Abstract
It is well known that hemiplegia is frequently observed in cerebral ischemia. It is important for the pathophysiologic study and development of drug therapies to establish a precise method investigating impairment of motor function with animal models. To develop a quantitative and objective method for evaluating impairment of motor function, we examined an inclined plane test after chronic focal cerebral ischemia in the rat. Standard scoring of neurologic deficits has limitations, including problems with quantification and objectivity. The purpose of this study was to establish a novel method for evaluating impairment of motor function in middle cerebral artery (MCA) occluded rats. The left MCA was permanently occluded at a proximal site, and sensorimotor performance was evaluated at the fifth day and every week for 11 weeks thereafter. The ability to maintain body position on an inclined plane was measured when rats were placed on a stainless steel slope in left-headed, right-headed, and up-headed positions. Neurologic examination based on hemiparesis and abnormal posture was also performed. After all behavioral examinations were completed, the degree of shrinkage of the left hemisphere to the contralateral was measured. The ability of MCA-occluded rats to maintain position on an inclined plane in the left-headed position was significantly restricted when compared with that of sham-operated rats throughout the test period (maximum angle of 37° versus 45°, respectively). Minimal natural recovery was observed for all position measurements. MCA-occluded rats showed a significantly higher neurologic score with natural recovery. The ability to maintain position on an inclined plane after MCA occlusion (MCAO) was significantly correlated with the degree of the shrinkage of the ischemic hemisphere and neurologic score. The angle for the left-headed position was most strongly correlated with ipsilateral shrinkage. In the present study, long-lasting impairment of motor function was detected in rats with MCAO, which correlated with the shrinkage of the ischemic hemisphere. Furthermore, a difference in performance depending on body position (left-headed versus right-headed) was also detected. The left-headed position was found to be most sensitive for evaluating this model. The inclined plane test is a quantitative, objective, and sensitive method for evaluating motor deficits after chronic focal cerebral ischemia in rats, and this method may be useful to investigate changes in motor function in hemiplegia.
It is important for the pathophysiologic study and development of drug therapies of stroke to establish convenient methods, and to evaluate impairment of neurologic and motor functions in rodent models because muscle weakness is a common complaint after stroke in humans. The rat middle cerebral artery occlusion (MCAO) model developed by Tamura et al. (Tamura et al., 1981a; Yamamoto et al., 1988; Hirakawa et al., 1994) appears to be a suitable stroke model that induces a relatively constant infarct size. Several investigators have detected a variety of behavioral deficits in this model (Tamura et al., 1985; Yamamoto et al., 1989a, 1989b; Markgraf et al., 1992; Wahl et al., 1992; Hirakawa et al., 1994; Okada et al., 1995; Yonemori et al., 1996). However, reliable methods to evaluate motor function are still under investigation. A neurologic examination based on posture and hemiparesis developed by Bederson et al. (1986), the motor score based on screen test, balance beam test, and prehensile-traction test developed by Combs and D'Alecy (1987) and other tests have been widely used to examine neurologic deficits or motor performance in animal models because they are simple and were developed according to clinical criteria. Although these tests include many parameters to examine total deficits, each parameter tends to be relatively coarse. There are also problems such as quantification and objectivity for measurement in the chronic phase because testing conditions and individuals can vary over time. Because it is thus necessary to develop a fine, objective, and quantitative method, this study was designed to establish a method to evaluate impairment of motor function, especially muscle strength, with middle cerebral artery (MCA)-occluded rats.
METHODS
Surgical procedure
This experiment complied with the Guidelines of Animal Experimentation of our laboratories. Male Wistar rats (Slc, Shizuoka, Japan) 10 weeks old and weighing 250 to 310 g were anesthetized with 1% to 2% halothane and the left MCA was permanently occluded at a proximal site (n = 18) with a microbipolar coagulator (Tamura et al., 1981a; Yamamoto et al., 1988). After the incision was sutured, the rats were allowed free access to laboratory chow and water. Sham-operated rats (n = 12) were subjected only to exposure of the MCA.
Inclined plane test
We evaluated motor performance in rats, using a sliding apparatus (model SN-453, Shinano Co. Ltd., Tokyo, Japan), at 5 days and 1, 2, 3, 4, 5, 6, 7, 8, 10, and 11 weeks after surgery. The sliding apparatus had a 60 × 40-cm stainless steel plane that could be inclined at an angle of 0° (horizontal) to 60°. Each rat was initially placed on the 30°-angled incline plane in the left-headed position (right side up orientation), right-headed position (left side up orientation), or up-headed position (head-up orientation), and the angle of the inclined plane was increased at a rate of 2°/sec from 30° to 60°. The maximum angle was then determined at the moment just when a limb of the rat slipped in order to maintain body position. The test was performed three times for each head position and averaged. Each trial was performed after a 1-minute interval. During this time the rat was returned to a cage filled with dry wooden chips, with which the limbs and body were wiped and dried so as not to influence the friction coefficient by urine and feces. Trials in which the rat turned its body uphill without a limb slipping were not included.
Neurologic examination and spontaneous locomotor activity
We evaluated sensorimotor performance in the rats using a neurologic deficit score based on the detection of hemiparesis and abnormal posture at 5 days and 1, 2, 3, 4, 5, 6, 7, 8, 10, and 11 weeks after surgery. The right hind limb of each rat was extended gently with round-tipped forceps and the flexor response was evaluated as 0 (normal), 1 (slight deficit), 2 (moderate deficit), or 3 (severe deficit). For assessment of posture, rats were suspended by the tail and forelimb flexion and body twisting were evaluated as 0 (normal), 1 (slight twisting), 2 (marked twisting), or 3 (marked twisting and forelimb flexion).
Spontaneous locomotor activity was measured every 5 minutes for 15 minutes at 1, 2, and 7 weeks after surgery using an automated activity box (Automex II; Columbus Instruments, OH).
Histopathology
After the behavioral studies were completed, the rats were anesthetized and fixed by perfusion with 10% formaldehyde in 0.1 mol/L phosphate buffer, and then their brains were removed. Five coronal blocks (2-mm thick) were cut from 12.2 to 4.2 mm anterior to the interaural line according to the brain atlas of Paxinos and Watson (1982), and were embedded in paraffin. Then 4-μm sections were cut from each block and stained with hematoxylin and eosin. The area of each cerebral hemisphere was determined on individual sections with a digitizer (Luzex FS, Nireco, Tokyo, Japan) and the total area of each hemisphere was calculated as the sum of the individual areas. The degree of shrinkage of the left cerebral hemisphere was calculated as a percentage relative to the right hemisphere area.
Statistical analysis
The results are expressed as mean ± SD for all parameters. To analyze parametric data, repeated measures analysis of variance (ANOVA) followed by Tukey-compromise test or Student's t-test was used. For testing the difference between orientations, we use the paired t-test. When dealing with nonparametric data, Mann-Whitney U-test was applied. Regression analysis was used to investigate correlations.
RESULTS
General behavior
There were no deaths in the MCAO (n = 18) or sham-operated group (n = 12) throughout the experimental period. Body weight of the MCAO group was significantly decreased compared with that of the sham-operated group at 5 days after surgery (F = 6.88, P < .05; one-factor ANOVA followed by Student's t-test), although subsequently there was no difference between the two groups (group: F1,28 = 2.10, P = .16; time: F10, 280 = 319.49, P < .001; group*time: F10, 280 = 0.69, P = .74; two-factor ANOVA for repeated measurements). At 1, 2, and 7 weeks after surgery, spontaneous locomotor activity did not significantly differ between the two groups (two-factor ANOVA for repeated measurements).
Inclined plane test
We evaluated motor performance in MCA-occluded rats using an inclined plane test at 5 days and 1, 2, 3, 4, 5, 6, 7, 8, 10, and 11 weeks after surgery. Figure 1 shows the results of the test in MCA-occluded and sham-operated rats. The maximum angles at which position was maintained in the sham-operated rats were 44.4 ± 3.5°, 44.0 ± 2.3° and 42.7 ± 3.2° for left-, right- and up-headed initial body positions, respectively, at 5 days after surgery. A slight increase in slope angle was noted for all initial body positions. Compared to the sham operation, MCAO disturbed performance in this task. First, for the left-headed initial position, the maximum angle to hold position was 37.9 ± 3.2° at 5 days after MCAO, significantly decreased compared with that of sham-operated rats (P < .01, Student's t-test). Thereafter, the angle slightly increased to about 40° at 3 weeks but was still significantly decreased compared with that of sham-operated rats (P < .01, Student's t-test). This decrease was detected throughout the test period, with significant differences between the sham-operated and MCAO groups (group: F1, 28 = 94.84, P < .01; time F10, 280 = 5.40, P < .01; group*time: F10, 280 = 2.08, P < .05; two-factor ANOVA for repeated measurements) (Fig. 1A). Second, for the right-headed initial position, the maximum angle was 41.7 ± 2.3° at 5 days after MCAO, and nearly the same as that of sham-operated rats. However, the performance gradually decreased to less than 40° after 4 weeks (group: F1, 28 = 102.64, P < .01; time F10, 280 = 0.51, P = .88; group*time: F10, 280 = 7.48, P < .01; two-factor ANOVA for repeated measurements) (Fig. 1B). Third, for the up-headed initial position, the level of performance was between that for left-headed and right-headed position (38.9 ± 2.8°, P < .01 versus sham group at 5 days; group: F1, 28 = 72.74, P < .01; time F10, 280 = 3.10, P < .01; group*time: F10, 280 = 4.37, P < .01; two-factor ANOVA for repeated measurements) (Fig. 1C). Concerning the differences between left-headed and right-headed positions seen in MCA-occluded rats, further statistical analysis indicated that there was a significant difference between the performance for the two positions during the first 2 weeks (P < .01 at 5 days and 1 week, P < .05 at 2 weeks; paired t-test), but not after 3 weeks. Sham-operated rats did not show any differences between left-headed and right-headed orientations.
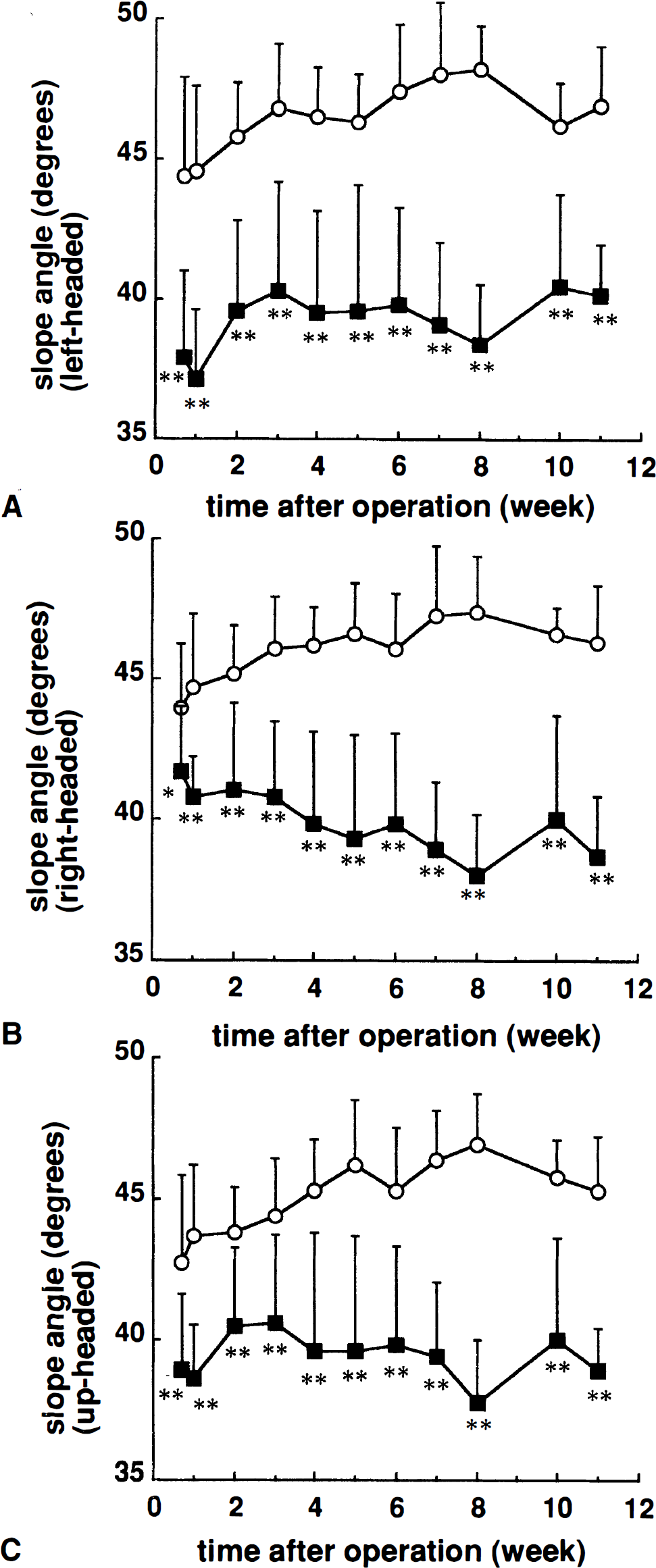
Ability to maintain body position on an inclined plane in sham-operated (○) and middle cerebral artery-occluded (▪) rats. The maximum angle was determined at the moment just when a limb of the rat slipped to maintain its position for the left-headed
Neurologic examination
MCA-occluded rats had a significantly higher neurologic score compared with sham-operated rats (group: F1, 28 = 31.94, P < .001; time: F10, 280 = 16.62, P < .001; group*time: F10, 280 = 12.36, P < .001; two-factor ANOVA for repeated measurements; P < .01 for each time point; Mann-Whitney U-test) (Fig. 2). MCAO also significantly disturbed both the posture and the flexor response of the contralateral hind limb (P < .01, Mann-Whitney U-test). Although the neurologic scores showed gradual recovery, MCA-occluded rats had significantly higher neurologic scores than sham-operated rats even at 11 weeks after surgery.
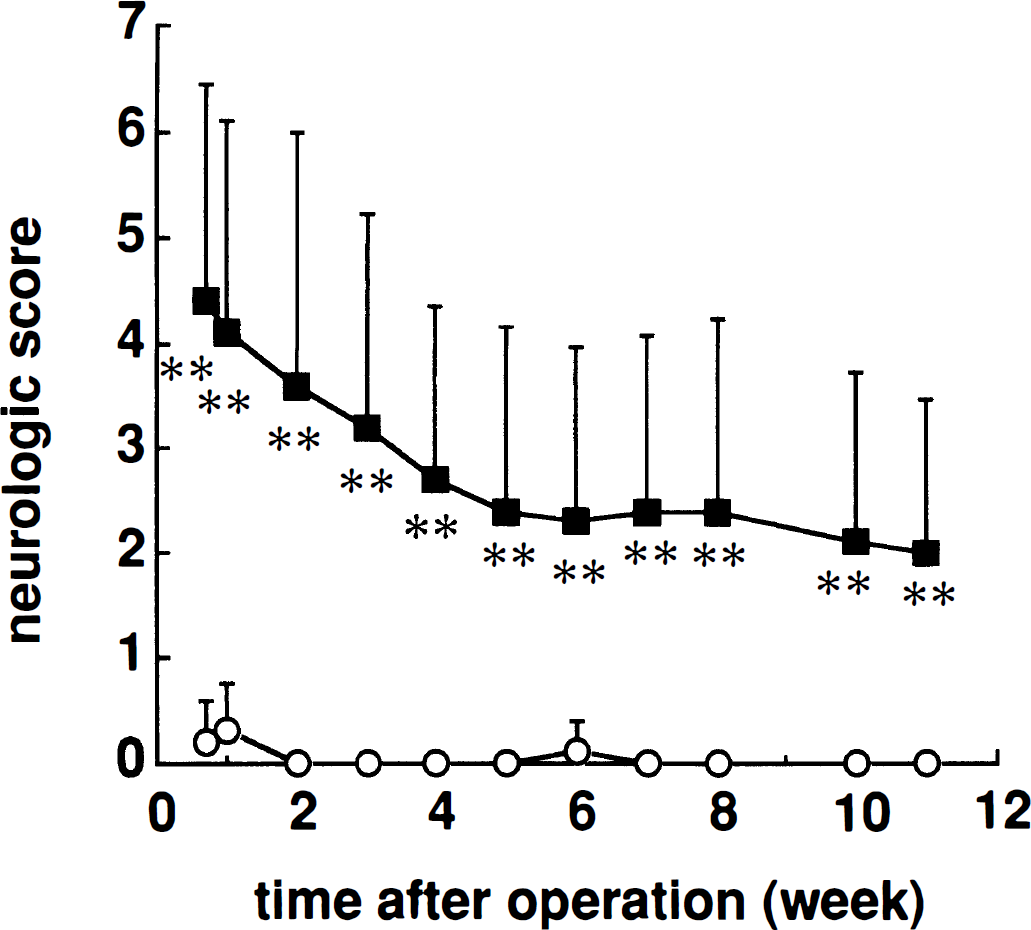
Neurologic deficits in sham-operated (○) and middle cerebral artery-occluded (▪) rats. Abnormal posture and hemiparesis were graded from 0 to 3, and the total scores were calculated. Each point represents the mean ± SD (n = 12 to 18) of total score. The neurologic score was significantly increased in the middle cerebral artery occlusion group compared with the sham-operated group throughout the test period.
Correlation analysis
The total area of ipsilateral and contralateral hemisphere were 122.9 ± 29.3 mm2 and 187.5 ± 10.2 mm2 in MCA-occluded rats, and 197.4 ± 11.9 mm2 and 199.6 ± 12.6 mm2 in sham-operated rats, respectively. The percent area of the left cerebral hemisphere relative to that of the right hemisphere in the MCAO group was 65.1 ± 12.8%, which was significantly smaller than that in the sham-operated group (98.9 ± 2.3%). The ability to maintain position on an inclined plane at 2 to 11 weeks after MCAO was significantly correlated to the ipsilateral shrinkages for all initial body positions (Fig. 3). The correlation was strongest in the slice at interaural 10.2 mm, and was stronger for the left-headed position (r = 0.84, P < .01 at 2 weeks) than for the right-headed (r = 0.65, P < .01) or up-headed (r = 0.69, P < .01) positions. Furthermore, there was also a strong correlation between the neurologic scores and the maximum slope angles 2 to 10 weeks after MCAO (Fig. 4). This correlation was seen in hemiparesis (r = 0.85, P < .01 for the left-headed position at 2 weeks), posture (r = 0.89, P < .01), and total score (r = 0.90, P < .01). This correlation was strongest for the left-headed position (r = 0.90, P < .01 versus r = 0.65, P < .01 for right-headed and r = 0.64 for up-headed position at 2 weeks).
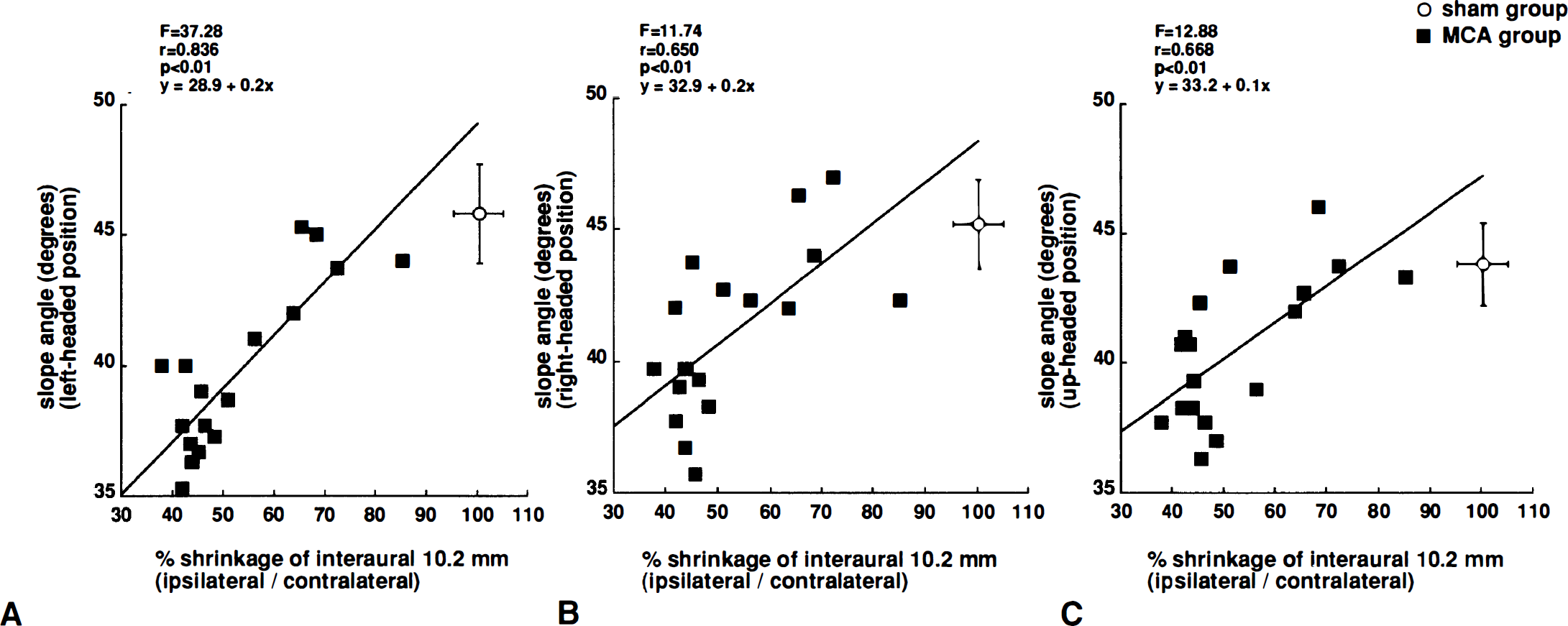
Relationship between ipsilateral shrinkage and maximum angle of inclined plane in rats with middle cerebral artery occlusion. Data from middle cerebral artery-occluded rats (n = 18; filled squares) were used for regression analysis, while data from sham-operated rats (n = 12; open circle; mean ± SD) were excluded. Ipsilateral shrinkage and maximum angle for the left-headed
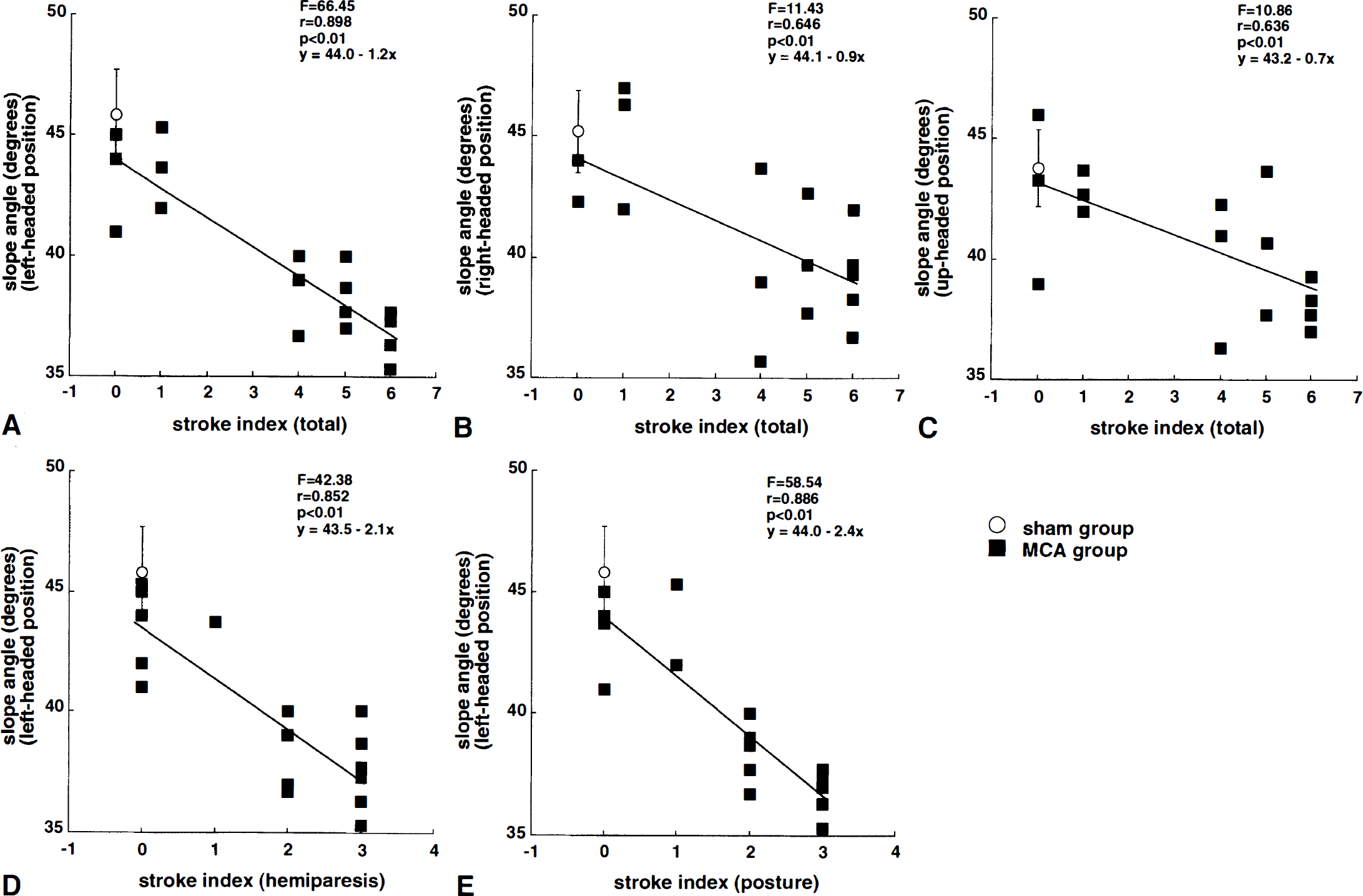
The relationship between maximum angle of inclined plane and neurologic score in rats with middle cerebral artery occlusion is shown. Data from middle cerebral artery-occluded rats (n = 18; filled squares) were used for regression analysis, while data from sham-operated rats (n = 12; open circle; mean ± SD) were excluded. Total neurologic score and maximum angle for the left-headed
DISCUSSION
We examined the ability of rats to maintain body position on an inclined plane after focal cerebral ischemia to develop a quantitative, objective, and sensitive method for evaluating impairment of motor function. We used the proximal permanent MCAO model developed by Tamura et al. (Tamura et al., 1981a; Yamamoto et al., 1988; Hirakawa et al., 1994) because the infarct size induced is relatively constant and because this model is widely used to evaluate behavioral deficits after MCAO (Yamamoto et al., 1989a, 1989b; Markgraf et al., 1992; Wahl et al., 1992; Hirakawa et al., 1994; Okada et al., 1995; Yonemori et al., 1996). Using the inclined plane test, we detected a long-lasting impairment of motor function in this model, and also observed performance differences depending on the position of the rat. Various methods have been developed and widely used to examine neurologic deficits or alterations in motor performance in animal stroke models, including the neurologic examination based on posture and hemiparesis developed by Bederson et al. (1986), motor score based on screen test, balance beam test and prehensile-traction test developed by Combs and D'Alecy (1987), beam walking test by Feeney et al (1982), and the limb-placing test developed by De Ryck et al. (1989). These methods were easily applied and improved on according to clinical criteria, although these tests include many parameters as a battery of tests to evaluate total deficits caused by ischemia; parameters such as time, frequency, and degree of tonus are converted into scores and coarsely evaluated. In this regard the method developed in this study is quantitative, objective, and sensitive, for the following reasons. First, the parameter was the numerical angle at which the animal maintained body position, which is also useful for statistical analysis. Second, the angle was mechanically obtained, which is useful for chronic deficits measurement after ischemia because long-lasting motor deficits can be recorded irrespective of who and when testing takes place. Finally, this test is sensitive enough to detect differences between left-headed and right-headed initial positions, as shown in this study. This inclined plane test in MCA-occluded rats may thus be useful in a battery of tests as an indicator of motor functions.
It has been reported that the inclined plane test is influenced by both the friction of inclined plane and the initial slope angle (Allmark and Bachinski, 1949). We used a stainless steel plane with which the rat was unable to grip, and between trials the rat was returned to a cage filled with dry wooden chips with which the limbs and body of the rat were wiped and dried so as not to influence the friction coefficient. We set the initial plane angle as 30° because rats could move spontaneously at angles less than 30°. Under the experimental conditions, the ability of MCA-occluded rats to maintain position on the inclined plane was significantly decreased as compared with that of sham-operated rats throughout the test period. Because spontaneous locomotor activity of the MCA group did not differ from that of the sham group throughout the test period, and because we measured the angle at the moment when a limb of the rat just slipped, it is considered that the decreased inclined plane performance in MCA-occluded rats represents a decrease in muscle strength. This conclusion is supported by reports that the inclined plane test serves as an indicator of general muscle strength by measuring maintenance of up-headed body position (Allmark and Bachinski, 1949; Dean et al., 1981; Combs and D'Alecy, 1987). However, the inclined plane has also been used in equilibrium evaluation, especially negative geotaxis, by measuring the time taken to orient head-up from downhill initial position (Wahl et al., 1992; Capdeville et al., 1986). Motor coordination, balance, or other motor abilities may be required for rats to perform this task in horizontal body positions (Adams et al., 1985; Altman and Sudarshan, 1975). As the performance in vertical initial position (up-headed position) may be less affected by negative geotaxis (Crozier and Pincus, 1926), the angle at the moment when a limb of the rat just slipped in up-headed position mainly reflected muscle strength. On the other hand, the performances in horizontal initial body position may be affected not only by muscle strength but also by postural adjustments and by limb placing which may be as adequate an explanation as muscle weakness (Kawamata et al., 1967). Exact measurement of muscle strength in a rodent model is important because muscle weakness is a common complaint after stroke in humans. Therefore, our method is considered useful to estimate clinical statement. Furthermore, this test seems to be very sensitive in the left-headed position for the left MCA-occluded rats. This specific character is useful for detecting the motor impairment of MCA-occluded rats because the impairment measured in the left-headed position was clearly detected earlier than that in the others position. Although it has been reported that proximal MCAO does not affect muscle strength as measured by rotating screen (Markgraf et al. 1992), Tominaga and Ohnishi (1989) reported that the inclined plane test to measure time spent on the board was affected by the reperfusion model of MCAO. These contradictory results may be due to differences in measuring methods, especially using a plane with or without a grid, and that impairment of muscle strength after proximal MCAO can be detected by our method of measuring the angle on an inclined plane. In this regard, our method is considered to be sensitive in detecting the impairment of limb muscle strength and/or motor coordination.
In the inclined plane test, there was a strong positive correlation between angle to maintain position on an inclined plane at 2 to 11 weeks after MCAO and shrinkage of the ischemic hemisphere. The relationship was most apparent for the coronal slice at interaural 10.2 mm, which contains the parietal cortex and caudate putamen, and was also most apparent for the left-headed position. In this study, posture, hemiparesis, and total score of neurologic examination also showed strong correlation with shrinkage of the ischemic hemisphere, especially with that of the coronal slice at interaural 10.2 mm (data not shown). These results are consistent with those of Bederson et al. (1986) and Persson et al. (1989). In the proximal MCAO model we used, frontal, sensorimotor, auditory and occipital cortex, and caudate putamen were infarcted (Tamura et al., 1981a,1981b). Taken together, it is considered that the deficits of motor performance observed in the inclined plane test after focal cerebral ischemia are due to damage of the frontal motor cortex center of the pyramidal tract or of the caudate putamen center of the extrapyramidal tract or to a combination of both. In addition, performance on the inclined plane in the left-headed position might reflect well the damage after left MCAO in rats, as compared with the other initial body positions.
In this study we also found differences with orientation, i.e., the ability to maintain body position on an inclined plane for the left-headed position had already decreased since 5 days after MCAO, while on the other hand, for right-headed position it was nearly the same as that of sham-operated rats at 5 days, and thereafter gradually decreased to that for the left-headed position. After traumatic spinal cord injury in rats, Faden (1989) showed differences in the performance between vertical and horizontal positions but found no differences between left and right positions. If this difference between Faden's result and ours is considered to be caused by differences of animal models, MCAO may be a suitable model for evaluating hemiparesis. The reason for the difference of orientation is still unknown, but two possibilities are considered. The first possibility is that the upper-side limbs are more important to maintain body on an inclined plane. In this case, the reason why inclined plane performance gradually decreased in the right-headed position is considered to be that the tonus of the normal side limb of MCA-occluded rat gradually decreased because of secondary damage or that the center of gravity was changed to maintain the body side balance caused by unbalance compensation. This idea is supported by (1) the finding that the performance for the left-headed initial body position (right side-up orientation; damaged limbs on upper side) was more affected in the left MCAO, and (2) the fact that no hemiparesis occurs on left side limbs in left MCAO in rats. In our preliminary study, there was a decrease of tonus of the right hind-limb in left MCA-occluded rats compared to that of sham-operated rats after 6 weeks, while there was no difference in left hind-limb tonus. On the other hand, our preliminary data showed that the tonus of the normal side hind-limb was slightly decreased compared with the sham-operated rat at 10 weeks after MCAO (data not shown). The second possibility is related to equilibrium, i.e., the conflict between negative geotaxis and ipsiversive movement asymmetry could cause the rats to slip more readily in the left-headed position. When the intact limb is uphill (right-headed position), the rats tend to initiate movement using the intact limb to be in the uphill direction due to geotaxis. Whereas in the left-headed position, the intact limb is on the downhill side and the rat would tend to move ipsiversively due to their injury-related asymmetry of movement, which would be in the nonpreferred downhill direction according to negative geotaxis. In this case, gradual decrease of performance in right-headed position may be attributed to asymmetry of movement or negative geotaxis or a combination of both. It is reported that negative geotaxis is not affected by MCAO (Wahl et al., 1992), and that asymmetry of movement is apparently detected after MCAO, and then gradually decreases. As side difference was detected within a week, it might be related to alteration of the substantia nigra (SN) after MCAO. Schallert et al. (1982) reported that electrical nigrostriatal damage affected negative geotaxis. Tamura et al. indicated that proximal MCAO did not infarct SN, but upregulated the ipsilateral regional CBF (Tamura et al., 1981a,1981b). We also detected circling behavior during the first 2 days, which is typical behavior after SN damage.
Limited natural recovery of motor deficits after MCAO was observed for the left-headed position, while the deficit gradually increased for the right-headed initial position. This phenomenon is distinguished from the neurologic examination (posture and hemiparesis) in which, although neurologic deficits remained at 11 weeks after MCAO, the deficits gradually decreased. This phenomenon is possibly related to changes in the intact hemisphere, which could improve functioning of the uninvolved limb, or which could cause chronic deficits based on secondary damage. It has been noted that after infarction in the territory of the MCA, in the chronic phase progressive shrinkage occurs in the ipsilateral thalamus and SN (Fujie et al., 1990; Tamura et al., 1990; Iizuka et al., 1990). Thus, secondary damage or diaschisis of the contralateral hemisphere may contribute to this phenomenon (Andrews et al., 1991; Feeney and Baron, 1986).
In summary, we developed a method to evaluate motor function after chronic focal cerebral ischemia in rats using an inclined plane test. After MCAO, a long-lasting impairment of motor function was detected that was correlated with shrinkage of the ischemic hemisphere. A difference caused by initial body position was also detected, with the left-headed initial position having the highest correlation. This method is quantitative, objective, and sensitive for evaluating impairment of motor function, and may be useful for pathophysiologic investigation and the development of drug therapies for hemiplegia after cerebral ischemia.
Footnotes
Acknowledgements
The authors thank Mr. Atsuhiro Uemura and Ms. Satomi Takeuchi for their technical assistance.
