Abstract
Cationic polypeptides are released by activated leukocytes and may play an important role in the regulation of vascular tone. Effects of cationic polypeptides on cerebral vascular tone have not been studied. The present experiments were designed to determine if synthetic cationic polypeptides, poly-L-arginine and poly-L-lysine, affect the function of cerebral arteries. Rings of canine basilar arteries with and without endothelium were suspended for isometric force recording. Poly-L-arginine (10–8–10–7 M) and poly-L-lysine (10–8–10–7 M) caused endothelium-dependent relaxations. A nitric oxide synthase inhibitor, NG-nitro-L-arginine methyl ester (10–4 M), and a nitric oxide scavenger, oxyhemoglobin (3 × 10–6 M), inhibited relaxations in response to cationic polypeptides. Negatively charged molecules, heparin (1 U/ml) and dextran sulfate (10 mg/ml), also inhibited relaxations to poly-L-arginine or poly-L-lysine. Higher concentrations of poly-L-arginine (10–6–10–5 M) and poly-L-lysine (10–6–10–5 M) induced endothelium-independent contractions. A protein kinase C inhibitor, staurosporine (10–8 M), abolished these contractions. Heparin (10 U/ml) and dextran sulfate (100 mg/ml) inhibited the contractile effect of cationic polypeptides but did not affect contractions to phorbol 12,13-dibutyrate. Poly-L-arginine (10–6 M) and poly-L-lysine (10–6 M) abolished endothelium-dependent relaxations in response to bradykinin (10–10–10–6 M) or calcium ionophore A23187 (10–9–10–6 M). Heparin (50 U/ml) and dextran sulfate (200 mg/ml) restored endothelium-dependent relaxations to bradykinin (10–10–10–6 M) in arteries exposed to poly-L-arginine (10–6 M) or poly-L-lysine (10–6 M). These studies demonstrate that in the lower concentration range (10–8–10–7 M), poly-L-arginine and poly-L-lysine induce endothelium-dependent relaxations by production of nitric oxide via charge-dependent activation of endothelial nitric oxide synthase. In the higher concentration range (10–6–10–5 M), cationic polypeptides cause endothelium-independent contractions as well as impairment of endothelium-dependent relaxations in response to bradykinin and A23187. These contractions and inhibition of endothelium-dependent relaxations are also mediated by a charge-dependent mechanism and may involve activation of protein kinase C.
Cytoplasmic granules of neutrophils contain several well characterized components of cationic peptides (Selsted et al., 1985; Henson and Johnston, 1987; Coyle et al., 1994). Cationic peptides, which are released from activated eosinophils (e.g., major basic protein, eosinophil peroxidase), can also increase microvascular permeability in the lung and contract airway smooth muscle (Frigas and Gleich, 1986; Coyle et al., 1994; Spina and Goldie, 1994; Minnicozzi et al., 1995). Previous studies in isolated rat aorta (Thomas et al., 1986, 1989; Rapoport et al., 1989) and in isolated bovine pulmonary arteries and veins (Ignarro et al., 1989) suggested that cationic polypeptides induce endothelium-dependent relaxations via increased production of nitric oxide. In contrast, several studies on peripheral arteries indicated that prolonged contact of blood vessels with cationic polypeptides inhibits relaxations induced by activation of endothelial cells (Chang et al., 1987; Ignarro et al., 1989; Rapoport et al., 1989), illustrating a complexity of vascular effects mediated by positively charged molecules. A more recent study demonstrated that electrostatic forces play an important role in the regulation of endothelial nitric oxide synthase activity (Venema et al., 1995). Because the effects of cationic polypeptides have not been studied in cerebral arteries, the present study was designed to determine if poly-L-arginine and poly-L-lysine affect cerebral arterial function including production, release, and vasodilator activity of nitric oxide.
METHODS
The experiments were performed on 4-mm basilar artery rings taken from mongrel dogs (15–20 kg) of either sex, anesthetized with 30 mg/kg i.v. pentobarbital sodium. All procedures were conducted in accordance with institutional guidelines. Rings were studied in modified Krebs–Ringer bicarbonate solution (control solution) of the following composition (mM): NaCl 118.3, KCl 4.7, CaCl2 2.5, MgSO4 1.2, KH2PO4 1.2, NaHCO3 25.0, calcium ethylenediaminetetraacetate (EDTA) 0.026, and glucose 11.1. In certain rings, the endothelium was removed mechanically (Katusic et al., 1984, 1989). Each ring was connected to an isometric force transducer (Grass FT03) and suspended in an organ chamber filled with 25 ml control solution (37°C, pH 7.4) bubbled with 94% O2/6% CO2 gas mixture. Each ring was then gradually stretched to the optimal point of its length–tension curve as determined by the contraction to uridine 5′-triphosphate (10–5 M). In most of the studied arteries, optimal tension was achieved at ∼3 g. The functional integrity of endothelium was evaluated by the presence of relaxation induced by vasopressin (10–7 M) (Katusic et al., 1984).
Preparation of oxyhemoglobin
Oxyhemoglobin was prepared by adding 1 mM solution of mixtures of reduced and oxidized hemoglobin and a 40-fold molar excess of sodium hydrosulfite. Sodium hydrosulfite was then removed by dialysis against 15 L of distilled water containing 1% EDTA bubbled with nitrogen at room temperature. After dialysis, the oxyhemoglobin content was determined using a co-oximeter (282 Co-oximeter; Instrumentation Laboratory, Lexington, MA, U.S.A.) (Martin et al., 1985; Katusic et al., 1989). Previous studies demonstrated that commercially available hemoglobin could be contaminated with endotoxin (Mayberg et al., 1990) and hemin (Letarte et al., 1993). In the present study, hemoglobin was used as a pharmacological tool to chemically inactivate nitric oxide, and under our experimental conditions we did not detect nonselective effects of hemoglobin.
Drugs
The following pharmacological agents were used: calcium ionophore A23187, arginine vasopressin, bradykinin, dextran sulfate, dimethyl sulfoxide (DMSO), EDTA, hemoglobin, heparin, indomethacin, L-arginine hydrochloride, NG-nitro-L-arginine methyl ester (L-NAME), PDBu, poly-L-arginine (MW 92,000–95,400), poly-L-lysine (MW 87,400–95,100), potassium chloride (KCl), papaverine hydrochloride, sodium hydrosulfite, staurosporine, uridine 5′-triphosphate (Sigma, St. Louis, MO, U.S.A.), and 3-morpholinosydnonimine (SIN-1) (Casella AG, Frankfurt, Germany). Drugs were dissolved in distilled water such that volumes of <0.2 ml were added to the organ chambers. Stock solutions of A23187 (10–6 M), PDBu (10–6 M), and staurosporine (10–8 M) were prepared in DMSO (0.5 × 10–4 M). Stock solutions of indomethacin (10–5 M) were prepared in equal molar concentrations of Na2CO3. The concentrations of drugs are expressed as final molar concentrations.
Concentration-response curves
Concentration–response curves were obtained in a cumulative fashion. Several rings cut from the same artery were studied in parallel. Only one concentration–response curve was made. All experiments except for those of concentration–response curves to KCl, PDBu, and uridine 5′-triphosphate were performed in the presence of indomethacin (10–5 M) to inhibit the production of prostanoids by the cyclooxygenase pathway. On average, 25, 60, and 60 min were required to construct concentration–response curves for KCl, PDBu, and uridine 5′-triphosphate, respectively. Responses to bradykinin, A23187, low concentrations of poly-L-arginine and poly-L-lysine, and SIN-1 were obtained during submaximal contractions to uridine 5′-triphosphate (10–5 M). Because L-NAME, oxyhemoglobin, and high concentrations of poly-L-arginine and poly-L-lysine increased resting tension, care was taken to match the contractions induced by uridine 5′-triphosphate in control and treated rings. The contractions were expressed as a percentage of the maximal contractions to KCl (60 mM), whereas the relaxations were expressed as a percentage of the maximal relaxations to papaverine (3 × 10–4 M). The incubation periods were 30 min for indomethacin and staurosporine, 15 min for L-arginine, L-NAME, oxyhemoglobin, poly-L-arginine, and poly-L-lysine (for pretreatment), and 5 min for heparin and dextran sulfate.
Statistical analysis
The data are expressed as means ± SD; n refers to the number of dogs from which the basilar artery was taken. Statistical analysis was performed using a one-way analysis of variance, followed by Scheffé's F test. Differences were considered to be statistically significant when p was <0.05.
RESULTS
Endothelium-dependent relaxations in response to poly-L-arginine and poly-L-lysine
Poly-L-arginine (10–8–10–7 M) and poly-L-lysine (10–10–10–7 M) caused endothelium-dependent relaxations (Fig. 1). The L-arginine analogue L-NAME (10–4 M) reduced relaxations in response to poly-L-arginine or poly-L-lysine (Figs. 2 and 3). Pretreatment with L-arginine (3 × 10–4 M) restored relaxations induced by the cationic polypeptides in arteries exposed to L-NAME (Figs. 2 and 3).
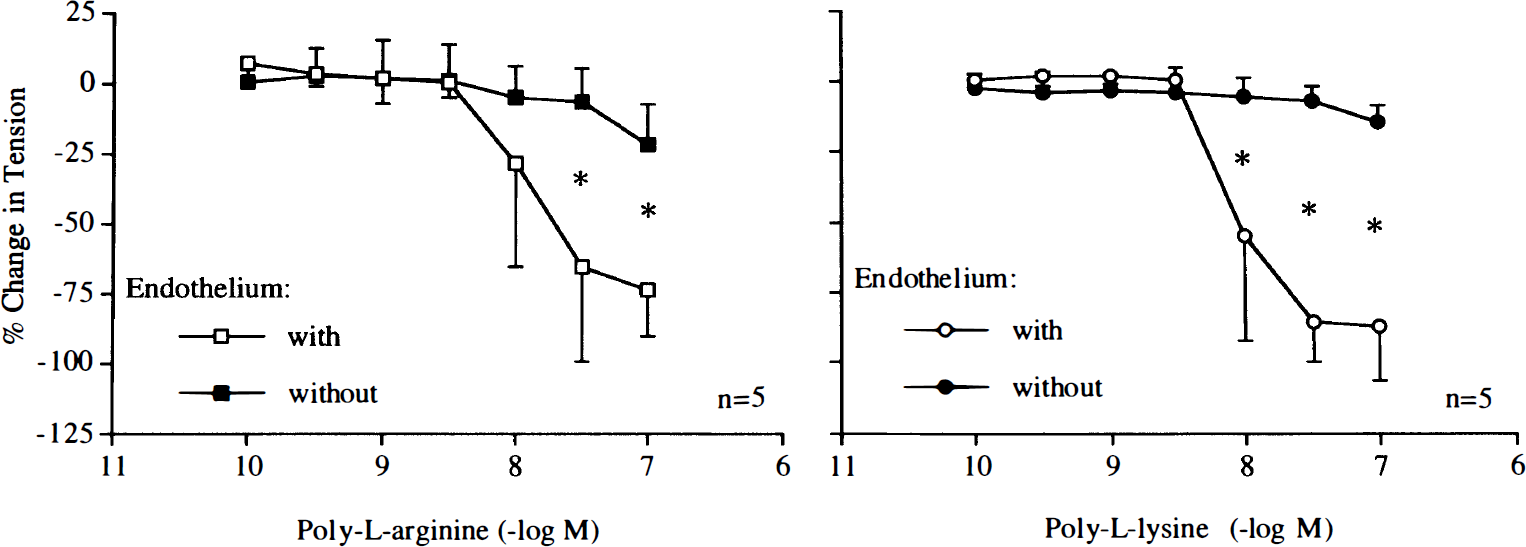
Concentration–response curves to poly-L-arginine and poly-L-lysine obtained in canine basilar arteries with and without endothelium. Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 3.5 ± 0.9 g (n = 5) and 3.7 ± 0.6 g (n = 5)] for rings with and without endothelium exposed to poly-L-arginine, 100% = 4.8 ± 0.6 g (n = 5) and 5.2 ± 1.1 g (n = 5) for rings with and without endothelium, exposed to poly-L-lysine, respectively). *Difference between rings with and without endothelium is statistically significant (p < 0.05).
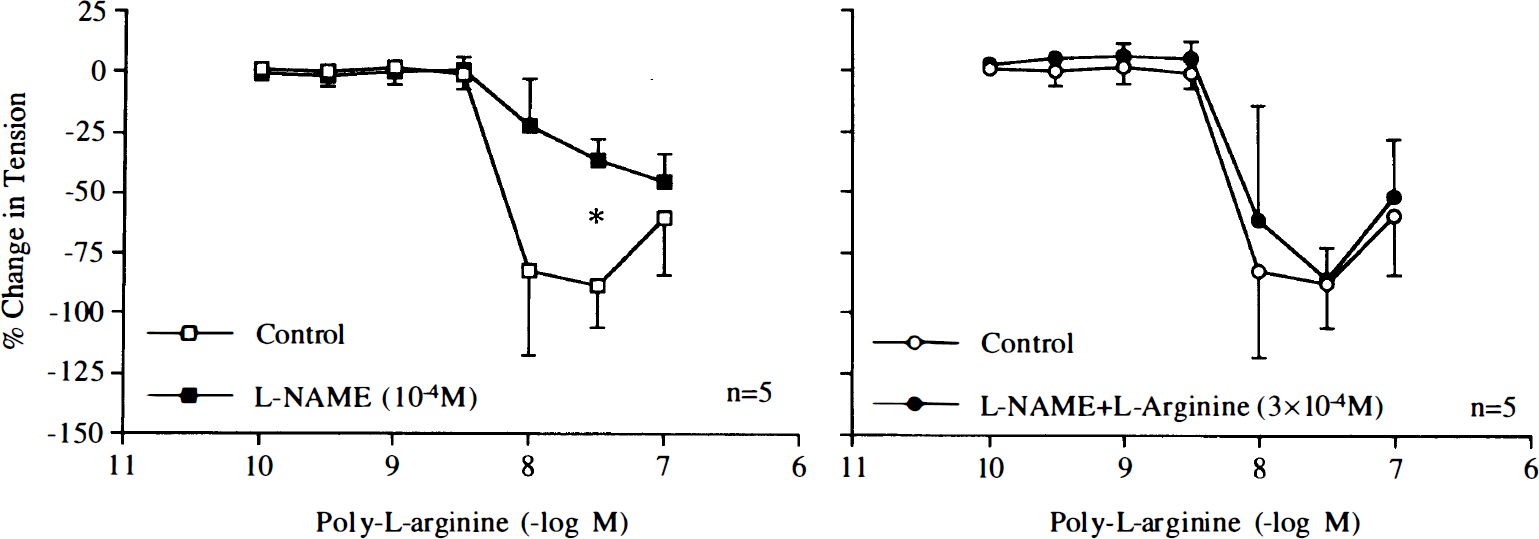
Concentration–response curves to poly-L-arginine obtained in canine basilar arteries with endothelium in the absence and presence of NG-nitro-L-arginine methyl ester (L-NAME; 10–4 M) or L-NAME plus L-arginine (3 × 10–4 M). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 4.4 ± 1.1 g (n = 5), 4.8 ± 0.7 g (n = 5), and 4.7 ± 0.6 g (n = 5) for control rings and rings treated with L-NAME or L-NAME plus L-arginine, respectively]. *Difference between control rings and rings treated with L-NAME is statistically significant (p < 0.05).
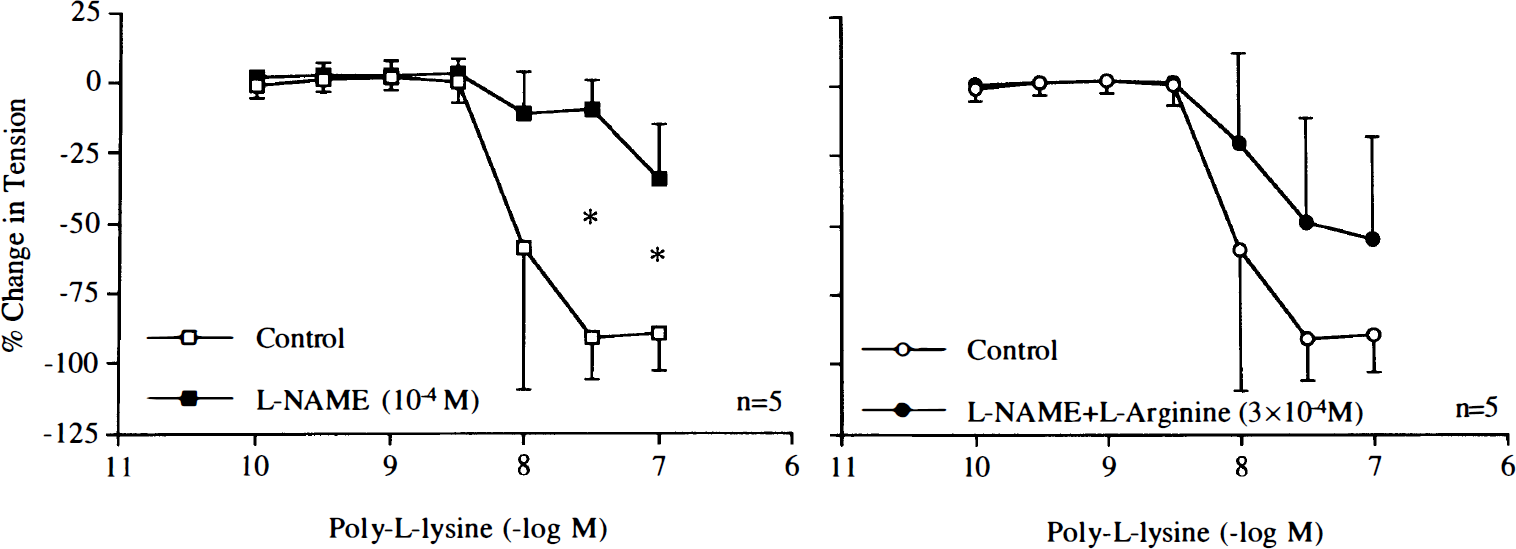
Concentration–response curves to poly-L-lysine obtained in canine basilar arteries with endothelium in the absence and presence of NG-nitro-L-arginine methyl ester (L-NAME; 10–4 M) or L-NAME plus L-arginine (3 × 10–4 M). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 4.0 ± 0.7 g (n = 5), 4.2 ± 0.8 g (n = 5), and 4.0 ± 0.8 g (n = 5) for control rings and rings treated with L-NAME or L-NAME plus L-arginine, respectively]. *Difference between control rings and rings treated with L-NAME is statistically significant (p < 0.05).
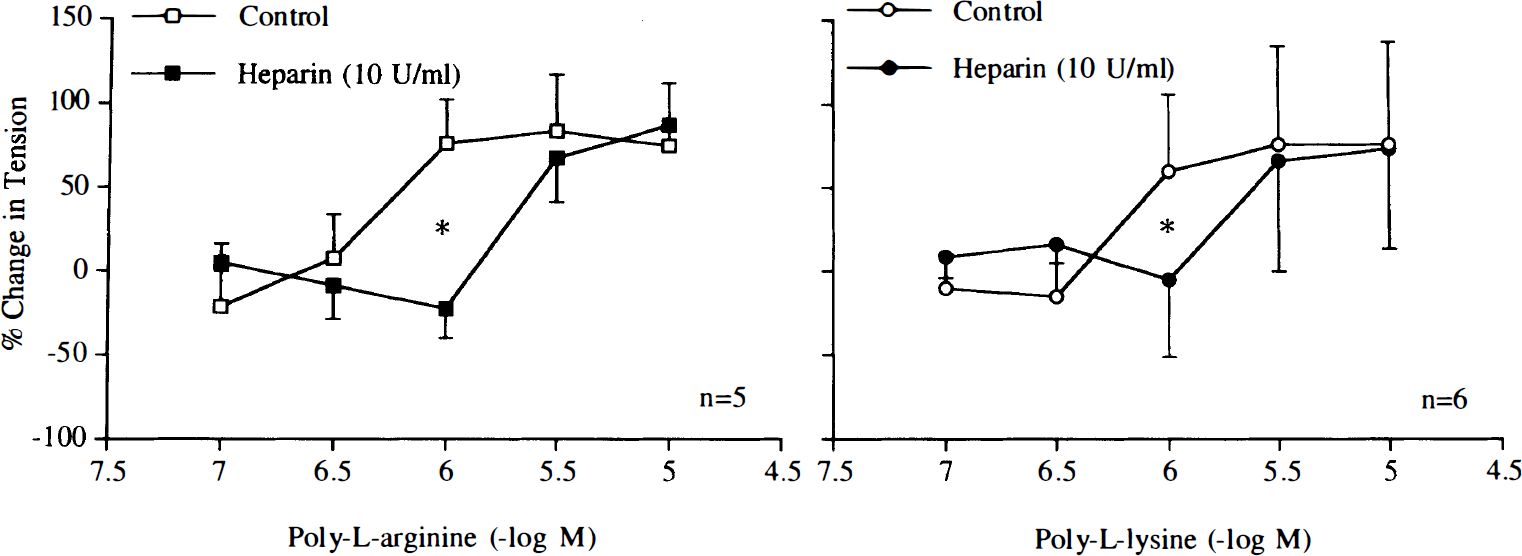
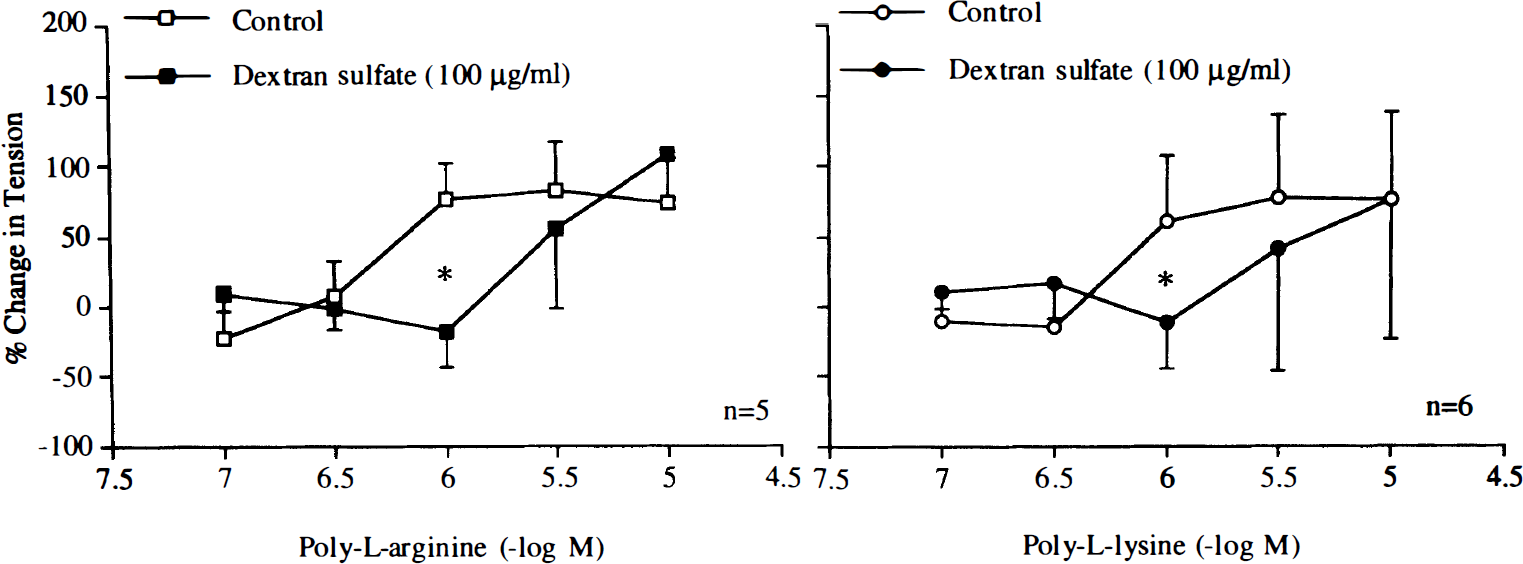
The nitric oxide scavenger oxyhemoglobin (3 × 10–6 M) inhibited relaxations induced by poly-L-arginine or poly-L-lysine (Fig. 4). The negatively charged molecules heparin (1 U/ml) and dextran sulfate (10 mg/ml) reduced and abolished these relaxations, respectively (Figs. 5 and 6).
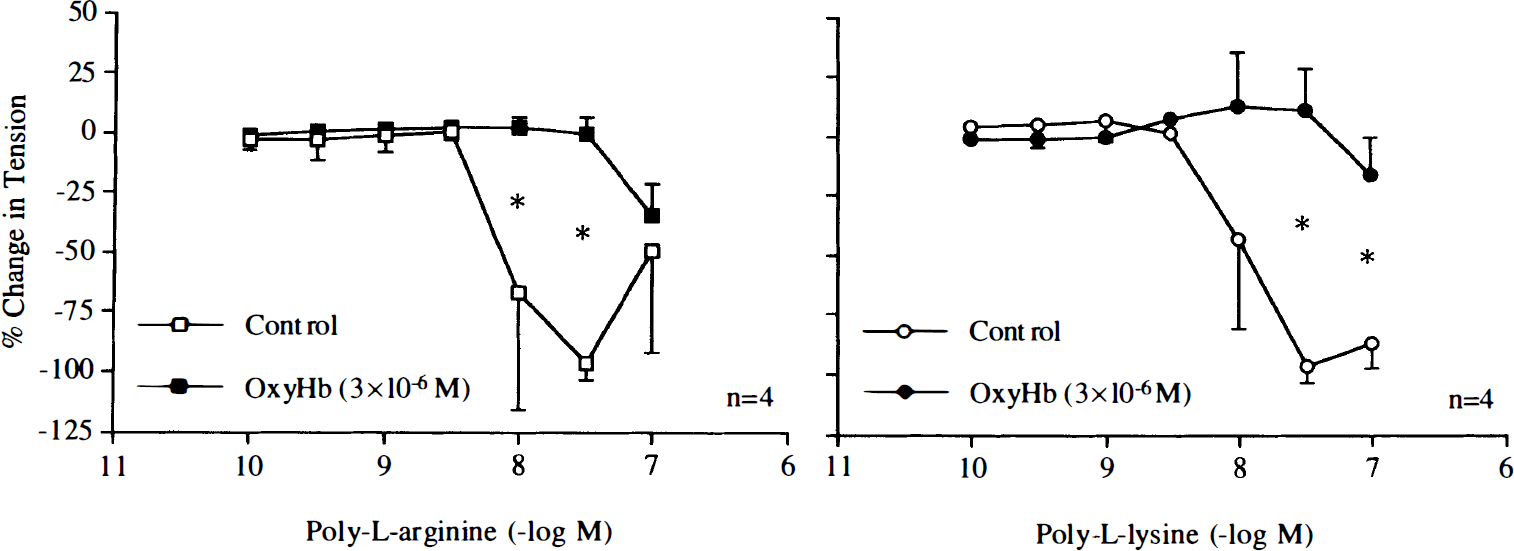
Concentration–response curves to poly-L-arginine and poly-L-lysine obtained in canine basilar arteries with endothelium in the absence and presence of oxyhemoglobin (oxyHb; 3 × 10–6 M). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 4.2 ± 3.2 g (n = 4) and 3.5 ± 1.5 g (n = 4) for control rings of poly-L-arginine and rings treated with oxyHb, 100% = 3.8 ± 1.8 g (n = 4) and 4.6 ± 2.6 g (n = 4) for control rings of poly-L-lysine and rings treated with oxyHb, respectively]. *Difference between control rings and rings treated with oxyHb is statistically significant (p < 0.05).
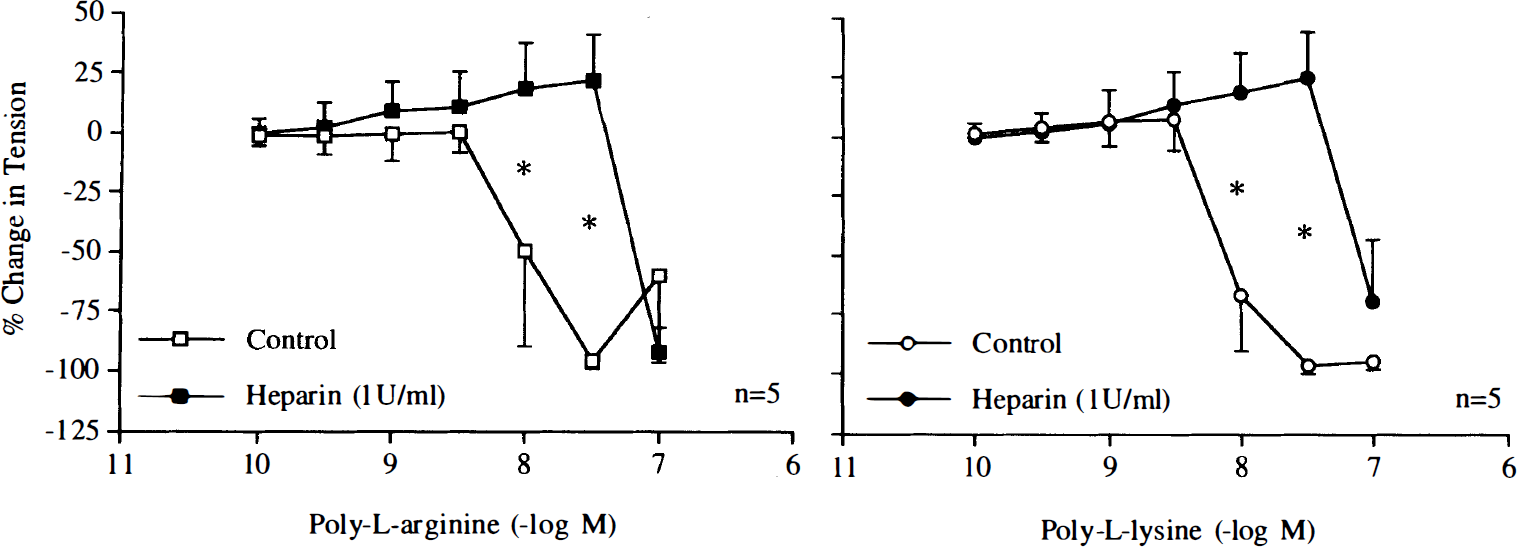
Concentration–response curves to poly-L-arginine and poly-L-lysine obtained in canine basilar arteries with endothelium in the absence and presence of heparin (1 U/ml). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 4.2 ± 1.2 g (n = 5) and 4.2 ± 1.2 g (n = 5) for control rings of poly-L-arginine and rings treated with heparin, 100% = 4.6 ± 1.2 g (n = 5) and 4.4 ± 1.3 g (n = 5) for control rings of poly-L-lysine and rings treated with heparin, respectively]. *Difference between control rings and rings treated with heparin is statistically significant (p < 0.05).
Concentration–response curves to poly-L-arginine and poly-L-lysine obtained in canine basilar arteries with endothelium in the absence and presence of dextran sulfate (10 mg/ml). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 4.0 ± 1.1 g (n = 5) and 4.2 ± 1.1 g (n = 5) for control rings of poly-L-arginine and rings treated with dextran sulfate, 100% = 3.7 ± 1.4 g (n = 5) and 3.8 ± 0.9 g (n = 5) for control rings of poly-L-lysine and rings treated with dextran sulfate, respectively]. *Difference between control rings and rings treated with dextran sulfate is statistically significant (p < 0.05).
Endothelium-independent contractions in response to poly-L-arginine and poly-L-lysine
Higher concentrations of poly-L-arginine and poly-L-lysine (10–6–10–5 M) induced endothelium-independent contractions (Fig. 7). Staurosporine (10–8 M) abolished contractions in response to the cationic polypeptides (Fig. 8). Similarly, staurosporine (10–8 M) significantly reduced contractions caused by PDBu (10–9–10–6 M), whereas this concentration of staurosporine did not affect endothelium-independent contractions caused by KCl (Fig. 9).
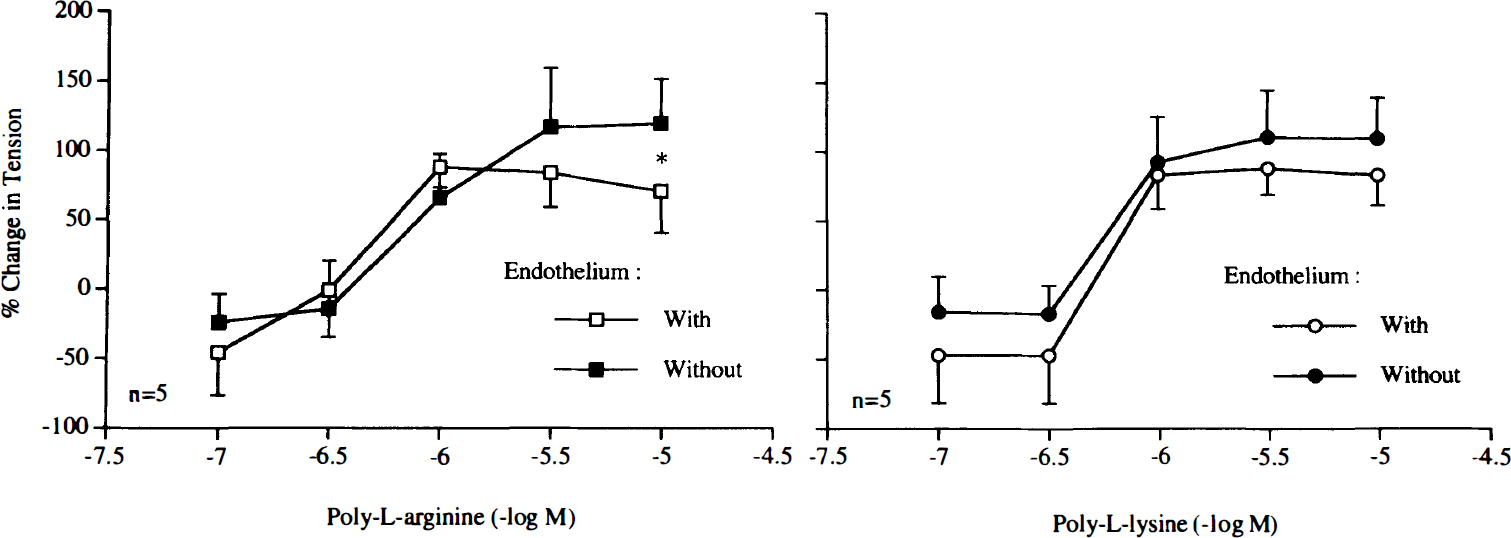
Concentration–response curves to poly-L-arginine or poly-L-lysine obtained in canine basilar arteries with and without endothelium. Data are shown as means ± SD and expressed as percent of maximal contraction induced by KCl [60 mM; 100% = 3.6 ± 1.5 g (n = 5) and 4.1 ± 2.3 g (n = 5) for rings with and without endothelium exposed to poly-L-arginine, 100% = 4.5 ± 1.3 g (n = 5) and 3.8 ± 2.3 g (n = 5) for rings with and without endothelium exposed to poly-L-lysine, respectively]. *In rings exposed to poly-L-arginine, difference between with and without endothelium is statistically significant (p < 0.05).
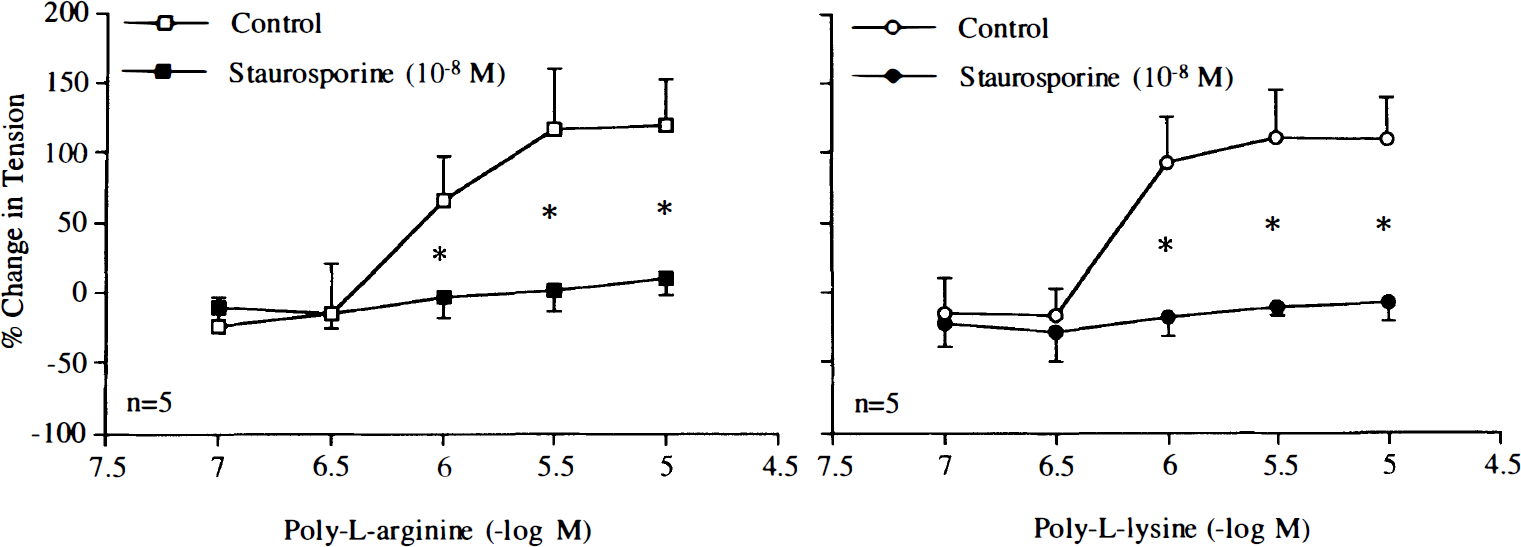
Concentration–response curves to poly-L-arginine or poly-L-lysine obtained in canine basilar arteries without endothelium in the absence and presence of staurosporine (10–8 M). Data are shown as means ± SD and expressed as percent of maximal contraction induced by KCl [60 mM; 100% = 4.1 ± 2.3 g (n = 5) and 3.3 ± 1.3 g (n = 5) for control rings of poly-L-arginine and rings treated with staurosporine, 100% = 3.8 ± 2.3 g (n = 5) and 2.7 ± 0.4 g (n = 5) for control rings of poly-L-lysine and rings treated with staurosporine, respectively]. *Difference between control rings and rings treated with staurosporine is statistically significant (p < 0.05).
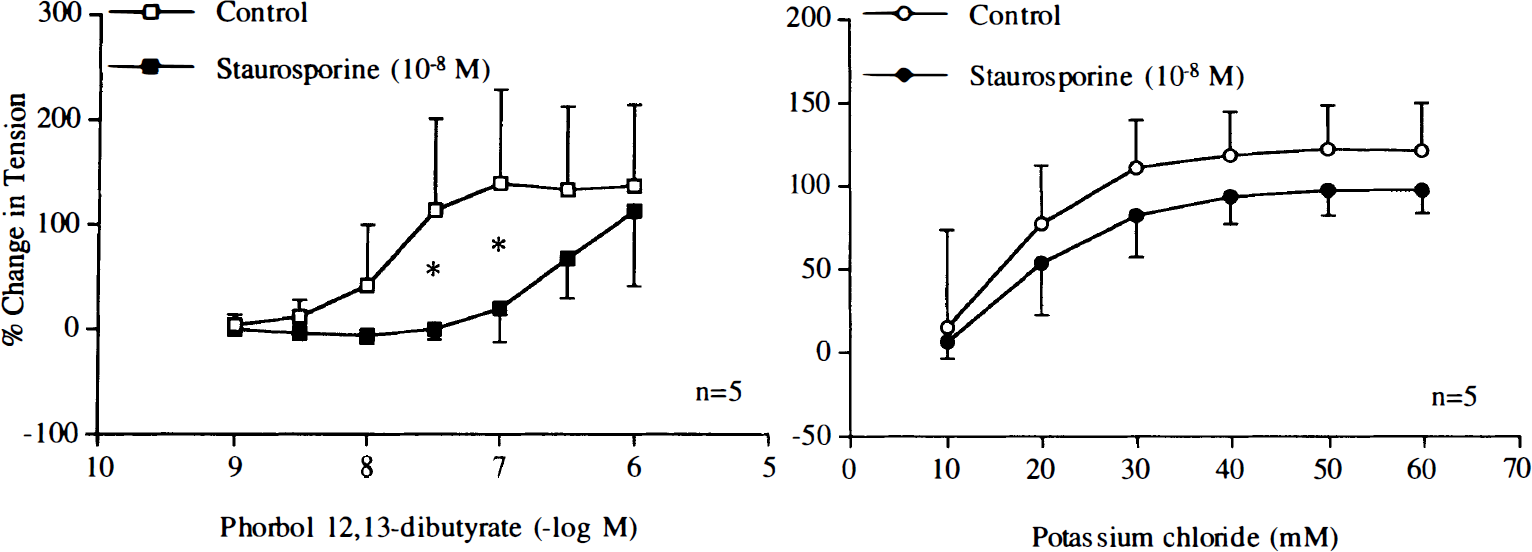
Concentration–response curves to phorbol 12,13-dibutyrate or potassium chloride (KCl) obtained in canine basilar arteries without endothelium in the absence and presence of staurosporine (10–8 M). Data are shown as means ± SD and expressed as percent of maximal contraction induced by KCl [60 mM; 100% = 3.4 ± 2.2 g (n = 5) and 3.0 ± 3.0 g (n = 5) for control rings of phorbol 12,13-dibutyrate and rings treated with staurosporine, 100% = 3.0 ± 1.9 g (n = 5) and 3.2 ± 1.8 g (n = 5) for control rings of KCl and rings treated with staurosporine, respectively]. *Difference between control rings of phorbol 12,13-dibutyrate and rings treated with staurosporine is statistically significant (p < 0.05).
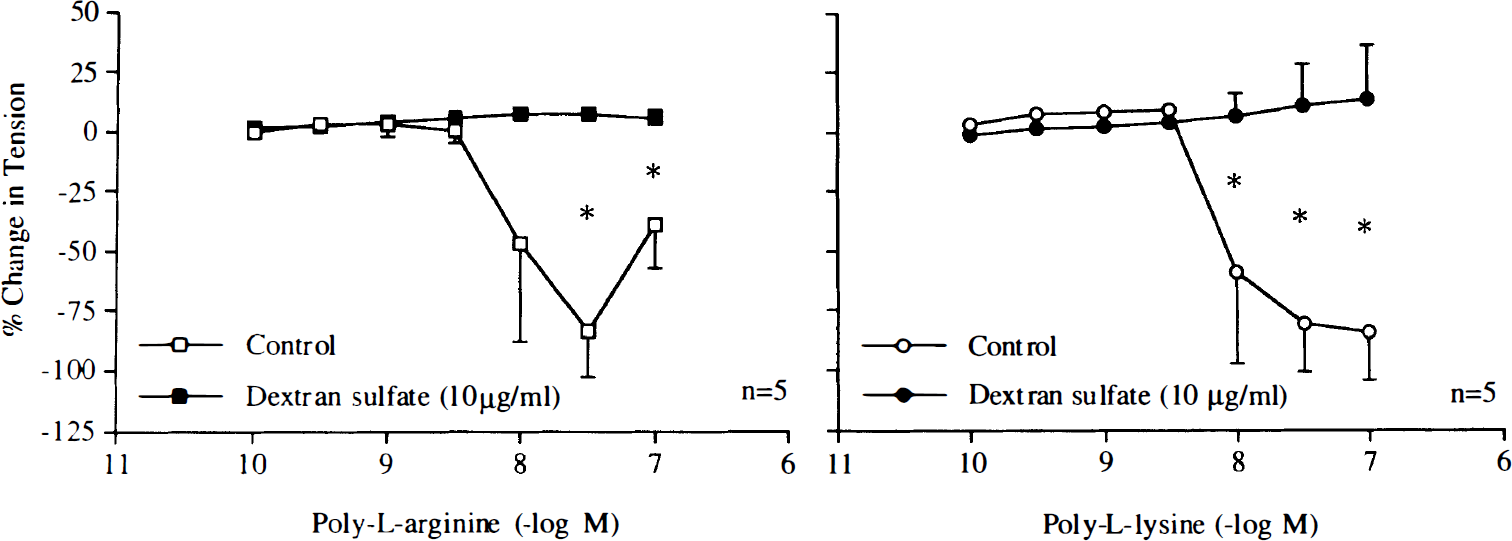
Heparin (10 U/ml) and dextran sulfate (100 mg/ml) inhibited contractions induced by poly-L-arginine or poly-L-lysine (Figs. 10 and 11). but did not affect contractions caused by PDBu (10–9–10–6 M; n = 5, data not shown), uridine 5′-triphosphate (10–8–10–3 M; n = 5, data not shown), or KCl (10–60 mM; n = 5, data not shown).
Concentration–response curves to poly-L-arginine (10–5 M) or poly-L-lysine (10–5 M) obtained in canine basilar arteries without endothelium in the absence and presence of heparin (10 U/ml). Data are shown as means ± SD and expressed as percent of maximal contraction induced by KCl [60 mM; 100% = 4.1 ± 2.0 g (n = 5) and 4.2 ± 1.1 g (n = 5) for control rings of poly-L-arginine and rings treated with heparin, 100% = 3.4 ± 0.9 g (n = 6) and 3.0 ± 1.7 g (n = 6) for control rings of poly-L-lysine and rings treated with heparin, respectively]. *Difference between control rings and rings treated with heparin is statistically significant (p < 0.05).
Concentration–response curves to poly-L-arginine (10–5 M) or poly-L-lysine (10–5 M) obtained in canine basilar arteries without endothelium in the absence and presence of dextran sulfate (100 mg/ml). Data are shown as means ± SD and expressed as percent of maximal contraction induced by KCl [60 mM; 100% = 4.1 ± 2.0 g (n = 5) and 4.5 ± 2.1 g (n = 5) for control rings of poly-L-arginine and rings treated with dextran sulfate, 100% = 3.4 ± 0.9 g (n = 6) and 3.2 ± 1.6 g (n = 6) for control rings of poly-L-lysine and rings treated with sulfate, respectively]. *Difference between control rings and rings treated with dextran sulfate is statistically significant (p < 0.05).
Effect of poly-L-arginine and poly-L-lysine on relaxations in response to bradykinin, A23187, and SIN-1
During contractions induced by poly-L-arginine (10–6 M) and poly-L-lysine (10–6 M), endothelium-dependent relaxations to bradykinin (10–10–10–6 M) or calcium ionophore A23187 (10–9–10–6 M) were abolished (Figs. 12 and 13). In rings without endothelium, the cationic polypeptides also reduced relaxations caused by the nitric oxide donor SIN-1 (10–9–10–4 M) (Fig. 14).
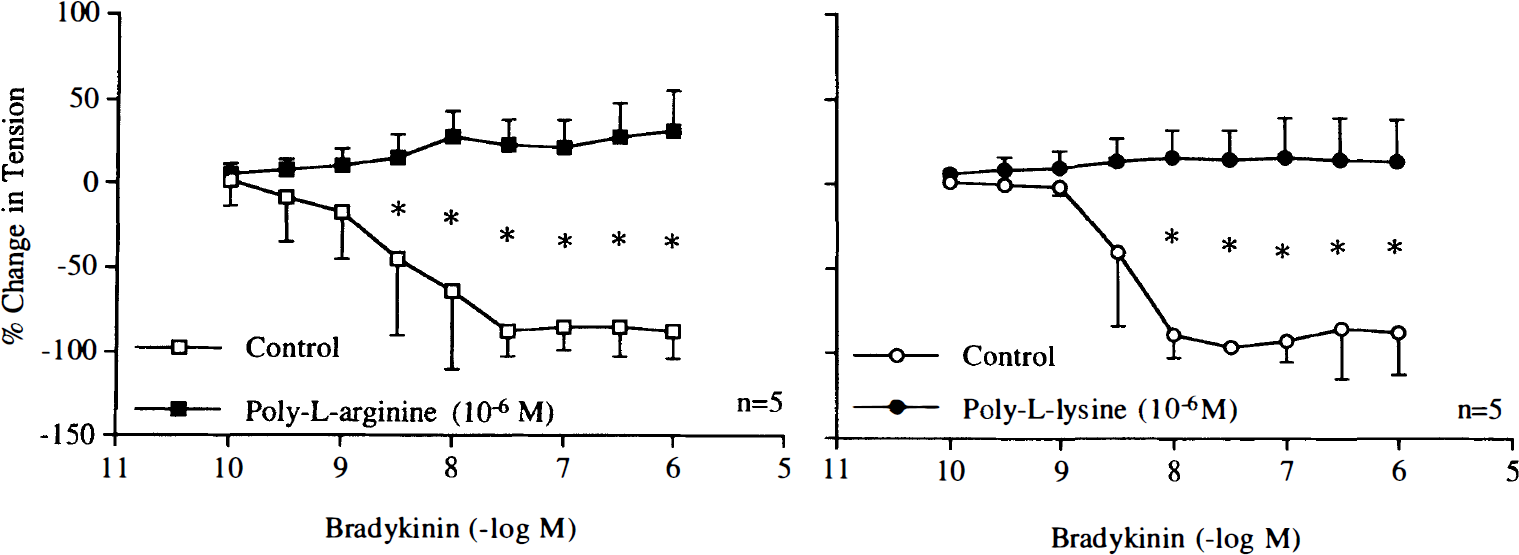
Concentration–response curves to bradykinin obtained in canine basilar arteries with endothelium in the absence and presence of poly-L-arginine (10–6 M) or poly-L-lysine (10–6 M). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 2.8 ± 1.3 g (n = 5) and 3.2 ± 1.9 g (n = 5) for control rings and rings treated with poly-L-arginine, 100% = 5.8 ± 1.4 g (n = 5) and 5.2 ± 1.5 g (n = 5) for control rings and rings treated with poly-L-lysine, respectively]. *Difference between control rings and rings treated with poly-L-arginine or poly-L-lysine is statistically significant (p < 0.05).
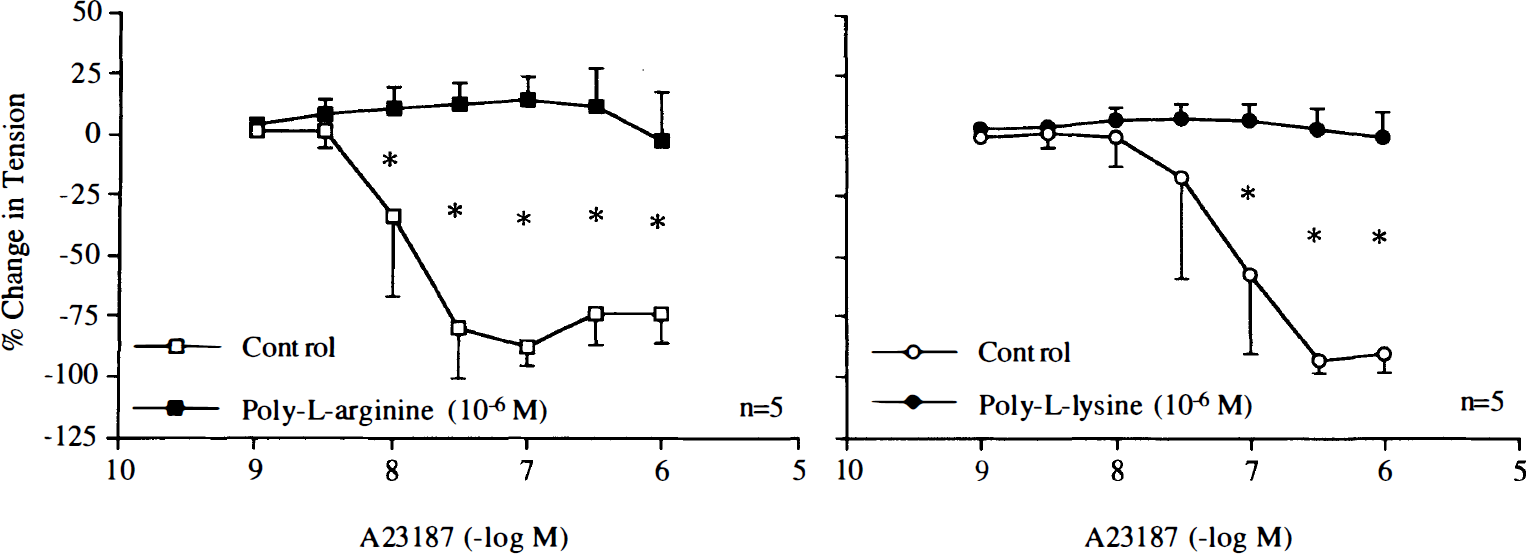
Concentration–response curves to A23187 obtained in canine basilar arteries with endothelium in the absence and presence of poly-L-arginine (10–6 M) or poly-L-lysine (10–6 M). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 6.0 ± 1.6 g (n = 5) and 5.7 ± 1.4 g (n = 5) for control rings and rings treated with poly-L-arginine, 100% = 5.1 ± 1.5 g (n = 5) and 5.0 ± 1.3 g (n = 5) for control rings and rings treated with poly-L-lysine, respectively]. *Difference between control rings and rings treated with poly-L-arginine or poly-L-lysine is statistically significant (p < 0.05).
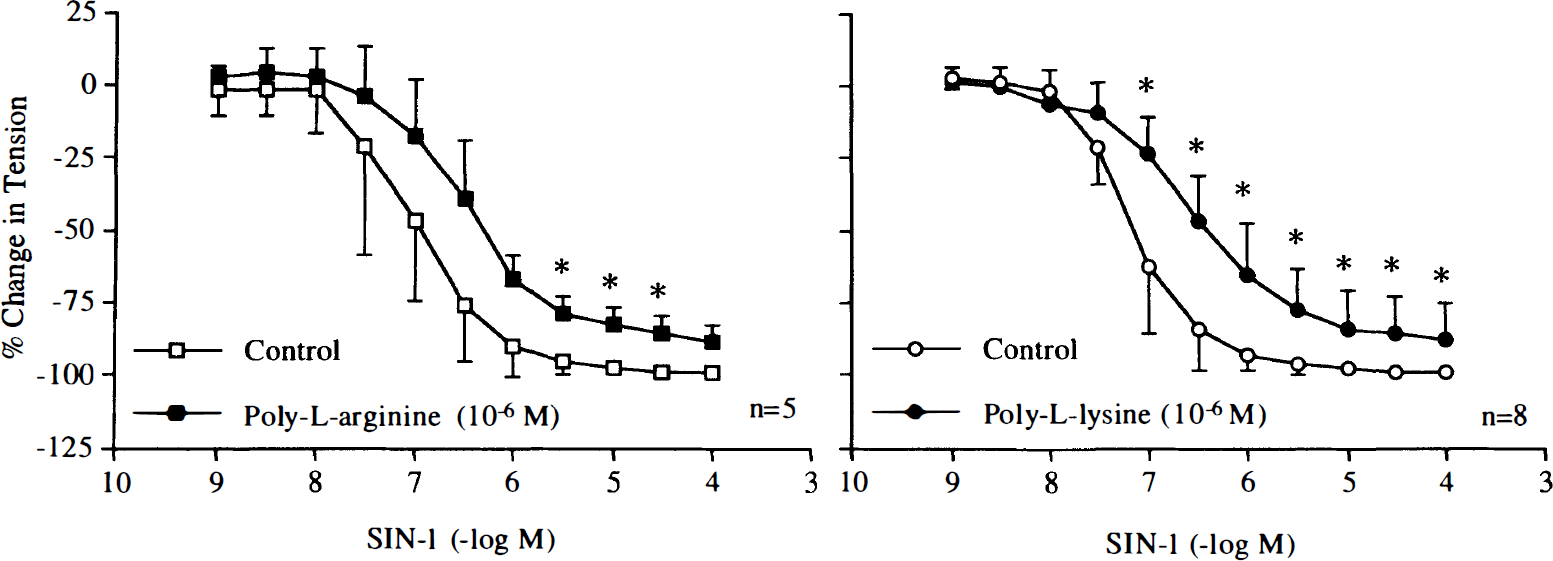
Concentration–response curves to 3-morpholinosydnonimine (SIN-1) obtained in canine basilar arteries without endothelium in the absence and presence of poly-L-arginine (10–6 M) or poly-L-lysine (10–6 M). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 2.4 ± 0.6 g (n = 5) and 2.9 ± 1.4 g (n = 5) for control rings and rings treated with poly-L-arginine, 100% = 4.2 ± 0.9 g (n = 8) and 3.5 ± 1.0 g (n = 8) for control rings and rings treated with poly-L-lysine, respectively]. *Difference between control rings and rings treated with poly-L-arginine or poly-L-lysine is statistically significant (p < 0.05).
In the presence of heparin (50 U/ml) or dextran sulfate (200 mg/ml), endothelium-dependent relaxations to bradykinin (10–10–10–6 M) were restored in arteries exposed to poly-L-arginine (10–6 M) or poly-L-lysine (10–6 M) (Figs. 15 and 16). These concentrations of heparin and dextran sulfate did not affect endothelium-dependent relaxations to bradykinin (10–10–10–6 M; n = 5, data not shown).
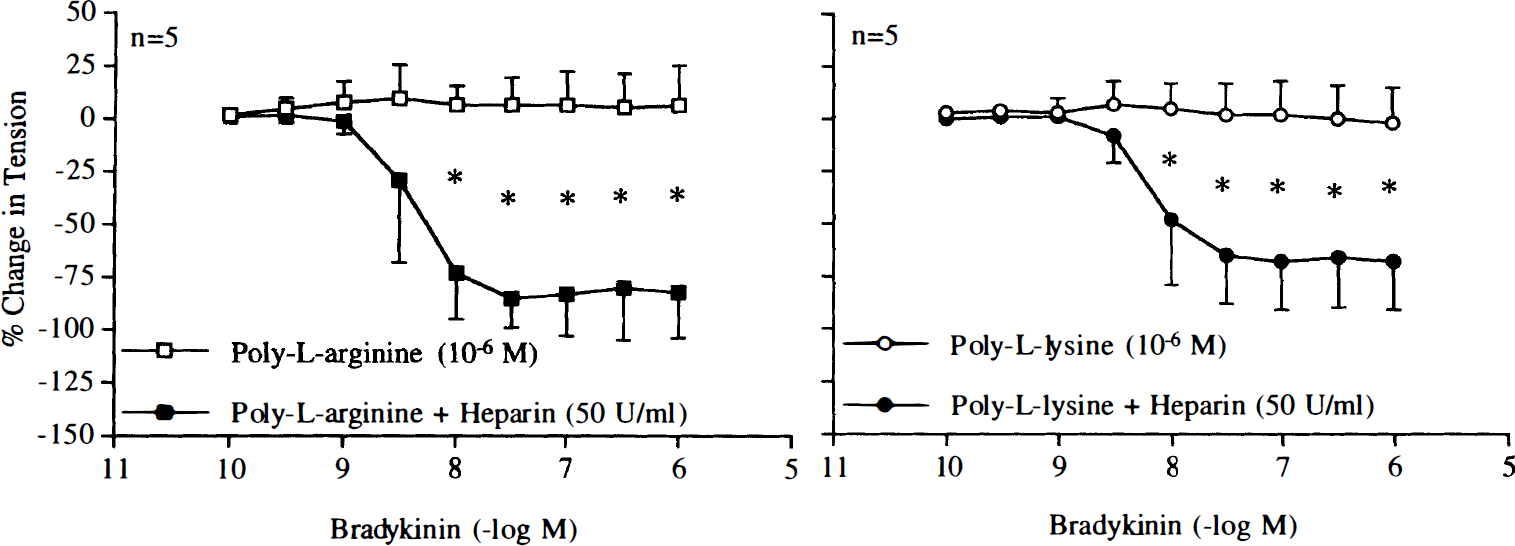
Concentration–response curves to bradykinin obtained in canine basilar arteries with endothelium pretreated with poly-L-arginine (10–6 M) or poly-L-lysine (10–6 M) in the absence and presence of heparin (50 U/ml). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 3.8 ± 1.3 g (n = 5) and 4.4 ± 1.9 g (n = 5) for control rings of poly-L-arginine and rings treated with heparin, 100% = 5.0 ± 0.8 g (n = 5) and 5.0 ± 0.8 g (n = 5) for control rings of poly-L-lysine and rings treated with heparin, respectively]. *Difference between control rings and rings treated with heparin is statistically significant (p < 0.05).
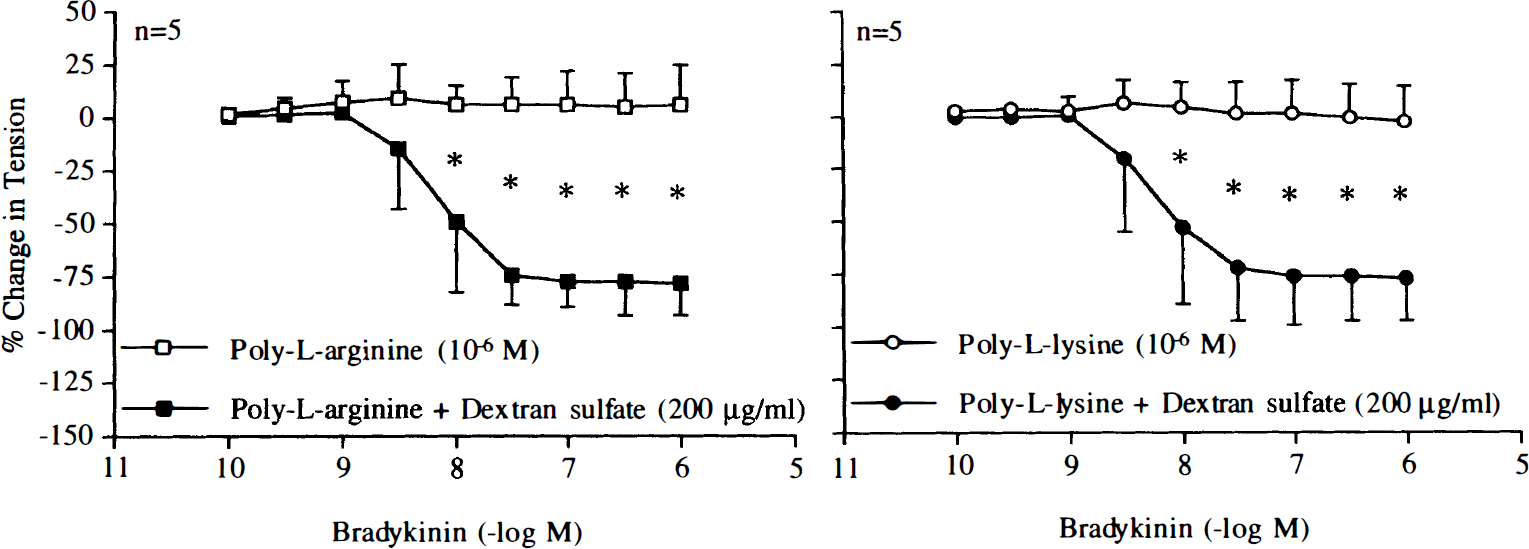
Concentration–response curves to bradykinin obtained in canine basilar arteries with endothelium pretreated with poly-L-arginine (10–6 M) or poly-L-lysine (10–6 M) in the absence and presence of dextran sulfate (200 mg/ml). Data are shown as means ± SD and expressed as percent of maximal relaxation induced by papaverine [3 × 10–4 M; 100% = 3.8 ± 1.3 g (n = 5) and 4.2 ± 1.0 g (n = 5) for control rings of poly-L-arginine and rings treated with dextran sulfate, 100% = 5.0 ± 0.8 g (n = 5) and 4.8 ± 0.9 g (n = 5) for control rings of poly-L-lysine and rings treated with dextran sulfate, respectively]. *Difference between control rings and rings treated with dextran sulfate is statistically significant (p < 0.05).
DISCUSSION
This is the first study to examine the effect of the synthetic cationic polypeptides poly-L-arginine and poly-L-lysine in cerebral arteries. Our results provide several novel observations: (a) In canine basilar arteries, low concentrations (10–8–10–7 M) of poly-L-arginine and poly-L-lysine cause endothelium-dependent relaxations; (b) in concentrations higher than 10–7 M, the cationic polypeptides activate smooth muscle cells, inducing endothelium-independent contractions; and (c) higher concentrations of the cationic polypeptides (10–6 M) impair endothelium-dependent relaxations in response to bradykinin and A23187.
Endothelium-dependent relaxations in response to poly-L-arginine and poly-L-lysine
In isolated canine basilar arteries, poly-L-arginine and poly-L-lysine caused endothelium-dependent relaxations. These relaxations were reduced by a nitric oxide synthase inhibitor, L-NAME, and abolished by a nitric oxide scavenger, oxyhemoglobin, demonstrating that the effects of cationic polypeptides are mediated by increased production of nitric oxide (Martin et al., 1985; Moore et al., 1990). This conclusion is also supported by the observation that L-arginine reversed the inhibitory effect of L-NAME, indicating that poly-L-arginine and poly-L-lysine produce nitric oxide via activation of L-arginine metabolism. Indeed, previous studies in isolated rat aorta (Thomas et al., 1986, 1989; Rapoport et al., 1989) and in isolated bovine pulmonary arteries and veins (Ignarro et al., 1989) also demonstrated that cationic polypeptides induce endothelium-dependent relaxations via production of nitric oxide.
In the present study, negatively charged molecules, heparin and dextran sulfate, inhibited endothelium-dependent relaxations in response to cationic polypeptides. These results are consistent with previous findings on isolated rat aorta (Thomas et al., 1989), suggesting that the cationic properties of poly-L-arginine and poly-L-lysine are responsible for activation of nitric oxide synthase. This conclusion is supported by several lines of evidence. The endothelial nitric oxide synthase is located close to or at the plasma membrane (Boje and Fung, 1990). Upon activation by bradykinin, it is translocated from cell membrane to cytosol, indicating that the intracellular translocation may have important implications for the regulation of the nitric oxide synthase activity (Michel et al., 1993). A recent study demonstrated that electrostatic interaction between anionic membrane phospholipids and basic residues in the endothelial nitric oxide synthase calmodulin-binding domain is an important mechanism responsible for enzyme membrane association (Venema et al., 1995). This finding also provided an explanation for the inability of calmodulin to activate membrane-bound endothelial nitric oxide synthase. It is conceivable that the positive charge of cationic polypeptides may abolish association between anionic membrane phospholipids and the endothelial nitric oxide synthase, resulting in translocation of the enzyme from cell membrane to cytosol, with subsequent stimulation of enzymatic activity and increased production of nitric oxide.
Endothelium-independent contractions in response to poly-L-arginine and poly-L-lysine
Poly-L-arginine and poly-L-lysine caused endothelium-independent contractions that were abolished in the presence of the protein kinase C inhibitor staurosporine (Sugawa et al., 1991). Staurosporine also inhibited contractions to a well established activator of protein kinase C, PDBu (Murray et al., 1992). Therefore, it appears that cationic polypeptides may induce cerebral vascular smooth muscle contractions via activation of protein kinase C. However, it is generally accepted that many protein kinase C inhibitors lack selectivity for protein kinase C over several other kinases (Ohara et al., 1995). Staurosporine is a potent protein kinase C inhibitor, but it may also affect activities of cyclic AMP-dependent protein kinase and myosin light-chain kinase (Ohara et al., 1995). In the present study, staurosporine did not affect contractions to KCl, demonstrating that the inhibitory effect on protein kinase C may be selective. These findings suggest that contractions in response to cationic polypeptides can be explained by the activation of protein kinase C. Our conclusion is also in agreement with the results of a biochemical study demonstrating that in the rat brain, the arginine-rich polypeptides poly-L-arginine and protamine can allosterically activate protein kinase C (Leventhal and Bertics, 1993).
What are the mechanisms responsible for activation of protein kinase C by cationic polypeptides? The negatively charged molecules heparin and dextran sulfate inhibited contractions in response to poly-L-arginine and poly-L-lysine but did not affect contractions to the protein kinase C activator PDBu. These findings indicate that the effects of heparin and dextran sulfate are not mediated by inhibition of protein kinase C. The ability of negatively charged molecules to neutralize the positive charge of poly-L-arginine and poly-L-lysine is the most likely explanation for the inhibition of contractions to cationic polypeptides. These results suggest that protein kinase C may be activated by changes in the electrical properties of the cell membrane exposed to cationic polypeptides.
Inhibitory effect of poly-L-arginine and poly-L-lysine on endothelium-dependent relaxations
Poly-L-arginine and poly-L-lysine abolished endothelium-dependent relaxations in response to bradykinin and calcium ionophore A23187. These results are consistent with previous findings indicating that prolonged contact of arteries with cationic polypeptides, including poly-L-arginine, poly-L-lysine, and protamine, inhibits relaxations mediated by activation of endothelial cells (Chang et al., 1987; Ignarro et al., 1989; Rapoport et al., 1989). The most likely explanation for the inhibitory effect of cationic polypeptides could be related to their ability to activate protein kinase C. A number of previous studies demonstrated that protein kinase C activators inhibit the production, release, and action of nitric oxide in the vascular wall (Cherry and Gillis, 1988; Rubanyi et al., 1989). Thus, poly-L-arginine and poly-L-lysine may activate protein kinase C in endothelial cells. This in turn may inhibit the production and release of nitric oxide (Cherry and Gillis, 1988; Rubanyi et al., 1989). In our experiments, relaxations in response to SIN-1 were also significantly reduced in arteries without endothelium. This finding is in agreement with the previously reported inhibitory effect of phorbol esters on nitric oxide-induced relaxations of vascular smooth muscle (Rubanyi et al., 1989).
Hu et al. (1995) reported that cationic polypeptides inhibit the enzymatic activity of neuronal nitric oxide synthase. This effect was observed in cytosolic preparations of rat cerebellum, indicating that it resulted from direct interaction between cationic polypeptides and nitric oxide synthase rather than activation of a signal transduction pathway in the cell membrane. Our observations do not allow any conclusion regarding the effect of cationic polypeptides on the enzymatic activity of endothelial nitric oxide synthase. Furthermore, we cannot rule out the possibility that this mechanism may contribute to inhibition of endothelium-dependent relaxations observed in the presence of poly-L-arginine or poly-L-lysine.
Higher concentrations of positively charged molecules (>2 × 10–4 M for poly-L-arginine or poly-L-lysine, >10–2 M for protamine) may morphologically damage endothelium, indicating the possible cytotoxicity of cationic polypeptides (Nagy et al., 1983; Chang et al., 1987; Rapoport et al., 1989). In contrast, Sunnergren et al. (1987) demonstrated that protamine at a concentration of 10–2 M does not induce morphological damage to endothelial cells. Thus, whether the 10–6 M concentration of poly-L-arginine and poly-L-lysine affects the morphology of cerebral arteries remains to be determined. However, since poly-L-lysine (10–7 M) can inhibit relaxations in response to endothelium-dependent agonists without morphological damage to endothelial cells (Ignarro et al., 1989), it is possible that a 10–6 M concentration of cationic polypeptides impairs function without affecting endothelial morphology.
Implications for vascular physiology and pathogenesis of cerebral arterial diseases
Endothelial nitric oxide synthase plays a key role in the regulation of cerebral arterial tone (Faraci and Brian, 1994). Shear stress as well as a number of circulatory hormones participate in control of nitric oxide production under physiological conditions. Our results suggest that positively charged molecules may affect nitric oxide synthase activity and smooth muscle cells reactivity to nitric oxide. More importantly, it is clear that electrostatic forces represent an important mechanisms involved in nitric oxide-mediated control of vascular tone.
Interaction between leukocytes and blood vessels results in extensive dilation, loss of endothelial integrity, and plasma extravasation (McEver, 1994). Leukocytes produce oxygen-derived free radicals, which inhibit endothelium-dependent relaxations (Henson and Johnston, 1987; McEver, 1994). In cerebral arteries, activated leukocytes can induce an acute impairment of the endothelium-dependent relaxations (Akopov et al., 1994) and a vasoconstrictor effect contributing to cerebral ischemia, especially in the presence of an atherosclerotic lesion (Faraci et al., 1991). Leukocytes may also play a role in the pathogenesis of cerebral vasospasm due to subarachnoid hemorrhage (Peterson et al., 1990) and hypoperfusion after brain ischemia (Grøgaard et al., 1989). Our results indicate that vascular effects attributed to activated leukocytes could be mimicked by synthetic cationic polypeptides. Indeed, in vivo concentrations of cationic polypeptides released from activated leukocytes can reach concentrations of >10–6 M (Frigas and Gleich, 1986). Thus, our findings may help to explain complex changes in cerebral arterial tone induced by positively charged molecules released from white blood cells.
Footnotes
Acknowledgment:
The authors thank Leslie Smith for technical assistance and Janet Beckman for typing the manuscript.
