Abstract
The enzyme argininosuccinate synthetase (ASS) initiates the metabolic pathway leading from L-citrulline to L-arginine, the only physiological substrate of all isoforms of nitric oxide synthases. The presence of ASS in glial cells in vivo was investigated by immunohistochemical methods in a model of rat brain inflammation. Phosphate-buffered saline or a mixture of bacterial lipopolysaccharide and interferon-γ was injected into the left striatum, and animals were killed 24 hours later. Ipsilateral and contralateral sides of brain sections were incubated with an antiserum against ASS or antibodies against cell-specific markers. In the three areas examined, striatum, corpus callosum, and cortex, a strong induction of ASS immunoreactivity was observed in glial cells after injection of immunostimulants. A detailed quantitative analysis of double-stained sections revealed that ASS was almost exclusively expressed in reactive, ED1-positive microglial cells/brain macrophages in immunostimulant- or sham-injected ipsilateral sides of the sections. Furthermore, ASS/ED1 costaining was observed in perivascular cells. Colocalization of ASS with astroglial marker glial fibrillary acidic protein was given only occasionally after immunostimulation. ASS-positive neurons were detected in control and experimental animals; staining intensity was comparable in both cases. The results suggest that neurons express ASS constitutively, whereas the enzyme is induced in glial cells in response to proinflammatory stimuli. This finding is the first demonstration of an induction of a pathway auxiliary to generation of nitric oxide in brain in response to immunostimulants and provides new insight into neural arginine metabolism.
The physiological importance of nitric oxide, NO, in nervous tissue is no longer debated. NO may act as a signaling molecule or may be part of a cytotoxic defense mechanism when generated in large amounts during inflammation or related processes (Hibbs et al., 1987; Stuehr and Nathan, 1989; Moncada et al., 1991). Enzyme-derived cellular production of NO is dependent on the presence of L-arginine, the only known substrate of all isoforms of nitric oxide synthase (NOS). Adequate supply of L-arginine may be secured by uptake of the amino acid from the extracellular space or by recycling from the NOS coproduct L-citrulline through the combined action of the urea cycle enzymes, argininosuccinate synthetase (ASS), and argininosuccinate lyase. In rat brain, astrocytes appear to be particularly enriched in L-arginine–like immunoreactivity (Aoki et al., 1991), and arginine is released from glial cells on stimulation with glutamate (Grima et al., 1997). ASS activity has been detected in primary glial cells (Jackson et al., 1996). Transport of L-arginine in cultured rodent astroglial cells is upregulated concomittantly with the increase of NO synthesis after induction of NOS by immunostimulants such as bacterial lipopolysaccharide (LPS) or interferon-γ (IFNγ) (Schmidlin and Wiesinger, 1995). Under the same proinflammatory conditions, expression of ASS is upregulated in glioma cells as well as in mixed glial and pure astroglial cultures and a functional role of the enzyme in recycling of L-citrulline for the generation of glial NO has been shown (Schmidlin et al., 1997; Schmidlin and Wiesinger, 1998).
In contrast to the results obtained in cell culture, ASS immunoreactivity in rat brain sections has been assigned almost exclusively to neurons under physiological conditions (Nakamura et al., 1991; Amt-Ramos et al., 1992). It may be reasoned that in nonchallenged animals ASS in glial cells is expressed at a low basal level not detectable by the immunohistochemical methods used. The objective of the present study was to investigate if expression of glial ASS is upregulated in vivo under proinflammatory conditions. Injection of LPS and IFNγ into the striatum of rats induced strong ASS immunoreactivity in several glial populations with the most prominent staining in ED1-positive microgliaibrain macrophages and perivascular cells. These results show for the first time that in central nervous tissue in vivo high-output production of NO is accompanied by the upregulation of the biochemical pathway which recycles L-citrulline to the NOS substrate L-arginine in response to immunostimulants.
MATERIALS AND METHODS
Materials
Phosphate-buffered saline (PBS) was purchased from GIBCO Life Technologies (Karlsruhe, Germany). Pentobarbital, LPS (E. coli, 026:B6) and mouse recombinant IFNγ were from Sigma (Deisenhofen, Germany). Acetic acid, formaldehyde, methanol, and all other chemicals were of analytical grade and purchased from E. Merck (Darmstadt, Germany). The generation of the antiserum against peptide sequence 196-222 of mouse liver ASS has been described elsewhere (Schmidlin et al., 1997).
Animals
Male Wistar rats (Interfauna, Tuttlingen, Germany) weighing 250 to 300 g, were housed in groups of four under standard conditions at a temperature of 22°C (± 1°C) and a 12-hour light-dark cycle (light from 7.00–19.00) with free access to standard food pellets (Altromin, Soest, Germany) and tap water.
Animal procedures
Rats (n = 6) were anesthetized using pentobarbital (50 mg/kg, dissolved in 0.9% sodium chloride; intraperitoneal) and placed in a stereotaxic frame. A mixture of LPS (1 µg/µL) and IFNγ (10 U/µL) dissolved in 1µL of PBS (pH 7.4) was injected over 60 seconds into the left striatum using a 5 µL Hamilton syringe, at AP 0.0, L −2.5, V 5.5 mm relative to bregma according to the stereotaxic atlas from König and Klippel (Konig and Klippel, 1963). Control animals (n = 6) received an injection of 1µL PBS. The needle was left in place for a further 5 minutes to prevent reflux up the needle tract. To maintain constant body temperature, animals were placed under a heating lamp until complete recovery from anesthesia. Twenty-four hours after intrastriatal challenge with LPS/IFNγ, animals were killed by an overdose of pentobarbital and then perfused transcardially with 200 mL heparinized sodium chloride (0.9%) and subsequently with 200 mL fixative containing 10% formaldehyde, 10% acetic acid, and 80% methanol. Brains were removed and immersed in fixative for 72 hours at room temperature, followed by paraffin embedding using standard protocols. All experiments were performed in accordance with the declaration of Helsinki and the animal welfare guidelines and laws of the Federal Republic of Germany and were approved by the local ethical committee for animal experiments.
Processing of brain tissue and immunohistochemistry
Serial coronal sections of the whole brain were cut 8 µm thick using a Leitz microtome and mounted on poly-L-lysine–coated slides. The following antibodies were used in the respective diluton: (1) Antiserum directed against the peptide sequence 196–222 of mouse liver ASS (1:250 [Schmidlin et al., 1997]); (2) Monoclonal antibody raised against glial fibrillary acid protein (GFAP) of astrocytes (1: 100, MCA 363, Serotec, Darmstadt, Germany); and (3) ED1, a monoclonal antibody raised against rat lysosomal membrane antigen of activated macrophage/microglia (1:500, MCA 341, Serotec, Darmstadt, Germany). For detection, slides were immersed in 10 mmol/L citrate buffer (pH 6.0) and heated in a microwave oven, four cycles of 5 minutes each, to unmask antigen sites. Thereafter, slides were removed and cooled for 20 minutes at room temperature before washing in PBS. Endogenous peroxidase activity was inhibited by rinsing slides in 0.1 % hydrogen peroxide for 10 minutes. After washing in PBS (pH 7.4), nonspecific binding was prevented by incubating sections in blocking solution containing 10% normal goat serum in PBS for 1 hour at room temperature. After washing in PBS, sections were incubated overnight or for 24 hours at 4 °C with antiserum (1). Thereafter, the primary antibody was removed and sections were washed intensively with PBS followed by incubation with a biotinylated anti-rabbit immunoglobulin G (IgG) (1:200, Vector Laboratories) for 30 minutes at room temperature. Immunohistochemical localization was performed using an avidin-biotinylated peroxidase complex method (Vecstatin ABC-Kit, Vector Laboratories, Burlingame, USA) with 3,3'-diaminobenzidine (DAB, Sigma, Germany) as peroxidase substrate. For costaining with a second antibody, slides were washed twice in PBS and blocked with 10% normal horse serum in the following. After incubation with the primary antibody (2) or (3) for 30 minutes and 1 hour, respectively, slides were washed in PBS and incubated with a biotinylated horse anti-mouse IgG. Immunohistochemical localization was detected as described above using VIP as substrate (Vector VIP substrate kit, Vector Laboratories, Burlingame, USA). Immunostained sections were counterstained with hemalaun Mayer, dehydrated, and cleared with xylene before mounting in DePeX (Fluka, Buchs, Switzerland). Coronal sections were examined using a Leitz Aristoplan microscope.
Quantification of immunohistochemistry and statistical analysis
Quantitative evaluation for ASS, ED1 antigen, and GFAP was performed in six animals injected with LPS/IFNγ and in six control animals. Immunoreactivities were detected on three sections with a defined distance to the level of intrastriatal injection. The sections chosen were as follows: the middle section was taken at the level of injection, with the injection site discernible. The other two sections were taken at the distance of 40 µm rostral and caudal of the injection site. Three different areas were defined and evaluated: striatum, corpus callosum and cortex ipsilateral and contralateral to the injection side. In each area three different fields were counted (Fig. 1). The number of cells within the respective fields was counted by using a counting grid. Data are shown as mean ± SD. Differences between LPS/IFNγ- and PBS-injected animals at the site of injection and at the contralateral site were assessed by one way analysis of variance followed by a Tukey test (SYSTAT, Evanston, USA).
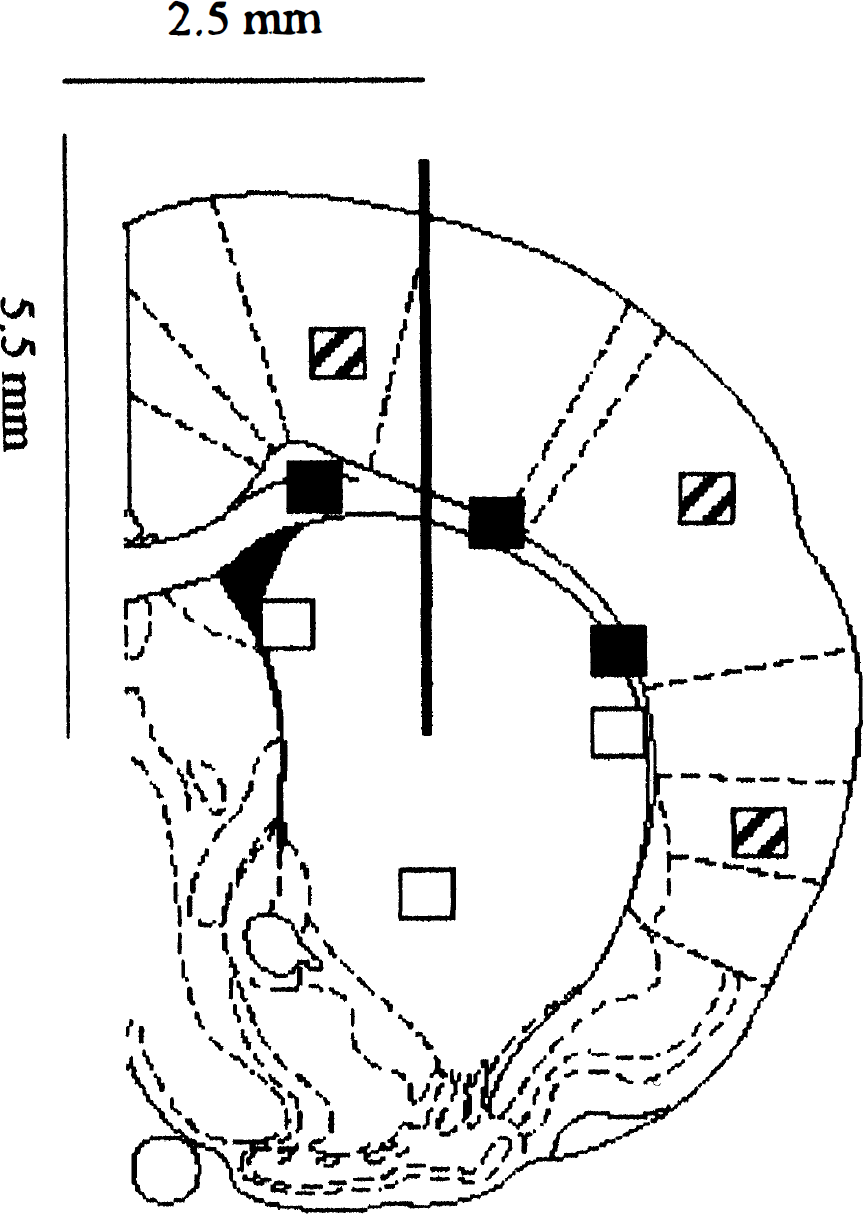
Areas and fields of counting. Immunoreactivities were detected on three sections with a defined distance to the level of intrastriatal injection. The sections chosen were as follows: the middle section was taken at the level of injection, with the injection site discernible at AP 0.0, L −2.5, V 5.5 mm relative to bregma according to the stereotaxic atlas of König and Klippel (1963). The other two sections were taken at the distance of 40 µm rostral and caudal of the injection site. Three different areas were defined and evaluated: Striatum (□), corpus callosum (▪) and cortex (▪) ipsilateral and contralateral to the injection site. In each area three different fields were counted.
RESULTS
To investigate the degree of participation in the response to intrastriatal injection of immunostimulants, three different brain areas (striatum, corpus callosum and cortex) were evaluated for ASS, ED1 antigen, and GFAP immunoreactivity (Fig. 1). Two types of control experiments were performed. Both the contralateral side of sections of LPS/IFNγ,-injected animals and the ipsilateral side of animals injected with PBS were investigated.
Injection of LPS/IFNγ, ipsilateral versus contralateral
Injection of LPS/IFNγ, increased the number of cells positive for ASS in the striatum and corpus callosum of all sections investigated (Table 1). The number of ED1-positive cells and cells costained for ED 1 antigen and ASS was increased significantly in all ipsilateral brain areas examined when compared to the contralateral hemisphere (Table 1; Figs. 2a and 2b). In the striatum (60%), corpus callosum (80%), and cortex (20%) ASS-positive cells were stained with ED 1 (Table 1). These cells were rounded or oval showing the characteristic morphology of activated microglia (Figs. 2e and 2f). Remaining ASS-positive cells were mainly neurons. The intensity and number of ASS immunostaining in neurons was not affected by the injection of LPS/IFNγ, (see below). Furthermore, the number of perivascular cells positive for ASS and ED1 increased substantially in response to LPS/IFNγ, (Fig. 2e). In both perivascular and parenchymal ED1-positive cells a granule-like reaction with ED1 was observed (arrowheads in Figs. 2e and 2f). Only occasionally perivascular cells costained for ASS and ED 1 antigen were detected in contralateral areas of LPS/IFNγ,-injected animals (not shown). In the ventricles of animals challenged with immunostimulants, an intense staining with ED 1 and for ASS was observed within the plexus choroideus, whereas the plexus of the contralateral ventricle remained unaffected (Figs. 2a and 2b).
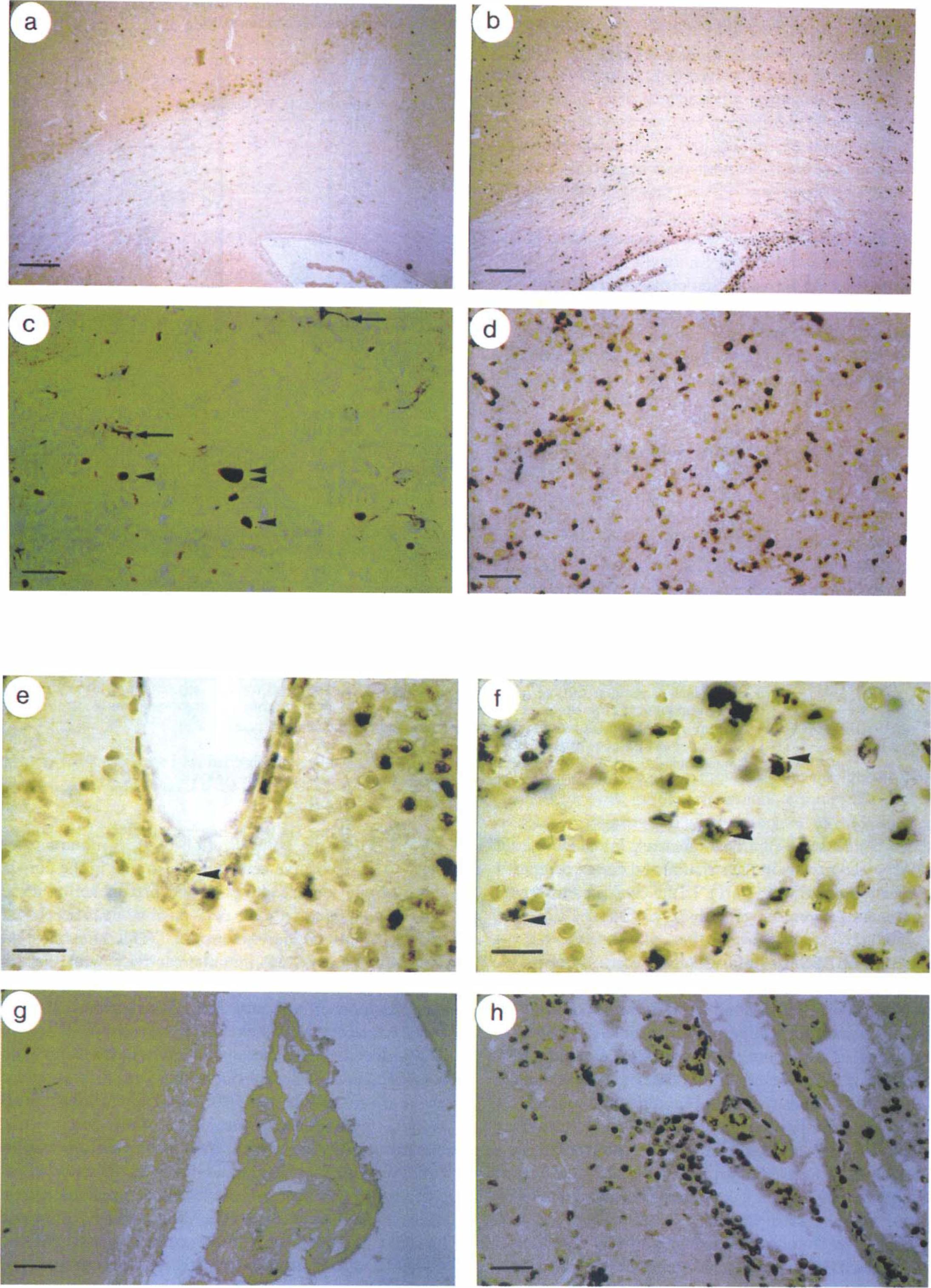
Immunohistochemical localization of ASS and ED1 antigen in paraffin wax sections of rat brain after intrastriatal injection of PBS or LPS/IFNγ. Sections prepared from contralateral
Quantification of ASS-, ED1 antigen- and GFAP-immunoreactive cells in paraffin wax sections of rat brain after intrastriatal injection of PBS or LPS/IFNγ
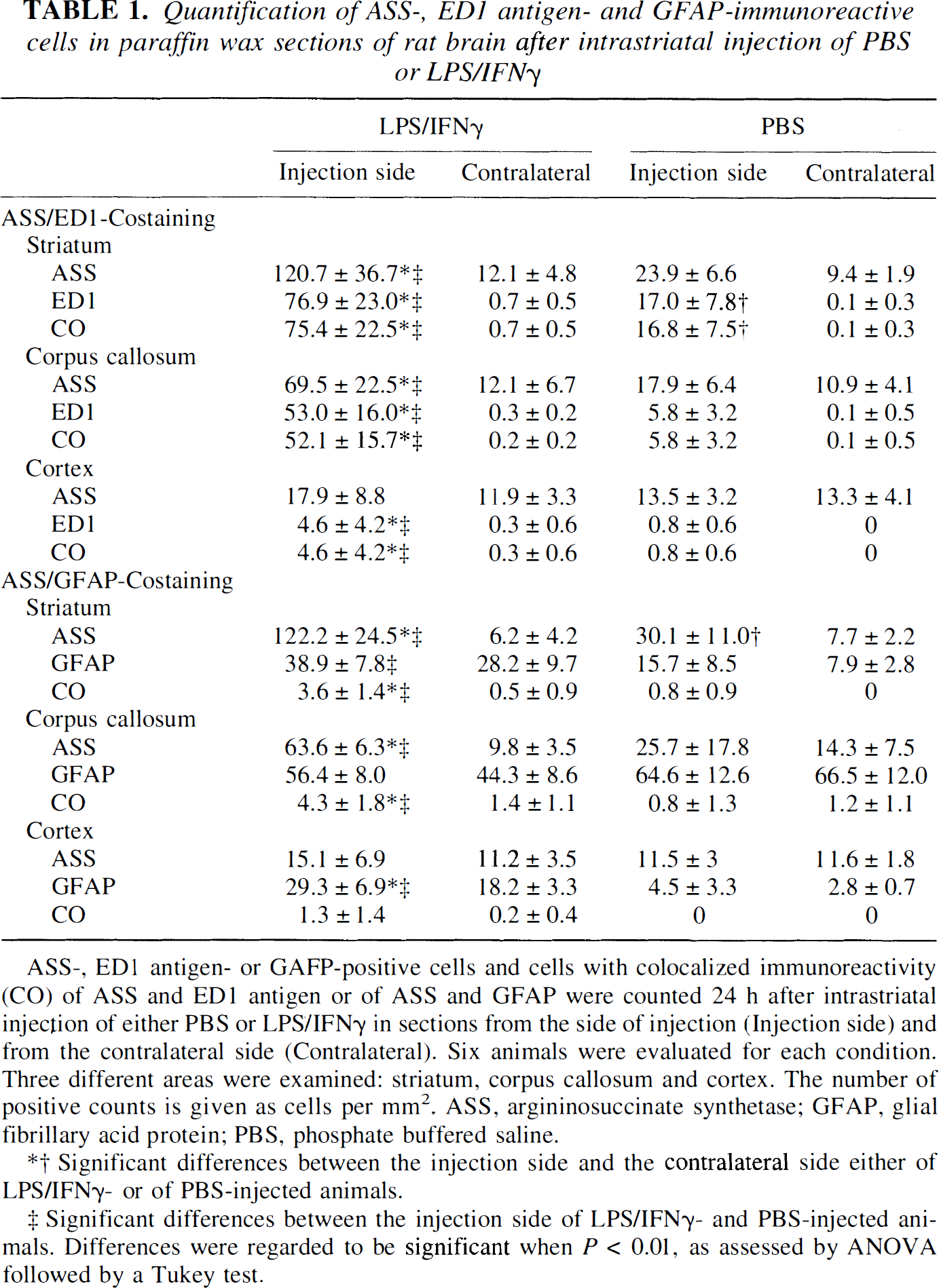
ASS-, ED 1 antigen- or GAFP-positive cells and cells with colocalized immunoreactivity (CO) of ASS and ED1 antigen or of ASS and GFAP were counted 24 h after intrastriatal injection of either PBS or LPS/IFNγ, in sections from the side of injection (Injection side) and from the contralateral side (Contralateral). Six animals were evaluated for each condition. Three different areas were examined: striatum, corpus callosum and cortex. The number of positive counts is given as cells per mm2. ASS, argininosuccinate synthetase; GFAP, glial fibrillary acid protein; PBS, phosphate buffered saline.
Significant differences between the injection side and the contralateral side either of LPS/IFNγ,- or of PBS-injected animals.
Significant differences between the injection side of LPS/IFNγ- and PBS-injected animals. Differences were regarded to be significant when P < 0.01, as assessed by ANOVA followed by a Tukey test.
Occasional localization of ASS immunoreactivity in the same cell with astroglial marker GFAP was questionable, but it was counted as costained in order not to underestimate a possible colocalization. In the corpus callosum GFAP expression was similar in both hemispheres, however, the number of ASS-positive cells ipsilateral to LPS/IFNγ, injection increased strongly (Table 1; Figs. 3a and 3b). In contrast, the number of GFAP-positive cells increased in the cortex ipsilateral to the injection in response to LPS/IFNγ, significantly (Table 1), whereas the number of ASS-positive cells in the cortex remained almost equal under the different conditions examined. In the striatum LPS/IFNγ, increased the number of GFAP-positive cells only in tendency ipsilateral to the injection (Table 1), whereas the size and arborization of GFAP-immunoreactive cells remained almost unaffected. However, the increase in the number of ASS-positive cells in the striatum was more pronounced (Table 1).
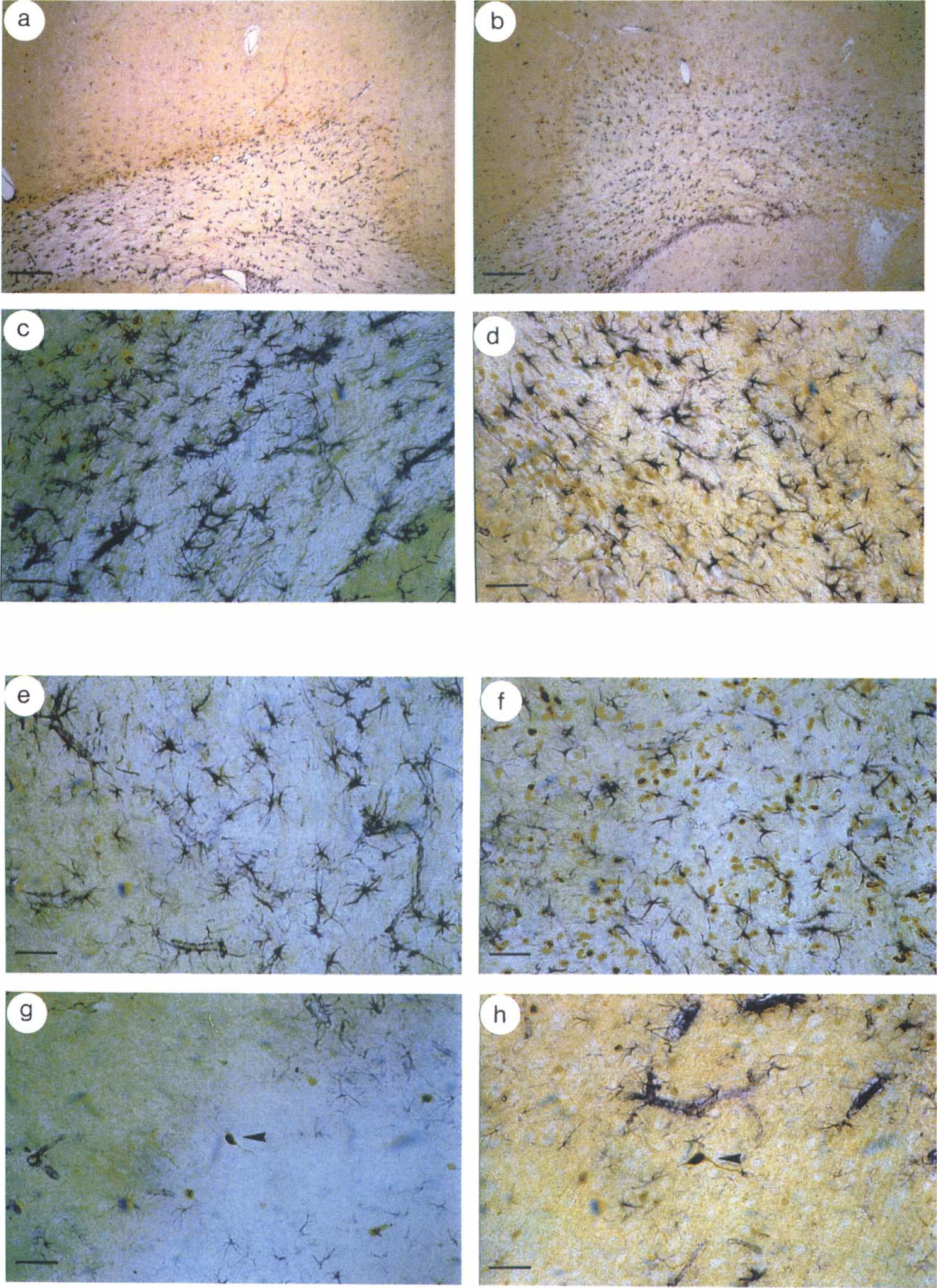
Immunohistochemical colocalization of ASS and GFAP in paraffin wax sections of rat brain after intrastriatal injection of PBS or LPS/IFNγ. Sections prepared from contralateral
Injection of LPS/IFNγ, ipsilateral versus injection of PBS, ipsilateral
Injection of LPS/IFNγ increased the number of cells positive for ASS in the striatum and corpus callosum of all sections investigated when compared to PBS-injected animals (Table 1; Figs. 2c, 2d, and 3c–3f). However, no difference in the number of ASS-positive cells was observed in the cortex of LPS/IFNγ,- or PBS-injected animals (Table 1; Figs. 3g and 3h). Most ASS-immunoreactive cells in the cortex were magnocellular and judged to be neurons by their morphology. The intensity of ASS immunoreactivity in neurons and the number of ASS-positive neurons were not affected by the injection of LPS/IFNγ, compared to PBS-injected brains (arrowheads in Figs. 3g and 3h). The number of ED1-positive cells and cells costained for ED1 antigen and ASS was increased significantly after injection of LPS/IFNγ, in all ipsilateral brain areas examined when compared to the ipsilateral hemisphere of PBS-injected animals (Table 1). In the ventricles of animals challenged with immunostimulants, an intense staining for ED 1 antigen and for ASS was observed within the plexus choroideus, whereas the plexus of PBS-injected animals remained unaffected (Figs. 2g and 2h). Ipsilateral to the injection, LPS/IFNγ caused a significant increase in the number of GFAP-positive cells in the striatum and cortex compared to injection of PBS (Table 1). In the corpus callosum no significant differences were observed (Table 1).
Injection of PBS, ipsilateral versus contralateral
Ipsilateral to the PBS injection a number of ASS-positive cells were detected in each of the three brain areas examined. In sections costained for ASS and ED1 antigen, all of the cells reacting with ED 1 exhibited ASS immunoreactivity in the striatum and comprised about 60% of all ASS-positive cells (Table 1). The majority of ASS- and ED1-positive cells showed a characteristic morphology of activated microglia. In contrast to quiescent arborized microglia (arrows in Fig. 2C), these cells were less ramified and occasionally oval or rounded (single arrowheads in Fig. 2C). In addition, some perivascular cells were also costained with anti-ASS antiserum and ED 1 ipsilateral to the PBS injection (not shown). The strict colocalization of ASS immunoreactivity in ED 1-positive cells holds also true in the corpus callosum and cortex, although the percentage of these cells as compared with all ASS-positive cells decreased and was almost negligible in cortex (Table 1). Almost no colocalization of ASS immunoreactivity with the astroglial marker GFAP was detectable (Table 1). Where colocalization of ASS and GFAP was questionable and could not be completely excluded, the cells were counted as costained in an attempt not to underestimate a possible colocalization. Some but not all ASS-positive cells not reacting with ED1 or anti-GFAP antibody had the typical morphology of neurons (arrowhead in Fig. 3g).
Although the number of ASS-positive cells was in tendency higher ipsilateral to the PBS injection compared to the contralateral side in the striatum and corpus callosum of all sections costained for either ED 1 antigen or GFAP and ASS, this difference reached the level of statistical significance only in the striatum of sections costained for ASS and GFAP (Table 1). No difference of GFAP immunoreactivity was observed between the ipsilateral and contralateral side of PBS-injected animals in all areas investigated, whereas PBS induced a significant increase in the number of ED1-positive cells and cells costained for ED 1 and ASS in the striatum ipsilateral to the injection (Table 1). A small number of ASS-positive cells was identified as neurons in each of the three areas in both, the ipsilateral and contralateral side of PBS-injected animals.
DISCUSSION
As one consequence of a variety of disease states of the CNS the inducible isoform of NOS (iNOS) is expressed in immunocompetent cells such as astrocytes and microglial cells (Murphy and Grzybicki, 1996). Being part of a possible defense mechanism the high output of NO over several hours is not affected by changes in the concentration of intracellular Ca++, the main regulatory agent of the constitutive isoforms of NOS. However, the concentration of essential cofactors or of the substrate L-arginine may become limiting for long-lasting NO production in brain cells and compensatory mechanisms may exist. Indeed, it has been shown that biosynthesis of NOS cofactor tetrahydrobiopterin is induced in glial cells by LPS and IFNγ (Sakai et al., 1995; D'Sa et al., 1996). In cultured murine astroglial cells, upregulation of L-arginine transport and of the expression of ASS in parallel with induction of NO synthesis has been shown (Schmidlin and Wiesinger, 1995; 1998). However, evidence for modulation of any of these pathways auxiliary to the biosynthesis of NO in CNS in vivo has been lacking so far. The present study was designed to investigate a possible upregulation of ASS, the enzyme which initiates recycling of L-citrulline to the NOS substrate L-arginine, in the brain.
The mixture of immunostimulants, LPS plus IFNγ, was chosen because it had elicited in cultured glioma cells as well as in primary astroglial cultures a maximal response in generation of NO, in induction of iNOS, and in expression of ASS (Schmidlin et al., 1997; Schmidlin and Wiesinger, 1998). It may be noted that T-cell-derived IFNγ can be found in the CNS after disruption of the blood-brain barrier during disease states (Benveniste, 1995) and that both microglial and astroglial cells express the LPS receptor CD14 (Becher and Antel, 1996; Galea et al., 1996), thereby enabling the cellular response to bacterial lipopolysaccharide (Ulevitch and Tobias, 1995). The antiserum used in the present study had been generated against a peptide representing amino acid sequence 196–222 of mouse liver ASS. It has been characterized as being monospecific for ASS by Western blot experiments with rat liver and brain homogenates and, therefore, may be taken as showing specific cellular presence of ASS in immunolocalization experiments in rat tissues (Schmidlin et al., 1997; Schmidlin and Wiesinger, 1998). Intrastriatal injections of either LPS/IFNγ or PBS were performed. To investigate if and given the case to which extent other brain areas participate in the response to LPS/IFNγ the striatum, corpus callosum and cortex of both injected and non-injected hemispheres were examined for ASS, ED1 antigen, and GFAP immunoreactivity.
The ipsilateral side of PBS-injected animals and the contralateral side of LPS/IFNγ-injected animals served as controls. Mainly in the first case cells were observed that reacted with antibody ED1. This antibody recognizes cells of the macrophage/monocyte lineage of the rat (Dijkstra et al., 1985) and in the CNS is specific for parenchymal microglial cells in various states of activation, brain macrophages and perivascular microglial cells (Graeber et al., 1990; Flaris et al., 1993; Streit, 1995). Most parenchymal cells reacting with ED1 were oval or rounded revealing the characteristic morphology of activated microglia and exhibited ASS immunoreactivity. This finding shows that PBS injection is sufficient to transform microglial cells, although at a low number, from their quiescent state to an activated one and that this transformation is accompanied by the induction of ASS. Similarly, the injection of PBS into rat hippocampus resulted in weak expression of iNOS in a few parenchymal cells (Garcion et al., 1998). A small number of perivascular cells expressing ASS and reacting with ED 1 were observed in PBS-injected animals ipsilateral to the injection. This is in line with previous findings reporting constitutive expression of ED1 antigen in a small number of perivascular cells in the brain (Graeber et al., 1989).
Lack of ASS immunoreactivity in glial cells of the contralateral side of PBS-injected brains as found in the present investigation is in agreement with two extensive previous studies on the presence of ASS in nonchallenged rat brain. Using a polyclonal antiserum against purified rat liver ASS, Nakamura et al. (1991) detected only weak immunoreactivity in white matter glial cells of a few selected brain areas. A rabbit antiserum against purified human ASS did not stain any glial cells in rat brain sections (Arnt-Ramos et al., 1992). In contrast, strong ASS immunoreactivity was found in murine and rat glial cells after incubation of respective primary cultures with immunostimulants (Schmidlin and Wiesinger, 1998). The hypothesis was suggested that basal ASS expression in glial cells of uninjured brain is too low to be detectable by immunohistochemical methods. In accordance with this reasoning, strong immunoreactivity was found in two populations of glial cells after injection of LPS/IFNγ into the striatum of the rat.
By far the vast majority of ASS-positive cells were costained with antibody ED 1. However, a difference between the areas examined was observed. In the striatum and corpus callosum about 60% and 80% of ASS-positive cells, respectively, were costained with ED 1, whereas in the cortex only 20% of ASS-positive cells were also stained with ED1. In line with this, the number of ASS-positive cells is more than doubled in the striatum and corpus callosum. In the cortex ASS-positivity increased only to a small extent. These results together with a consideration of the morphology of the cells suggest that the majority of ASS-positive cells detected in the cortex of both hemispheres are neurons. In similar rat models of brain inflammation, strong iNOS immunoreactivity was detected in microglial cells 24 hours after intrahippocampal injection of LPS (Garcion et al., 1998) or LPS plus IFNγ (Kitamura et al., 1996). Although colocalization of ASS and iNOS was not attempted in the present study, it must be emphasized that in vivo microglial cells can be induced to express an L-arginine-generating (ASS; this study) as well as an L-arginine-consuming enzyme (iNOS; Kitamura et al., 1996; Garcion et al., 1998) and, therefore, may exhibit a functional NO/citrulline-cycle (Hattori et al., 1994). Whether this cycle operates indeed in the same cell remains to be investigated.
Very few cells exhibiting ASS immunoreactivity were identified as astrocytes by colocalization with the astroglial marker protein GFAP. As observed for ASS and ED1, a variable pattern of GFAP expression was found in the brain regions investigated. GFAP immunoreactivity was induced by LPS/IFNγ in the striatum and cortex of both hemispheres evaluated when compared to PBS injection. The corpus callosum showed no modulation of the basal GFAP expression by immunostimulants. Again, a conformity with iNOS expression must be noted: iNOS immunoreactivity was not colocalized with GFAP-positive astrocytes after intrahippocampal injection of LPS plus IFNγ (Kitamura et al., 1996). If the few ASS expressing astrocytes found in this study do not consume arginine for generation of NO, they may recycle citrulline derived from other cells, e.g., microglial cells. However, the operation of an intercellular NO/L-citrulline-cycle in brain remains an open question (Schmidlin and Wiesinger, 1998).
The finding that ASS is widely induced in activated microglia in vivo is in contrast to results obtained in mixed glial primary cultures derived from newborn rat brain: after incubation of the cultures with the same immunostimulants which were used in the present study only a minority of GFAP-negative cells exhibited ASS immunoreactivity (Schmidlin and Wiesinger, 1998). Absence of ASS from microglial cells in culture even after immunostimulation is in contrast to findings about induction of ASS in peripheral macrophages. ASS immunoreactivity and ASS mRNA were expressed in rat alveolar macrophages (Hammermann et al., 1998) and cells of the murine macrophage line RAW 264.7 (Nussler et al., 1994) after immunostimulation. Even more striking is the opposite finding: immunostaining for ASS, although at varying intensity, was detectable in almost all astroglial cells in primary culture (Schmidlin and Wiesinger, 1998), but scarcely colocalization of ASS with astroglial marker GFAP was detected in vivo. A number of possible explanations can be tested experimentally. Varying degrees of astroglial differentiation may affect expression of ASS. Indeed, cultured astroglial cells treated with dibutyryl cyclic AMP were unable to use citrulline for LPS-stimulated NO synthesis and no ASS immunoreactivity was detected in Western blot experiments (Vogel and Wiesinger, unpublished results, October, 1998). However, it should be noted that astroglial cells in coculture with neurons expressed ASS even in the absence of proinflammatory stimuli (Schmidlin and Wiesinger, 1998). As a further explanation, additional signals in brain may downregulate ASS expression or prevent induction of the enzyme. One such candidate as an inhibitory factor for iNOS expression in the CNS is transforming growth factor β (Vincent et al., 1997), and experiments on the effects of this cytokine on ASS expression in cultured glial cells are under way. Finally, the complex regulation of iNOS expression in astrocytes as a consequence of other primary events may also hold for the induction of ASS, and, therefore, ASS in astrocytes in vivo may only be expressed after prolonged exposure to immunostimulants. After intrahippocampal injection of LPS, iNOS immunoreactivity was induced in a sequential order, e.g., in ependymal cells after 5 hours, in reactive microglia between 15 and 24 hours, but in astrocytes not before 72 hours after injection (Garcion et al, 1998). However, the investigation of the time course of induction of ASS in brain cells was beyond the scope of the present study.
In contrast to glial cells, unequivocal evidence concerning the presence of ASS in neurons throughout the brain in the absence of immunostimulation has been presented (Nakamura et al., 1991; Arnt-Ramos et al., 1992). Distribution and size of ASS-positive cells pointed to localization of the enzyme mainly in interneurons (Nakamura et al., 1991). Colocalization of ASS with the constitutive isoform of NOS in neurons of the cat hypothalamus, selected regions of rat brain, and in porcine cerebral perivascular nerves has been reported (Arnt-Ramos et al., 1992; Isayama et al., 1997; Yu et al., 1997), although ASS in general appears to be more widely distributed than NOS (Nakamura et al., 1991). In the present study neurons in sections obtained from vehicle-injected animals as well as from the contralateral side of stimulant-injected animals exhibited strong ASS immunoreactivity in all brain regions examined and revealed the morphologic characteristics of interneurons especially in the striatum. ASS immunoreactivity was also found in neurons after injection of LPS/IFNγ. However, the number and intensity of staining was comparable to that found in neurons of control brains. This finding points to a constitutive expression of ASS in neurons that is not modulated by proinflammatory cytokines and LPS.
Conversion of L-citrulline to L-arginine with the reaction catalyzed by ASS being the first and rate-limiting step appears to be a constitutive mechanism in a variety of peripheral and central neurons. The recycling of the coproduct of NOS to the substrate of this enzyme may be a prerequisite for NOergic neurotransmission (Pow, 1994; Shuttleworth et al., 1997; Grima et al., 1997) and regulation of cerebral vascular tone (Yu et al., 1997). A partly differential localization of ASS and constitutive NOS (Arnt-Ramos et al., 1992; Isayama et al., 1997) implies that generation and consumption of arginine do not necessarily occur in the same cell and neuronalneuronal or neuronal-endothelial cell trafficking of the respective amino acids may occur. Although the histochemical evidence obtained in the present study must be supported by functional data, the results suggest that glial cells in vivo must also be considered arginine-regenerating cells. However, this ability appears to be negligible under normal physiological conditions, but it is substantially increased under conditions mimicking an inflammation of the brain. Induction of glial NO synthesis is accompanied by glial induction of ASS, the enzyme that initiates the recycling of L-citrulline to L-arginine. Thus, it is shown that in vivo a mechanism is upregulated that supports long-lasting generation of NO by brain glial cells.
Footnotes
Acknowledgments
This article is dedicated to Prof. B. Hamprecht on occasion of his 60th birthday. The authors thank Drs. D. Pellegrino and D. Feinstein for fruitful discussions and critical reading of the manuscript.
