Abstract
Physiologic and anatomic evidence suggest that 5-hydroxytryptamine (5-HT) neurons regulate local cerebral blood flow and blood-brain barrier permeability. To evaluate the possibility that some of these effects occur directly on the blood vessels, molecular and/or pharmacologic approaches were used to assess the presence of 5-HT receptors in human brain microvascular fractions, endothelial and smooth muscle cell cultures, as well as in astroglial cells which intimately associate with intraparenchymal blood vessels. Isolated microvessels and' capillaries consistently expressed messages for the h5-HT1B, h5-HT1D, 5-HT1F, 5-HT2A but not 5-HT7 receptors. When their distribution within the vessel wall was studied in more detail, it was found that capillary endothelial cells exhibited mRNA for the h5-HT1D and for the 5-HT7 receptors whereas microvascular smooth muscle cells, in addition to h5-HT1D and 5-HT7, also showed polymerase chain reaction products for h5-HT1B receptors. Expression of 5-HT1F and 5-HT2A receptor mRNAs was never detected in any of the microvascular cell cultures. In contrast, messages for all 5-HT receptors tested were detected in human brain astrocytes with a predominance of the 5-HT2A and 5-HT7 subtypes. In all cultures, sumatriptan inhibited (35–58%, P < .05) the forskolin-stimulated production of cyclic AMP, an effect blocked by the 5-HT1B/1d receptor antagonists GR127935 and GR55562. In contrast, 5-carboxamidotryptamine induced strong increases (≥ 400%, P < .005) in basal cyclic AMP levels that were abolished by mesulergine, a nonselective 5-HT7 receptor antagonist. Only astroglial cells showed a ketanserin-sensitive increase (177%, P < .05) in IP3 formation when exposed to 5-HT. These results show that specific populations of functional 5-HT receptors are differentially distributed within the various cellular compartments of the human cortical microvascular bed, and that human brain astroglial cells are endowed with multiple 5-HT receptors. These findings emphasize the complex interactions between brain serotonergic pathways and non-neuronal cells within the CNS and, further, they raise the possibility that some of these receptors may be activated by antimigraine compounds such as brain penetrant triptan derivatives.
Keywords
A large body of physiologic evidence suggests that brainstem serotonin (5-hydroxytryptamine, 5-HT) neurons can regulate local cerebral blood flow (CBF) and possibly blood-brain barrier (BBB) permeability (Cohen et al., 1996). The predominant vascular response to 5-HT is a reduction in local CBF (i.e., cerebral vasoconstriction), although increases in flow (i.e., vasodilatation) have also been observed (Bonvento and Lacombe, 1993; Cohen et al., 1996). This dual effect has been related to the initial tone of the blood vessels (Rosenblum and Nelson, 1990) as well as to the ability of different regions within the dorsal raphe nucleus to trigger opposite vasomotor responses (Underwood et al., 1992). However, it cannot be excluded that different populations of cerebrovascular 5-HT receptors which elicit opposite vasomotor responses are responsible for these two distinct effects. In support of these observations, ultrastructural studies have documented close associations between 5-HT nerve terminals and intracortical penetrating arteries, microarterioles, and capillaries in the frontoparietal cortex (Cohen et al., 1995b), a region in which manipulations of 5-HT neurons result in significant CBF changes (Bonvento et al., 1997). Taken together, these observations suggest that neurally released 5-HT can exert effects on the microcirculation through interactions with specific 5-HT receptors located on the blood vessel wall.
However, despite these proposed vascular effects in response to central changes in 5-HT neurotransmission, virtually no information is available on the nature and location of 5-HT receptors in the cerebral microvascular bed. Early radioligand binding studies performed on isolated intracortical microvessels from human brains suggested the presence of 5-HT1 binding sites (O'Neill et al., 1988) while addition of 5-HT to human brain endothelial cells in culture was found to stimulate the production of cyclic AMP (cAMP) (Bacic et al., 1991), an effect now attributed primarily to activation of 5-HT6 or 5-HT7 receptor subtypes (Hoyer et al., 1994, Martin, 1994). In addition, these studies could not discriminate which subtypes of 5-HT receptors were involved in these responses due to the lack of selective pharmacologic agents. With the advances in molecular cloning techniques and the development of novel pharmacologic tools, several new 5-HT receptors and their pharmacologic profiles have been identified (Hoyer et al., 1994). Some of these receptors are likely to be involved in the modulation of cerebral vascular functions. For instance, 5-HT1B, 5-HT 1D, 5-HT2A, and 5-HT7 receptors have been found in major cerebral arteries and/or in peripheral blood vessels of various species including human, and have been implicated in either constriction or relaxation (Hamel et al., 1993; Martin, 1994; Leung et al., 1996; Terron, 1996).
The aim of the present study was to evaluate the expression of putative vascular 5-HT receptors in the human brain microvascular bed, to identify their cellular localization within the vascular and/or astroglial compartments, and to assess their ability to mediate functional responses. The results indicate that different 5-HT receptor subtypes coupled to distinct cellular signaling pathways are expressed in specific endothelial, smooth muscle and/or astroglial compartments of the human cortical microcirculation. Furthermore, the nature of 5-HT receptors identified in various cellular constituents of the cerebromicrovascular bed suggests that 5-HT is likely involved in the regulation of both cerebral microcirculation and astrocyte functions, as they pertain to local brain perfusion and BBB permeability. Some aspects of this study have previously been presented in abstract form (Cohen et al., 1995a; Cohen and Hamel, 1996).
MATERIALS AND METHODS
Tissue and cell culture preparation
Isolated microvessels and capillaries. Human brain cortices (n = 6) were obtained at autopsy from the Royal Victoria Hospital (Montréal, Canada) from men (n = 4) and women (n = 2) who died from diseases not affecting the CNS (post-mortem delay of 16 ±2 hours). Microvessel (MV) and capillary (CAP) fractions were isolated from the cerebral cortex by centrifugation in 15% dextran, and their relative purity confirmed by microscopic examination and biochemical enrichment of the endothelial marker enzymes δ-glutamyltranspeptidase and alkaline phosphatase, as previously described in detail (Linville and Hamel, 1995).
Human brain microvascular smooth muscle and endothelial cell cultures. Human brain microvascular smooth muscle (HBSM) and endothelial cell (HBEC) cultures were generated using previously described techniques (Stanimirovic et al., 1996; Abounader et al., 1999). In short, MV and CAP fractions were isolated from small samples of human temporal lobe surgically removed for the treatment of idiopathic epilepsy. Cortical homogenates were sequentially filtered through 350-µm and 112-µm mesh screens. The large MVs collected on these meshes were dislodged in cold M199 medium (Gibco BRL, Gaithersburg, MD) and used to generate HBSM cultures (see below) while the filtrates were further resuspended in 20% dextran, centrifuged, and collected on a 20-µm (CAP fraction) mesh and used to generate HBEC cultures (see below).
Microvessels from the larger meshes were washed, dissociated with type IV collagenase, and seeded into 0.5% gelatin-coated tissue culture plates, maintained in growth media containing 65% M199, 10% fetal calf serum, 5% human serum, 20% murine melanoma cell (mouse melanoma, Cloudman S91, clone M-3, melanin-producing cells)–conditioned media. The dissociated microvessels were allowed to grow in primary culture for 4 to 5 weeks, by which time the slow-proliferating, spindle-shaped smooth muscle cells largely outgrew the initial sprouting of endothelial cells. Characteristic criss-crossing and palisading of several smooth muscle cell layers were observed throughout the cultures. Routine immunostaining for the smooth muscle cell protein α-actin (Accurate Chemical and Scientific Corp, Westbury, NY) showed that about 85% of the cells were positive for α-actin while the 15% non-muscular cells incorporated acetylated-low density lipoprotein (DiI-Ac-LDL), a specific marker for endothelial cells. A total of six primary HBSM cell cultures derived from either 112-µm (n = 2) or 350-µm (n = 4) microvascular fractions were used in the present study.
Microvessels on the 20-µm mesh were collected and similarly dissociated with the type IV collagenase, seeded and maintained in the same growth medium as above with 5 µg/mL of insulin, 5 µg/mL of transferrin, 5 ng/mL of selenium, and 10 µg/mL of endothelial cell growth supplement. Endothelial cell colonies emerging from attached CAPs were removed using cloning rings and two to three of these cloned colonies were pooled and grown to confluence. Confluent cultures from six different human biopsy specimens (passages 3 to 7) were used in this study. The morphologic, biochemical, and functional properties of these HBEC cultures have been previously described at length (Stanimirovic et al., 1996; Abounader et al., 1999). Endothelial cells represented more than 95% of the culture, as estimated by factor VIII–related antigen immunocytochemistry.
Human brain astrocytes. Human brain astrocyte (HBA) cultures (n = 5) prepared as described previously (Yong et al., 1992) were generated from fetal human brains, obtained from 10- to 18-week-old fetuses aborted for medical indications with the approval from the Institutional Ethics Review Committee. Cells were pelleted after filtration of dissociated brain tissue through a 130-µm mesh, resuspended in a feeding medium containing 95% Dulbecco's modified Eagle's medium and 5% fetal bovine serum, then plated onto poly-l-lysine-coated dishes. Contaminating neuronal clumps completely disappeared after initial passaging. Essentially all cells (> 95%) in HBA cultures stained positively for glial fibrillary acidic protein and were devoid of contaminating oligodendrocytes and/or microglia. Passages 3-5 were used for the experiments in this study.
Reverse transcriptase-polymerase chain reaction
Isolated MV and CAP fractions were homogenized with a polytron (Brinkmann, Westbury, NY) while the cell cultures were lysed either by passage through a pipette in the TRIzol Reagent (Gibco BRL). Isolated total RNA was primed with random hexamers (Gibco BRL) and reverse transcribed in a 50 µL final volume containing avian myeloblastosis virus reverse transcriptase (Promega, Madison, WI). Polymerase chain reaction (PCR) amplifications, performed in 50 µL using an MJ Research thermal cycler, were performed for 35 to 40 cycles (94°C for 40 seconds, 56°C to 58°C for 40 seconds depending on receptors, and 72°C for 60 seconds) with a pre-incubation and post-incubation of 94°C for 5 minutes and 72°C for 5 minutes, respectively. PCR products were size fractionated on a 1% agarose gel in Tris/borate/ethylenediamine-tetraacetic acid containing ethidium bromide and photographed under ultraviolet light. RNA extracts from each tissue were concurrently used for amplification of all receptors studied, normally repeated three times, but occasionally repeated twice. Positive and negative controls were always run in parallel. Positive controls consisted of a tissue known to express the appropriate receptors such as human cerebral cortex for 5-HT1F, 5-HT2A and 5-HT7 receptors and caudate nucleus for h5-HT1B and h5-HT1D receptors (Hoyer et al., 1994). Negative controls, consisting of reactions without reverse transcriptase, were used to monitor for the presence of contaminating genomic DNA.
Primers. Primers for 5-HT receptors of potential interest in cerebrovascular tissue (Martin, 1994; Cohen et al., 1996) were designed using the NBI OLIGO 5.0 program based on the published nucleotide sequences. Human 5-HT1Dα and 5-HT1Dβ receptors will be referred to as h5-HT1D and h5-HT1B receptors (Hartig et al., 1996), respectively, and 5-HT1F and 5-HT7 will be treated as functional receptors in biological tissues (Eglen et al., 1997; Phebus et al., 1997). The primers were synthesized using an Applied Biosystems synthesizer and purified using an OPC column. All oligonucleotide primers were flanked with the T7 and SP6 RNA polymerase promoter sequences for use in other experiments. PCR was performed with the following sets of oligonucleotide primers: for h5-HT1B (ACC # M89478) sense, AAGCCTTCTCCTCAAGCA (base position 75), and antisense, AGGTGATGAGCGCCAATA (base position 616); for h5-HT1D (ACC # M81589) sense, CACCATCTACTC-CACCTGTG (base position 634), and antisense, CAGAAATCCTCTTGCGTTC (base position 920); for 5-HT1F (ACC # L05597) sense, CTTGAAGCCTTCTCTGAACTG (base position 139) and antisense, AGAGAT GCAAGATGGAGCAC (base position 567); for 5-HT2A (ACC # X57830) sense, GAGTGTGGGTACATCAAGG (base position 57) and antisense TAAGGAGAGACACGACGGT (base position 349); for 5-HT7 (ACC # L21195) sense, GGAACAGATCAACTACGGCAGAGT (base position 240) and antisense, GGTGGTGGCTGCTTTCTGTTCTCGCTTAAA (base position 1011). These sets of primers would be expected to generate PCR products of 595, 340, 482, 346 and 825 bp, respectively.
Sequencing analyses. Representative PCR products from HBEC (h5-HT1D receptor), HBSM (h5-HT1B and 5-HT7 receptors), and HBA (5-HT1F and 5-HT2A receptors) were treated with Klenow enzyme (Gibco BRL) and digested with the appropriate restriction enzymes. These fragments were then ligated with T4 DNA ligase (Promega) into the appropriate bacteriophage or plasmid. Competent DH5αF' (Gibco BRL) cells were transformed for preparation of single-stranded or double-stranded DNA for sequencing. Nucleotide sequence analysis was performed using the Sanger dideoxy-nucleotide chain termination method and Sequenase (U.S. Biochemical) in an automated sequencer (ALF, Pharmacia).
Second messenger assays
cAMP production. To measure 5-HT1 receptor–mediated responses on cAMP production, HBEC, HBSM, and HBA cells were incubated with forskolin (1 µmol/L, 15 minutes) either alone, or in the presence the 5-HT1 receptor agonist sumatriptan (1 µmol/L, 15 minutes), with or without the 5-HT1B/1D receptor antagonists, GR127935 and GR55562 (1 µmol/L, Skingle et al., 1996; Connor and Beattie, 1996), in phosphate buffered saline containing 0.2% bovine serum albumin (BSA) and 1 mmol/L 3-isobutyl-1-methyl-xanthine. Sumatriptan, GR127935, and GR55562 were generously provided by GlaxoWellcome Inc (Hertfordshire, UK). The 5-HT7 receptor-mediated responses were assessed by incubating (15 minutes) the cells with 5-carboxamidotryptamine (5-CT, 10 µmol/L; Research Biochemicals Inc [RBI], Natick, MA) alone or in the presence of the 5-HT receptor antagonist, mesulergine (10 to 100 µmol/L; RBI), both compounds exhibiting high affinity at the cloned 5-HT7 receptor (Bard et al., 1993). The cells were incubated with antagonists for 10 minutes (37°C) before the addition of agonists. The reaction was stopped by removal of the reaction mixture and addition of 65% (v/v) ice-cold ethanol. The ethanol extraction was repeated twice, the extracts combined, dried (vacuum oven at 80°C), and dissolved in 200 µL of the assay buffer for determination of cAMP content using a cAMP enzyme-immunoassay kit (Biotrak, Amersham). The cell pellets were dissolved in 0.1N NaOH and protein content was measured using BSA as the standard (Lowry et al., 1951).
Inositol phosphate formation. Coupling of 5-HT2 receptors to inositol phosphate (IP) formation was determined according to the protocol originally described by Berridge et al (1982). Briefly, confluent HBEC, HBSM, and HBA were pre-labeled with [3H]myo-inositol (2.5 µCi/mL; New England Nuclear, Boston, MA) for 16 to 18 hours in serum- and inositol-free M199. Unbound 33H]myo-inositol was removed by washing in M199. The cells were pre-incubated or not with the 5-HT2A/2C receptor antagonist ketanserin (100 µmol/L, 10 minutes) and then exposed to 5-HT (100 µmol/L, 15 minutes) in the presence of 20 mmol/L LiCl. Histamine (100 µmol/L) and endothelin-1 (100 nmol/L) were used as positive controls as they stimulate IP3 production in HBEC. The reaction was stopped by replacing the medium with cold 0.3-mol/L trichloroacetic acid and the cells were scraped, briefly sonicated, and sedimented by centrifugation. Aliquots of the supernatants were treated twice with anhydrous diethyl ether, the aqueous phases were separated by centrifugation, and the remaining ether was evaporated under nitrogen. The samples were neutralized with 6.25 mmol/L sodium tetraborate, applied to a 1-mL DOWEX-AG 1×8-formate form anion exchange column and the IP fractions were eluted from the columns and quantified, as previously described (Stanimirovic et al., 1996). The IP3 fraction determined by this technique represents a mixture of IP3 isomers. Protein content of the cellular pellets was measured as above.
Statistical analyses. The effects of the various treatments on cell cultures were assessed by a one-way analysis of variance and followed by Schéffé's post hoc test for multiple comparisons using the STATISTICA program. Values of P < .05 were considered statistically significant.
RESULTS
Expression of 5-HT receptors
Isolated MVs and CAPs. Specific signals for the h5-HT1B and h5-HT1D receptors were consistently detected by gel electrophoresis in both isolated MV and CAP fractions (Fig. 1; Table 1). Similarly, PCR products of the expected size for the 5-HT2A receptor were detected in a majority of both fractions (Fig. 1; Table 1). Message for 5-HT1F receptors was found in some MV and more frequently in the CAP fraction (Fig. 1; Table 1). No mRNA transcripts for the 5-HT7 receptor were detected by gel electrophoresis in any of the microvascular fractions (Fig. 1; Table 1). In all preparations, the intensity of the PCR products generated from vascular tissues was generally lower than that found in their respective control brain tissues (not shown).
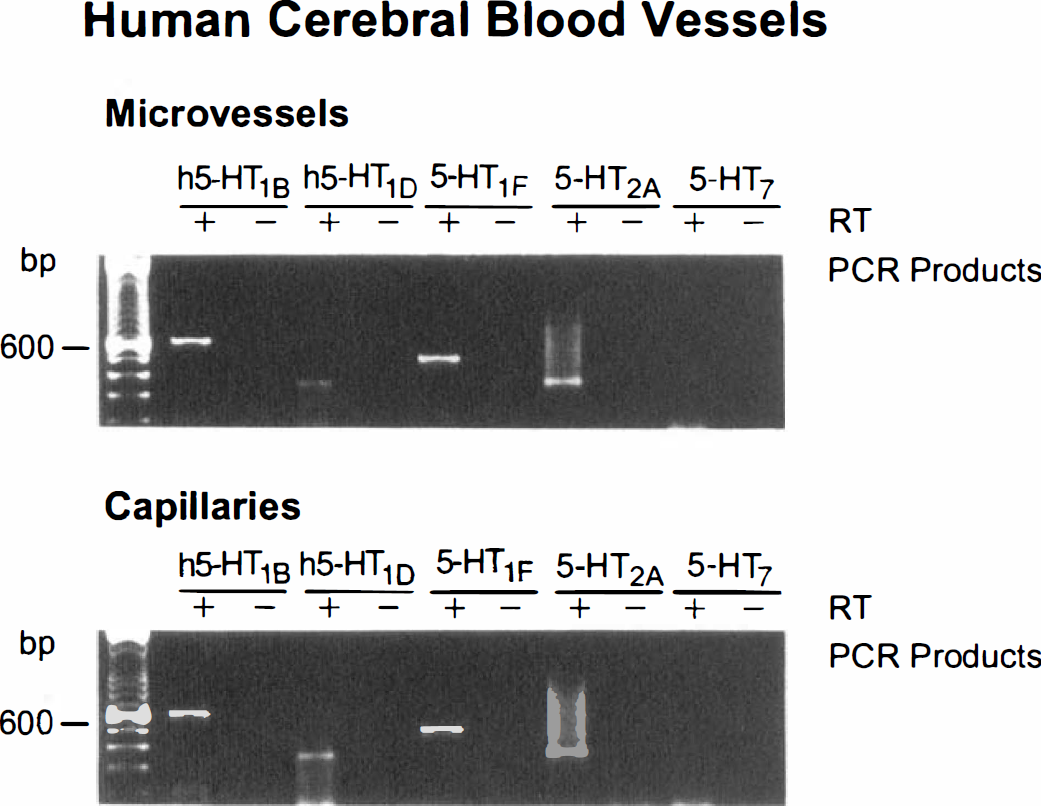
Identification of 5-HT receptors in isolated human brain MVs and CAPs by RT-PCR. Agarose gel electrophoresis of PCR-amplified cDNA from representative tissues using specific oligonucleotide primers for h5-HT1B, h5-HT1D, 5-HT1F, 5-HT2A, and 5-HT7 receptors. To control for genomic DNA and/or PCR contamination, amplification was performed with or without reverse transcriptase enzyme (+ or – RT lanes) added in cDNA synthesis reaction (see Materials and Methods). PCR products of the appropriate size (see Materials and Methods for exact size) were observed for all except the 5-HT7 receptors.
Semiquantitative expression of mRNA for various 5-HT receptors in human isolated intracortical blood vessels and cultured vascular cells and astrocytes

Relative expression for the various 5-HT receptors in all tissues based on 1) number of preparations expressing these receptors and 2) abundance with respect to control amplification. (–) denotes absence of PCR products in all or a majority (5/6) of tissue preparations. –/+: signal barely detected but present consistently, +, ++, +++, ++++: based on intensity and expression in a majority (4 to 6) of preparations studied. CAPs, capillaries; HBA, human brain astrocytes; HBEC, human brain endothelial cells; HBSM, human brain smooth muscle cells; MVs, microvessels.
Human brain microvascular cells and astrocytes in culture. When expression of 5-HT receptors was studied in these cultured cells, specific PCR products corresponding in size to the h5-HT1D and 5-HT7 receptors were detected at varying intensities by gel electrophoresis in virtually all HBEC cultures examined (Fig. 2, Table 1). Amplified fragments for the h5-HT1B, 5-HT1F, and 5-HT2A receptor were not observed in these cells (Table 1). Strong intensity PCR products corresponding in size to the h5-HT1B and 5-HT7 receptors were evident in all HBSM cells (Fig. 3) while a specific but weak signal for the h5-HT1D receptor was detected in a majority of HBSM cell cultures (Fig. 3). However, similar to what was observed in the HBEC, messages for the 5-HT1F and 5-HT2A receptors were not detected by gel electrophoresis in any of the HBSM cell cultures (Table 1). In contrast, the HBA cultures, in addition to mRNAs corresponding to the h5-HT1B, h5-HT1D, and 5-HT7 receptors, further exhibited specific PCR products of moderate and strong intensity for the 5-HT1F and the 5-HT2A receptors (Fig. 4; Table 1). In all PCR experiments, no products were observed if amplifications were performed in cDNA samples devoid of reverse transcriptase (RT) enzyme (-lanes, in Figs. 1–4). The sequence of the PCR products obtained from tissue mRNA as template, matched unambiguously, i.e., 99.8% (115-HT1B), 98.8% (h5-HT1D), 98.6% (5-HT1F), 99.1% (5-HT2A), and 99% (5-HT7), the published sequences for their respective cloned receptors.
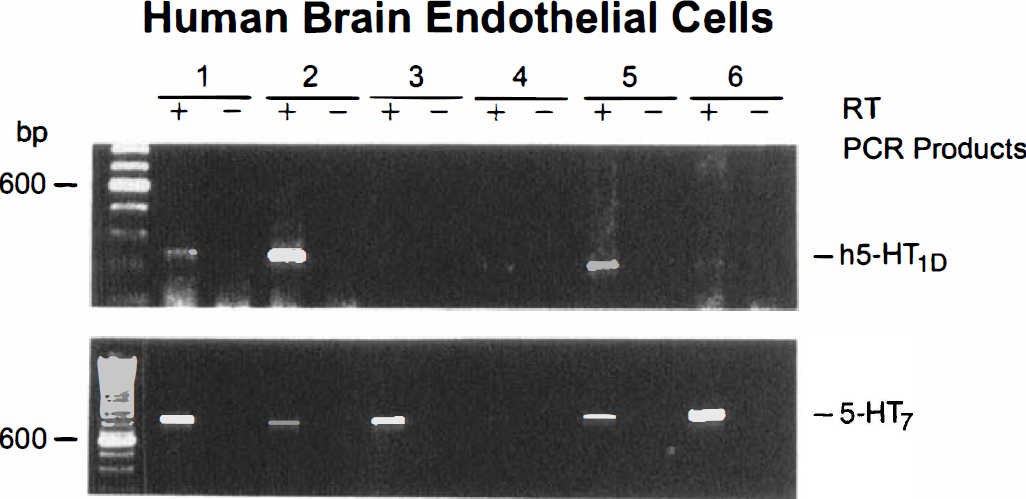
Agarose gel electrophoresis of RT-PCR products from six different cultures of HBEC. Only messages for the h5-HT1D and 5-HT7 receptors were detected by RT-PCR in these cells. The presence or absence of RT enzyme in the cDNA reaction was always used to monitor possible DNA contamination (+ and – RT on Figure).
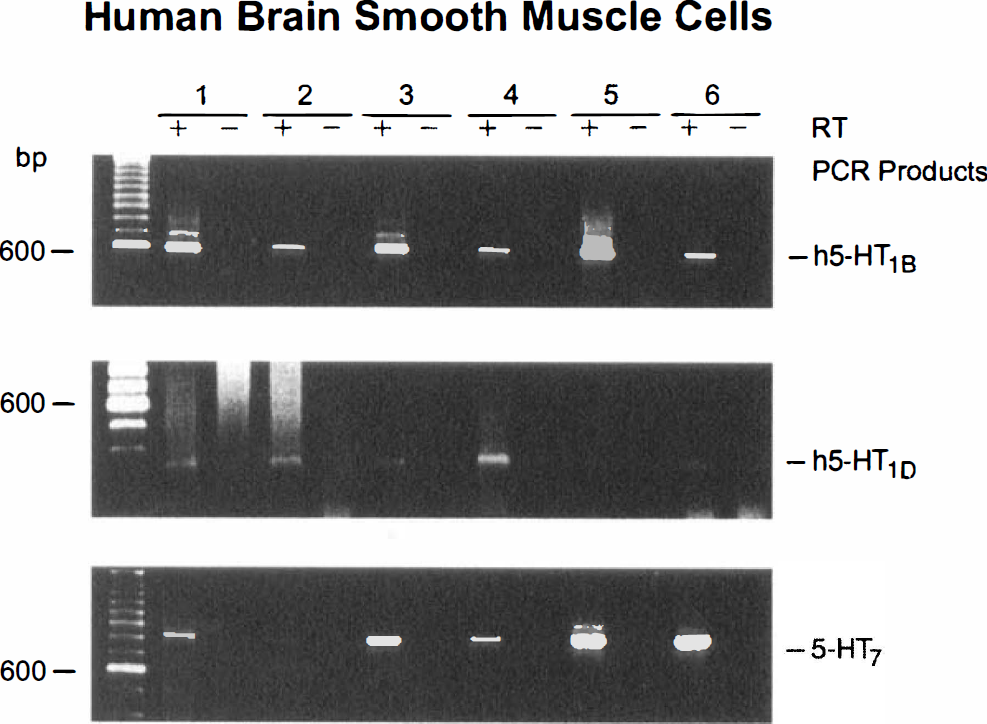
Identification of 5-HT receptors in six primary cultures of HBSM cells by RT-PCR. As seen by gel electrophoresis, h5-HT1B, h5-HT1D, and 5-HT7, but not 5-ht1F or 5-HT2A receptor messages were detected in these cells. Note the weak signal intensity for the h5-HT1D receptor. Controls for genomic DNA contaminations (+ or – RT) are indicated.
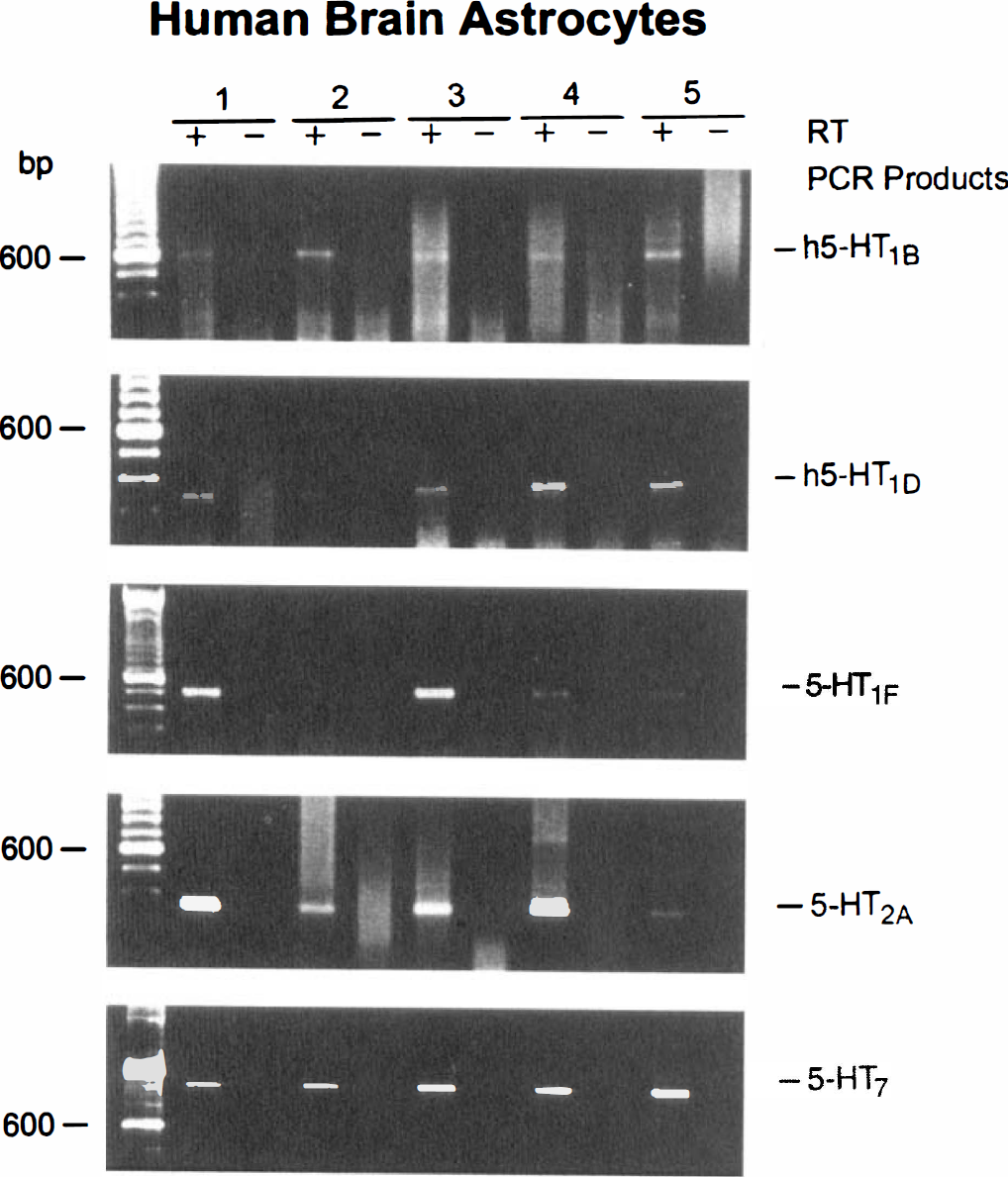
Gel electrophoresis of PCR products for the 5-HT receptors expression from five different cultures of HBA. Messages for all 5-HT receptors were detected in these cells with stronger signals for the 5-HT2A and 5-HT7 receptors. Samples with and without reverse transcriptase (+ or – RT) are shown.
Functional and pharmacologic assessment of 5-HT receptors
cAMP production. Independently of the cell type, basal levels of cAMP varied slightly from one cell preparation to another and levels normally range between 10 and 50 fmol/µg protein (26.3 ± 30.4, n = 22 different experiments). Stimulation of adenylate cyclase by 1 µmol/L forskolin resulted in a 6- to 10-fold increase in basal cAMP levels in HBEC, HBSM, and HBA. Sumatriptan effectively inhibited (35% in HBEC, P < .005; 47% in HBSM, P < .05; 58% in HBA, P < .05) the forskolin-stimulated cAMP production in all three cell types (Fig. 5) and this effect was significantly antagonized (P < .05) by the 5-HT1B/1D receptor antagonists, GR127935 and GR55562 (Fig. 5). On the other hand, addition of 5-CT alone resulted in a potent stimulation (446% to 3593% above basal levels, P < .005) of cAMP production (Fig. 6) in all three cell types; an effect completely inhibited by addition of the nonselective 5-HT7 receptor antagonist, mesulergine (Fig. 6).
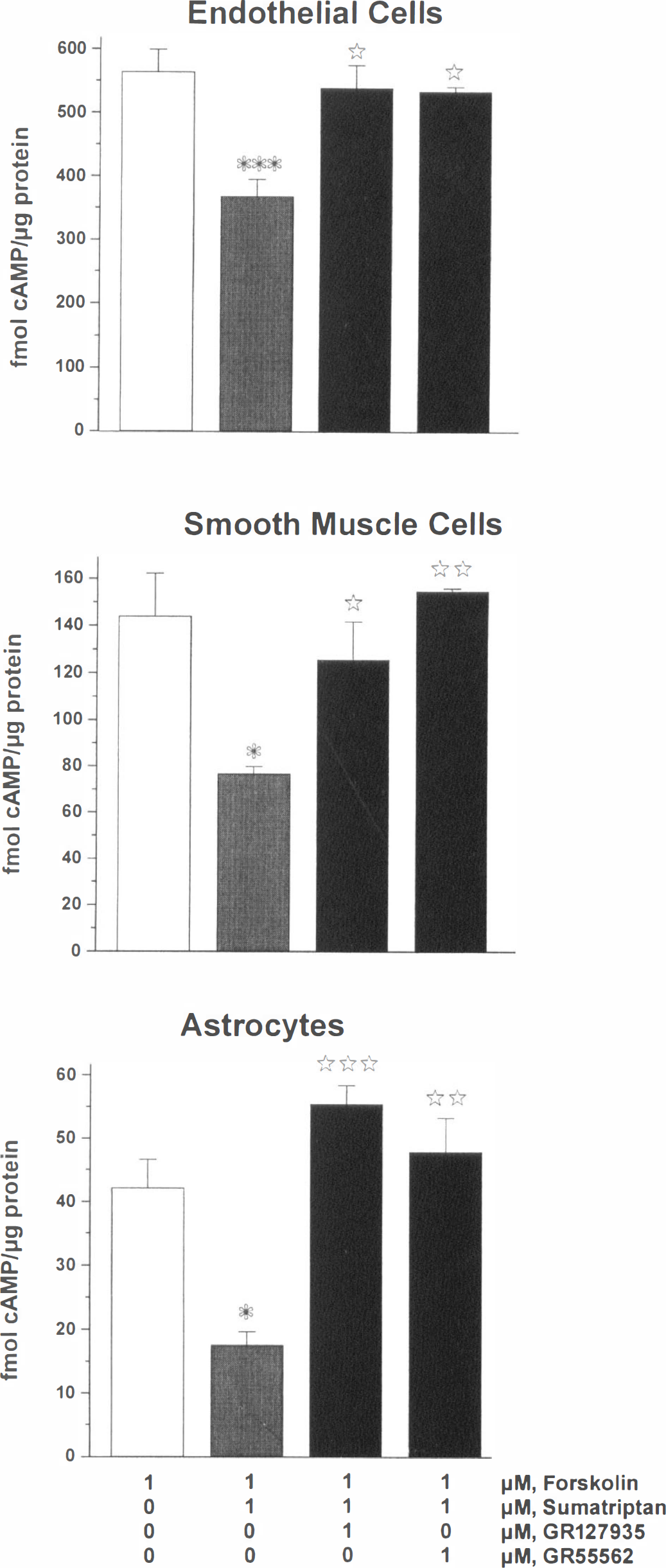
Effects of the 5-HT1 receptor agonist, sumatriptan, on the forskolin-stimulated cAMP production in HBEC, HBSM, and HBA cultures. Forskolin-stimulated cAMP levels were taken as controls (open box). In all three cells types, sumatriptan significantly inhibited the stimulatory effect of forskolin, a response partially (HBEC) or totally (HBSM and HBA) blocked by the 5-HT1B/1D receptor antagonists, GR127935 and GR55562. Comparisons were made between cells treated with forskolin alone and with sumatriptan (* and *** P < .05; P < .005, respectively; analysis of variance followed by Schéffé's post hoc test) and between forskolin/sumatriptan treated cells to those also treated with the antagonists (*, **, ***: P < .05; P < .01; P < .005, respectively; analysis of variance followed by Scheffe's post hoc test).
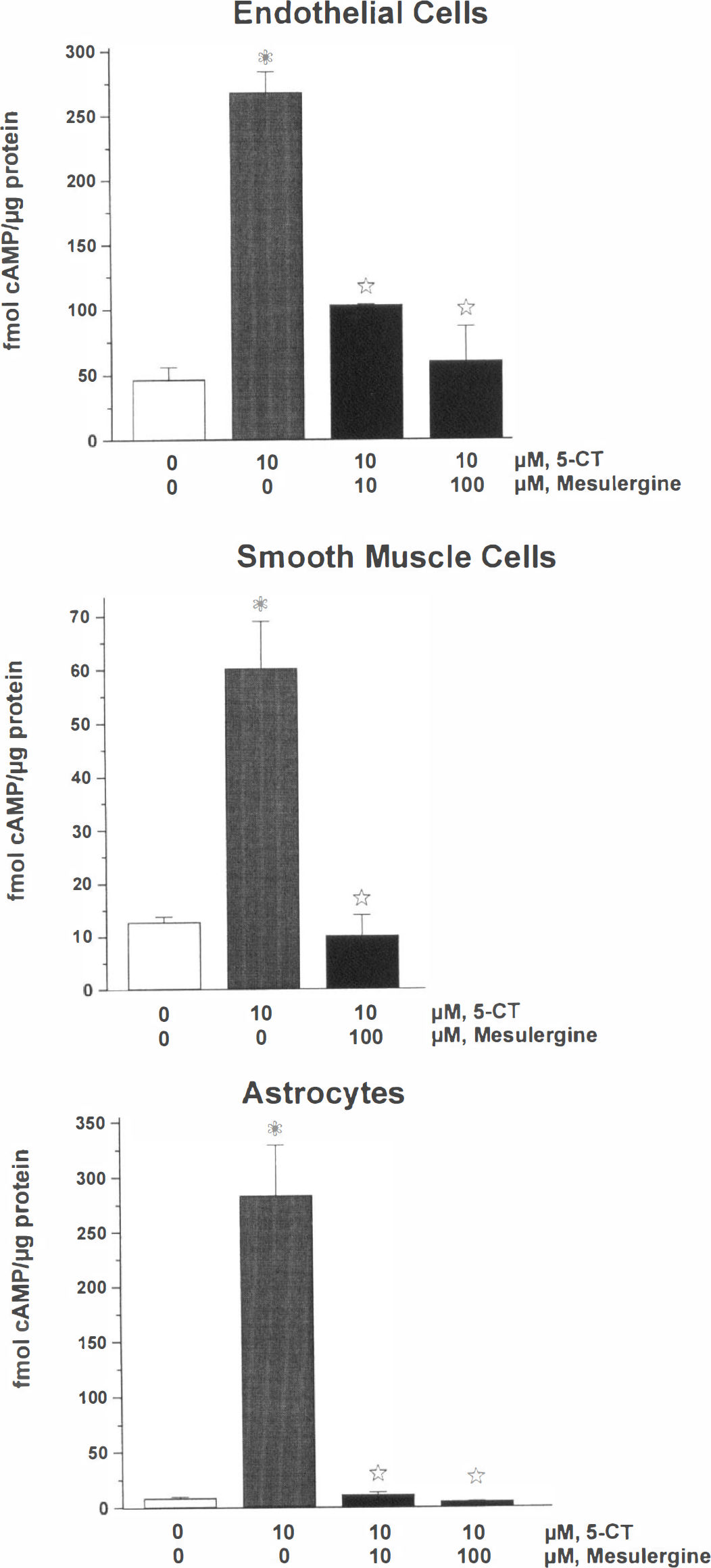
Effects of 5-HT, 5-CT, and mesulergine on cAMP production in HBEC, HBSM, and HBA cultures. The basal levels of cAMP in the cells were taken as controls (open box) and the stimulatory effect of 5-CT was significant (P < .005; analysis of variance followed by Schéffé's post hoc test) in all cultures. The non-selective 5-HT7 receptor antagonist, mesulergine, abolished the agonist-mediated increase in cAMP levels in all cells (P < .005; analysis of variance followed by Schéffé's post hoc test).
IP3 production. In HBEC and HBSM cultures, 5-HT (1-100 µmol/L) did not induce any significant changes in IP3 formation, although significant increases in IP3 levels were observed in the same cultures stimulated by histamine (100 µmol/L) and endothelin-l (100 nmol/L) (287% and 1268%, respectively) (data not shown). In contrast, 100 µmol/L 5-HT induced a 177% increase (P < .05) in IP3 levels in HBA and this stimulation was completely blocked by the 5-HT2A/2C receptor antagonist, ketanserin (Fig. 7).
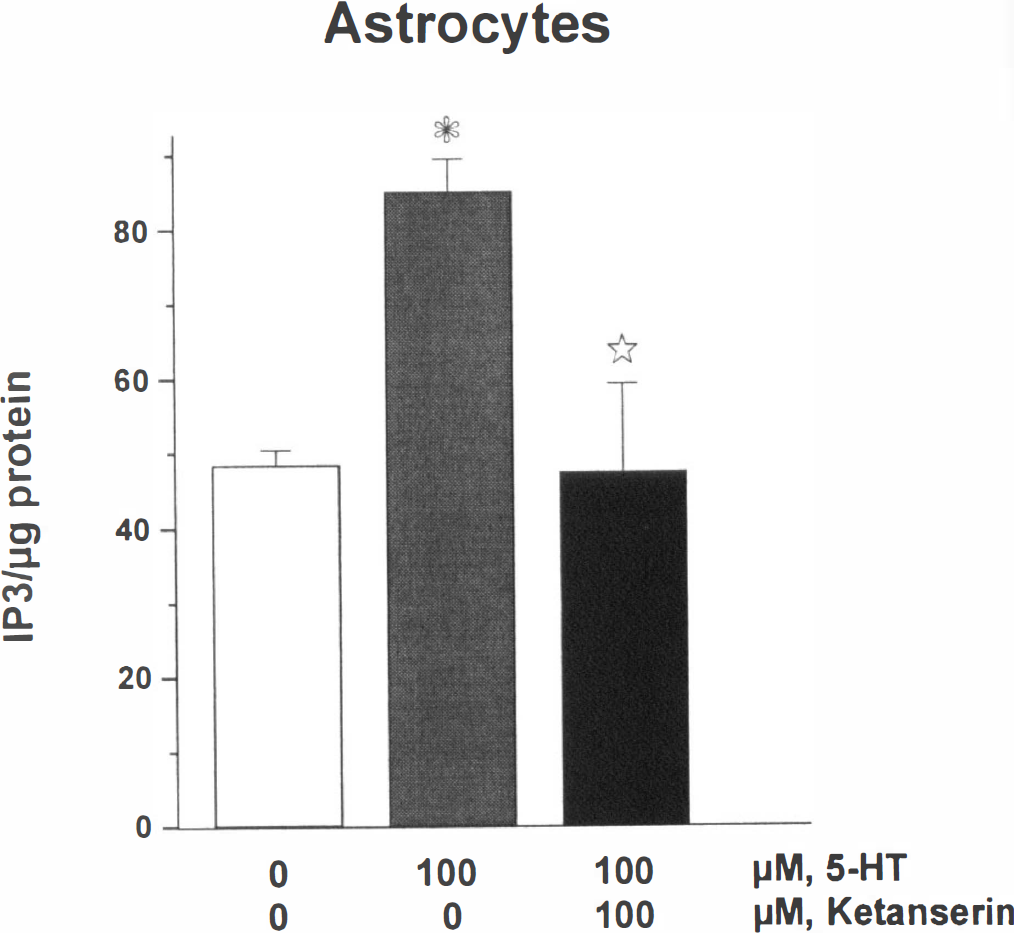
Significant stimulation of IP3 formation (expressed as DPM per µg protein) in HBA cultures in the presence of 5-HT (P < .05), and potent blockade by the selective 5-HT2A receptor antagonist, ketanserin (P < .05; analysis of variance followed by Schéffé's post hoc test).
DISCUSSION
The present data show that different cellular compartments within the microvascular bed of the human cerebral cortex express distinct populations of 5-HT receptors, findings that provide a strong basis to the concept that central serotonergic pathways can regulate cerebromicrovascular tone, local CBF and BBB permeability. In fact, not only were messages for specific 5-HT receptors known to exert vasomotor functions detected by RT-PCR in a given compartment of the blood vessel wall but the encoded proteins were translated at levels sufficient to functionally couple to their expected signaling pathways (Hoyer et al., 1994) and, therefore, to potentially respond to changes in central serotonergic neurotransmission. Furthermore, we conclude that non-neuronal 5-HT1F and 5-HT2A receptors are astroglial but not vascular receptors in the human cerebral cortex.
Heterogeneity of 5-HT receptors in cerebral MV and cells in culture
5-HT1 receptors. Sumatriptan, a nonselective 5-HT1 receptor agonist that exhibits high affinity at the h5-HT1B, h5-HT 1D, and 5-HT1F receptors (Connor and Beattie, 1996), inhibited the forskolin-induced cAMP formation in all cell cultures, a finding that suggested the presence of functional 5-HT1 receptors. The involvement of h5-HT1B and/or h5-HT1D receptor subtypes in mediating this response was confirmed pharmacologically by the ability of the 5-HT1B/1D receptor antagonists, GR127935 and GR55562 (Skingle et al., 1996; Connor and Beattie, 1996), to block the effects of sumatriptan. Based on the RT-PCR data, it appears that the receptor linked to adenylate cyclase inhibition in capillary HBEC is the h5-HT1D while in the HBSM cell cultures, this response would be primarily mediated by a h5-HT1B receptor, although 5-HT1D receptors may also play a role in this response. Because an effect at the 5-HT1F receptor cannot be totally excluded for the GR compounds at the concentration used in the present study (Connor and Beattie, 1996), a role for these receptors in modulating adenylate cyclase activity in human brain astrocytes will only be confirmed when selective 5-HT1F receptor agonists or antagonists are made available (Johnson et al., 1997; Phebus et al., 1997).
Our finding of h5-HT1D and lack of h5-HT1B receptor messages in HBEC is at variance with previous reports in human and porcine endothelial cells of peripheral vessels (Ullmer et al., 1995; Schoeffter et al., 1995), The HBEC used in the present study are derived from small intracortical capillaries and exhibit phenotypic characteristics of the BBB endothelium (Stanimirovic et al., 1996), As such, they may harbor receptor populations different from those present in endothelial cells from larger MVs and macrovascular peripheral or cerebral blood vessels, This contention is supported by our recent ultrastructural immunocytochemical localization of 5-HT1B receptor protein in human smooth muscle and endothelial cells of intracortical resistance MVs but not in endothelial cells of neighboring capillaries (Riad et al, 1998). Whether endothelial 5-HT1D receptors in the CAP fraction are involved in permeability mechanisms (Sharma et al., 1990) or growth-promoting effects (Pakala et al., 1994) and the endothelial 5-HT1B receptors from larger MVs (Riad et al, 1998) in vasomotor responses as reported in peripheral vessels (Schoeffter et al., 1995) remains unknown and further experiments will be required to test these possibilities.
The strong expression of h5-HT1B receptor in HBSM cells is fully consistent with the recent immunocytochemical muscular localization of this receptor in human meningeal arteries (Longmore et al., 1997) and brain resistance MVs (Riad et al., 1998), as well as with the previous identification of this receptor as the mediator of the 5-HT–induced constriction of human cerebral arteries (Hamel et al., 1993; Kaumann et al., 1993; Connor and Beattie, 1996; Bouchelet et al., 1996). Despite the possibility that endothelial cells may contribute to the PCR detection of h5-HT1B receptor mRNA in primary smooth muscle cultures derived from large MVs (Riad et al., 1998), the second messenger data argue for a muscular receptor as the very high coupling efficacy of sumatriptan in these cultures cannot be related to the low percentage (10% to 15%) of contaminating endothelial cells. The smooth muscle location of the h5-HT 1B receptor and its ability to inhibit cAMP production, two characteristics compatible with a vasocontractile effect (Zgombick et al., 1993; Sweeney et al., 1995), suggest that microvascular h5-HT m receptors may also mediate vasoconstriction. Preliminary results in isolated human brain intracortical arterioles indeed support this conclusion (Elhusseiny and Hamel, 1998). An interesting finding from the present study was the absence of 5-HT1F receptor mRNA in either the endothelial or smooth muscle cell cultures, suggesting that this receptor is likely not involved in the regulation of vascular tone in the human brain microcirculation. Such a statement would agree with previous pharmacologic studies that excluded this receptor subtype as the mediator of the contraction elicited by either 5-HT or sumatriptan in human cerebral arteries (Hamel et al., 1993), and with more recent observations on the failure of 5-HT1F receptor agonists to elicit vasocontraction in peripheral (Johnson et al., 1997) and human brain (Bouchelet et al., 1999) vessels.
The multiplicity of 5-HT1 receptor messages identified in HBA points to various possibilities through which 5-HT can regulate astroglial cells. Functional 5-HT1A receptors have been associated with astrocytes and found to stimulate the production of the serotonergic glial growth factor S-100β (Whitaker-Azmitia et al., 1993). The roles of other 5-HT1 receptors detected in HBA and whether these receptors are expressed in the same cells in vivo remain unknown.
5-HT2A receptors. Expression and effective coupling of 5-HT2A receptors to IP3 formation was exclusively found in astroglial cells, a finding supported by previous reports in other species (Deecher et al., 1992). Because vasomotor responses in human brain vessels, in contrast to peripheral vessels (see Martin, 1994), do not appear to involve 5-HT2A receptors (Hamel and Bouchard, 1991; Kaumann et al, 1993), it is likely that astroglial 5-HT2A receptors regulate specific glial functions such as glycogenolysis (Poblete et al., 1995), astrocyte proliferation and maturation (Le Prince et al., 1990; Pauwels et al., 1996), and the development of central serotonergic pathways (Whitaker-Azmitia et al., 1993). Alternatively, activation of astroglial receptors could lead to the synthesis and release of vasoactive signaling molecules (Alkayed et al., 1997) that could indirectly influence the microcirculation.
5-HT7 receptors. When administered directly to HBEC, HBSM, and HBA, 5-CT significantly increased cAMP levels. This effect is probably due to activation of 5-HT7 receptors (Bard et al., 1993) whose message was detected by RT-PCR in all three cell types. The absence of 5-HT7 receptor mRNA in MV and CAP fractions isolated from post-mortem brains remains unexplained but is unlikely an artifact of cell culturing conditions since the 5-HT7 receptor message has previously been shown in peripheral blood vessels and smooth muscle cells (Ullmer et al., 1995; Schoeffter et al., 1996). 5-HT7 receptor message may be expressed at lower levels in vivo and/or be more susceptible than other 5-HT receptors to downregulation (Shimizu et al., 1997), or postmortem degradation.
The 5-HT receptor antagonist, mesulergine, which exhibits high affinity at human 5-HT7 receptor (pKi, 7.7) and a much lower (about 100-fold) affinity at 5-HT6 receptors (pKi, 5.8; Bard et al., 1993), inhibited the 5-CT–stimulated cAMP levels in all three cell types, strongly suggesting the presence of functional 5-HT7 receptors in both microvascular and astroglial cells. In agreement with our findings, expression of 5-HT7 receptor mRNA and/or stimulation of adenylate cyclase by 5-HT have previously been reported in both human brain endothelial and rat astroglial cells (Bacic et al., 1991; Hirst et al., 1997; Shimizu et al., 1997), or human peripheral vascular smooth muscle cells (Ullmer et al., 1995; Schoeffter et al., 1996). Microvascular 5-HT7 receptors could mediate an endothelium-independent relaxation, an effect similar to that observed in peripheral blood vessels (Leung et al., 1996; Terron, 1996) and fully compatible with their ability to increase cAMP levels. It is also possible that these receptors regulate vascular smooth muscle proliferation as documented in peripheral smooth muscle cells (Lee et al., 1991). Astroglial 5-HT7 receptors might be involved in the regulation of ion and pH homeostasis, BBB, as well as neuronal-astroglial-vascular interactions (Sharma et al., 1990; Hertz, 1992; Cohen et al., 1995b).
Physiologic implications
The expression of heterogeneous microvascular and astroglial 5-HT receptors in human cerebral cortex implies that non-neuronal cells within the brain parenchyma can respond to changes in brain serotonergic neurotransmission, and possibly to circulating 5-HT in the case of endothelial 5-HT receptors, although their luminal localization still has to be established. The finding of functional h5-HT1B and 5-HT7 receptors, respectively known to mediate constriction and dilation in several vascular beds, is consistent with the previous observations, albeit in the rat, of decreases and occasional increases in cortical blood flow following stimulation of serotonergic raphe neurons (Bonvento et al., 1993; Cohen et al., 1996). Therefore, the final and integrated vasomotor response to 5-HT is likely to depend on the proportion of vascular h5-HT1B, 5-HT7 and, possibly, other subtypes of 5-HT receptors (Cohen et al., 1996), the initial tone of the vessel (Rosenblum and Nelson, 1990) and, perhaps, the subdivision of the raphe nucleus being activated (Underwood et al., 1992). Our data raise the possibility of cross-talks between different 5-HT receptors within a given cell type (Tournois et al., 1998). Further, the distribution of non-neuronal 5-HT receptors identified in this study raises the possibility that new antimigraine drugs that penetrate into the brain parenchyma (Martin, 1997; Goadsby, 1997) and may access and activate some of these microvascular and astroglial 5-HT receptors.
Footnotes
Acknowledgments
The authors thank Drs. V.W. Yong and J.P. Antel for their generous supply of HBA cultures, Dr. B. Case for the post-mortem tissue, and Dr. P. Séguéla for the design of some oligonucleotide primers. The authors are also grateful to M. Arts, H. Bernhard and J.P. Acco for expert photographic work and L. Michel for preparing the manuscript.
