Abstract
Interleukin-6 (IL-6) may play multiple roles in angiogenesis and vascular remodeling. Our previous study showed that a promoter polymorphism (174G>C) in IL-6 is associated with brain arteriovenous malformation hemorrhage; tissue expression is related to genotype. In this study, we investigated the effects of IL-6 on human cerebral smooth muscle cells (HCSMCs) and smooth muscle cells isolated from brain arteriovenous malformation surgical specimens (AVM SMCs) and surgical controls (control HCSMCs—from structurally normal temporal lobe taken during surgical treatment of epilepsy patients). We found that IL-6 (1.1±0.27 versus 0.37±0.04 pg/mL, n=5, P<0.05) and endogenous vascular endothelial growth factor (VEGF) receptor II (kinase domain-containing receptor (KDR), 15±3 versus 1.5±3 pg/mL, n=5, P<0.05) were increased in brain AVM SMCs compared with control HCSMCs. Further research revealed that IL-6 could stimulate SMC proliferation, VEGF release, and KDR activation in control HCSMCs. It could also stimulate KDR phosphorylation in control HCSMCs, further confirming a unique role of IL-6 in the triggering of KDR. Interleukin-6 could increase matrix metalloproteinase-9 (MMP-9) secretion through activating KDR in control HCSMCs (P<0.05 versus control). Inhibiting IL-6-induced KDR could reduce MMP-9 activity at least 50% compared with the control group (P<0.05). Increased MMP-9 activity was accompanied by increased control HCSMC proliferation, and blocking MMP-9 activity significantly reduced IL-6-induced control HCSMC proliferation (P<0.05). Collectively, our results show that IL-6 could activate, amplify, and maintain the angiogenic cascade in HCSMCs. A novel role of IL-6 during HCSMC proliferation is upregulating KDR expression and phosphorylation. The results may contribute to the angiogenic phenotype of human brain vascular diseases, such as brain AVM.
Keywords
Introduction
Brain arteriovenous malformations (AVMs) are an important cause of hemorrhagic stroke in young adults, but how brain AVMs develop in adults remains poorly understood. Human cerebral smooth muscle cells (HCSMCs), a major cell type in the brain vascular wall, are involved in vascular remodeling and angiogenesis. Proliferation and migration of HCSMCs are the important determinants of brain vascular diseases including AVMs. Many growth factors and cytokines play essential roles during vascular remodeling and angiogenesis, and among these interleukin-6 (IL-6) has received particular attention. Interleukin-6, a multifunctional cytokine, is a critical factor in inducing inflammatory, immunoregulatory, and hematopoietic responses (Akira et al, 1993; Taga and Kishimoto, 1997). It plays an important role in both physiologic and pathologic angiogenesis (Wang and Newman, 2003). Our previous studies showed that IL-6 174C>G polymorphism is associated with brain AVM patients (Pawlikowska et al, 2004). Brain AVM patients with IL-6 174GG polymorphism have higher IL-6 protein in both blood samples and surgical tissue (Chen et al, 2006). Recent in vitro studies show that IL-6 can activate SMC, which has profound effects on the proliferation, migration, and secretion of cytokines (Wang and Newman, 2003). These processes have been most often related to the action of vascular endothelial growth factor (VEGF) (Wang and Newman, 2003).
Vascular endothelial growth factor is the most important growth factor in inducing vascular remodeling and promoting angiogenesis. Expression of VEGF is greatly increased during normal physiologic conditions (e.g., embryo development and wound healing) and abnormal physiologic conditions (e.g., tumor angiogenesis and brain AVMs) (Hashimoto et al, 2005). Vascular endothelial growth factor exerts its biologic function through specific membrane receptors: kinase domain-containing receptor (KDR) and the fin-like tyrosine kinase 1 (flt-1) (Plouet and Moukadiri, 1990). On ligand binding, VEGF receptors initiate autophosphorylation and induce tyrosine kinase activity, subsequently stimulating cellular responses (Miao et al, 1999). Although KDR and flt-1 are structurally highly similar, their mechanisms in vascular remodeling and angiogenic signal transduction are different: KDR is responsible for mediating the proliferation response to exogenous VEGF (Liu et al, 1995), whereas flt-1 is involved in SMC migration (Soker et al, 2001). Until now, there is no evidence that VEGF through KDR plays an angiogenic action in HCSMCs.
Smooth muscle cell activation is involved in increased production of matrix metalloproteinase-9 (MMP-9), which is a member of the family of zinc-dependent endopeptidase. It may promote angiogenesis through regulating cell attachment, proliferation, and migration. These actions of MMP-9 could be produced through direct or indirect growth factor release from the extracellular membrane (Jia et al, 2000). Growth factors and cytokines could stimulate MMP-9 expression during abnormal angiogenesis (Fabunmi et al, 1996). However, the relationship between growth factor release and MMP-9 activation, which may be involved in HCSMC proliferation, is unknown.
In the present study, we investigated VEGF and KDR changes in IL-6-stimulated HCSMCs, and evaluated functional activity of KDR by monitoring its phosphorylation. We further measured MMP-9 activity with IL-6 stimulation. Finally, we explored the role of IL-6 on brain AVM SMC and surgical control HCSMC proliferation.
Materials and methods
Smooth Muscle and Endothelial Cell Culture
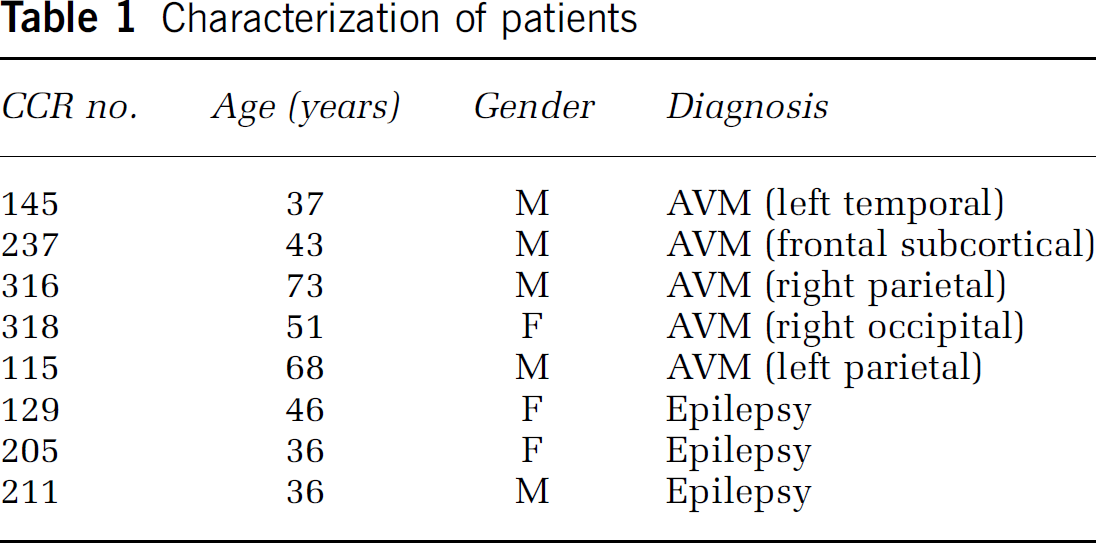
The study was IRB-approved and patients gave informed consent. Brain AVM characteristics such as size, location, and venous drainage pattern were documented based on standardized guidelines (Joint Writing Group of the Technology Assessment Committee, 2001). Control cerebral cortex was obtained from patients undergoing surgical treatment of epilepsy as previously described (Table 1) (Hashimoto et al, 2004, 2005). Three types of brain cells were used: (1) AVM SMCs—cells isolated from brain AVM surgical specimens (n=5); (2) surgical control SMCs—cells isolated from structurally normal temporal lobe taken during surgical treatment of epilepsy patients (n=6), of which three were a gift from Dr Harry V Vinters of UCLA; these SMCs are also called control HCSMCs because no abnormal phenotype was detected; (3) normal HCSMCs and human cerebral endothelial cells (HCECs) purchased from Cell Systems (St Katharinen, Germany). The SMCs' homogenous nature was confirmed by immunostaining with anti-SM-α-actin, and no significant difference in morphology was found (Chemicon, Temecula, CA, USA; Figure 5A). All experiments were performed on control HCSMC two to five passages. In IL-6 treatment experiments, control HCSMCs were grown in 10% fetal bovine serum, whereas HCECs were grown in ECs growth medium (Clonetics, Walkersville, MD, USA) in 35 mm polystyrene plates to 90% confluent state, and rinsed three times with a serum-free medium before arresting with serum-free medium. The cells were then incubated with IL-6 and other antibodies in specified conditions.
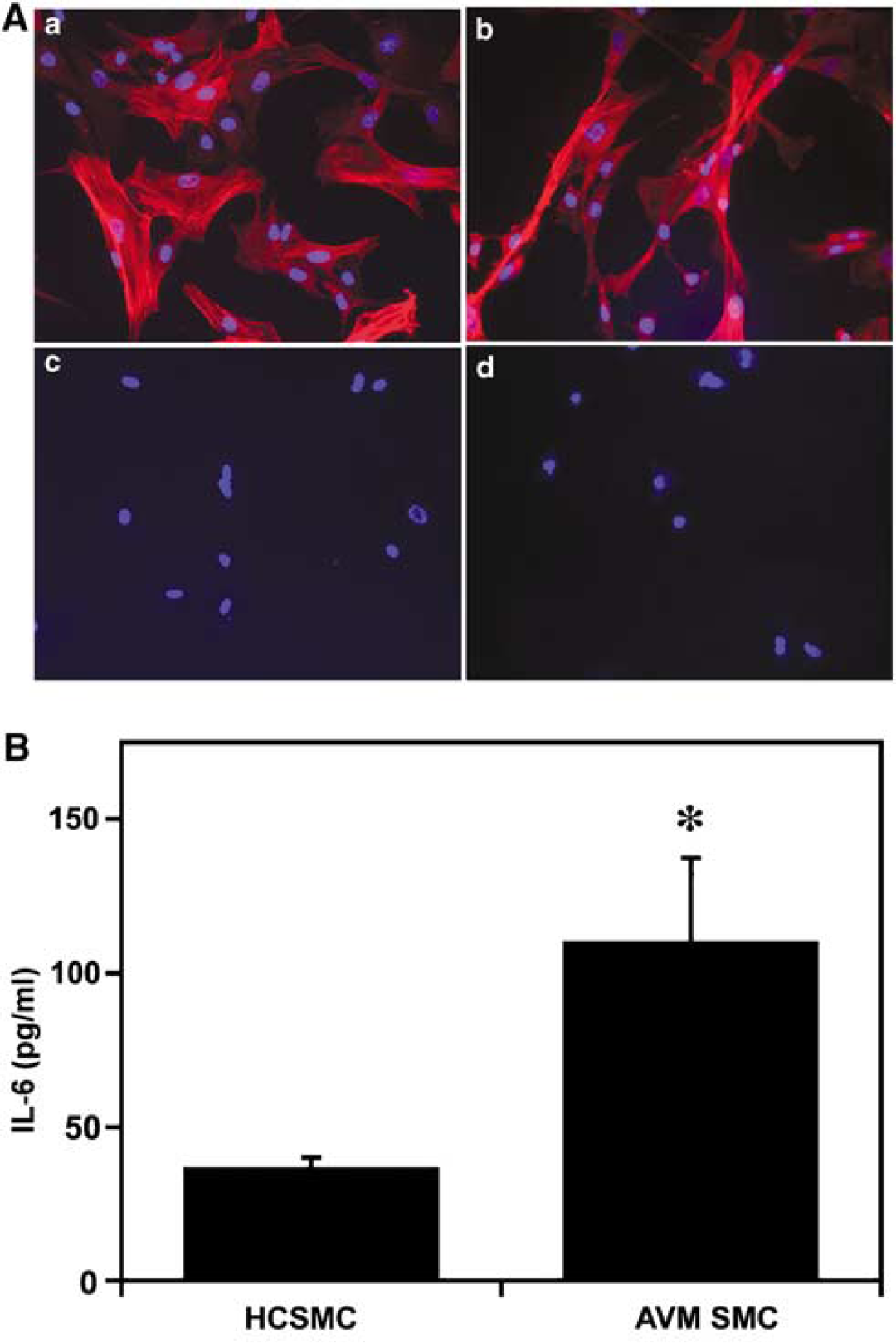
Increase of IL-6 in AVM SMC culture. (
Characterization of patients
Antibody Cytotoxicity was Determined using Colorimetric Thiazolyl Blue
Antibody cytotoxicity was determined using MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) assay (Lokeshwar et al, 2002). The HCSMCs and HCECs were grown in 96-well microtiter plates with 100 μL of culture medium for 24 h. The cells were treated with IL-6 at increasing concentration in 0.2% bovine serum albumin/Dulbecco's modified Eagle's medium. The plates were incubated for 36 h and the media were replaced with 100 μL fresh Dulbecco's modified Eagle's medium containing 0.2% bovine serum albumin. Next, 2 mg/mL MTT solution was added and the plates were incubated again for 3 h. Finally, MTT-containing medium was aspirated off and 150 μL of dimethyl sulfoxide solution was added. Crystals were solvated by dimethyl sulfoxide and the absorbance was measured at 570 nm using an enzyme-linked immunosorbent assay (ELISA) reader (E max, Molecular Devices, Sunnyvale, CA, USA).
HCSMC and HCEC Proliferation Assay
The HCSMCs and HCECs grew to 90% confluence in T-25 flash, and the cells were harvested using 0.025% trypsin. The cells (1 × 104) were seeded onto 96-well gelatin-coated plate overnight, treated with different antibodies for another 24 h, followed by incubation with 5-bromodeoxyuridine for 2 h (BrdU cell proliferation ELISA kit, Roche, Penzberg, Germany) and measurement of absorbance at 450 nm (reference wavelength 690 nm) using an ELISA reader (E max).
Real-Time Polymerase Chain Reaction
Total RNA was extracted from cultured cells using Trizol reagent (Invitrogen, Carlsbad, CA, USA). Reverse transcription was performed using a RETROscript kit (Ambion, Austin, TX, USA). Oligonucleotide primers are as follows: MMP-9 forward: 5′-CCGCAGGGCCCCTTCCTTAT-3′; reverse: 5′-CCGCAGGGCCCCTTCCTTAT-3′; VEGF forward: 5′-TGTGAATGCAGACCAAAGAAAGA-3; reverse: 5′-GCTTTCTCCGCTCTGAGCAA-3′; probe: AGAGCAAGACAAGAAAATCCCTGTGGGC; Glyseraldehyde-3-phosphate dehydrogenase (GAPDN) forward: 5′-GAAGGTGAAGGTCGGAGTC-3′; reverse: 5′-GAAGATGGTGATGGGATTTC-3′; probe: CAAGCTTCCCGTTCTCAGCC. Real-time polymerase chain reaction (RT-PCR) was determined using 2.5 μL (5 ng) cDNA, 12.5 μL SYBR Green PCR Master Mix (2 ×) for MMP-9, or Taqman PCR Master Mix (2 ×) for VEGF, 2.5 μL primer pair mix (5 pmol each primer), and water to a 25 μL final volume (ABI-prism 7000 sequence detector, Foster City, CA, USA). Thermocycler conditions consisted of an initial holding at 50°C for 2 mins and then 95°C for 3 mins. This was followed by 94°C for 15 secs, 56°C for 45 secs, and 72°C for 30 secs for 45 cycles for MMP-9. For VEGF, thermocycler conditions consisted of an initial holding at 50°C for 2 mins and then 95°C for 10 mins. This was followed by a two-step TaqMan PCR program consisting of 95°C for 15 secs and 60°C for 60 secs for 35 to 40 cycles. Results were analyzed using ABI sequence detection software, and MMP-9 and VEGF mRNA levels were normalized to glyceraldehyde 3-phosphate dehydrogenase (18S rRNA endogenous control). The RT-PCR products were verified on agarose gels.
Western Blot Analysis
After harvesting, HCSMCs were homogenized in a lysis buffer. The protein concentration was determined using a Bio-Rad system. An equal amount of protein (70 μg) was loaded on 4% to 12% gradient gel (Invitrogen) for electrophoresis of protein. Subsequently, protein was electroblotted onto a polyvinylidene difluoride membrane (Bio-Rad, Richmond, CA, USA) in a transfer buffer. The membrane was placed in 5% nonfat milk TBS with 0.1% Tween 20 for 1 h to block nonspecific binding, and was then immunoprobed overnight with anti-VEGF antibody (1:200 dilution; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA), anti-KDR antibody (1:200 dilution; Santa Cruz), or anti-flt-1 antibody (1:300 dilution; Santa Cruz). After washing, the membrane was incubated with horseradish peroxidase (HSP)-conjugated anti-rabbit antibody or anti-mouse antibodies (Amersham). Finally, the membrane was incubated with an enhanced ECL (Amersham), and exposed to Kodak film. Protein bands were scanned and semiquantified by densitometry using NIH 1.63 image analysis software.
Phospho-KDR Expression
Phospho-KDR expression was semiquantified using Western blot analysis. The HCSMCs grew to approximately 80% confluent, and made quiescence with serum-free medium for 24 h. Then, IL-6 (100 ng/mL) was added and plates were incubated for 5 to 45 mins. The cells were washed with phosphate-buffered saline and scraped into a lysis buffer. Phospho-KDR was detected by incubating membranes overnight with rabbit polyclonal phospho-KDR antibody (1:100 dilution; Santa Cruz); secondary horseradish peroxidase-labeled anti-rabbit antibody (1:2,000 dilution; Amersham) was added for 1 h.
Gelatin Zymography
Zymography was performed as we have previously described (Hashimoto et al, 2003). A 70 μg sample protein was separated under nonreducing conditions in a 10% zymogram gel (Invitrogen) containing 0.1% gelatin as a substrate. After electrophoresis, gels were washed and incubated in developing buffer overnight at 37°C. The gels were then stained with 0.5% Coomassie blue R-250 and destained. The activity of MMP can be detected as white bands of lysis against the Coomassie blue-stained gel. Protein activity bands in zymography were quantitated through scanning densitometry.
Immunohistochemistry
The cell sections were fixed using 4% paraformaldehyde and quenched using 0.3% H2O2 in methanol for 30 mins. After washing, sections were incubated with primary antibodies: mouse anti-SM-α-actin (1:200 dilution; Chemicon, Temecula, CA, USA), mouse anti-human CD-31 (1:1,000 dilution; DAKO, Carpinteria, CA, USA), mouse anti-human-glial fibrillary acidic protein (1:500 dilution; Sigma, St. Louis, MO, USA), mouse anti-human flt-1 (1:500 dilution; Santa Cruz), and mouse anti-human-KDR (1:200 dilution; Santa Cruz). After incubating at 4°C overnight and washing in phosphate-buffered saline, the sections were incubated with biotinylated goat anti-mouse (Vector Laboratories, Burlingame, CA, USA) diluted 1:5,000 for 1 h at room temperature. The sections were treated with the ABC streptavidin detection system (Vector Laboratories) for 1 h. The resulting horseradish peroxidase signal was detected using 3,3′-diaminobenzidene. For fluorescent staining, after incubating overnight at 4°C with primary antibodies, sections were incubated with Alexa Fluor 594-conjugated goat anti-mouse or Alexa Fluor 488-conjugated goat anti-rabbit IgG (Molecular Probes, Eugene, OR, USA) at 1:500 dilution for 1 h at room temperature. The sections were then mounted and photographed with a fluorescent microscope. Negative controls were performed by omitting the primary antibodies.
Enzyme-Linked Immunosorbent Assays
Interleukin-6 expression from human brain AVM SMCs were measured using an ELISA kit according to the protocols provided by the manufacturer (R&D Systems Inc., Minneapolis, MN, USA). Cultured brain AVM SMCs were maintained at 37°C in 5% CO2/95% ambient mixed air, and the culture media were changed every 2 days. The same amount of protein was examined for each sample. All experiments were performed on brain AVM SMC two to five passages.
Statistical Analysis
Brain AVM SMCs were compared with controls by analysis of variance. Results of the dose-dependent proliferation and inhibition experiments were analyzed using an analysis of variance with post hoc multiple comparison test. All data are represented as mean±standard deviation (mean±s.d.). A random probability value less than 0.05 is considered statistically significant for the comparisons.
Results
IL-6 Upregulation Stimulates VEGF Expression in Control HCSMCs
To investigate the effects of IL-6 on control HCSMCs, we exposed HCSMCs to IL-6 for 24 h. Interleukin-6 at 50 ng/mL (2 nmol/L) could stimulate the increase of VEGF protein in a dose-dependent manner (P<0.05, IL-6 versus control; Figures 1A and 1B). Consistent with the protein expression, IL-6 could also stimulate VEGF mRNA expression in a dose-dependent manner (P<0.05; Figure 1C). To determine if increased VEGF expression was because of IL-6 stimulation, we neutralized IL-6 with concentrations equivalent to 100 ng/mL of IL-6 in cultured HCSMCs. As anticipated, neutralizing IL-6 could abolish VEGF expression in both protein and mRNA levels (Figures 1D–1F), whereas the same concentration of nonimmune goat IgG could not reduce VEGF expression (data not shown). MTT assay shows no detectable cytotoxicity of tested antibodies in the experiment, indicating that the changes in cellular events were a result of neither nonspecific IgG nor cytotoxicity.
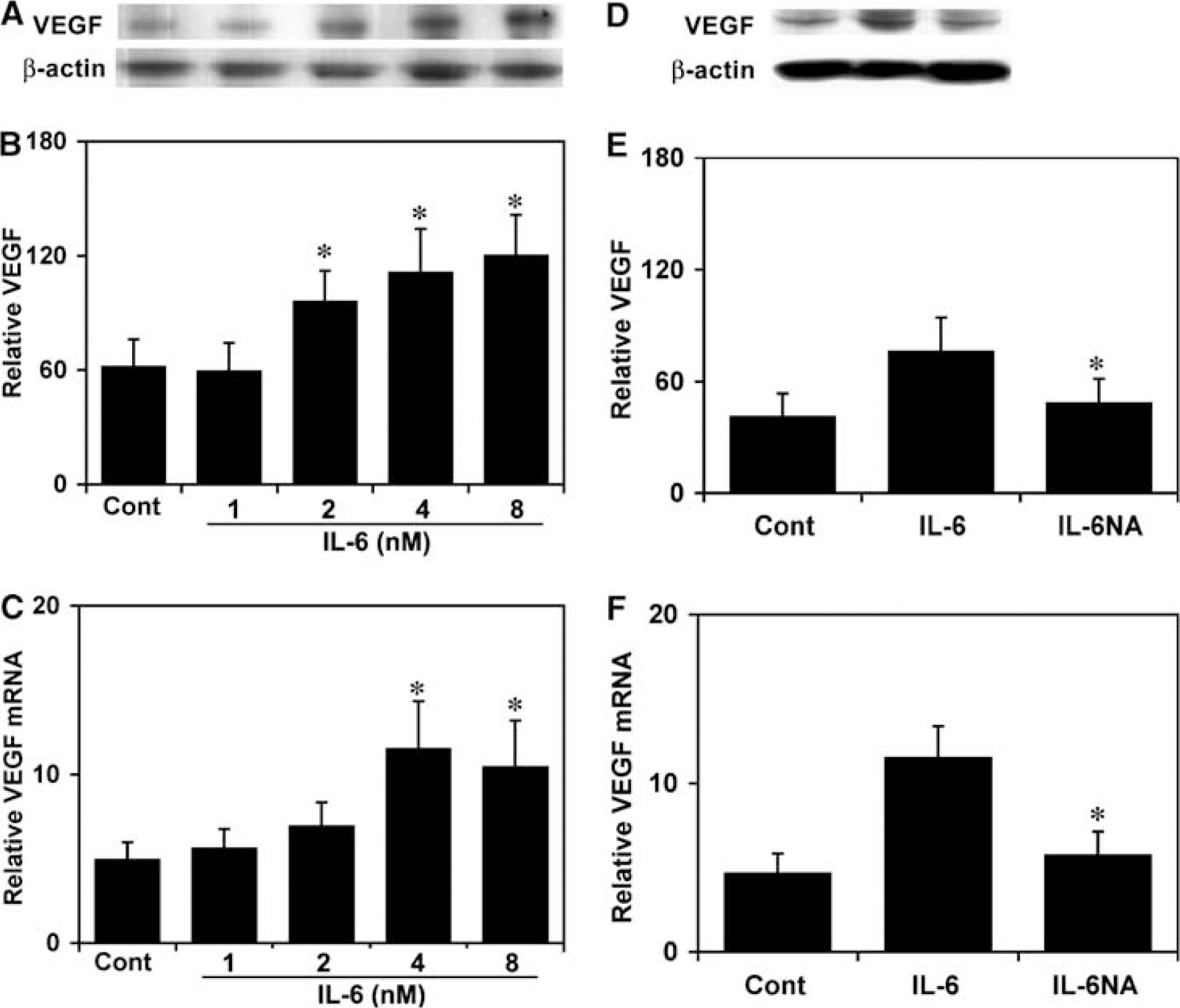
Interleukin-6 upregulates VEGF expression in control HCSMCs. (
IL-6 Stimulates KDR Expression and Phosphorylation in HCSMCs
To explore possible signal transduction pathway for VEGF, we examined VEGF receptors flt-1 and KDR expression in control HCSMCs. There was no statistical significance in flt-1 expression between the control and the treated groups after IL-6 stimulation (Figure 2A). However, KDR expression was upregulated in a dose-dependent manner in control HCSMCs (P<0.05; Figures 2A and 2B). The parallel result was obtained in immunofluorescent staining (Figure 2C). Prolonged exposure to IL-6 produced no further enhancement of KDR (data not shown). Treatment with human VEGF of 20 ng/mL for 24 h could upregulate KDR in normal HCECs but not in control HCSMCs; interestingly, IL-6 could upregulate KDR expression in both normal HCECs and control HCSMCs (Figure 2D). To ascertain that KDR expression was not because of an increase of endogenous VEGF induced by IL-6, we used anti-VEGF antibody to block endogenous VEGF. We found that blocking endogenous VEGF failed to abolish IL-6-induced KDR expression (Figures 2E and 2F); furthermore, nonimmune goat IgG (2 μg/mL) did not influence KDR expression (data not shown). Taken together, these data indicate that IL-6 upregulating KDR was independent of VEGF. To find out if IL-6 upregulating KDR expression was functional, phospho-KDR was examined in the cell extracts from non-IL-6-stimulated or IL-6-stimulated control HCSMCs. The results showed that IL-6 retained the ability to activate KDR, which peaked after 15 mins of IL-6 stimulation (Figure 2G).
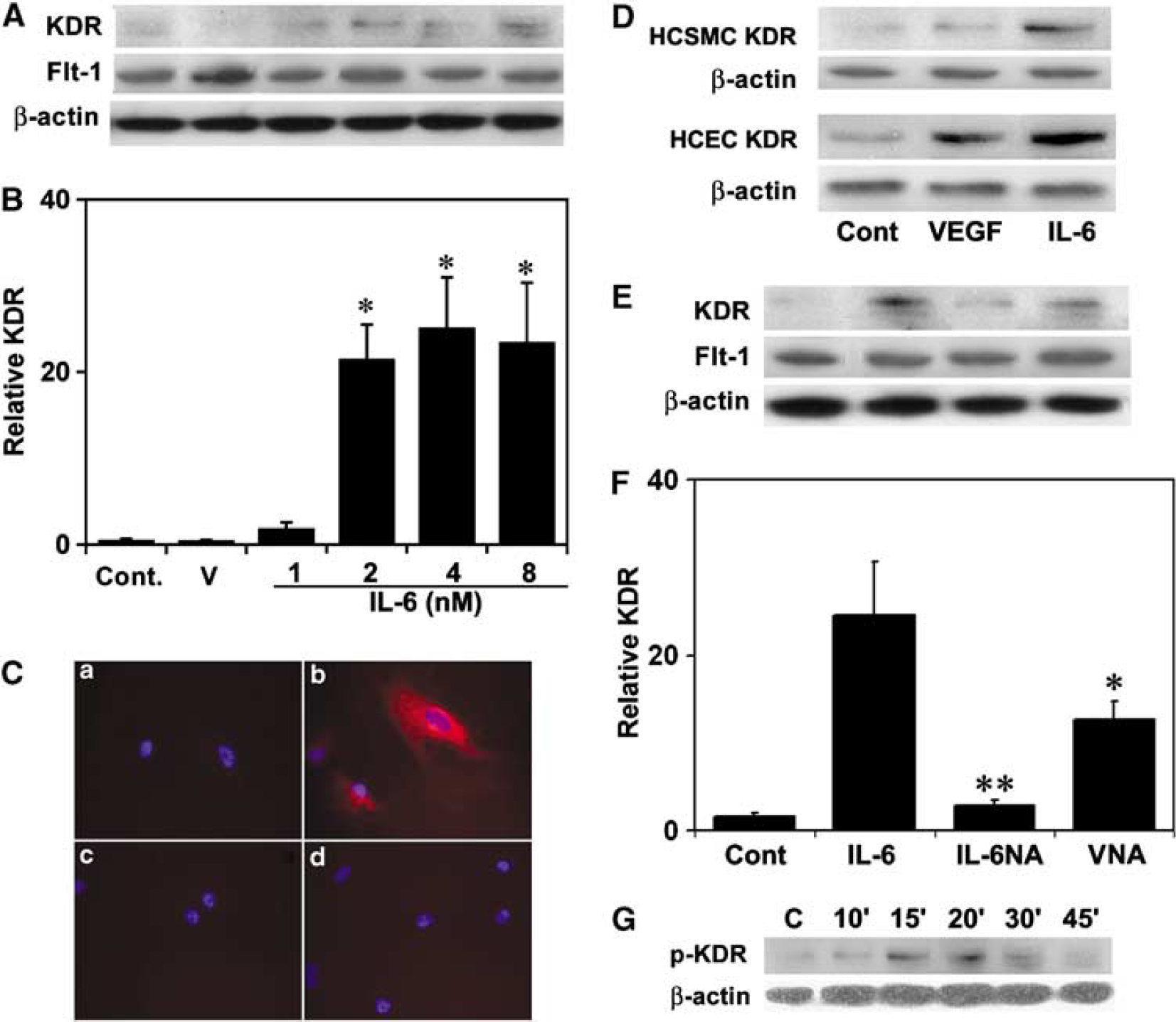
Interleukin-6 upregulates KDR expression and phosphorylation in control HCSMCs. (
IL-6 Enhances MMP-9 Expression
To examine whether IL-6 enhances MMPs expression, we measured MMP-2 and -9 mRNA level in control HCSMC culture. Interleukin-6 did not increase MMP-2 mRNA expression (data not shown), but it greatly increased MMP-9 mRNA in control HCSMCs (P<0.05; Figure 3C). Because gelatin MMPs are secreted as inactive zymoyens, it has to be activated by cleavage of N-terminal sequences to achieve its proteolytic activity. We therefore performed zymographic analyses in the serum-free HCSMC cultured supernatants. Our results indicate that IL-6 did not change MMP-2 activity; however, it increased MMP-9 activity in control HCSMCs (P<0.05; Figures 3A and 3B).
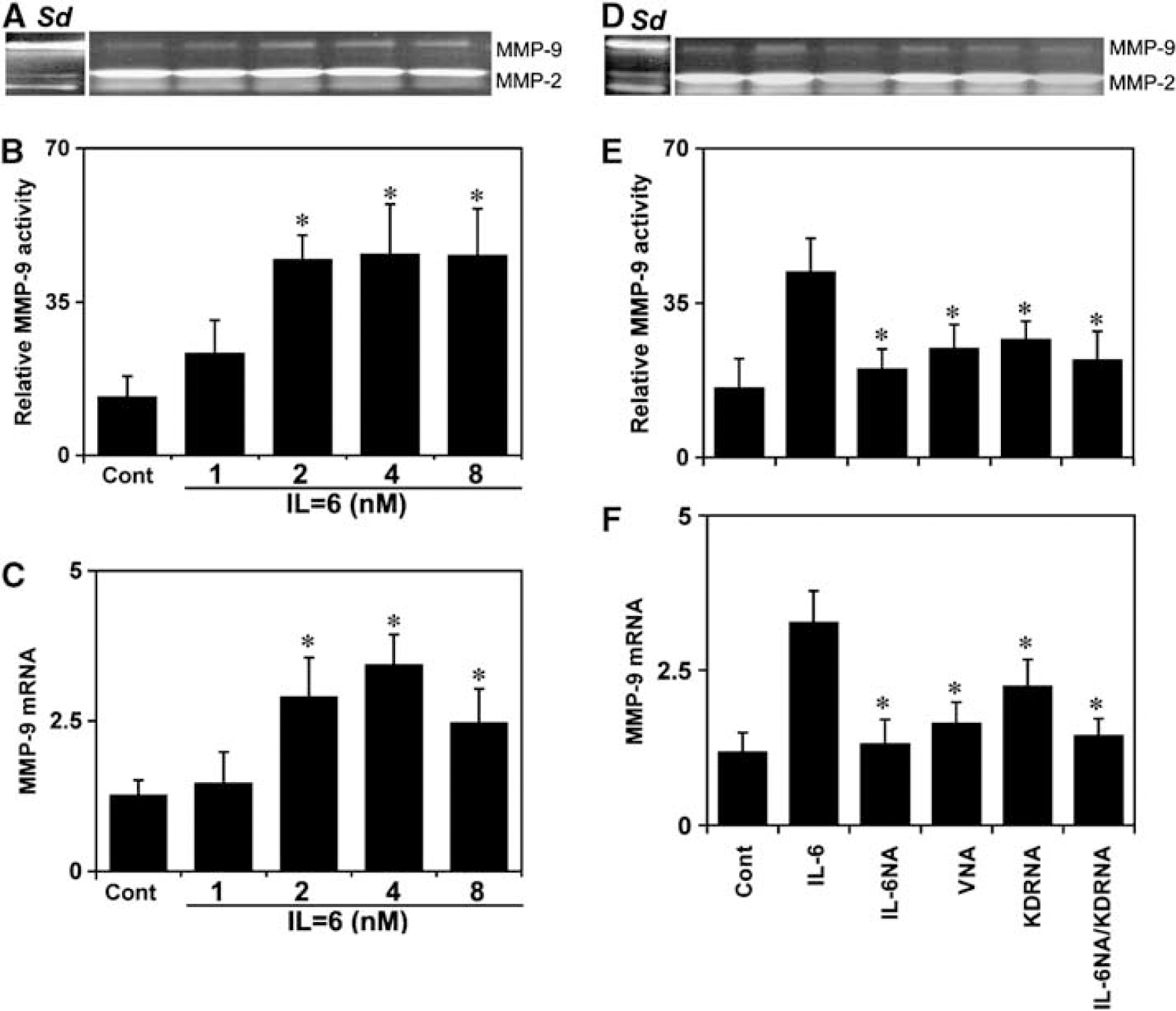
Interleukin-6 enhances MMP-9 expression and activity. (
To determine the relationship among IL-6, VEGF, KDR, and MMP-9 activity, neutralizing antibodies against IL-6, VEGF, and KDR were used in the IL-6-treated HCSMCs. The result clearly indicated that anti-IL-6 antibody effectively reversed MMP-9 overexpression; however, inhibiting KDR and VEGF only partially reduced (≈50%) IL-6 effect in both protein and mRNA levels, suggesting a direct role of IL-6 on MMP-9 activity (Figures 3C, 3D, and 3F). No changes were detected in the control cells treated with goat or mouse IgG at the same concentration, and additional administration of these inactivated antibodies failed to induce MMP-9 inhibition (data not shown).
IL-6 Stimulation Causes HCSMC Proliferation Through Upregulation of KDR
The effect of IL-6 on HCSMC proliferation showed that IL-6 at 100 ng/mL could nearly double proliferation rates of control HCSMCs. This effect is comparable with the effect of platelet-derived growth factor, which is a well-known cell proliferation stimulator (Figure 4A). A similar result was obtained when HCECs were stimulated by IL-6 (Figure 4B). Interestingly, VEGF failed to induce HCSMC proliferation but could stimulate HCEC proliferation (Figures 4C and 4D). Anti-VEGF and anti-KDR antibodies, in part, decreased IL-6-mediated HCSMC proliferation, which suggests that KDR activation is a downstream event in IL-6-induced proliferation. Furthermore, the increased proliferating cells were partially inhibited by MMP inhibitor GM6001, supporting a role of MMP-9 in the HCSMC proliferation process (Figure 4E) (McCawley and Matrisian, 2001).
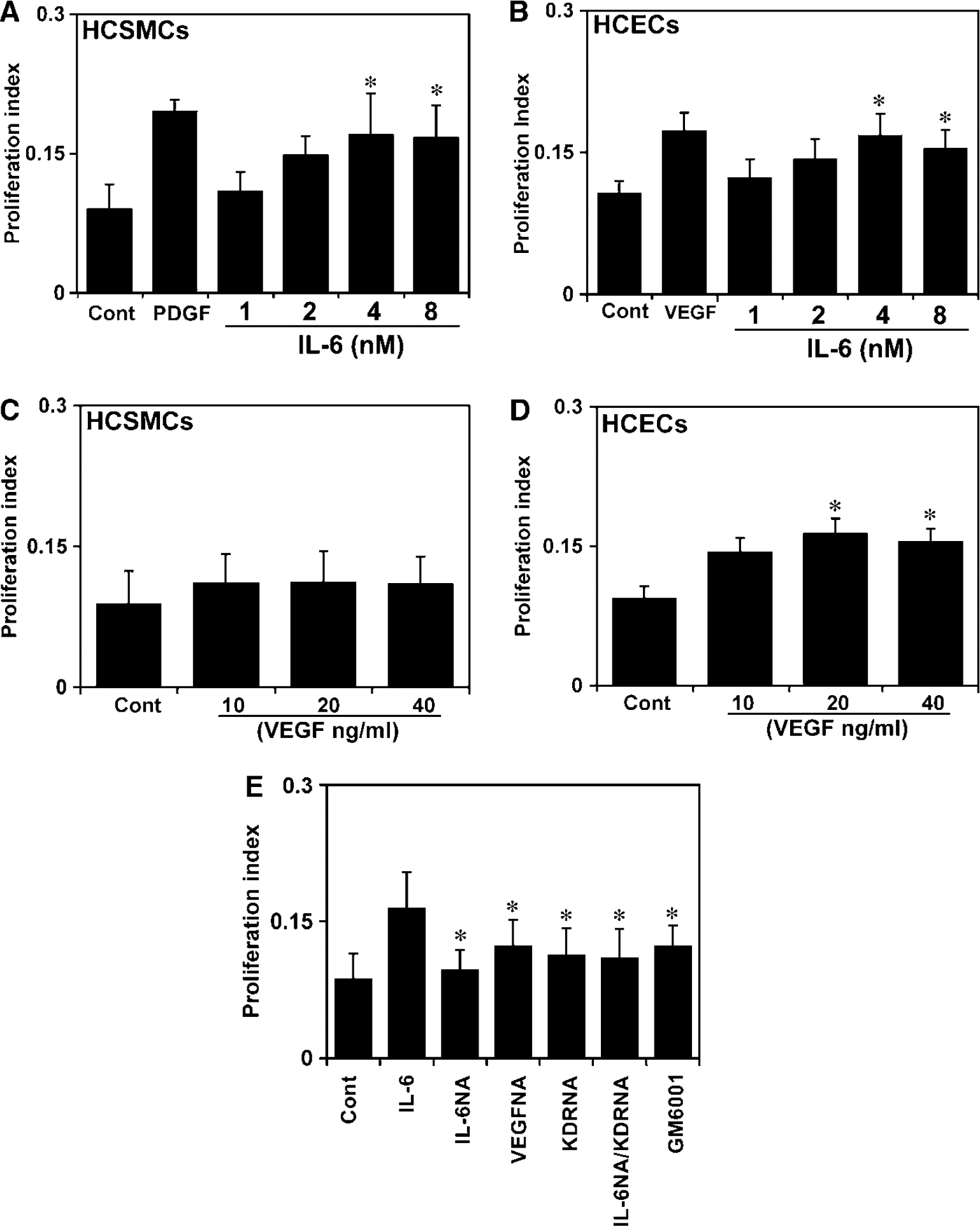
Interleukin-6 stimulation causes control HCSMC proliferation through upregulating KDR. Bar graphs show that IL-6 stimulates both HCSMC (
Increase of IL-6 Expression in the Brain AVM Tissue
Here, we investigated if basic IL-6 level is higher in the AVM SMCs. We first confirmed that the isolated cells from human brain AVM tissue were SMCs (Figure 5A). An ELISA assay then showed that IL-6 level in AVM SMCs was three times higher than in control HCMSCs (P<0.05; Figure 5B).
KDR Expression in AVM SMCs
We then examined whether VEGF receptors flt-1 and KDR expression exists in brain AVM SMCs. Immunostaining showed that all HCECs, control HCSMCs, and AVM SMCs express flt-1 in the normal condition, and that there are no significant differences among cell sources. We showed that KDR-positive staining was detected in the HCEC and AVM SMCs; however, control HCSMCs showed negative KDR staining (Figure 6A). Semiquantitative Western blot result showed a similar result; KDR level in AVM SMCs was 10 times higher than that in control HCSMCs (P<0.05; Figure 6B).
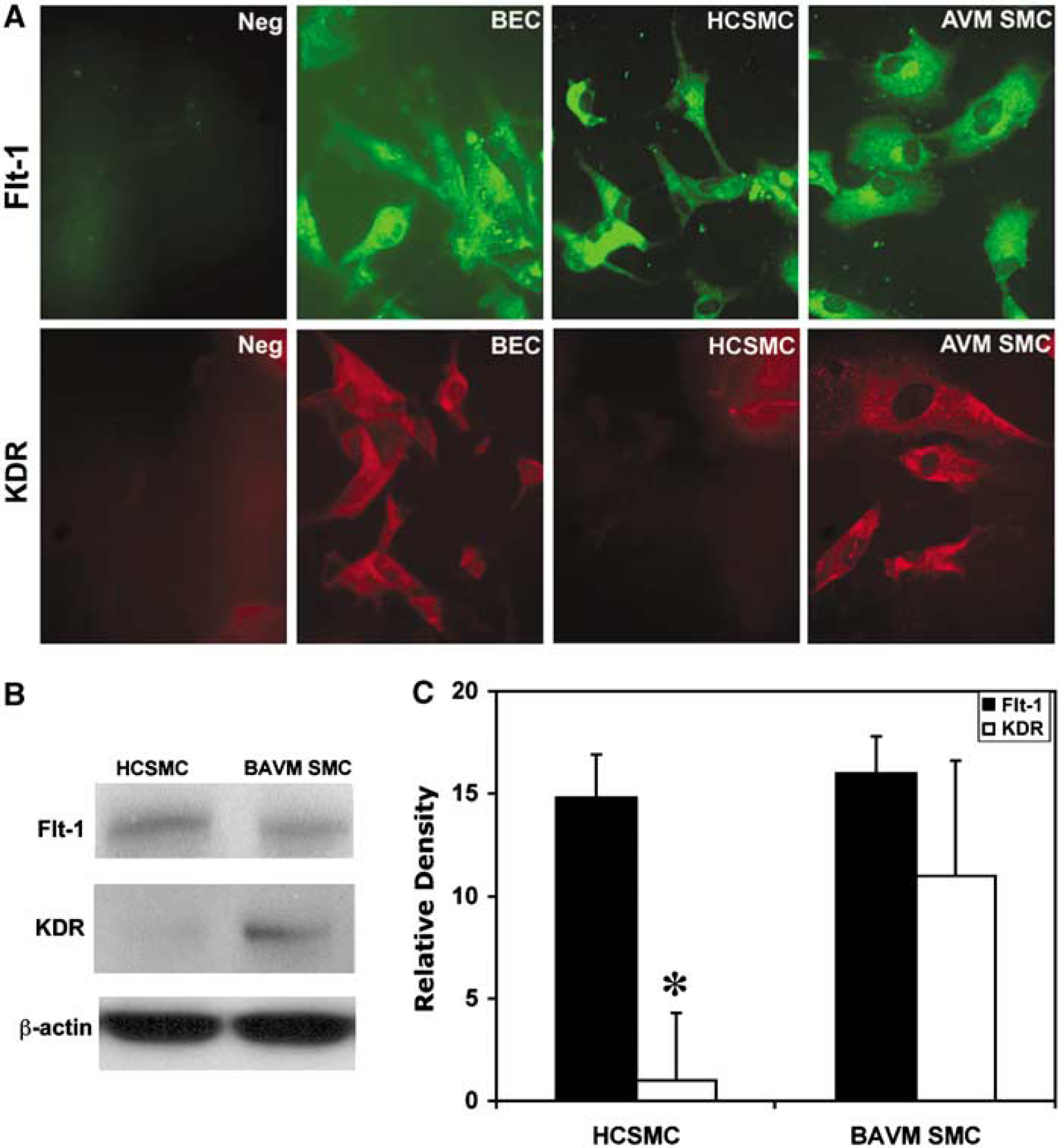
The expression of KDR in AVM SMCs. (
Discussion
In the present study, we revealed for the first time that IL-6 could trigger cultured HCSMC proliferation induced by increased endogenous VEGF and KDR in both mRNA and protein levels. This effect was completely abrogated by IL-6 antibody neutralizing in HCSMCs, which verified the direct trigger effect of IL-6. Exogenous VEGF-treated HCSMC failed to activate KDR, suggesting a specific role of IL-6 in the upregulation of KDR. Interleukin-6 stimulating KDR phosphorylation further supported the role of IL-6 in upregulating KDR. In addition, the role of KDR was identified through IL-6 activating MMP-9. Activating MMP-9 was accompanied by increased HCSMC proliferation, which implies that MMP-9 plays a crucial function in HCSMC proliferation. Finally, we showed that AVM SMCs have higher IL-6 and KDR levels compared with control HCSMCs, indicating that increased IL-6 and KDR are involved in brain AVM development. It is unclear if increased IL-6 is the cause or the effect. However, our genomic study shows that GG genotype of the IL-6−174G>C promoter polymorphism is associated with clinical presentation of brain AVM patients (Chen et al, 2006), which suggests that IL-6 likely plays a role in the development of brain AVM. Our results provided strong evidence that IL-6 could trigger, amplify, and maintain HCSMC proliferation, which may be a major contribution to IL-6-regulated vascular remodeling and induced angiogenesis. Interleukin-6 is a potent preinflammatory cytokine in normal and neoplastic angiogenesis, and has been implicated in all stages of vascular diseases (Campbell et al, 1993). Its overexpression in the central nervous system is correlated to pronounced vasculization in vivo (Campbell et al, 1993). Interleukin-6 polymorphism is closely related to vascular diseases such as abdominal aortic aneurysms and brain AVMs (Jones et al, 2001; Pawlikowska et al, 2004). Increased IL-6 is beneficial to giant-cell arthritis patients, indicating that IL-6 activates a functional program related to angiogenesis (Hernandez-Rodriguez et al, 2003). It also increases in patients after stroke, which may reflect these patients' changing inflammatory-angiogenesis status (Heep et al, 2003). Monocyte-derived macrophage, T cells, ECs, and cardiac myocytes could synthesize IL-6 (Ikeda et al, 1992). We found that IL-6 expression is increased in AVM SMCs, an indication that both ECs and SMCs play a role in triggering pathologic angiogenesis in the brain. The IL-6 level we measured in the SMC culture medium is around 30 to 110 pg/105 SMCs in 1 mL culture medium. The result is similar to our previous study from AVM tissue samples (Chen et al, 2006). The concentration of IL-6 we chose for the stimulation study (25 to 200 ng/mL) is based on our previous research (Yao et al, 2006) and that of other groups (Huang et al, 2004; Loeffler et al, 2005). We did not observe VEGF and KDR expression in the low IL-6 level (less than 25 ng/mL). We believe that a higher dose of IL-6 appears necessary because only by maintaining a higher level of IL-6 within a short period of time can the therapeutic level of IL-6 be reached.
Interleukin-6 may activate signal transducers and activators of transcription 3 signal pathway by binding to gp130 subunit, which then transduces intracellular signals and produces various biologic functions (Kishimoto et al, 1995). This signal pathway widely exists in HCECs and HCSMCs, and mediates pathogenesis of vasculization (Seino et al, 1994).
Involvement of IL-6 in angiogenesis is associated with upregulation of VEGF in many cell types (Cohen et al, 1996). Vascular endothelial growth factor, an essential angiogenic growth factor, plays a crucial role in promoting angiogenesis in HCSMCs. We showed that IL-6 could greatly upregulate VEGF in both transcription and protein levels. Increased VEGF is related to the increase of HCSMC proliferation and migration. Our data provide additional evidence that IL-6 acts as an indirect regulator in HCSMC angiogenic changes. The most important finding in our study is that IL-6 not only induces VEGF release but also directly upregulates KDR. The neutralizing antibody against endogenous VEGF only partially downregulates KDR, indicating that activating KDR is not because of endogenous VEGF. Furthermore, treating HCSMCs with VEGF alone cannot upregulate KDR, presumably because a very low level of KDR in HCSMCs may not interact with exogenous VEGF. We found that IL-6 treatment does not change flt-1 expression in HCSMCs, and neutralizing flt-1 does not influence IL-6-induced HCSMC proliferation. This result implies that IL-6 promoting HCSMC proliferation is through activation of KDR but not flt-1.
Matrix metalloproteinase-9 plays an important role in local microenvironments during embryonic development and growth, as well as in tissue remodeling processes (Mason et al, 1999). Our data showed the role of KDR in upregulating MMP-9 release. Many reports have established that MMP-9 influences HCSMC migration but few have shown that MMP-9 affects HCSMC proliferation. Inhibiting KDR with the specific KDR inhibitor SU5416 decreases HCEC secreting MMP-9 (Wagner et al, 2003). This result strongly supports our similar finding in HCSMCs. Activated KDR is associated with signaling intermediates such as phospholipase C-1γ, GTPase-activating protein, Nck-Grb2, and Shc-Grb2 (Thakker et al, 1999). These downstream events of KDR phosphorylation could contribute to MMP gene regulation. Endothelial cell proliferation is restricted when MMP-9 is absent, whereas EC proliferation is greatly increased when functional MMP-9 is added (Werb, 1997). These results match our finding that upregulating MMP-9 is accompanied by an increased HCSMC proliferation. The mechanism of this process is unknown. One potential explanation could be that MMP-9 stimulates matrix-bound cytokines and growth factors, which act in a paracrine manner to influence cell growth (Bergers et al, 2000; Imai et al, 1997). There are convincing data that these growth factors are necessary for cell proliferation. Another explanation may be that MMP-9 directly cleaves active growth factors. Matrix metalloproteinase-9 directly processes transforming growth factor-β and IL-1β into an active ligand (Schonbeck et al, 1998; Yu and Stamenkovic, 2000), and enables them to diffuse more widely, thereby inducing cell proliferation. The final explanation is that MMP-9 may disrupt cell-to-cell interaction in HCSMC culture. This process may relate to expression of cadherin and β-catenin (Novak and Dedhar, 1999). Neutralizing endogenous VEGF and KDR partially inhibits MMP-9 expression, suggesting that upregulating MMP-9 by IL-6 is not involved in KDR signal pathway. Interleukin-6 could induce angiogenic molecules that play a role in the release of MMP and in stimulating HCSMC proliferation. Therefore, this would explain why blockage of VEGF and KDR, in part, inhibits HCSMC proliferation. Current evidence is clear that IL-6, as one of the principal angiogenic molecules, acts both as a direct and an indirect stimulator of HCSMC proliferation.
The sample size in our study is relatively small, and the results might not be representative of the whole population of brain AVM patients. This would be a limitation if we found no effect, as there might be many confounding factors that could influence the range of values. A false negative would be highly suspect. However, unlike complicated clinical or experimental in vivo studies, our in vitro study was on one type of HCSMC only, and there was a rather robust and uniform difference from control, which suggests a general effect. Therefore, it seems unlikely that we are dealing with a type I false positive effect. Nevertheless, we found a significant difference in IL-6 expression between AVM and controls. In future studies, a large sample size will have to be considered.
In summary, our findings provide evidence that IL-6 stimulating HCSMC proliferation depends on upregulation of KDR. The activation of KDR promotes MMP-9 release from HCSMCs. Interleukin-6 triggering MMP-9 production may have broad implications for brain vascular generation. All our data suggest that IL-6 can activate, amplify, and maintain the angiogenic cascade in HCSMCs. We further showed that AVM SMCs have higher IL-6 and KDR compared with control HCSMCs. As AVM has been considered a continuously progressive angiogenic change and vascular remodeling, and SMCs are one of the main components of the vascular wall, our results provide novel evidence that IL-6 as a chemoattractant can stimulate HCSMC proliferation by upregulation of KDR. This process may be relevant to AVM development in adults.
