Abstract
We investigated the impairment of retention of spatial memory in rats with chronic focal cerebral ischemia, and examined the correlation between this impairment and pathological outcomes. A preoperative acquisition trial of the Morris water-maze task was performed twice a day for 14 successive days, and then the middle cerebral artery (MCA) was occluded. A retention trial was performed 8 weeks after MCA occlusion. Escape latency and swimming path length to the platform were significantly increased in MCA-occluded rats compared with those of sham-operated rats, and these deficits significantly correlated with massive shrinkage of the brain. Retention latency of a passive avoidance task, which was trained preoperatively, was also significantly shortened in MCA-occluded rats compared with that in sham-operated rats. These results suggest that chronic focal cerebral ischemia causes prolonged spatial memory disturbance in rats and is associated with pathological changes, and that this rat model may be useful for assessing not only anterograde but also retrograde amnesia caused by focal cerebral ischemia.
Keywords
Patients with cerebrovascular disease occasionally exhibit cognitive disturbances, such as amnesia and topographical disorientation, as well as neurological symptoms, such as hemiparesis (Caplan et al., 1985; Caplan et al., 1990). In order to investigate the pathophysiology of such symptoms and to develop effective drug therapy, it is necessary to establish a chronic animal model. Spatial cognitive performance is known to be altered by pharmacological agents, aging, and brain lesions (Miyamoto et al., 1987; Rapp et al., 1987; Whishaw et al., 1987; DiMattia and Kesner, 1988; Mandel and Thai, 1988; Nakamuraand Ishihara, 1990; McNamaraand Skelton, 1993). The middle cerebral artery (MCA) occlusion model developed in rats by Tamura and coworkers (Tamura et al., 1981; Yamamoto et al., 1988) appears to be a suitable stroke model with a relatively constant infarct size. Several authors have detected the disturbance of memory acquisition after MCA occlusion in rats using avoidance tasks and maze tasks (Yamamoto et al., 1989a, b ; Markgraf et al., 1992; Wahl et al., 1992; Okada et al., 1995b). However, only a few studies have focused on memory retention (Hirakawa et al., 1994).
The aim of the present study was to clarify the influence of chronic cerebral infarction on the retention of spatial memory in rats using the water navigation task developed by Morris (Morris, 1981; Morris et al., 1982), chosen because it is not readily influenced by olfactory sensation or motivation. We also investigated correlations between the extent of brain damage and behavioral outcome.
METHODS
Surgical procedure
Male Wistar rats (Japan Sic Inc., Shizuoka, Japan), aged 10 weeks and weighing 250–310 g, performed acquisition procedures of the Morris water-maze task and passive avoidance task for ∼2 weeks. Then, 18 rats were anesthetized with 1–2% halothane and the left MCA was permanently occluded at a proximal site using a micro-bipolar coagulator (Tamura et al., 1981; Yamamoto et al., 1988). After the incision was sutured, rats were allowed free access to laboratory chow and water. Sham-operated rats (n = 12) were subjected only to exposure of the MCA after induction of anesthesia.
Assessment of general behavior
Locomotor activity
Spontaneous locomotor activity was measured every 5 min for 90 min at 1, 2, and 7 weeks after surgery using an automated activity box (Automex II; Columbus Instruments, Columbus, OH, U.S.A.). Locomotor activity was evaluated over the initial 15- and 90-min periods.
Neurological examination
We evaluated sensorimotor performance in rats using a neurological deficit score based on detection of hemiparesis and abnormal posture at 5 days and 1, 2, 3, 4, 5, 6, 7, 8, 10, and 11 weeks after surgery. The right hind limb of each rat was extended gently using round-tipped forceps and the flexor response evaluated as 0 (normal), 1 (slight deficit), 2 (moderate deficit), or 3 (severe deficit). For assessment of posture, rats were suspended by the tail, and forelimb flexion and body twisting were evaluated as 0 (normal), 1 (slight twisting), 2 (marked twisting), or 3 (marked twisting and forelimb flexion).
Passive avoidance task
Rats were tested using a step-through type of passive avoidance task (Jarvik and Kopp, 1967; Yamamoto et al., 1988). The apparatus (Neuroscience Inc., Tokyo, Japan) consisted of two compartments: an illuminated chamber (10 × 30 cm) and a dark chamber with a grid floor of stainless steel rods (30 × 30 cm). Rats initially underwent habituation by being placed in the illuminated chamber, with an open door between chambers, so that they could enter the dark chamber. In preoperative acquisition trials, rats performed the same task, but a 0.6 mA foot shock was delivered through the grid floor for 1 s once they entered the dark chamber. Acquisition trials were carried out once a day for 3 days before MCA occlusion, while postoperative retention trials were performed without foot shock 10, 17, and 24 days after occlusion. Step-through latency was recorded to a maximum of 600 s.
Morris water-maze task
Spatial memory retention was examined using the Morris water-maze. The apparatus used was a 144 cm diameter cylindrical tank, filled with water to a depth of 32 cm, with a transparent 10 cm diameter platform placed at a constant position in the center of one of the four quadrants within the tank. The platform was set 2 cm below the water level where the rats could not see it directly. Seventeen days before MCA surgery, rats were allowed to swim freely for 1 min to become habituated to the apparatus. From the next day, acquisition trials were carried out twice a day for 14 successive days. In each trial, rats were placed into the water at a fixed starting position, and the time taken to escape onto the hidden platform and the swimming path length were measured using a Video Image Motion Analyzer (AXIS30; Neuroscience Inc). Rats were given 120 s to find the hidden platform during each acquisition trial and were allowed to rest on the platform for 30 s after finding it. Rats failing to find the platform within the allotted period were placed onto it by the experimenter and a time of 120 s assigned. Postoperative retention trials were carried out twice a day for 3 days at 8 weeks after MCA occlusion. Each rat was made to perform the same procedure as described above but was not placed onto the platform by the experimenter.
Histopathology
After the behavioral studies were completed, rats were anesthetized with diethyl ether and fixed by perfusion with 10% formaldehyde in 0.1 M phosphate buffer, and their brains removed. Five coronal blocks (2 mm thick) were cut 12.2–4.2 mm anterior to the interaural line, according to the brain atlas of Paxinos and Watson (1982), and embedded in paraffin. Then, 4-μm sections were cut from each block and stained with hematoxylin and eosin. The area of each cerebral hemisphere was determined on individual sections with a digitizer (Luzex-FS, Nireco Corp., Tokyo, Japan) and the total area of each hemisphere was calculated as the sum of the individual areas. Then, the degree of shrinkage of the left cerebral hemisphere was calculated as a percent relative to that of the right hemisphere area.
Statistical analysis
To analyze parametric data, repeated measures analysis of variance (ANOVA), followed by Scheffe's-test, was used. When dealing with nonparametric data, the Mann–Whitney U-test was used. Regression analysis was used to investigate correlations.
RESULTS
General behavior
There were no deaths in the MCA occlusion group (n = 18) or the sham-operated group (n = 12) throughout experimental period. The body weight of the MCA occlusion group was significantly decreased compared with that of the sham-operated group at 5 days after surgery (F = 6.88, p < 0.05; one-factor ANOVA followed by Scheffe's-test), although there was no difference between the two groups subsequently (group: F1,28 = 2.10, p = 0.16; time: F10,280 = 319.49, p < 0.001; group · time: F10,280 = 0.69, p = 0.74; two-factor ANOVA for repeated measurements).
At 1, 2, and 7 weeks after surgery, spontaneous locomotor activity was not significantly different between the two groups (two-factor ANOVA for repeated measurements) when assessed using the initial 15- (Fig. 1A) and 90-min intervals (Fig. 1B).
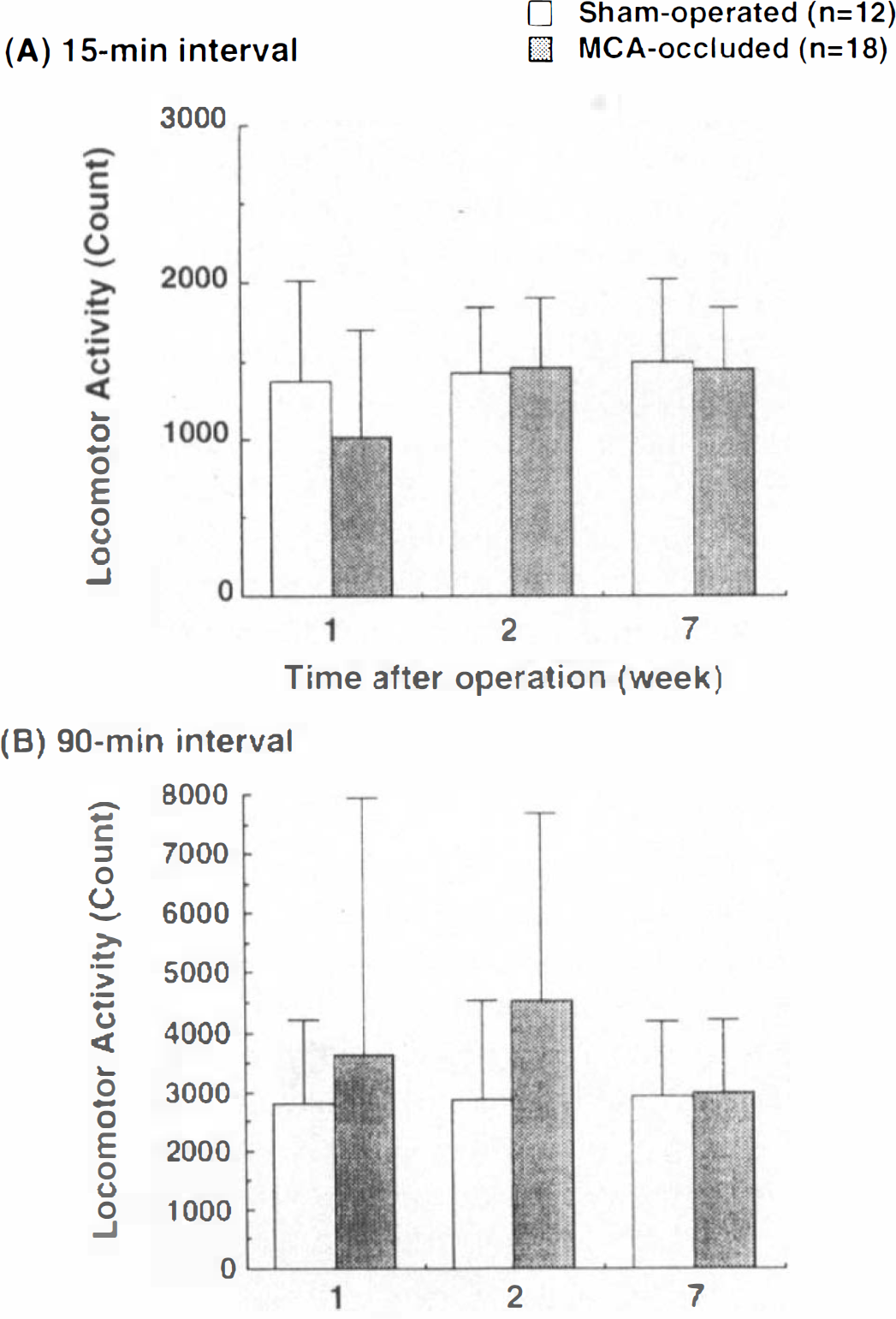
Locomotor activity in sham-operated and MCA-occluded rats at 1, 2, and 7 weeks after operation. Data were analyzed for the initial 15-min interval (
MCA occlusion significantly disturbed both posture (p < 0.01, Mann–Whitney U-test; Fig. 2A) and flexor response of the contralateral hind limb (Fig. 2B). Although neurological scores showed gradual recovery, MCA-occluded rats had significantly higher neurological scores than did sham-operated rats even at 11 weeks after surgery. Sham-operated rats did not show any abnormalities during the observation period.
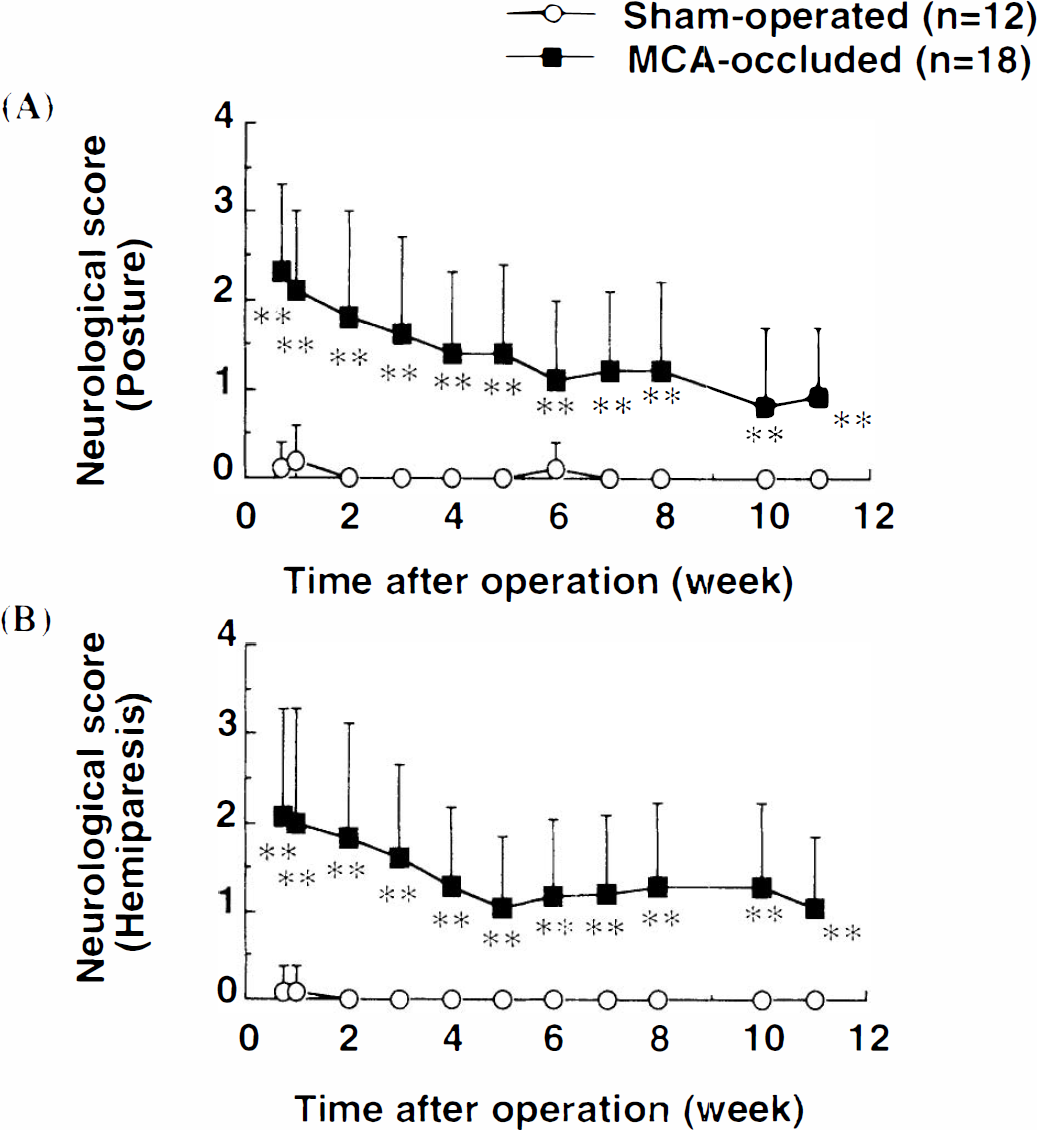
Neurological deficits in sham-operated and MCA-occluded rats. Abnormal posture (
Passive avoidance task
Preoperative acquisition trials of the passive avoidance task were carried out once a day for 3 days before MCA occlusion; the retention latencies of all rats was >600 s. Postoperative retention trials were performed 10, 17, and 24 days after MCA occlusion. Results obtained are shown in Fig. 3. In the first retention trial, retention latency of the MCA occlusion group (229.1 ± 278.6 s; mean ± SD) was significantly shorter than that of the sham-operated group (599.1 ± 3.2 s; p < 0.01, Mann–Whitney U-test). In the second and third retention trials, significantly shorter latencies were also observed.
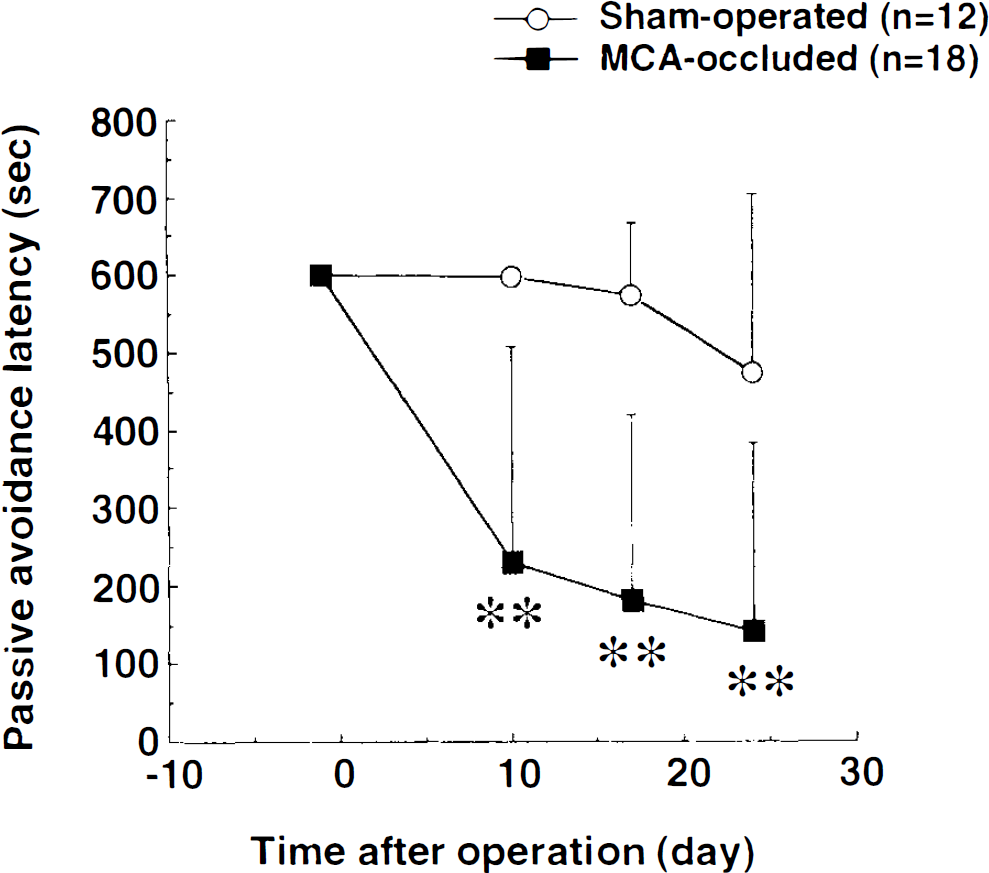
Passive avoidance responses of sham-operated and MCA-occluded rats. Acquisition trials were done once a day for 3 days before MCA occlusion, and retention trials were performed 10,17, and 24 days after the operation. Each point represents the mean ± SD(n = 12–18). At 10,17, and 24 days postoperatively, the retention latency of the MCA occlusion group was significantly shorter than that of the sham-operated group (**p < 0.01; Mann–Whitney U-test).
Morris water-maze task
Escape latency, swimming path length, and mean swimming velocity to the hidden platform in preoperative acquisition trials and postoperative retention trials are shown in Fig. 4. Both groups performed this task rapidly during preoperative trials. Two-factor ANOVA showed no differences between the MCA occlusion group and the sham-operated group in escape latency (Fig. 4A), path length (Fig. 4C), and velocity (Fig. 4E). In the last preoperative session, the sham-operated group took 4.5 ± 2.1 s (mean ± SD) to reach the platform and the MCA occlusion group, 4.6 ± 1.7 s. Path lengths of the two groups were 126.6 ± 33.0, and 133.7 ± 32.4 cm, respectively, and velocities were 30.8 ± 7.6 and 32.3 ± 6.1 cm/s, respectively. There were no significant differences in any parameter at the last session (Mann–Whitney U-test).
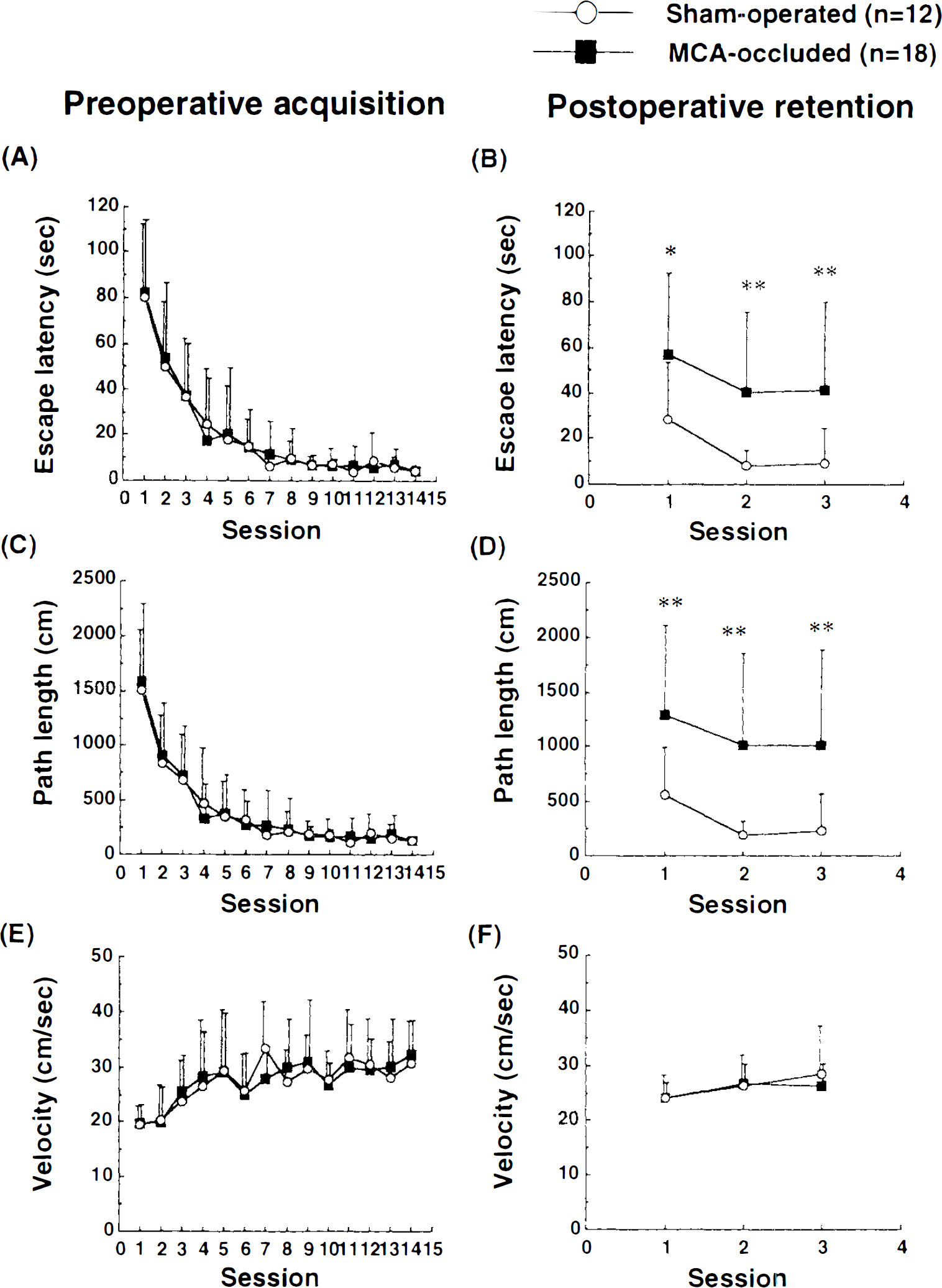
Effects of focal cerebral ischemia on spatial memory retention in the Morris water-maze task. Each animal was tested twice a day for 14 consecutive days, and escape latency (
Retention trials were performed twice a day for 3 days at 8 weeks after MCA occlusion. MCA-occluded rats did not perform this task well. When two-factor ANOVA was used to compare the last preoperative acquisition session and all of the postoperative retention sessions, there was a significant difference between MCA-occluded and sham-operated rats with respect to escape latency (Fig. 4B) and path length (Fig. 4D), but not velocity (Fig. 4F). In the first postoperative retention trial, escape latency and path length of the sham-operated group were 28.4 ± 25.1 s and 562.6 ± 431.3 cm, respectively, significantly longer compared to those of the last preoperative acquisition trial (p < 0.01, Mann–Whitney U-test), but still significantly shorter than those of the first preoperative acquisition trial (p < 0.01, Mann–Whitney U-test). Both parameters decreased to the preoperative level within three retention sessions. On the other hand, escape latency and path length of MCA-occluded rats were 56.7 ± 36.0 s and 1296.9 ± 812.6 cm, respectively, in the first retention session, significantly larger values than those for sham-operated rats (p < 0.01, Mann–Whitney U-test) and similar to results of the first preoperative acquisition trial. In contrast to those of the sham-operated group, these two parameters did not improve within three sessions in the MCA occlusion group, even though the rats swam well and adopted a searching strategy. There was no significant difference in velocity between the two groups in any session.
Correlation analysis
After the behavioral investigation, the percent area of the left cerebral hemisphere relative to that of the right hemisphere was determined. In the MCA occlusion group, the value was 65.1 ± 12.8%, significantly smaller than that of the sham-operated group (98.9 ± 2.3%). There was a positive correlation between neurological score and degree of brain shrinkage (r = 0.50, p < 0.05; Fig. 5A). There was also a significant correlation between the sum of three retention swimming path lengths (r = 0.55, p < 0.05; Fig. 5B) in the Morris water-maze task and the degree of brain shrinkage. The sum of three retention escape latencies also tended to correlate with brain shrinkage (r = 0.43, p < 0.10; Fig. 6C), but velocity did not (r = 0.02, p = 0.92). Retention latency in the passive avoidance task did not correlate with the retention swimming path length in the Morris water-maze task (r = 0.30, p = 0.23; Fig. 6D).
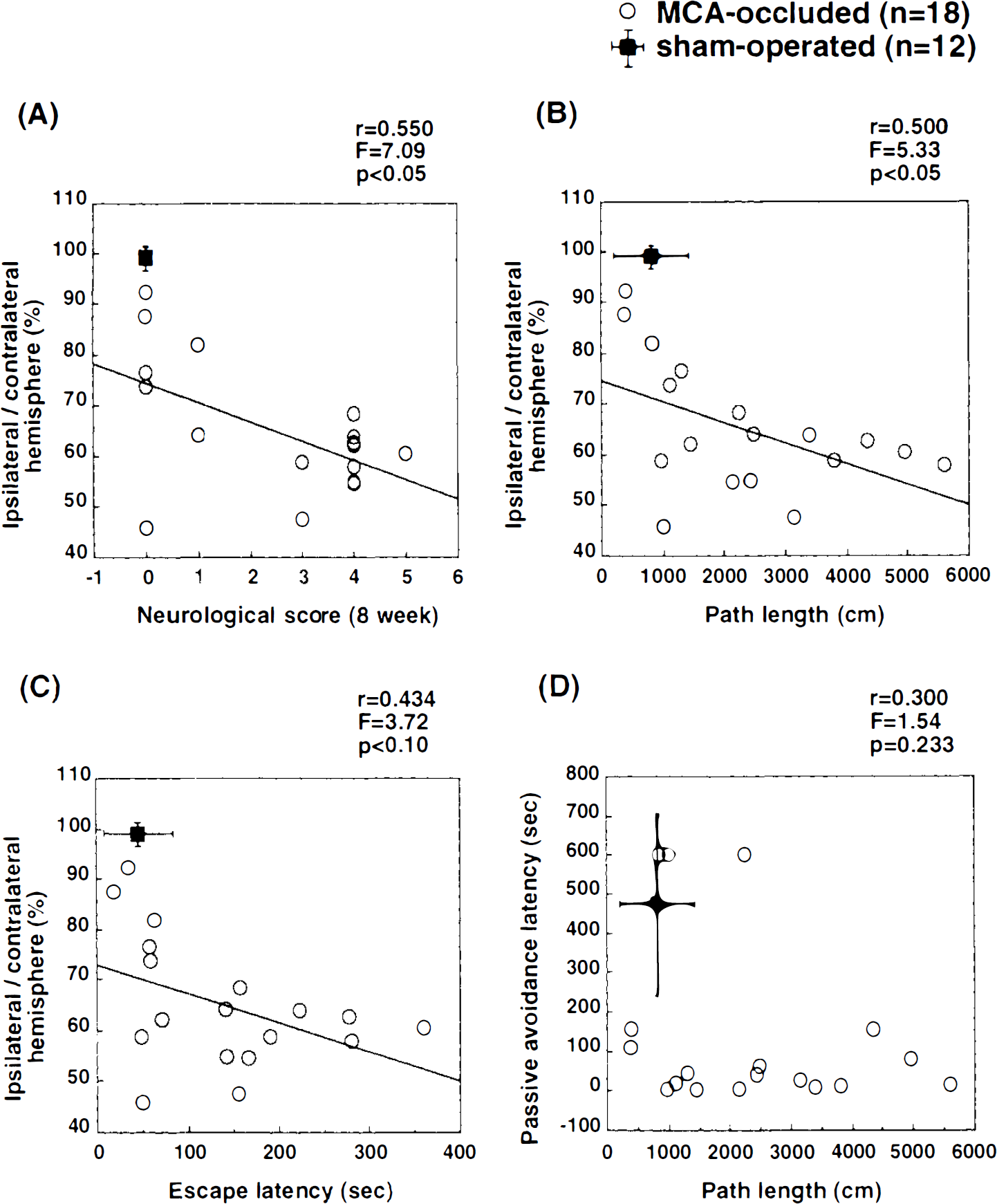
Relationship between lesion size and behavioral outcome in rats subjected to MCA occlusion. Data from MCA-occluded rats (n = 18, open circles) were used for regression analysis, and data from sham-operated rats (n = 12, filled squares; mean ± SD) were excluded. Neurological score (
DISCUSSION
Behavioral changes occurring in both acute and chronic phases of focal cerebral ischemia following MCA occlusion have already been studied in rats (Robinson, 1979; Tamura et al., 1985; Yamamoto et al., 1988; Person et al., 1989). Several authors have reported that memory and learning were disturbed in the passive avoidance task (Yamamoto et al., 1989a, b ; Hirakawa et al., 1994), the Y-maze task (Wahl et al., 1992), and the maze task (Markgraf et al., 1992; Okada et al., 1995b). Memory retention is just as important as memory acquisition, but there has been no previous study dealing specifically with spatial memory retention in rats with focal cerebral ischemia and using the Morris water-maze task as well as passive avoidance task. In the present study, we investigated the disturbance of memory retention in rats with focal cerebral ischemia and examined the correlations between behavioral and pathological changes.
Changes of body weight and locomotor activity as well as neurological deficits of MCA-occluded rats observed in this study are in agreement with previous findings (Bederson et al., 1986; Obana et al., 1988; Yamamoto et al., 1988; Person et al., 1989; Yamamoto et al., 1989a, b ). By 6 weeks after MCA occlusion, neurological deficits were fairly stable and showed little subsequent change.
We evaluated the retention of spatial memory using the Morris water-maze task at 8 weeks after surgery, a time when residual neurological deficits were mild and stable and when MCA occlusion had no effect on body weight or locomotor activity. Swimming speed did not differ between the two groups before or after the operation, indicating that neurological deficits, body weight, and locomotor activity did not influence performance in the water-maze task. However, MCA-occluded rats took significantly more time and used a longer route to reach the platform compared with sham-operated rats throughout all of the retention sessions, even though all of the MCA-occluded rats exhibited searching behavior in the task and escaped onto the hidden platform by themselves at least once during the retention sessions. In addition, MCA-occluded rats showed a shorter step-through latency in the passive avoidance task compared with sham-operated rats at 10, 17, and 24 days after surgery. Our results are in good agreement with the findings of Hirakawa et al. (1994). These findings indicate that not only disturbance of acquisition performance but also disturbance of retention performance is a chronic result of focal cerebral ischemia.
It has been reported that permanent MCA occlusion disturbs postoperative training for the Morris water-maze task (Markgraf et al., 1992; Yonemori et al., 1995), and the eight-arm radial maze task (Okada et al., 1995b). In the four-vessel occlusion forebrain ischemia model, cognitive performance was also found to be impaired in the Morris water-maze task (Hagan and Beaughard, 1990; Jaspers et al., 1990; Nunn et al., 1994) and in the radial-maze task (Volpe et al., 1989). Furthermore, Gionet et al. (1991) reported that 10 min of two-vessel forebrain ischemia impaired retention of a radial-maze discrimination task. Our present findings showed that proximal MCA occlusion caused prolonged and severe disturbance of spatial memories that had been acquired preoperatively.
In the Morris water-maze task, there was a significant positive correlation between postoperative swimming path length and shrinkage of the ischemic hemisphere. The relationship was most apparent for the coronal slice, which contained the parietal cortex and caudate-putamen. Swimming path length in the postoperative Morris water-maze task did not correlate with retention latency of the passive avoidance task. We previously reported that acquisition of the Morris water-maze task, but not the passive avoidance task, was correlated with brain infarct volume (Yonemori et al., 1995). Tamura et al. (1993) also reported that active avoidance retention latency, but not passive avoidance retention latency, was correlated with cortical infarct volume in MCA-occluded rats. These findings indicate the possibility that different regions of the brain contribute to cognitive procedures and memory retention and/or retrieval procedures, with the cortex and/or caudate-putamen having an important role. In the four-vessel occluded forebrain ischemia model, transient spatial cognitive impairment in the Morris water-maze task has been related to damage of the CA1 region (Hagan and Beaughard, 1990; Jaspers et al., 1990), although Nunn et al. (1994) reported that CA1 damage may not be the only determinant of water-maze deficits. In rats with hippocampal lesions (Morris et al., 1982) and rats with small parietal cortex lesions (Kolb et al., 1983), acquisition of the Morris water-maze task is impaired, while in rats with entorhinal cortex lesions (Schenk and Morris, 1985), of both acquisition and retention are impaired. It is reported that bilateral lesions of the parietal lobe (Kesner et al., 1987; DiMattia and Kesner, 1988) and caudate-putamen (Whishaw et al., 1987) also produce deficits of spatial memory in rats. In the MCA occlusion model used in this study, the cerebral cortex and striatum were infarcted, but the hippocampus was spared (Tamura et al., 1981; Okada et al., 1995a). Although the possibility remains that secondary damage to the hippocampus contributed to the impairment of spatial memory retention, we consider that this impairment is more likely related to cortical and/or caudate-putamen infarction.
In summary, we demonstrated that focal cerebral ischemia causes chronic spatial memory disturbance in rats and is correlated with shrinkage of the ischemic hemisphere. These findings are considered to be relevant to the clinical situation, and this rat model may be useful for assessing not only anterograde but also retrograde amnesia caused by focal cerebral ischemia.
Footnotes
Acknowledgment:
We wish to thank Ms. Satomi Takeuchi for her technical assistance.
