin levels by SERT inhibition and by promotion of carrier-mediated exchange (Garattini et al, 1986; Rudnick and Clark, 1993). Microdialysis studies in unanesthetized monkeys have shown that an intravenous dose of 5 mg/kg fenfluramine increases serotonin levels by 20-fold (Udo de Haes et al, 2006). The observed fenfluramine-induced decrease in [11C]AZ10419369 binding, in both displacement and pretreatment studies, is therefore likely related to elevated serotonin levels.
The here reported mean fenfluramine effect on [11C]AZ10419369 binding across eight brain regions (–12% at 1.0 mg/kg; −33% and −34% at 5.0 mg/kg) is in line with our previous study, in which [11C]AZ10419369 was administered as a bolus injection (–27% at 1.0 mg/kg; −50% at 5.0 mg/kg; Finnema et al, 2010). A comparable effect of fenfluramine on radioligand binding has more recently also been showed using the 5-HT1A receptor radioligand [11C]CUMI-101 (Milak et al, 2010) and the 5-HT1B receptor radioligand [11C]P943 (Cosgrove et al, 2011; Ridler et al, 2011). Importantly, the extent of the displacement effect of the three radioligands has been shown to be comparable, at equivalent (±)-fenfluramine doses (approximately −25% at 1.0 mg/kg; −40% at 5.0 mg/kg). Although the three radioligands provide similar results in the fenfluramine challenge, the methodology used for quantification has been different across the radioligands. Only for [11C]AZ10419369 the reference region approach has so far been validated. This simplified quantitative approach is an advantage when advancing to clinical studies, as no blood sampling is required.
The CB has been considered suitable for estimation of nondisplaceable binding of [11C]AZ10419369, based on several previous findings. First, postmortem autoradiography studies have showed that the 5-HT1B receptor density is negligible in the CB of the primate brain (Varnäs et al, 2001, 2004). Second, pretreatment studies with 5-HT1B receptor antagonists have shown no evident effect on [11C]AZ10419369 binding in the CB of guinea-pig and monkey (Maier et al, 2009; Pierson et al, 2008). Third, the cerebellar distribution volume (
A classical displacement paradigm is typically applied to show reversibility of radioligand binding. In our previous study we showed a fenfluramine displacement effect on [11C]AZ10419369 binding using bolus injection of the radioligand (Finnema et al, 2010). A limitation of such a design is that a separate PET measurement is required to obtain baseline
Microdialysis studies have previously shown that serotonin levels are very dynamic after intravenous administration of fenfluramine. In monkey, a rapid intravenous bolus injection of fenfluramine has been shown to induce a peak in serotonin levels ~ 15 minutes after administration, followed by a rapid decrease in serotonin levels over time. One hour after administration, serotonin levels were less than half of the peak value, with recovery to baseline levels ~ 4 hours after fenfluramine administration (Udo de Haes et al, 2006). The time effect in the fenfluramine-induced decreases in [11C]AZ10419369
Examination of regional differences in fenfluramine effect is of interest to further understand the applied methodology. A number of biological aspects should however be considered when comparing regional changes in [11C]AZ10419369
One alternative interpretation we recognized is that fenfluramine has a direct competitive effect on [11C]AZ10419369 receptor binding. The in the literature reported 5-HT1A and 5-HT1B receptor affinity of fenfluramine are, however, in the micro molar range (Mennini et al, 1991). In addition, fenfluramine was reported not to influence the 5-HT1A receptor binding of the antagonist radioligand [18F]MPPF (Udo de Haes et al, 2006). We therefore argue that the observed effect of fenfluramine on radioligand binding is most likely due to elevated serotonin levels, but further confirmation with other serotonin level modifying agents is required to fully exclude direct competitive effects of fenfluramine and its metabolites. First results of this approach have very recently shown that administration of the stimulant
Follow up studies, applying the here evaluated methodology and other serotonin level modifying drugs, are currently ongoing to further explore the limit of sensitivity toward modified serotonin levels in nonhuman primates. Ultimately, human studies are required to fully assess the utility of [11C]AZ10419369 and PET to determine serotonin release in the living brain of humans. When using the mean plasma input function and regional brain time activity curves obtained after bolus injection of [11C]AZ10419369 in human subjects from a previous reported study, we estimate that a
In summary, the current PET study confirms that fenfluramine decreases [11C]AZ10419369 receptor binding in nonhuman primates. The displacement studies confirm the reversibility of [11C]AZ10419369 binding and show that the
Footnotes
Acknowledgements
We thank AstraZeneca for providing the radioligand precursor and H Lundbeck A/S (Dr Benny Bang-Andersen) for providing (±)-fenfluramine. We also gratefully acknowledge the Karolinska PET group, including special thanks to Gudrun Nylen, for excellent technical assistance.
Lars Farde is a shareholder with AstraZeneca. All other authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
