Abstract
Results from biochemical and pharmacologic studies suggest that Lcitrulline is taken up by cerebral perivascular nerves and is converted to Larginine for synthesizing nitric oxide (NO). The current study was designed using morphologic techniques to determine whether Lcitrulline is taken up into axoplasm of perivascular nerves and to explore the possibility that conversion of Lcitrulline to Larginine in these nerves is through the argininosuccinate pathway in porcine cerebral arteries. Results from light and electron microscopic autoradiographic studies indicated that dense silver grains representing L-[3H]citrulline uptake were found in cytoplasm of perivascular nerves, smooth muscle cells, and endothelial cells. The neuronal silver grains were significantly decreased in arteries pretreated with glutamine, which has been shown biochemically to block neuronal uptake of Lcitrulline. Results from light and electron microscopic immunohistochemical and histochemical studies indicate that dense nitric oxide synthase-immunoreactive (NOS-I), argininosuccinate synthetase-immunoreactive (ASS-I), and argininosuccinate lyase-immunoreactive (ASL-I) fibers were found in the adventitia of cerebral arteries. NOS-, ASS-, and ASL-immunoreactivities fibers were found in the axoplasm and in the endothelium. In whole-mount preparations, the NOS-I, ASS-I, and ASL-I fibers were completely coincident with NADPH diaphorase fibers, suggesting that axoplasmic ASS, ASL, and NOS were co-localized in the same neurons. These studies provide the first morphologic evidence indicating that Lcitrulline is taken up into cytoplasm of cerebral perivascular nerves and that the axoplasmic enzymes catalyzing the conversion of Lcitrulline to Larginine (for synthesizing NO) by argininosuccinate pathway always are co-localized in same neurons. These results support the hypothesis that Lcitrulline, the by-product of NO synthesis, is recycled to form Larginine for synthesizing NO in perivascular nerves to mediate cerebral neurogenic vasodilation. Results of the current morphologic studies also support the presence of Lcitrulline-Larginine cycle in cerebral vascular endothelium.
Keywords
Cerebral blood vessels from several species have been shown to receive dense nitric oxidergic innervation, and the neurogenic vasodilation in isolated cerebral arteries is mediated predominantly by nitric oxide (NO) (Lee, 1994; Farraci and Brian, 1994; Iadecola et al., 1994). Nitric oxide is synthesized in the presence of nitric oxide synthase (NOS) from L-arginine with L-citrulline as a by-product in various types of cells (Buniatian and Davtian, 1966; Palmer and Moncada, 1989; Bredt and Snyder, 1990). Based on biochemical and pharmacologic studies, we propose that citrulline in cerebral perivascular nerves is actively converted to form L-arginine for synthesizing NO. Thus, an active citrulline-arginine cycle exists in cerebral perivascular nerves. Recycling of L-citrulline to form L-arginine, therefore, provides a means to maintain an adequate supply of NO during neurovascular transmission. This is based primarily on biochemical and pharmacologic evidence that exogenously administered L-citrulline is taken up by cerebral perivascular nerves and is converted to L-arginine (Chen and Lee, 1995; Lee et al., 1996). Direct evidence indicating that exogenously administered citrulline is taken up by cerebral perivascular nerves for conversion to L-Arginine, however, has not been presented, nor is the pathway of citrulline conversion to L-arginine clarified.
The known pathway of L-citrulline conversion to L-arginine is by way of the production of argininosuccinate. In this pathway, Argininosuccinate synthetase (ASS) catalyzes the formation of argininosuccinate from citrulline and aspartate. Argininosuccinate then is cleaved to form arginine and fumarate by argininosuccinate lyase (ASL). Citrulline conversion to arginine has been demonstrated in cytokine-activated macrophages (Wu and Brosnan, 1992), vascular endothelial cells (Hecker et al., 1990; Wu and Meininger, 1993), nonvascular smooth muscle cells, and vascular smooth muscle cells (Hattori et al., 1994). In rat CNS, argininosuccinate synthetase immunoreactivity, citrulline immunoreactivity, and NADPH diaphorase staining were reported to be co-localized in some but not all neurons (Pasqualotto et al., 1991; Arnt-Ramos et al., 1992). Both ASS and ASL immunoreactivities were not always co-localized in rat CNS neurons either (Nakamura et al., 1990). These results suggest that only certain neurons in CNS can recycle citrulline to form arginine.
The current study was designed to investigate, by autoradiography, the distribution of L-[3H]citrulline in porcine cerebral perivascular nerves and to explore, by immunocytochemical and histochemical study, the possibility of the presence of a complete citrulline-arginine cycle in these nerves. Thus, possible co-localization of NADPH diaphorase staining (a marker for NOS), ASS, and ASL was examined.
MATERIALS AND METHODS
Chemicals
L-glutamine, N-2-hydroxyethylpiperazine-N'-2-ethanesulfonic acid (HEPES) and bovine serum albumin (fatty-acid free) were purchased from Sigma Chemical Co. (St. Louis, MO, U.S.A.). L-[3H]citrulline (specific activity 5.5 Ci/mmol) was obtained from Moravok Biochemicals, Inc. (Brea, CA, U.S.A.).
Biochemical assay of citrulline uptake in porcine cerebral arteries
After removing surrounding tissues, the arteries were cut into segments. The uptake of L-citrulline into cerebral arteries was carried out by incubating the arteries with L-[3H]citrulline (6.4 µCi/mL) at 37°C for 30 minutes in oxygenated (95% oxygen-5% carbon dioxide) Krebs' bicarbonate solution supplemented with 5 mmol/L D-glucose, 20 mmol/L HEPES (pH 7.4), and 1% bovine serum albumin. For glutamine-pretreated group, arteries were incubated with L-[3H]citrulline (6.4 µCi/mL) in the presence of 0.5 mmol/L L-glutamine. The latter has been shown to block uptake of L-citrulline into cerebral perivascular nerves (Chen and Lee, 1995; Lee et al., 1996).
After incubation, the labeled tissues were washed with phosphate-buffered saline (PBS) and each piece of artery was cut into two parts. One part was blotted and weighed immediately and then digested with 1 mL of BTS-450 (Beckman, Beckman Instruments, Inc., Fullerton, CA, U.S.A.) at 37°C in closed vials until the tissue dissolved, and the radioactivity was measured with a liquid scintillation counter (Beckman LS-5800). The other part of the artery was processed for autoradiography.
Light microscopic autoradiography
Radiolabeled arteries described above were fixed in 2.5% glutaraldehyde in 0.13 mol/L sodium cacodylate buffer at 4°C for 4 hours. The tissues then were transferred to cold buffer for 20 hours and followed by postfixation in 1.5% OsO4 at 4°C for 2 hours. After dehydration through a graded series of ethanol, tissues were embedded in Poly-bed 812 resin. Cross-sections of the Poly-bed 812-embedded radiolabeled arteries of 1.5-µm thickness were cut and transferred to cleaned glass slides. The slides were dipped into melted Ilford L4 emulsion diluted 1:1 with distilled water. After the emulsion was dried, the slides were placed in light-tight boxes containing CaCl2 as a drying agent and left exposed in a refrigerator at 4°C. After 2 to 3 weeks, one of the slides in each group was removed and developed with Kodak D19 (Eastman Kodak Co., Rochester, NY, U.S.A.) for 2 to 3 minutes at room temperature, rinsed in distilled water, and fixed with 30% sodium thiosulfate for 15 minutes (Yu and Lee, 1989).
Electron microscopic autoradiography
Ultrathin sections of Poly-bed 812-embedded radiolabeled arterial preparations were cut and placed on formvar-coated nickel grids and then coated with 1:2 diluted Ilford L4 emulsion. The grids were held in a dry, light-tight box at 4°C for several months for exposure, and then developed and fixed as described for light microscopic autoradiography. The ultrathin sections were stained with 2.5% uranyl acetate and lead citrate and observed under a Hitachi 700 transmission electron microscope (Yu and Lee, 1989).
Quantitative analysis of silver grains
The silver grains representing citrulline uptake at light microscopic autoradiography were quantified from slides by MCID/M2 Image Analysis System (Imagining Research, Inc., St. Catharines, Ontario, Canada). The threshold to recognize the maximum number of silver grains with minimum nonspecific readings was established. The number of turned “on” pixels over total pixels in the area examined was obtained. This ratio (grain proportional area) was equivalent to the density of silver grains per defined area.
Immunohistochemistry
Avidin-biotin-peroxidase complex method.
Specific anti-sera to purified ASS and ASL were raised in rabbits. The procedures of enzyme purification, characterization, and specificity of antisera were described elsewhere (O'Brien, 1979; O'Brien and Barr, 1981; Morris et al., 1991; Arnt-Ramos et al., 1992; Shuttleworth et al., 1995). The isolated arteries were fixed either in periodate-lysine-paraformaldehyde or periodate-paraformaldehyde-picric acid-formaldehyde-lysine solution (Miao and Lee, 1990) for 4 hours, and then cleaned of adhering connective tissues. The fixed arteries were washed in 0.01 mol/L PBS, incubated in 0.5% hydrogen peroxide for 30 minutes at room temperature, and were blocked with 1% normal goat serum diluted in 0.05% Triton-X/PBS for 30 minutes at room temperature. After washing in PBS, the specimens were incubated in rabbit antiserum against ASS 1:2000 (O'Brien, 1979; Morris et al., 1991; Arnt-Ramos et al., 1992), ASL 1:2000 (O'Brien and Barr, 1981; Morris et al. 1991), or brain NOS 1:1000 (Affinity BioReagents) for 24 to 48 hours at 4°C. After another wash in PBS, specimens were incubated in affinity purified biotinylated goat anti-rabbit IgG antibody (1:200 diluted in PBS, Vector Labs, Burlingame, CA, U.S.A.) at room temperature for 1 hour and were incubated in 1:100 avidin-biotin-peroxidase complex (Vector Labs) at room temperature for another hour (Hsu et al., 1981). Specimens then were placed in a solution of diaminobenzidine peroxidase substrate (Vector Labs) for 2 to 5 minutes. Immunoreactivities, as indicated by brownish end products of diaminobenzidine, were examined under a Zeiss universal light microscope and were photographed. To examine possible co-localization of ASL, ASS, and NOS, the specimens were washed in PBS and processed for NADPH diaphorase biochemical study, as described later (Chen and Lee, 1995).
Immunofluorescence method.
To further examine possible co-localization of NOS, ASS, and ASL in cerebral perivascular nerves, a second method, indirect immunofluorescence (Yu and Lee, 1989), was used. After fixation and incubation with specific antibodies as described earlier, specimens were incubated with biotinylated goat anti-rabbit IgG antibody for 4 hours at 4°C, washed in PBS, and incubated for 4 hours at 4°C with fluorescein avidin D (Vector Labs). The specimens then were rinsed in PBS and coverslipped with Vectashield mounting medium (Vector Labs) for photography under a Zeiss universal fluorescence microscope fitted with proper filters. After taking photographs of immunofluorescence fibers, the specimens were washed in PBS and processed for NADPH diaphorase histochemical study, as described later.
NADPH diaphorase histochemistry
After fixation and incubation with specific antibodies to demonstrate immunoperoxidase labeling, or immunofluorescence labeling and photographing as described earlier, arterial specimens were incubated in 0.1 mol/L phosphate buffer (pH 8.0) containing 0.5 mg/mL of NADPH (reduced form) (Sigma), 0.1 mg/mL of nitro blue tetrazolium, and 0.3% Triton X-100 at 37°C for 2 hours (Chen and Lee, 1995). The tissues were rinsed with PBS, mounted in buffered glycerol (PBS:glycerol = 1:3), and examined under a Zeiss universal light microscope.
Immunoelectron microscopy
As described elsewhere (Yu and Lee, 1992), after immunoperoxidase staining, specimens were postfixed with 1.5% osmium tetroxide for 1 hour and dehydrated with ethanol after diaminobenzidine reaction, and then embedded in poly-bed 812 resin. Ultrathin sections were cut and stained with 2.5% uranyl acetate for 30 minutes and observed under a Hitachi 700 electron microscope.
Controls
The specificity of the immunolabeling was tested by omitting ASS or ASL antiserum and biotinylated goat anti-rabbit IgG independently, as well as by using nonimmune normal rabbit serum or nonimmune normal goat serum. For control of NADPH diaphorase activity, NADPH was omitted from the incubation medium. This resulted in elimination of NADPH diaphorase reaction product in perivascular nerves (Chen and Lee, 1995).
Statistical analysis
Statistical analysis was performed using Student's t-test. A P value of less than 0.05 was considered as significant.
RESULTS
Citrulline uptake in cerebral arteries
Light microscopic autoradiographic examination of 30 minutes' L-[3H]citrulline uptake in cerebral arteries showed that silver grains were distributed densely in perivascular nerves, smooth muscle cells, and endothelial cells (Fig. 1). At the ultrastructural level, silver grains were found in the axoplasm of perivascular nerves and Schwann cells (Fig. 2A). The grains found in extracellular space account for less than 5% of those found in the nerves and Schwann cells. Silver grains also appeared in the cytoplasm of smooth muscle cells and endothelial cells (Figs. 2B and 2C). Uptake of L[3H]citrulline by measuring silver grains in perivascular nerves, smooth muscle cells, and endothelial cells was significantly reduced in the presence of L-glutamine (0.5 mmol/L; P < 0.001) (Fig. 3).
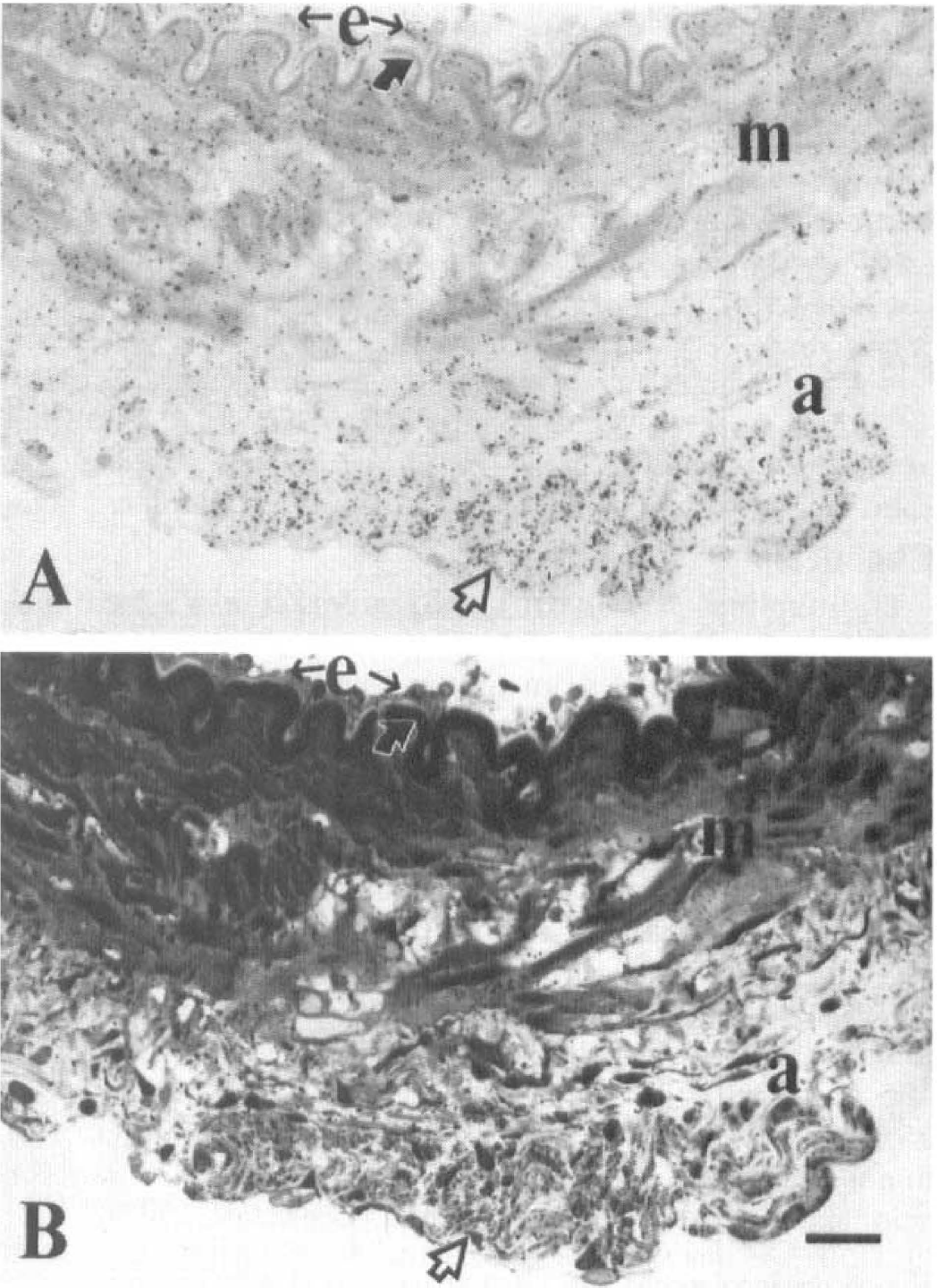
Light microscopic autoradiograph of L-[3H]citrulline uptake (30 minutes) in a basilar artery.
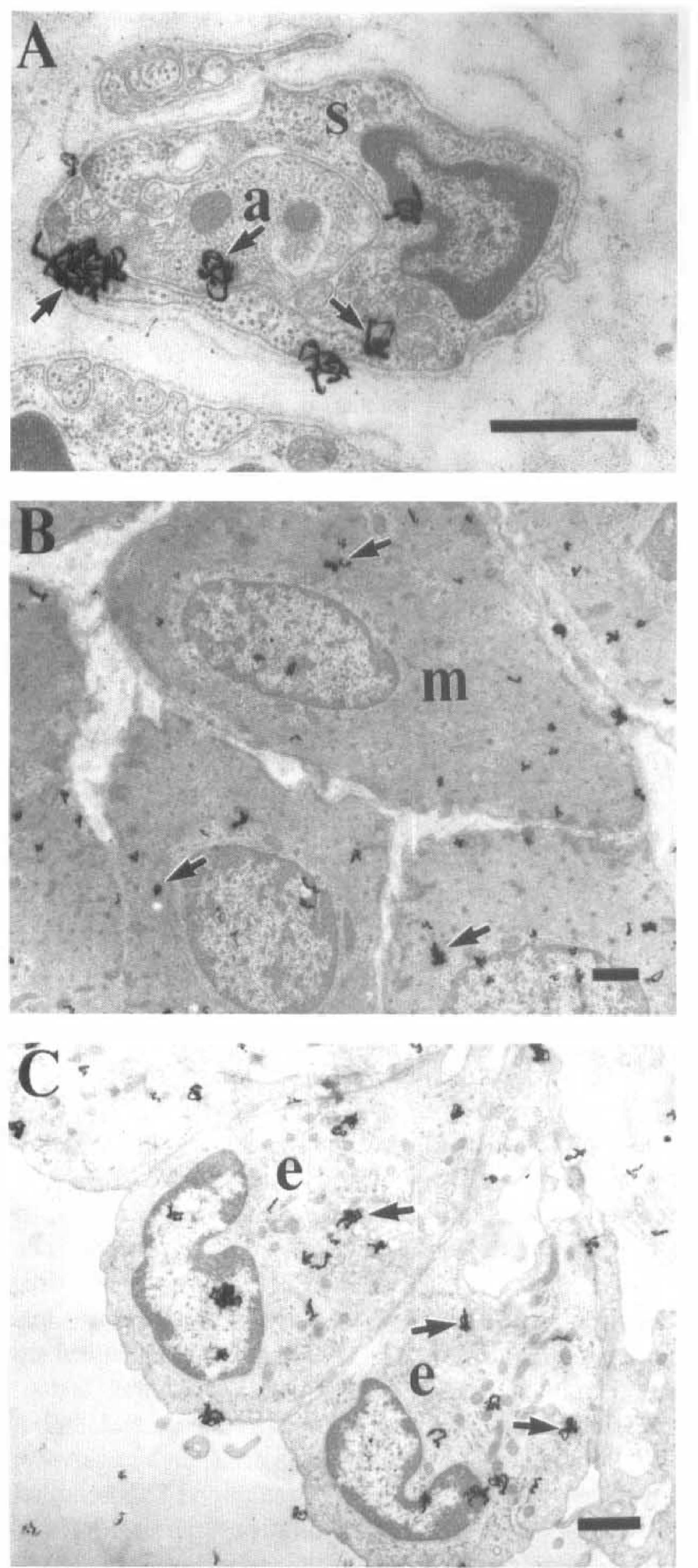
Electron microscopic autoradiographs of L-[3H]citrulline uptake (30 minutes) in the basilar artery. Silver grains (arrows) were found in axoplasm (a)
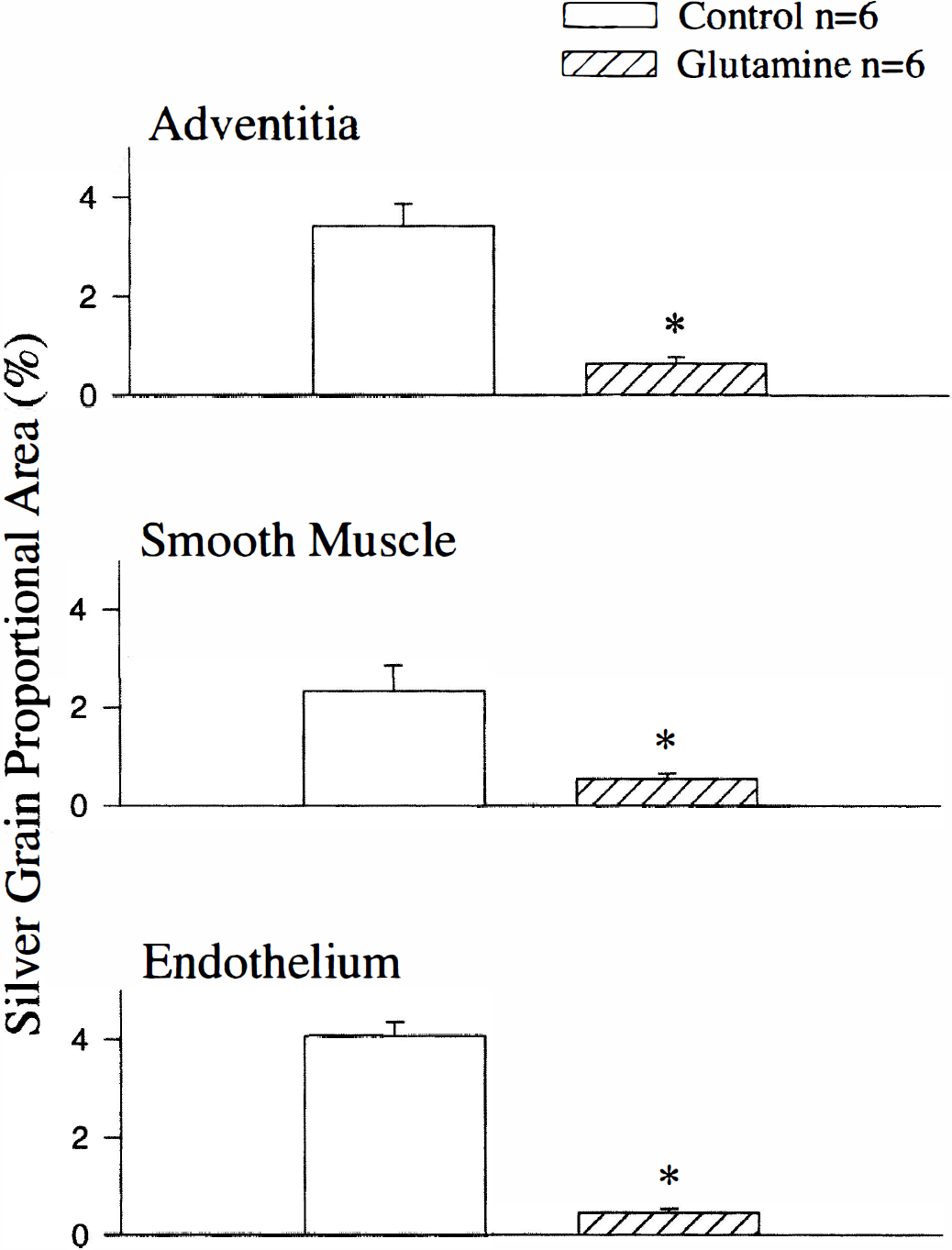
Histogram depicting the density of [3H]-citrulline silver grains at light microscopic level in perivascular nerve-containing adventitia, smooth muscle cells, and endothelial cells in basilar arteries. Glutamine (0.5 mmol/L) pretreatment resulted in a significant decrease of silver grains in the adventitia (*P < 0.001), smooth muscle cells (*P < 0.001), and endothelial cells (*P < 0.001). Data represent mean ± SEM; n indicates number of experiments in each group.
Distribution of NOS-I, ASS-I, ASL-I, and NADPH diaphorase fibers in cerebral arteries
Using immunoperoxidase method, the whole mount cerebral arteries were found to express nitric oxide synthase-immunoreactive (NOS-I), argininosuccinate synthetase-immunoreactive (ASS-I), argininosuccinate lyase-immunoreactive (ASL-I) (as indicated by the brown color), and NADPH diaphorase (as indicated by the blue color) fibers (Fig. 4). These fibers were contained in bundles of various sizes, and fine fibers ran longitudinally or spirally along the walls of the arteries. The density and distribution pattern of NOS-I, ASS-I, ASL-I, and NADPH diaphorase fibers were similar. ASS-I, ASL-I, NOS-I and NADPH diaphorase fibers were more abundant in large arteries than their smaller branches (not shown). Examination of cross-sections of the arteries showed that immunoreactivities of NOS, ASS, and ASL, and NADPH diaphorase was present in nerve profiles in the adventitia and in the endothelium (Fig. 5). At the ultrastructural level, NOS-I, ASS-I, and ASL-I were found in the axoplasm but were not found in association with Schwann cells (Fig. 6). Immunoreactivities of these three enzymes were not observed in both whole-mount preparations and transverse sections by omitting primary antibody or incubating tissues with only normal rabbit or normal goat serum (data not shown).
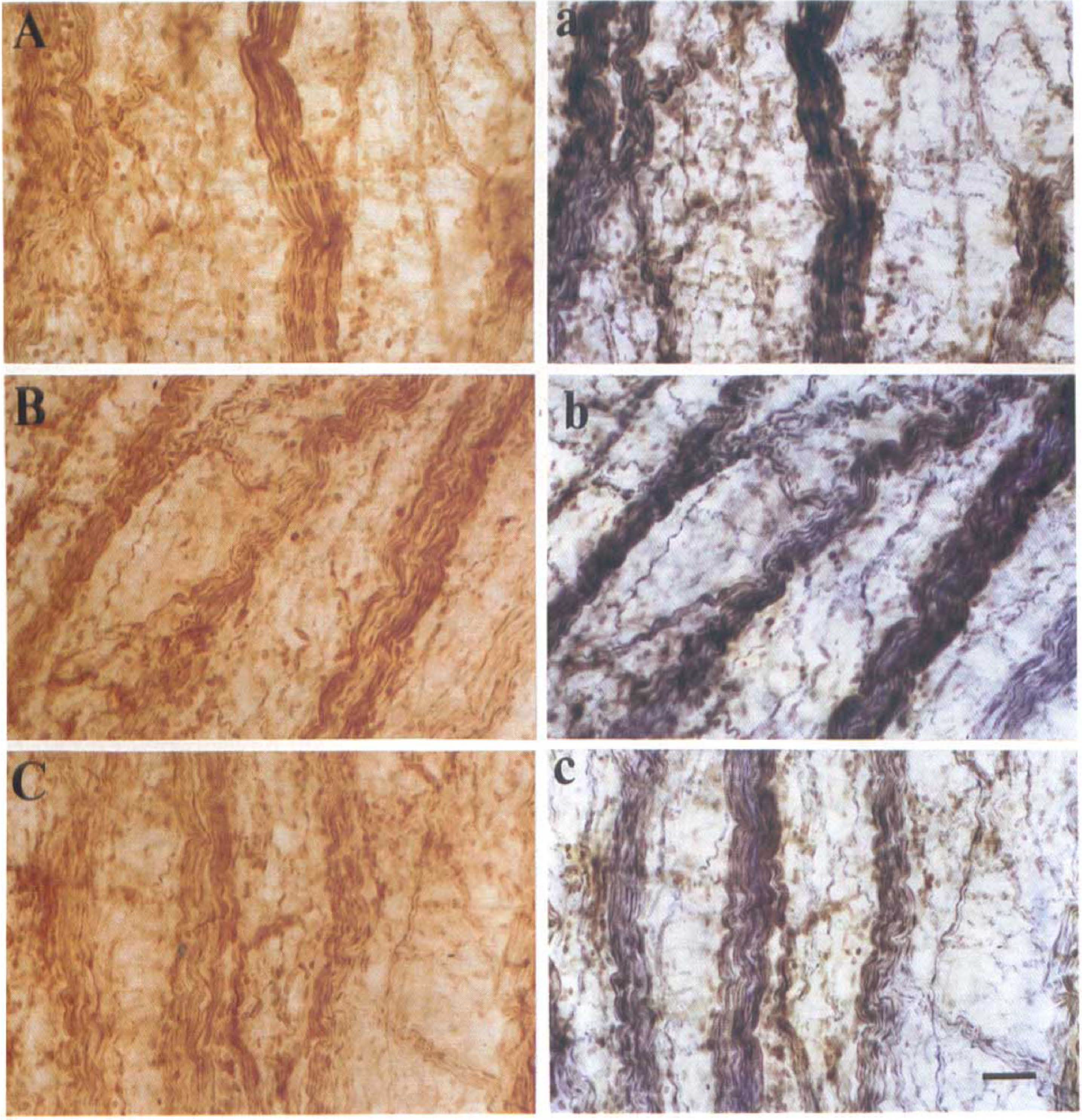
Light photomicrographs showing complete coincident pattern of nitric oxide synthase-immunoperoxidase
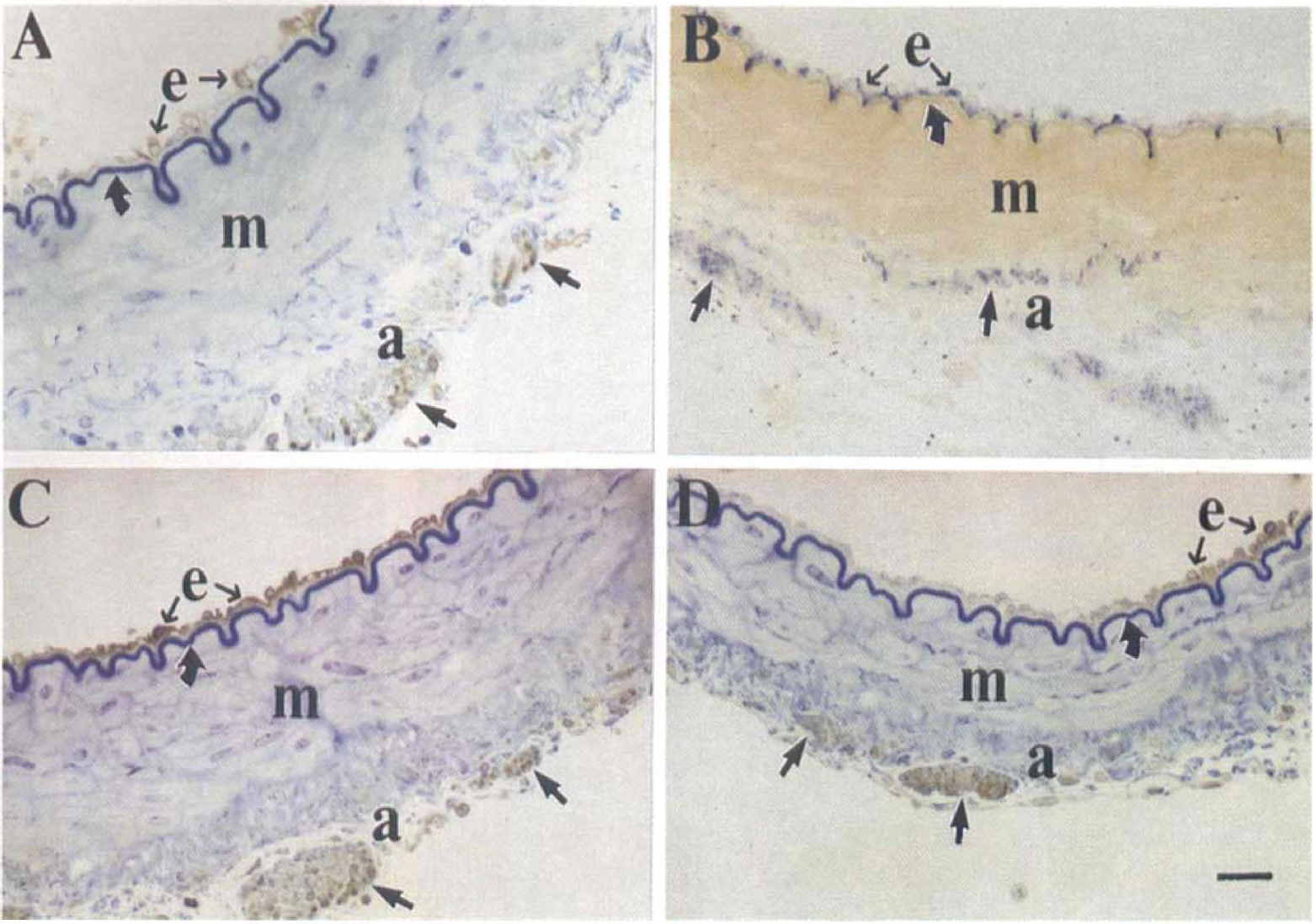
Cross-sections of nitric oxide synthase immunoreactivities
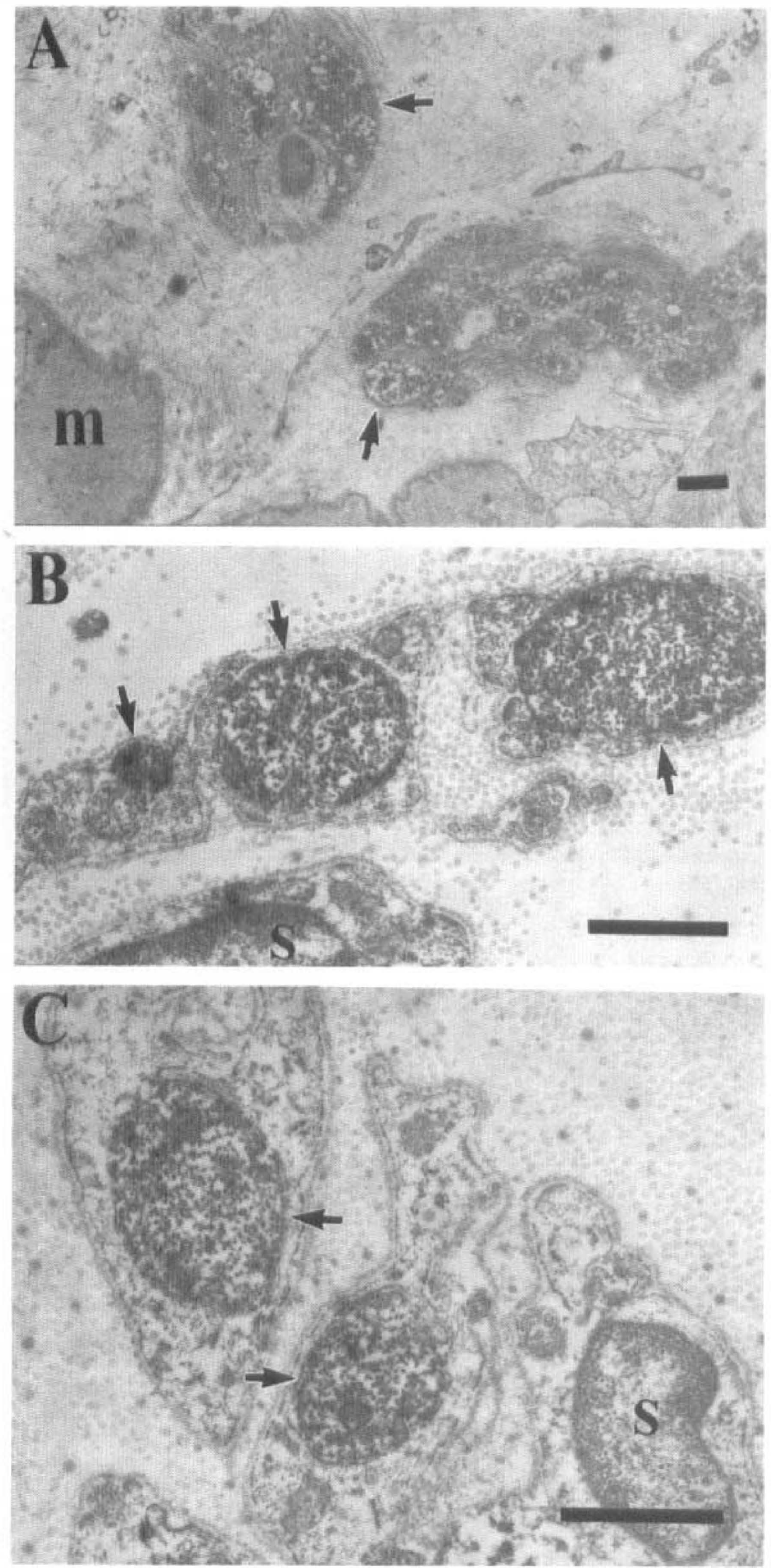
Electron photomicrographs showing axon profiles of perivascular nerves in porcine middle cerebral arteries. Nitric oxide synthase immunoreactivities
Coincident pattern of NOS-I, ASS-I, ASL-I, and NADPH diaphorase fibers
Using a combination of immunoperoxidase labeling and NADPH diaphorase staining, and a combination of immunofluorescence labeling and NADPH diaphorase staining methods, identical results were obtained. That is, all NOS-I fibers and all NADPH diaphorase fibers (both bundles and fine fibers) were found to coincide completely with each other (Figs. 4 and 7). Similar results were found for all ASS-I (or ASL-I) and all NADPH diaphorase fibers (Figs. 4 and 7).
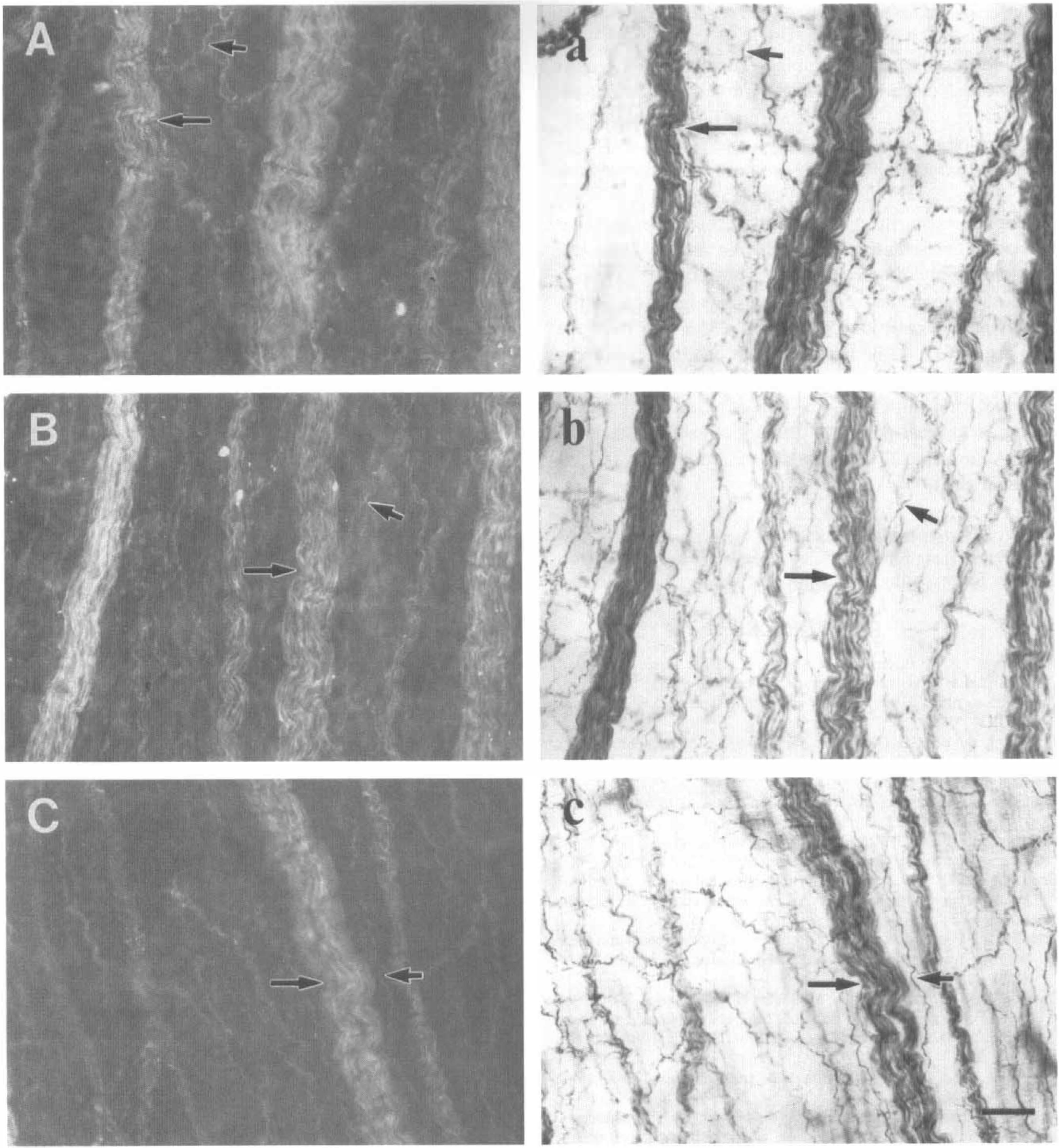
Light micrographs showing complete coincident pattern of nitric oxide synthase immunofluorescence
DISCUSSION
The current study demonstrated that L-citrulline was taken up into cytoplasm of cerebral perivascular nerves. It also demonstrated that NOS, ASS, and ASL are axoplasmic enzymes and are co-localized in the same neurons innervating cerebral arteries. These results, together with our previous pharmacologic and biochemical findings, suggest that L-citrulline, the byproduct of NO synthesis, is recycled to form L-arginine by the argininosuccinate pathway in cerebral perivascular nerves.
The presence of a vasodilator innervation to cerebral circulation was first described as cholinergic in nature, and that acetylcholine was suggested to be the transmitter mediating relaxation of the smooth muscle cells (Chorobski and Penfield, 1932). The neurogenic vasodilation in isolated cerebral arteries from several species, however, was not blocked by atropine or enhanced by physostigmine (Lee et al., 1978). Furthermore, in isolated cerebral arterial ring segments without endothelial cells, acetylcholine induced constriction exclusively, whereas the transmural nerve stimulation (TNS)-elicited relaxation (neurogenic vasodilation) persisted (Lee, 1980). These results indicate that acetylcholine is not the primary transmitter for cerebral vasodilation.
Hemolysates (the membrane-free erythrocytes) were the first substance shown to specifically abolish cerebral neurogenic vasodilation (Lee et al., 1984). Similar results were obtained with hemoglobin, the major component of hemolysates (Linnik and Lee, 1989). Neither hemolysates nor hemoglobin, however, affected relaxation induced by exogenous vasoactive intestinal polypeptide (VIP), calcitonin gene-related peptide (CGRP) or β-adrenoceptor agonists (Lee et al., 1988). These results suggest that the vasodilator mediating TNS-elicited relaxation was sensitive to hemoglobin preparations (Lee et al., 1985). Together with the finding by Martin and others (1985), that endothelium-derived NO was inactivated by hemoglobin, and that both endothelium-derived NO (Martin et al., 1985) and TNS-elicited relaxation in cerebral arteries were accompanied by increased synthesis of cGMP (Lee et al., 1989), it was suggested that a neuronal substance similar to endothelial-derived NO or a related substance mediated cerebral neurogenic vasodilation (Lee et al., 1989).
Subsequently, the neurogenic vasodilation in isolated cerebral arteries was found to be abolished by inhibiting NOS, and the inhibition was specifically reversed by L-arginine, the substrate of NOS (Toda et al., 1990; Lee and Sarwinski, 1991). Thus, in isolated cerebral arteries, the TNS-elicited vasodilation is predominantly mediated by NO (Lee and Sarwinski, 1991; Lee 1994). Additional work has been established that the chemical nature of the endogenously released vasodilator substance on TNS is identical to that of the exogenous NO (Chen and Lee, 1993).
There are, however, some questions as to whether NO is released directly from the perivascular nitric oxidergic nerves. Some reports indicate that in bovine cerebral arteries NO is indirectly released from endothelial cells in response to an unknown substance originating in perivascular nerves (Gonzalez and Estrada, 1991) or in the goat cerebral arteries from enzymatic degradation of neural peptides such as VIP and CGRP after their release in the synapses (Gaw et al., 1991). These two possibilities, at least in the porcine cerebral arteries, are unlikely (Lee, 1994). The TNS-elicited cerebral vasodilation in the pig and cat was enhanced in arteries without endothelial cells (Lee, 1982; Lee et al., 1984). Also, inhibition of NOS by nitro-L-arginine did not affect exogenous VIP- or CGRP-induced relaxation in the cat and pig cerebral arteries (Lee et al., 1993). This latter finding suggests that VIP and CGRP, which possess two molecules of L-arginine, do not release NO in the synapse in cerebral arteries of the cat and pig. These results favor the hypothesis that NO is released directly from cerebral perivascular nitric oxidergic nerves.
Evidence for release of NO directly from cerebral perivascular nerves was first presented based on biochemical and pharmacologic findings that there is an active conversion of exogenously administered citrulline to arginine in cerebral perivascular nerves (Chen and Lee, 1995), suggesting that on synthesis and release of NO from L-arginine, the byproduct L-citrulline is recycled to form L-arginine. This is further supported by results of the current study that L-citrulline is indeed taken up into axoplasm, where ASS, ASL, and NOS are located. Silver grains also were found in Schwann cells, suggesting that Schwann cells also can take up L-citrulline. The Schwann cells, however, did not possess NOS, ASS, and ASL immunoreactivity. Lack of NOS immunoreactivity in Schwann cells also was found in the Sprague-Dawley rat basilar arteries (Loesch et al., 1994). The uptake of L-citrulline into Schwann cells may therefore provide additional source of L-citrulline to be transported into axoplasm for NO synthesis.
Our previous biochemical studies demonstrated that L-glutamine inhibited L-citrulline uptake into normal cerebral arterial walls, but did not significantly affect that in arteries after cold-storage denervation, suggesting that glutamine blocked L-citrulline uptake into perivascular nerves (Chen and Lee, 1995). This is supported by the current autoradiographic studies. The silver grains indicative of L-citrulline uptake in the adventitia were found primarily (95%) in the cytoplasm of perivascular nerves and Schwann cells. The decrease by about 80% of silver grains (Fig. 3) in the adventitia in the presence of L-glutamine suggests that L-glutamine has to block the neuronal uptake of L-citrulline. The current study also demonstrates that L-glutamine blocks L-[H3] citrulline uptake into smooth muscle and endothelial cells. This is different from results of biochemical studies where glutamine did not significantly affect L-[C14] citrulline uptake into vascular smooth muscle in cold storage-denervated arteries or into endothelial cells (Chen and Lee, 1995). This was explained by lack of sensitivity of the method in measuring differences in L-citrulline uptake, particularly in the monolayer endothelial cells (Chen and Lee, 1995).
According to the known urea cycle, L-citrulline is converted to L-arginine through the argininosuccinate pathway catalyzed by ASS and ASL (Ratner, 1973). This also appears to be true in cerebral perivascular nerves, based on results of the current study. The distribution pattern of NOS-I, ASS-I, and ASL-I fibers is similar to that of NADPH diaphorase fibers. NADPH diaphorase has been shown to be identical to NOS (Hope et al., 1991; Dawson et al., 1991; Schmidt et al., 1992), although several reports indicate that NADPH diaphorase staining in certain cells is not because of NOS (Kashimoto et al., 1993; Santer and Symons, 1993). Results of our current studies indicate that all NOS-I fibers (fine and bundle fibers) coincide with NADPH diaphorase fibers. This is based on identical results using two different combination “staining” methods: immunoperoxidase labeling combined with NADPH diaphorase staining, and immunofluorescence labeling combined with NADPH diaphorase staining. Our results support that NOS and NADPH diaphorase in cerebral perivascular nerves are identical enzymes. Similarly, most ASS-I and ASL-I fibers are coincident with NADPH diaphorase fibers using both staining methods. These results suggest that axoplasmic ASS, ASL, and NOS are co-localized in the same neurons, providing essential enzymes for conversion of L-citrulline to L-arginine for synthesis of NO in the same neurons in cerebral arteries.
Simultaneous localization of NOS, ASS, and ASL immunoreactivities in the same tissue was not performed because the available primary antibodies to these three proteins all were raised in the rabbit. The combined NADPH diaphorase histochemistry with immunoperoxidase and with immunofluorescence methods for investigating possible co-localization of ASS and ASL were therefore used as alternatives.
Applying similar antibodies as those used in the current study, Shuttleworth and others (1995) report that all NOS-containing enteric neurons also contain ASS and ASL-a result similar to that found in the current study. Using these same antibodies, Arnt-Ramos and associates (1992) and Nakamura et al. (1990), however, reported that in the rat CNS only certain neurons contain both ASS and NADPH diaphorase, whereas some neurons contain only ASS, ASL, or NOS. In these CNS neurons, regeneration of arginine from citrulline will does not occur in the same neurons, but may involve interneuronal transport of intermediate metabolites.
Several studies show that some NO-releasing cells such as endothelial cells, macrophage, and vascular smooth muscle cells are capable of regenerating arginine from citrulline (Hecker et al., 1990; Wu and Brosnan, 1992; Wu and Meininger, 1993; Hattori et al., 1994). In the current studies, cerebral vascular endothelial cells express NOS-I, ASS-I, and ASL-I and are positive for NADPH diaphorase staining. The presence of NOS-I endothelial cells suggests that cerebral arterial endothelial cells also contain type I NOS. A similar result was found in the rat basilar arteries (Loesch et al., 1994). Although co-localization of NOS, ASS, and ASL in endothelial cells was not examined, our results from previous pharmacologic studies (Lee et al., 1996) indicate that L-citrulline, like L-arginine, reverses the enhanced basal tone induced by nitro-L-arginine. Together these results suggest that cerebral vascular endothelial cells are capable of recycling L-citrulline to form L-arginine for synthesizing NO.
In summary, results of our previous and current work indicate that an active conversion of L-citrulline to form L-arginine exists in cytoplasm in cerebral perivascular nerves and endothelial cells, allowing maintenance of a constant supply of L-arginine for synthesizing NO to mediate neurogenic and endothelium-dependent vasodilation. The co-localization of NOS, ASS, and ASL in the axoplasm of the same neurons of all cerebral perivascular nerves suggests that the conversion of citrulline to arginine for synthesizing NO in cerebral perivascular nerves is at least partly by the argininosuccinate pathway. A similar pathway also appears to exist in the endothelial cells.
Footnotes
Abbreviations used
Acknowledgements
We thank Ms. Susan Sarwinski and Ms. Xiao-Feng Chang for their technical assistance and Ms. Dawn Melcher for preparing the manuscript.
