Abstract
The ischemic tolerance is known to show protective effects on the neurons and the restricted Ca2+ influx through Ca2+ channels might be involved. In α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptor, ribonucleic acid (RNA) editing of the GluR2 subunit determines receptor desensitization and Ca2+ permeability. The authors investigated the effect of ischemic tolerance on the messenger RNA editing of Q/R and R/G sites of GluR2 subunit in hippocampus. It was found that the rate of RNA editing in Q/R site showed no change (100% edited), whereas that in R/G site decreased significantly (83.3% normal editing level to 60.4%) at day 3 (preconditioning period) and returned to normal level at day 14 (after preconditioning period). Further investigation revealed that the decrease of editing rate in ischemic tolerance resulted mainly from the decrease of editing in CA1 area.
Keywords
Ischemic tolerance that is caused by preceding nonlethal ischemic insult is known to exhibit protective effects against delayed neuronal death in hippocampal neurons (Kitagawa et al., 1990; Kirino et al., 1991; Kitagawa et al., 1991; Glazier et al., 1994). As an established model of ischemic tolerance, 2-minute ischemic treatment of gerbil hippocampus has been used and studied (Kitagawa et al., 1990; Kirino et al., 1991; Shimazaki et al., 1998). This phenomenon is intriguing in view of the induction of tolerance to ischemia in neuronal cells of brain, but its mechanism still is unclear. The restriction of Ca2+ influx into neuronal cells through Ca2+ channels may be involved in this mechanism (Nakata et al., 1992). The initial major event triggered in the process of ischemic neuronal damage is a breakdown of ion homeostasis and a rise in intracellular Ca2+ concentration initiated a cascade of events, leading to cell death (Haddad and Jiang, 1993; Tsubokawa et al., 1994). To prevent damage caused by this Ca2+ toxicity, it is supposed that the Ca2+ permeability of N-methyl-D-aspartate (NMDA) and α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors may be reduced in ischemic tolerance, even with a increase of glutamate concentration in the extracellular fluid. Depolarization using AMPA receptor activation removes Mg2+ from the channel of NMDA receptor (Nowak et al, 1984; Schoepfer et al, 1994), and Ca2+-permeability of NMDA receptor can be increased. The AMPA receptor is impermeable to Ca2+ as long as it contains at least one subunit of GluR2, which is edited in Q/R site (Hollmann et al., 1991; Verdoorn et al., 1991). An ionic permeability and a rectification of GluR2 are controlled by the ribonucleic acid (RNA) editing in the pore region (Q/R site). In addition, desensitization and activity of GluR2 are determined by the editing of GluR2 at R/G site (Lomeli et al., 1994).
In the current study, we examined the possibility that ischemic tolerance might be led to the change of RNA editing in GluR2 (at Q/R and R/G sites) or its expression level, both of which may cause the change of Ca2+ permeability in NMDA or AMPA receptors.
MATERIALS AND METHODS
Animal experiments for molecular biology
Adult Mongolian gerbils (body weight 60 to 70 g) were anesthetized with ether inhalation. Both common carotid arteries were exposed, and bilateral cerebral ischemia was produced by occlusion of these arteries with miniature aneurysmal clips. During the operation, body and tympanic temperature were monitored and controlled at 36.5 to 37.0°C using warming blankets. After 1, 2, or 5 minutes of ischemia, clips were removed and restoration of blood flow was visually verified. Animals were divided into a sham-operated group (without ischemia), and 1-, 2-, and 5-minute ischemic groups. Animals of the sham-operated and 2-minute ischemic groups were decapitated at postischemic recirculation intervals of 3 days and 14 days (n = 3 per time point). Animals of the 1-minute ischemic group were killed at the postischemic interval of 3 days only (n = 3). Animals of 5-minute ischemic group were killed at the postischemic interval of 1 day only (n = 3).
Histologic measurement after hippocampal ischemia
Total 30 male gerbils (weighing 60 to 70 g) were divided into six groups by preconditioning times and intervals of subsequent 5-minute ischemia: 5-minute ischemia after 3 days of 1- or 2-minute ischemia or sham operation; 5-minute ischemia after 14 days of 2-minute ischemia; 5-minute ischemia only; and sham operation only (normal control). Animals were anesthetized with ether inhalation after 1 week of second ischemia or sham operation, and were transcardially perfused with 4% phosphate-buffered paraformaldehyde after a flush of 0.1 mol/L phosphate-buffered saline. Paraffin sections were made in the coronal plane at a thickness of 10 μm with a microtome and stained with hematoxylin-eosin. The average value of neuronal density from both sides of hippocampal CA1 area in one section (corresponding roughly to 1.7 mm posterior to the bregma) was counted for each animal by a person who was blind to the study.
Ribonucleic acid isolation and reverse transcriptase-polymerase chain reaction
Bilateral hippocampi were dissected from brains after each interval from operation. Total RNA was extracted by Trizol reagent (Gibco-BRL, Rockville, MD, U.S.A.), and 3 μg of total RNA from each gerbil was reverse transcribed at 42°C for 60 minutes with SuperScript II Reverse Transcriptase (Gibco-BRL) using oligo (dT) primer. Cyclic deoxyribonucleic acid (DNA) solution (1 μL; equivalent to 150 ng of total RNA) was used for amplifying a fragment spanning the edited base located in second (Q/R site) and fourth (R/G site) transmembrane region of gerbil GluR2. Polymerase chain reaction (PCR) was carried out according to standard protocols (Taylor, 1991). To quantify GluR2 messenger RNA, the PCR condition of 95°C, 30 seconds; 58°C, 30 seconds; 72°C, 20 seconds; 28 cycles was used with GluR2-specific primers (G1-F and G1-R primers). For the analysis of RNA editing, the PCR condition of 95°C, 30 seconds; 58°C, 30 seconds; 72°C, 30 seconds; 28 cycles was used with Q/R site-specific primers (G1-F and G1-R primers) and R/G site-specific primers (G2-F and G2-R primers). The sequence of primers were as follows: G1-F (upper strand primer of Q/R site) 5′-AGCCCCTACGAGTGGCACACTG-3′, G1-R (lower strand primer of Q/R site) 5′-TCAGGGTAAAGAACCACCACAC-3′, G2-F (upper strand primer of R/G site) 5′-GAGTACATCGAGCAGAGGAAGC-3′, and G2-R (lower strand primer of R/G site) 5′-ATCAAAGCCACCAGCATTGCCA-3′. Simultaneously, G3PDH gene was amplified using G3PDH-specific primers (Clontech, Palo Alto, CA, U.S.A.) for 28 cycles (95°C, 30 seconds; 60°C, 30 seconds; 72°C, 20 seconds) as a control of GluR2 quantification. Between 25 and 31 amplification cycles, the amount of cyclic DNA in each PCR product was verified to give linear signal with both GluR2 (G1-F:G1-R and G2-F:G2-R) and G3PDH primers.
Semiquantitative reverse transcriptase-polymerase chain reaction of GluR2 in hippocampus
For quantification of GluR2, PCR product (10 μL each) of sham-operated and ischemic condition at each time point was run on a 2% agarose gel and stained by ethidium bromide. The intensity of PCR bands was measured by a dual-wavelength TLC scanner. The abundance of PCR products was calculated with the normalization relative to the intensity of the G3PDH PCR product (10 μL of each product). Cyclic DNA sample of each animal was PCR amplified three times independently, and the densitometric ratios in Fig. 1 were obtained by comparing the density of each PCR product band against the density of one control G3PDH PCR product (average of three PCR products). After the normalization, the densitometric data of each animal were analyzed statistically.
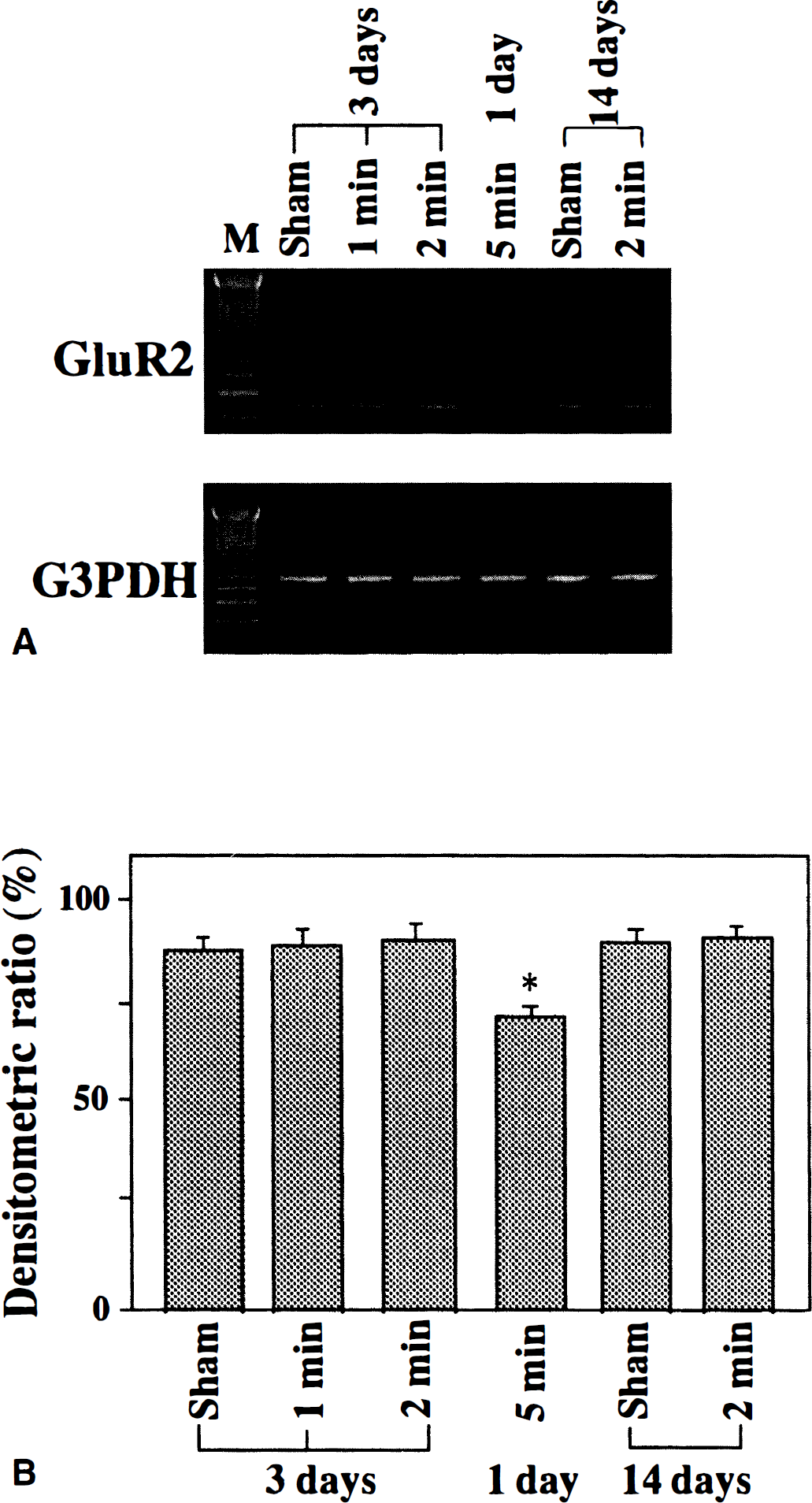
Effect of ischemic tolerance on expression levels of GluR2 messenger ribonucleic acid (mRNA).
Analysis of ribonucleic acid editing by sequencing
The PCR products from Q/R site and R/G site were ligated into pCR-Script SK(+) vector (Stratagene, La Jolla, CA, U.S.A.) and transformed into Escherichia coli. Colonies were processed and sequence determined by ABI 377 DNA sequencer by the Sanger method (Sanger et al., 1977). About 86 to 90 colonies from each experimental group (n = 3, one PCR for each animal in one group, total of three PCR in one group) were analyzed and the editing rate was calculated statistically.
Measurement of editing ratio in each hippocampal area by microdissection
The CA1, CA3, and dentate gyrus region of both hippocampi were microdissected from each animal (n = 3) of shamoperated (day 3) and 2-minute ischemic (day 3) groups. The RNA was isolated from each sample following the method described earlier, and sequencing analysis was done as the same procedure (68 to 75 sequence data in each group).
Analysis of ribonucleic acid editing by polymerase chain reaction
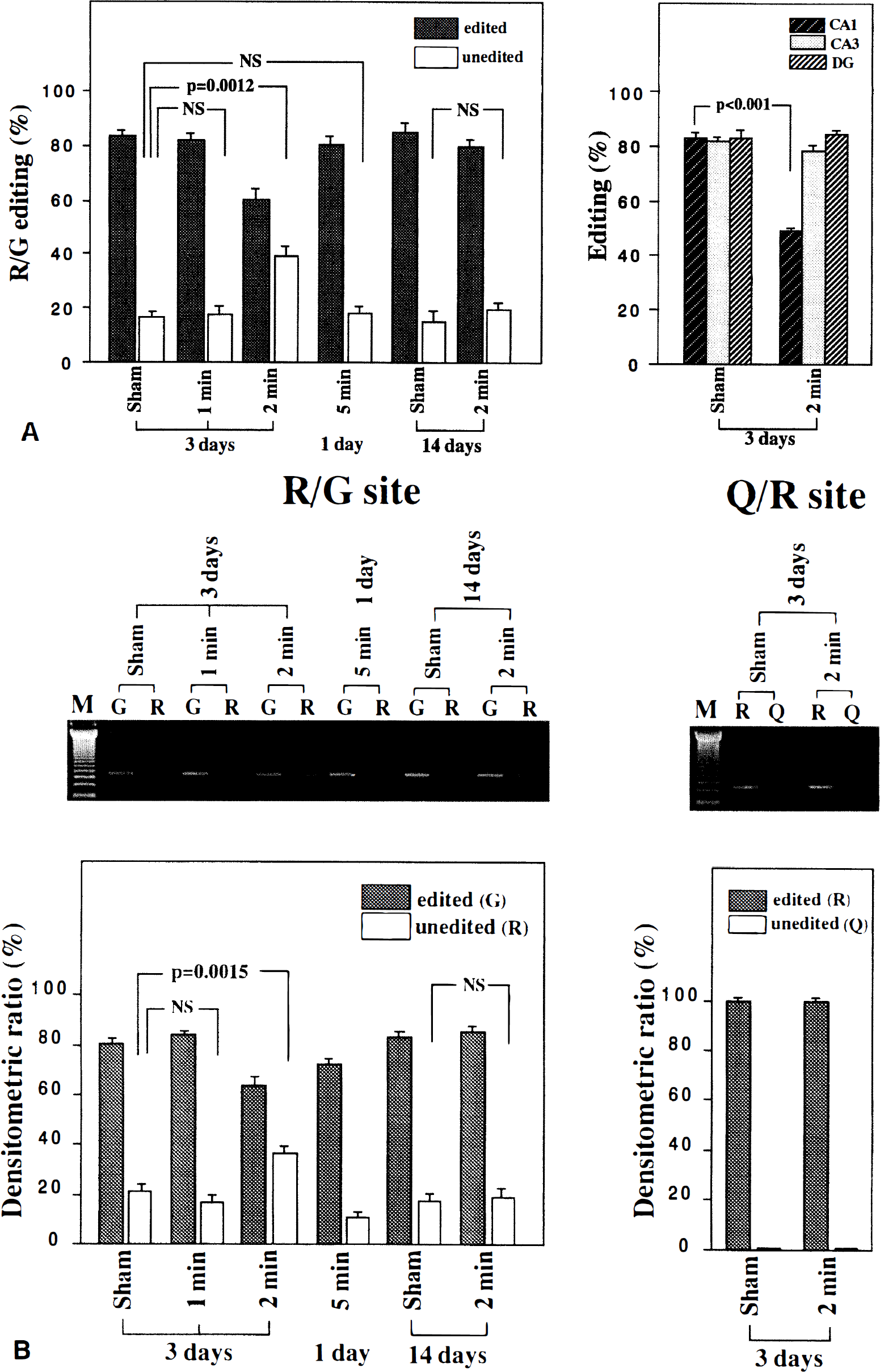
The RNA editing at the R/G site also was analyzed by primer extension method, which was described previously (Wisden et al., 1991), with some modifications. Cyclic DNA solutions (see earlier) were amplified with an upper strand primer Glu-RTM4 (5′-CCACACCTAAAGGATCCTCATTAA-3′) or a primer Glu-GTM4 (5′-CCACACCTAAAGGATCCTCATTAG-3′) and a lower strand primer Glu-LTM4 (5′-TAACACTCTCGATGCCATATACG-3′), or amplified with an upper strand primer Glu-QTM2 (5′-GTTTTCCTTGGGTGCCTTTATGCA-3′) or a primer Glu-RTM2 and a lower strand primer Glu-LTM2 (5′-CCACATTTTATCAAACACTGCGAT-3′). The PCR condition was as follows: 95°C, 30 seconds; 65°C, 30 seconds; 72°C, 20 seconds; 25 cycles. The PCR products (20 μL each) were run on a 2% agarose gel and stained by ethidium bromide. The intensity of signals on the gel was measured as described earlier. The densitometric ratios in Fig. 2B were obtained from the average of three PCR products in each cyclic DNA sample of various conditions. The density of a sham-operated (day 3) sample in R/G site and Q/R site was designated to be 100% when the density of edited and unedited were summed, and the densitometric ratio of the other samples in each site was calculated.
Statistical analysis
Data are presented as means ± SD. Statistical analysis was carried out by analysis of variance followed by Scheffé's F test.
RESULTS
The survival ratio of neuronal cells in CA1 area after 1 week of 5-minute ischemia with or without the pretreatment of 1- or 2-minute ischemia was measured. The 5-minute ischemia was given after 3 days or 14 days of the first ischemia. The histologic data in Table 1 represents that ischemic tolerance was present at 3 days after 2-minute ischemia (66.8% of neuronal cells survived in CA1 area after 5-minute ischemia against 4.4% in shamoperated control) and absent at 14 days after 2-minute ischemia (3.5% survived). In contrast, 1-minute ischemia did not show tolerance after 3 days from operation (6.7% survived).
Neuronal cell density in CA1 region
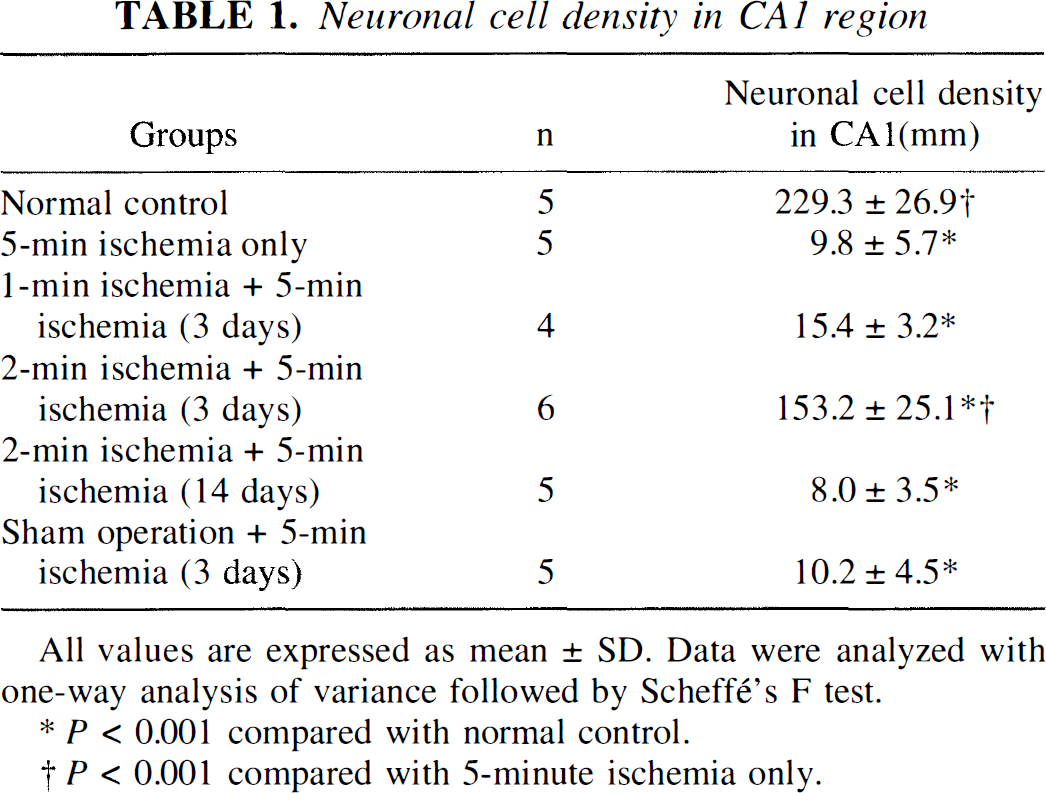
All values are expressed as mean ± SD. Data were analyzed with one—way analysis of variance followed by Scheff's F test.
P < 0.001 compared with normal control.
P < 0.001 compared with 5-minute ischemia only.
The expression level of GluR2 messenger RNA in ischemic tolerance (3 days after 2-minute ischemia), nonischemic tolerance (sham-operated) or out of the course of ischemic tolerance (14 days after 2-minute ischemia) showed no difference (Fig. 1). However, the expression level of GluR2 messenger RNA at 1 day after 5-minute ischemia showed about a 15% decrease.
Using animals treated in the same time course and conditions as those in Fig. 1, RNA editing at the R/G site of GluR2 was evaluated by sequencing (Fig. 2A). It was indicated that only at the state of ischemic tolerance (3 days after 2-minute ischemia) did a proportion of RNA editing change significantly (P = 0.0012 compared with sham-operated control). The percentage of edited and unedited forms at each time course are as follows: edited and unedited forms in sham-operated, 1-minute ischemia, and 2-minute ischemia at 3 days after operation were 83.3 ± 4.1%:16.7 ± 4.1%, 81.6 ± 4.5%:18.4 ± 4.5%, and 60.4 ± 6.2%:39.6 ± 6.2%, respectively. Those in 5-minute ischemia at 1 day after operation was 80.3 ± 4.5%:19.7 ± 4.5%. Whereas those in sham-operated and 2-minute ischemia at 14 days after operation were 84.5 ± 6.4%:15.5 ± 6.4% and 79.8 ± 3.2%:20.2 ± 3.2%, respectively. Microdissected tissue RNA samples revealed that the change of R/G editing in 2-minute ischemic group (3 days after operation) was mainly caused by the decrease of editing in CA1 area. The percentage of edited form at each area in sham-operated group was CA1, 83.2 ± 2.1%; CA3, 82.1 ± 1.8%; and dentate gyrus, 83.3 ± 3.4%; whereas that in 2-minute ischemic group was CA1, 48.5 ± 1.7%; CA3, 78.6 ± 3.8%; and dentate gyrus, 83.9 ± 2.1%, respectively.
In addition, primer extension method with site-specific primers (G-specific and R-specific primers of R/G site, and R-specific and Q-specific primers of Q/R site) was performed (Fig. 2B). Densitometric ratios of edited and unedited forms in sham-operated and 1- and 2-minute ischemia at 3 days after operation were 81.5 ± 3.8%:22.2 ± 4.3%, 83.7 ± 1.9%:18.7 ± 2.9%, and 66.3 ± 5.9%:38.7 ± 3.7%, respectively. Those in 5-minute ischemia at 1 day after operation were 76.3 ± 3.1%:16.7 ± 1.9%. Whereas those in sham-operated and 2-minute ischemia at 14 days after operation were 83.6 ± 2.9%:23.3 ± 3.6% and 84.3 ± 3.8%:22.5 ± 4.9%, respectively. There was a significant difference between the group of 3 days after 2-minute ischemia and controlled sham-operated groups (P = 0.0015). The group of 5-minute ischemia (1 day after operation) showed decrease of signals both in edited and unedited group. This result might be caused by the decrease of expression level of GluR2 with 5-minute ischemia as shown in Fig. 1. The ratio of edited and unedited GluR2 RNA after 5-minute ischemia shown in Fig. 2B and the ratio of sham-operated groups (3 days and 14 days) in Fig. 2B had no significance. The results shown in Fig. 2A and ratios of edited and unedited GluR2 in each group shown in Fig. 2B were representative.
On the other hand, the RNA editing of Q/R site in each group also was studied by sequencing and primer extension method (Fig. 2B). By sequencing analysis, it was found that the Q/R site in all groups was mostly edited and was independent of ischemic condition or from the length of the recirculation interval (data not shown). Primer extension method in Fig. 2B showed the same result in sham-operated and 2-minute ischemic groups (3 days after operation).
DISCUSSION
We have shown that ischemic tolerance by the pretreatment of 2-minute ischemia significantly decreased the death of hippocampal cells in CA1 area caused by following 5-minute ischemia as presented in Table 1. This protective effect is prominent at day 3 after 2-minute ischemia but is less effective at day 14. At day 3 after 2-minute ischemia, the editing rate in R/G site in ischemic tolerance changed significantly and unedited form of GluR2 subunit increased, then it decreased to the normal level reversibly by day 14 of treatment when no tolerance was observed. This change of editing rate did not occur when the ischemic tolerance was not formed (sham-operated) or after 5-minute ischemia treatment only. The microdissection study in Fig. 2A revealed that the change in editing rate occurred mainly in the CA1 area of hippocampus, where the neuronal cells are most sensitive to ischemic insult. The distribution and expression of GluR2 subunit were well studied, and it was known that GluR2 is expressed mainly in neuronal cells in hippocampus (Kohama et al., 1998; He et al., 1998). From these observations, our data may reflect mostly the change in the editing rate of GluR2 in CA1 neuronal cells. The increase of unedited GluR2 RNA, which is consistent with the time course of the cell protection effect, suggests that RNA editing may play an important role in ischemic tolerance.
The precise mechanism and role of RNA editing in ischemic tolerance are unknown. It has been reported that the unedited GluR2 in R/G site showed a slower recovery rate from desensitization than that of edited one (Lomeli et al., 1994). Therefore, the increase of unedited GluR2 in R/G site observed during ischemic tolerance is supposed to cause the functional modification of GluR2 subunit. This functional change of GluR2 may have an effect on the total activity of AMPA receptors and, furthermore, on decrease of Ca2+ permeability through NMDA receptors, which is important for survival of neuronal cells.
It also is intelligible that the editing of Q/R site of GluR2 was not affected by 2-minute ischemia. The Q/R site had been edited almost 100% in normal condition, and this edited form of GluR2 makes Ca2+ impermeable (Paschen et al., 1996; Rump et al., 1996). Therefore, a lack of change of RNA editing in the Q/R site in ischemic tolerance was in agreement with the physiologic meaning of cell protection from excessive Ca2+ influx.
As well as this RNA editing, the expression level of GluR2 in ischemic tolerance also was examined, and it was clarified that ischemic tolerance did not affect the expression of GluR2 messenger RNA. However, the treatment of 5-minute ischemia caused a decrease of GluR2 expression, which is consistent with the report by Gorter et al. (1997).
In conclusion, our data suggest that the modification of RNA editing of R/G site in GluR2 subunit rather than the alteration of its expression level may play an important role in ischemic tolerance. This study contributes to understanding the mechanism of ischemic tolerance and the physiologic roles of RNA editing in brain.
