Abstract
Tissue type plasminogen activator (tPA) can be effective therapy for embolic stroke by restoring cerebral perfusion. However, a recent experimental study showed that tPA increased infarct size in a mouse model of transient focal ischemia, suggesting a possible adverse effect of tPA on ischemic tissue per se. In this report, the effects of tPA in two rat models of cerebral ischemia were compared. In experiment 1, rats were subjected to focal ischemia via injection of autologous clots into the middle cerebral artery territory. Two hours after clot injection, rats were treated with 10 mg/kg tPA or normal saline. Perfusion-sensitive computed tomography scanning showed that tPA restored cerebral perfusion in this thromboembolic model. Treatment with tPA significantly reduced ischemic lesion volumes measured at 24 hours by >60%. In experiment 2, three groups of rats were subjected to focal ischemia via a mechanical approach in which a silicon-coated filament was used intraluminally to occlude the origin of the middle cerebral artery. In two groups, the filament was withdrawn after 2 hours to allow for reperfusion, and then rats were randomly treated with 10 mg/kg tPA or normal saline. In the third group, rats were not treated and the filament was not withdrawn so that permanent focal ischemia was present. In this experiment, tPA did not significantly alter lesion volumes after 2 hours of transient focal ischemia. In contrast, permanent ischemia significantly increased lesion volumes by 55% compared with transient ischemia. These results indicate that in these rat models of focal cerebral ischemia, tPA did not have detectable negative effects. Other potentially negative effects of tPA may be dependent on choice of animal species and model systems.
Clinical trials have suggested that the tissue type plasminogen activator (tPA) can be efficacious in ischemic stroke if administered within hours after acute stroke onset (NINDS rt-PA Stroke Study Group, 1995). The thrombolytic effects of tPA beneficially produce clot dissolution and reperfusion of ischemic territories (Busch et al., 1998; Jiang et al., 1998). However, a recent study has suggested that tPA may also have neurotoxic effects. Wang and colleagues (1998) have shown that infusion of tPA increased infarct size in a mouse model of focal cerebral ischemia. Furthermore, knockout mice that lack the capacity to produce endogenous tPA showed reduced infarct sizes compared with their wild-type littermates (Wang et al., 1998).
As tPA is used for the treatment of acute ischemic stroke, it is critical to identify potential neurotoxic effects of this compound. In the present study, we compared the effects of tPA in two different models of focal cerebral ischemia in rats. In experiment 1, the effects of tPA were examined in rats subjected to thromboembolic focal ischemia via injection of autologous clots into the middle cerebral artery territory. In this model, we expected that the thrombolytic properties of tPA would have the predominant effects on outcome. In experiment 2, we examined tPA in a mechanical model of arterial occlusion. A standard model was used in which an intraluminal silicon-coated nylon filament is inserted up the internal carotid artery to occlude the origin of the middle cerebral artery. Reperfusion is achieved by withdrawal of the filament occluder. In this second experiment, we expected that the potentially neurotoxic effects of tPA could be assessed separately from its thrombolytic properties.
MATERIALS AND METHODS
General methods
Adult male Sprague-Dawley rats were used, following an institutionally approved protocol in accordance with guidelines set by the NIH Guide for the Care and Use of Laboratory Animals. Rats were anesthetized with 1 to 2% halothane under free-breathing conditions in a 2:1 mixture of air/oxygen. Catheters were inserted into the femoral artery and vein. Physiologic parameters including rectal core temperature, mean arterial blood pressure, pH, and gases were monitored throughout all experiments. Human recombinant tPA (Activase) was purchased from Genentech (South San Francisco, CA, U.S.A.).
Animal model for experiment 1
In this experiment, a thromboembolic approach was used to induce focal ischemia in rats (300 to 400 g). A small burr hole was placed into the skull at bregma to serve as a fiducial marker for computed tomography (CT) image localization (see below). A surgical cutdown was performed via the ventral surface of the neck to expose the internal and external carotid arteries. The external carotid artery, occipital artery, and pterygopalatine artery were ligated. A 24-gauge catheter was inserted into the external carotid artery until the tip was positioned at the internal carotid junction. Focal ischemia was induced according to methods first described by Kudo et al. (1982).
Under microscopic guidance, 200 μL of autologous micro-clot solution was slowly injected into the cerebral circulation to induce focal ischemia. Microclot suspensions were made by passing 48-hour aged clots through a 27-gauge needle three times to ensure that particle sizes were in the 50-μm range. Two hours after occlusion, rats were treated with either tPA (10 mg/kg slowly infused intravenously over 30 minutes) or normal saline (n = 8 rats per group).
Animal model for experiment 2
In this experiment, a mechanical method was used to induce focal ischemia in rats (270 to 310 g) via a standard intraluminal approach (Lo et al., 1998; Zea Longa et al., 1989). In brief, a surgical cutdown was performed in the ventral neck region and the internal, external, and common carotid arteries were visualized. A 4.0 nylon monofilament suture coated with silicon was inserted into the external carotid artery and advanced up the internal carotid artery until the tip was placed ∼ 19 to 20 mm from the internal-external carotid bifurcation. After 2 hours of transient focal ischemia, the monofilament suture was withdrawn to allow for cerebral reperfusion. Immediately on mechanical reperfusion, rats were given either tPA (10 mg/kg infused over 30 minutes) or normal saline (n = 8 rats per group). A third group of rats (n = 8) were left untreated and subjected to permanent focal ischemia; that is, the filaments were not withdrawn. The purpose of this third group was to establish that maximal ischemic lesions are not achieved after 2-hour occlusions of the middle cerebral artery. This is an important point because if lesion volumes are indeed maximal after 2 hours of transient focal ischemia, then potentially negative effects of tPA may not be detectable because the lesions cannot get any larger.
Perfusion-sensitive CT scanning
To document the distribution of focal ischemia after thromboembolic occlusions, rats were subjected to perfusion-sensitive CT scanning at −30 to 60 minutes after clot injections to image relative cerebral perfusion. This was necessary to confirm that appropriate occlusions within the middle cerebral artery territories had occurred. Perfusion-sensitive CT was not performed in the second experiment, in which standard and more reproducible mechanical methods of middle cerebral artery occlusion were used. Rats were immobilized with a custom-made head holder constructed from a 10-mL syringe and scanned in a helical slip-ring CT scanner (TCT 900SX; Toshiba, Tokyo, Japan). Anesthesia was maintained with 1 to 1.5% halothane in a 2:1 air/oxygen mix. Sagittal plane scout images were used to locate the burr hole in the skull at bregma. Thereafter, an axial slice at the level of bregma was selected, corresponding to the caudate putamen level in rat brain (Paxinos and Watson, 1982).
Functional CT scanning was performed using previously described techniques (Hamberg et al., 1996; Lo et al., 1996; Shimizu-Sasamata et al., 1998). In brief, bolus injections of an iodinated contrast agent (1.2 mL/kg Omnipaque 350; Sanofi-Winthrop, Paris, France) were performed via femoral vein. Dynamic CT scans (120 kV, 150 mA) were acquired at a rate of 1 image/second for 35 seconds. Relative cerebral perfusion images were then calculated by integrating under first-pass transit profiles of the contrast agent boluses (Hamberg et al., 1996; Lo et al., 1996; Shimizu-Sasamata et al., 1998). This procedure was then repeated after treatment with either normal saline or tPA to assess the profiles of reperfusion.
Quantitative CT imaging data were analyzed as follows. First, average hemispheric perfusion values were calculated from the contralateral side. Then, a threshold was set at 50% of these contralateral perfusion levels. Ischemic brain areas with perfusion below this 50% threshold were then measured in the ipsilateral hemisphere and expressed as percent area of the ipsilateral side.
Quantitation of ischemic lesion sizes
Rats from both experiments were allowed to recover after ischemic surgery in individual cages. Twenty-four hours later, rats were killed with an overdose of sodium pentobarbital (100 mg/kg intraperitoneally). Brains were extracted, cut into 2-mm-thick axial sections, and stained with 2% 2,3,5-triphenyltetrazolium chloride. Ischemic lesion volumes were quantified using standard computer-assisted image analysis techniques, in which lesion areas from each axial slice were integrated to yield total volumes (Lo et al., 1998). To eliminate the effects of brain swelling and edema, an indirect method was used to calculate lesion volumes (Lin et al., 1993). The Shapiro-Wilks test for normality was performed and confirmed that data distributions were normal. Therefore, parametric two-tailed Student t tests were used to compare results from control rats and tPA-treated rats. P values of <0.05 were considered significant.
RESULTS
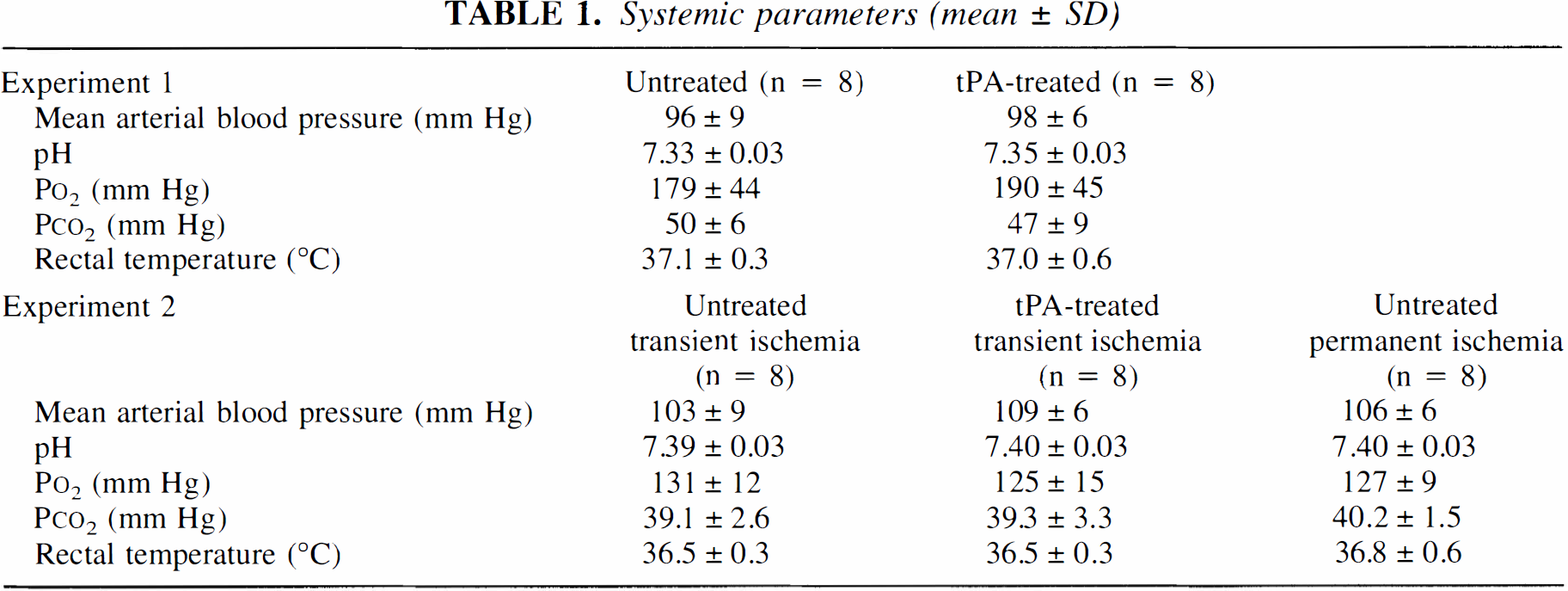
In all experiments, systemic parameters (Table 1) were within the range expected for halothane-anesthetized rats under free-breathing conditions (Lo et al., 1998; Memezawa et al., 1992). There were no significant differences between control and tPA-treated rats. In experiment 1, functional CT scans performed between 30 and 60 minutes after clot injection showed that focal ischemia was present primarily within the middle cerebral artery territories (Fig. 1). In untreated rats, the extent of focal ischemia either worsened or remained unchanged after 2 to 3 hours (Figs. 1A and 1B). Ischemic brain areas with perfusion below 50% of contralateral hemispheric levels significantly increased (Table 2). However, in tPA-treated rats, partial restoration of cerebral perfusion was observed (Figs. 1C and 1D). After tPA infusion, ischemic brain areas with perfusion below 50% of contralateral hemispheric levels were significantly decreased (Table 2). Twenty-four-hour ischemic lesion sizes were significantly reduced by tPA (50 ± 26 mm3 in tPA-treated rats versus 141 ± 42 mm3 in controls, P < 0.05; Fig. 2).
Systemic parameters (mean ± SD)
Perfusion deficit areas after thromboembolic occlusion*

Brain regions with cerebral perfusion below 50% of contralateral levels. These areas were measured and data expressed as percent of the ipsilateral hemspheric areas.
P < 0.05 versus pretreatment values by paired t-test.
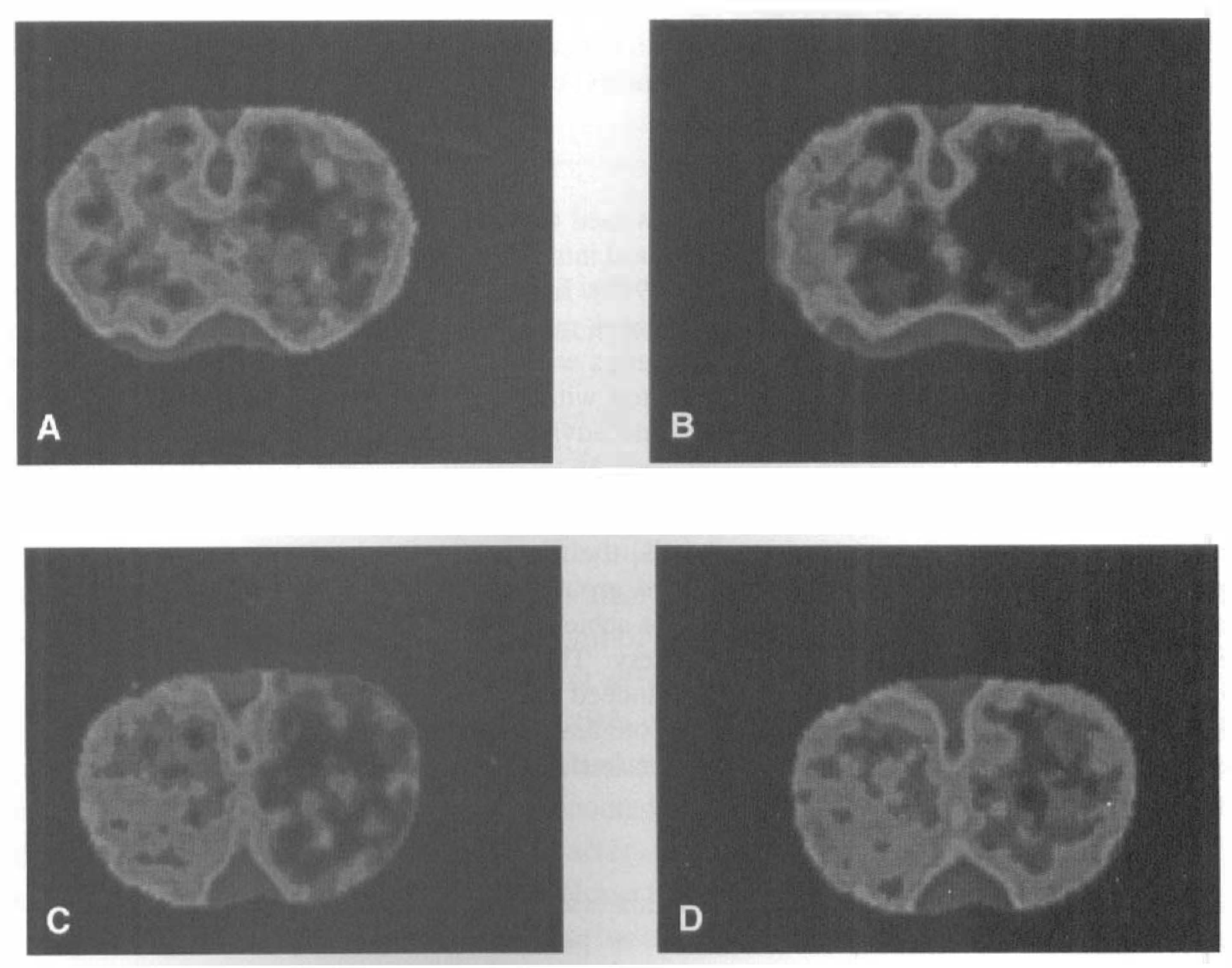
Perfusion-sensitive computed tomography (CT) of thromboembolic stroke in rat brain.
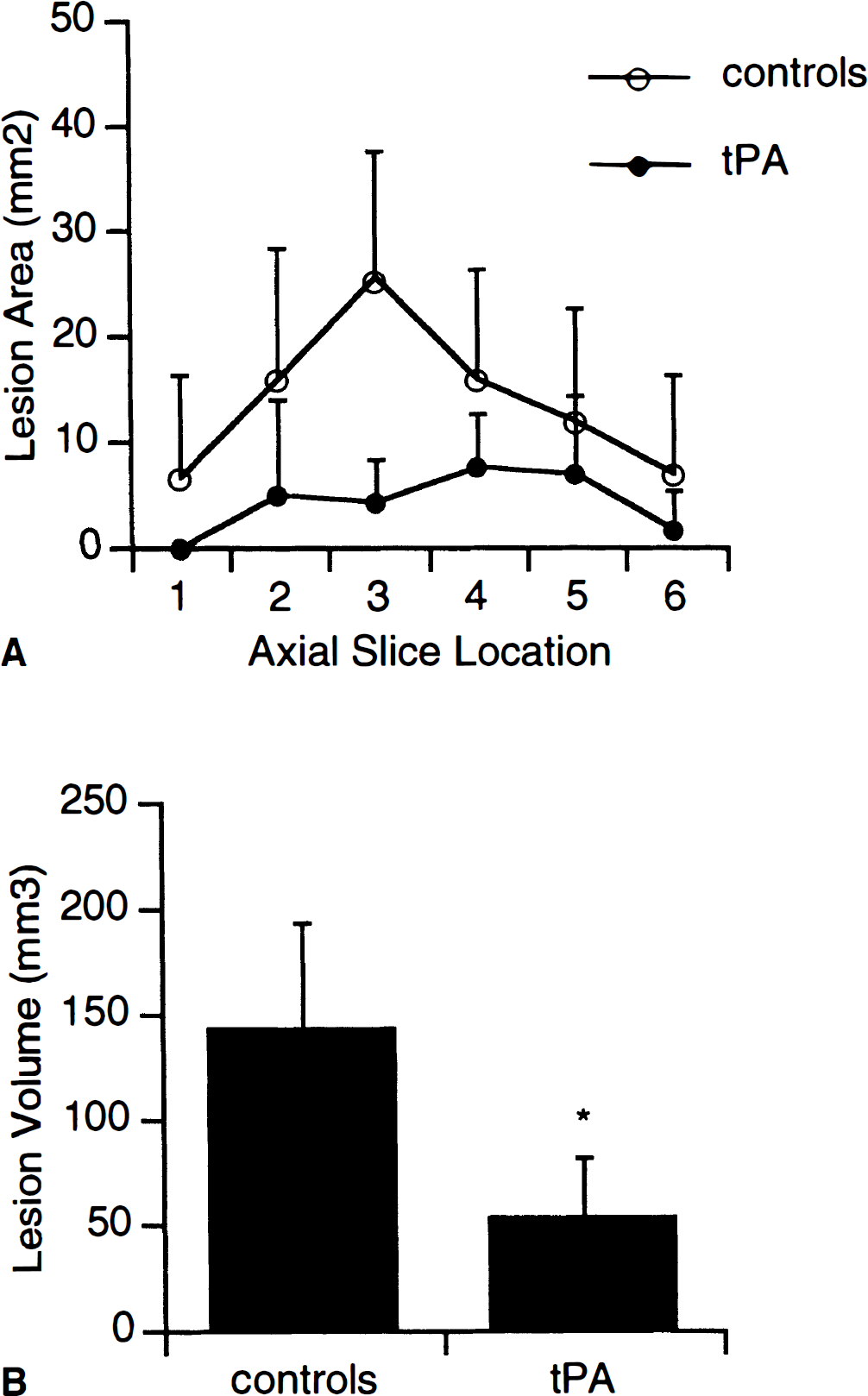
Ischemic lesion sizes after focal ischemia in rats produced by injection of autologous clots into the middle cerebral artery distribution. Tissue type plasminogen activator significantly reduced lesion sizes.
In experiment 2, mechanically induced focal ischemia and reperfusion resulted in large regions of ischemic injury localized to the middle cerebral artery territory. Total lesion volumes in this model were greater than those obtained with the thromboembolic occlusions in experiment 1. There were no significant differences in 24-hour lesion sizes between control and tPA-treated rats after 2 hours of transient focal ischemia (Fig. 3). However, permanent occlusion of the middle cerebral artery resulted in lesions that were 55% larger (P < 0.05) than those in the 2-hour occlusion groups (Fig. 3).
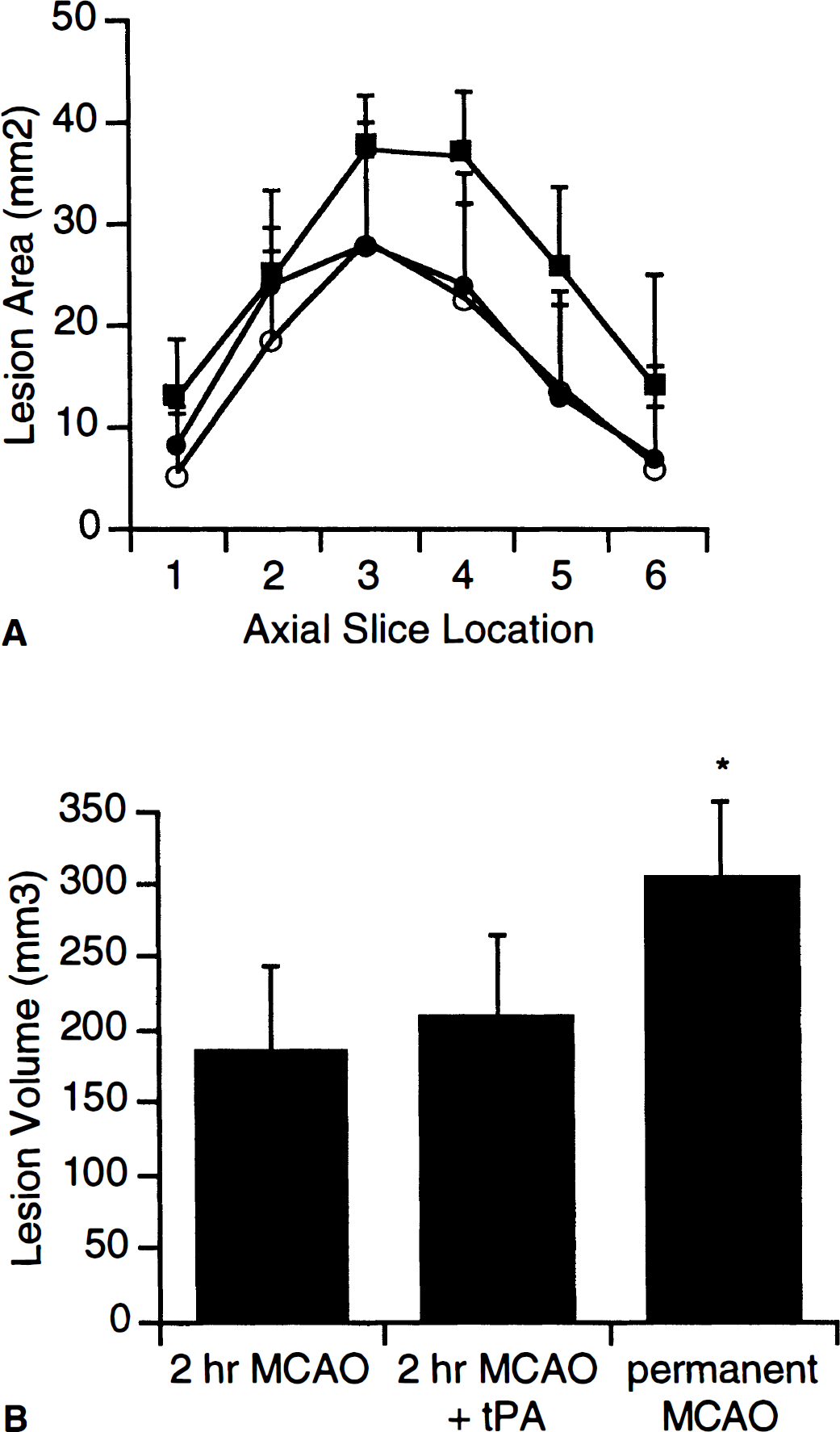
Ischemic lesion sizes after focal cerebral ischemia in rats produced using a mechanical intraluminal occluder (mean ± SEM). Tissue type plasminogen activator had no effect on lesion sizes after 2-hour transient occlusions. However, rats subjected to permanent occlusions had significantly greater lesion sizes.
DISCUSSION
Recently, Wang and colleagues (1998) reported that infusion of tPA significantly increased infarct size in both wild-type and tPA knockout mice after focal cerebral ischemia. Potential negative effects of tPA may be based on its ability to activate plasminogen and induce damaging extracellular proteolytic pathways (Tsirka et al., 1997a, b ). Specifically, nonfibrin substrates for plasmin, such as laminin, may be degraded (Chen and Strickland, 1997). Proteolysis of laminin and other proteins in the extracellular matrix may amplify excitotoxicity. It has been shown that knockout mice deficient in tPA or plasminogen were resistant to excitotoxic injury after intracerebral injections of kainate, N-methyl-D-aspartate, and α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (Tsirka et al., 1995, 1996, 1997a, b ). As excitotoxicity plays a central role in ischemic pathophysiology (Choi and Rothman, 1990; Lipton and Rosenberg, 1994), it is conceivable that tPA can have detrimental effects on neuronal survival after stroke.
In the present study, we compared the effects of tPA in two rat models of focal cerebral ischemia. Specifically, we expected that tPA would be neuroprotective in thromboembolic models, whereas in mechanical models of arterial occlusion, potentially neurotoxic effects of tPA might be examined. Our methodology reproduced results found in prior clinical and animal studies, namely, that tPA effectively restored cerebral perfusion and reduced infarct size. However, we did not find that tPA was neurotoxic in the mechanical models of arterial occlusion. There were no significant differences in infarct size after ischemia-reperfusion in rats induced by the standard intraluminal occlusion technique. These results stand in contrast to those reported by Wang et al. (1998), in which tPA increased infarct volumes after transient focal ischemia produced by similar mechanical methods of arterial occlusion.
There may have been two possible reasons for these different results. First, the doses of human recombinant tPA used were different. In the study by Wang et al. (1998), a dose of 1 mg/kg was used. Here, we used a much higher dose (10 mg/kg) based on the fact that there is a 10-fold difference in fibrin-specific enzyme activity of human recombinant tPA in human versus rodent systems (Korninger and Collen, 1981). However, there is no obvious reason why one would expect neurotoxic effects of tPA to be expressed only at lower rather than higher doses. A second difference lies in the choice of animal species. Wang et al. (1998) used mouse models of focal ischemia, whereas rat models were used in the present study. Species differences in critical ischemic events have been shown before. For example, rates of DNA damage after ischemia-reperfusion are different in baboon versus rat (Tagaya et al., 1997), and activation profiles of inducible nitric oxide synthase are critically different in rat versus mouse models of focal ischemia (Yoshida et al., 1995). It remains possible that species differences in tPA response after ischemia-reperfusion may explain some of the differences between the results of Wang et al. (1998) and the present study.
In this study, rats subjected to permanent focal ischemia had significantly larger lesions than those subjected to transient occlusions. This shows that after 2 hours of occlusion, the middle cerebral artery territory is not maximally damaged. This is an important point because if maximal lesions were achieved after 2 hours of transient ischemia, then potentially negative effects of tPA would not have been detectable as the lesions would not have been able to get any larger. A related issue worth considering is whether we had sufficient statistical power to detect negative effects of tPA. A standard power analysis of our data (α = 0.05, β = 0.8) showed that we would have been able to detect increases of infarction of −40%. This is within the range of expected effects, as in the Wang et al. study (1998), tPA increased infarct volumes by 100% in tPA-deficient knockout mice and by 33% in wild-type mice. However, it must be acknowledged that we would not have been able to reliably detect much smaller changes in infarct size after tPA in our models. Furthermore, as we assess outcomes only at 24 hours, we cannot exclude a role for delayed events. Finally, a technical caveat must be emphasized in the present study. The primary purpose of using two different models was to compare a thromboembolic occlusion method against a mechanical one. It has been shown that the intraluminal mechanical occluder does not induce thrombosis in the middle cerebral artery (Zhang et al., 1995). However, one cannot unequivocally exclude the possibility that the intraluminal filament might perturb the endothelial surface and induce downstream micro-thrombotic events that are not detected.
tPA has been shown to be effective in human clinical trials, but severe complications involving intracerebral hemorrhage occur in some patients (NINDS rt-PA Stroke Study Group, 1997). Although we did not observe any macroscopic evidence of hemorrhagic transformations in our studies, it is conceivable that, under certain conditions, tPA may damage the basal lamina and weaken vascular walls after stroke, thus increasing the probability of hemorrhage after reperfusion. Plasmin can cleave and activate matrix metalloproteinases (Mazzieri et al., 1997) that degrade cerebrovascular basal lamina (Heo et al., 1999; Rosenberg et al., 1996). One might speculate that after ischemia, plasminogen activators can excessively up-regulate these matrix proteinases and lead to alterations in vessel integrity. Furthermore, it is well established in nonhuman primate models of stroke that spontaneous microvascular hemorrhage may play a critical role in reperfusion injury (del Zoppo, 1994; Hamman et al., 1996). Precise mechanisms that mediate tPA's extracellular proteolytic effects after ischemia warrant additional investigation.
In conclusion, our results support prior observations that tPA represents effective therapy for thromboembolic occlusions. In a mechanical model of middle cerebral artery occlusion, however, we did not find evidence of a direct neurotoxic effect of tPA in cerebral tissue. Reported potentially negative effects of tPA may be critically dependent on the choice of animal species and model systems.
