Abstract
Transient ischemia increases tyrosine phosphorylation of N-methyl-
Keywords
Transient global ischemia induces several pathophysiologic changes that may lead to neuronal cell death. A role for ionotropic glutamate receptors of both the N-methyl-
The NMDAR subunits NR2A and NR2B are phosphorylated on tyrosine residues (Moon et al., 1994; Lau and Huganir 1995). Tyrosine phosphorylation of the NMDAR may be altered under a variety of conditions including long-term potentiation (Rosenblum et al., 1996; Rostas et al., 1996), taste aversion learning (Rosenblum, et al., 1997), acute exposure to ethanol (Miyakawa et al., 1997), 6-hydroxydopamine lesioning of nigrostriatal dopaminergic neurons (Menegoz et al., 1995), and transient global ischemia (Takagi et al., 1997). In the latter case, the onset of reperfusion after 15 minutes of ischemia was accompanied by a rapid and sustained increase in the tyrosine phosphorylation of NR2A and NR2B in the rat hippocampus, with smaller increases occurring in the striatum and frontal cortex. One consequence of altered tyrosine phosphorylation may be modulation of Ca2+ entry through the NMDAR (Wang and Salter, 1994; Wang et al., 1996).
In addition to regulation of the NMDAR ion channel, tyrosine phosphorylation may link the NMDAR to proteins that contain Src homology 2 (SH2) domains (Gurd and Bissoon, 1997), thereby contributing to the assembly of signaling complexes at the postsynaptic density (PSD). The SH2 domain recognizes phosphotyrosine in the context of specific amino acid sequences, (Songyang et al., 1993; Songyang et al., 1995; Kavanaugh et al., 1995) and is found in a variety of proteins involved in signal transduction including pp60c-src and related tyrosine kinases, phosphatidylinositol 3-kinase p85, Grb2, PLC-γ, and SH2-containing tyrosine phosphatases (reviewed in Pawson, 1995). Src is present in PSDs (Cudmore and Gurd, 1991) and is associated with the NMDAR in hippocampal cells in culture and in embryonic rat spinal cord (Yu et al., 1997). Src has been implicated in regulation of the NMDAR ion channel (Yu et al., 1997) and in the induction of long-term potentiation (Lu et al., 1998). Fyn is a member of the Src family of tyrosine kinases and also is present in the PSD. Fyn phosphorylates the NMDAR and is involved in the regulation of a variety of neuronal activities, including the induction of LTP (Grant, et al., 1992; Suzuki and Okumura-Noji, 1995; Kojima et al., 1997; Köhr and Seeberg, 1996). These findings suggest that the increase in tyrosine phosphorylation of NR2 subunits, which occurs after transient global ischemia (Takagi et al., 1997), might alter the interaction of the NMDAR with Src and Fyn, thereby affecting signaling pathways, which are initiated at the PSD. To investigate this possibility, we analyzed the effect of ischemia on the interaction of NMDAR subunits with Src and Fyn SH2 domains expressed as glutathione-S-transferase (GST) fusion proteins. Because the NMDAR is localized to the PSD, we also examined the effect of ischemia on the presence of Src and Fyn in an isolated PSD fraction. The results demonstrate that transient global ischemia results in an increase in the ability of NR2A and NR2B to bind to the SH2 domains of Src and Fyn and, further, that the association of both tyrosine kinases with PSDs is increased after an ischemic episode.
MATERIALS AND METHODS
Animal surgical procedures and tissue preparation
All procedures using animals were approved by the Animal Care Committee of the University of Toronto and were in accordance with the guidelines established by the Canadian Council on Animal Care. Male Wistar rats (200 to 250 g) were given free access to food and water before surgery. Transient global ischemia was induced as reported (Zhang et al., 1997) using a modified version of the method of Pulsinelli and Brierley (1979) and Kirino (1982). After 1 hour of reperfusion, rats were killed by decapitation. Heads were quickly frozen by immersion in −42°C isopentane for 5 minutes and were stored at −72°C. Before dissection, tissue was warmed to −5° to 0°C. The hippocampus was removed and homogenized in ice cold 0.32 mol/L sucrose containing 0.1 mmol/L sodium orthovanadate, 0.1 mmol/L phenylmethylsulfonyl fluoride, and 5 µg/mL each of antipain, aprotinin, and leupeptin. Protein was determined by the method of Lowry and associates (1951). Homogenates were stored at −72°C until use.
For the preparation of PSDs, animals were treated as described earlier except that the reperfusion time was 6 hours. Frozen heads were warmed to 0°C, and the forebrains from five ischemic animals or five sham-operated animals were dissected and pooled for the preparation of PSDs according to the method of Cho and coworkers (1992). Preliminary experiments indicate that although the yield of PSD from frozen brains was decreased relative to the yield from unfrozen brains, the levels of several PSD-associated proteins, including NR1, NR2A and NR2B, PSD-95, and Src, were unaffected by the freezing step.
Glutathione-S-transferase–SH2 fusion proteins and SH2 binding assay
To prepare the SH2 fusion proteins, bacterial cells containing pGEX expression vectors into which cDNA encoding the SH2 domain of Src or the SH2 domain of Fyn had been subcloned were provided by Dr. T. Pawson (Samuel Lumenfeld Institute, Mount Sinai Hospital, Toronto, Canada). Protein expression was induced with isopropyl β-
Immunoprecipitation and Western blotting
The immunoprecipitation of NR2A and NR2B subunits was performed with antibodies specific for NR2A and NR2B (see later) as described by Takagi and colleagues (1997). For immunoblotting, SDS-solubilized samples from SH2-binding experiments or immunoprecipitations were separated on 8% polyacrylamide gels and transferred to nitrocellulose as described by Gurd and coworkers (1992). Protein blots were reacted with 2A or 2B specific antibodies or antibodies against phosphotyrosine and immunoreactive proteins detected by enhanced chemiluminescence (Pierce Super Signal). Preliminary experiments demonstrated that binding of NMDAR subunits to the SH2 domains was proportional to the amount of homogenate protein between 100 and 400 µg. For quantification, exposed x-ray film was scanned using a Bio-Rad GS 700 gel scanner, and care was taken to ensure that protein loads were within the linear range of response (Gurd et al., 1992). When quantitative comparisons of sham and ischemic tissue were made, smaller amounts of the latter were analyzed to maintain both samples within the linear response range. Statistical comparison between ischemia and sham-operated samples was done using the unpaired Student's t-test.
Antibodies
For immunoprecipitation, antibodies specific for NR2A or NR2B were provided by Dr. R.J. Wenthold (NIH, Bethesda, MD, U.S.A.). For immunoblotting, anti-NR2A from Upstate Biotechnology (Lake Placid, NY, U.S.A.), or R.J. Wenthold, anti-NR2B from Transduction Laboratories (Lexington, KY, U.S.A.), anti-NR1 from Pharmingen (San Diego, CA, U.S.A.), and antibodies to the various NR1 cassettes provided by R.J. Wenthold were used. Antibodies to phosphotyrosine (clone 4G10) and to Src were from Upstate Biotechnology. Antibodies to Fyn and to PSD95 were obtained from Transduction Laboratories and Affinity Bioreagents (Golden, CO, U.S.A.), respectively.
RESULTS
N-methyl-d-aspartate receptor subunits NR2A and NR2B bind to the SH2 domains of Src and Fyn
Both NR2A and NR2B bind to the SH2 domains of PLC-γ, suggesting a possible direct linkage between the receptor and signaling pathways in the postsynaptic cell (Gurd and Bissoon, 1997). The nonreceptor tyrosine kinases Src and Fyn have been implicated in the tyrosine phosphorylation of NMDAR subunits and in the regulation of synaptic activity (Grant et al., 1992; Suzuki and Okumura-Noji, 1995; Köhr and Seeberg, 1996; Yu et al., 1997; Lu et al., 1998). The presence of potential binding sites for the SH2 domains of Src family tyrosine kinases in NR2A (Tyr1292-Asp-Asn-Ile) and NR2B (Tyr1069-Gly-Asn-Ile) (Songyang et al., 1993) suggests that Src and Fyn might bind to the NMDAR through their SH2 domains. To assess this possibility, we incubated solubilized hippocampal proteins from naive animals with the SH2 domains of Src and Fyn expressed as GST fusion proteins and bound to glutathione-agarose. The results in Fig. 1 show that NR2A and NR2B each bound to both SH2 domains. Neither subunit bound to GST-sepharose alone, indicating that binding required the presence of an SH2 domain.
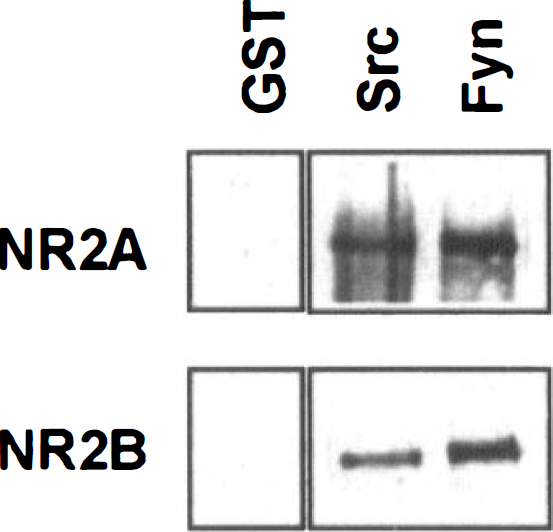
N-Methyl-
Transient global ischemia results in an increase in the binding of NR2A and NR2B to the SH2 domains of Src and Fyn
Transient global ischemia followed by 6 hours of reperfusion induces a marked increase in the tyrosine phosphorylation of the NMDAR subunit NR2A and a smaller increase in the tyrosine phosphorylation of NR2B in the rat hippocampus (Takagi et al., 1997). A similar differential increase in the reactivity of NR2A and NR2B with antiphosphotyrosine antibodies was evident after only 1 hour of reperfusion, the phosphorylation of NR2A and NR2B being elevated sixfold to sevenfold, and threefold over sham-operated controls, respectively (Figs. 2A and 2B). Similar amounts of NR2A and NR2B were immunoprecipitated from sham-operated and ischemic animals, indicating that tyrosine phosphorylation of the receptor subunits was increased after ischemia (Figs. 2C and 2D).
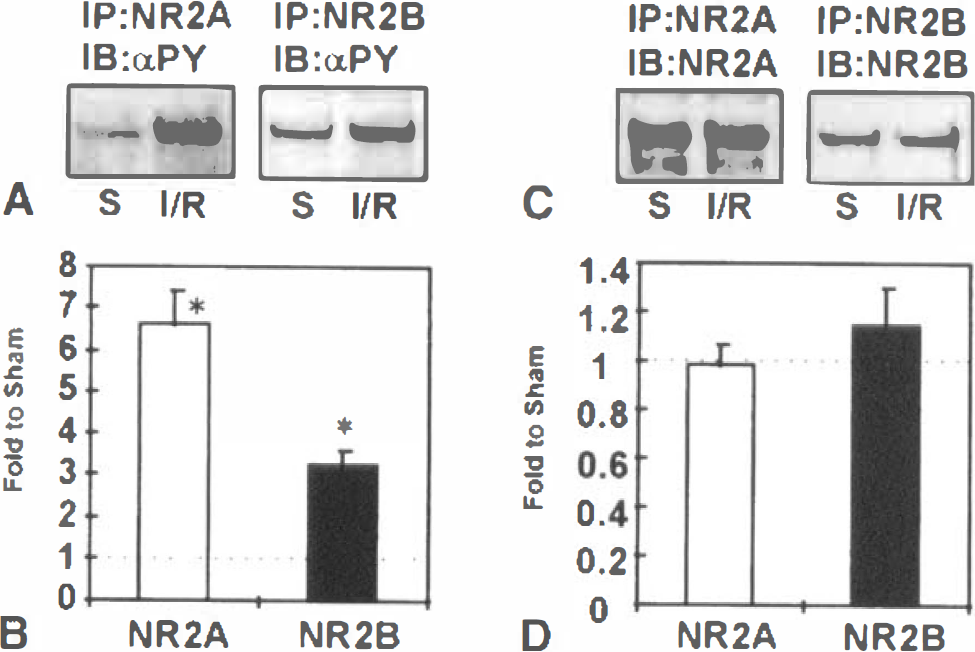
Ischemia/reperfusion enhances the tyrosine phosphorylation of NR2A and NR2B.
The effect of the ischemia-induced increase in tyrosine phosphorylation on the ability of NR2A and NR2B to bind to the SH2 domains of Src and Fyn is shown in Fig. 3. After ischemia and reperfusion, the binding of the two NMDAR subunits to the SH2 domains was enhanced 1.5- to 2.5-fold. To rule out the possibility that the binding reaction might have been limited by the capacity of the SH2 beads, homogenates from ischemic hippocampus were incubated for 10 minutes with ATP to increase tyrosine phosphorylation levels (Gurd et al., 1992) before incubation with SH2-sepharose. Under these conditions, there was a further increase in binding (not shown), demonstrating that the binding of endogenously phosphorylated receptor subunits was not limited by the capacity of the SH2-sepharose.
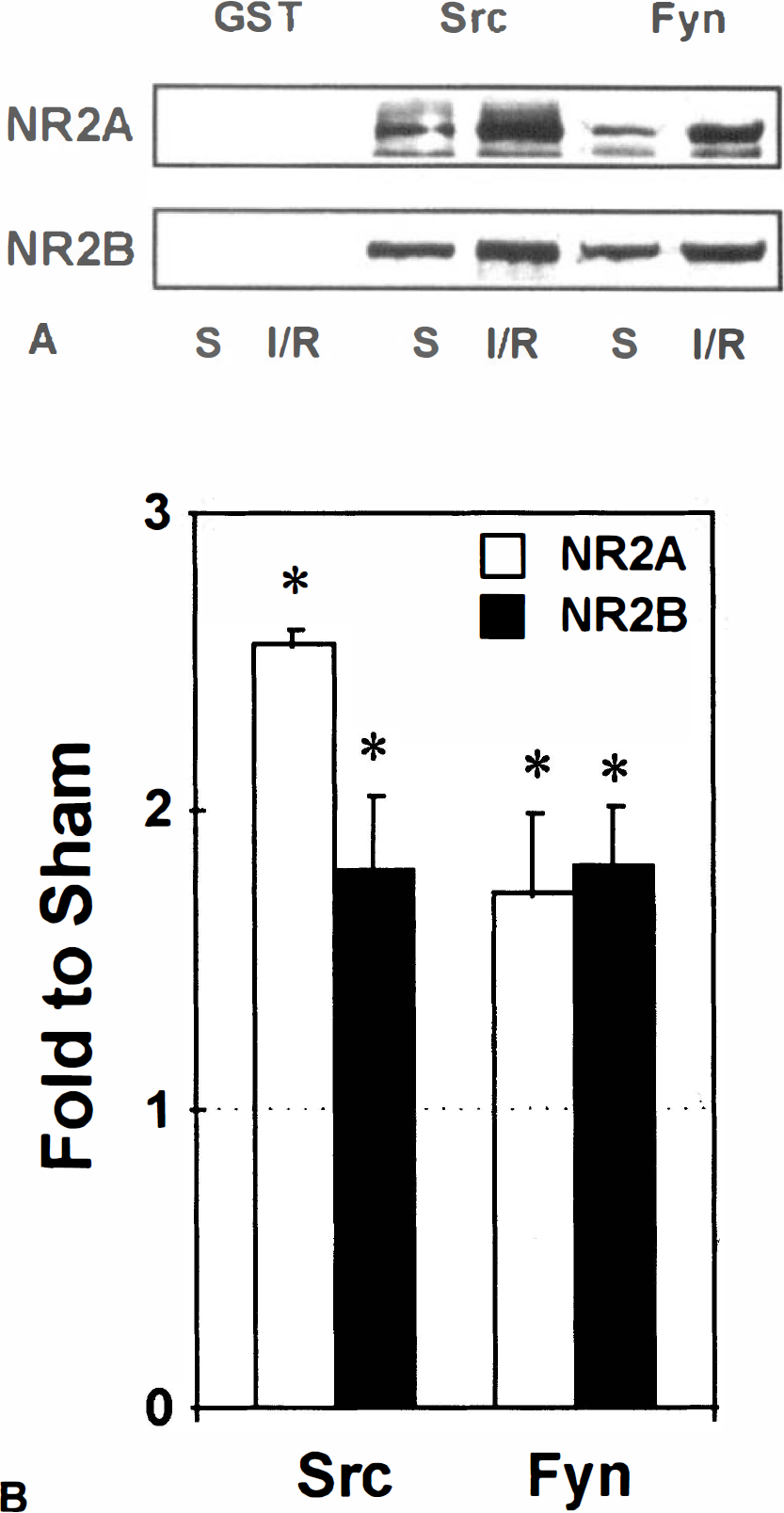
Increased binding of NR2A and NR2B to the SH2 domains of Src and Fyn after ischemia.
To confirm that the interaction of the NMDAR with the SH2 domains was dependent on tyrosine phosphorylation and did not result from binding with peptide sequences that do not contain phosphorylated tyrosine residues (Raffel et al., 1996; Joung et al., 1996; Oligino et al., 1997), proteins from the postischemic hippocampus were dephosphorylated in vitro with an exogenous protein tyrosine phosphatase before incubation with Src SH2-agarose. This treatment prevented binding of NR2A and NR2B to the SH2 domain (Fig. 4A). The amounts of the individual subunits were not altered as a consequence of the dephosphorylation reaction (Fig. 4C), and analysis of the unbound fraction confirmed the presence of NR2A and NR2B subunits that were not tyrosine phosphorylated (Fig. 4B). We also assessed the ability of the high affinity Src SH2-binding peptide, EPQpYEEIPIA (Liu et al., 1993), to inhibit the interaction of NMDAR subunits with the Src SH2 domain. The presence of 10 µmol/L EPQpYEEIPIA completely abolished the binding of both NR2 subunits to Src SH2-sepharose (Fig. 4A). In contrast, the control, nonphosphorylated peptide (EPQYEEIPIA), had no effect on the binding reaction, even at a concentration of 100 µmol/L (Fig. 4A). These results demonstrate that binding of the NMDAR to the Src SH2 domain was dependent on tyrosine phosphorylation of the receptor and, further, that binding required the presence of a free phosphotyrosine binding site in the SH2 domain.
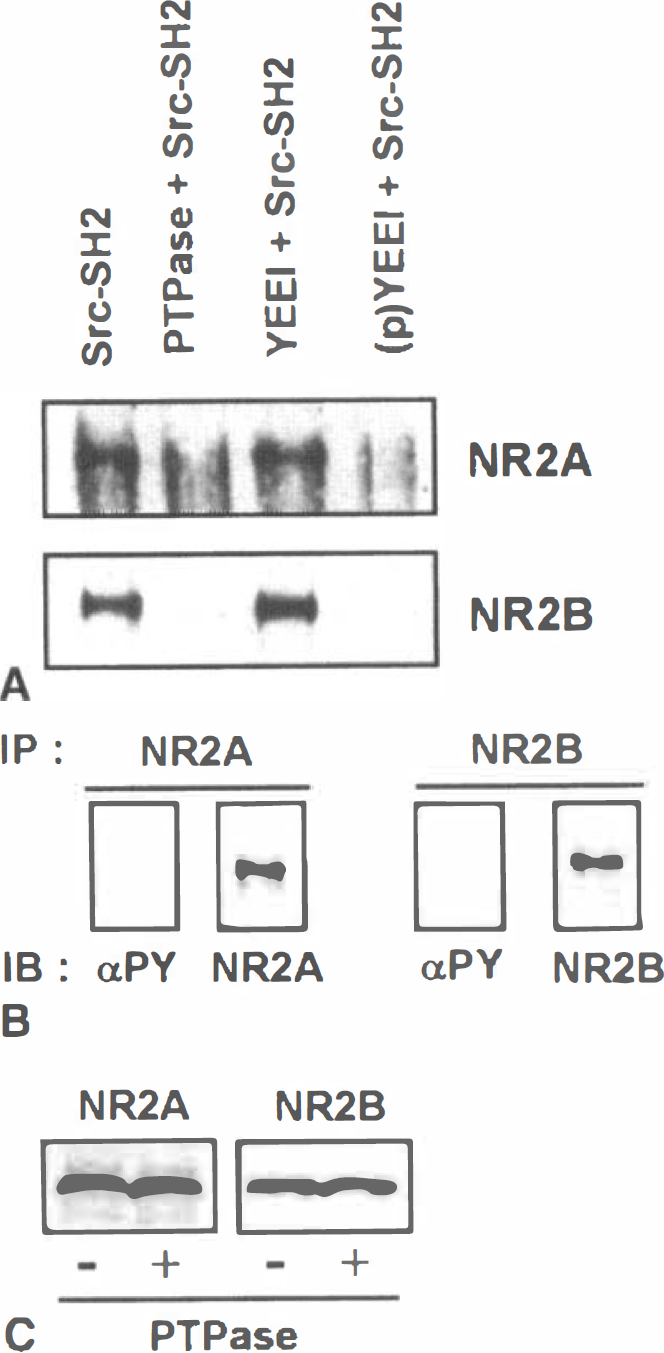
The effect of treatment with protein tyrosine phosphatase or high-affinity Src-binding peptide on the binding of Src-SH2-sepharose to NR2A or NR2B.
Association of Src and Fyn with postsynaptic densities increases after ischemia
Because the NMDAR is concentrated in the PSD (Moon et al., 1994), the enhanced ability of Src and Fyn SH2 domains to bind to the NMDAR after ischemia suggests that ischemia might result in an increase in the association of the corresponding proteins with PSDs. To assess this possibility, we isolated PSDs from sham-operated animals and from animals that had received 15 minutes of ischemia followed by 6 hours of reperfusion. To obtain adequate amounts of protein for analysis, PSDs were prepared from rat forebrains rather than from the hippocampus. We reported previously that tyrosine phosphorylation of NR2A and NR2B is increased in the frontal cortex and the striatum after ischemia, although the increases are smaller than those which occur in the hippocampus (Takagi et al., 1997). The overall protein composition of PSDs from sham-operated and challenged animals was similar (Fig. 5A). Western blotting with anti-PSD95 antibodies showed that the level of PSD95 was lower in PSDs from ischemic compared with sham-operated animals (Fig. 5C, ratio of PSD95 in ischemic to PSD95 in sham-operated animals equals 0.6 ± 0.2, average ± SD, n = 3), which is in accord with the recent observations of Hu and coworkers (1998). The amounts of NR2A and NR2B in the PSD fractions were unaffected by ischemia (Fig. 5C), as were the levels of NR1 and of the individual NR1 cassettes N1, C1, C2, and C2‘ (Fig. 5C). Ischemia resulted in a marked increase in the tyrosine phosphorylation of several PSD proteins, including NR2A and NR2B (Figs. 5A and 5B). In accord with the results described earlier for the hippocampus, the enhanced tyrosine phosphorylation of NR2A and NR2B in the PSD fraction was associated with an increase in the ability of both subunits to bind to the SH2 domain of Src (Fig. 5D). The effect of ischemia on the presence of Src and Fyn in the PSD fraction is shown in Fig. 6. In two different experiments, ischemia resulted in twofold and 2.5-fold increases in the amount of Src that was present in the PSD fraction relative to sham-operated controls. Fyn exhibited a similar ischemia-related increase in its association with the PSD (Fig. 6). In contrast to the results obtained with PSDs, the levels of Src and Fyn in total homogenates, in the P2 fraction, and in synaptosomes were similar in the ischemic and sham-operated samples (Fig. 6), indicating that the increase was selective for the PSD and did not represent a general increase in protein levels.
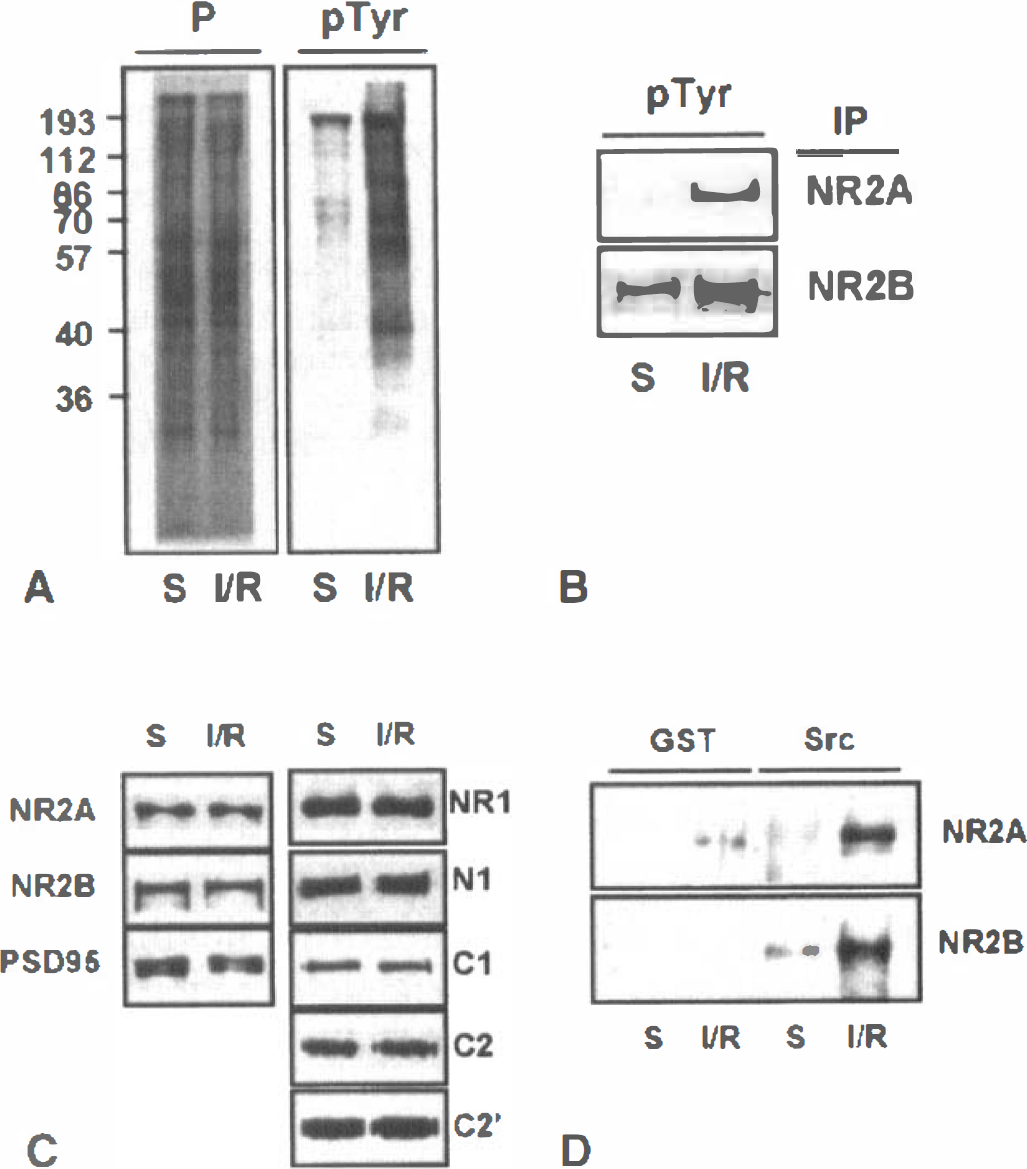
Ischemia results in increased tyrosine phosphorylation and SH2-binding of NR2A and NR2B in postsynaptic densities.
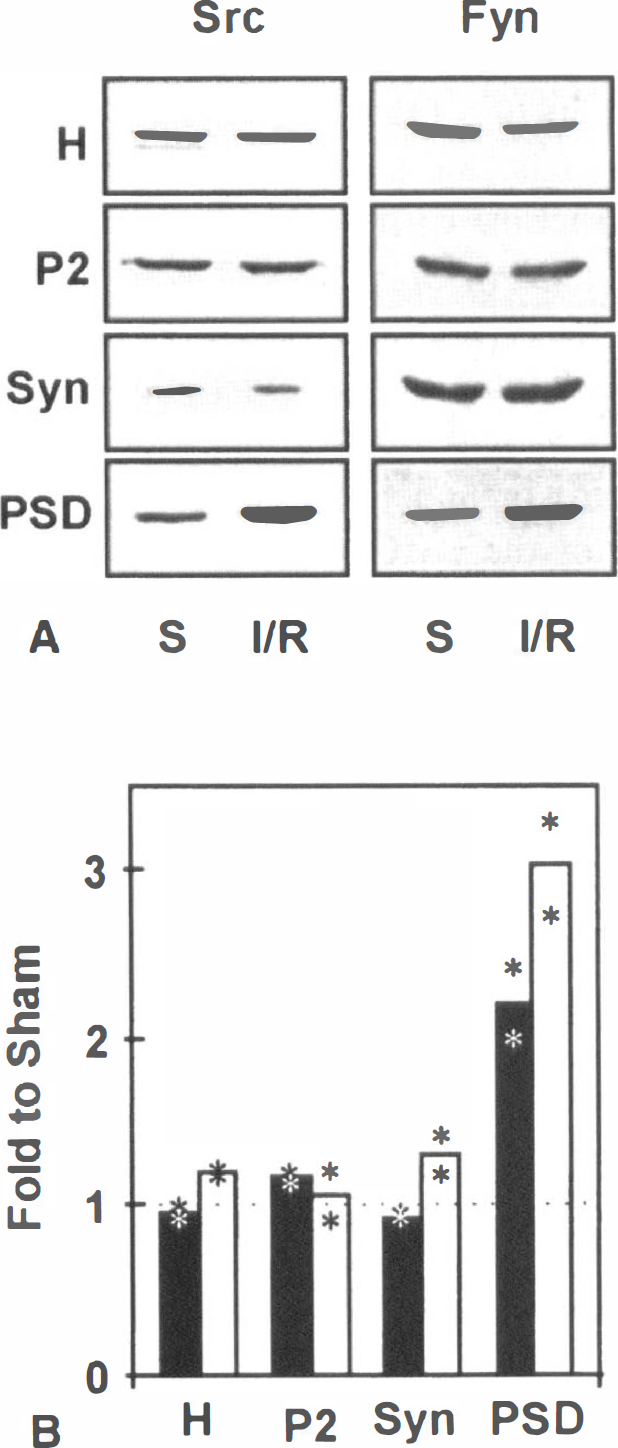
Increased association of Src and Fyn with postsynaptic densities after ischemia.
DISCUSSION
Transient cerebral ischemia sets in motion a series of events that may lead over several days to the death of neurons in sensitive brain regions, including the hippocampus. Whereas the precise molecular events responsible for initiating the processes that result in delayed neuronal death remain to be identified, one of the early changes subsequent to the start of reperfusion is an increase in tyrosine phosphorylation of several proteins, including the NMDAR subunits NR2A and NR2B (Hu and Wieloch, 1994; Yokota et al., 1994; Takagi et al., 1997). Tyrosine phosphorylation modulates the properties of the NMDAR ion channel (Wang and Salter, 1994; Kohr and Seeburg, 1996) and can link the NMDAR to signaling proteins that contain SH2 domains (Gurd and Bissoon, 1997). The results of the current study demonstrate that ischemia results in increased tyrosine phosphorylation of several PSD proteins, including NR2A and NR2B, that the increase in tyrosine phosphorylation of NR2A and NR2B enhances their ability to bind to the SH2 domains of Src and Fyn, and that these changes are paralleled by an increase in the association of Src and Fyn with the PSD.
The generally accepted membrane topology of glutamate receptors consists of three transmembrane domains and one pore loop region corresponding to the TMII sequence (Bennet and Dingledine, 1995). The carboxy-terminal domain, located on the cytoplasmic side of the membrane, constitutes the likely site of tyrosine phosphorylation. The SH2 domain of members of the Src family of tyrosine kinases preferentially selects phosphopeptides with the sequence pYEEI as well as peptides that have asparagine in the +2 position relative to pY (Songyang et al., 1993). The subunits NR2A and NR2B each contain 25 tyrosine residues in their carboxy-terminal regions. The YEEI sequence is not present in either 2A or 2B; however, potential binding sites for Src SH2 domains are present in NR2A at Tyr1292-Asp-Asn-Ile (YDNI) and in NR2B at Tyr1069-Gly-Asn-Ile (YGNI). Although Src family SH2 domains exhibit similar specificities in peptide selection assays (Songyang et al., 1993), there is variability in the sites recognized by individual SH2 domains, and closely related Src family members may discriminate between different phosphoproteins (Malek and Desiderio, 1993). Thus, the sequences recognized by the Src and Fyn SH2 domains on NR2A and NR2B may not correspond to the preferred sequences identified in the peptide binding assay. The increases in tyrosine phosphorylation of NR2A (6.6-fold relative to sham-operated animals) and NR2B (3.3-fold) were, in each case, greater than the ischemia-induced increase in binding to the Src and Fyn SH2 domains, suggesting that not all of the newly phosphorylated tyrosine residues are recognized by the SH2 domains. Identification of the tyrosine phosphorylation sites and, in particular, those involved in binding to Src and Fyn, becomes an important objective.
The current results were obtained with NMDAR subunits that had been denatured by solubilization of homogenates or PSDs with SDS. The effect of increased tyrosine phosphorylation on the interaction of the native NMDAR with SH2-containing proteins in situ might differ, being influenced by such factors as accessibility of phosphotyrosine residues in the heteromeric receptor complex to SH2 domains, the synaptic localization of proteins that contain SH2 domains, and competition between multiple SH2-containing proteins for potential binding sites. In this regard, we have found in preliminary experiments that the binding of NMDAR present in 1% deoxycholate extracts of the hippocampus to the Src SH2 domain is enhanced twofold after ischemia, indicating that phosphorylated tyrosine residues are available for interaction with SH2 domains in “native” receptor complexes, albeit in a soluble form. Finally, although the current results are consistent with the interaction of the NR2 subunits with the SH2 domains of Src and Fyn, the possibility that the association is mediated through one of more intervening proteins that remain associated with the receptor after solubilization in SDS cannot be excluded.
The current findings suggest that ischemia might result in enhanced association of Src and Fyn with the NMDAR. Although Src is associated with the NMDAR in immature neurons (Yu et al., 1997), the biochemical basis of this, association is unknown. A similar association between Src and the NMDAR in adult tissue has not been reported, and our attempts to demonstrate an association between the NMDAR and Src in extracts of the adult hippocampus by co-immunoprecipitation have been unsuccessful. As an alternate approach to determining the possible biological significance of the increased ability of Src and Fyn to bind to the NMDAR after ischemia, we considered that it might result in the recruitment of tyrosine kinases to the PSD. Whereas the increase in both Src and Fyn in PSD after ischemia is consistent with this proposal, the molecular basis of their association with the PSD is not known, and mechanisms other than, or in addition to, SH2 interactions are possible. In preliminary experiments, we have found that the phosphorylation of PSD proteins by exogenous Src is prevented by the inclusion of the SH2 domain-binding peptide EPQ(p)YEEIPIA but not by the nonphosphorylated homologue in the phosphorylation assay, suggesting that the phosphorylation reaction is dependent on Src binding to the PSD through its SH2 domain (Cheung and Gurd, unpublished observations). Whether or not Src and Fyn associate with the NMDAR through their SH2 domains, their recruitment to the PSD after an ischemic episode might result in enhanced tyrosine phosphorylation of PSD-associated proteins, including the NMDAR itself. Phosphorylation by Src (Yu et al., 1997) and Fyn (Kohr and Seeburg, 1996) potentiates the NMDAR ion channel so that an increase in the local concentration of these kinases at the PSD may contribute to receptor activation. In general accord with this suggestion, we have found that at short times of reperfusion (6 hours), the open probability and the mean open time of NMDA ion channels in CA1 neurons were greater in postischemic than in sham-operated controls (Zhang et al., 1997), changes that generally are consistent with the effects of enhanced tyrosine phosphorylation (Yu et al., 1997). In addition to modulation of the receptor ion channel, the recruitment of Src family tyrosine kinases to the PSD also may represent the first step in activating signal transduction pathways leading to ischemia-induced necrosis, apoptosis, or both. Inhibition of tyrosine phosphorylation was neuroprotective after global ischemia in the gerbil (Kindy, 1993; Ohtuski et al., 1996), and tyrosine phosphorylation has been implicated in apoptosis in a variety of systems (Lavin et al., 1996), Ischemia also results in increased levels of the brain-derived neurotrophic factor receptor gp145 trkB (Hu et al., 1998) and of the tyrosine kinase Pyk2 (Cheung and Gurd, in preparation) in the PSD, indicating that there may be more than one mechanism by which ischemia leads to changes in the tyrosine phosphorylation of PSD proteins.
It has become apparent that the NMDAR is present at the PSD as part of a macromolecular complex of receptors, ion channels, and signaling proteins linked together by their interaction with the PSD-95 family of proteins (Ehlers et al., 1996; Sheng, 1997; Ziff, 1998). The current and earlier (Gurd and Bissoon, 1997) findings suggest that SH2-containing proteins may contribute to this protein network and further that ischemia-induced increases in tyrosine phosphorylation may modulate the composition of the postsynaptic receptor/signaling protein complex, Prevention or disruption of these interactions with reagents that selectively interfere with SH2 domain binding reactions (e.g., Hall et al., 1996) may prove beneficial in reducing the damaging effects of an ischemic event.
