Abstract
The authors investigated the impairment of spatial cognitive performance in rats with chronic focal cerebral ischemia using the Morris maze, and examined the correlation between this deficit and other behavioral changes, such as step-through latency in passive avoidance task and neurologic score, or pathologic changes. The authors focused on the relationship between the damaged brain region and the affected spatial learning behavior. In the Morris maze task at 8 weeks after the middle cerebral artery (MCA) occlusion, escape latency, swimming path length, and percent time spent in goal quadrant of MCA-occluded rats were impaired, which correlated with shrinkage of the cortex involving parietal cortex, but not caudate-putamen (CP). Middle cerebral artery-occluded rats were also impaired in the percent time spent in the outermost annulus and in turning ratio, which significantly correlated with shrinkage of CP, but not cortex. Middle cerebral artery-occluded rats showed two typical search patterns; one was almost the same as that of sham-operated and intact rats, and the other was round shaped and had less turning behavior. Both subgroups of MCA-occluded rats divided by turning ratio had significantly impaired spatial cognitive performance, which indicates that the changes of search pattern did not affect cognitive performance in the Morris maze. The neurologic deficits recovered gradually after MCA occlusion, which correlated with shrinkage of cortex and CP. The step-through latency in passive avoidance task of the MCA-occluded rats was impaired, but did not correlate with shrinkage of cortex or CP. These results suggest that the long-term spatial cognitive deficit of MCA-occluded rats is in part associated with damage to the cortex involving parietal cortex, and that the change of search strategies is associated with damage to CP. These findings support the idea that different brain regions contribute differently to cognitive performance, search strategies, avoidance task, and neurologic performance, and may be useful for estimating the related region of functional disorder in the clinical situation.
Keywords
Patients with cerebrovascular disease may exhibit cognitive disturbances such as amnesia and topographic disorientation as well as neurologic symptoms (Caplan et al., 1985, 1990). To study the pathophysiology of such symptoms and develop an effective drug therapy, it is necessary to establish a chronic animal model, characterize it using several parameters, and estimate the related brain regions of functional disorders. The middle cerebral artery (MCA) occlusion model in rats developed by Tamura et al. (Tamura et al., 1981a, 1981b; Yamamoto et al., 1988) appears to provide a suitable stroke model, because it produces a relatively uniform infarct area, and behavioral disturbances of learning and memory can be assessed using active and passive avoidance tasks (Hirakawa et al., 1994; Yamamoto et al., 1989a, 1989b), radial maze (Okada et al., 1995b), and Morris maze tasks (Markgraf et al., 1992; Yonemori et al., 1996; Smith et al., 1997), in addition to neurologic deficits (Robinson, 1979; Tamura et al., 1985; Yamamoto et al., 1988; Person et al., 1989).
It is important to study the relationship between infarcted region and functional deficits to estimate the related brain regions of the deficit after MCA occlusion. We previously reported that active avoidance retention latency but not passive avoidance latency correlates with cortical infarct volume (Tamura et al., 1993), and furthermore that in rats trained preoperatively retention of the Morris maze task correlated with brain infarct volume in MCA-occluded rats (Yonemori et al., 1996). Precise and systematic studies using brain-lesioned rats have suggested that different brain regions contribute to spatial cognitive functions (Thompson, 1978; Kolb and Tees, 1990). However, because the damaged area after MCA occlusion in rats is widely spread, it is still unknown what areas within the region of infarction are involved in spatial deficits. The few studies that have focused on this relationship suggest that MCA occlusion is capable of producing spatial cognitive disturbance in rats without any evidence of neurobiologic damage in the hippocampus (Okada et al., 1995a).
In this study, we demonstrate the influence of chronic cerebral infarction on spatial cognition as well as on an avoidance task and neurologic performance in rats, and estimate the location of the infarcted region that correlated with spatial cognitive performance.
MATERIALS AND METHODS
Surgical procedure
This experiment complied with the guidelines of animal experimentation of our laboratories. Male Wistar rats (Slc, Shizuoka, Japan), aged 10 weeks and weighing 260 to 350 g, were anesthetized with 1% to 2% halothane, and the left MCA was permanently occluded at a proximal site (n = 28) with a microbipolar coagulator (Tamura et al., 1981a; Yamamoto et al., 1988). After the incision was sutured, the rats were allowed free access to laboratory chow and water. Sham-operated rats (n = 19) were subjected only to exposure of the MCA, whereas intact control rats (n = 11) underwent no surgery.
Assessment of general behavior
Neurologic examination. Sensorimotor performance was evaluated using a neurologic deficit score based on hemiparesis and abnormal posture at 1, 3, 5, 1, and 10 days and 2, 3, 4, 6, 8, and 10 weeks after surgery. The right hind limb of each rat was extended gently with round-tipped forceps, and the flexor response was evaluated as 0 (normal), 1 (mild deficit), or 2 (severe deficit). For assessment of abnormal posture, rats were suspended by the tail; then forelimb flexion and body twisting were evaluated as 0 (normal), 1 (mild deficit), or 2 (severe deficit). Total score was calculated by adding hemiparesis score and abnormal posture score.
Locomotor activity. Spontaneous locomotor activity was measured for 90 minutes at 1 weeks after surgery using an automated activity box (Automex II; Columbus Instruments, Columbus, OH, U.S.A.).
Passive avoidance task. Rats were tested using a step-through type of passive avoidance task (Yamamoto et al., 1988; Jarvik and Kopp, 1967) at 7 weeks after surgery. The apparatus (Neuroscience Inc., Tokyo, Japan) consisted of two compartments: an illuminated chamber (10 cm × 30 cm) and a dark one (30 cm × 30 cm). The rats initially underwent habituation by being placed in the illuminated chamber and then a door between the chambers was opened so that they could enter the dark chamber. In the acquisition trial, the rats performed the same task, but a 0.60-mA foot shock was delivered through the grid floor for 1 second once they entered the dark chamber. Retention trials were performed without foot shock 1 day and 17 days after the acquisition trial. The step-through latency was recorded to a maximum of 300 seconds.
Morris maze task
Spatial cognition was examined using the Morris maze (Morris, 1981; Morris et al., 1982). A cylindrical tank 144 cm in diameter was filled with water to a depth of 32 cm, and a transparent platform 10 cm in diameter was placed at a fixed position in the center of one of the four quadrants (quadrant 2) and the center of one of the three annuluses (annulus B). The platform was set 2 cm below the water level where the rats could not see it directly. Eight weeks after MCA occlusion, the rats were allowed to swim freely for 1 minute to become habituated. From the next day, acquisition trials were carried out once a day for 10 days. In each trial, the rat was placed into the water at a fixed starting position and the swimming path to escape onto the hidden platform was measured using a Video Image Motion Analyzer (AXIS30; Neuroscience Inc.). The rats were given 120 seconds to find the hidden platform during each acquisition trial, and were allowed to rest on the platform for 30 seconds after they found it. Rats that failed to find the platform within the allotted period were placed onto it by the experimenter, and a time of 120 seconds was assigned. In each trial, the latency to escape onto the hidden platform, the swimming path length, the mean swimming velocity, the heading angle at one body length from start point, and the percent time spent in quadrants and annuluses were measured. To perform further analysis, the number of turns during each trial was measured. Turning was defined as when the rat changed swimming direction more than 90 degrees within one body length, including at the pool wall. Swimming in circles at the same position was counted as one turn. To normalize, turning ratio was calculated as the path length divided by the number of turns.
Histopathology
After all behavioral examinations were completed, the rats were anesthetized with diethyl ether and fixed by perfusion with 10% formaldehyde in 0.1 mol/L phosphate buffer, and their brains were removed. Five coronal blocks of 2 mm thick were cut from 11.2 to 1.2 mm anterior to the interaural line according to the brain atlas of Paxinos and Watson (1982) and embedded in paraffin. Then 4-µm sections were cut coronally from each block and stained with hematoxylin and eosin. The area of cerebral cortex and caudate-putamen (CP) of each hemisphere was determined for individual sections with a digitizer (Nikon, Tokyo, Japan), and the respective total areas were calculated as the sum of individual sections. The degree of shrinkage of left cerebral hemispheric regions was calculated as a percentage relative to right hemispheric regions, to negate for changes in size of infarcted tissue.
Statistical analysis
The results were expressed as mean ± SD for all parameters. To analyze parametric data, repeated measures of analysis of variance (ANOVA) followed by Tukey's post-hoc test was used. When dealing with nonparametric data, Kruskal-Wallis type of ANOVA was used, and comparisons were done by Mann-Whitney U test. Regression analysis was used to investigate correlations.
RESULTS
General behavior
Body weight. No deaths occurred in the MCA occlusion (n = 28), sham operation (n = 19), or intact groups (n = 11) throughout the experimental period. The body weight of the MCA-occluded rats was significantly decreased compared with that of sham-operated and intact rats (group effect, F2,55 = 8.37, P < 0.01; time effect, F11,605 = 779.71, P < 0.01; group * time interaction, F22,605 = 7.14, P < 0.01; two-factor ANOVA for repeated measurements). At 1 week after operation, body weight of MCA-occluded group was 283.7 ± 20.6 g, which was significantly decreased (P < 0.01) from that of the sham-operated (309.7 ± 14.9 g) and intact groups (302.0 ± 10.4 g). However, there was no difference between the MCA-occluded and sham-operated groups at 4, 8, and 10 weeks after surgery (one-factor ANOVA followed by Tukey's test).
Neurologic examination. Middle cerebral artery occlusion significantly disturbed both the posture (Fig. 1A) and flexor response of the contralateral hind limb (P < 0.01, Kruskal-Wallis test followed by Mann-Whitney U test; Fig. 1B) 1 week after surgery. Although the neurologic scores showed gradual recovery, MCA-occluded rats had significantly higher neurologic scores than sham-operated and intact rats even at 10 weeks after surgery. Sham-operated rats showed no apparent abnormalities during the observation period.
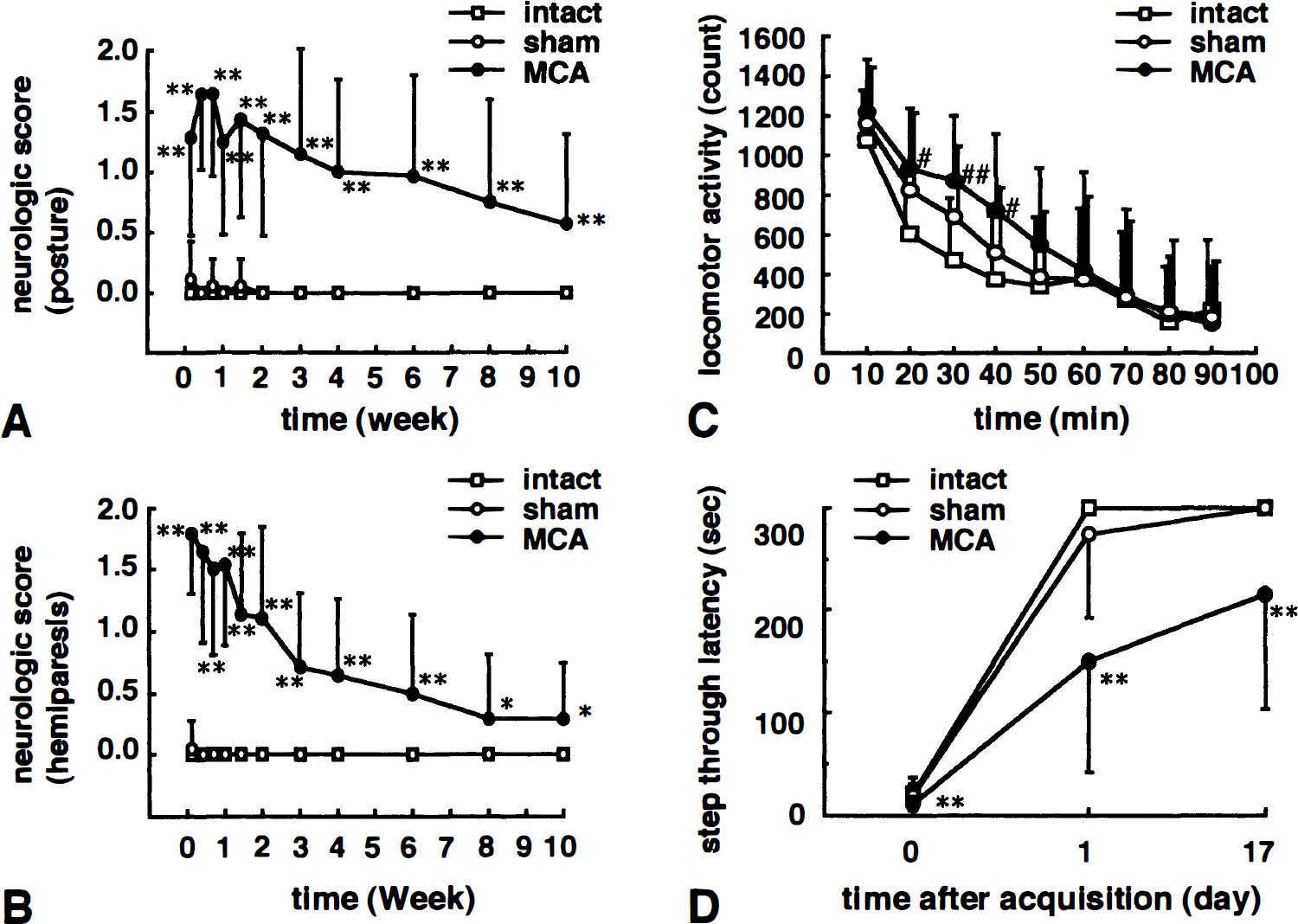
Neurologic and passive avoidance performance in intact, sham-operated, and middle cerebral artery (MCA)- occluded rats. The degree of abnormal posture (
Locomotor activity. At 7 weeks after surgery, 90-minute spontaneous locomotor activity was not significantly different between the three groups (group effect, F2,55 = 2.02, P = 0.142; time effect, F8,440 = 66.63, P < 0.01; group * time interaction, F16,440 = 1.68, P < 0.05; two-factor ANOVA for repeated measurements). But whereas locomotor activity of the three groups was almost the same at the initial 10-minute interval, locomotor activity of MCA-occluded rats at the second to fourth 10-minute intervals tended to be higher than that of intact rats, hence the mild group * time interaction (Fig. 1C).
Passive avoidance task. Seven weeks after surgery, performance in the passive avoidance task was evaluated using the step-through test (Fig. 1D). The acquisition latency of MCA-occluded, sham-operated, and intact rats was 11.2 ± 6.4, 19.0 ± 11.2, and 21.5 ± 15.7 seconds, respectively. In the first retention trial at 1 day after acquisition, MCA-occluded rats had a shorter step-through latency (150.8 ± 108.8 seconds) than sham-operated (273.1 ± 80.7 seconds) and intact rats (300.0 ± 0.0 seconds) (P < 0.01; Kruskal-Wallis test followed by Mann-Whitney U test). When rats underwent the second retention trial 17 days after the acquisition trial, a significantly shorter latency was still observed (P < 0.01; Kruskal-Wallis test followed by Mann-Whitney U test). There were no significant differences between sham-operated and intact rats.
Morris maze task
Search behavior. Eight weeks after surgery, spatial cognitive function was evaluated using the Morris maze. Typical swimming locus of intact, sham-operated, and MCA-occluded rats are shown in Fig. 2. It is noteworthy that MCA-occluded rats showed two typical search patterns; one almost the same as that of sham-operated and intact rats, with several clear turns across the pool (MCA-n), and the other, quite different from sham-operated and intact rats, with a round-shaped locus and fewer turns (MCA-r).
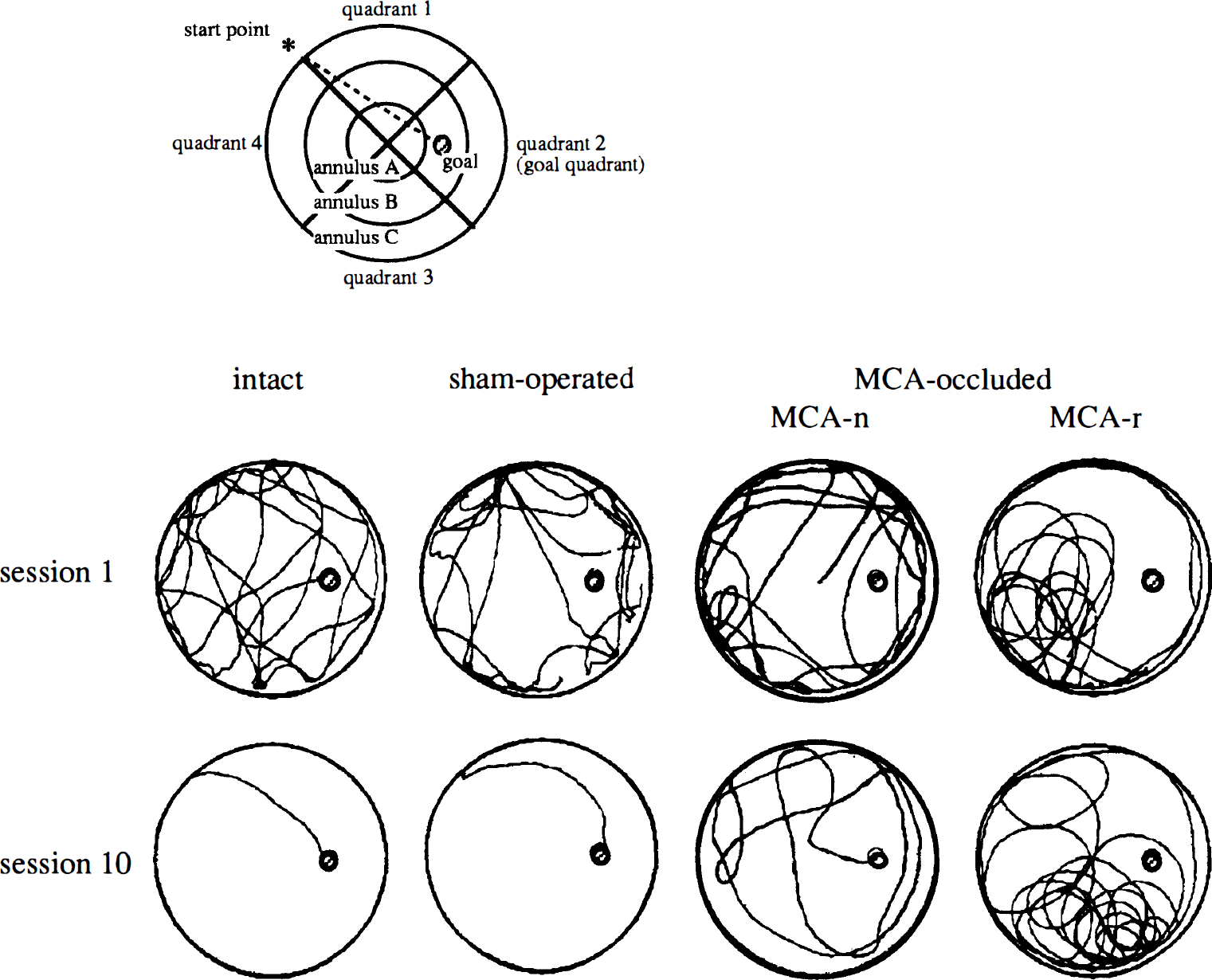
Typical swimming locus of intact, sham-operated, and MCA-occluded rats at first and last session. Open circle indicates the edge of swimming pool, 144 cm in diameter, which was filled with water to a depth of 32 cm (top view). Hatched small circle indicates the transparent goal platform 10 cm in diameter, which was placed at a fixed position in the center of one of the four quadrants (quadrant 2) and the center of one of the three annuluses (annulus B). Rats of all groups swam well and showed search behavior at first session (top panel). Middle cerebral artery-occluded rats took a longer route than intact and sham-operated rats at last session (bottom panel). Middle cerebral artery-occluded rats showed two typical search patterns: one was almost the same as that of sham-operated and intact rats (MCA-n), and the other was round shaped and had less turning behavior (MCA-r).
Cognitive performance. The mean escape latency, swimming path length and swimming velocity onto the hidden platform, heading angle error at one body length from start point, percent time spent in goal quadrant (quadrant 2) and outermost annulus, and turning ratio (path length/number of turns) are shown in Fig. 3. Intact and sham-operated rats performed this task rapidly; 10.0 ± 8.1 and 20.6 ± 24.0 seconds, respectively, to reach the platform in the last session. In contrast, the mean time in the last session for MCA-occluded rats was 62.6 ± 37.9 seconds, with 6 of 28 rats failing to find the platform within 120 seconds. The escape latency showed significant differences (group, F2,55 = 18.26, P < 0.01; session, F9,495 = 11.18, P <0.01; group * session interaction, F18,495 = 2.47, P < 0.01; two-factor ANOVA repeated measurements; Fig. 3A), as did path length (group, F2,55 = 33.41, P < 0.01; session, F9,495 = 14.07, P < 0.01; interaction, F18,495 = 1.34, P = 0.16; Fig. 3B). The percent time spent in goal quadrant (group, F2,55 = 18.30, P < 0.01; session, F9,495 = 4.52, P < 0.01; interaction, F18,495 = 1.92, P < 0.05; Fig. 3C), and heading angle error (group, F2,55 = 22.36, P < 0.01; session, F9,495 = 3.17, P < 0.01; interaction, F18,495 = 3.40, P < 0.01; Fig. 3D) were also significantly affected, indicating that MCA occlusion affected cognitive performance in the Morris maze. Percent time spent in the outermost annulus (group, F2,55 = 23.78, P < 0.01; session, F9,495 = 33.91, P < 0.01; interaction, F18,495 = 4.27, P < 0.01; Fig. 3E) as a parameter for circling around the pool, and turning ratio (group, F2,55 = 10.23, P < 0.01; session, 9,495 = 0.65, P = 0.75; interaction, F18,495 = 0.84, P = 0.65; Fig. 3F) as a parameter for swimming direction change, were also significantly affected. On the other hand, there was no significant difference with respect to swimming velocity (group, F2,55 = 0.14, P = 0.87; session, F9,495 = 1.41, P = 0.18; interaction, F18,495 = 2.52, P < 0.01; Fig. 3G). Further analysis using Kruskal-Wallis test followed by Mann-Whitney U test revealed that these changes in MCA-occluded rats were seen during the last four or five sessions, whereas there was no significant difference between intact rats and sham-operated rats.
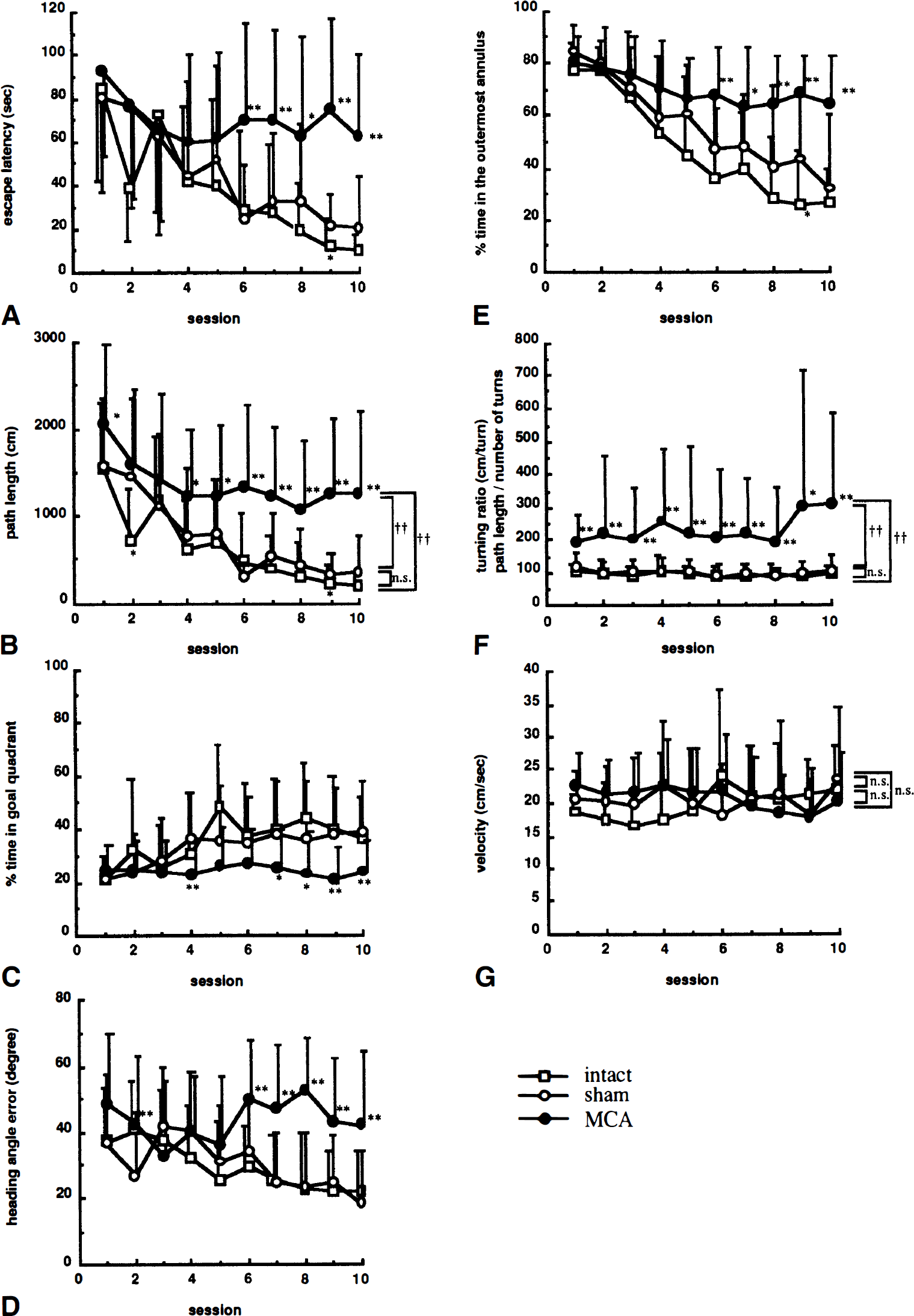
Effects of focal cerebral ischemia on acquisition of the Morris maze task. Each animal was tested one trial per day for 10 consecutive days, and escape latency (
Analysis of differences in search pattern. To investigate whether the two different search patterns seen in MCA-occluded rats affected cognitive performance, we divided the MCA-occluded group into two subgroups according to mean turning ratio (path length/number of turns) with a threshold of 200 cm/turn, because greater than 200 cm/turn was rarely seen in sham-operated or intact rats. Results are shown in Fig. 4. Turning ratio of 12 MCA-occluded rats was more than 200 cm/turn (MCA-r), whereas 16 were less than 200 cm/turn (MCA-n). By definition, the turning ratio, the group dividing parameter, of MCA-r group but not that of MCA-n group was different from the sham-operated and intact groups (group, F3,54 = 48.86, P < 0.01; session, F9,486 = 2.28, P < 0.05; interaction, F27,486 = 2.41, P < 0.01; Fig. 4A). On the other hand, escape latency (group, F3,54 = 13.18, P < 0.01; session, F9,486 = 8.88, P < 0.01; interaction, F27,486 = 1.85, P < 0.01; Fig. 4B), percent time spent in goal quadrant (group, F3,54 = 13.36, P <0.01; session, F9,486 = 2.90, P < 0.01; interaction, F27,486 = 1.36, P = 0.11; Fig. 4C), percent time spent in outermost annulus (group, F3,54 = 15.57, P < 0.01; session, F9,486 = 26.79, P < 0.001; interaction, F27,486 = 3.10, P < 0.01; Fig. 4D), and heading angle error (group, F3,54 = 15,51, P < 0.01; session, F9,486 = 2.54, P < 0.01; interaction, F27,486 = 2.93, P < 0.01; Fig. 4E) of both MCA-occluded groups were significantly different from those of the sham-operated group, with no differences between MCA-r and MCA-n groups (two-factor ANOVA repeated measurement followed by Tukey's test). These results indicate that regardless of whether MCA-occluded rats changed search pattern or not, cognitive performance of MCA-occluded rats was impaired.
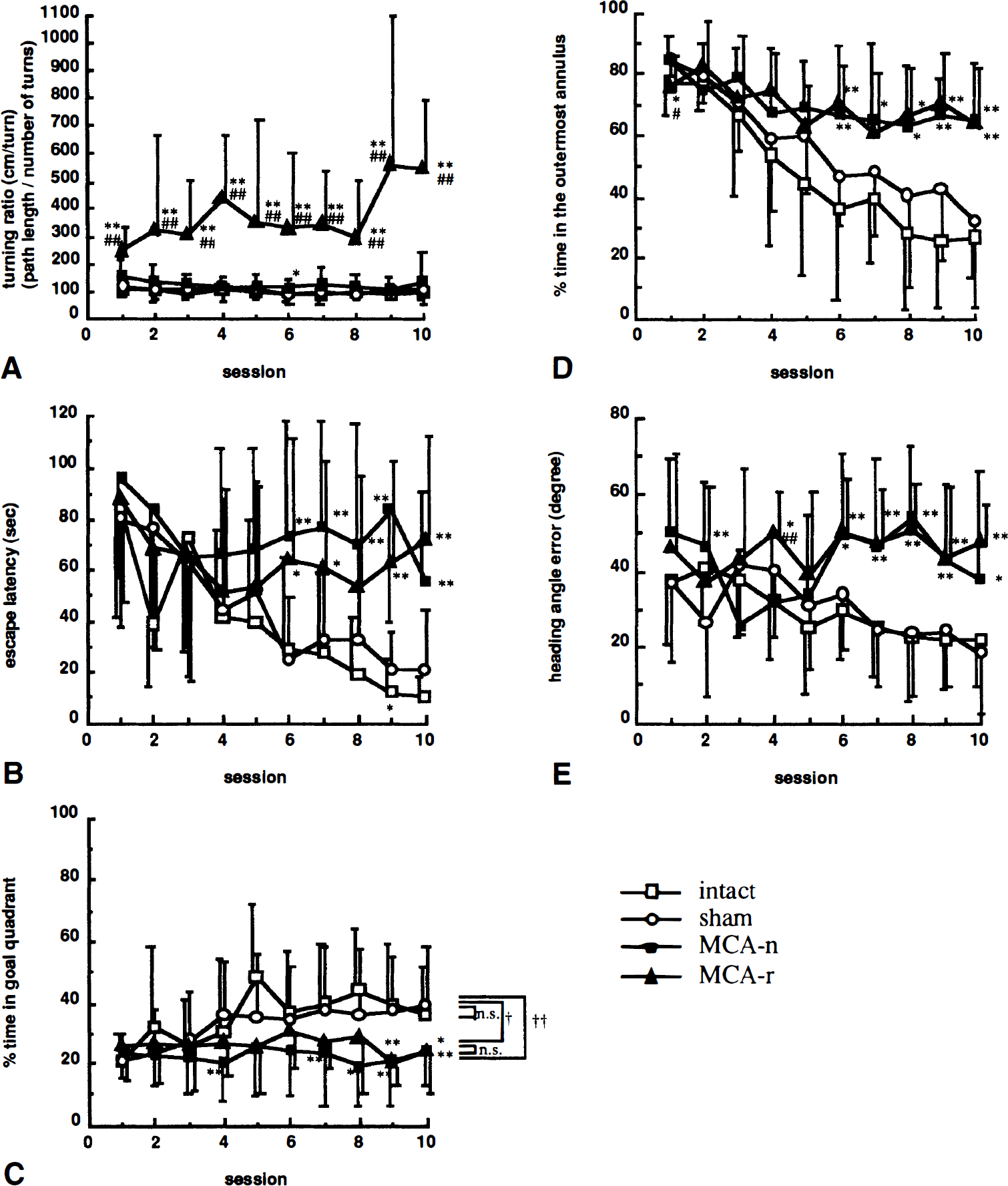
Effect of differences of search pattern on acquisition of the Morris maze task. Middle cerebral artery-occluded rats were divided by turning ratio into two subgroup, MCA-n and MCA-r. Turning ratio (
Correlation analysis
After the behavioral investigation, the degree of shrinkage of cerebral cortex and CP of the ipsilateral hemisphere was determined as a percentage relative to corresponding right hemispheric regions. As the typical example in Fig. 5 shows, the area of cerebral cortex and CP was decreased in MCA-occluded rats. Although there was significant shrinkage in MCA-occluded rats (73.5% ± 10.4%) compared with sham-operated (99.3% ± 3.4%), the ipsilateral hippocampus of MCA-occluded rats was not damaged, as demonstrated in Fig. 6A, including pyramidal cells of the CA1 region (Fig. 6B). There were no significant difference between the shrinkage of MCA-n group (76.4% ± 11.6%) and that of MCA-r group (69.6% ± 7.4%; Student's t test).
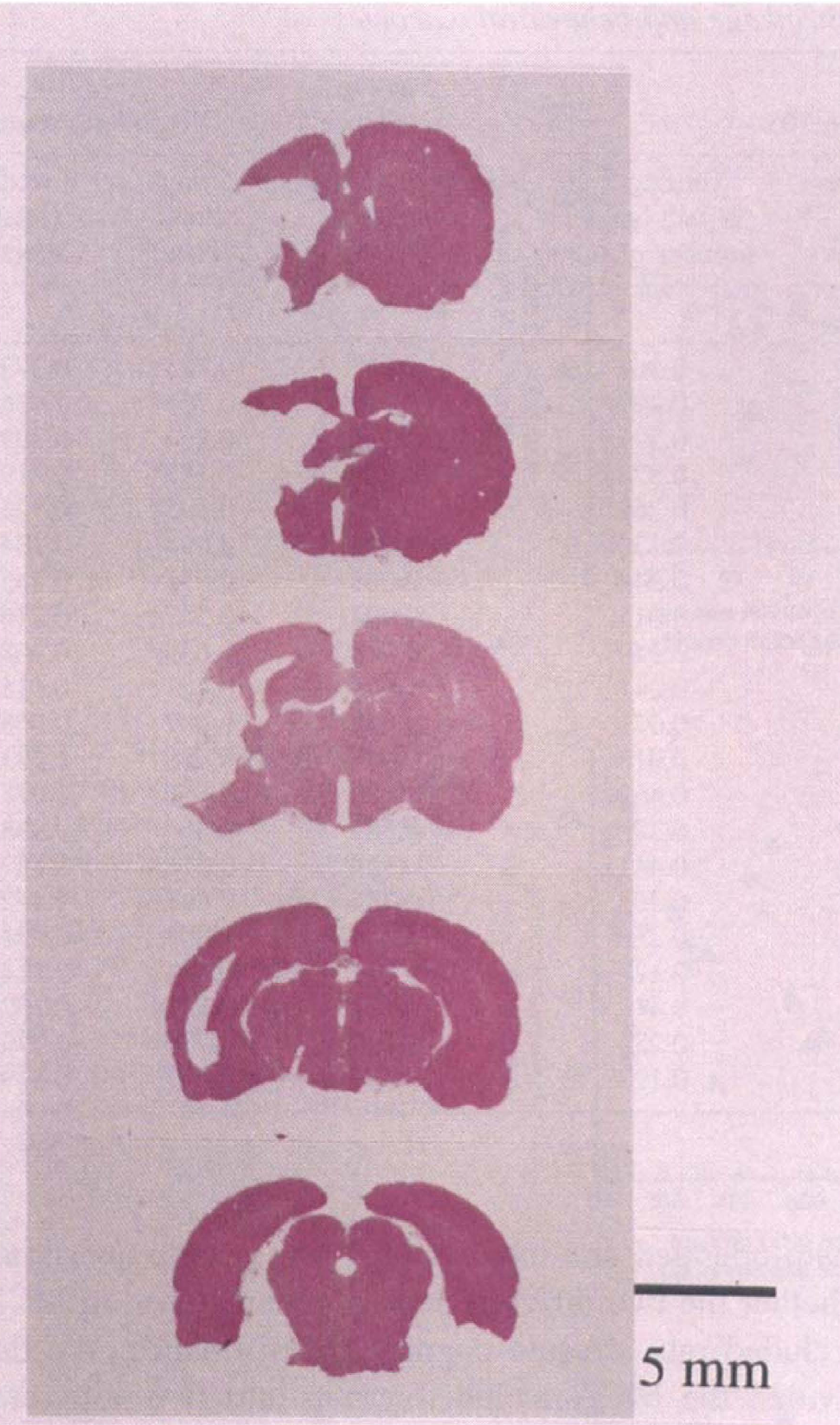
Typical photograph of coronal section of infarcted brain for MCA-occluded rat.
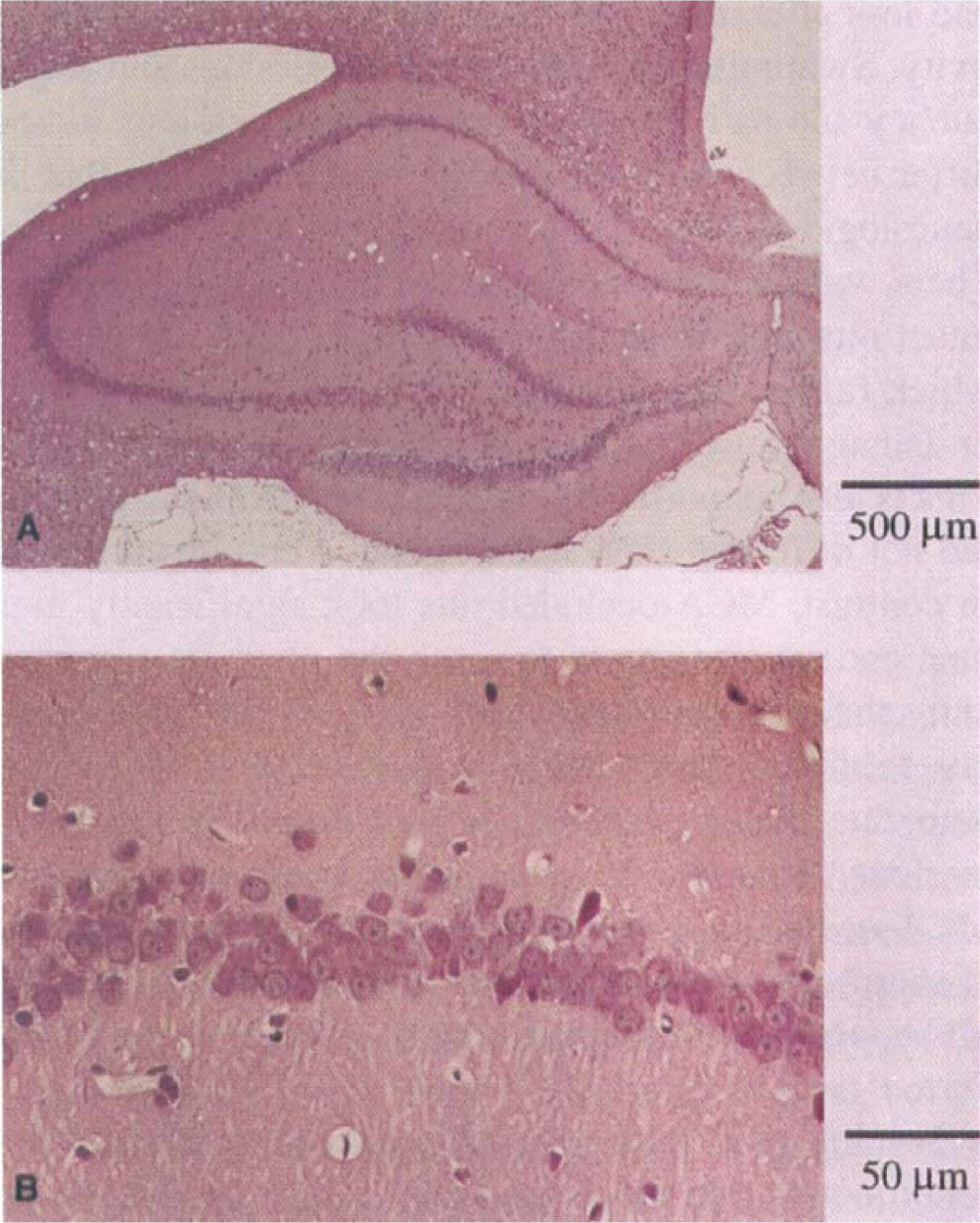
Microscopic photograph of ipsilateral hippocampus region for MCA-occluded rat (same sample as Fig. 5). Hippocampus (
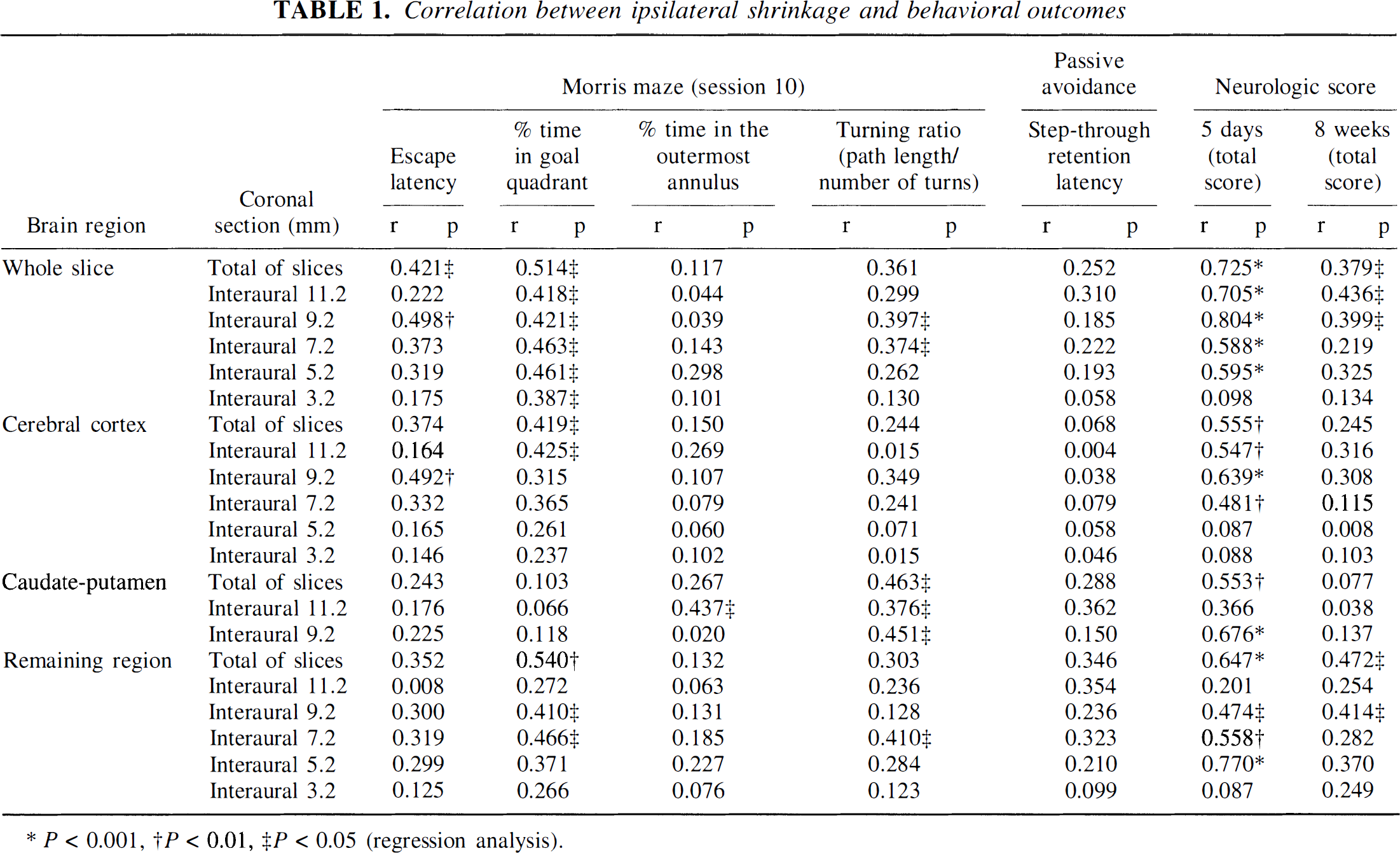
A summary of correlation analysis is shown in Table 1. It was found that the escape latency (r = 0.421, P < 0.05), swimming path length (r = 0.408, P < 0.05), and percent time spent in goal quadrant (r = 0.514, P < 0.05) of last session modestly but significantly correlated with shrinkage of the whole slice, especially with the slice at interaural 9.2 mm (r = 0.498, P < 0.01 for latency; r = 0.521, P < 0.01 for path length; r = 0.421, P < 0.05 for percent time in goal quadrant). We also found that escape latency (r = 0.492, P <0.01; Fig. 7A) and swimming path length (r = 0.483, P < 0.01) significantly correlated with shrinkage of cerebral cortex section from interaural 9.2 mm, but not with that of the CP (r = 0.225, P = 0.25 for latency, Fig. 7D; r = 0.257, P = 0.19 for path length), and that the percent time spent in goal quadrant (r = 0.425, P < 0.05) significantly correlated with shrinkage of cerebral cortex section from interaural 11.2 mm, but not with that of CP (r = 0.066, P = 0.74). On the other hand, turning ratio and percent time spent in the outermost annulus significantly correlated with shrinkage of CP (r = 0.451, P < 0.05; 9.2-mm slice for turning ratio, Fig. 7E; r = 0.437, P < 0.05; 11.2-mm slice for percent time spent in the outermost annulus), but not with shrinkage of cortex (r = 0.349, P = 0.07; 9.2-mm slice for turning ratio, Fig. 7B; r = 0.269, P = 0.17; 11.2-mm slice for percent time spent in the outermost annulus). The swimming velocity and heading error did not correlate with shrinkage of any brain region.
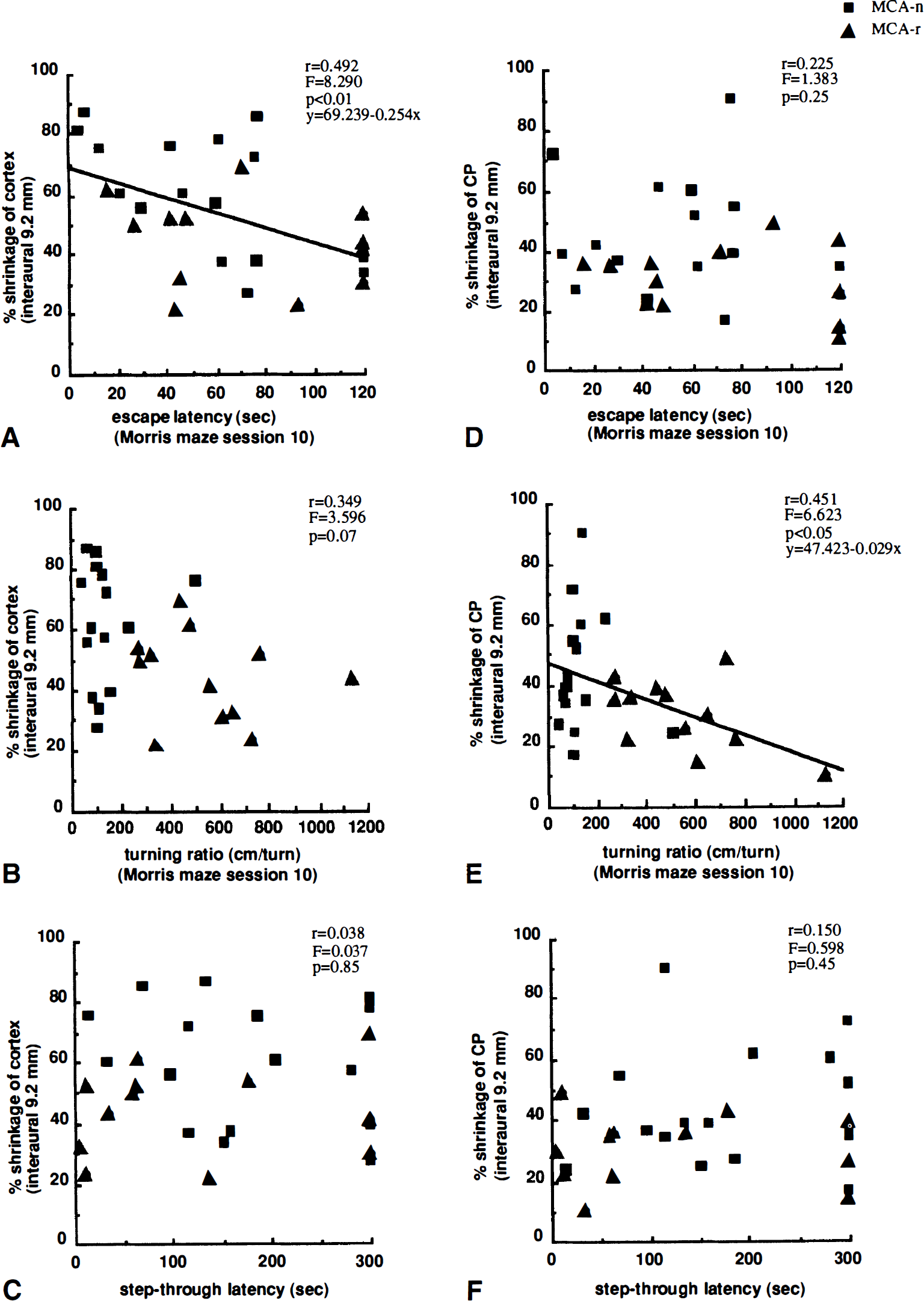
Relationship between shrinkage of brain regions and behavioral outcomes in rats subjected to MCA occlusion (n = 28). Left panels (
Correlation between ipsilateral shrinkage and behavioral outcomes
P < 0.001
P < 0.01
P < 0.05 (regression analysis).
There was also a strong positive correlation between the total neurologic score at day 5 and shrinkage of whole slice (r = 0.725, P < 0.001), cerebral cortex (r = 0.555, P < 0.01), CP (r = 0.553, P < 0.001), and the remaining region (r = 0.647, P < 0.001). The strongest correlation was for the section at interaural 9.2 mm. At 8 weeks after MCA occlusion, although a correlation remained (r = 0.379, P < 0.05; total score for whole slice), posture score (r = 0.400, P < 0.05; whole slice) more strongly correlated with shrinkage than hemiparesis score (r = 0.152, P = 0.44; whole slice). In contrast, the step-through latency in the passive avoidance task did not correlate with shrinkage of whole slice (r = 0.252, P = 0.20), cortex (r = 0.068, P = 0.73), CP (r = 0.288, P = 0.14), or the remaining region (r = 0.346, P = 0.07), including the coronal section at interaural 9.2 mm (Fig. 7C and F). No correlations was observed among Morris maze escape latency, turning ratio, passive avoidance latency, and neurologic scores (Fig. 8).
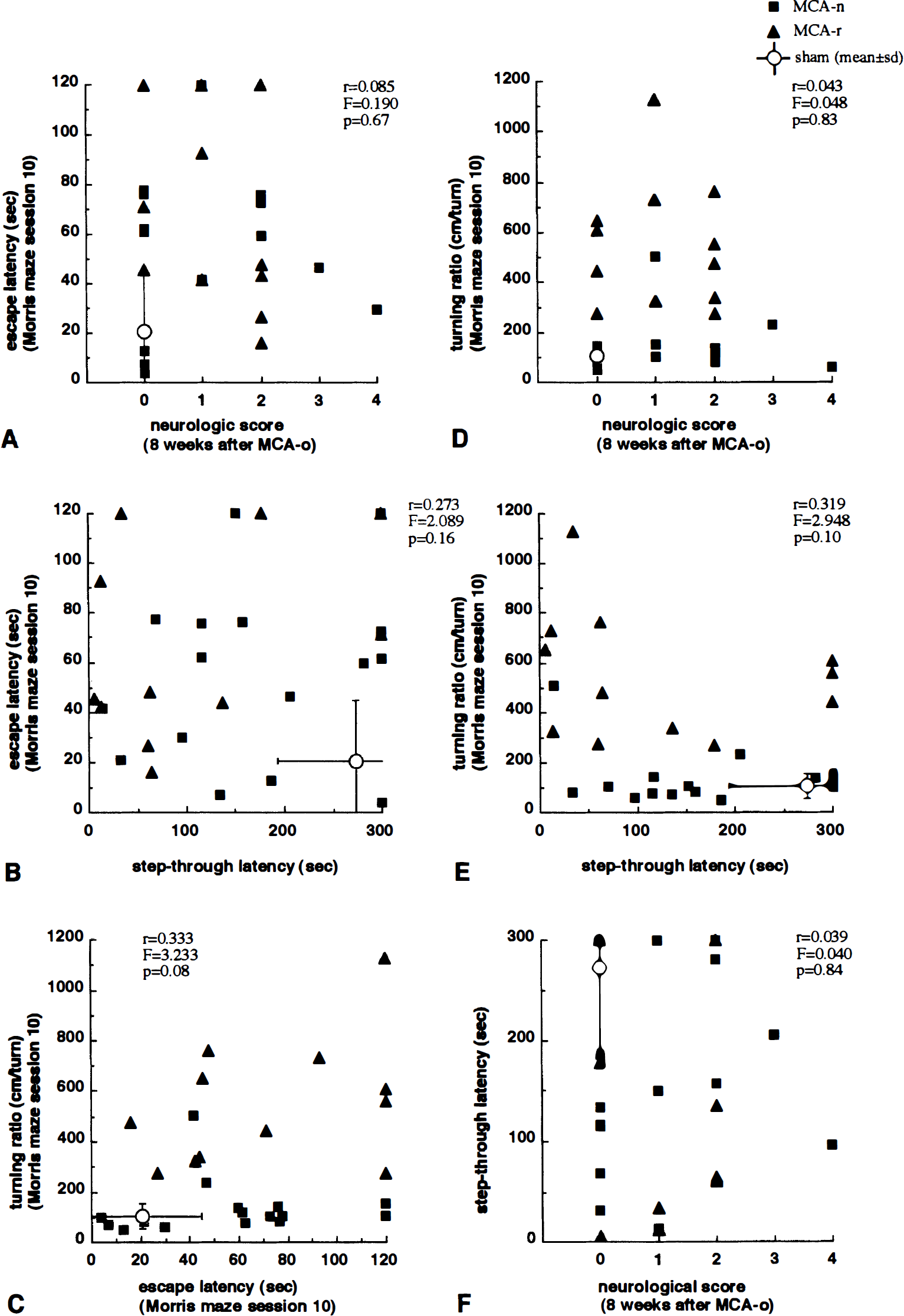
Relationship between performance of Morris maze and other behavioral outcomes in rats with MCA occlusion (n = 28). No correlations were observed among Morris maze escape latency, turning ratio, passive avoidance latency, and neurologic scores.
DISCUSSION
It is well known that functional and pathologic changes are observed in both the acute and chronic phases of focal cerebral ischemia after MCA occlusion in rats (Robinson, 1979; Tamura et al., 1985; Yamamoto et al., 1988; Person et al., 1989). It has also been reported that permanent MCA occlusion disturbs postoperative training of Morris maze (Markgraf et al., 1992), and preoperative trained memory for the Morris maze (Yonemori et al., 1996) and the 8-arm radial maze task (Okada et al., 1995b). Although it is important to identify how ischemic damage in a specific brain region affects behaviors related to learning and memory, few studies have focused on this relationship. Recently Okada et al. (1995a) reported that long-term spatial cognitive performance was disturbed after MCA occlusion in rat without any evidence of neurobiologic damage in the hippocampus. In this study, we investigated impairment of spatial cognitive performance of Morris maze as well as neurologic performance in rats with chronic focal cerebral ischemia, and using correlation analysis, estimated how infarction of brain region related to spatial cognitive function.
Changes in body weight, locomotor activity, neurologic deficits, and passive avoidance task of the MCA-occluded rats observed in this study are in agreement with previous findings (Bederson et al., 1986; Obana et al., 1988; Yamamoto et al., 1988, 1989a, 1989b; Person et al., 1989; Yonemori et al., 1996; Wahl et al., 1992; Tamura et al., 1993; Hirakawa et al., 1994). We evaluated spatial cognitive performance of Morris maze at 8 weeks after the operation, when the residual neurologic deficits were mild and stable and when MCA occlusion had less of an effect on body weight and locomotor activity. Swimming velocity during all sessions and escape latency and path length of the first four sessions did not differ between the three groups, which indicates that the neurologic deficits did not influence this performance. There was no difference between the sham-operated and intact rats with respect to path length or escape latency, which further indicates that loss of sight on the left side or damage caused by surgery did not affect performance. This is also supported by the fact that heading angle error did not differ between sham-operated and intact groups. In contrast, MCA-occluded rats took significantly more time and a longer route to reach the platform compared with sham-operated rats, although all MCA-occluded rats exhibited search behavior and successfully escaped onto the hidden platform at least once during the 10 sessions. Percent time spent in the goal quadrant of the MCA-occluded group was decreased compared with that of sham-operated and intact groups, which indicates that MCA occlusion affected spatial cognition in Morris maze.
Turning ratio (path length/number of turns) and percent time spent in the outermost annulus, which indicates changing of search pattern of rats, were influenced, with MCA-occluded rats swimming circles around the pool and not changing swimming direction compared with sham-operated and intact rats. These results questioned whether the two different search patterns seen in MCA-occluded rats affected cognitive performance. We thus divided the MCA-occluded group into two subgroups according to mean turning ratio. Turning ratio was used as a dividing parameter because circling swimming of some MCA-occluded rats was not restricted to the pool wall, and thus percent time spent in the outermost annulus was not a representative parameter for the changing of search pattern of MCA-occluded rats. When MCA-occluded rats were divided into the two subgroups (MCA-n and MCA-r), the escape latency, swimming path length, percent time spent in goal quadrant and in the outermost annulus, and heading angle error of both subgroups were impaired, with no differences between the two subgroups, suggesting that changes of search pattern did not affect cognition performance, but affected search strategies.
We found that spatial cognitive performance (escape latency, path length, and percent time spent in goal quadrant) in the Morris maze task significantly correlated with shrinkage of cortex, but not with that of CP. The relationship was most apparent in the coronal section from interaural 9.2 to 11.2 mm, involving parietal cortex. This finding indicates that the cortex, at least in part, contributes to spatial cognition in the Morris maze. We also found that turning ratio and percent time spent in the outermost annulus, parameters indicating changes of search pattern, significantly correlated with shrinkage of CP, but not cortex. This indicates that CP, at least in part, contributes to search patterns in the Morris maze. Cognitive performance and search pattern did not correlate with retention latency of passive avoidance task nor neurologic performance, which supports the view that neither disturbance of passive avoidance task nor neurologic deficits affected spatial cognitive performance and search pattern. Whishaw et al. (1987) showed that bilateral lesion of CP produced both impairment of spatial cognition and memory retention of Morris maze in rats, but not prolonged impairment, and suggested that when necessary, medial CP-lesioned rats, like normal rats, can use place strategies for spatial navigation, but if an alternative, perhaps simpler, taxon strategy is available they seemingly ignore place information and navigate using a simpler strategy. They also suggested that the medial CP neural system is involved in the selection of alternative strategies in spatial navigation tasks. Considering that cognitive performance of MCA-occluded rats was impaired regardless of changing search pattern, it is suggested that deficits of spatial cognitive performance of MCA-occluded rat in Morris maze are mainly associated with damage to cortex involving parietal cortex, with less contribution from CP, and that changes of search strategy are mainly associated with CP damage.
Retention latency of passive avoidance task did not correlate with shrinkage of cortex or CP. We have previously reported that preoperative trained retention of Morris maze task but not the passive avoidance task correlates with brain infarct volume in MCA-occluded rats (Yonemori et al., 1996). We also reported that active avoidance retention latency but not passive avoidance latency correlates with cortical infarct volume in MCA-occluded rats (Tamura et al., 1993). Taken together, these findings support the idea that different brain regions contribute differently to cognitive performance, search strategies, avoidance task, and neurologic performance.
In the proximal MCA occlusion model we used, frontal, sensorimotor, auditory, and occipital cortex and CP were infarcted, but the cingulate cortex, hypothalamus, globus pallidus, thalamus, hippocampus, subthalamic nuclei, cerebellum, and pons were spared (Tamura et al., 1981a, 1981b). Systematic investigation using lesion techniques in rats has shown that bilateral lesion of parietal cortex affects Morris maze, card displacement, and active avoidance performances, bilateral lesion of occipitotemporal cortex affects maze, visual discrimination, and active avoidance performances, bilateral lesion of cingulate cortex affects maze and vestibulokinesthetic discrimination performances, and bilateral lesion of frontal cortex affects card displacement but not maze performance (Thompson, 1978). Kolb et al. (1983) suggest that small parietal cortex lesions produce deficits in spatial behavior when a task requires that a particular route be learned, and that small frontal lesions produce deficits when the spatial location of the Morris maze goal must be learned. It has also been reported that bilateral lesions of the parietal lobe (Kesner et al., 1987; DiMattia and Kesner, 1988) produce spatial memory impairment of the task in rats, and Kolb and Tees (1990) further suggested that spatial learning is particularly affected in rats with temporal association cortex lesion. Entorhinal cortex lesions (Schenk and Morris, 1985) produce impairment of both acquisition and retention. Together with these reports and the results of our correlation analysis, the impairment in spatial cognitive performance noted in our study may be associated with damage to frontal, parietal, or occipital cortex after focal cerebral ischemia.
In contrast to these findings, (Kolb et al. 1983, 1990) have suggested that severe impairment in performance of the Morris maze by rats with frontal lesions are of the same magnitude as those observed in rats with hippocampal lesions, but that the frontal cortex is not essential for postoperative retention of learning. This appears to be different from MCA-occluded rats, first, because the medial portion of frontal cortex (cingulate cortex) is spared in MCA occlusion, and second, as we previously reported, because preoperative trained memory retention of Morris maze is also impaired after MCA occlusion (Yonemori et al., 1996). The damage after MCA occlusion in our study appeared strongest in the parietal area, whereas the correlation between spatial cognitive deficits and cortical shrinkage was most apparent in the coronal section from interaural 9.2 mm, which involves parietal, but not occipital cortex (Zilles, 1985). Together, this suggests that damage in parietal cortex after MCA occlusion is most likely associated with the deficits in spatial cognition and that frontal cortex is less important.
The hippocampus, which is not primarily damaged in MCA-occluded rats, is reported to play an important role in spatial memory (Morris et al., 1982). However, Okada et al. (1995a), using histologic and electrophysiologic techniques, observed that MCA occlusion was capable of producing long-term spatial cognitive deficits in rats without significant damage to the hippocampus. We also histologically showed in this study that hippocampus including CA1 region was not affected by MCA occlusion. As there is no evidence that the septohippocampal system receives secondary damage after chronic focal cerebral ischemia, it seems unlikely that the hippocampus is responsible for the spatial cognition deficits in MCA-occluded rats. It is also known that after infarction in the territory of the MCA, in the chronic phase, progressive shrinkage occurs in the ipsilateral thalamus and substantia nigra (Fujie et al., 1990; Tamura et al., 1990; Iizuka et al., 1990), indicating that these areas may also contribute to spatial deficits. Prolonged spatial cognitive disturbance may likely result from damage to several neuronal network systems at the same time rather than a specific brain region.
In summary, we have demonstrated that focal cerebral ischemia causes a prolonged spatial cognitive disturbance in rats that is correlated with shrinkage of the ischemic cortex, especially the coronal section involving parietal cortex, and that a change of search strategies occurs that is correlated with shrinkage of the ischemic CP, and is distinct from neurologic or passive avoidance deficits. The results suggest that the long-term spatial cognitive deficit of MCA-occluded rats is in part associated with damage to the cortex involving parietal cortex, and that change of search strategies of the rats is mainly associated with damage to the CP. These findings support the idea that different brain regions contribute differently to cognitive performance, search strategies, avoidance task, and neurologic performance, and may be useful for estimating the origin of functional disorder in the clinical situation.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Mr. Atsuhiro Uemura and Ms. Satomi Takeuchi for their technical assistance.
