Abstract
A short duration of ischemia (i.e., ischemic preconditioning) results in significant brain protection to subsequent severe ischemic insult. Because previous studies suggest that tumor necrosis factor-α (TNF-α) plays a role in both promoting ischemic damage and neuroprotection, the present work aimed to evaluate the expression of TNF-α mRNA in an established model of ischemic preconditioning using a transient 10-minute occlusion of the middle cerebral artery. Because the level of TNF-α mRNA expression in the brain was too low to be consistently detected by Northern technique, a real-time polymerase chain reaction method was applied to quantitate the absolute copy number of TNF-α transcript in rat brain after the preconditioning procedure. TNF-α mRNA was induced in the ipsilateral cortex as early as 1 hour (27 ± 1 copies of mRNA per microgram of tissue compared to 11 ± 3 copies in sham-operated samples) after preconditioning, reached a peak level at 6 hours (49 ± 10 copies of transcript, n = 4, P < 0.01), and persisted up to 2 days. These data not only demonstrate the utility of real-time polymerase chain reaction for sensitive and accurate measurement of mRNA expression in normal and injured tissues but also suggest a potential role of TNF-α in the phenomenon of ischemic preconditioning.
Tumor necrosis factor α (TNF-α) is a pleitrophic cytokine with diverse biologic activities. TNF-α is expressed in monocytes/macrophages as well as many other cells including glia and neurons (Akira et al., 1990; Liu et al., 1994; Munozfernandez and Fresno, 1998). Whereas normal brain tissue has little or no TNF-α expression, this cytokine is significantly induced after transient or permanent focal cerebral ischemia (Liu et al., 1994; Wang et al., 1994; Buttini et al., 1996). In addition, TNF-α receptors are also present in the brain and may mediate neuronal and glial activation, proliferation, differentiation, and survival (Munozfernandez and Fresno, 1998). Furthermore, the expression of TNFR2 was demonstrated to be upregulated during cerebral infection (Lucas et al., 1997).
Although the precise role of TNF-α in brain function and especially in stroke is not fully understood, it has been suggested that TNF-α expression in brain ischemia is detrimental to neurons because the administration of anti-TNF-α antibodies or TNF receptor linked to polyethylene glycol (TNFbp, which binds and inhibits TNF-α) significantly reduced focal cerebral ischemic injury (Dawson et al., 1996; Barone et al., 1997; Nawashiro et al., 1997a). In addition, in vivo administration of TNF-α into brain parenchyma produced significant inflammatory reaction in brain capillaries including pericapillary edema and significant leukocyte accumulation (Feuerstein et al., 1994; Liu et al., 1994). TNF-α administration into the cerebroventricular space before ischemia augments the extent of tissue damage and neurologic deficits (Barone et al., 1997). These results are in agreement with early studies that suggested TNF-α might play a crucial role in tissue injury during disease processes when it is released from activated lymphocytes, macrophages of peripheral origin, and/or astrocytes and microglia of the brain parenchyma (Martin et al., 1995; Probert et al., 1995).
On the other hand, there is increasing evidence to suggest that TNF-α may be involved in neuroprotection (Barger et al., 1995). For example, TNF receptor knock-out mice developed larger lesions after middle cerebral artery occlusion (MCAO) than nontransgenic controls (Bruce et al., 1996). Furthermore, mice treated with TNF-α are resistant to focal cerebral ischemia injury (Nawashiro et al., 1997b). A similar neuroprotective function (Ohtsuki et al., 1996) has been demonstrated for another inflammatory cytokine, interleukin-1β, that was induced after ischemic injury and ischemic tolerance (Minami et al., 1992; Rothwell and Strijbos, 1995; Yamasaki et al., 1995; Ohtsuki et al., 1996; Wang et al., 1999).
Taken together, these data suggest that TNF-α may play a role in both ischemic injury and in tolerance induction. To further explore the role of TNF-α in ischemic tolerance, in the present study we examined TNF-α mRNA expression in a rat model of preconditioning. In this preconditioning model, a transient 10-minute MCAO (used as preconditioning) induced tolerance to subsequent (24 hours later) permanent MCAO, which persists up to 7 days (Barone et al., 1998). Under these conditions, both infarct size and neurologic deficits are remarkedly reduced by preconditioning. This preconditioning paradigm is dependent on de novo protein synthesis (Barone et al., 1998).
Because our pilot studies revealed that the levels of TNF-α mRNA expression after preconditioning was inconsistently detectable by standard Northern hybridization or semiquantitative polymerase chain reaction (PCR) methods (Wang et al., 1997), we adopted a novel real-time PCR technique (Gibson et al., 1996; Heid et al., 1996) for precise quantitation of TNF-α mRNA expression in brain tolerance. This method utilizes a specific fluorogenic probe to hybridize the target sequence that is amplified with a pair of specific PCR primers. The hybridization signal is detected during PCR cycling in proportion to the accumulation of the PCR products. The end point measurement of fluorescence in each sample provides a homogeneous signal that is specifically associated with the amplified target and quantitatively related to the amount of PCR products. By using a cloned cDNA as a template standard and meanwhile normalizing RNA samples with a housekeeping gene for the real-time PCR as described in this report, the absolute copy number of TNF-α transcripts in the cortical samples after preconditioning was determined.
MATERIALS AND METHODS
Animals
Rats were housed and cared for in accordance with the Guide for the Care and Use of Laboratory Animals [DHEW (DHHS) Publication No. (NIH) 85-23, revised 1985, Office of Science and Health Reports, DRR/NIH, Bethesda, MD 20205]. Procedures using lab animals were approved by the Institutional Animal Care and Use Committee of SmithKline Beecham Pharmaceuticals.
Focal ischemic preconditioning
Transient MCAO or sham surgery was carried out in spontaneously hypertensive rats, 300 to 350 g, under sodium pentobarbital anesthesia as described previously (Barone et al., 1992). Briefly, after craniotomy using stereotaxic procedures, the dura was opened over the right MCA. The bent tip of a platinum-iridium wire mounted on a micromanipulator was placed under the MCA and used to lift the artery away from the brain surface to temporarily occlude blood flow, as verified by monitoring cortical microvascular perfusion (Barone et al., 1992). A period of 10 minutes of temporary MCAO was utilized for focal preconditioning because this short ischemic period results in no tissue injury but protects the tissue from late ischemia (Barone et al., 1998).
TaqMan real-time reverse transcription-polymerase chain reaction assay
Rat cortex (210 to 260 mg each) was dissected and immediately frozen in liquid nitrogen and stored at −80°C. Total RNA was prepared by homogenizing the cortical tissues in an acid guanidinium thiocyanate solution and extracted with phenol and chloroform as previously described (Chomczynski and Sacchi, 1987). Average RNA yield was about 1 μg RNA per milligram brain tissue.
The PCR primers and TaqMan probes for TNF-α and rpL32 were designed using a software program from Perkin-Elmer (Foster City, CA, U.S.A.) according to the published rat TNF-α and rpL32 cDNA sequences (S40199; Rajchel et al., 1988) and summarized in Table 1. The specificity of PCR primers was tested under normal PCR conditions in a thermocycler (Model 9700, Perkin-Elmer) before TaqMan quantitation.
Summary of the PCR primers and TaqMan probes
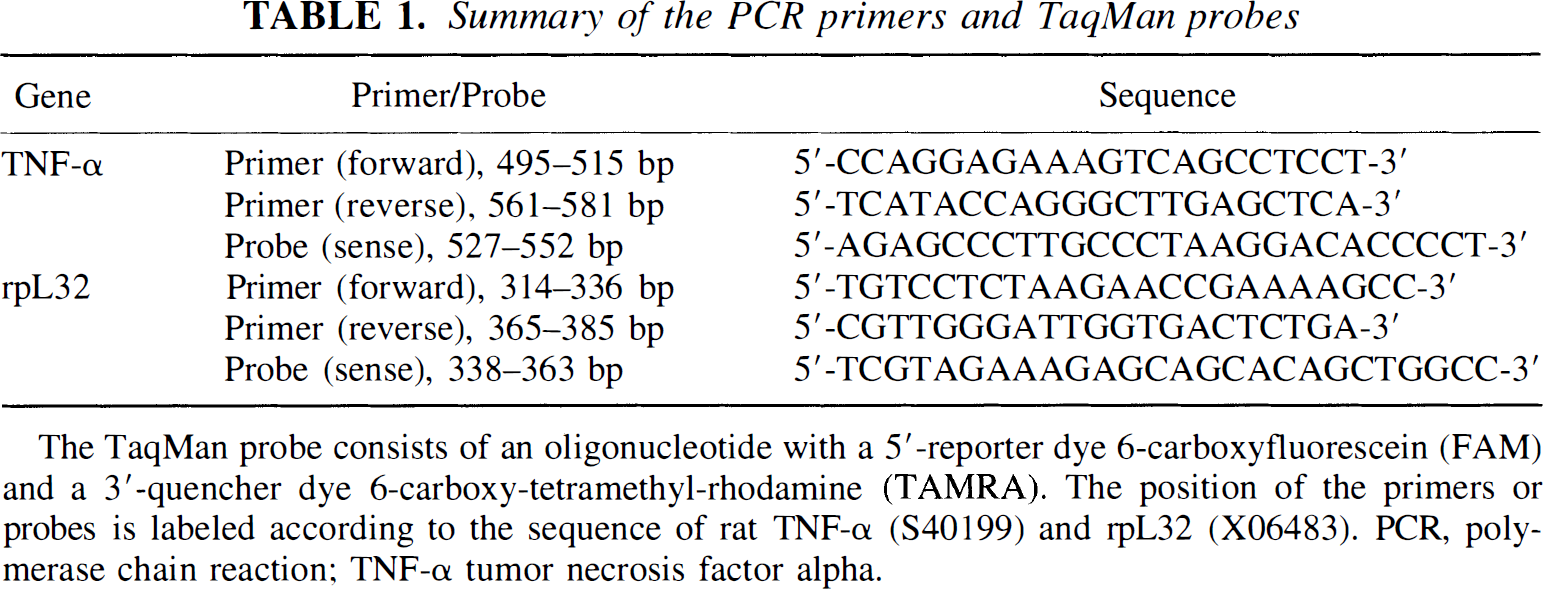
The TaqMan probe consists of an oligonucleotide with a 5'-reporter dye 6-carboxyfluorescein (FAM) and a 3'-quencher dye 6-carboxy-tetramethyl-rhodamine (TAMRA). The position of the primers or probes is labeled according to the sequence of rat TNF-α (S40199) and rpL32 (X06483). PCR, polymerase chain reaction; TNF-α tumor necrosis factor alpha.
Reverse transcription (RT) and PCR were carried out using a TaqMan Reverse Transcription Reagents and TaqMan Gold RT-PCR kit (Perkin-Elmer) according to the manufacturer's specification. A two-step RT-PCR was performed. RT reaction used 3 μg total RNA in a total volume of 40 μL containing 1 × TaqMan ET buffer, 5.5 mmol/L MgCl2, 500 μmol/L of each dNTP, 2.5 μmol/L oligo d(T)16 primers, 0.4 U/μL RNase Inhibitor and MultiScribe Reverse Transcriptase. RT reaction was carried out at 25°C for 10 minutes, 48°C for 30 minutes, and 95°C for 5 minutes.
A thermal stable AmpliTaq Gold DNA Polymerase was used for the second strand cDNA synthesis and DNA amplification. Real-time PCR was performed with 4 μL of RT products (300 ng total RNA), 1× TaqMan buffer A, 5.5 mmol/L MgCl2, 200 μmol/L dATP/dCTP/dGTP, 400 μmol/L dUTP, 200 nmol/L primers (forward and reverse), 100 nmol/L TaqMan probe, 0.01 U/μL AmpErase and 0.025 U/μL AmpliTaq Gold DNA Polymerase in a total volume of 50 μL. PCR was performed at 50°C for 2 minutes (for AmpErase UNG incubation to remove any uracil incorporated into the cDNA), at 95°C for 10 minutes (for AmpliTaq Gold activation) and then run for 40 cycles at 95°C for 15 seconds, and at 60°C for 1 minute on the ABI PRISM 7700 Detection System.
The principle of the real-time detection is based on the fluorogenic 5' nuclease assay as illustrated in Fig. 1. During the PCR reaction, the AmpliTaq Gold DNA Polymerase cleaves the TaqMan probe at the 5' end and separates the reporter dye from the quencher dye only if the probe hybridizes to the target. This cleavage results in the fluorescent signal generated by the cleaved reporter dye and directly monitored by the ABI PRISM 7700 Detection System. The increase in the fluorescence signal is proportional to the amount of the specific PCR product.
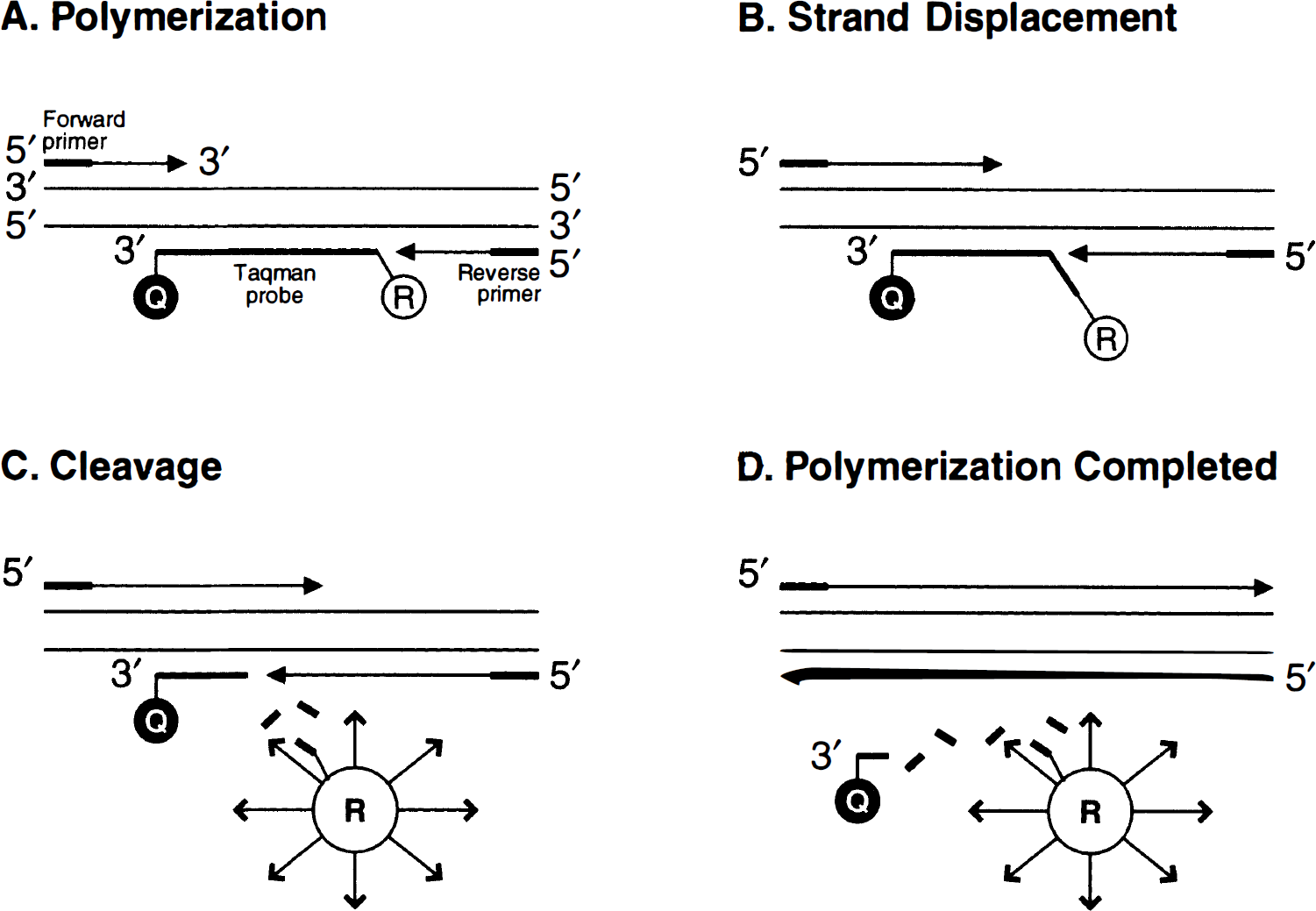
Schematic illustration of 5' nuclease assay for detecting PCR products. The nonextendable oligonucleotide probe is labeled with a reporter fluorescent dye (FAM) at the 5' end and a quencher-fluorescent dye (TAMRA) at the 3' end. When the probe is intact, the reporter dye emmission is quenched because of the physical proximity of the reporter and quencher dyes. During PCR, the 5' nuclease activity of the DNA polymerase cleaves the hybridized probe and releases the reporter dye from the probe. The emission of the released reporter dye can now be detected by a sequencer detector and the relative signal increase in real-time during PCR amplification.
Real-time polymerase chain reaction standard curves for TNF-α and rpL32
To determine the absolute copy number of the target transcript, a cloned plasmid DNA for TNF-α or rpL32 (Wang et al., 1994) was used to produce a standard curve. TNF-α and rpL32 plasmids contain 710 and 465 base pair (bp) of cDNA insert, respectively, in a pBluescript cloning vector (2958 bp). To determine the copy number of DNA templates, the molecular weights of the plasmids were calculated according to 6.6 × 105 per kilobase (kb) average value and then converted into the copy numbers based on the Avogadro's number, i.e., 1 mol = 6.022 × 1023 molecules. The cloned plasmid DNA was serially diluted (for every 5-fold). Each sample was run in duplicates, and the ΔRn (the ratio for the amount of reporter dye emission to the quenching dye emission) and threshold cycle (Ct) values were averaged from each reaction. Data were analyzed using a Sequence Detector V1.6 program (Perkin-Elmer).
Statistical analysis
Statistical comparisons were made by analysis of variance (ANOVA; Fisher's protected least squares difference) and values were considered to be significant when P was <0.05.
RESULTS
Determination of TNF-α and rpL32 standard curves with cloned plasmid DNA using real-time polymerase chain reaction
As shown in Fig. 2, TNF-α and rpL32 plasmids were serially diluted (five-fold dilution ranging from 1.65 to 7.24 log molecules or 16.4 fg to 64 pg for TNF-α template; 3.04 to 7.93 log molecules or 0.409 pg to 0.32 ng for rpL32), and real-time PCR data were plotted using cycle numbers versus Rn (Fig. 2, upper panels) or template copy numbers versus Ct values (Fig. 2, lower panels). The amplification (as indicated by the Ct value) was in a linear relationship with the initial template concentration (Fig. 2, lower panels). All of our testing samples were located within this linear amplification range (Fig. 2, lower panels).
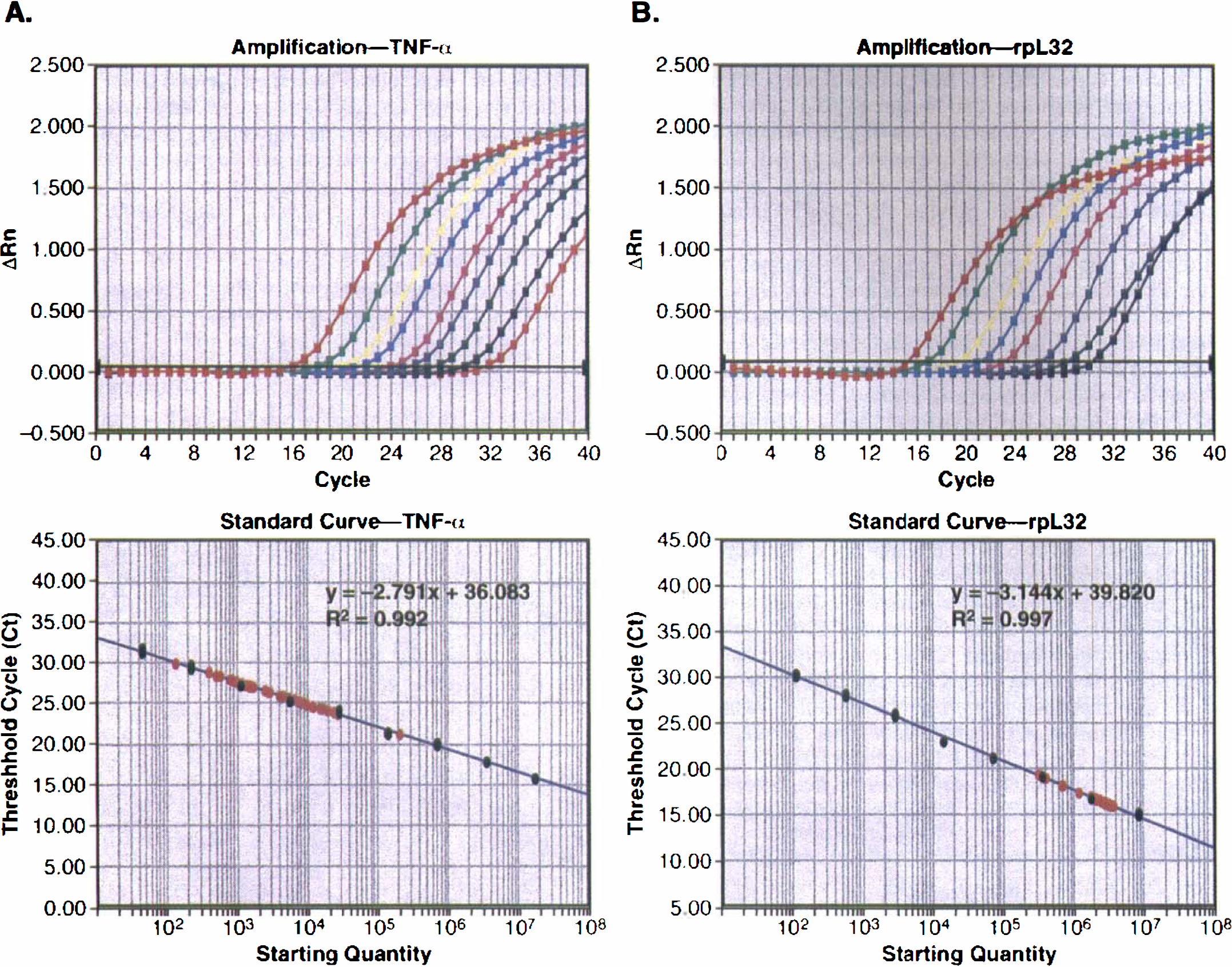
Real-time PCR of TNF-α
Temporal expression of TNF-α mRNA after ischemic preconditioning
TNF-α mRNA expression after preconditioning was examined using real-time PCR. The copy numbers of TNF-α mRNA in each sample were determined by the Ct value of the TaqMan PCR according to its DNA standard. To exclude the inaccuracy of an RNA concentration measurement that may result in false information on TNF-α RNA expression, rpL32 was selected as the internal control for the normalization of RNA concentration because it was demonstrated to be consistently expressed in cortical samples after preconditioning (Wang et al., 1998; Fig. 3B). The levels of rpL32 mRNA expression in the cortical samples are approximately 1.01 to 1.53 × 105 copies of transcript per nanogram of total RNA or microgram of tissue (Fig. 3B). After normalizing to the rpL32 transcript, the quantitative data (n = 4) for TNF-α mRNA expression in cortical samples after preconditioning or sham-operated animals are illustrated in Fig. 3A. Low levels of TNF-α mRNA (2 to 6 copies of mRNA per nanogram of RNA or microgram of tissue) were observed in the contralateral cortex. The levels of TNF-α mRNA expression were rapidly increased (27 ± 1 copies at 1 hour) in the ispsilateral cortex after preconditioning. Significant induction in TNF-α mRNA was observed at 6 hours (49 ± 10 copies of mRNA per microgram of tissue, or 4.4-fold increase compared to sham, P < 0.001) and 12 hours after preconditioning (37 ± 9 copies, P < 0.05) (Fig. 3).
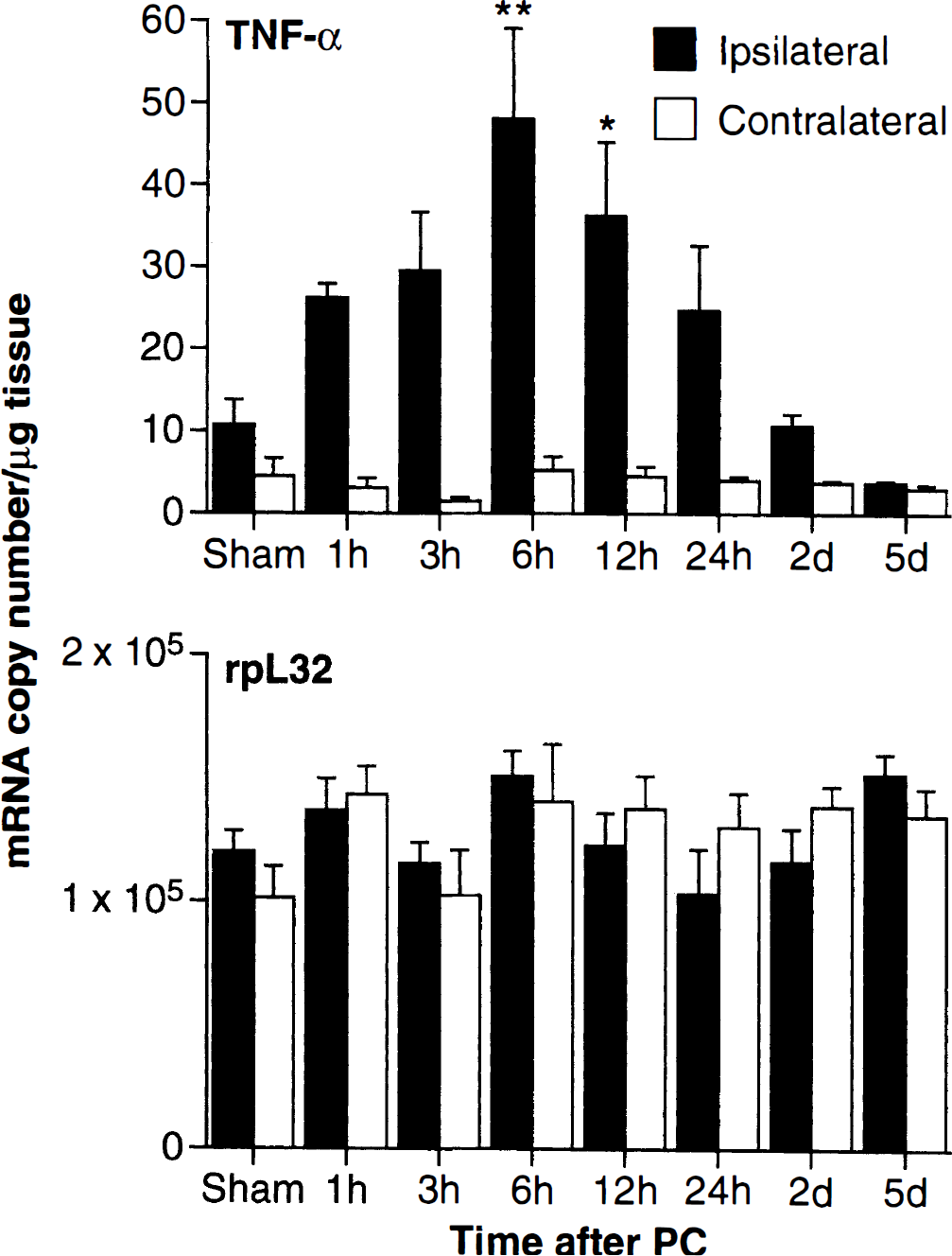
Real time PCR analysis of temporal expression of TNF-α mRNA in rat cortical samples after preconditioning. Total cellular RNA isolated from ipsilateral and contralateral cortex of rats subjected to sham surgery (6 hours) or after 1, 3, 6, 12 and 24 hours, and 2 and 5 days of preconditioning as described in Materials and Methods. RT and PCR was carried out using TaqMan quantitation (illustrated using 1 mg brain tissue or 1 ng RNA for the template). The absolute copy numbers of TNF-α transcripts (
DISCUSSION
Using a novel, exquisitely sensitive and quantitative real-time PCR technique, the present work describes for the first time a significant induction of TNF-α mRNA expression in brain subjected to a preconditioning procedure. TNF-α mRNA induction was observed as early as 1 hour and sustained up to 2 days in the ipsilateral cortex after preconditioning. The induced expression of TNF-α mRNA in ischemic tolerance induced by preconditioning suggests that this cytokine may contribute to the neuroprotective effect of preconditioning, a procedure that produces significant ischemic tolerance (Barone et al., 1998). It should also be pointed out that the increased expression of TNF-α mRNA may not represent the level of its increase in protein synthesis, and that even if the increase in the mRNA is translated into protein after preconditioning, the physiologic significance of this small-magnitude change of TNF-α still in our preconditioning model remains to be evaluated. The beneficial role of TNF-α in an lipopolysaccharide-induced tolerance model in rats has been demonstrated by concurrent administration of TNFbp (Tasaki et al., 1997). In addition, pretreated mice with TNF-α induced significant protection against focal cerebral ischemia injury (Nawashiro et al., 1997Z?), suggesting a role of TNF-α in the tolerance to ischemia induced by both lipopolysaccharide and TNF-α itself.
The molecular mechanisms for the effect of TNF-α in preconditioning are unknown. As it has been demonstrated previously, because the overall effect of TNF-α expression after brain ischemia appears to be detrimental (Dawson et al., 1996; Barone et al., 1997; Nawashiro et al., 1997a), the low level of TNF-α expression after preconditioning may result in sublethal stress, whereas the sublethal stress may lead to an undefined adaptation that increases resistance to subsequent ischemia. The involvement of a noxious but sublethal stress in preconditioning has been demonstrated with various chemical and physical stimuli (Kitagawa et al., 1991; Kobayashi et al., 1995; Chen and Simon, 1997; Riepe et al., 1997). It should also be mentioned that a low level of TNF-α expression after preconditioning may be acting to stimulate brain cells to undergo further adjustments that are the basis for ischemic tolerance.
The real-time PCR represents a novel technology based on the 5' nuclease activity of the Taq DNA polymerase. Early methods of quantitative PCR either rely on a housekeeping gene to serve as an internal control (such as the use of aldolase A and rpL32) for co-amplification (Chelly et al., 1988; Wang et al., 1997) or use of the same PCR primers and templates (with a small insertion or deletion in the templates) (Wang et al., 1989; Gilliland et al., 1990). All these methods have disadvantages for providing either semi- or inaccurate quantitation because of the involvement of amplification efficiency. The critical difference between the real-time PCR and the previous methods is the use of the threshold cycle numbers as a measure of the initial concentration of the target nucleic acid (Gibson et al., 1996; Heid et al., 1996). Thus, the effect of amplification efficiency differences is minimal as long as the target templates are in the linear range of PCR amplification. This advantage makes TaqMan PCR sensitive, accurate, and able to produce high throughput for quantitation of mRNA, especially in its application to detect those of a low number of expressed genes or when limited amount of tissue is available.
In conclusion, our present data illustrate the application of a sensitive and quantitative real-time PCR method for the accurate measurement of TNF-α mRNA expression in the brain and demonstrate for the first time the presence of TNF-α mRNA (though at very low levels) in normal tissue and its induction at a state of tolerance to ischemic insult. The induced expression of TNF-α by preconditioning suggests that TNF-α may play a role in ischemic tolerance.
