Abstract
This study evaluates the validity of the transfer function analysis of spontaneous fluctuations of arterial blood pressure (ABP) and blood flow velocity of the middle cerebral artery (MCAFV) as a simple, convenient method to assess human cerebral autoregulation in patients with carotid stenosis. Eighty-three consecutive patients with various degrees of carotid stenosis and 37 healthy controls were enrolled. The carotid stenosis was graded based on the diagnostic criteria of duplex ultrasound. Instantaneous bilateral MCAFV and ABP of all participants were assessed noninvasively using transcranial Doppler sonography and the servocontrolled infrared finger plethysmography, respectively. Spectral analyses of ABP and MCAFV were performed by fast Fourier transform. The fluctuations in ABP as well as in MCAFV were diffracted into three components at specific frequency ranges designated as high-frequency (HF; 0.15 to 0.4 Hz), low-frequency (LF; 0.04 to 0.15 Hz), and very low-frequency (VLF; 0.016 to 0.04 Hz). Cross-spectral analysis was applied to quantify the coherence, transfer phase, and magnitude in individual HF, LF, and VLF components. Transcranial Doppler CO2 vasomotor reactivity was measured with 5% CO2 inhalation. The LF phase angle (r = −0.53, P < 0.001); magnitude of VLF (r = −0.29, P = 0.002), LF (r = −0.35, P < 0.001), and HF (r = −0.47, P < 0.001); and CO2 vasomotor reactivity (r = −0.66, P < 0.001) were negatively correlated with the severity of stenosis. Patients with unilateral high-grade (greater than 90% stenosis) carotid stenosis demonstrated significant reduction in LF phase angle (P < 0.001) and HF magnitude (P = 0.018) on the ipsilateral side of the affected vessel compared with their contralateral side. The study also revealed a high sensitivity, specificity, and accuracy using LF phase angle and HF magnitude to detect a high-grade carotid stenosis. A strong correlation existed between the LF phase angle and the CO2 vasomotor reactivity test (r = 0.62, P < 0.001), and the correlation between the HF magnitude and the CO2 vasomotor reactivity (r = 0.44, P < 0.001) was statistically significant as well. We conclude that transfer function analysis of spontaneous fluctuations of MCAFV and ABP could be used to identify hemodynamically significant high-grade carotid stenosis with impaired cerebral autoregulation or vasomotor reserve.
Keywords
Cerebral perfusion pressure autoregulation (CA) and vasomotor reserve are two important homeostatic mechanisms for controlling cerebral blood flow. For many years, the evaluation of CA was performed under a steady-state condition called the “static autoregulation test.” Not until the late 1980s did Aslid and coworkers (1989) develop a “dynamic autoregulation test” that greatly promoted the evaluation of CA. They used transcranial Doppler (TCD) sonography to record cerebral blood flow velocity (CBFV) during stepwise reductions (deflation of leg cuffs) in arterial blood pressure (ABP). Using the dynamic autoregulation test, Newell et al. (1994) revealed that the time course in CBFV expressed as percentage deviations from the baseline was nearly identical to responses in CBF measured directly within the internal carotid artery, suggesting that the relative changes in CBFV seem to reflect the relative changes in CBF. As a result, TCD is recognized as a good evaluation tool to assess the dynamics of CA. Later, Tiecks et al. (1996) developed autoregulatory indices based on the changes in blood flow velocity and blood pressure during the Valsalva maneuver. Under the assumption that the dynamics of the cerebral autoregulatory system could be described in terms of a high-pass filter model, Diehl et al. (1995) demonstrated a phase relationship between CBFV and blood pressure during deep breathing at the rate of 6 breaths per minute.
Furthermore, extensive studies of cardiovascular regulation using frequency domain analysis of heart rate and blood pressure fluctuations expanded the insight into how the human body functions. To the best of our knowledge, our laboratory is among the earliest research teams using frequency domain analysis of the spontaneous fluctuation of CBFV and ABP to evaluate cerebral hemodynamics (Kuo et al., 1998). The methodology of using transfer function analysis to evaluate cerebral hemodynamics was further tested by Blaber et al. (1997) and Zhang et al. (1998) in different settings and populations. In our previous work (Kuo et al., 1998), frequency domain analysis was applied to assess and quantify spontaneous fluctuations in the blood flow velocity of the middle cerebral artery (MCAFV) and ABP. We have demonstrated that the high-frequency (HF) and low-frequency (LF) fluctuations of MCAFV exhibited a good coherence with those of ABP. This finding indicated a great similarity of MCAFV and ABP fluctuation within the two frequency ranges, suggesting that the HF and LF spontaneous fluctuations of MCAFV may be secondary to those of ABP, or may come from the same origin as ABP. Thus, frequency domain analysis may offer an opportunity to assess CA without any application of special maneuvers, such as CO2 inhalation (Widder et al., 1986), carotid compression (Giller, 1991; Smielewski et al., 1995), deep breathing (Diehl et al., 1995), Valsalva maneuver, or artificial perturbation of ABP (Tiecks et al., 1995). Such maneuvers require not only cooperation of patients, but also may produce discomfort or even pose a risk in patients with cerebrovascular disease.
The present study was undertaken to evaluate the validity of autospectral and cross-spectral analysis techniques (i.e., transfer function analysis of CBFV and its correlation with ABP) as a simple, convenient test in the assessment of human CA in patients with carotid artery disease. More specifically, the aim of this study was to answer the question whether the phase shift angle and magnitude in the LF and HF ranges could be applied as valid parameters to assess CA and, hopefully, cerebral vasomotor reserve in patients with carotid artery disease.
SUBJECTS AND METHODS
The study took place in a large urban teaching hospital (2,700 beds). From November 1, 1996, to December 31, 1997, all consecutive patients with a clinical diagnosis of transient ischemic attack or minor stroke who were referred to our neurovascular laboratory for the screening of carotid artery stenosis were evaluated for eligibility for the study. The degree of the carotid artery stenosis was determined by color-coded duplex ultrasound using published criteria (Bluth et al., 1988; Hu et al., 1993). Patients qualifying for the study were those who had occlusive carotid artery disease with (1) unilateral or bilateral stenosis of greater than 50% or occlusion of the internal carotid artery (ICA), (2) an adequate temporal bone window for sufficient TCD examination, and (3) no additional intracranial stenosis on the carotid siphon, the middle cerebral artery, and the anterior cerebral artery. The diagnosis of carotid artery stenosis and the exclusion of the intracranial stenosis in the major arteries in the brain base were performed by color-coded duplex ultrasound (Acuson 128 XP; Acuson, CA, U.S.A.) with 7.5-MHz probe for extracranial examination and 2-MHz probe for intracranial examination. Using the color-coded duplex ultrasound, the extracranial findings for each of the ICA were classified as follows: normal (group 1), less than 50% stenosis (group 2), stenosis of 50% to 75% (group 3), 75% to 90% (group 4), 90% to 99% (group 5), and occlusion (group 6). The vertebral arteries and basilar artery were insonated by color-coded duplex ultrasound, but the findings of vertebral and basilar arteries were not included in the analysis.
For comparison, healthy subjects in a similar age range (48 to 75 years old) were recruited as the control group. All healthy subjects were carefully screened for not having any history of diabetes mellitus and cardiovascular disease, which have been reported to have effects on cardiovascular fluctuations. A complete physical examination was given and the color-coded duplex ultrasound was performed to exclude carotid artery stenosis and stenosis in the major arteries in the brain base. The study protocol was approved by the Human Ethics Committee and each of the participants signed an informed consent. All measurements for spectral analysis were performed in a supine position for 15 minutes. The detailed methodology for spectral and transfer analyses of MCAFV and ABP has been reported elsewhere (Kuo et al., 1998). In brief, instantaneous bilateral MCAFV of all study subjects was detected using TCD (Multidop-X, DWL; Sipplingen, Germany), the transducers were fixed in place, and MCAFV was continuously monitored at the depth of the best signal (44 to 55 mm). Instantaneous ABP was recorded noninvasively by servocontrolled infrared finger plethysmography (Finapres, model 2300, Ohmeda Monitoring Systems, Englewood, CT, U.S.A.). Both ABP and MCAFV signals were obtained, displayed, and stored to floppy disk by multidop-X. Spectral analyses of MCAFV and ABP were performed by fast Fourier transform. The fluctuations in MCAFV as well as ABP were diffracted into three components at specific frequency ranges designated as HF (0.15 to 0.4 Hz), LF (0.04 to 0.15 Hz), and very low-frequency (VLF) (0.016 to 0.04 Hz). Cross-spectral analysis was applied to quantify the coherence, transfer phase, and magnitude in individual HF, LF, and VLF components. For each of the frequency ranges, an oscillating frequency was first defined at which the coherence function exhibited the largest value, then coherence, transfer phase, and magnitude of the oscillating frequency were selected. This algorithm ensured the highest reliability to quantify the transfer function.
After monitoring ABP and MCAFV for 15 minutes in a supine position, CO2 vasomotor reactivity was measured for each of the study subjects. To produce hypercapnia, 5% CO2 was inhaled through a mouthpiece with a one-way valve connected to a respiratory balloon. The concentration of CO2 in the breathing gas was continuously monitored with an infrared gas analyzer (Respina 1H26, NEC Medical Systems, Japan). Hypocapnia was achieved by having the subject hyperventilate. With off-line data processing, samples from stable normocapnic, hypercapnic, and hypocapnic conditions were extracted. The CO2 reactivity is expressed as the percentage of change in velocity per unit change in PCO2 in kilopascals (1 kPa equals 1 vol%) using an individual's normocapnic state as the baseline. The mean of the maximal (mean) MCAFV during the stable spectral TCD recordings was used in this calculation. The results of hypocapnia were not used for calculation owing to the difficulty of achieving stable hyperventilation in many of the study participants.
All values are given as a mean ± SD unless otherwise indicated. Pearson product moment correlation coefficients were calculated to provide quantitative measures of the dependence between the two groups of data obtained from the different measures. The receiver operating characteristic curves were constructed from the data of transfer function and the area under the receiver operating characteristic curves was calculated to provide the best index for detection (or discrimination) of a hemodynamically significant carotid artery stenosis on the basis of the results of color-coded duplex ultrasound as a surrogate gold standard. The sensitivity, specificity, and predictive values of the transfer function were also calculated.
RESULTS
Of the 102 referral patients who met the study criteria, 19 refused to participate for various reasons. A total of 37 normal healthy adults were recruited for control subjects. The resulting sample size for final analysis included 83 patients (mean age, 69.7 ± 7.4 years; 82 were men) and 37 normal control subjects (mean age, 60.9 ± 8.7 years; 28 were men). Twenty-two of 37 normal subjects and 71 of 83 patients could be examined bilaterally; the remaining subjects had a suitable temporal bone window that was unilateral. Thus, a total of 213 middle cerebral arteries were investigated. Occlusive carotid artery disease was unilateral in 75 and bilateral in 8 of the 83 patients. With respect to the classification of the groups, group 1 consisted of 59 hemispheres, group 2 of 52, group 3 of 34, group 4 of 22, group 5 of 23, and group 6 of 23.
Results from the transfer function analysis and CO2 vasomotor reactivity test for each group are shown in Table 1. The LF phase angle (r = −0.53, P < 0.001); magnitude of VLF (r = −0.29, P = 0.002), LF (r = −0.35, P < 0.001), and HF (r = −0.47, P < 0.001); and CO2 reactivity (r = −0.66, P < 0.001) were all reduced and were negatively correlated with the severity of carotid stenosis (Table 1). Because both the North American Symptomatic Carotid Endarterectomy Trials (NA-SCET) and the European Carotid Surgery Trial demonstrated the benefit of carotid endarterectomy for symptomatic patients with a high-grade (greater than 70% linear diameter) carotid artery stenosis (European Carotid Surgery Trialists' collaborative Group, 1991; Barnett et al., 1992), a carotid artery stenosis greater than 90% as measured by duplex ultrasound, which corresponds to an approximately 75% reduction in diameter determined by angiography (Bluth et al., 1988; Ricotta et al., 1987), was considered as a hemodynamically significant high-grade carotid artery stenosis in this study. The LF phase angle (P < 0.001) and HF magnitude (P = 0.018) were both significantly reduced on the ipsilateral side of the affected carotid artery compared with the contralateral side in patients with high-grade unilateral carotid artery stenosis (Fig. 1).
Correlation of transfer function phase, magnitude, and CO2 reactivity with the severity of carotid stenosis
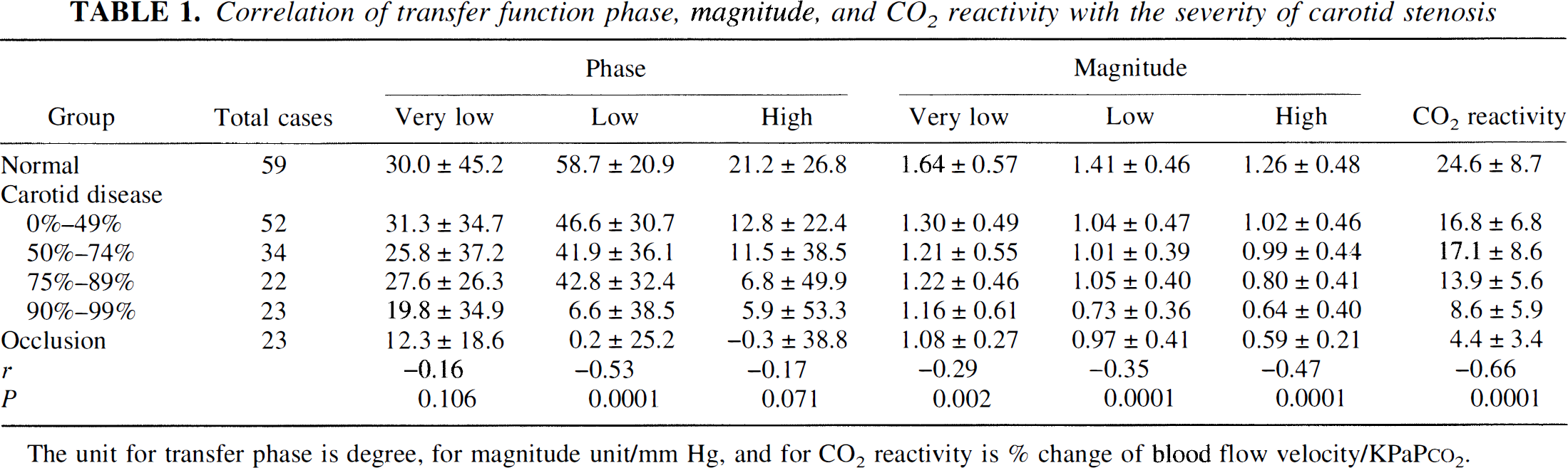
The unit for transfer phase is degree, for magnitude unit/mm Hg, and for CO2 reactivity is % change of blood flow velocity/KPaPco2
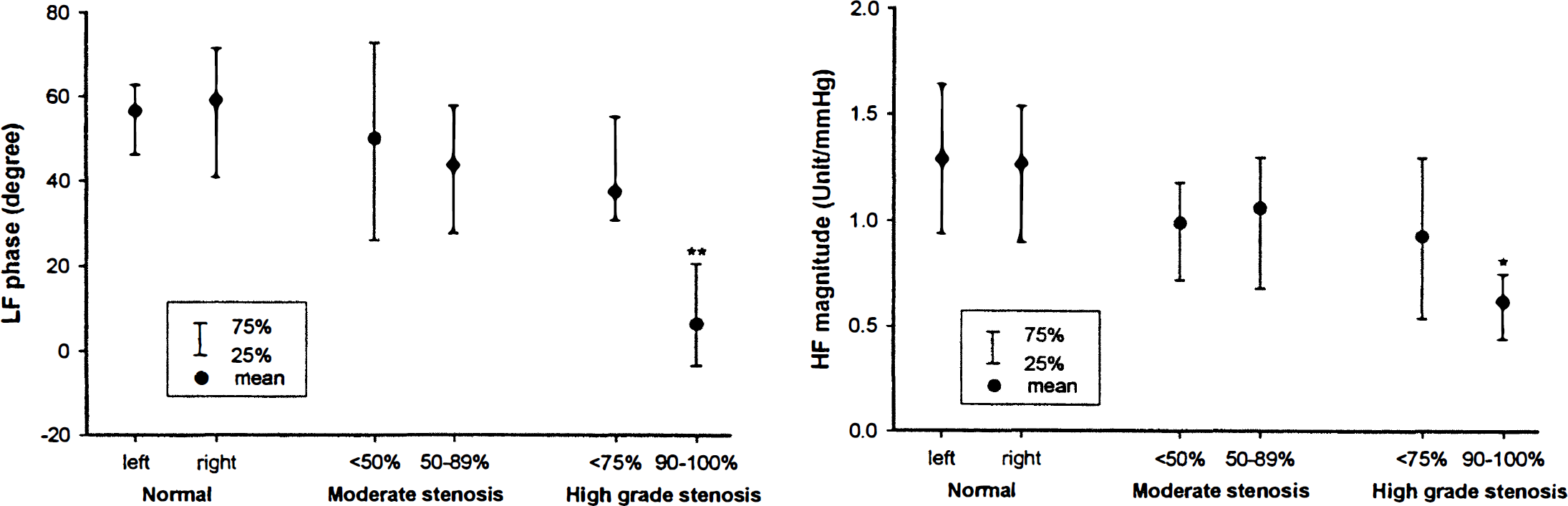
Comparison of the transfer function phase angle (low frequency, LF) and magnitude (high frequency, HF) to demonstrate the left-right comparison in normal volunteers and side-side difference in patients with unilateral moderate-degree and high-grade carotid artery stenosis. (* indicates a significant side-side difference, P = 0.018; ** indicates a significant side-side difference, P < 0.001.)
Table 2 shows the sensitivity, specificity, and accuracy of using the LF phase angle, HF magnitude, and CO2 reactivity as parameters of impaired CA or impaired vasomotor reserve to predict a hemodynamically significant high-grade carotid artery stenosis.
The sensitivity, specificity, and predictive values to detect a high-grade carotid stenosis/occlusion
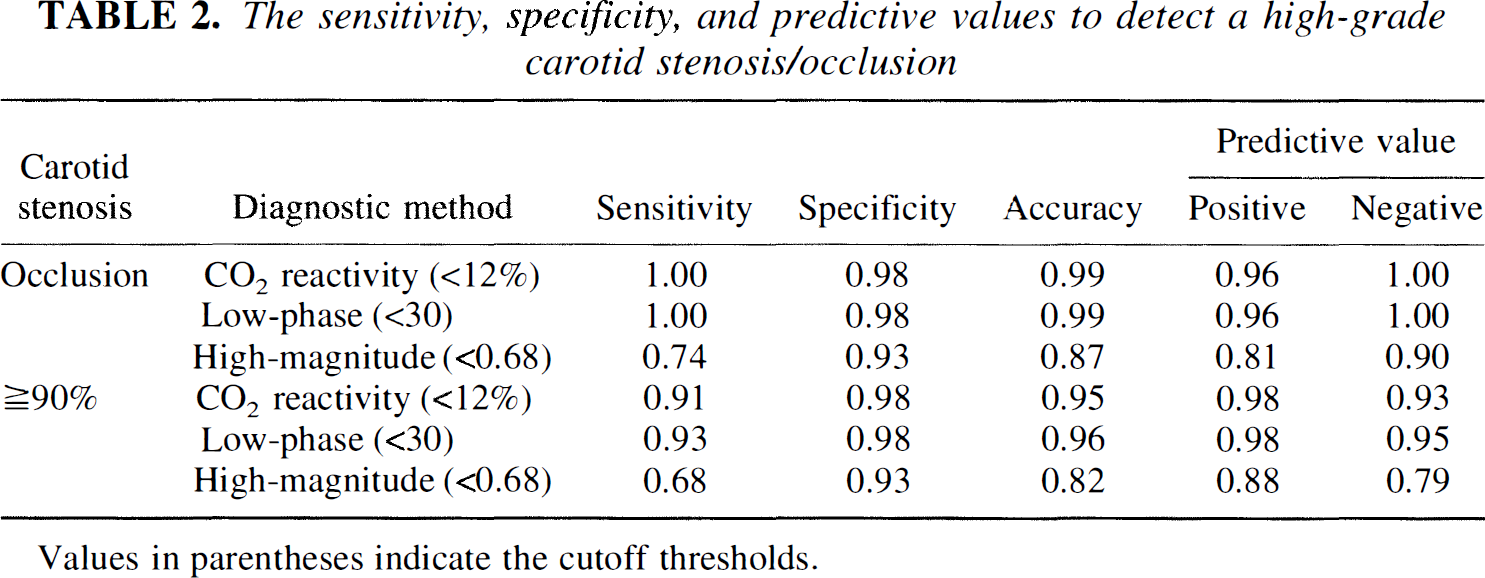
Values in parentheses indicate the cutoff thresholds.
There was a strong positive correlation between the LF phase angle and the CO2 vasomotor reactivity test (r = 0.62, P < 0.001; Fig. 2) and a moderate positive correlation between the HF magnitude and the CO2 vasomotor reactivity test (r = 0.44, P < 0.001; Fig. 3).
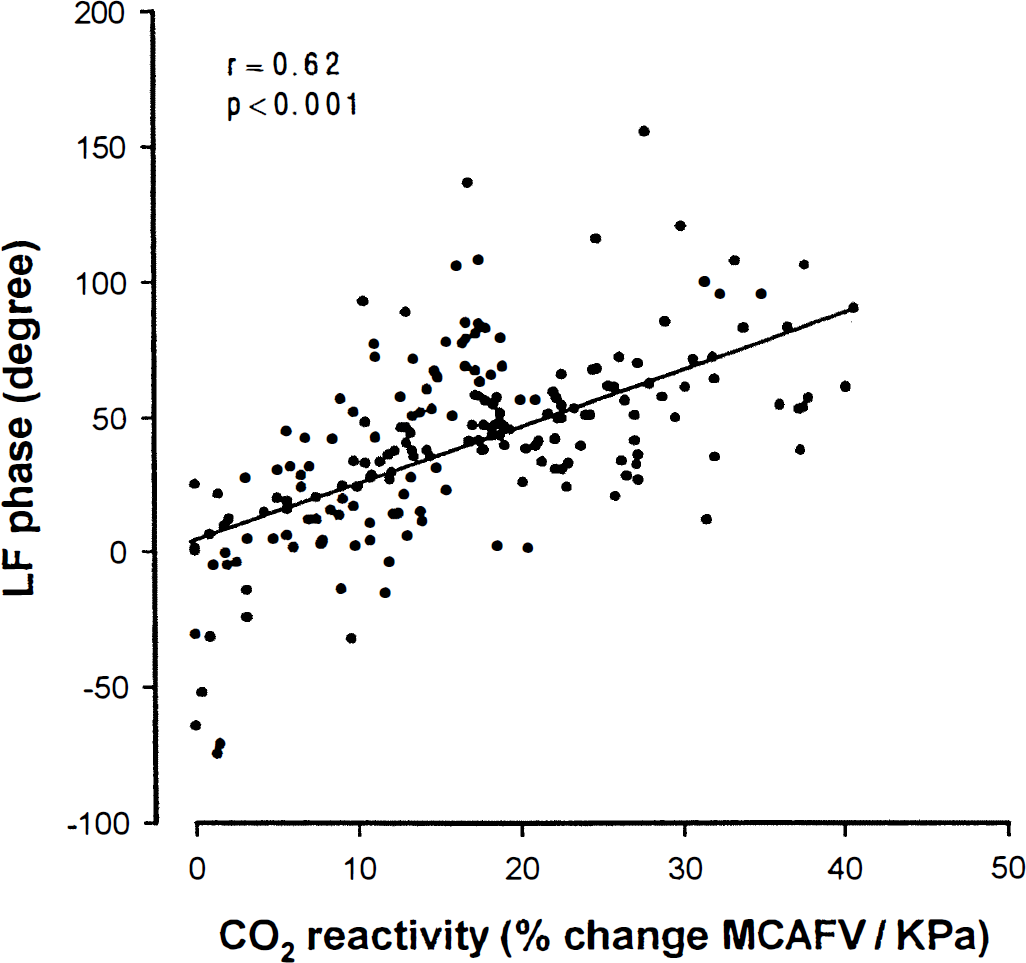
Correlation between the transfer function phase angle (LF) and the CO2 test on the side of each carotid artery from normal to varying degrees of stenosis (occlusion). The × axis shows percent change in middle cerebral artery blood flow velocity (MCAFV) per unit change in end-expiratory PCO2 in kilopascals, the y axis, transfer function phase angle (LF).
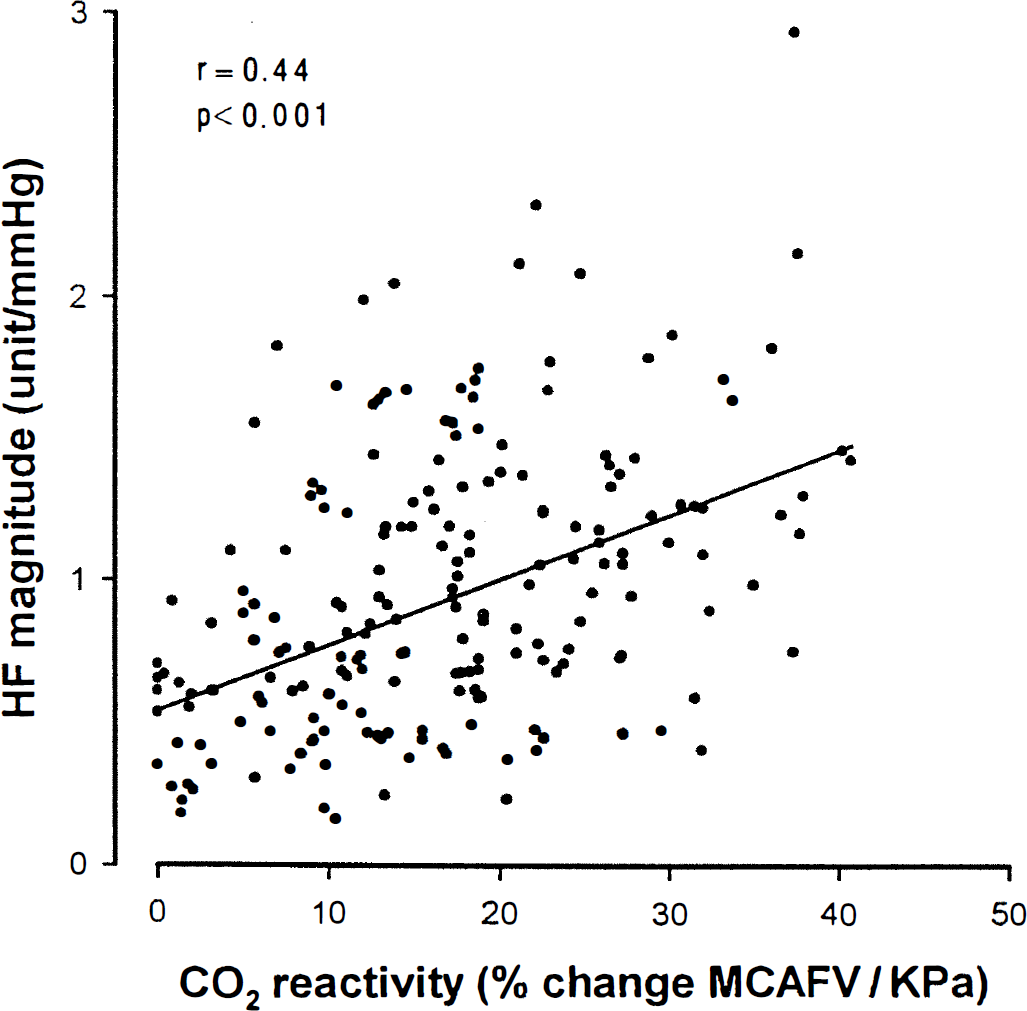
Correlation between the transfer function magnitude (HF) and the CO2 test on the side of each carotid artery from normal to varying degrees of stenosis (occlusion). The × axis shows percent change in middle cerebral artery blood flow velocity (MCAFV) per unit change in end-expiratory PCO2 in kilopascals, the y axis, transfer function magnitude (HF).
DISCUSSION
Using transfer function analysis of MCAFV and ABP, we found that measures of LF phase angle and HF magnitude were positively correlated with the traditional CO2 vasomotor reactivity test and all the measures of LF phase, HF magnitude, and CO2 reactivity were negatively correlated with the level of severity of carotid artery stenosis. These results support our previous findings that frequency domain analysis may have a great potential in the evaluation of CA (Kuo et al., 1998). The technique we used to assess the study participants with normal to various degrees of carotid artery stenosis allowed us to identify the impaired CA and vasomotor reserve ipsilateral to a hemodynamically significant high-grade carotid artery stenosis quickly and easily. During the past decade, most noninvasive techniques along with the use of TCD to assess CA require the application of special maneuvers (Widder et al., 1986; Giller, 1991; Diehl et al., 1995; Tiecks et al., 1995). Our method requires less cooperation from patients and has much less discomfort and risk for patients already experiencing cerebrovascular disease.
The result of this study is comparable with others in that CA in response to change in ABP is similar to the response of a high-pass filter and has a phase angle between the fluctuations of ABP and MCAFV within the LF range (Blaber et al., 1997; Diehl et al., 1995; Panerai et al., 1995). Because of the fact that this phase angle was reduced and negatively correlated with the degree of ipsilateral carotid artery stenosis, it could be used to evaluate pressure autoregulation.
The oscillating frequency at which the coherence function exhibited the largest values was chosen because this algorithm ensured the highest reliability to quantify the transfer function. Because the phase function is linear to the oscillating frequency in the LF range, the phase angle measured at a lower frequency with the largest coherence (in the LF range) is theoretically greater than the angle measured at a higher frequency in the same person; therefore, making an angle correction before doing any comparison or correlation analysis is justified. For example, all the obtained angles could be adjusted to a “corrected value” theoretically at 0.1 Hz, which has been used to study CA with regular respiration at 6 breaths per minute (Diehl et al., 1995). However, in this study, the correction of angle value improved little to the correlation coefficients in the analysis of the results because most of our study subjects already had the largest coherence value at a rather lower frequency in the LF range. Therefore, the correction of angle value may be useful only when study subjects have a wide distribution in the frequency of the largest coherence in the LF range. Further study is needed to correlate the LF phase shift angle of spontaneous fluctuation and the provoked fluctuation of ABP and MCAFV at 0.1 Hz to assess whether they measure the same autoregulatory function.
The magnitudes have been viewed as an indicator of the change of amplitude in MCAFV corresponding to the change in ABP. The belief is that with autoregulation changes in arteriole vessel diameter attempt to minimize the effect of blood pressure changes (Blaber et al., 1997). If this is the case, a smaller magnitude indicates more effective autoregulation and a greater magnitude represents an impaired autoregulation. On the contrary, in this study, we found that magnitudes of all three frequencies were greater ipsilaterally to normal carotid arteries, which are expected to have better autoregulation and intact vasomotor reserve (Table 1). A large reduction in spontaneous MCAFV oscillation in a frequency between 0.4 and 9 cycles/min ipsilaterally to the ICA lesion has been reported by Diehl et al. (1991). Oscillation in a frequency between 0.4 and 9 cycles/min covers both the LF and VLF ranges. Our result is consistent with Diehl et al. (1991) in that the magnitudes of both the LF and VLF ranges were reduced on the side ipsilateral to the hemodynamically stenotic carotid artery. Moreover, we found that the HF magnitude was not only reduced in carotid artery stenoses, but also significantly correlated with the vasomotor reserve assessed by the CO2 reactivity test. We agree with Diehl et al. (1991) that intracranial small vessels in the ipsilateral ICA territory are expected to be dilated, leading to a reduction in cerebrovascular resistance. The reduction in response to the provocation study that used hypercapnia to produce dilation of cerebral small vessels suggests that the capacity of small vessels to change their diameter over the affected ICA is reduced. The magnitude reduction ipsilateral to the affected ICA in patients with carotid artery stenosis is in excellent concordance with these characteristics of small vessels. In this study, we demonstrated that reduction of the HF magnitude could be interpreted as a function of vasomotor reserve and also could be used as a parameter for diagnosis of hemodynamically significant carotid artery stenosis.
The fact that HF magnitude correlated well with CO2 vasomotor reactivity, but the LF and VLF magnitudes were less prominent, needs further discussion. The HF fluctuations of MCAFV and ABP were both found to be associated with respiration (Newell et al., 1992; Novak et al., 1993; Brown et al., 1993). Respiratory activity continually perturbs cardiovascular hemodynamics through a number of direct and indirect mechanisms. One of these mechanisms is that changes in arterial PCO2, (resulting from short- and long-term changes in respiratory activity) modulate the activity of both afferent and efferent autonomic nerves in the brain stem and the periphery and central sensitivity to afferent impulses (Haymet and McCloskey, 1975; Saul et al., 1991). Thus, changes of PCO2 may influence both the results of the CO2 vasomotor reactivity test and HF fluctuations of MCAFV and ABP. This could partially explain our findings-a better correlation between the CO2 vasomotor reactivity test and the HF magnitude. Whether or not the magnitude of LF and VLF may be more correlated to other types of vasomotor stimuli, e.g., acetazolamide and mental stress, needs to be further investigated.
In conclusion, transfer function analysis of spontaneous fluctuations of ABP and MCAFV is a simple, convenient autoregulation test and is valid and reliable. We will continue our efforts to test its clinical application, such as for predicting the prognosis of asymptomatic carotid stenosis, separating of high- and low-risk patients, and determining whether a patient with carotid artery stenosis would benefit from extracranial-intracranial bypass surgery. In addition, we would also like to put our efforts toward assessing whether this simple autoregulation test is equivalent to or even better than a combination of the TCD dynamic autoregulation test and the TCD CO2 vasomotor reactivity test.
