Abstract
Calcitonin gene-related peptide (CGRP) and adrenomedullin (ADM) are potent dilators of human brain arteries, and they have been implicated in the neurogenic inflammation underlying migraine headache and in the evolution of stroke, respectively. However, little is known about the presynaptic and postsynaptic distribution of their respective receptors in the human cerebrovascular bed and trigeminovascular system. In the current study, the expression of mRNA for ADM and the two cloned human CGRP1 receptors (identified here as A-CGRP1 receptors [Aiyar et al., 1996] and K-CGRP1 receptors) [Kapas and Clark, 1995] were evaluated in human brain vessels and trigeminal ganglia. Further, the ability of CGRP and ADM to activate adenylate cyclase in cerebromicrovascular and astroglial cell cultures was determined, and the receptors involved were characterized pharmacologically. Isolated human pial vessels, intracortical microvessels, and capillaries, as well as cultures of brain endothelial (EC), smooth muscle (SMC), and astroglial (AST) cells, all expressed mRNA for the two cloned CGRP1 receptors; however, message for the K-CGRP1 receptor was barely detectable in microvascular tissues and cells. In contrast, only isolated capillaries and cultured AST exhibited message for the ADM receptor. In human trigeminal ganglia, mRNA for ADM and the two CGRP1 receptors was systematically present The CGRP dose-dependently increased (up to 50-fold) cAMP formation in cell cultures, an effect significantly blocked by 0.1 to 10 μmol/L of the CGRP1 receptor antagonist CGRP8–37. The ADM receptor agonist, ADM13–52 (1 μmol/L), similarly increased cAMP production in all cell types, and this response was virtually abolished by 1 μmol/L CGRP8–37. Low concentrations (1 to 10 μmol/L) of the ADM receptor antagonist ADM22–52 blocked the ADM13–52-induced cAMP formation in AST (26% at 10 μmol/L, P < 0.05), whereas they potentiated this response in brain EC and SMC (40% and 100%, P < 0.001, respectively). Even at a higher dose (50 μmol/L), ADM22–52 inhibited the ADM13–52 effect in vascular cells (45%) much less effectively than in AST (95%). These results indicate that both CGRP and ADM can affect human brain vessels through a CGRP1 receptor, and they further suggest the presence of functional ADM receptors in human astroglial cells.
Calcitonin gene-related peptide (CORP) is a 37 -amino acid peptide generated from the alternative splicing of the calcitonin gene and, among other functions, it is an exquisitely potent dilator of cerebrovascular smooth muscles (Edvinsson et al., 1987; Jansen-Olesen et al., 1996). It displays about 20% sequence homology with adrenomedullin (ADM), another vasoactive peptide purified and sequenced from human pheochromocytoma (Van Rossum et al., 1997), Both peptides have been implicated in cerebrovascular disorders: CORP in either the etiology of migraine headache (Moskowitz, 1992) or in vasospasm after subarachnoid hemorrhage (Juul et al., 1990), and ADM in the evolution of stroke (Wang et al. 1995).
Calcitonin gene-related peptide exerts its effects through G-protein—coupled receptors, which have been classified into CORP1 and CORP2 receptors based on the differential biological activity of various CORP analogues, but primarily of the C-terminal fragment CORP8–37, which displays antagonistic properties with higher affinity at the CORP1 than CORP2 receptors (Van Rossum et al., 1997). Only the CORP1 receptor has been cloned, and two different genes encoding a pharmacologically defined CGRP1-type receptor have been identified (Aiyar et al., 1996; Kapas and Clark, 1995), which are referred to as A-CGRP1 and K-CGRP1 receptors, respectively, in the current study. Their localization and physiologic implications are largely unknown, and no pharmacologic tools are available to discriminate between them.
Major cerebral arteries and small pial vessels are richly innervated by CGRP-containing nerve fibers, which originate mainly in the ophthalmic division of the trigeminal ganglia (Uddman et al., 1985). The presence of regulatory presynaptic CGRP receptors has not been explored in the trigeminovascular system, but message for the A-CGRP1 receptor has been found in human trigeminal ganglia (Edvinsson et al., 1997). In both large cerebral arteries (Edvinsson et al., 1987; Jansen-Olesen, et al., 1996) and small intraparenchymal microvessels (Edwards et al., 1991; Fergus et al., 1995), CGRP is a potent and long-lasting vasodilator. This response is likely mediated by a smooth muscle CGRP1 receptor, since it is insensitive to endothelial denudation (Jansen-Olesen et al., 1996) and it is blocked by the CGRP1 receptor antagonist CGRP8–37 (Edwards et al., 1991; Jansen-Olesen et al., 1996). However, the participation of a CGRP2 receptor in this response has not been excluded (Jansen-Olesen et al., 1996).
Adrenomedullin also induces a potent relaxation of cerebral arteries (Baskaya et al., 1995; Mori et al., 1997) and reportedly increases cAMP production in vascular smooth muscle cells (Ishizaka et al., 1994). However, ADM is not contained within perivascular fibers but has been found in circulating blood (Kitamura et al., 1994) and can be constitutively synthesized and secreted by endothelial and vascular smooth muscle cells (Sugo et al., 1994a, b ). Although a specific ADM receptor has been cloned and sequenced in human tissues (Hänze et al., 1997), pharmacologic evidence suggests that ADM-induced vasomotor activity most likely is mediated through interaction with vascular CGRP1 receptors, which bind ADM with a similar (Baskaya et al., 1995) or slightly lower (Nuki et al., 1993) affinity than CGRP itself.
The current study (1) evaluates the expression of ADM and the two cloned CGRP1 receptors in the human trigeminal ganglia and in different compartments of the cerebrovascular bed, (2) identifies their cellular (endothelial, muscular, or astroglial) localization within the vessel wall, and (3) assesses their functional coupling to adenylate cyclase in human cerebromicrovascular and astroglial cell cultures. Part of these results have appeared in abstract form (Moreno et al., 1997,1998).
MATERIALS AND METHODS
Preparation of tissues and cell cultures
Human trigeminal ganglia, and cerebrovascular and brain tissues. Human brain tissues (n = 5 halves cerebral hemisphere) and trigeminal ganglia (n = 5) were obtained at autopsy (postmortem delay of 2 to 24 hours) from the Royal Victoria Hospital (Montreal, QC, Canada) or the Human Brain Bank from the Douglas Hospital Research Centre (Verdun, QC, Canada) from both male and female subjects who died from diseases not related to the CNS. All tissues were obtained with approval from the Institutional Research Ethics Committee. When required, samples of striatum and cerebellum were dissected out for use as control tissues (Van Rossum et al., 1997). Trigeminal ganglia were removed from the skull base and cleaned of any attached tissue. Pial vessels (PV) were carefully removed from the surface of the cortical mantle in cold Krebs-Ringer buffer under a dissecting microscope. Isolated cortical microvascular fractions, corresponding to resistance microvessels (MV) (l50-μm mesh) or small microvessels and capillaries (CAP, 50-μm mesh) were prepared as described previously (Linville and Hamel, 1995). These have been shown to be highly enriched in vascular elements by light microscopic study and in endothelial marker enzymes γ-glutamyltranspeptidase and alkaline phosphatase (Linville and Hamel, 1995). All tissues were frozen at −45°C in isopentane and kept at −80°C until use.
Human brain microvascular and astroglial cell cultures. Cultures of human brain smooth muscle cells (SMC, n = 5 primary cultures) and endothelial cells (EC, n = 5, passages 3 through 5) were generated from microvascular fractions isolated from human temporal cortex surgically removed from patients undergoing treatment for idiopathic epilepsy, as approved by the Institutional Research Ethics Committee (for details, see Stanimirovic et al., 1996; Abounader et al., 1999). Briefly, SMC cultures were grown from the microvessels isolated by a sequential filtration of cerebral cortex homogenates on 350- and 112-μm nylon mesh screens. The trapped resistance microvessels were resuspended in cold medium 199, dissociated by incubation with type IV collagenase (1 mg/mL, 15 minutes), then seeded and maintained at 37°C in an atmosphere of 5% CO2 in air. The cells were allowed to grow as primary culture for 4 to 5 weeks until the SMC outgrew the initial sprouting of EC. More than 85% of cells stained positively for the SMC protein, α-actin (Accurate Chemical and Scientific Corp., Westerbury, NY, U.S.A.), whereas about 10% demonstrated EC morphologic features and incorporated acetylated low-density lipoproteins.
Endothelial cells were obtained by further sieving the 112-μm mesh screen filtrates through a 20-μm mesh. The recovered small microvessels and CAP were dissociated with type IV collagenase (15 minutes, 37°C), subcloned, and maintained at 37°C in the same atmosphere and medium as described earlier for SMC, but also containing 10 mg/mL EC growth supplements (see Stanimirovic et al., 1996). In addition, EC were harvested from two different segments of pial vessels (PVEC) obtained from one postmortem sample by perfusing 0.5% trypsin through the vessel lumen with a syringe and keeping it in place 5 minutes after clampling one end of the vessel segment. Intraluminal content was collected into culture medium (as described earlier for EC) and seeded onto 0.5% gelatin-coated tissue culture dishes. Endothelial cell colonies emerged after 4 to 5 days and reached confluence after 2 weeks. Both EC and PVEC cultures were characterized morphologically and exhibited high activities of the enzymes specific for the blood-brain barrier: γ—glutamyltranspeptidase and alkaline phosphatase. They were highly pure (more than 95%), as defined by immunostaining for angiotensin II-converting enzyme and factor VIII–related antibodies, and by incorporation of fluorescently labeled acetylated low-density lipoproteins.
Human brain astrocytes (AST, n = 5, passages 2 through 5) were isolated from fetal human brains (10 to 18 weeks old) aborted for medical indications with the approval from the Institutional Research Ethics Committee. The cultures were provided by Dr. J. Antel from the Montreal Neurological Institute. Brains were trypsinized (0.25% trypsin), homogenized, and filtered through a 130-μm mesh. Cell pellets were resuspended in a feeding medium containing 95% Dulbecco's modified Eagle's medium and 5% fetal bovine serum, and then plated onto poly-l-lysine-coated dishes. Contaminating neurons were eliminated by trypsinization. More than 95% of these cells were immunopositive for the glial fibrillary acidic protein.
Reverse transcriptase-polymerase chain reaction
Total RNA was isolated from tissues and cells with TRIzol Reagent (Gibco BRL, Gaithersburg, MD, U.S.A.) (see Abounader et al., 1999 for details) and residual genomic DNA removed with RQ1-DNase (Promega, Madison, WI, U.S.A.) for 30 minutes at 37°C. The RNA then was primed with random hexamers (200 ng/mg RNA, Gibco BRL, U.S.A.) and reverse-transcribed with 1 to 3 U of avian myeloblastosis virus reverse transcriptase (AMV RT, Promega) in a final volume of 50 μL. The cDNA was amplified with primers selective for each human receptor and designated as follows: A-CGRP1 for the calcitonin receptor-like receptor cloned in human tissue by Aiyar et al. (1996); K-CGRP1 for the canine orphan receptor RDC-1 cloned by Kapas and Clark (1995), and ADM-R for the ADM receptor sequence taken from Hänze et al. (1997). The same cDNA samples prepared from each tissue or cell type were used in parallel for polymerase chain reaction (PCR) amplification of the three receptor genes, thus allowing for internal control of the RNA preparations. The oligonucleotides were designed using the NBI OLIGO 5.0 program and were as follows: A-CGRP1 (ACC #L76380):5′-CAA GAC CCC ATT CAA CAA G-3′ (upper primer, base position 714) and 5′-ACA GCC CAT CAG GTA AAG AT-3′ (lower primer, base position 1210), yielding a 516-base pair (bp) product; K-CGRP1(ACC #X14048):5′-CAG GCA ACT TCT CGG ACA TC (upper primer, base position 32) and 5′ -CCG GCA GTA GGT CTC ATT GT-3′ (lower primer, base position 572), yielding a 560-bp product; and ADM-R (ACC #Y13583):5′-CAG TGT CGA CCG CTA TGT CA-3′ (upper primer, base position 535) and 5′-CGG AAG TGT GGG CTG AGA A-3′ (lower primer, base position 1059), yielding a 543-bp product. The reaction mixture contained 1 to 3 μL of cDNA samples, 0.2 mmol/L of each deoxinucleotide triphosphate, 5% of dimethylsulfoxide, 2 to 4 mmol/L MgCl2, 0.5 mmol/L of each primer, and 1 to 2 U of Taq DNA polymerase (Promega) in a buffer containing 50 mmol/L KCl, 10 mmol/L Tris HCl, and 0.1 % Triton X-100 for a total volume of 50 μL. The PCR amplification conditions were as follows: hot start at 94°C for 5 minutes, denaturation at 94°C for 40 seconds, annealing at 56°C for 40 seconds, and extension at 72°C for 60 seconds. After 40 cycles in these conditions, a final extension at 72°C for 5 minutes was performed.
In each experiment, a tissue known to express the appropriate receptor was run in parallel as a positive control. The PCR products were electrophoretically separated on a 1.2% agarose gel in Tris/borate/ethylenediamine tetraacetic acid containing 0.5 mg/mL ethidium bromide (ICN Biomedicals, Costa Mesa, U.S.A.) and a 100-bp DNA ladder to confirm the molecular size of the products. For a similar amount of control tissue cDNA samples, the A-CGRP1 and K-CGRP1 yielded PCR products of comparable intensity, as evaluated on the agarose gel. Control reactions lacking the reverse transcriptase enzyme were run in parallel for all samples to monitor for potential DNA contamination of the RNA samples. Representative PCR products from EC (A-CGRP1 and K-CGRP1 receptors) and AST (ADM-R) were purified using the QIAquick PCR Purification Kit (Qiagen, Germany) and subjected to automatic fluorescence sequencing by the Sanger method (W.N. Keck Foundation Biotechnology Resource Laboratory, Yale University, New Haven, CT). Their sequences were checked with BLAST program of the NCBI database to reveal the percent homology with the corresponding fragments of the published cloned human receptor genes (Aiyar et al., 1996; Sreedharan et al., 1991; Hänze et al., 1997).
cAMP production
The effect of α-CGRP (human α-CGRP; Boehringer Ingelheim PharmaKG, Biberach, Germany) and ADM13–52 (human-ADM13–52; Peninsula Labs., Belmont, CA, U.S.A. or Dr. S. St.-Pierre, UQAM, Montréal, QC, Canada) in the production of cAMP was assayed in SMC, EC, and AST grown in 24-well tissue culture plates. The cells were incubated (10 minutes) with increasing concentrations (0.001 to 1 μmol/L) of hα-CGRP or a single concentration (1 μmol/L) of h-ADM13–52 in phosphate-buffered saline containing 0.2% bovine serum albumin and 1 mmol/L 3-isobutyl-1-methyl-xanthine. Activation of adenylate cyclase by 1 μmol/L forskolin (10 minutes) was used as a control. Coupling of α-CGRP and ADM13–52 to their corresponding receptor was evaluated by preincubating (10 minutes) the cells with either 0.1 to 10 μmol/L of the CGRP1 receptor antagonist CGRP8–37, (human α-CGRP8–37, Peninsula Labs.) or 1 to 50 μmol/L of the ADM receptor antagonist ADM22–52, (human ADM22–52, Dr. S. St.-Pierre, UQAM, Montréal, QC, Canada) before addition of either 50 nmol/L hα-CGRP or 1 μmol/L ADM13–52. The PVEC cultures were similarly incubated with 50 nmol/L α-CGRP in the absence or presence of 0.1 to 10 μmol/L CGRP13–52. Levels of cAMP were determined with a commercial enzyme immunoassay kit (Biotrak, Amersham, Buckinghamshire, England). The cell pellets were dissolved in 0.1 N NaOH, and protein content was measured as before (Stanimirovic et al., 1996). The cAMP levels were expressed as a function of protein content in cell extracts. Values are expressed as mean ± SD. One-way analysis of variance was used followed by a Newman-Keuls multiple comparison test. A P < 0.05 was considered significant.
RESULTS
Expression of CGRP1 and ADM receptors
The PCR products for A-CGRP1, K-CGRP1, and ADM receptors were consistently amplified in the control tissues (data not shown). Contamination with genomic DNA was excluded from all RNA preparations, since no PCR product could be amplified in samples that were not reverse transcribed (−RT in Figs. 1 and 2). Without exception, all preparations (n = 5) of human trigeminal ganglia were found to express PCR products corresponding in size with A-CGRP1, K-CGRP1, and ADM receptors, with bands being consistently of similar intensity for the three receptors (Fig. 1).
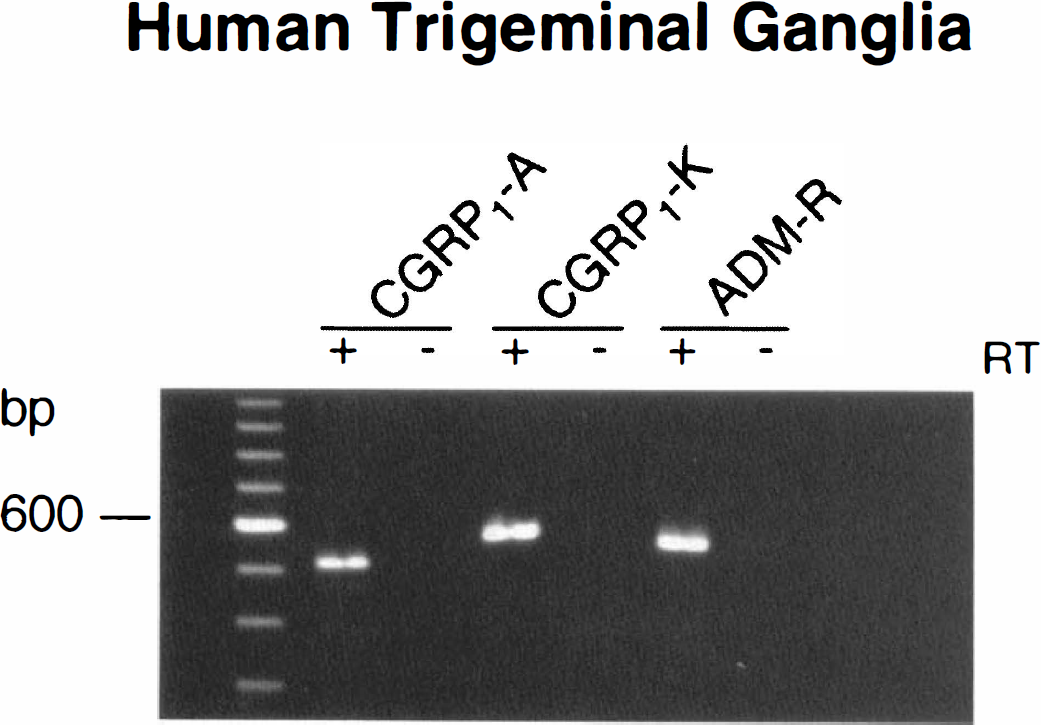
Expression of A-CGRP1, K-CGRP1, and adrenomedullin (ADM-R) receptor messages in human trigeminal ganglia as visualized by gel electrophoresis of the polymerase chain reaction (PCR) products obtained using selective oligonucleotide primers. All five ganglia showed similar results. Samples either were (+) or were not (−) submitted to reverse transcriptase (RT) treatment before PCR amplification to monitor for DNA contamination.
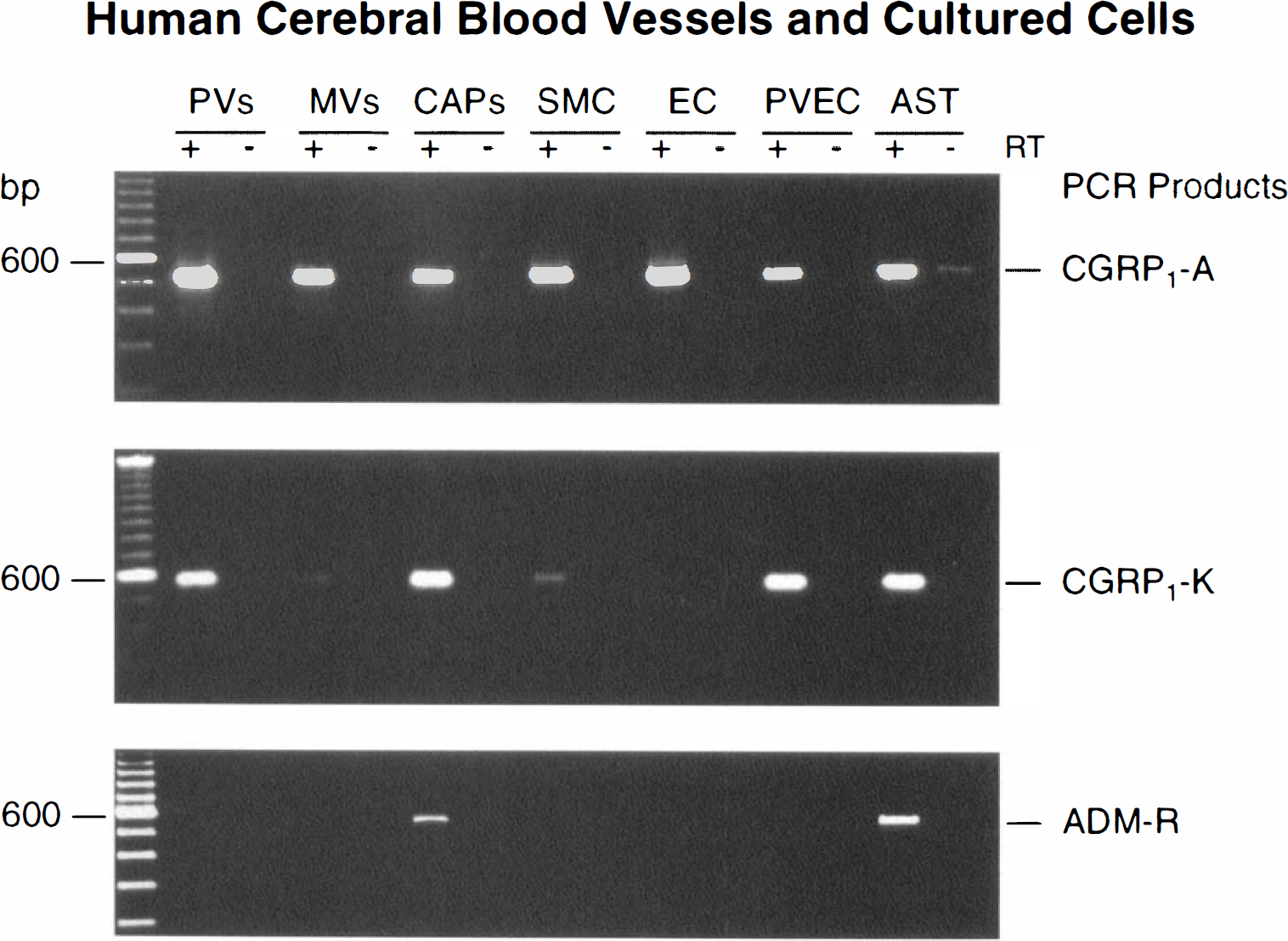
Gel electrophoresis of representative PCR products for the A-CGRP1, K-CGRP1, and adrenomedullin (ADM-R) receptors obtained from human pial vessels (PV), isolated cortical microvessels (MV), and capillaries (CAP), cultures of human brain microvascular smooth muscle (SMC) and endothelial (EC) cells, as well as in endothelial cells derived from pial vessels (PV) and fetal human brain astrocytes (AST). A similar expression pattern was obtained in all tissues and cells tested (n = 5 in each case). The presence (+) or absence (−) of reverse transcriptase (RT) treatment was used to monitor for genomic contamination.
The PCR products of the expected size for A-CGRP1 and K-CCRP1 receptors also were systematically detected by gel electrophoresis in all PV, isolated MV, and CAP, as well as microvascular and astroglial cell cultures (Fig. 2). Reproducibly, the bands of the PCR products were of comparable intensity for the two CGRP1 receptor genes in PV, CAP, PVEC, and AST, but they were of much lower intensity for the K-CCRP1 compared with th6 A-CCRP1 receptor in all preparations of MV and brain microvascular SMC and EC cultures (Fig. 2). In contrast, ADM-R mRNA was consistently detected only in isolated CAP and AST cultures, with none of the other vascular tissues or cells expressing message for this receptor (Fig. 2). Sequence analysis of selected PCR products confirmed their identity with a 99% (K-CGRP1 and ADM receptors) and 98% (A-CGRP1 receptor) homology with the published sequences of the corresponding human receptor genes.
cAMP production
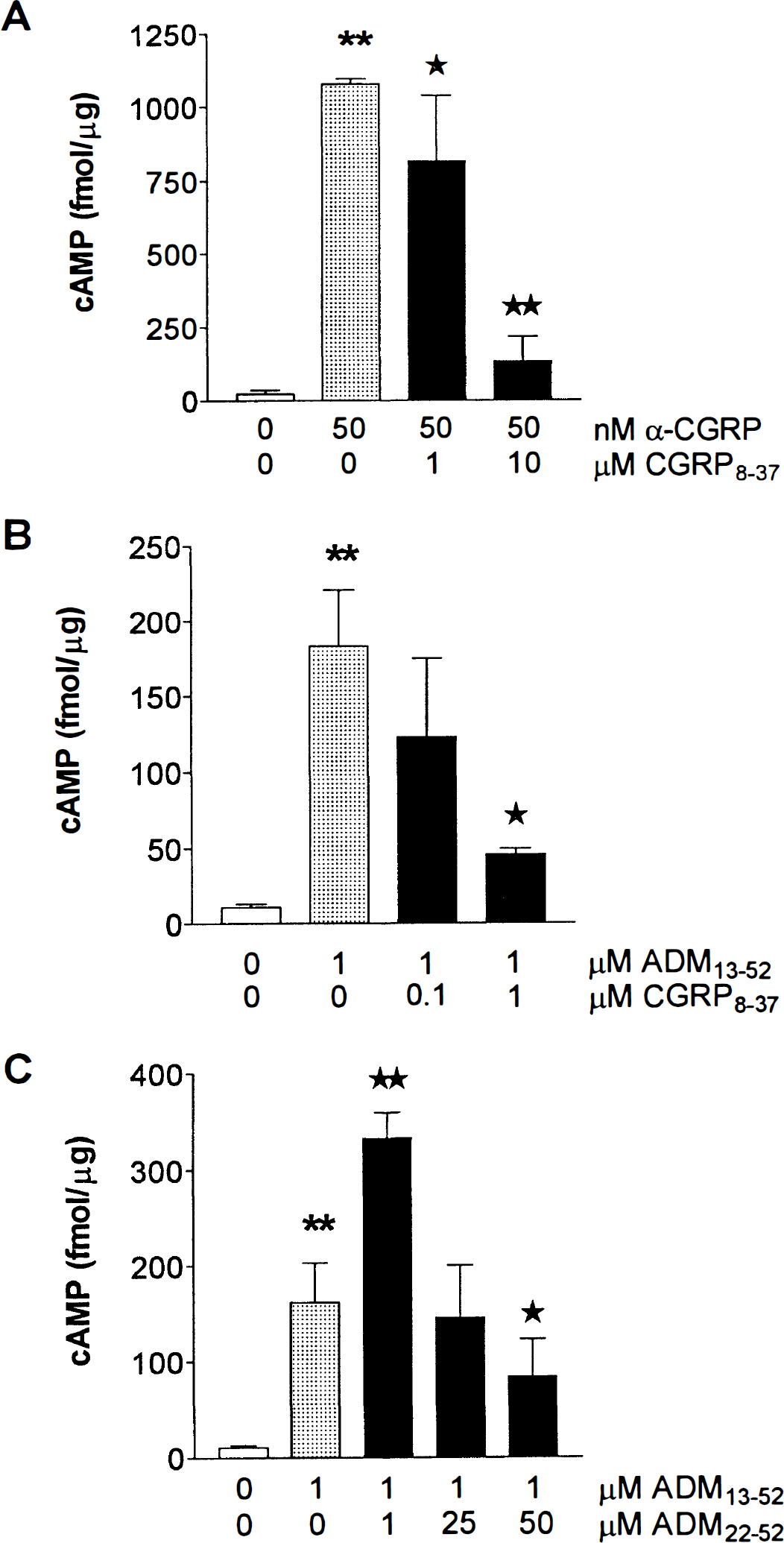
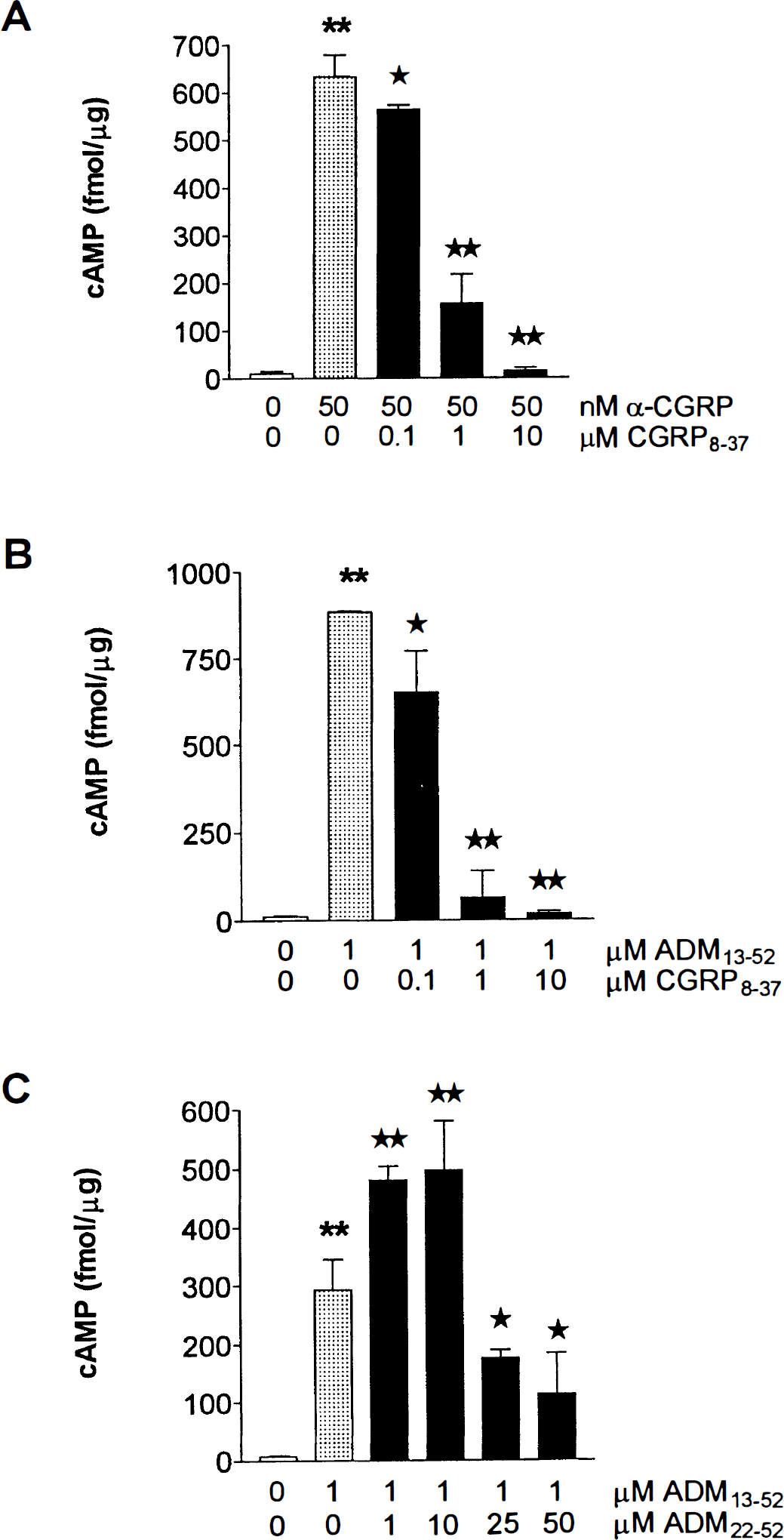
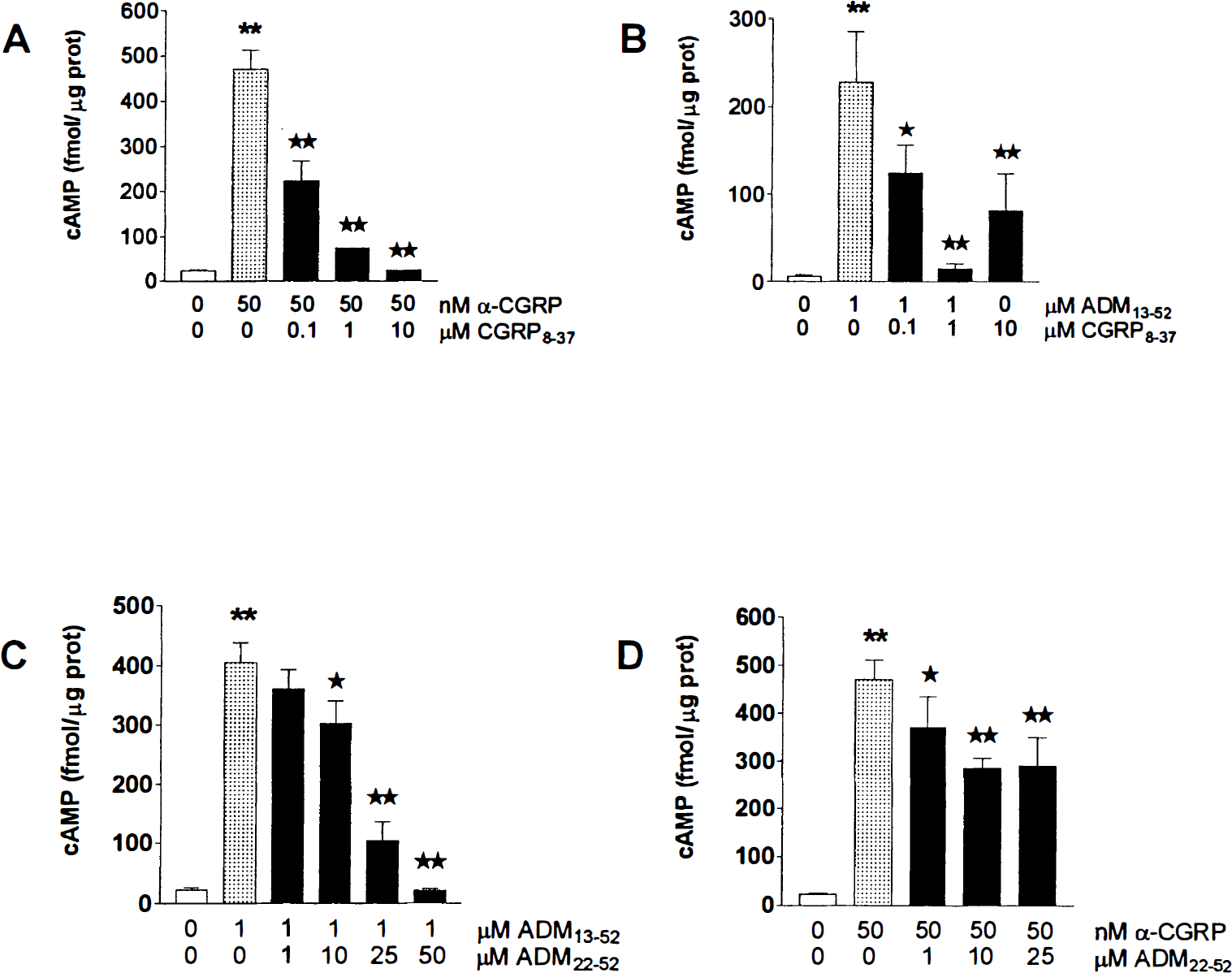
Basal levels of cAMP were similar in all cells studied, irrespective of their vascular or astroglial origin, with values ranging between 6 and 19 fmol/μg protein. In all cultures, α-CGRP elicited a dose-dependent production of cAMP, with a maximum effect at 50 nmol/L (10- to 50-fold above basal levels, P < 0.001) (Figs. 3 and 4A through 6A). This response generally was greater (twofold to threefold, P < 0.05) than that obtained with 1 μmol/L forskolin (data not shown). The α-CGRP-induced cAMP accumulation was partially inhibited by 0.1 μmol/L CGRP8–37 in both EC (11% inhibition from 633 ± 45 fmol/μg protein, P < 0.05) and AST (52% inhibition from 466 ± 57 fmol/μg protein, P < 0.001) (Figs. 3, 5A, and 6A), and it was more potently antagonized by 1 μmol/L CGRP8–37 in all cells (50% in PVEC, P < 0.01; 24% in SMC, P < 0.05; 76% in EC, P < 0.001; and 84% in AST, P < 0.001). The inhibition was almost complete at 10 μmol/L CGRP8–37 in all cell types (Figs. 3 and 4A, 5A, and 6A).
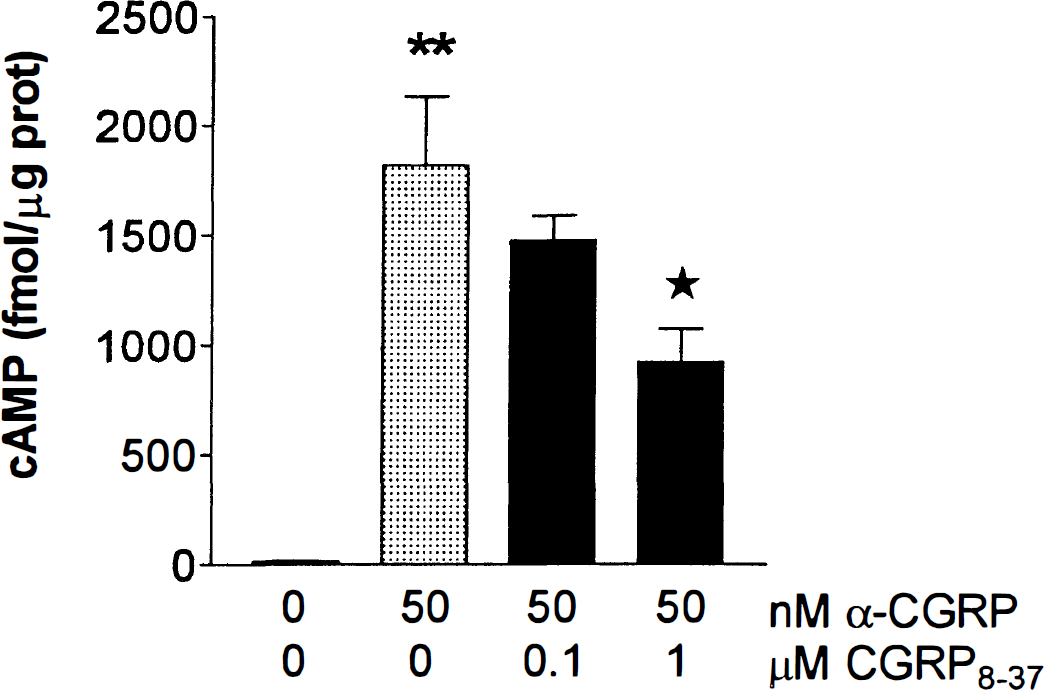
Effects of α-CGRP on cAMP formation in human pial vessel endothelial cell cultures in the presence and absence of the CGRP1 receptor antagonist CGRP8–37 (0.1 and 1 μmol/L). **P < 0.001 when comparing cAMP levels before and after incubation with 50 nmol/L α-CGRP and *P < 0.05 when comparing cAMP levels between cells treated with α-CGRP alone and those preincubated with 1 μmol/L CGRP8–37, as determined by analysis of variance (ANOVA) followed by a Newman-Keuls multiple comparison test. Bars are means ± SD.
Effects of α-CGRP
Stimulating effect of α-CGRP
Increased cAMP production in cultures of human brain astrocytes by α-CGRP
The ADM13–52 (1 μmol/L) also elicited a strong increase (20- to 50-fold, P < 0.01) in cAMP production in all cultured cells (Figs. 4B, 5B, and 6B). Similar to what was observed with α-CGRP, this effect was partially antagonized by 0.1 μmol/L CGRP8–37 in EC (27% inhibition from 885 ± 0.28 fmol/μg protein, P < 0.01) and AST (52% inhibition from 228 ± 57 fmol/μg protein, P < 0.001), whereas it was potently antagonized in all cell types at higher concentrations of CGRP8–37 (Figs. 4B, 5B, and 6B). In contrast, the ADM receptor antagonist ADM22–52 dose-dependently inhibited the ADM-induced cAMP formation only in AST (Fig. 6C). At low concentrations (1 to 10 μmol/L), this antagonist exerted a significant stimulatory effect of the response to ADM in both SMC and EC cultures (Figs. 4C and 5C). At 50 μmol/L, ADM22–52 significantly inhibited the ADM13–52-mediated effect in SMC and EC (45% inhibition in these two cell types, from 161 ± 42 and 293 ± 52 fmol/μg protein, respectively, P < 0.05), but this inhibition was much less potent than that in AST (95%, P < 0.01) (Figs. 4C, 5C, and 6C). In AST, 25 μmol/L ADM22–52 potently inhibited (75% inhibition from 405 ± 33 fmol/μg protein, P < 0.001) the increase in cAMP production elicited by ADM13–52, whereas it only partially antagonized (38%, P < 0.01) that induced by α-CGRP (Figs. 6C and 6D). Incubation of the cells with either ADM22–52 (1 μmol/L) and α-CGRP8–37 (0.1 and 1 μmol/L) alone resulted in a small but significant (P < 0.05) agonistic effect on adenylate cyclase activity, increasing cAMP levels about twofold with respect to baseline (data not shown).
DISCUSSION
The current results indicate that human extracerebral and intraparenchymal blood vessels express messages for the two cloned CGRP receptors, which exhibit a CGRP type 1 receptor pharmacology when transfected in cells (Kapas and Clark, 1995; Aiyar et al., 1996). Further, the data show that the A-CGRP1 mRNA receptor is ubiquitously expressed with high intensity PCR products in all vascular tissues, trigeminal ganglia, and astroglial cells, whereas PCR products for the K-CGRP1 receptor were expressed at comparatively lower intensity in the same microvascular tissues and cells, but not in pial vessels and trigeminal ganglia, These findings suggest that the difference in expression arises from the tissues and not from the performance of the primers or quality of the RNA extracts. Together, these observations may suggest a predominant expression of the A-CGRP1 receptor in the human cerebromicrovascular bed. Moreover, the findings indicate that human brain AST but not micro-vascular tissue and cells express functional ADM-R. A clearer definition of the respective roles of these putative receptors in the human microcirculation and astroglial cells will be possible only when better pharmacologic tools and more information on these clones become available.
Our finding of A-CGRP1 and, to a lesser extent, K-CGRP1 receptor messages in cerebromicrovascular SMC cultures and the ability of CGRP to elicit cAMP production in these cells is fully compatible with its reported ability in the rat to dilate isolated intraparenchymal arterioles (Edwards et al., 1991; Fergus et al., 1995) and in vivo to induce a long-lasting increase in local CBF (Suzuki et al., 1989; Kobari et al 1995). It is thus possible that CGRP may exert similar effects in the human brain microcirculation. In human pial vessels, the CGRP receptor mediating dilation seems to be located in the smooth muscle layer, as evidenced by the capacity of CGRP to relax endothelium-denuded cerebral arteries (Jansen-Olensen et al., 1996) and the expression of A-CGRP1 mRNA in similarly prepared vessels (Edvinsson et al., 1997). Based on these observations, a novel and unexpected finding from the current study was the expression of the two human CGRP1 receptor genes in human EC obtained from both pial and intracortical vessels, and the prominent CGRP8–37 sensitive CGRP-induced cAMP production in these cells. The strong expression of both A-CGRP1 and K-CGRP1 receptor genes in pial vessel EC, although only the A-CGRP1 receptor was expressed at high intensity in microvascular EC cultures, may suggest that these receptors are differentially distributed in extraparenchymal and intraparenchymal EC. Although additional work is needed to determine their respective roles in brain EC from various origins, preliminary results from our laboratory suggest that CGRP may alter permeability of capillary EC (Moreno et al., 1999).
The existence of functional CGRP1 receptors in human brain AST, as shown here, together with the clear expression of both A-CGRP1 and K-CGRP1 receptor mRNA in these cells, concurs with previous studies, albeit in rodents, supporting the presence of astroglial CGRP receptors (Yeung et al., 1996). In view of the suggested importance of these cells in the regulation of brain metabolic and ionic homeostasis (Tsacopoulos and Magistretti, 1996), local CBF (Alkayed et al., 1997), and blood-brain barrier function (Tao-Cheng et al., 1987), CGRP most likely has the ability to modulate some of these various astrogial functions. The possibility also exists that CGRP can exert differential effects through the two CGRP1 receptors found in these cells, although further work is required to clarify this issue.
In contrast to the widespread distribution of CGRP1 receptors in cerebrovascular and astrogial cells, ADM receptor mRNA was detected only in isolated CAP and AST cultures, suggesting that its expression in CAP most likely results from the attached astrogial cells (see Linville and Hamel, 1995; Abounader et al., 1999). Such a localization agrees with previous studies that showed the presence of specific binding sites for ADM together with its positive coupling to adenylate cyclase in mouse and rat AST (Yeung et al., 1996). In this regard, notice that ADM mRNA and peptide are overexpressed after focal cerebral ischemia (Wang et al., 1995), a situation in which astroglial cells may become activated, possibly to contribute to the spontaneous recovery of ischemic neurons (Zoli et al., 1997). Our data showing that ADM22–52 more effectively inhibited the ADM13–52-mediated increase in cAMP production in AST than in vascular cells, where this response was either potentiated (1 to 10 μmol/L) or partially inhibited (50 μmol/L), provide convincing evidence for functional ADM receptors in human brain AST. This also is supported by the fact that ADM22–52 only partially (40%) blocked the CGRP-mediated cAMP production in AST, an observation suggesting that no more than 40% of the inhibition exerted by ADM22–52 in these cells can be ascribed to its documented interaction with CGRP receptors (Champion et al., 1997).
No evidence could be obtained in the current study to suggest the expression of functional ADM receptors in SMC and EC cultures, or in any other vascular preparations if we take into consideration the contaminating AST in the isolated CAP, as discussed earlier. This conclusion may be at variance from that obtained in cultured EC of human umbilical vein (Kato et al., 1995), but it is further supported by the ability of the CGRP1 receptor antagonist CGRP8–37 to block the ADM-mediated cAMP increase in brain EC at concentrations (0.1 to 1 μmol/L) known to act selectively at CGRP1 receptors (Quirion et al, 1992). Moreover, in these cells and in SMC, ADM22–52 exerted comparatively weaker antagonistic properties than CGRP8–37 on the ADM13-s2-mediated cAMP response, and this at concentrations 50-fold higher than those of CGRP8–37, at which interaction with CGRP1 receptors could be taking place (Champion et al., 1997). Together, these molecular and pharmacologic findings allow us to conclude that the vasodilator effect of ADM is most likely mediated by CGRP1 receptors for which it has a relatively high affinity (EC50 values of 10−7 mol/L for ADM compared with 3 × 10−9 mol/L for CGRP [see Kapas and Clark, 1995]). In this respect, notice that the human calcitonin-receptor-like receptor, which was cloned and identified by Aiyar et al. (1996) as a CGRP, receptor and referred to as A-CGRP1 receptors in the current study, seems to function either as a CGRP1 or an ADM receptor, depending on which member of a new family of single-transmembrane domain proteins (called receptor activity-modifying proteins [RAMPs]) is coexpressed (McLatchie et al., 1998). It thus would be interesting to identify which receptor activity-modifying protein is present in human cerebrovascular and astroglial cells and if these could explain the distinct pharmacologic behavior observed in these cells for the ADM and CGRP compounds.
Our results show expression of ADM and of the two cloned CGRP1 receptors in human trigeminal ganglia, observations that are supported, at least in part, by a recent report on A-CGRP1 receptor mRNA in this tissue (Edvinsson et al., 1997). Presynaptic CGRP, receptors that regulate CGRP release from perivascular nerves are present in rat mesenteric resistance vessels (Nuki et al., 1994), and a similar regulation could operate at the level of CGRP-containing trigeminovascular afferents. Interestingly, CGRP is released from these sensory nerves in migraine concomitantly with the development of head pain, and both phenomena are normalized on acute treatment with the 5-HT1B/1D receptor agonist sumatriptan (Goadsby and Edvinsson, 1993). The clinical efficacy of sumatriptan is believed to result from its ability to presynaptically block the release (Moskowitz, 1992) or the synthesis (Durham et al, 1997) of CGRP. The possibility of acting directly on presynaptic CGRP1 receptors, as suggested by the presence of message for the two CGRP type 1 receptors in human trigeminal ganglion neurons, may offer a novel approach to counteract the CGRP-mediated vasodilation and neurogenic inflammation associated with migraine headache. Alternatively, inhibition of the postsynaptic smooth muscle CGRP1 receptor-mediating dilation could represent an interesting approach to block the vascular symptoms of an acute migraine attack (Moskowitz, 1992).
In addition to clarifying the distribution and cellular localization of the ADM and the two cloned CGRP1 receptors in the human cerebrovascular bed and astroglial cells, the current results raise further questions regarding their respective roles in EC and SMC and how CGRP derivatives could interact at the vascular and neuronal sites to modulate the development or manifestation of head pain. Further, they underscore the urgent need to better correlate the molecular and functional aspects of the two cloned Numan CGRP1 receptors.
Footnotes
Acknowledgments:
The authors thank Drs. A. Olivier and J. Antel, Montreal Neurological Institute, McGill University, for their precious collaboration for obtaining human brain tissue and astroglial cell cultures. The authors also thank Ms. Rita Ball for skillful technical assistance, and Ms. Linda Michel for preparing the manuscript.
