Abstract
Neuropeptide Y (NPY) has been suggested as an important regulator of CBF. However, except for the presence of Y1 receptors in large cerebral arteries, little is known about its possible sites of action on brain vessels. In this study, we sought to identify the NPY receptors present in the human cerebrovascular bed. Specific Y1 receptor binding sites, localized on the smooth muscle of human pial vessels and potently competed by NPY, polypeptide YY (PYY), and the selective Y1 receptor antagonist BIBP 3226, were identified by quantitative radioautography of the Y1 radioligand [125I]_[Leu31, pro34]-PYY. In contrast, no specific binding of the Y2-([125I]-PYY3-36) and Y4/Y5-(125I-human pancreatic polypeptide [hPP]) radioligands could be detected. By in situ hybridization, expression of Y1 receptor mRNA was restricted to the smooth muscle layer of pial vessels, whereas no specific signals were detected for either Y2, Y4, or Y5 receptors. Similarly, using reverse transcriptase-polymerase chain reaction (RT-PCR), mRNA for Y1 but not Y2, Y4, or Y5 receptors was consistently detected in isolated human pial vessels, intracortical microvessels, and capillaries. In human brain microvascular cells in culture, PCR products for the Y1 receptors were exclusively found in the smooth muscle cells. In cultures of human brain astrocytes, a cell type that associates intimately with brain microvessels, PCR products for Y1, Y2, and Y4 but not Y5 receptors were identified. Finally, NPY significantly inhibited the forskolin-induced cAMP production in smooth muscle but not in endothelial cell cultures. We conclude that smooth muscle Y1 receptors are the primary if not exclusive NPY receptors associated with human brain extraparenchymal and intraparenchymal blood vessels, where they most likely mediate cerebral vasoconstriction.
Keywords
Neuropeptide Y (NPY) is one of the most widely distributed peptides in the brain (Adrian et al., 1983), and it is known to exert a variety of functions including the regulation of the cerebrovascular tone (Edvinsson et al., 1987). In both rat and human brain, NPY neurons and projection fibers have been shown to associate with local microvessels and their perivascular astrocytes, and could therefore affect brain perfusion (see Abounader and Hamel, 1997a, for additional references). In fact, intracarotid or local intraparenchymal (Suzuki et al., 1989; Tuor et al., 1990) injection of NPY in the rat is known to result in a strong reduction in CBF in the ipsilateral hemisphere or locally in the ipsilateral striatum and adjacent areas. These contractile in vivo effects are fully compatible with the ability of NPY to induce long-lasting and sustained vasoconstriction of isolated brain extraparenchymal (Edvinsson et al., 1987) and intraparenchymal (Dacey et al., 1988) blood vessels. However, a transient NPY -mediated cerebral vasodilation, presumably mediated by nitric oxide, also has been reported after intracarotid injection of NPY in the cat (Kobari et al., 1993).
In large cerebral arteries of various species including man, the NPY-induced contractile effect has been shown to be mediated by the NPY-Y1 receptor subtype (Abounader et al., 1995; Nilsson et al., 1996). In peripheral vascular beds from some species, however, pharmacologic studies suggest that Y1 and possibly Y2 receptors are involved in this contractile response (Grundemar et al., 1992; Tessel et al., 1993). Alternatively, Y2 receptors could mediate the vasodilatory effects of NPY either directly (Neild and Lewis, 1995) or indirectly through presynaptic regulation (Hashim and Tadepalli, 1995). In the cerebral microvascular bed, little is known on the types of NPY receptors present on these nonneuronal cells. Such information is crucial to a better understanding of the role of NPY in cerebrovascular physiology and, possibly, pathophysiology, as suggested by the marked increases in NPY mRNA expression and immunoreactivity after focal ischemia in rats (Cheung et al., 1995).
Six different NPY receptors (Y1, Y2, Y3, Y4, Y5, and Y6) have been described with different affinities for NPY, polypeptide YY (PYY), the pancreatic polypeptides (PP) and their derivatives (Wahlestedt et al., 1986; Dumont et al., 1995; Gerald et al., 1996; Weinberg et al., 1996). With the exception of the Y3 subtype, all receptors have been cloned (Herzog et al., 1992; Larhammar et al., 1992; Gerald et al., 1996; Rose et al., 1995; Bard et al., 1995; Weinberg et al., 1996). In cerebral arteries, a Y1 receptor has been unequivocally identified pharmacologically (Abounader et al., 1995) and by expression of its mRNA (Nilsson et al., 1996; Abounader and Hamel, 1997b); message for the Y4 receptor type has been detected only in human coronary artery (Bard et al., 1995).
In the current study, we used pharmacologic and molecular approaches to investigate the expression of NPY-Y1, NPY-Y2, NPY-Y4, and NPY-Y5 receptor messages and proteins in human pial vessels, intracortical blood vessels, and cerebromicrovascular and astroglial cells in culture. The results indicate that only the Y1 receptor is consistently expressed in human cerebrovascular tissues, and that it is exclusively localized to smooth muscle cells where NPY application is coupled to inhibition of adenylyl cyclase activity. Furthermore, they show the expression of Y1, Y2, and Y4 receptor types in human brain astroglial cells in culture. Part of these data has appeared in an abstract form (Abounader and Hamel, 1997b).
METHODS
Human cerebrovascular tissues
Human tissues were obtained with approval from the Institutional Research Ethics Committee from male and female individuals who either died from diseases not related to the CNS (n = 9, postmortem delay 2 to 18 hours; Brain Bank from the Douglas Hospital Research Centre, Verdun, QC, Canada) or were undergoing temporal lobe surgery at the Montreal Neurological Institute for the treatment of epilepsy (n = 6). Pial vessels (from postmortem brains) were dissected out and cleaned, and underlying cortical brain tissue then was immediately used for isolation of intraparenchymal microvessels (MV) and capillaries (CAP), as previously described (Linville and Hamel, 1995). Pial vessels, MV, and CAP were either frozen in cold isopentane (−45°C; for radioautography and in situ hybridization) or in liquid nitrogen (for reverse transcriptase-polymerase chain reaction [RT-PCR]) and stored at −80°C until use.
Cell cultures
Human pial vessel endothelial cells were obtained by enzymatic stripping of pial arteries. After removing intraluminal debris with Ca2+- and Mg2+-free phosphate-buffered saline, 0.5% trypsin was injected into the lumen and kept in place for 5 minutes. The intraluminal content then was recovered into a small volume of endothelial cell culturing media (65% medium M199 containing Earle's salts, 25 mmol/L Hepes, 4.35 g/L sodium bicarbonate, and 3 mmol/L
Cultures of human cerebromicrovascular smooth muscle (SMC) and endothelial (END) cells were generated from surgically removed brain tissue. The SMC cultures were derived from large intraparenchymal microvessels isolated by a sequential filtration of brain homogenate through 350- and 112-µm mesh screens. These resistance microvessels then were dislodged from the mesh screens with cold medium M199, washed, dissociated with the type IV collagenase (1 mg/mL, 15 minutes), and seeded onto 0.5% gelatin-coated culture plates in the medium containing 65% medium M199, 10% fetal calf serum, 5% human serum, 5 µg/mL insulin, 5 µg/mL transferrin, and 5 ng/mL selenium. After 4 to 5 weeks in culture, more than 85% of cells stained positively for the smooth muscle a-actin, whereas about 10% of cells demonstrated endothelial cell morphologic features and incorporated acetylated low density lipoprotein. Six SMC-enriched cultures derived from the microvessels harvested from the 350 µm (n = 4) and 112 µm (n = 2) mesh screens were used in the current study.
To initiate brain microvessel and capillary END cell cultures, brain homogenate depleted of large microvessels was centrifuged in 20% dextran (3000 g, 15 minutes), the pellet was resuspended in cold medium M199, and microvessels collected on a 20-µm mesh. The vessels were dissociated with the type IV collagenase, seeded, and maintained at 37°C in endothelial cell culturing media described earlier. The END cells were purified from the culture 4 to 5 days after seeding using cloning rings (BELLCO Glass, Inc., Vineland, NJ, U.S.A.). More than 95% of cells derived by these procedures were immunocytochemically positive for factor VIII-related antigen and expressed high levels of the cerebral endothelium-specific enzymes, γ-glutamyl-transpeptidase and alkaline phosphatase (Stanimirovic et al., 1996). The morphologic, phenotypic, biochemical, and functional characteristics of END cultures used in this study have been described in detail previously (Stanimirovic et al., 1996). The END cells (passages 3 to 7) from six different human brain biopsy specimens were used in this study.
Cultures of fetal (10 to 18 weeks of gestation) human brain astrocytes (AST) (n = 5) were provided by Drs. J. Antel and W. Yong (Montreal Neurological Institute, Montréal, QC, Canada) and prepared using established procedures (Yong et al., 1992). Brains were trypsinized (0.25%), homogenized, and filtered through a 130-µm mesh. Cell pellets were resuspended in a 95% Dulbecco's modified Eagle's medium (4500 g/L glucose) containing 5% fetal bovine serum, and plated onto poly-
Radioautography and in situ hybridization
Pial vessel cross-sections (20-µm thick) were cut on a cryostat, mounted either on gelatin-coated slides, and stored at −90°C until use (radioautography) or on clean RNAase-free slides (Superfrost plus, Fisherbrand) and stored at −20°C for no more than 24 hours (in situ hybridization).
Radioautography. For radioligand binding studies, sections were preincubated (60 minutes, room temperature) in 1.2 mmol/L Krebs-Ringer phosphate buffer (containing 0.12 mol/L NaCl, 4.7 mmol/L KCl, 2.5 mmol/L CaCl2, 1.2 mmol/L MgSO4, 11 mmol/L dextrose, and 25 mmol/L NaHCO3) and then incubated (2 hours, room temperature) in the same buffer supplemented with 0.1% bovine serum albumin and 0.05% bacitracin, and containing about 1.104 cpm/mL of either the Y1-(125I-[Leu31, Pro34]-PYY or 3H-BIBP 3226), Y2-(125I-PYY3-36), or Y4/Y5-(125I-hPP) radioligands. Incubations were carried out in the presence (nonspecific binding) or absence (total binding) of 1 µmol/L NPY. After washing (3 × 10 minutes) in 1.2 mmol/L Krebs-Ringer phosphate buffer, the sections were fixed in 3% glutaraldehyde in 0.1 mol/L phosphate buffer (phosphate-buffered saline, 10 minutes), washed, dehydrated, and air dried before being dipped in photographic emulsion (Kodak NTB-2; 1:2 with water, Photoservice, Montreal, Quebec, Canada) and exposed at 4°C for 7 to 30 days. The cellular distribution of Y1, Y2, or Y4/Y5 binding sites within the vessel wall was evaluated under light microscopic study on 0.5% cresyl violet-counterstained sections by comparing the specificity and location of the silver grains in sections incubated under total and nonspecific (10−6 mol/L NPY) conditions. For competition binding experiments, sections were incubated in the presence of increasing concentrations (10−10 to 10−6 mol/L) of either NPY, PYY, or the selective Y1 receptor antagonist BIBP 3226 in the case of Y1 sites labeled with125I-[Leu31, Pro34]-PYY; NPY for those labeled with the selective Y1 radioligand 3H-BIBP 3226; NPY, NPY13-36, or [Leu31, Pro34]-NPY for sections incubated with the Y2 radioligand 125I-PYY3-36; and NPY or human pancreatic polypeptide (hPP) for displacement of the Y4/Y5 sites labeled with 125I-hPP. After rinsing and dehydration, sections were directly apposed to Kodak XAR films and exposed for 2 to 15 days (4°C). Film radioautograms were used for quantitative densitometry performed on a microcomputer-based image analysis system (The Image Calculator, Soquelec, Ltd., Montréal, QC, Canada). The pIC50 values (mean ± SD, n = 2 or 3 different experiments) for each agonist or antagonist were determined from the competition curves, and the total binding was set at 100%.
In situ hybridization. Pial vessel sections were thawed, fixed (5 minutes in 2% paraformaldehyde in 4 × standard saline citrate [SSC]) and washed (3 × 5 minutes in 4 × SSC) before immersion (10 minutes) in 0.1 mol/L triethanolamine containing 0.25% acetic anhydride. The dried sections were incubated (overnight in a humidified chamber at 56°C) in hybridization buffer (4 × SSC, 50% formamide, 10% dextran sulfate, 1 mol/L dithiothreitol, 1 × Denhardt's solution, 500 µg/mL denatured salmon sperm DNA, and 250 µg/mL yeast tRNA) containing approximately 5.106 cpm/mL of [35S]-labeled cRNA probes (200 µL of probe per section). Adjacent sections were used for incubation with sense and antisense cRNA probes transcribed from cDNA complementary to the full coding region of the human Y1, Y2, Y4, and Y5 receptors (Larhammar et al., 1992; Rose et al., 1995; Bard et al., 1995; Gerald et al., 1996). The cDNA was subcloned in PGEM plasmids (constructed and kindly provided by Dr. Y. Tong at the Douglas Hospital Research Centre, Verdun, QC, Canada). The plasmids were linearized, antisense and sense cRNA probes transcribed (60 minutes at 37°C) in 20 µL of 1 × transcription buffer (40 mmol/L Tris-HCl, 6 mmol/L MgCl2, 2 mmol/L spermidine, and 10 mmol/L NaCl; pH = 7.9) containing 20 mmol/L dithiothreitol; 10 units of RNAsin (Promega); 2 mmol/L ATP, γ-glutamyl-transpeptidase, and CTP; 2.5 mmol/L [35S]-UTP; 1 µg linearized plasmid template; and 10 units of SP6, T7, or T3 RNA polymerase, depending on the cloning orientation in the respective plasmid. The cDNA template was removed by incubation (15 minutes, 37°C) with 10 units of RNAase-free RQ1-DNAase (Promega). The probes were purified by phenol/chloroform extraction and precipitated (−90°C, 60 minutes) in ethanol before resuspension of the pellet in 50 µL of RNAase-free deionized water for addition in hybridization buffer. After hybridization, the sections were subjected to high-stringency washes (4 × SSC/50% formamide, 30 minutes, 65°C and then 4 × SSC, 2 × 30 minutes at 65°C) and the remaining unhybridized probes removed with RNAase A (10 µg/mL) in 2 × SSC (30 minutes, 37°C). The sections then were washed (2 hours, 65°C in 0.5 × SSC), cleared in 70% ethanol, dried, and dipped into Kodak NTB-2 emulsion and exposed (4°C, 4 to 30 days). After development in Kodak D19, the sections were stained with 0.5% cresyl violet and observed under a Leitz Aristoplan microscope. Expression of NPY receptor mRNA was evaluated by comparing the density and location of silver grains between antisense- and sense-treated sections.
Reverse transcriptase-polymerase chain reaction
Frozen pial vessels, MV, and CAP were first powdered on dry ice whereas cell cultures were used directly for total RNA extraction, after homogenization in TRIzol Reagent (Gibco BRL, Gaithersburg, MD, U.S.A.). The suspension was treated with RQ1-DNAase (3U, Gibco BRL) and cDNA synthesized from 5 to 30 µg of total RNA (5- to 10-µg cells, 20- to 30-µg tissues) using random primers (200 ng/µg RNA, Gibco BRL) and avian myeloblastosis virus reverse transcriptase (Promega, Madison, WI, U.S.A.) (1 hour at 42°C). cDNA prepared from human cerebral cortex obtained at surgery was included in all polymerase chain reaction (PCR) studies as positive controls. The PCR amplification was performed in an MJ Research minicycler (Fisher Scientific, Montréal, Quebec, Canada) under the following conditions: 2 minutes at 95°C, 5 minutes at 72°C, 40 seconds at 95°C, 40 seconds at 56° to 58°C (depending on the primer used), and 40 seconds at 72°C for 37 cycles, followed by a 5-minute final extension at 72°C. The reaction mixture contained 3 to 5 µL of cDNA, 2 U of Taq DNA polymerase, and 0.5 µmol/L of each primer in a total volume of 50 µL. Specific oliginucleotide primers for the human Y1, Y2, Y4, and Y5 receptor genes were designed (NBI Oligo 5.0) according to published sequences (Larhammar et al., 1992; Rose et al., 1995; Bard et al., 1995; Gerald et al., 1996). They were synthesized in an Applied Biosystems synthesizer and purified using an OPC column (Applied Biosystems). Some oligonucleotide primers were flanked with the restriction enzyme EcoR1 (GAATTC) and BamH1 (GGATCC) sequences. The primers were as follows: Y1 (Genbank accession #M88461), Y1-Forward: 5ʹ-TCAGGATCCAACATCCTGATTGTGAAAC-3ʹ and Y1-Reverse: 5ʹ-TACTGCAGCACCAAGAGGAG-3ʹ, yielding a PCR product of 435 base pairs; Y2: (Genbank accession #36269), Y2-Forward: 5ʹ-TGAATTCATATTGGCCTACTGCTCC-3ʹ and Y2-Reverse: 5ʹ-CTGGATCCTGCCATAGATGCTCTTCTCC-3ʹ, PCR product of 492 base pairs; Y4 (Genbank accession #U35232), Y4-Forward: 5ʹ-TGAATTCTTCATCGTCACTTCCTACAGC-3ʹ and Y4-Reverse: CTGGATCCTAACAGACCAGGATGAAGCC-3ʹ with PCR product of 575 base pairs; and Y5 (Genbank accession #U56079), Y5-Forward: 5ʹ-TGAATTCGACACTAGGTTTTGCCATCTG-3ʹ and Y5-Reverse: 5ʹ-CTGGATCCGGACCCCTGGTATGAACTTA-3ʹ, yielding a PCR product of 510 base pairs. The PCR products were size-fractionated by electrophoresis on a 1.5% Tris/borate/ethylenediamine-tetraacetic acid agarose gel containing ethidium bromide, their size confirmed, and then photographed under ultraviolet light. Representative PCR fragments for the Y1 (SMC), Y2, and Y4 (AST) receptors were used for sequencing analysis using direct automated fluorescent sequencing (W.N. Keck Foundation Biotechnology Resource Laboratory, Yale University, New Haven, CT, U.S.A.).
Second messenger assays
The presence of functional NPY receptor proteins in microvascular cells was evaluated by measuring the ability of NPY to affect cAMP production in cultured human brain SMC (n = 4) and END (n = 3) cells, a pathway previously correlated to the vasoconstrictor activity of NPY (Lundberg et al., 1988). For comparison, one preparation of endothelial cells derived from human pial vessels also was used. Cells grown to confluence in 24-well tissue culture plates were incubated (15 minutes, 37°C) in phosphate-buffered saline containing 0.2% bovine serum albumin and 1 mmol/L 3-isobutyl-1-methyl-xanthine with NPY (0.1 and/or 1 µmol/L) either alone or in the presence of 1 µmol/L forskolin. The reaction was stopped the double extraction with 65% (v/v) ice-cold ethanol. The extracts were combined, dried (vacuum oven at 80°C) and dissolved in 200 µL of the assay buffer for measurement of cAMP levels with a commercial enzyme immunoassay kit (Biotrak, Amersham). The cell pellets were solubilized in 0.1 N NaOH and used for protein measurement by the method of Lowry and coworkers (1951). Statistical differences (P ≤ 0.05) were evaluated by Student's t test.
RESULTS
Localization of neuropeptide Y receptor mRNA in human pial vessels
As shown in Fig. 1, sections incubated with a selective Y1 receptor [35S]-labeled cRNA antisense probe exhibited a high density of silver grains, located primarily if not exclusively on the smooth muscle layer of human pial vessels (Fig. 1A). Although silver grains could accumulate at the border of the elastic lamina (Fig. 1A, arrow), endothelial cells remain unlabeled. No specific signal was observed in sections incubated with either the Y1 sense riboprobe (Fig. 1B) or the sense and antisense probes for the Y2, Y4, and Y5 receptor types (Figs. 1C through H).
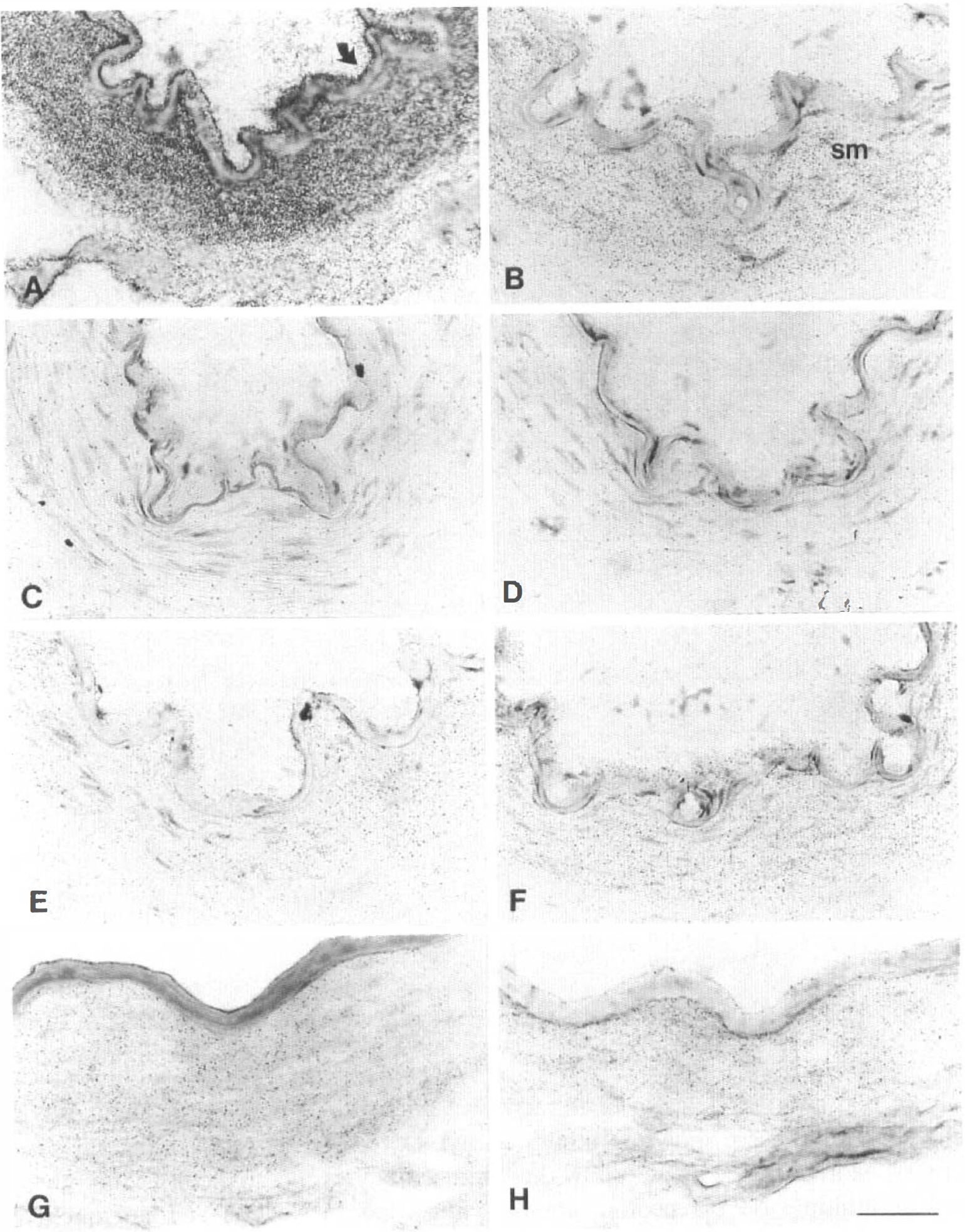
Photomicrographs of emulsion-dipped sections of human pial vessels incubated with antisense (left panels) or sense (right panels) cRNA probes complementary to the coding sequence of the human neuropeptide Y (NPY)-Y1
Localization of neuropeptide Y receptor proteins in human pial vessels
In sections incubated with the Y1 radioligand 125I-[Leu31,Pro34]-PYY, a high level of specific binding sites (reflecting the presence of Y1 receptor protein) was observed in the smooth muscle layer, as evidenced by the difference in silver grain density in sections incubated under total and nonspecific conditions (Figs. 2A and B). In contrast, incubation with the Y2-(125 I-PYY3-36) and Y4/Y5-(125I-hPP) receptor ligands yielded no specific binding in any of the cellular compartments of the human pial vessels, as shown by the similar distribution and low density of silver grains in sections incubated under both total and nonspecific binding conditions (Figs. 2C through F).
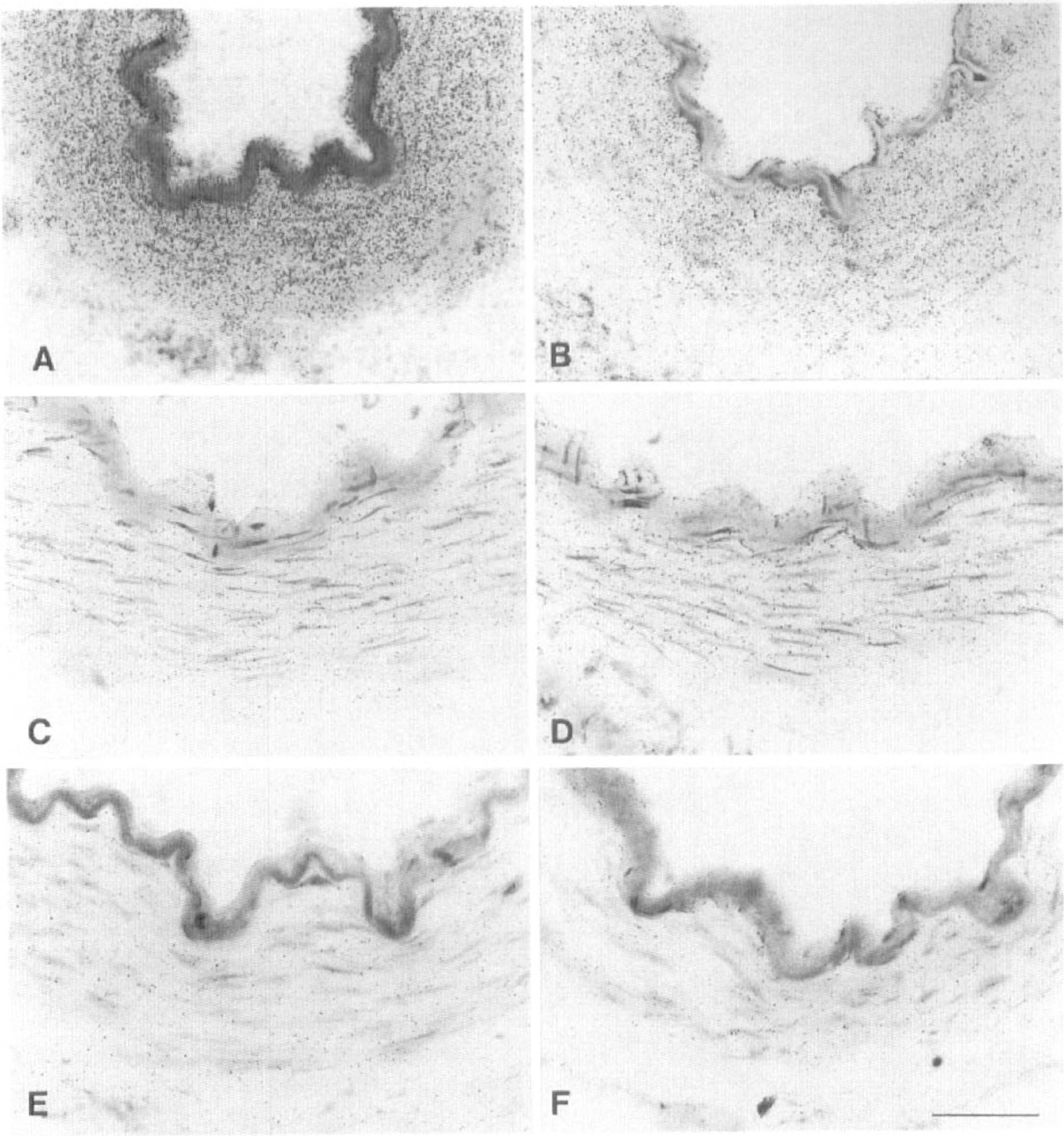
Photomicrographs of emulsion-dipped human pial vessel sections incubated with radioligands for the NPY-Y1 (125I-[Leu31, Pro34]-PYY)
Quantitative densitometry of film radioautograms from human pial vessel sections incubated with the Y1 radioligand 125I-[Leu31, Pro34]-PYY showed competition of the bound ligand from the muscle layer by increasing concentrations of unlabeled NPY, PYY, and the selective Y1 receptor antagonist BIBP 3226 (Fig. 3A). Their respective pIC50 values were of 7.86 ± 0.06, 7.43 ± 0.04, and 8.42 ± 0.21 (mean ± SD). Nonspecific binding (evaluated in the presence of 10−6 mol/L of NPY, PYY, or BIBP, respectively) corresponded to about 25% to 35% of total binding (Fig. 3A). The Y1 radioligand 3H-BIBP 3226 was competed from the pial vessel sections in a concentration-dependent manner by NPY with a calculated pIC50 value of 8.01 ± 0.01 (mean ± SD) (Fig. 3B). In contrast, film densitometry showed no significant difference in the density of binding sites between sections incubated with the radioligand alone or in the presence of various concentrations (10−9 to 10−6 mol/L) of either NPY, [Leu31-Pro34]-NPY, or NPY13-36 for Y2-(125I-PYY3-36) sites, or hPP (10−7 to 10−10 mol/L) in the case of Y4/Y5-(125I-hPP) binding sites.
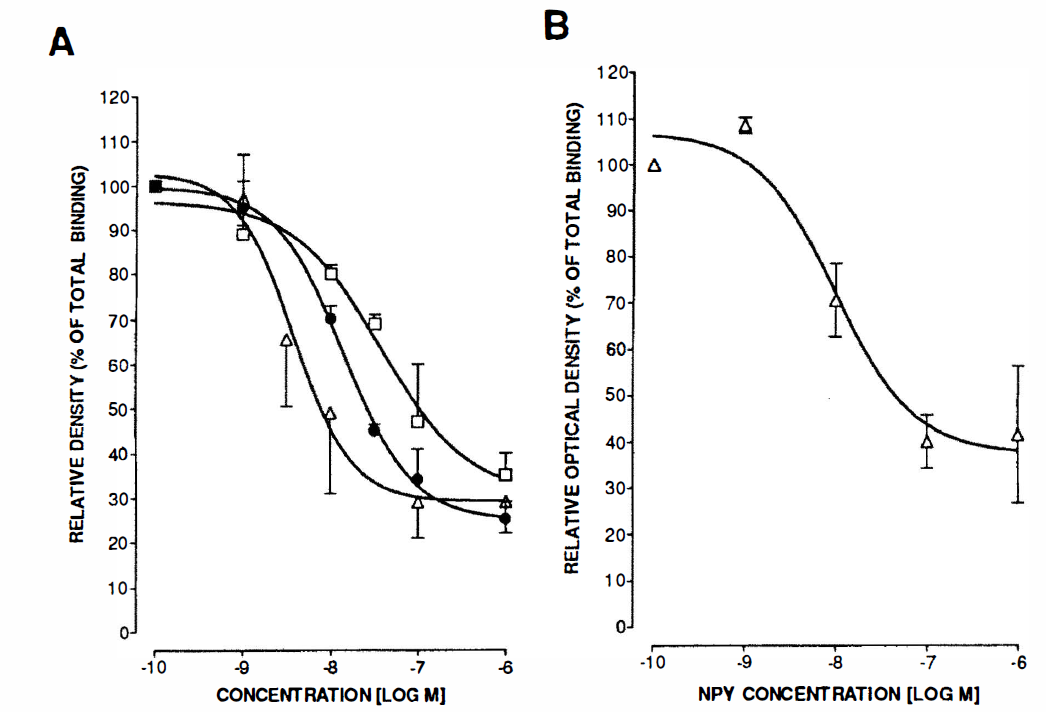
Competition of 125I-[Leu31-Pro34]-PYY–labeled smooth muscle specific binding sites by increasing concentrations (10−10 to 10−6 mol/L) of NPY (•), PYY (□), or the selective Y1 receptor antagonist BIBP 3226 (Δ)
Neuropeptide Y receptors expression in cerebrovascular tissues and microvascular and astroglial cells
In agreement with radioautographic and in situ hybridization experiments, specific PCR products of the expected size (435 bp) for the Y1 receptor were detected in most (67% to 100%) human pial vessels, isolated MV, and CAP, as well as in most SMC and AST cell cultures (Fig. 4, top panel). However, no amplification of Y1 receptor message could be evidenced in endothelial cells derived from pial vessels or in any of the cerebromicrovascular END cell cultures tested (Fig. 4, top panel). Message for the Y2 receptor was detected in a few (approximately 20%) of vascular preparations and a single culture of microvascular endothelial cells. Messages for the Y4 and Y5 receptors could not be detected in any of the vascular tissues, END, or SMC cultures (Fig. 4). However, PCR products of the expected size for the Y2 (491 bp) and Y4 (578 bp) receptors were detected by gel electrophoresis in most (80% and 67%, respectively) of AST cells in culture (Fig. 4, middle and bottom panels). Sequence analysis of the selected PCR products matched the published sequences for their respective cloned human Y1 (99.8%), Y2 (99%), and Y4 (99.5%) NPY receptors.
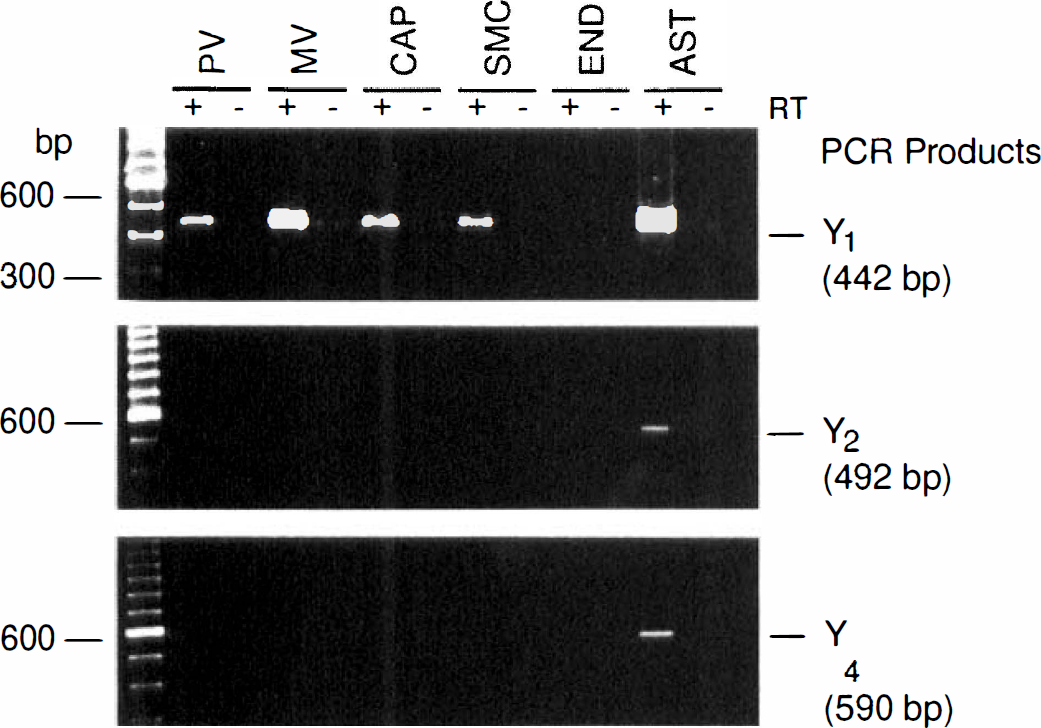
Gel electrophoresis of polymerase chain reaction (PCR) products for the NPY-Y1, NPY-Y2, and NPY-Y4 receptors amplified from reverse transcribed DNA (+) from human pial vessels (PV), isolated intracortical microvessels (MV) and capillaries (CAP), as well as from human microvascular smooth muscle (SMC), endothelial (END), and astroglial (AST) cells in culture. Expression of the Y1 receptor was detected in every segment of the human cerebrovascular bed and cells in culture except for the END cells. Expression of Y5 receptor mRNA could not be detected in any vascular or astroglial preparations, despite a positive and specific amplification in human cerebral cortex (not shown). Samples without reverse transcriptase were included (–) to monitor for background amplification and possible contamination.
Functional neuropeptide Y receptors in microvascular cell cultures
As shown in Fig. 5, functional NPY receptor proteins were detected in the muscular compartment of cerebral microvessels. The basal release of cAMP was slightly inhibited by NPY in SMC (about 30%, not significant) but not in END cells. Forskolin (1 µmol/L) resulted in a 10- to 20-fold increase in cAMP production in these cultures (Fig. 5). The NPY (0.1 and 1 µmol/L) significantly (P < 0.05) inhibited the forskolin-stimulated cAMP production in SMC (40% and 51%, respectively), whereas a 1-µmol/L NPY concentration had no effect on the evoked cAMP production in microvascular END cell cultures (Fig. 5) or in endothelial cells derived from human pial vessels (data not shown).
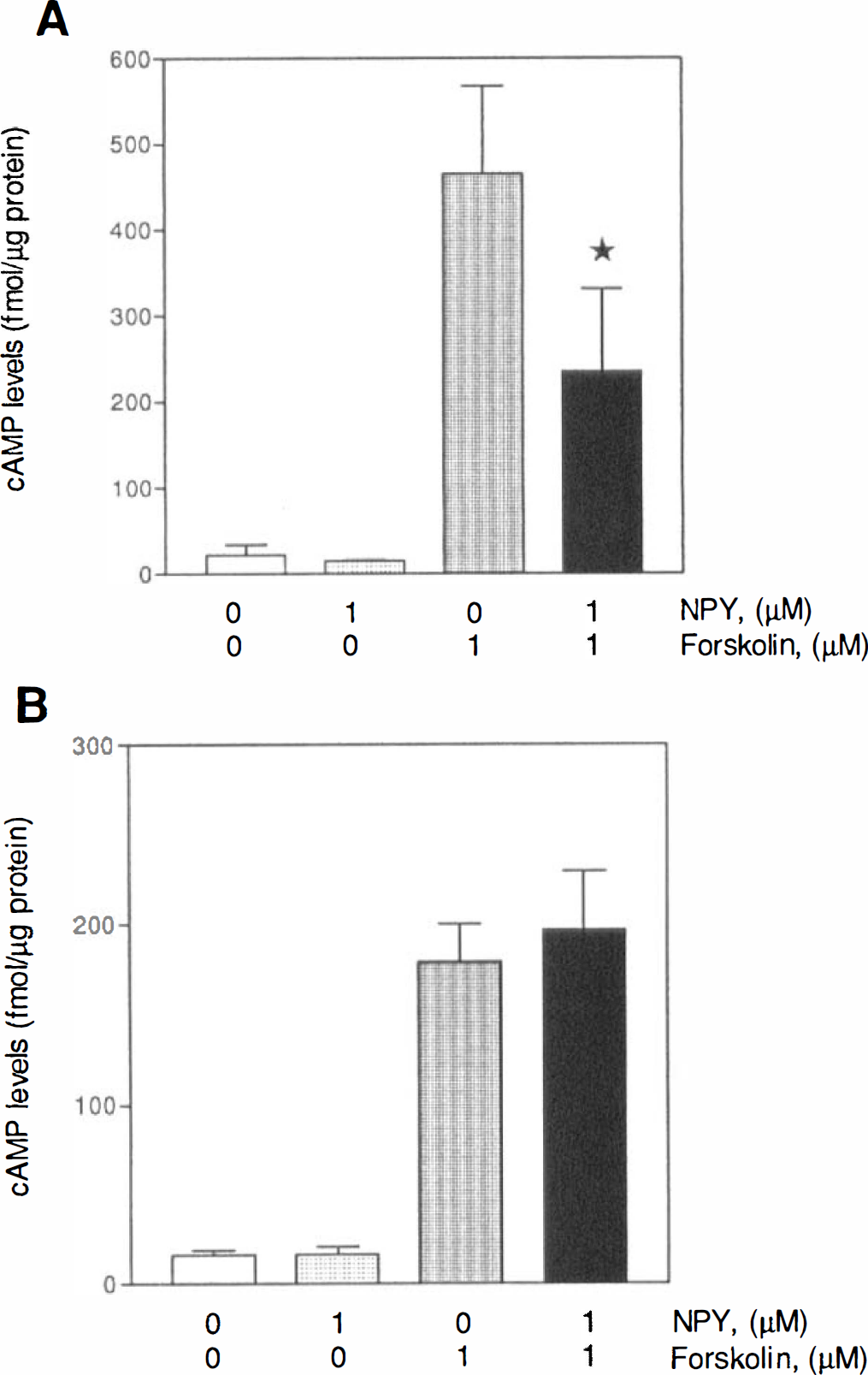
Effects of NPY on the basal and forskolin-stimulated production of cAMP in microvascular SMC
DISCUSSION
This study provides the first comprehensive analysis of NPY receptors message and protein distribution, as well as cellular localization in human cerebral blood vessels. The results obtained by both pharmacologic and molecular approaches suggest that the Y1 receptor subtype is the only receptor, of the known and cloned human NPY receptors, to be significantly expressed in human brain vessels, irrespective of their extraparenchymal or intraparenchymal location. These NPY receptors are exclusively located on the smooth muscle cell layer not only of pial arteries, but also of intracortical microvessels, where they were shown to inhibit adenylyl cyclase activity. Moreover, the results show that brain astrocytes express multiple NPY receptor subtypes, identified as Y1, Y2, and Y4 by RT-PCR. Taken together, these data indicate that NPY has the ability to influence nonneuronal cells within the human brain by stimulating vascular Y1 and heterogeneous astroglial NPY receptors and, as such, could participate in the regulation of brain perfusion and metabolic or ionic homeostasis.
The identification of Y1 receptors in human pial vessels by in situ hybridization and radioautography agrees with previous pharmacologic and RT-PCR studies (Abounader et al., 1995; Nilsson et al., 1996). However, the results further indicate that the Y1 receptor is the only NPY receptor associated with human cerebral arteries, and that it has an exclusive muscular localization. This localization concurs with a recent immunocytochemical study of cerebrovascular Y1 receptors in the rat (Bao et al., 1997). The Y1 nature of the human cerebrovascular NPY receptor is pharmacologically supported by the potency of NPY, PYY, and the Y1 receptor antagonist BIBP 3226 (respective pIC50 values of 7.86, 7.43, and 8.42) in competing for 125I-[Leu31-Pro34]-PYY–labeled specific smooth muscle binding sites. These compare well with the affinities reported for these compounds at the cerebrovascular Y1 receptor mediating contraction of human cerebral arteries (Abounader and Hamel, 1995; Nilsson et al., 1996). In addition to vasoconstriction, other roles for cerebrovascular smooth muscle Y1 receptors could be considered, such as mediation of mitogenesis, as reported in peripheral vascular smooth muscle cells (Zukowska-Grojec et al., 1993).
The results suggest that human cerebral arteries differ from some peripheral (femoral and intestinal) vascular beds in animal species in which both muscular Y1 and Y2 receptors have been implicated in the control of arterial or arteriolar vascular tone (Tessel et al., 1993; Neild and Lewis, 1995). Indeed, no evidence was obtained from the current molecular or anatomical studies for the presence of smooth muscle Y2 receptors in human brain vessels. Whereas the Y3 receptor also has been pharmacologically excluded as the mediator of the NPY-induced human cerebral vasoconstriction (Abounader and Hamel, 1995), additional work is required to verify its status at the cerebrovascular level when the human clone becomes available. Based on the current molecular and radioligand binding data, it is unlikely that Y4 and Y5 receptors exist in the human cerebrovascular bed in any significant amount.
A novel finding from the current study is that of a selective expression of Y1 receptor mRNA in the human brain microvascular bed, more specifically, at the level of the smooth muscle cells where the presence of functional receptor proteins was confirmed by the ability of NPY to inhibit cAMP production. Such association of microvascular Y1 receptors with the smooth muscle cell layer of human intracortical microvessels is fully compatible with the recent immnocytochemical demonstration of Y1 receptor in rat brain microarterioles (Bao et al., 1997) and with previous reports in rodents suggesting that Y1 receptors mediate the endothelium-independent NPY vasoconstriction in the microcirculation, albeit of peripheral origin (Kim et al., 1994). The presence of smooth muscle Y1 receptors in human intracortical microvessels strongly suggests that perivascular NPY nerve terminals (Abounader and Hamel, 1997a) could induce local cerebral vasoconstriction, a hypothesis supported by previous observations in the rat of a local decrease in CBF after intraparenchymal application of NPY (Tuor et al., 1990) and of an NPY-induced contractile response in isolated intracortical arterioles (Dacey et al., 1988). The ability of NPY to inhibit the forskolin-stimulated increase in cAMP levels in human cerebromicrovascular SMC cells, a signaling pathway associated with a direct vasocontractile response to NPY (Lundberg et al., 1988; Larhammar et al., 1992), further supports such a role for NPY at the microvascular level.
The failure to detect NPY receptors in a significant proportion of endothelial cells further suggests that muscular Y1 receptors are the primary if not exclusive mediators of the NPY-induced vascular responses in human brain vessels located both outside and within the cortical mantle. Interestingly, in endothelial cells from human umbilical vein, the Y1 receptor mRNA is only detectable during cell differentiation (Karwatoska-Prokopezuk et al., 1996). Therefore, although neither the NPY-Y1 receptor's message nor protein were detected in any preparation of human brain endothelial cells, it cannot be excluded that these receptors can be induced during development or by certain physiologic conditions.
In addition to being abundantly expressed in human brain cells, the current study shows that Y1 receptors appear to be an integral and functional component of brain vascular and astroglial cells. This distribution contrasts with that of the other cloned NPY receptors, which were either never present (Y4, Y5) or detected in only a few (Y2) cerebrovascular tissues or microvascular endothelial cell cultures. The detection of a Y2 message in isolated microvascular fractions is likely attributable to the associated perivascular astrocytes (see later). The apparent absence of endothelial NPY receptors is consistent with previous studies in nonhuman vessels, both in peripheral arteries (Pernow and Lunberg, 1988) and microvessels (Kim et al., 1994). However, it cannot be excluded that Y2 receptors could regulate vascular functions through receptors located presynaptically on nerve terminals, as documented in the peripheral circulation (Hashim and Tadepalli, 1995) or, alternatively, on astroglial cells, a cell type that is intimately associated with the microvascular bed.
We observed expression of multiple subtypes of NPY receptors (i.e., Y1, Y2, and Y4 receptors) in fetal human brain astrocytes. The presence of astroglial NPY receptors had been reported previously in the rat, either by identification of specific binding sites for NPY in cultures of rat brain astrocytes or by the observation of a membrane potential depolarization and an increase in [Ca2+]i after application of NPY (Hosli and Hosli, 1993; Gimpl et al., 1993). The possibility that these receptors are still expressed and functional in adult human brain deserves further investigation. Since astroglial cells are an integral component of isolated MV and CAP, the relative or complete absence of PCR products for the Y2 or Y4 receptor in adult human isolated MV and CAP suggests that these receptors are expressed at low levels in adult astrocytes or, alternatively, that their primary location is not the perivascular astrocytes.
In conclusion, the current study demonstrates the existence of Y1 receptors not only on smooth muscle of human cerebral arteries, but also of intracortical microvessels. In view of the potent effect of NPY on normal brain perfusion and the documented increase in brain NPY levels or expression in focal ischemia (Cheung et al., 1995), the exclusive presence of NPY-Y1 receptor on human brain myocytes may offer a unique opportunity to target the cerebrovascular system. Moreover, these data underscore the possibility that NPY released from human brain neurons can, through vascular smooth muscle Y1 receptors, directly influence cerebrovascular functions related to regulation of local cerebral perfusion. They also suggest that NPY interactions could occur through astroglial receptors, which may be involved not only in the control of microvascular functions, but also of brain metabolic and ionic homeostasis, gliosis, and recovery after injury (Zoli et al., 1997).
Footnotes
Acknowledgments
The authors thank Drs. Danielle Jacques, Yvan Dumont, and Yiai Tong, Douglas Hospital Research Centre, Verdun, Quebec, Canada, for invaluable technical advice, the iodinated radioligands, and the plasmids with human NPY receptor cDNAs; Dr. Henri N. Doods, Dr. Karl Thomae GmbH, Biberach, Germany, for 3H-BIBP 3226, NPY, [Leu31-Pro34]-NPY, PYY, and BIBP 3226; Drs. Alain Fournier and Serge St-Pierre, INRS-Santé, Pointe-Claire, Quebec, Canada for NPY13-36 and hPP; and Dr. Philippe Séguéla, Montréal Neurological Institute, Montréal, Quebec, Canada for the design of some PCR primers. Finally, the authors thank Ms. Linda Michel for preparation of the manuscript.
