Abstract
To investigate the chronic effects of a novel thyrotropin-releasing hormone analog, JTP-2942 (Nα-[(1S, 2R)-2-methyl-4-oxocyclopentylcarbonyl]-
Keywords
Patients with cerebrovascular disease may exhibit neurologic symptoms such as hemiplegia as well as cognitive disturbances (Caplan et al., 1985, 1990). Pathologic and functional changes not only during the acute phase but also during the subacute and chronic phases of focal cerebral ischemia have been extensively investigated (O'Brien et al., 1974; Ginsberg et al., 1977; Hayakawa and Waltz, 1975; Tamura et al., 1980; Tamura et al., 1981a,b; Tamura et al., 1985; Yamamoto et al., 1988; Yamamoto et al., 1989a,b; Markgraf et al., 1992; Wahl et al., 1992; Okada et al., 1995; Yonemori et al., 1996; Yonemori et al., 1998). However, for the development of therapeutic agents, there are only a few reports that evaluate the effect on motor or neurologic deficits during subacute or chronic phase of cerebral ischemia (Yamamoto et al., 1989a,b).
Thyrotropin-releasing hormone (TRH:
Chemical structure of JTP-2942
The present study was performed to investigate the effects of JTP-2942 on motor and neurologic deficits during the subacute and chronic phase of focal cerebral ischemia using an middle cerebral artery (MCA) occlusion model in rats and compared the effects of JTP-2942 with the effect of CDP-choline, a therapeutic agent for the disturbance of consciousness and hemiparesis after stroke (D'Orlando and Sandage., 1995; Secades and Frontera, 1995).
METHODS
Surgical procedure
This experiment complied with the Guidelines of Animal Experimentation of the Teikyo University School of Medicine.
Male Wistar rats (Slc, Shizuoka, Japan), 10 weeks of age and weighing 250 to 310 g, were anesthetized with 1 to 2% halothane and the left MCA was permanently occluded at a proximal site using a microbipolar coagulator (Tamura et al., 1981a; Yamamoto et al., 1988). After the incision was sutured, the rats were allowed free access to laboratory chow and water. Shamoperated rats were subjected only to exposure of the MCA.
Drugs
JTP-2942 (Lot Q; Central Pharmaceutical Research Institute, Japan Tobacco Inc.) was dissolved in physiologic saline (Otsuka Pharmaceutical Co., Ltd.) for intravenous administration at a dose of 1.0 mL/kg. For CDP-choline, nicholine H 25% solution for injection (Takeda Chemical Industries, Ltd.) was used at a dose of 2.0 mL/kg.
Behavioral examination
Inclined plane test. We evaluated the motor performance of the rats using an inclined plane (model SN-453, Shinano Co. Ltd., Tokyo, Japan), once a week for 9 weeks from 1 week after surgery according to the method described by Yonemori et al. (1998). The sliding apparatus had a 60 × 40 cm stainless steel plane that could be inclined at an angle of 0° (horizontal) to 60°. Each rat was initially placed on a 30° inclined plane in the left-headed position (right side up orientation), right-headed position (left side up orientation), or up-headed position (head up orientation), and the angle of the inclined plane was increased at a rate of 2°/s from 30 to 60°. The maximum angle was then determined at the moment when a limb of the rat slipped to maintain body position. The test was carried out three times for each head position and averaged. Each trial was carried out at 1-minute intervals. During this time the rat was returned to a cage filled with dry wood chips, which were used to wipe and dry the urine and feces from the limbs and body so as not to influence the friction coefficient. Trials in which the rat turned its body uphill without slipping movement of a limb were not included.
Neurologic examination. We evaluated the sensorimotor performance in the rats using a neurologic deficit score based on the detection of hemiparesis and abnormal posture. The right hind limb of each rat was extended gently with round-tipped forceps and the flexor response was evaluated as 0 (normal), 1 (slight deficit), 2 (moderate deficit), or 3 (severe deficit). For the assessment of posture, the rats were suspended by the tail, and forelimb flexion and body twisting were evaluated as 0 (normal), 1 (slight twisting), 2 (marked twisting), or 3 (marked twisting and forelimb flexion).
Passive avoidance task. The rats were tested using a step-through type of passive avoidance task (Jarvik & Kopp, 1967; Yamamoto et al., 1988). The apparatus (Neuroscience Inc., Tokyo, Japan) consisted of two compartments: an illuminated chamber (10 cm × 30 cm) and a dark one (30 cm × 30 cm). The rats initially underwent habituation by placing them in the illuminated chamber, and a door between the chambers was then opened so that they could enter the dark chamber. In the acquisition trial, the rats performed the same task, but a 0.60-mA foot shock was delivered through the grid floor for 1 second once they entered the dark chamber. Retention trials were performed without foot shock 1 day after the acquisition trial. The step-through latency was recorded to a maximum of 300 seconds.
Experimental procedure
The general protocol of this experiment is shown in Fig. 1B. The MCAs of 50 rats were occluded and 7 rats were sham-operated. MCA-occluded rats whose total neurologic score at 1 week after operation was 3 to 4 were selected, and these rats received JTP-2942 (0.003 mg/kg, n = 10; 0.01 mg/kg, n = 10; 0.03 mg/kg, n = 10), CDP-choline (500 mg/kg, n = 10), or saline (n = 10) intravenously from 1 week after operation once a day for 4 weeks. The sham-operated group (n = 7) was given only physiologic saline intravenously. Neurologic examination and the inclined plane test were performed once a week for 8 weeks, and the passive avoidance test was performed 2 weeks after operation. All behavioral tests were performed blindly before daily drug administration.
Histopathology
After the behavioral studies were completed, the rats were anesthetized and fixed by perfusion with 10% formaldehyde in 0.1 mol/L phosphate buffer, and the brains were removed. Five coronal blocks (2 mm thick) were cut from 12.2 to 4.2 mm anterior to the interaural line, according to the brain atlas of Paxinos and Watson (1986), and embedded in paraffin. Then, 4-μm sections were cut from each block and stained with hematoxylin and eosin. The area of each cerebral hemisphere was determined on individual sections using a digitizer (Luzex FS, Nireco, Tokyo, Japan), and the volume of each hemisphere was calculated from the individual areas. The infarcted volume was calculated by subtracting the volume of the left hemisphere from that of the right hemisphere.
Statistical analysis
The values are expressed as mean ± SD for all parameters. To analyze parametric data, repeated measures of analysis of variance (ANOVA) followed by Tukey's test or Student's t test was used. When dealing with nonparametric data, Kruskal-Wallis test followed by Mann-Whitney U test was applied.
RESULTS
General behavior
Eight rats with MCA occlusion died (vehicle, 2; JTP-2942 0.003, 2; JTP-2942 0.01, 1; JTP-2942 0.03, 1; CDP-choline, 2) with no significant differences among the groups. There was also no difference in body weight among the six groups (group: F5,43 = 1.865, P =0.121; time: F9,387 = 34.164, P < 0.001; group*time: F45,387 = 1.087, P = 0.331; two-factor ANOVA for repeated measurements).
Inclined plane test
The motor performance using the inclined plane test was evaluated once a week for 8 weeks. Figure 2 shows the results of the test. The maximum angles at which body position was maintained in the sham-operated rats were 43.8 ± 1.4°, 43.3 ± 1.7°, and 42.9 ± 1.7° for left-, right-, and up-headed initial body positions, respectively, at 1 week after surgery. When compared to the sham-operated rats, the MCA-occluded rats exhibited disturbed performance in this task. First, for the left-headed initial position, the maximum angle to hold position, 36.8 ± 0.8° at 1 week after MCA occlusion, significantly decreased compared with that of the sham-operated rats (P < 0.01, Tukey's test). This decrease was detected throughout the test period (Fig. 2A). Administration of JTP-2942 (0.01 to 0.03 mg/kg, administered intravenously) significantly improved this deficit in a time- and dose-dependent manner compared to the vehicle group, and the maximum angle at which body position was maintained on the inclined plane increased to that of sham-operated rats at 3 to 5 weeks after the start of the drug administration (group: F5,43 = 117.90, P < 0.001; time: F8,344 = 57.19, P < 0.001; group × time: F40,344 = 11.50, P < 0.001; two-factor ANOVA for repeated measurements). It is noteworthy that this improvement still persisted 4 weeks after the cessation of JTP-2942 treatment. On the other hand, CDP-choline did not improve the deficit throughout the test period.
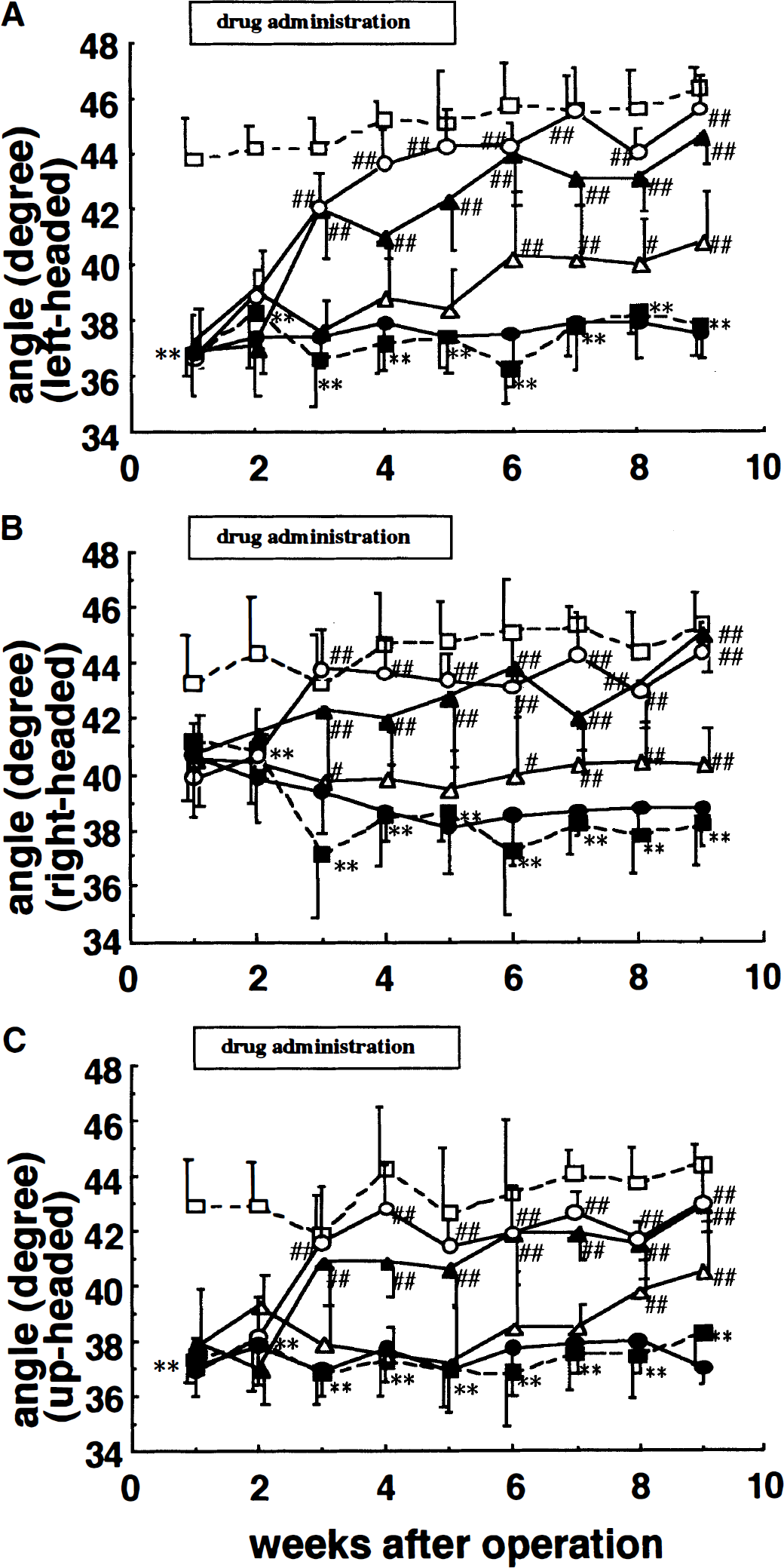
Effects of JTP-2942 and CDP-choline on the ability to maintain body position on an inclined plane in MCA-occluded rats. The maximum angle was determined at the moment when the rat failed to maintain its position for left-headed
Second, for the right-headed initial position, the maximum angle was 41.2 ± 2.1° (mean ± SD) at 1 week after MCA occlusion which was similar to that of the sham-operated rats. However, the maximum angle gradually decreased to below 40° after 3 weeks (Fig. 2B). The maximum angle at which the JTP-2942 (0.01 to 0.03 mg/kg, administered intravenously)-treated rats could maintain their body position on the inclined plane was over 40° throughout the test period and did not decrease even after the cessation of JTP-2942 treatment. This angle was similar to the maximum angle of the sham-operated group. The angle of the CDP-choline-treated group decreased to below 40° after 3 weeks and did not improve throughout the test period (group: F5,43 = 62.20, P < 0.001; time: F8,344 = 2.08, P < 0.05; group × time: F40,344 = 5.60, P < 0.001; two-factor ANOVA for repeated measurements).
Third, for the up-headed initial position, the level of performance was the same as that for the left-headed position (Fig. 2C). The sham-operated rats did not exhibit any deficit in ability throughout the test period (group: F5,438 = 81.35, P < 0.001; time: F8,344 = 19, P < 0.001; group × time: F40,344 = 11.50, P < 0.001; two-factor ANOVA for repeated measurements).
Neurologic examination
Figure 3 shows the results of the neurologic examination. The MCA-occluded rats had a significantly higher neurologic deficit score at 1 week after MCA occlusion than the sham-operated rats (P < 0.01, Kruskal-Wallis followed by Mann-Whitney U test). MCA occlusion also significantly disturbed both the posture and the flexor response of the contralateral hindlimb (P < 0.01, Mann-Whitney U test). Although the neurologic deficit scores showed gradual recovery, MCA-occluded rats had significantly higher neurologic deficit scores than sham-operated rats even at 9 weeks after surgery. Administration of JTP-2942 (0.003 to 0.03 mg/kg, intravenously) significantly improved this deficit in a time- and dose-dependent manner compared to the vehicle group. CDP-choline (500 mg/kg, administered intravenously) also improved this deficit.
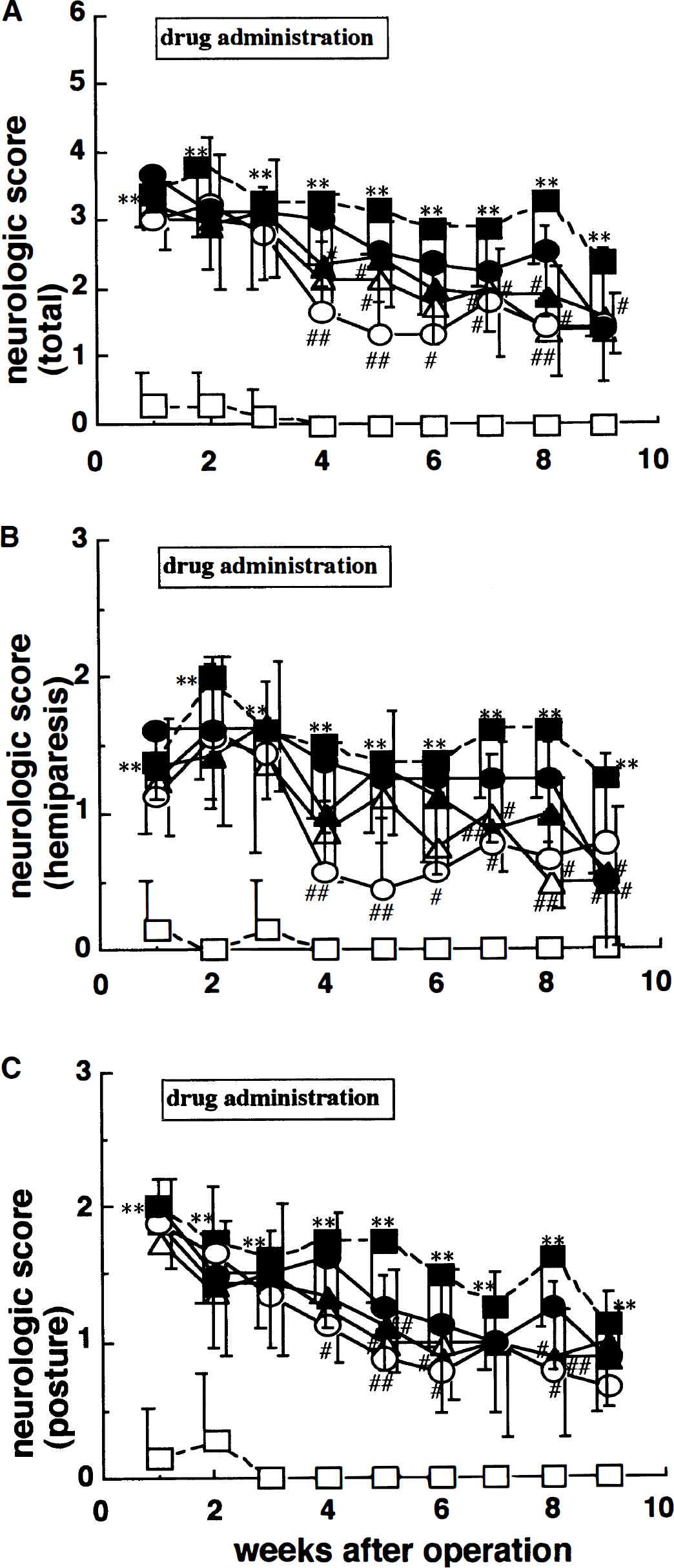
Effects of JTP-2942 and CDP-choline on neurologic deficits in MCA-occluded rats. Abnormal posture
Passive avoidance task
The step-through test was performed 2 weeks after MCA occlusion. The retention latency of the sham-operated rats was 300 seconds, which indicated that all these rats performed this test well (Fig. 4). On the other hand, the MCA-occluded rats did not; the retention latency of the vehicle group was 129 ± 127 seconds, which was significantly decreased compared to that of the sham-operated group. Administration of JTP-2942 (0.003 to 0.03 mg/kg, intravenously) significantly improved this deficit in a dose-dependent manner compared to the vehicle group. CDP-choline (intravenous 500 mg/kg) also improved this deficit.
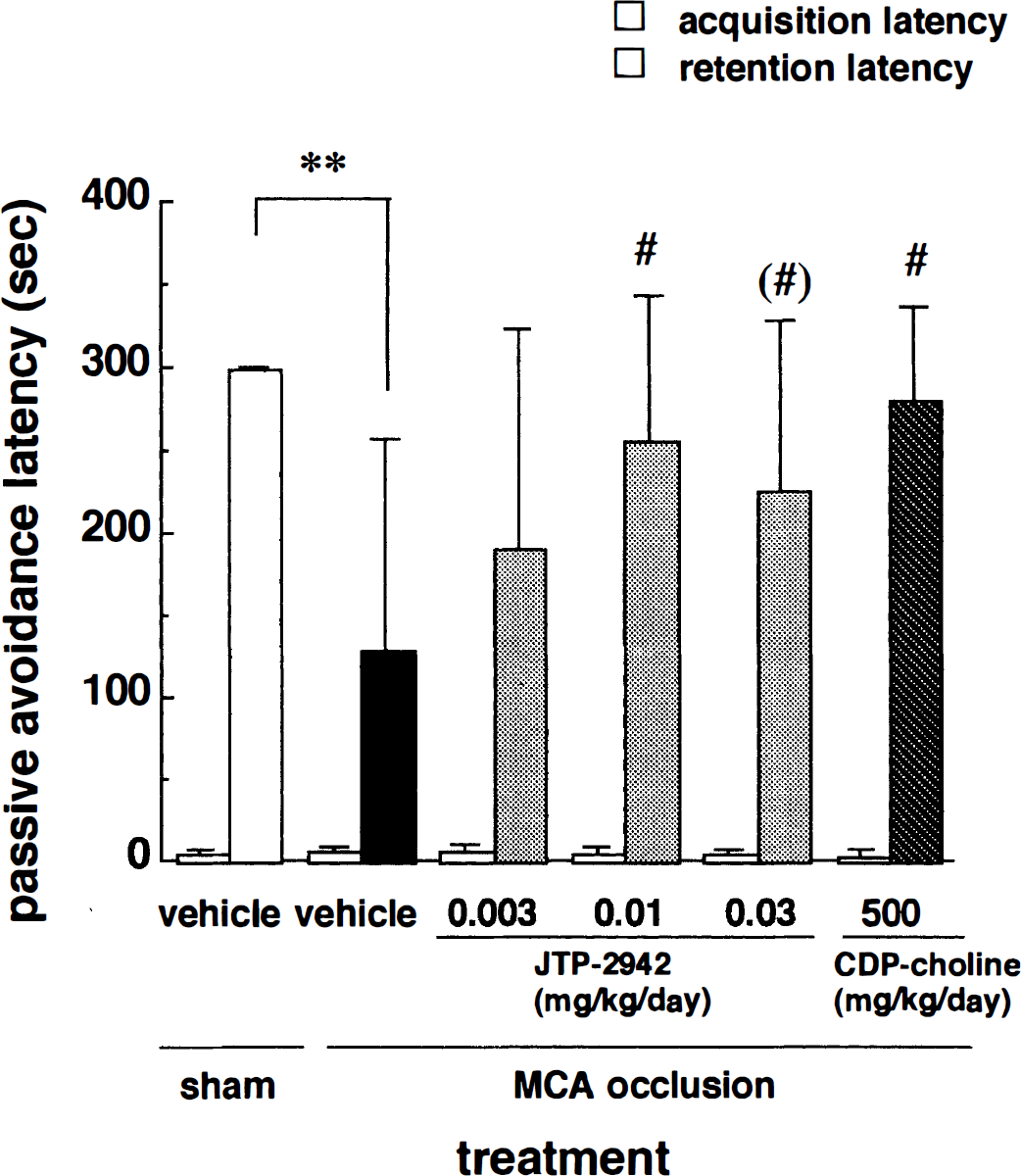
Effects of JTP-2942 and CDP-choline on passive avoidance latency in MCA-occluded rats. The left column in each pair represents the acquisition latency, and the right column represents the retention latency. Each column represents the mean ± SD (n = 7 to 9). **P < 0.01 versus the sham-operated group; (#)P < 0.10; #P < 0.05 versus the MCA-occluded and vehicle-treated group (Kruskal-Wallis followed by Mann-Whitney U test).
Histology
Infarcted volumes were calculated from the ipsilateral to the contralateral hemisphere volume in the coronal slices from interaural 12.2 to 4.2 mm. The infarcted volume of the MCA-occluded group was 154 ± 30 mm3 which was significantly increased compared to that of the sham-operated group (15 ± 30 mm3). Administration of JTP-2942 (0.003 to 0.03 mg/kg) or CDP-choline (500 mg/kg) did not affect the infarcted volumes compared to that of the vehicle group (ANOVA followed by Tukey's test; Fig. 5).
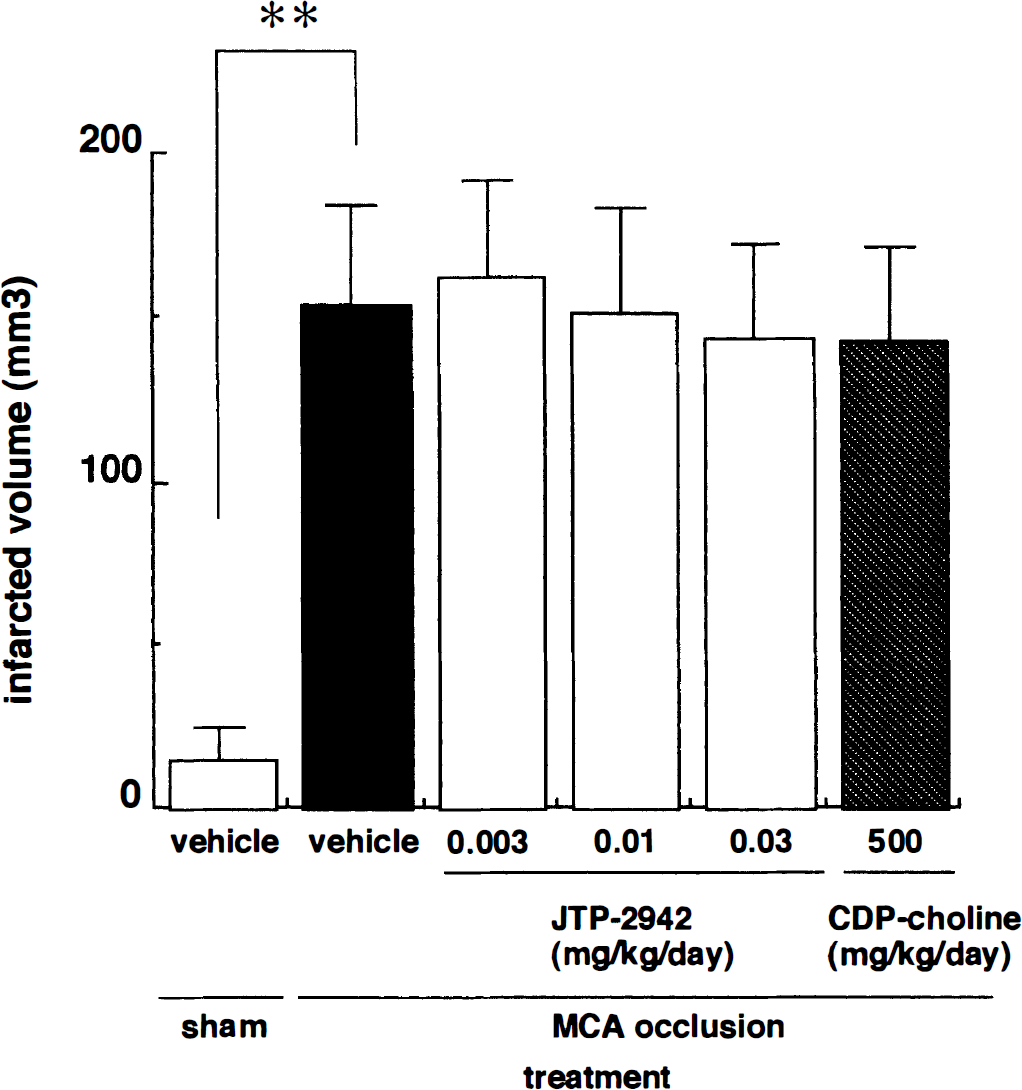
Effects of JTP-2942 and CDP-choline on massive cerebral shrinkage. Each column represents the infarcted volume. Each column represents the mean ± SD (n = 7 to 9). There were no significant differences among the MCA-occluded groups.
DISCUSSION
Patients with cerebrovascular disease frequently exhibit various clinical symptoms, including motor deficits such as neurologic disorder, and cognitive disturbances as well as psychosomatic manifestations (Caplan et al., 1985, 1990). Motor deficits comprise the core symptoms and are the most important targets for clinical therapy. However, there are few effective drugs for these deficits.
Thyrotropin-releasing hormone (TRH:
The present study was performed to investigate the effect of JTP-2942 on motor and neurologic deficits during the subacute and chronic phases of focal cerebral ischemia because development of a delayed treatment is very important in stroke patients. Yamamoto et al. (1989b) reported that a TRH analog improved behavioral response in a model of focal ischemia when the drug administration was started 1 week after ischemia. Similarly, the administration of JTP-2942 was started 1 week after ischemia in this study. We used a proximal MCA occlusion model in rats as reported by Tamura et al. (Tamura et al., 1981a,b; Yamamoto et al. 1988) because of the constant size of the infarct in this model and because of the similarities between the anatomy of the cranial circulation in the rat and humans (Yamori et al., 1976). In the inclined plane test, the maximum angle at which the body position was maintained on the plane was improved when JTP-2942 was administered for 4 weeks. On the other hand, CDP-choline, which is widely used as an agent for hemiparesis and disturbance of consciousness (D'Orlando and Sandage, 1995; Secades and Frontera, 1995), did not affect the maximum angle of the inclined plane. Impaired sensorimotor performance, based on the evaluation of posture and hemiparesis developed by Bederson et al. (1986), and the passive avoidance task were improved with administration of JTP-2942 and CDP-choline. The improvements were not related to a reduction in the infarcted area.
Because the inclined plane test serves as an indicator of the muscle strength in MCA-occluded rats (Yonemori et al., 1998), the improvement effect of JTP-2942 observed in the inclined plane test was considered to reflect an improvement in the muscle weakness caused by MCA occlusion. On the other hand, inasmuch as CDP-choline did not affect the angle of the inclined plane, it is considered to be effective in improving sensorimotor performance but not muscle strength. Thus, it is considered that JTP-2942 may aid in the recovery of motor deficits after cerebral ischemia because of muscle weakness, which constitutes a common complaint after stroke in humans. Additionally, JTP-2942 was effective in the improvement of neurologic performance and the passive avoidance task in rats, as was CDP-choline, indicating that JTP-2942 might be effective for treating memory disturbance.
It is noteworthy that the effect on muscle strength detected in the inclined plane test still persisted after the cessation of JTP-2942 administration. This indicates that JTP-2942 caused not only the activation of damaged neuronal networks but also structural changes. This is first because the measurement was performed before daily administration of drugs and second because it was considered that JTP-2942 did not remain in the rats 4 weeks after cessation of drug administration (t1/2 of JTP-2942 is about 30 minutes in rats). This indicated that although JTP-2942 directly activated cholinergic, dopaminergic, noradrenergic, serotonergic neurons, and other neuronal types for several hours after administration (Shinoda et al., 1992; Toide et al., 1993), these activations did not directly affect the motor performance on the inclined plane. On the other hand, administration of JTP-2942 was only started 1 week after MCA occlusion, when the presence of an infarcted area was confirmed. It is not considered that JTP-2942 administration in this protocol reduced the size of infarct. Indeed, the shrinkage observed in JTP-2942-treated groups was not different from that in the vehicle-treated group in this study. From this point of view we concluded that there may be some mechanism for JTP-2942 to improve the motor deficit caused by MCA occlusion and that this may be associated with plastic changes, such as changes in synaptic connections or activation of a compensation mechanism. In support of this idea it has been reported that TRH promotes neurite development in cultured neurons (Schmidt-Achert et al., 1984; Iwasaki et al., 1992), and our findings show that JTP-2942 has a similar effect (data not shown). Such improvement associated with synaptic connection changes or activation of a compensation mechanism may involve increased metabolic turnover related to oxygen and glucose supply. Recently, Katsumata et al. (1996a,b) confirmed that JTP-2942 improved local cerebral blood flow and local glucose utilization in rats with cerebral ischemia.
CDP-choline, which is approved in Europe and Japan for treatment of patients suffering from stroke, head trauma, and other neurologic disorders (D'Orlando and Sandage, 1995; Secades and Frontera, 1995), improved sensorimotor performance, based on posture and hemiparesis evaluations, and improved performance in the passive avoidance task but did not affect the angle on the inclined plane, an indicator for muscle strength. CDP-choline may thus affect sensorimotor performance but not muscle strength. In this regard, JTP-2942 is expected to serve as an agent to improve not only sensorimotor performance but also muscle strength, which is the most important target for clinical therapy.
In conclusion, a novel TRH analog, JTP-2942, improved motor deficits, particularly muscle strength, as well as sensorimotor performance. These effects were not attributable to a change in the size of the infarct and were not diminished 4 weeks after cessation of drug administration. The mechanisms of the improving effect of JTP-2942 may be associated with changes in synaptic connections or activation of a compensation mechanism.
