Abstract
Reactive oxygen species (ROS) is massively produced in the brain after cerebral ischemia and reperfusion. It reacts strongly with cellular components, which has detrimental effects and leads to neuronal cell death. DJ-1, which was found to be the causative gene of familial Parkinson's disease PARK7, is a multifunction protein, which plays a key role in transcriptional regulation, and a molecular chaperone. In this study, we investigated the neuroprotective effect of DJ-1 against neurodegeneration caused by ischemia/reperfusion injury. Cerebral ischemia was induced in rats by 120 mins of middle cerebral artery occlusion (MCAO) using an intraluminal introduction method. The intrastriatal injection of recombinant glutathione
Keywords
Introduction
Cerebral ischemia occurs as a result of a local reduction or an arrest of blood supply and can lead to neuronal cell death in the ischemic region. The pathophysiologic mechanisms of the ischemia/reperfusion injury are complex. Neurons and glial cells are lethally damaged by several events such as periinfarct depolarization, which occur within hours after the onset of ischemia, and the production of reactive oxygen species (ROS), which occurs immediately after ischemia/reperfusion, is followed by more delayed postischemic inflammation and apoptosis, and these events are involved in the progression and expansion of brain injury (Hata et al, 2000; Peters et al, 1998). This burst of ROS is involved in direct cytotoxic effects, including protein and lipid peroxidation, oxidative DNA damage, and postischemic inflammatory injury, through redox-mediated signaling pathways (Andersen, 2004; Margaill et al, 2005). The oxidation of proteins can have wide-ranging damaging effects such as disruption of the active sites of enzymes or alteration of the conformation of structural proteins. Oxidative modification of susceptible unsaturated fatty acids can result in the generation of lipid peroxides, which can further oxidize nearby unsaturated fatty acids in a chain-reaction event. This event can disrupt both the plasma membrane and membranes that are components of cellular organelles, such as mitochondria. Thus, the components of ROS are believed to make a contribution to neuronal loss after ischemia/reperfusion (Andersen, 2004; Margaill et al, 2005). Therefore, the control of ROS production is important for achieving neuroprotection against ischemia/reperfusion injury.
DJ-1 was originally identified as a novel oncogene in collaboration with activated small G-protein
In this study, to evaluate the neuroprotective effect of DJ-1 against ischemia/reperfusion injury, we performed the intrastriatal injection of recombinant human DJ-1 protein in rats that had been subjected to middle cerebral artery occlusion (MCAO) and reperfusion. We then investigated its effects on infarct size, neuronal survival, and behavioral outcome. Moreover, we also examined the effects of DJ-1 on ROS-mediated oxidative stress in SH-SY5Y cells.
Materials and methods
Preparation of Recombinant Human DJ-1 Protein and Anti-DJ-1 Antibody
Recombinant glutathione
Animals
Wistar rats were purchased from Japan SLC Inc. (Hamamatsu, Japan). The animals were acclimated to and maintained at 23°C under a 12-h light/dark cycle (light from 0800 to 2000 hours). Rats were housed in standard laboratory cages and had free access to food and water throughout the study period. All animal experiments were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals, and the protocols were approved by the Committee for Animal Research at Kyoto Pharmaceutical University.
Rat Model of Focal Ischemia
Male Wistar rats weighing 260 to 300 g were used in this study. Focal cerebral ischemia was induced by the intraluminal introduction of a nylon thread (Nagasawa and Kogure, 1989) as described previously (Kitamura et al, 2005). Briefly, animals were anesthetized with 4% halothane (Takeda Pharmaceutical, Osaka, Japan) and maintained on 1.5% halothane using a face mask. After a midline neck incision was made, 20mm of 4-0 nylon thread, with its tip rounded by heating and coated with silicon (Xantopren M; Heraeus Kulzer, Hanau, Germany), was inserted into the left internal carotid artery as far as the proximal end using a globular stopper. The origin of the middle cerebral artery was then occluded by a silicon-coated embolus. Anesthesia was discontinued, and the development of right hemiparesis with upper limb dominance was used as a criterion for ischemic insult. After 120 mins of MCAO, the embolus was withdrawn to allow reperfusion of the ischemic region through the anterior and posterior communicating arteries. Body temperature was maintained at 37 to 37.5°C with a heating pad and lamp during surgery. In the sham operation, a midline neck incision was made to expose the arteries, but the nylon thread was not inserted into the carotid artery.
Administration of Glutathione S-Transferase-Tagged Human DJ-1
For stereotaxic microinjection, rats were anesthetized (sodium pentobarbital; 50 mg/kg, intraperitoneally, Abbott Laboratories, North Chicago, IL, USA) and immobilized in a Kopf stereotaxic frame. Subsequently, rats were injected with GST (320 pmol), GST-DJ-1 (4, 40, 160, or 320 pmol), or phosphate-buffered saline (PBS) as the vehicle control in a final volume of 4 μL of PBS at 110 mins after the onset of MCAO (10 mins before reperfusion). To investigate the time window of efficacy, we injected GST-DJ-1 (320 pmol) at 60, 110, 180, and 300 mins after the onset of MCAO. The intrastriatal injection coordinates (1.0 mm anterior, 4.0 mm left lateral, 5.0 mm ventral from the bregma) were taken from a rat brain atlas (Paxinos and Watson, 2005). Injection was given by a motor-driven 10-μL Hamilton syringe using a 26-gauge needle. The infusion rate was 1 μL/min, and the injection cannula was kept in place for a further 5 mins after the injection.
Measurement of Infarct Size
At 3 days after MCAO, the brains were removed and cut into 2-mm-thick coronal sections. These sections were immersed in a 2% solution of 2,3,5-triphenyltetrazolium chloride (TTC; Wako Pure Chemical Industries, Osaka, Japan) in saline at 37°C for 20 mins and then fixed in 4% paraformaldehyde in 100 mmol/L phosphate buffer at 4°C. For quantification of infarct size, the brain sections were scanned using a camera (KY-F55MD; Victor, Tokyo, Japan), and the infarct size was then analyzed by computerized image analysis (WinRoof; Mitani, Fukui, Japan). Briefly, the infarct area in each section was calculated by subtracting the area of the normally stained ipsilateral hemisphere from the area of the contralateral hemisphere to reduce errors because of cerebral edema (Lin et al, 1993; Swanson et al, 1989). The infarct volume was calculated by multiplying the sum of the infarct areas by the distance between the sections.
Magnetic Resonance Imaging
Rats were sham-operated or operated on the MCAO and then received an intrastriatal injection with PBS (a vehicle control) or GST-DJ-1 (320 pmol) at 110 mins after the onset of MCAO (10 mins before reperfusion). After 7 days, magnetic resonance (MR) images of these rat heads were acquired with a 7T Unity Inova MR scanner (Varian, Palo Alto, CA, USA). A T2-weighted MR sequence was used with acquisition parameters of 2,000 ms repetition time, 40 ms echo time, 35 × 35 mm2 field of view, 1-mm slice thickness (total 24 slices) and 256 × 128 matrices. During the MR sessions, spontaneously breathing animals were anesthetized with 1.5% isoflurane in 50% O2 and 50% air.
The infarct area was calculated from T2-weighted MR images using imaging software (WinRoof), as described above. For each slice, areas of high intensity in T2-weighted MR images were marked as ischemic lesion areas, and the infarct volume was calculated by taking slice thickness (1 mm/slice) into account.
Neurologic Evaluation
Neurologic deficits were assessed at 30 mins after the onset of MCAO (80 mins before injection of PBS, GST, or GST-DJ-1), and subsequently every day until the rats were killed at 7 days after MCAO. Neurologic findings were graded as severe, moderate, mild, or absent on a 4-point scale, according to the method described by Bederson et al (1986); a grade 3 (severe) neurologic deficit indicates reduced resistance to lateral pushing toward the right side and forelimb flexion with consistent circling toward the paretic side; a grade 2 (moderate) neurologic deficit indicates reduced resistance to lateral pushing toward the right side and forelimb flexion but without circling movement; a grade 1 (mild) neurologic deficit indicates any amount of consistent forelimb flexion but no other abnormality; and grade 0 indicates no observable neurologic deficit. In this study, the protection against ischemia-induced neurologic deficits was evaluated by this method, similar to previous studies (Gao et al 2005; Suzuki et al, 2005).
Rota-rod Test
Spontaneous motor activity was measured using the rota-rod treadmill for rats (accelerating model 7750; Ugo Basile, Varese, Italy), and the accelerating rotor mode was used (10 speeds from 4 to 40 r.p.m. for 5 mins). The interval from when the animal mounted the rod to when it fell off was recorded as the performance time (Kitamura et al, 2005). The animals were trained for five trials per day for 2 days before MCAO to obtain stable baseline values. The mean duration on the rod was recorded with five trials at 1 day before surgery. Performance on the rota-rod test was measured five times a day at 2 and 5 days after MCAO, and these data are presented as the mean duration on the rod. Subsequently, time-dependent change in the percentage (%) of walking rats on the rotating rod was calculated by the Kaplan—Meier method.
Methamphetamine-Induced Rotation Test
At 6 days after MCAO, the methamphetamine-induced rotation test was assessed in rotometer bowls as described previously (Inden et al, 2006; Kitamura et al, 2005). Briefly, the total number of full-turn rotations in the ipsilateral direction was counted for 60 mins after the intraperitoneal administration of methamphetamine (2.5 mg/kg; Dainippon Pharmaceutical, Osaka, Japan), which stimulates dopamine release on the intact right side and induces marked ipsilateral circling in animals with stroke.
Immunohistochemistry
The animals were perfused through the aorta with 150 mL of 10 mmol/L PBS, followed by 300 mL of a cold fixative consisting of 4% paraformaldehyde in 100 mmol/L phosphate buffer under deep anesthesia with sodium pentobarbital (50 mg/kg, intraperitoneally). After perfusion, the brains were quickly removed and postfixed for 2 days with 4% paraformaldehyde in 100 mmol/L phosphate buffer and then transferred to 15% sucrose solution in 100 mmol/L phosphate buffer containing 0.1% sodium azide at 4°C. The brain pieces were cut 20 μm thick using a cryostat and collected in 100 mmol/L PBS containing 0.3% Triton X-100. The free-floating sections were treated with 0.3% hydrogen peroxide in PBS containing 0.3% Triton X-100 to eliminate endogenous peroxidase activity. After several washes, the sections were incubated for 3 days at 4°C with the following primary antibodies: mouse monoclonal antibodies against DJ-1 (dilution, 1:10,000), microtubule-associated protein-2 (MAP2) (1:3,000; Sigma, St Louis, MO, USA), and tyrosine hydroxylase (TH) (1:3,000; Sigma); rabbit polyclonal antibodies against GST (1:10,000; Cell Signaling Technology, Danvers, MA, USA); and nitrotyrosine (1:500; Chemicon International, Temecula, CA, USA). After several washes, the sections were incubated with biotinylated antibodies against mouse or rabbit IgG (1:2,000; Vector Laboratories, Burlingame, CA, USA) as appropriate for 2 h at room temperature. The sections were then incubated with avidin peroxidase (1:4,000; Vectastatin ABC Elite kit, Vector Laboratories) for 1 h at room temperature. All of the sections were washed several times with PBS containing 0.3% Triton X-100 between each incubation, and labeling was then revealed by 3,3′-diaminobenzidine (Dojindo Laboratories, Kumamoto, Japan), with nickel ammonium, which yielded a dark blue color.
For the semiquantitative analysis of GST-immunoreactive areas, the immunostained sections at +4, +3, +2, +1, ±0, −1, and −2 mm anterior—posterior from the bregma were also scanned using a camera, and these immunoreactive areas were measured by computerized image analysis (WinRoof).
To evaluate the level of oxidative and nitrosative stress, the sections that had been immunostained with anti-nitrotyrosine antibody at +1 mm anterior from the bregma were scanned using a camera. The number of nitrotyrosine-immunopositive cells was counted in a predefined area (2.015 mm2) within the boundary zones of the ischemic core in the cerebral cortex and the striatum and in the ischemic core in the striatum using computerized image analysis (WinRoof).
Double-Immunofluorescence Staining
For double-immunofluorescence staining, the sections were simultaneously incubated for 3 days at 4°C with the following primary antibodies: mouse monoclonal antibodies against neuronal nuclei (NeuN, 1:5,000; Chemicon International), glial fibrillary acidic protein (1:3,000; Chemicon International), and CD11b (1:500; Serotec); and a rabbit polyclonal anti-GST antibody (1:5,000). The primary antibodies were detected with anti-mouse IgG antibody conjugated with Alexa Fluor 488 and a anti-rabbit IgG antibody conjugated with Alexa Fluor 546 (1:500; Molecular Probes, Eugene, OR, USA) for 2 h at room temperature. In addition, the sections were incubated with Hoechst 33342 (Molecular Probes) for 15 mins at room temperature. Fluorescence was then detected using a laser scanning confocal microscope (LSM410; Carl Zeiss, Jena, Germany)
Establishment of DJ-1-Knockdown Cell Line
The human neuroblastoma cell line SH-SY5Y was cultured in Dulbecco's modified Eagle's medium supplemented with 10% (v/v) fetal calf serum, 50 μg/mL penicillin, and 100 μg/mL streptomycin, and kept at 37°C in humidified 5% CO2/95% air.
We established DJ-1-knockdown SH-SY5Y cell line by essentially similar method in mouse Flp-In3T3 cells (Takahashi-Niki et al, 2004). In brief, the nucleotide sequence of the upper strand of the oligonucleotide used for construction of an siRNA vector targeting human DJ-1 gene is 5′-GGATCCCGTCAAGGCTGGCATCAGGACAATTGATATCCGTTGTCCTGATGCCAGCCTTGATTTTTTCCAAAAGCTT-3′. After annealing oligonucleotides corresponding to the upper and lower strands of DNA, they were inserted into
6-carboxy-2′,7′-Dichlorodihydrofluorescein Diacetate Di(acetoxymethyl) Ester Staining
To detect H2O2-induced ROS production, we used a redox-sensitive dye, 6-carboxy-2′,7′-dichlorodihydrofluorescein diacetate di(acetoxymethyl) ester (Molecular Probes) (Kitamura et al, 1998). 6-Carboxy-2′,7′-dichlorodihydrofluorescein diacetate di(acetoxymethyl) ester is readily taken up by cells. When it reacts with ROS, it is converted into 6-carboxy-2′,7′-dichlorodihydrofluorescence (C-DCF), and subsequently illuminates at an excitation wavelength of 488 nm. After the SH-SY5Y cells were prepared in uncoated glass-bottomed microwells (inner diameter, 18 mm), 6-carboxy-2′,7′-dichlorodihydrofluorescein diacetate di(acetoxymethyl) ester was added to the cell culture to a final concentration of 2 μmol/L for 10 mins at 37°C. After two rinses with a serum-free medium, the fluorescence intensity of C-DCF was scanned under a confocal microscope (LSM410). As illumination at an excitation wavelength of 488 nm causes increased fluorescence due to oxidation of this dye (Kitamura et al, 1998), each field was exposed to light for exactly the same amount of time. Fluorescence intensity was quantified by computerized image analysis (WinRoof).
Measurement of Cell Viability
To evaluate cell viability, we performed a 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyltetrazolium bromide (MTT; Dojindo Laboratories) assay as an index of surviving cells. In living cells, MTT is converted to formazan, which has a specific absorption maximum. SH-SY5Y cells, in which endogenous DJ-1 expression was normal or knocked down by an siRNA vector, were seeded at 3 × 104 cells/well in a 96-microwell plate and treated with 50 or 100 μmol/L H2O2 at 24 h after plating. At 24 h after treatment, the culture medium was changed to a medium containing 5 mg/mL MTT, and the cells were incubated for an additional 4 h. They were then mixed thoroughly with an equal volume of isopropanol/0.04 mol/L HCl and sonicated to dissolve formazan completely. Absorbance was then measured at 570 nm with a microplate reader (Bio-Rad Laboratories, Hercules, CA, USA).
Western Blot Analysis
Cell lysates were diluted with Laemmli's sample buffer and subjected to sodium dodecyl sulfate—polyacrylamide gel electrophoresis (15% polyacrylamide gel), and immunoblotting was then carried out using antibodies against human DJ-1 (1:5,000) and β-actin (1:10,000). For semiquantitative analysis, the bands of these proteins on radiographic films were scanned with a charge-coupled device color scanner (DuoScan; AGFA, Leverkusen, Germany), and then analyzed. Densitometric analysis was performed using the public domain program NIH Image 1.56 (written by Wayne Rasband at the US National Institutes of Health and available from the Internet through anonymous FTP from zippy.nimh. nih.gov).
Statistical Analysis
Results are given as the mean±s.e.m. The significance of differences was determined by Student's
Results
Effect of Recombinant Glutathione S-Transferase-Tagged Human DJ-1 on Infarct Size
In a preliminary experiment, to detect the human DJ-1 protein, which was exogenously injected into the rat striatum, we compared immunohistochemical staining between anti-human DJ-1 antibody and anti-GST antibody. Anti-human DJ-1 antibody induced nonspecific staining with moderate intensity of some neural staining, but this antibody did not recognize rat DJ-1 by immunoblotting (Supplementary Figure S1). While anti-GST antibody specifically reacted with GST-DJ-1 without nonspecific staining in the rat brain (Supplementary Figure S1). As we reported previously antioxidative and cellular protective effects of GST-free DJ-1 (Taira et al, 2004) and GST-DJ-1 (Inden et al, 2006), we basically consider that GST-DJ-1 has almost the same properties as GST-free DJ-1. On the basis of these observations, we used GST-DJ-1 in this study, while we utilized PBS and GST (320 pmol) as vehicle controls, as GST-DJ-1 (4 to 320 pmol) was injected with PBS.
At 3 days after MCAO, although a regional loss of TTC staining was observed in the ipsilateral cerebral cortex and the striatum in PBS- and GST-injected rats, the area of TTC staining lost was smaller with the administration of GST-DJ-1 at 110 mins after the onset of MCAO (10 mins before reperfusion; Figure 1A). In the quantitative analysis, the infarct area was reduced in a dose-dependent manner by the administration of GST-DJ-1, and the total infarct volume was significantly reduced by the administration of GST-DJ-1 at 160 and 320 pmol, compared with PBS- and GST-injected rats (Figures 1B and 1C).
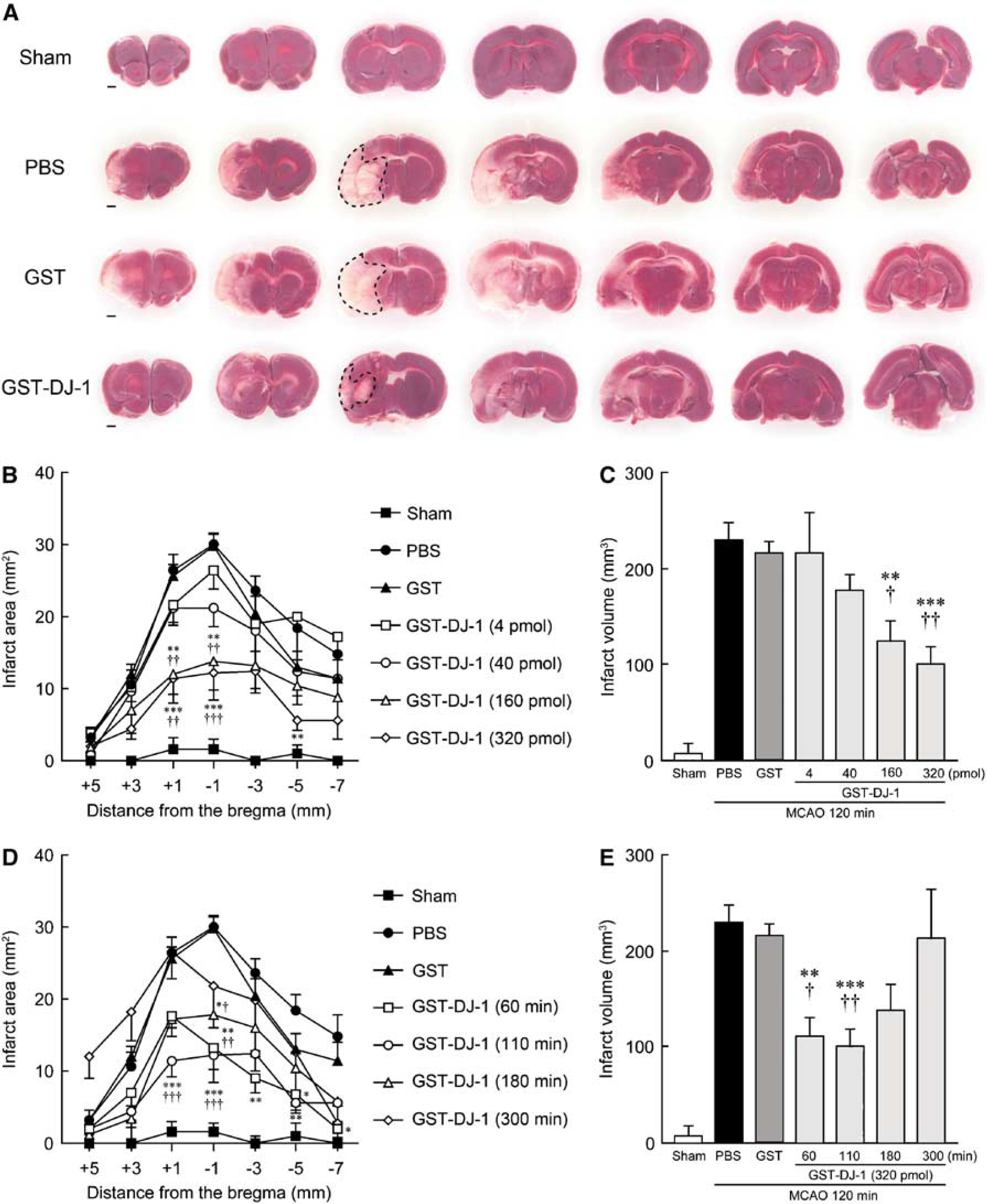
Glutathione
The delayed administration of GST-DJ-1 (320 pmol) significantly reduced the infarct volume when the injection was performed at 60 and 110 mins after the onset of MCAO. However, this effect was lost when the injection was delayed to 180 mins (Figures 1D and 1E). At that time, although the total infarct volume did not significantly change, the infarct area at the level of −1 mm from the bregma was significantly smaller with the administration of GST-DJ-1 at 180 mins after the onset of MCAO (Figure 1D).
Quantification of Infarct Lesion Using Magnetic Resonance Imaging
We further investigated the neuroprotective effect of GST-DJ-1 using MR imaging. In sham-operated rats, which had been injected with PBS and GST-DJ-1 (320 pmol), any difference of MR signal between PBS- and GST-DJ-1-injected rats was not detected on T2-weighted MR images at 7 days after the injection (Figure 2A). In ischemic rats, which had been injected with PBS at 110 mins after the onset of MCAO, the infarct lesion, as represented by an area of hyperintensity (white), was markedly observed in the ipsilateral cortex and striatum at 7 days after MCAO (Figure 2B and Supplementary Movie S2). In contrast, the infarct lesion was markedly smaller in ischemic rats, which had been injected with GST-DJ-1 (320 pmol) at 110 mins after the onset of MCAO (Figure 2B and Supplementary Movie S2). In the quantitative analysis, GST-DJ-1-injected rats showed a significant reduction of infarct size, compared with PBS-injected rats (Figures 2C and 2D).
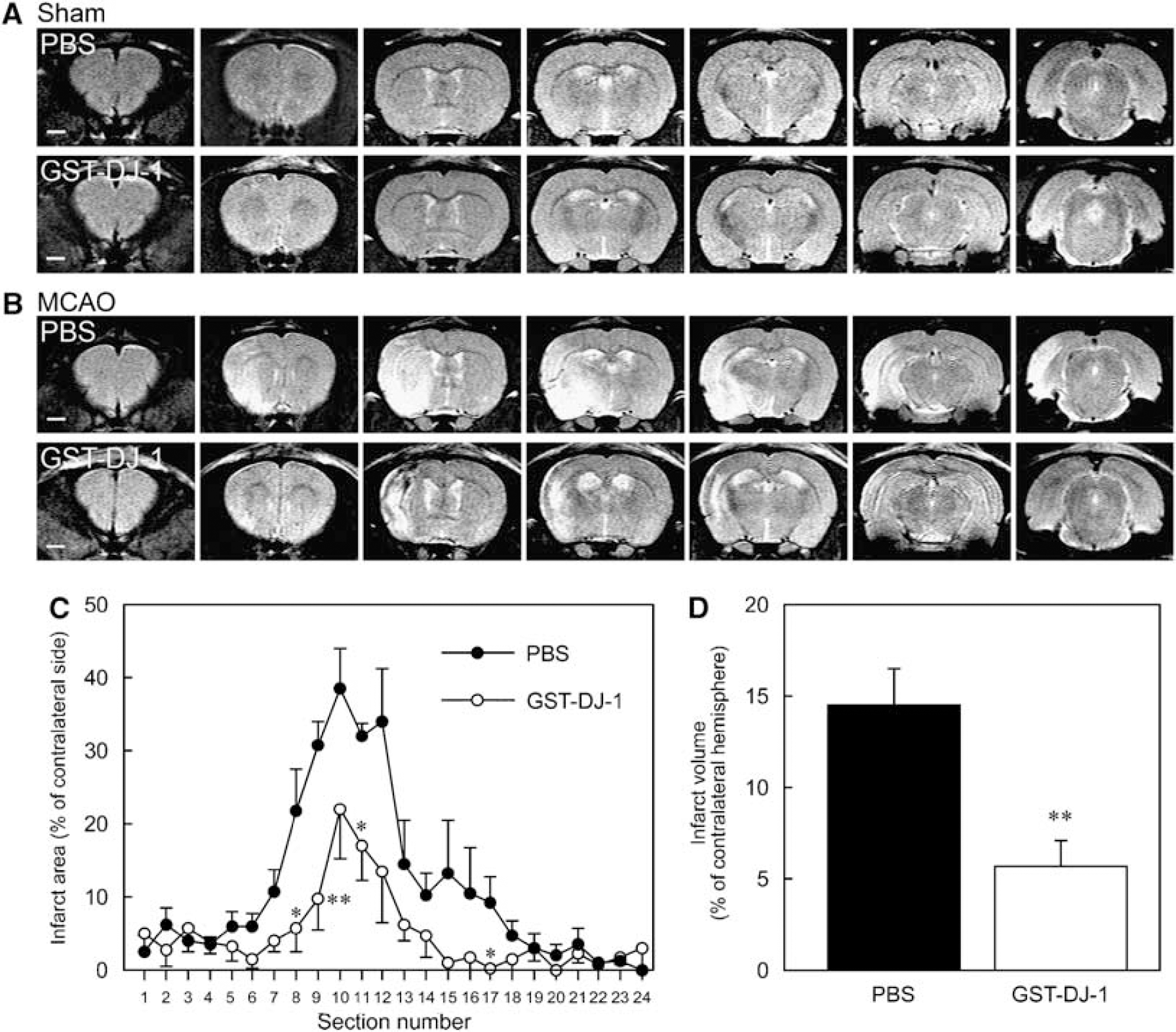
Changes in T2-weighted magnetic resonance (MR) images after the injection of GST-tagged human DJ-1 (GST-DJ-1).
Effect of Glutathione S-Transferase-Tagged Human DJ-1 on Ischemia-Induced Behavioral Dysfunction
To evaluate the effect of GST-DJ-1 on behavior, the animals were subjected to sham operation or MCAO, followed by injection with PBS, GST (320 pmol), or GST-DJ-1 (320 pmol) at 110 mins, and reperfusion at 120 mins after the onset of MCAO. Neurologic evaluation was assessed at 30 mins after the onset of MCAO (0 day) and subsequently every day until the rats were killed at 7 days after MCAO. Sham-operated rats showed no abnormal behavior at any time points. At 30 mins after the onset of MCAO (80 mins before injection of PBS, GST, or GST-DJ-1), ischemic rats caused severe neurologic deficits and did not show any difference in neurologic score among these rats (at 0 day). Subsequently, the neurologic grade in PBS- and GST-injected ischemic rats gradually decreased. However, GST-DJ-1-injected rats showed more improvement of neurologic grade than PBS- and GST-injected rats. In brief, neurologic deficits were significantly recovered at 3, 4, 5, and 6 days by GST-DJ-1 injection, in comparison with those by injection of PBS or GST (Figure 3A).
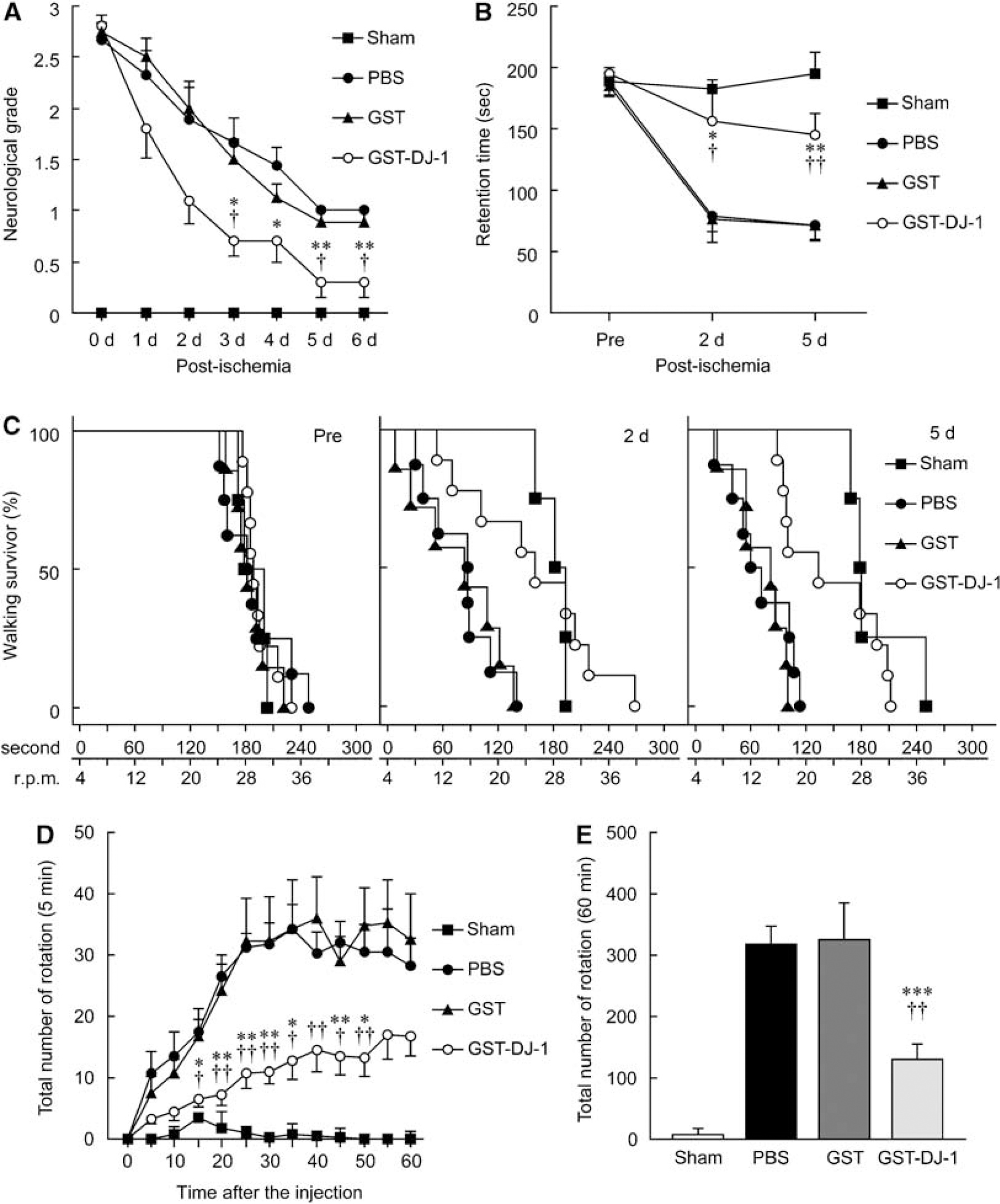
Effect of GST-tagged human DJ-1 (GST-DJ-1) on behavioral dysfunction induced by cerebral ischemia and reperfusion. Rats were subjected to MCAO followed by injection with PBS (
In the rota-rod test, there was no difference among PBS-, GST-, and GST-DJ-1-injected rats at 1 day before MCAO (preperformance). At 2 and 5 days after MCAO, the retention time on the rod markedly shortened in PBS- and GST-injected rats, compared with preperformance. In contrast, shortening of retention time was significantly inhibited by administration of GST-DJ-1 (Figure 3B). In addition, the population of ischemic rats, which walked for a long time on the accelerating rotating rod, significantly increased by GST-DJ-1 injection at 2 and 5 days after MCAO, in comparison with that by PBS injection (
We further examined methamphetamine-induced rotation asymmetry. In PBS- or GST-injected rats, the total number of rotations every 5 mins gradually increased until 30 mins after methamphetamine injection, and then the rats showed stable rotation of approximately 30 turns for 5 mins (Figure 3D). The total number of rotations in 60 mins reached 300 turns in PBS- and GST-injected rats. In contrast, the total number of rotations in GST-DJ-1-injected rats was significantly reduced (Figure 3E).
Changes in Immunoreactivities for Microtubule-Associated Protein-2, Tyrosine Hydroxylase, and Nitrotyrosine
It is known that damaged tissues sometimes cause nonspecific staining. To reduce this possibility, we usually performed the incubation at 4°C (in a cold room) with a lower concentration of primary antibody. In addition, when brain slices were incubated with mouse IgG1 or rabbit IgG as control, instead of the primary antibody, nonspecific staining was undetectable in the ischemic core and in the boundary zone of the ischemic region, under our experimental condition (Supplementary Figure S3).
At 7 days after MCAO, the treated rats were killed. For immunohistochemical analysis, sections were prepared and immunostaining with antibodies against MAP2, TH, and nitrotyrosine was performed. In sham-operated rats, although MAP2 immunoreactivity was detected in the cerebral cortex and the striatum (Figures 4A and 4E), this immunoreactivity was markedly reduced in the ipsilateral cerebral cortex and the striatum in PBS- and GST-injected rats (Figures 4B, 4C, 4F, and 4G). In contrast, GST-DJ-1-injected rats showed a reduction in the loss of MAP2 immunoreactivity in both the cerebral cortex and the striatum (Figures 4D and 4H). In addition, we evaluated TH immunoreactivity as an index of the level of dopaminergic neuronal injury. Presynaptic and neural fiber TH immunoreactivity was mainly detected in the striatum (Figure 4I), and TH-immunopositive neural cell bodies were detected in the SNpc in sham-operated rats (Figure 4M). Although fibrillar TH immunoreactivity in the striatum and the number of TH-immunopositive neurons in the SNpc were reduced by MCAO (Figures 4J, 4K, 4N, and 4O), these reductions were markedly inhibited in GST-DJ-1-injected rats (Figures 4L and 4P). By semiquantitative analysis, similar results were obtained (Supplementary Figure S4).
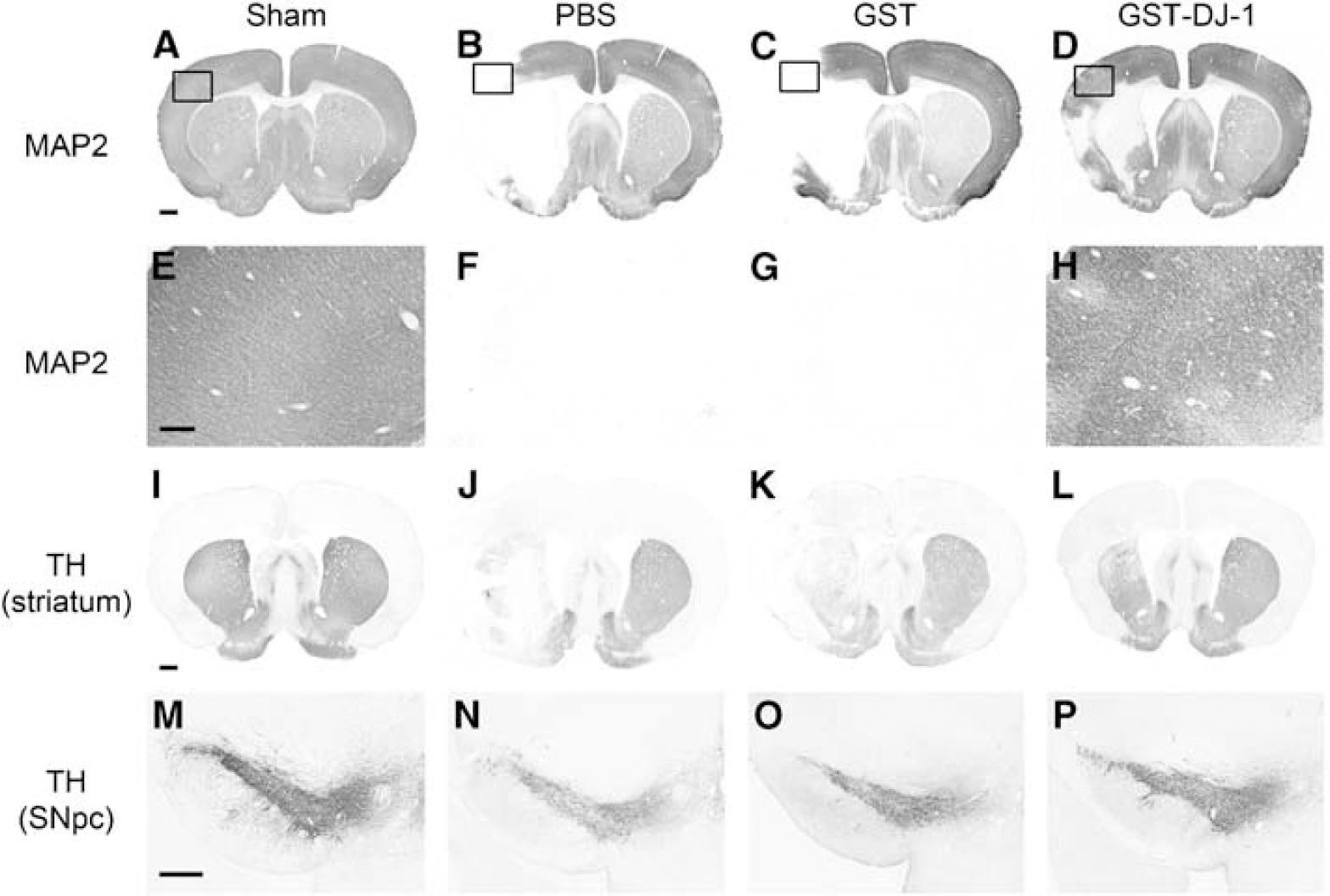
Immunohistochemical changes after the injection with GST-tagged human DJ-1 (GST-DJ-1) in ischemic rats.
However, normal rats that were injected with GST-DJ-1 did not exhibit any abnormal behavior (data not shown). In addition, as MAP2-immunoreactive neural fibers in GST-DJ-1-injected ipsilateral hemisphere did not change for 7 days after injection (Supplementary Figure S5), GST-DJ-1 may not affect naive neurons.
We further estimated nitrotyrosine immunoreactivity as an index of the level of oxidative and nitrosative stress. Although immunoreactivity for nitrotyrosine was hardly detected in sham-operated rats (Figures 5A, 5E, 5I, and 5M), this immunoreactivity was markedly increased in PBS- and GST-injected rats in the boundary zones of the ischemic core in the cerebral cortex and the striatum and in the ischemic core in the striatum (Figures 5B, 5C, 5F, 5G, 5J, 5K, 5N, and 5O). In contrast, in GST-DJ-1-injected rats, the number of nitrotyrosine-immunopositive cells was significantly reduced in the boundary zones of the ischemic core in the cerebral cortex and striatum, but not in the ischemic core in the striatum (Figures 5D, 5H, 5L, 5P, 5Q, 5R, and 5S).
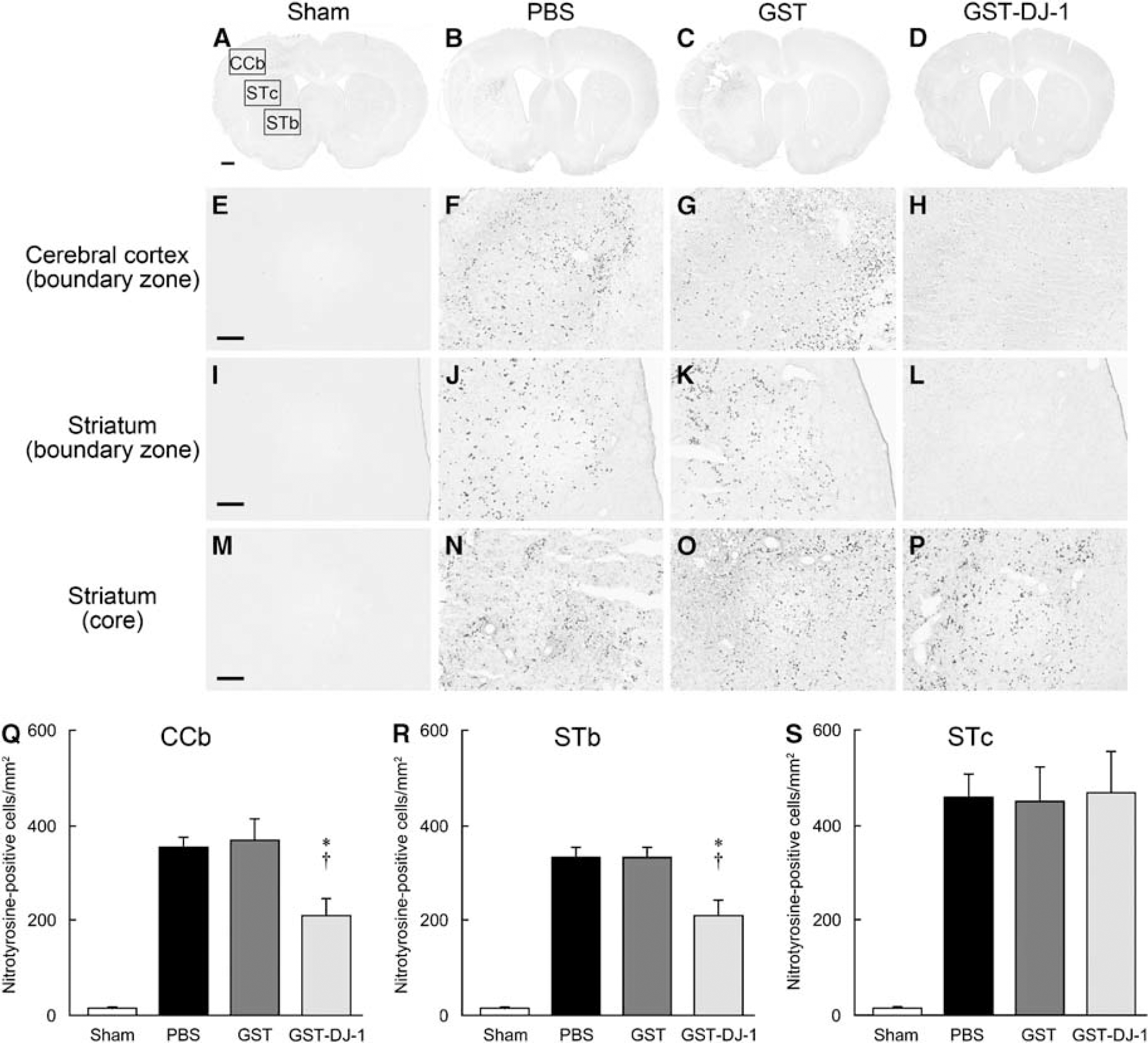
Effect of GST-tagged human DJ-1 (GST-DJ-1) on oxidative and nitrosative stress induced by cerebral ischemia and reperfusion. (
Distribution of Glutathione S-Transferase-Tagged Human DJ-1 after Intrastriatal Injection
To investigate the distribution of exogenous human DJ-1 in the rat brain, we performed the intrastriatal injection of GST-DJ-1 (320 pmol). Exogenous GST-DJ-1 was then detected using an anti-GST antibody. GST immunoreactivity for exogenous GST-DJ-1 was detected throughout the striatum and reached the cerebral cortex at 0.5 h after injection (Figure 6A). Thereafter, GST immunoreactivity gradually decreased until 24 h after injection and was hardly detected at 72 h (Figures 6B and 6C).
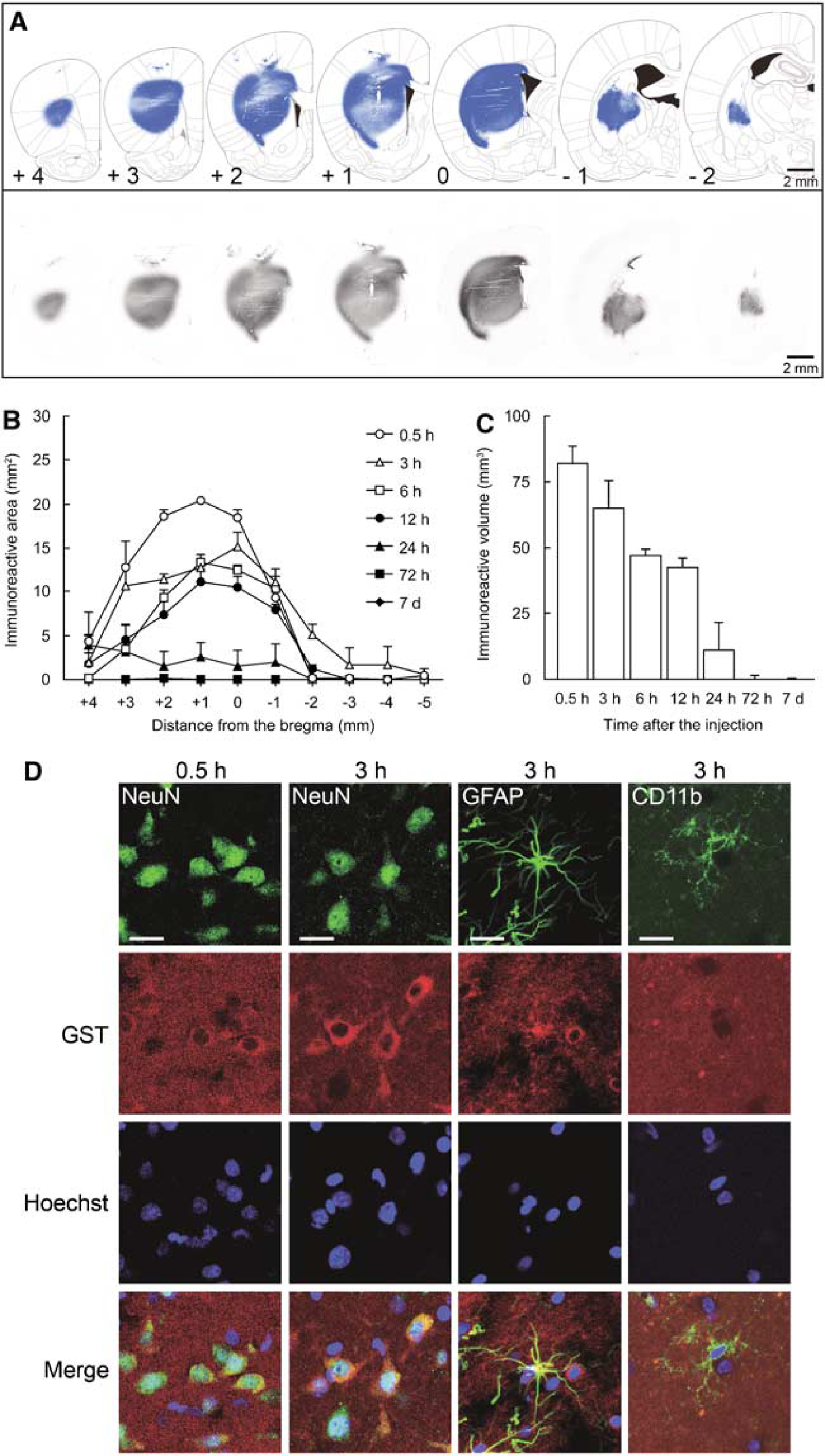
GST immunoreactivity for exogenous GST-tagged human DJ-1 (GST-DJ-1) in the rat brain. (
To clarify whether exogenous GST-DJ-1 was taken up by neurons and/or glial cells, we performed double staining using antibodies against GST and NeuN, glial fibrillary acidic protein, or CD11b. At 0.5 h after injection, GST immunoreactivity was markedly detected at the extracellular space and in NeuN-immunopositive neurons (Figure 6D). At 3 h after injection, GST immunoreactivity was detected at the extracellular space and was stronger in NeuN-positive neurons (Figure 6D). Conversely, at 3 h after injection, GST immunoreactivity was partially detected in glial fibrillary acidic protein-immunopositive astrocytes and was scarcely detectable in CD11b-immunopositive microglia (Figure 6D).
Glutathione S-Transferase-Tagged Human DJ-1 Inhibits H2O2-Induced Reactive Oxygen Species Production and Cell Death in human SH-SY5Y cells and DJ-1-Knockdown Cells
Recent structural and biochemical analyses have shown that DJ-1 forms a homodimer and that loss of the homodimer formation of L166P-DJ-1 results in loss of its function (Taira et al, 2004; Takahashi-Niki et al, 2004; Honbou et al, 2003). To investigate whether GST, GST-DJ-1, or GST-L166P-DJ-1 affected the level of ROS production, we performed a confocal analysis with a ROS-specific fluorogen C-DCDHF-DA. H2O2-induced intracellular C-fluorescence markedly increased when normal human SH-SY5Y cells were treated with 100 μmol/L H2O2 for 1 h (Figure 7A).
At that time (after 1 h), H2O2-induced intracellular ROS production was significantly inhibited by simultaneous treatment with 1 μmol/L GST-DJ-1 but not by treatment with GST or GST-L166P-DJ-1 (Figures 7A to 7C). Furthermore, after 24 h, H2O2-induced cell death was also significantly inhibited by 1 μmol/L GST-DJ-1 but not by GST and GST-L166P-DJ-1 (Figure 7D).
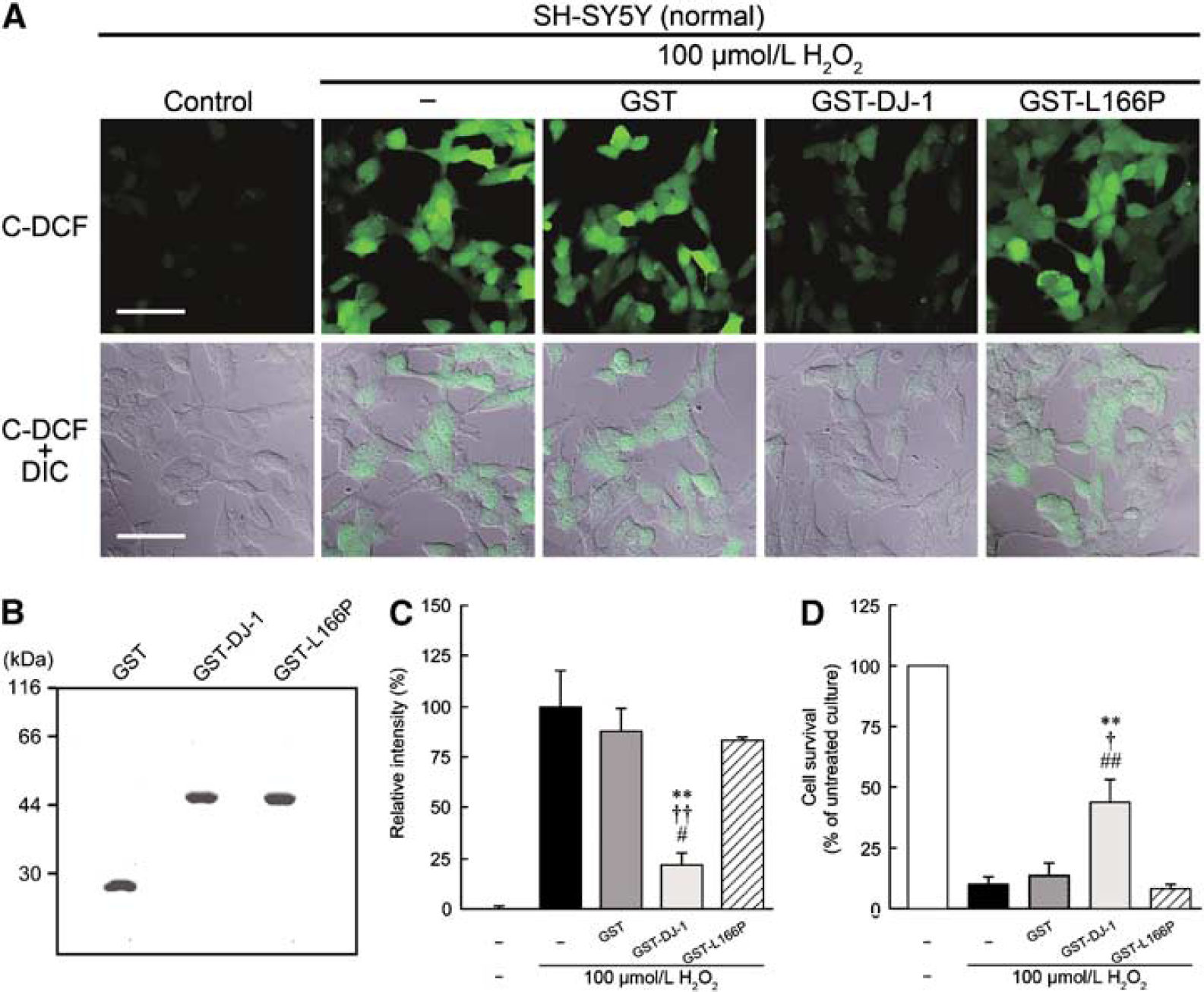
Effect of GST-tagged human DJ-1 (GST-DJ-1) on H2O2-induced oxidative stress in normal SH-SY5Y cells. 6-Carboxy-2′,7′-dichlorodihydrofluorescein diacetate di(acetoxymethyl) ester (C-DCDHF-DA) staining was performed at 1 h, and MTT assay for cell survival at 24 h after treatment with 100 μmol/L H2O2 in the presence or absence of 1 μmol/L GST, 1 μmol/L GST-DJ-1, or 1 μmol/L GST-tagged L166P-mutant human DJ-1 (GST-L166P). (
We also established DJ-1-knockdown SH-SY5Y cell line, in which the expression of endogenous human DJ-1 was suppressed by approximately 76% (Figure 8A). In normal SH-SY5Y cells, ROS production was induced markedly by 100 μmol/L H2O2 (Figure 7), it was induced moderately by 50 μmol/L H2O2 (Figures 8B and 8C). In DJ-1-knockdown cells, even 50 μmol/L H2O2 induced significant ROS production after 1 h. In addition, massive cell death of DJ-1-knockdown cells was caused by 50 μmol/L H2O2 in comparison with normal cells (Figure 8D). H2O2-induced intracellular ROS production and cell death in DJ-1-knockdown cells were significantly inhibited by GST-DJ-1 (Figures 8C and 8D). In contrast, GST and GST-L166P-DJ-1 had no effect.
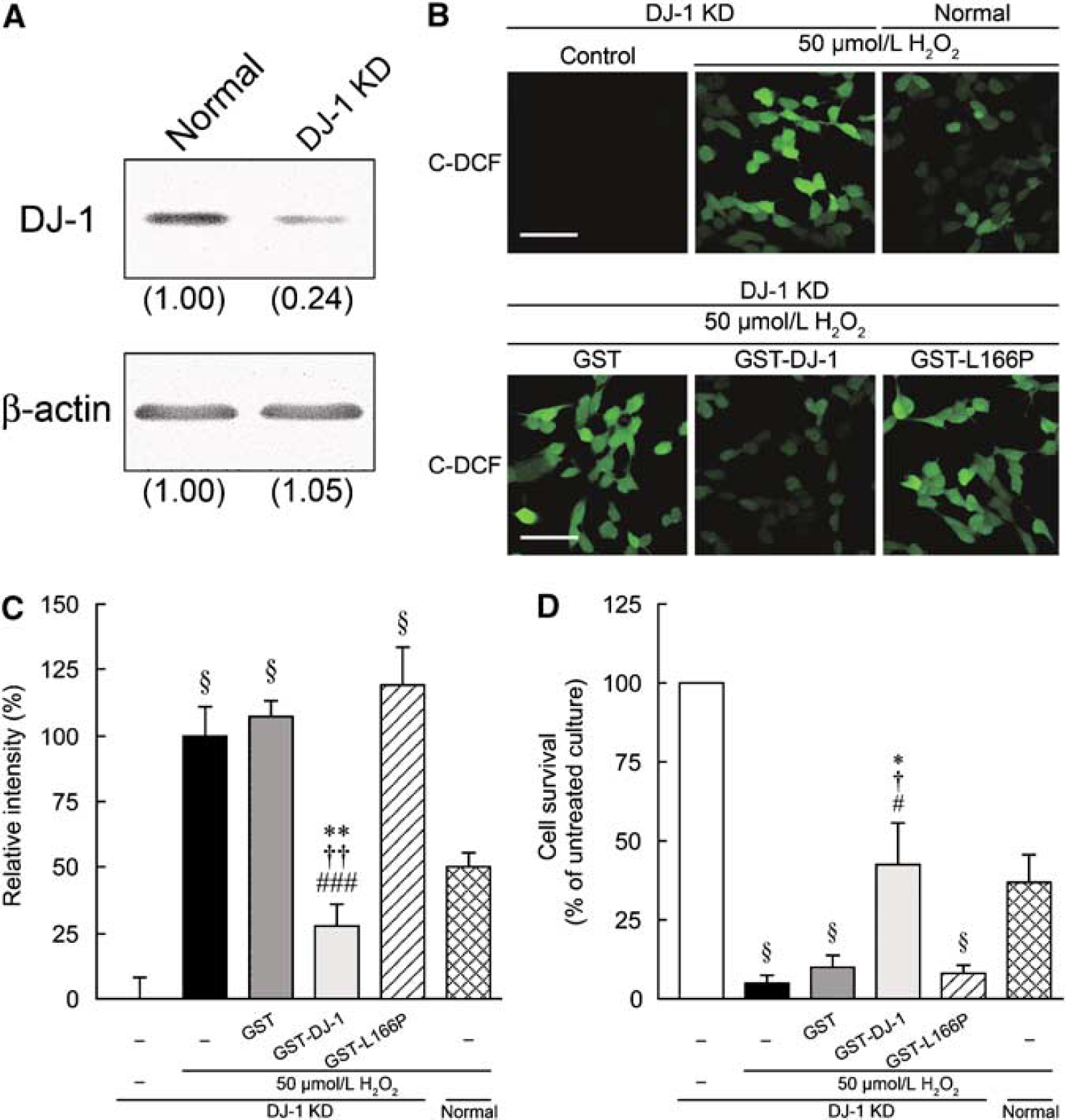
Effect of GST-tagged human DJ-1 (GST-DJ-1) on H2O2-induced oxidative stress in DJ-1-knockdown SH-SY5Y cells. (
Discussion
Although oxygen is necessary for life, it paradoxically produces ROS, which is highly toxic to cells, as a by-product of its metabolism. Reactive oxygen species is massively produced in the brain after cerebral ischemia and reperfusion. All of these species are redox active and can interact with nearby cellular components, such as proteins, lipids, and DNA (Andersen, 2004; Margaill et al, 2005). In this study, we revealed that the intrastriatal injection of recombinant GST-DJ-1 inhibited both ischemic neuronal cell loss and behavioral dysfunction induced by 120 mins of MCAO and reperfusion. It has been reported that cells in which endogenous DJ-1 was knocked down or cells which overexpressed point-mutated DJ-1 (such as L166P and C106S) were vulnerable to oxidative stress (Taira et al, 2004; Takahashi-Niki et al, 2004). More recently, we showed that treatment with GST-DJ-1 protected against nigral dopaminergic neuronal cell death induced by 6-hydroxydopamine-mediated oxidative stress (Inden et al, 2006). These observations suggest that the intrastriatal injection of GST-DJ-1 inhibits ROS-induced neurodegeneration caused by ischemia and reperfusion.
In this study, we injected GST-DJ-1 at 60, 110, 180, or 300 mins after the onset of MCAO. Significant reductions in infarct size were observed when the injection was performed at 60 and 110 mins, and partial reductions were seen at 180 mins, but no reductions in size were seen with the injection at 300 mins. Previous reports have suggested that during 120 mins of MCAO, the levels of 2,3-dihydroxybenzoic acid and 2,5-dihydroxybenzoic acid, as indicators of hydroxyl radicals, increased to values that were 2- to 2.5-fold above baseline and further increased to nearly fourfold at 30 mins after reperfusion (Morimoto et al, 1996). Another group reported that a short-lasting increase in lucigeninenhanced chemiluminescence (an indicator of superoxide production) by nearly fivefold occurred immediately after the initiation of reperfusion in 120 min-MCAO rats (Peters et al, 1998). Moreover, it was reported that the level of NOx (including NOO− ONOO−) increased immediately after the onset of reperfusion, and this elevation continued more than 120 mins (Hashiguchi et al, 2003). These observations suggest that ROS production begins to increase gradually after the onset of ischemia, and massively and immediately after reperfusion. Therefore, it seems that when the intrastriatal injection of GST-DJ-1 was performed at 300 mins after the onset of MCAO, ROS production was massively induced, and brain tissue had already been damaged. Furthermore, although exogenous GST-DJ-1 was detected throughout the striatum and partially in the cerebral cortex, the region in which GST-DJ-1 was detected was limited and smaller than the middle cerebral artery-ischemic territory. On the basis of these observations, in the case of intrastriatal injection, the time window of the neuroprotective efficacy of GST-DJ-1 is 110 mins (10 mins before reperfusion) or partially until 180 mins after the onset of MCAO.
In this study, we further performed a noninvasive evaluation of infarct size, using a 7T Unity Inova MR scanner, and found a reduction in infarct size after the administration of GST-DJ-1. Thus, we suggest that MR imaging is a powerful tool for noninvasive measurement of the extent of ischemic brain injury.
We used a redox-sensitive dye 6-carboxy-2′, 7′-dichlorodihydrofluorescein diacetate di(acetoxymethyl) ester to detect H2O2-mediated ROS production in the
In the immunohistochemical analysis, the formation of nitrotyrosine, which is considered to be a long-lived footprint of NO and ONOO− (Osuka et al, 2001), was inhibited by the administration of GST-DJ-1. Ischemia/reperfusion injury produces massive amounts of superoxide anion and nitric oxide. Nitric oxide reacts with superoxide anion and is then converted to peroxynitrite, which strongly reacts with cellular components. When proteins react with peroxynitrite, protein nitration occurs and this results in cellular injury. DJ-1 protein harbors three cysteine residues. It has been shown that DJ-1 forms a homodimer and that Cys106 is selectively oxidized under oxidative stress to eliminate ROS (Canet-Aviles et al, 2004; Honbou et al, 2003; Huai et al, 2003; Kinumi et al, 2004; Taira et al, 2004; Tao and Tong, 2003; Wilson et al, 2003). Furthermore, it has been reported that Cys46 and Cys53, but not Cys106, are susceptible to
To evaluate a neuroprotective effect of DJ-1, we investigated neural cell injury in the immunostained sections. Recently, unbiased stereological technique (such as three-dimensional stereological method) has come to be known a powerful tool with high potential for the counting of critical neural cell numbers in brain sections. However, the aim of this study was to evaluate neural cell injury, but not to determine changes in the critical number of neural cells. Therefore, the number of immunostained neurons was measured using a computerized image analysis based on a two-dimensional morphologic method (Figure 5 and Supplementary Figure S4), which is also one of the applied unbiased methods that is even now widely used for cell counting in damaged tissues (Inden et al, 2006). In this technique, each immunostained neuron is recognized and counted independently to cell size by the computerized image software. It is known that atrophy of the substantia nigra occurs in MCAO rats. In permanent MCAO rats, atrophy did not detect at 1 week, but it markedly occurred at 2 weeks (Tamura et al, 1990). As we performed 2-h MCAO and reperfusion (but not permanent) and immunohistochemical assay at 7 days after MCAO, atrophy in the cerebral cortex, striatum, and substantia nigra was undetectable. Therefore, although the stereological method is now the gold standard to evaluate cell loss, we assessed neuroprotective effect of DJ-1 by a computerized image analysis in this study.
We showed that DJ-1 had a neuroprotective effect against cerebral ischemia and reperfusion in rats. DJ-1 is widely expressed in the human and rodent brain (Bandopadhyay et al, 2004; Olizmann et al, 2007; Yanagida et al, 2006). Furthermore, it has been suggested that DJ-1 plays an important role in systems that defend against oxidative stress (Canet-Aviles et al, 2004; Kinumi et al, 2004; Mitsumoto et al, 2001; Taira et al, 2004). Therefore, it is believed that the development of drugs or treatments that enhance the expression of endogenous DJ-1 in the brain may be a novel therapeutic strategy in neurodegenerative disorders. Recently, peptide carriers, such as TAT and PEP-1, have been studied with regard to their ability to promote protein transduction not only
In conclusion, this is the first report to show that GST-DJ-1 has a neuroprotective effect in an
Conflict of interest
The authors declare no conflict of interest.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
