Abstract
These studies tested the hypothesis that the cerebral vasospasm that follows subarachnoid hemorrhage (SAH) is due to alterations in endothelin (ET) and ET receptor expression. Eight monkeys underwent cerebral angiography and induction of SAH. Angiography was repeated 7 days later to confirm the presence of cerebral vasospasm, and animals were killed. RNA was isolated from right (vasospastic) and left (control) side middle cerebral arteries and surrounding cerebral cortex. The levels of prepro (PP) ET-1 (ppET-1) and ppET-3 and ETA and ETB receptor mRNAs were determined using a quantitative reverse transcriptase polymerase chain reaction-based assay. ET-1 peptide was also measured in CSF at baseline and after 7 days. Specific agonist binding to ETA and ETB receptors in both middle cerebral arteries and in surrounding brain cortex was measured in three animals by autoradiographic binding assays. Levels of ETB receptor mRNA were 3.4 ± 2.2-fold higher in the right than in the left cerebral arteries (p < 0.01). There were no significant differences in the levels of ppET-1, ppET-3, or ETA receptor mRNA in cerebral arteries. ET-1 peptide was not elevated in CSF. Levels of ETA and ETB receptor mRNAs were 2.6 ± 1.1- and 2.1 ± 1.3-fold higher, respectively, in the right than in the left cerebral cortex, while the level of ppET-3 mRNA was 2.1 ± 1.0-fold lower. There were no differences in ppET-1 mRNA levels between right and left cerebral cortex. Binding to ETA and ETB receptors in cerebral arteries and cortex did not differ significantly between right and left sides. These results do not support the hypothesis that overexpression of ET-1 is the principal cause of vasospasm, but rather they suggest that SAH causes complex changes in the ET system that together are responsible for the cellular response to SAH.
Keywords
The endothelins (ETs) are a family of three related vasoconstricting peptides that have been implicated in the pathogenesis of the vasospasm that complicates aneurysmal subarachnoid hemorrhage (SAH) (Sokolovsky, 1992; Cosentino and Katusic, 1994). The ETs act on at least two ET receptor subtypes (ETA and ETB receptors) (Hori et al., 1992; Sokolovsky, 1992; Elshourbagy et al., 1993). Evidence that ETs are involved in cerebral vasospasm includes the finding that ET-1 and ET-3 are elevated in the CSF in some cases of vasospasm (Masaoka et al., 1989; Suzuki et al., 1990; Gaetani et al., 1994; Seifert et al., 1995). They are potent vasoconstrictors, and oxyhemoglobin, a possible cause of vasospasm, increases the release of ET-1 from endothelial and smooth muscle cells (Ohlstein and Storer, 1992; Fuwa et al., 1993; Kasuya et al., 1993). Several studies have found that drugs that antagonize the action of ETs prevent or reverse experimental vasospasm (Itoh et al., 1994; Zuccarello et al., 1994; Hino et al., 1995).
We have examined the molecular basis of vasospasm using a competitive reverse transcriptase (RT) polymerase chain reaction (PCR)–based assay to measure the levels of mRNAs for two ET peptide precursors [prepro ET-1 (ppET-1) and ppET-3] and two receptor subtypes (ETA and ETB) in cerebral arteries and cortex of monkeys exposed to SAH. We also carried out autoradiographic binding assays to measure the specific binding of ETA and ETB receptors in cerebral arteries and cortex and assays to measure ET-1 peptide in CSF.
MATERIALS AND METHODS
Angiography and SAH
Unilateral SAH was created in eight cynomolgus monkeys (Macaca fascicularis) as detailed previously (Macdonald et al., 1992; Hino et al., 1995). On day 0, animals were sedated with ketamine 10 mg/kg i.m. and intubated and ventilated on O2 with 0.5–3% isofluorane. Arterial blood pressure, heart rate (Criticon Dinamap Research Monitor; Criticon, Tampa, FL, U.S.A.), end-tidal Pco2 (Nellcor N-100 pulse oximeter, Hayward, CA, U.S.A.), and body temperature (Yellow Springs 43TA; Yellow Springs Instrument Co., Yellow Springs, CO, U.S.A.) were continuously monitored. Cerebral angiography was performed by injection of 60% iothalamate meglumine into a catheter inserted into the right axillary artery. A right frontotemporal craniectomy was then carried out, and the arachnoid over the right internal carotid, anterior cerebral, and middle cerebral arteries was dissected off to completely expose the arteries. Five milliliters of clotted autologus arterial blood was placed around these arteries. Seven days later (day 7), angiography was repeated to confirm the presence of vasospasm. In this model, the right cerebral arteries develop vasospasm and the left arteries remain of normal caliber and represent control arteries for each animal. The animals were given pentobarbital 100 mg/kg. The brains and cerebral arteries were removed and stored at −80°C.
All procedures on animals were approved by the Animal Care and Use Committee of the University of Chicago.
Isolation of RNA and preparation of primers for RT-PCR
The modified acid/guanidinium thiocyanate/phenol/chloroform method was used to isolate total RNA from each tissue sample from each animal (Chomczynski, 1993). The amount of RNA was 2.4 ± 1.3 μg for cerebral artery and 14 ± 4 μg for cerebral cortex (mean ± SD, n = 16). The wet weight of the tissue samples was <1 mg for cerebral artery and 3–6 mg for cerebral cortex. One microgram of RNA from each sample was reverse-transcribed as described later. In control experiments, the efficiency of reverse transcription was 42% (Y. Tokuyama, personal communication, 1994).
The monkey sequences of ppET-1, ppET-2, ppET-3, and ETA and ETB receptor mRNAs were not known so partial nucleotide sequences for these genes as well as for β-actin were determined. Genomic DNA or RNA was isolated from monkey thyroid, lung, heart, or aortic tissue (Iwashima et al., 1993). RNA was reverse-transcribed into cDNA using murine myeloma leukemia virus RT (Gibco BRL, Gaithersburg, MD, U.S.A.) and oligo dT16 primer (Perkin Elmer, Norwalk, CT, U.S.A.). The genomic DNAs or cDNAs of interest were amplified using PCR and degenerate oligoprimers based on the human nucleotide sequences of the target genes. The PCR products were subcloned into pGEM-3Z (Promega, Madison, WI, U.S.A.) and sequenced (Sequenase version 2.0; United States Biochemical, Cleveland, OH, U.S.A.). There were 97, 87, 98, and 99% identity between the sequences of the partial cDNA sequences of monkey ppET-1 and −3 and ETA and ETB receptors, respectively, and the corresponding human sequences. The high degree of sequence homology between human and monkey ETs and ET receptors makes it extremely unlikely that sequences were amplified for proteins other than those desired. Specific primers for RT-PCR were selected using the monkey cDNA sequences (Table 1). A search of the GenBank, using the primer sequences, did not reveal sequences other than the desired ET or ET receptors that exhibited high homology with the primers. Competitor DNA templates for quantitative RT-PCR assays were prepared from the cloned cDNA using PCR-based mutagenesis to introduce a small deletion so that the PCR products from the cDNA target and competitor template could be distinguished because of their difference in size (Vallette et al., 1989). The size difference was <10% so as to minimize possible differences in efficiency of PCR amplification. Each competitor DNA molecule was subcloned into pGEM 3Z and sequenced.
Nucleotide sequences of specific oligoprimers for competitive RT-PCR and PCR conditions
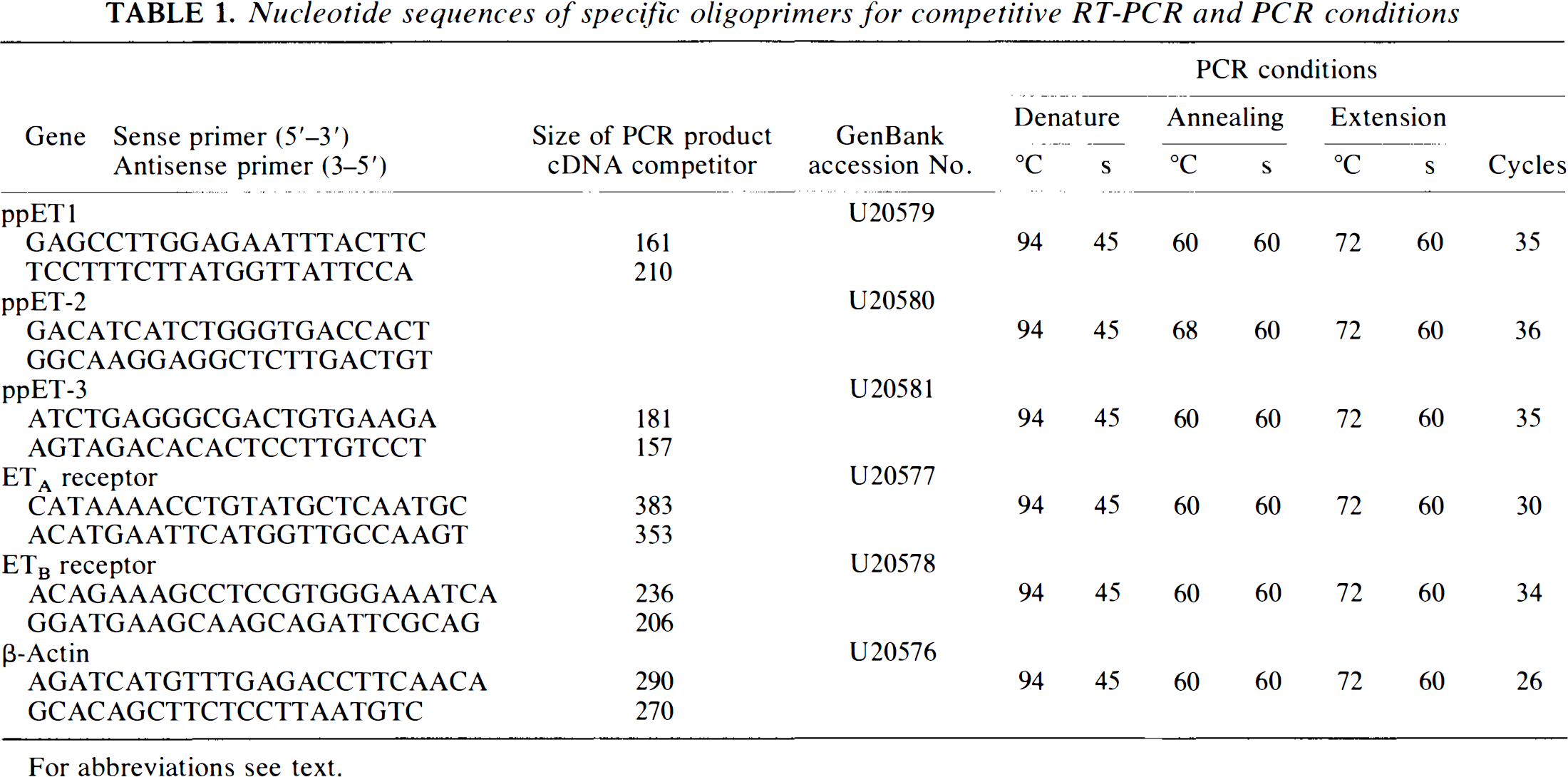
For abbreviations see text.
Competitive RT-PCR
Competitive RT-PCR was used to quantify the levels of mRNAs for ppET-1, ppET-3, ETA and ETB receptors, and β-actin. Known amounts of a competitor DNA molecule are added to each PCR, and the amount of target cDNA present in the sample is determined from the added competitor, which gives an equimolar amount of PCR product as the target cDNA (Siebert and Larrick, 1992; Iwashima et al., 1993).
PCR was carried out as described previously using 1 μCi of [α-32P]dCTP (3,000 Ci/mmol; Amersham, Arlington Heights, IL, U.S.A.), primers, and PCR conditions shown in Table 1 and using a Perkin Elmer GeneAmp System 9600 (Iwashima et al., 1993). The PCR products were separated by electrophoresis on a 5 or 7% Polyacrylamide gel, and the intensity of the bands corresponding to each specific PCR product was analyzed using a computed phosphoimage analyzer (Phospholmager; Molecular Dynamics, Sunnyvale, CA, U.S.A.).
Receptor binding assay and autoradiography
In three animals, 10-μm frozen sections of right and left middle cerebral artery and surrounding brain cortex were thaw mounted on poly-L-lysine-coated slides. After incubation at room temperature for 15 min, tissue sections were incubated for 90 min with 50 pM 125I-ET-1 in the presence of 0.5 μM ET-1 (to monitor nonspecific binding) or 0.5 μM BQ-3020 to measure ETA receptors. ETB receptors were labeled by incubating tissue sections with 100 pM 125I-BQ-3020 in the presence (for detection of nonspecific binding) or absence of 0.5 μM BQ-3020, a selective ETB receptor agonist. Following binding, sections were washed twice in buffer, coated with liquid emulsion film NBT2 (Eastman Kodak, U.S.A.), and exposed for 14 days at 10°C. The slides were developed in a Kodak D-19 developer at 19°C for 5 min. To obtain standard values, 125I-ET-1 at 20 and 200 nCi was added to rat brain homogenates (200 mg wet wt) and then frozen. Frozen tissues were cut at 10-μm thickness, mounted on coated slides, treated with liquid emulsion, and exposed for 14 days at 10°C. The slides were developed as described already. The binding of 125I-ET-1 and BQ-123 was measured as optical density (OD) in an image analyzer (BIOCOM 200; Computer Built, France) connected to a microscope. The amount of binding was calculated from the values of standard sections. Specific binding of ETA and ETB was calculated as follows: ETA = (125I-ET-1 + BQ-123) — (125I-ET-1 + ET-1) and ETB = 125I-BQ-123 -(125I-BQ-3020 + BQ-3020). Consecutive sections were stained with 0.1% toluidine blue for detection of morphological structures.
Measurement of ET-1 peptide
CSF was aspirated from the right-sided basal cisterns prior to induction of SAH and from the cisterna magna immediately prior to death on the 7th day. The level of ET-1 peptide was assayed using a commercially available enzyme immunoassay (Peninsula Laboratories, Belmont, CA, U.S.A.).
Statistical analysis
Angiographic arterial diameters were measured using an optical micrometer at predetermined points along each artery. Vasospasm was quantified by determining the percent change in arterial diameters between day 0 and day 7 angiograms. Comparison of amounts of mRNA between right and left cerebral arteries and cortex was made by paired Wilcoxon test and of day 0 and day 7 angiograms by paired t test. Comparison of ET-1 levels in CSF was by unpaired t test. All values are given as means ± SD. Significance was taken at p < 0.05.
RESULTS
Angiography
Comparison of angiograms taken before and 7 days after creation of SAH showed that there was a significant reduction in the diameter of the right middle cerebral artery (–43% ± 19%; p < 0.005) (Table 2). There were no significant changes in arterial diameters of the left-sided cerebral arteries. Delayed ischemic neurologic deficits did not develop.
Changes in angiographic arterial diameters and endothelin-1 (ET-1) levels in CSF
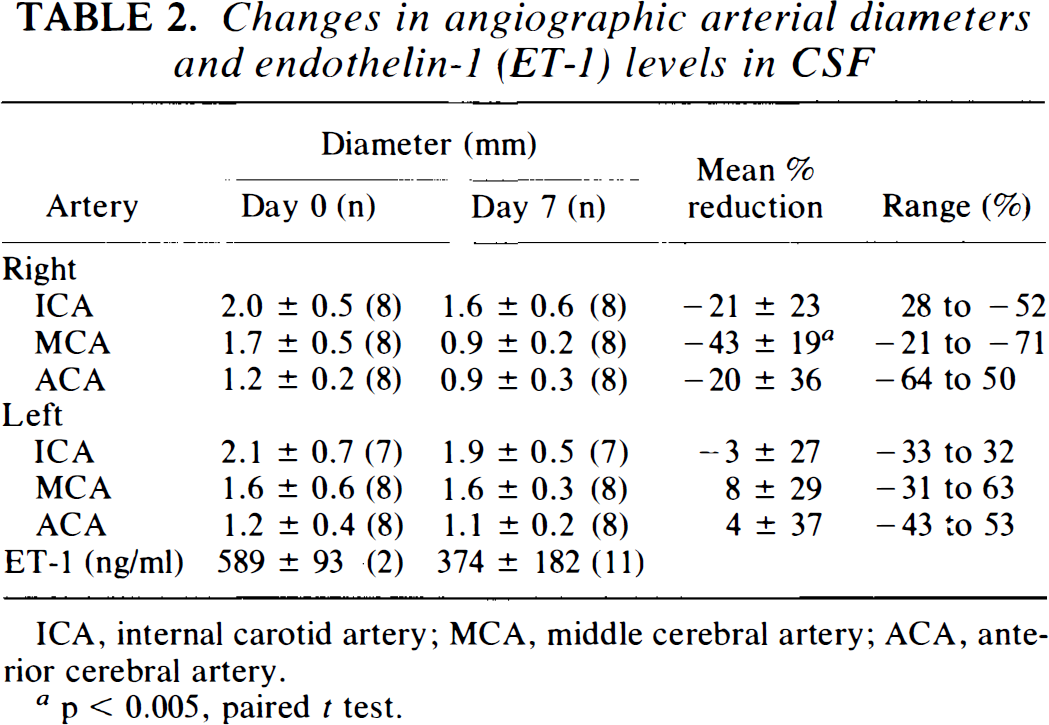
ICA, internal carotid artery; MCA, middle cerebral artery; ACA, anterior cerebral artery.
p< 0.005, paired t test.
Competitive RT-PCR
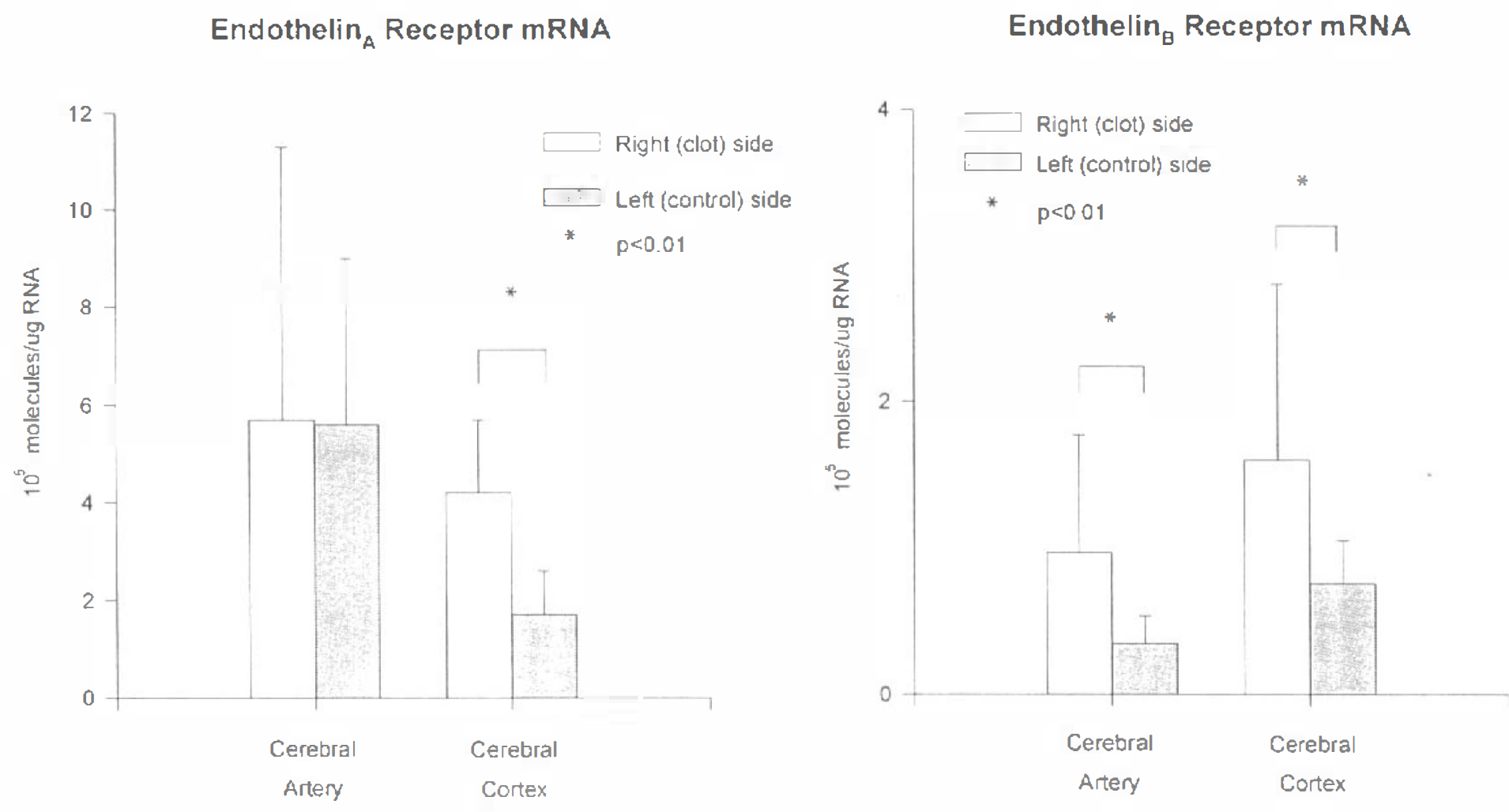
PreproET-1 and ETA and ETB receptor mRNA could be readily detected by RT-PCR in cerebral ateries, whereas ppET-3 mRNA was detected in some arterial samples and not others. The mRNA for the ETA receptor was 47 ± 70 times more abundant in cerebral artery than that for the ETB receptor. There was a significant 3.4 ± 2.2-fold increase in ETB receptor mRNA in vasospastic arteries (9.7 ± 2.8 × 104 molecules/μg RNA) compared with control left-side arteries (3.5 ± 0.7; p < 0.01) (Figs. 1–4). There were no significant changes in levels of mRNA for the ETA receptor, for ppET-1, or for β-actin, a control housekeeping transcript. PreproET-2 mRNA could be detected by RT-PCR in monkey cerebral artery and cortex. However, we were not able to quantify the levels of this mRNA by competitive RT-PCR because multiple bands were generated that DNA sequencing showed resulted from alternative splicing of monkey ppET-2 mRNA.
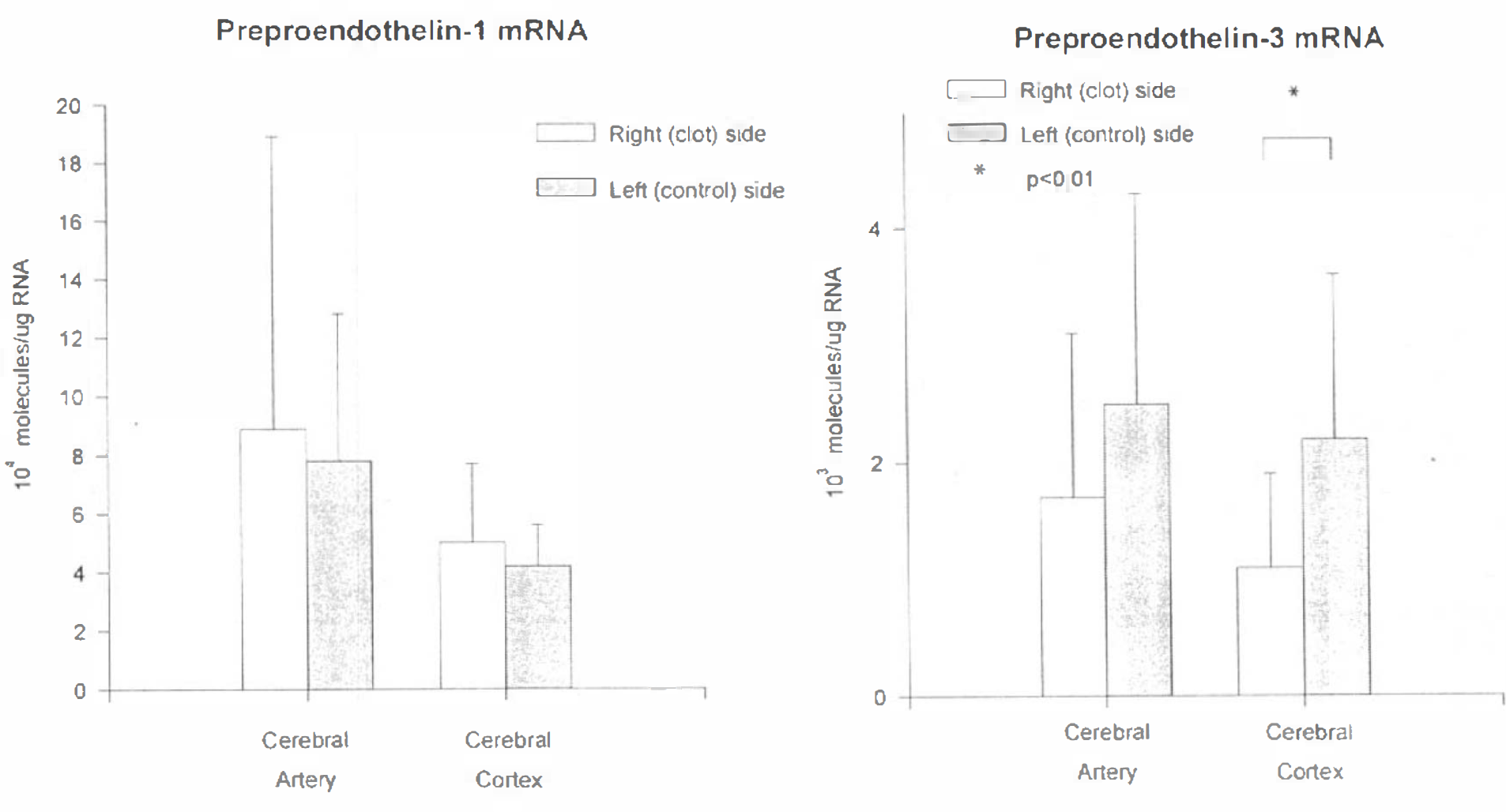
Bar graph showing amounts of preproendothelin-1 (ppET-1)
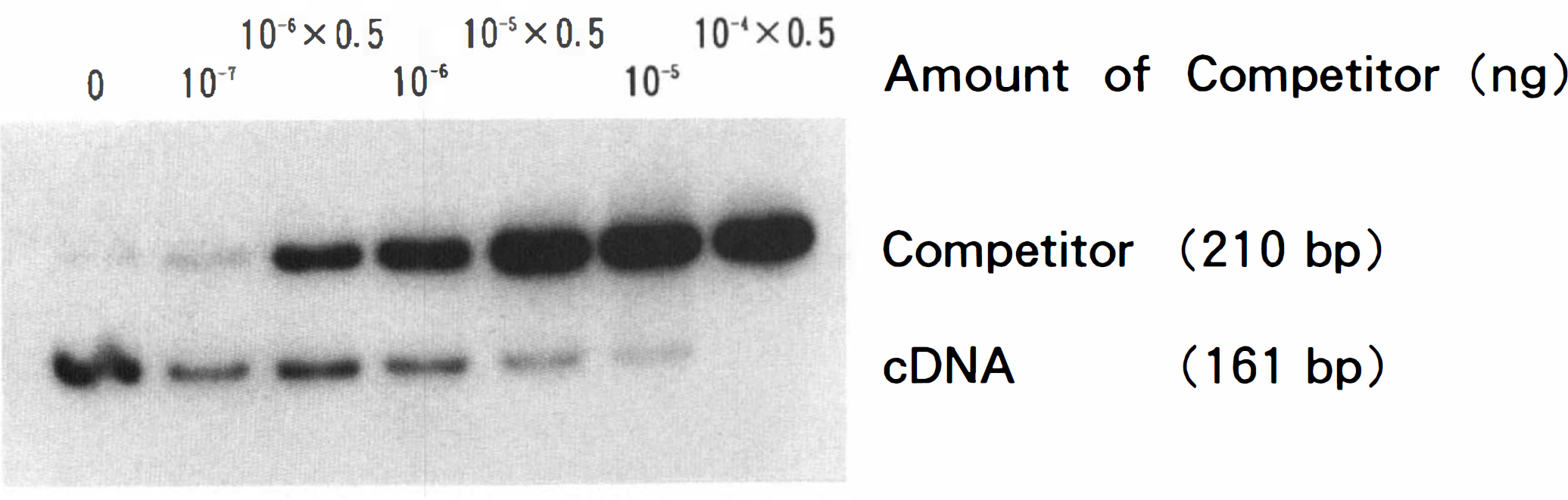
Image of example of competitive RT-PCR for ppET-1 mRNA in the left cerebral artery of a monkey. Upper bands are competitor cDNA and lower bands are target cDNA. Sixfold serial dilutions of the competitor were coamplified in each PCR with a constant amount (5 ng) of cDNA synthesized from monkey cerebral artery. The level of ppET-1 mRNA was determined by calculating the amount of competitor required to achieve equimolar amounts of products. Intensity of bands was quantified by image analysis, and values were corrected for difference in molecular size between competitor and target cDNA. For abbreviations see text.
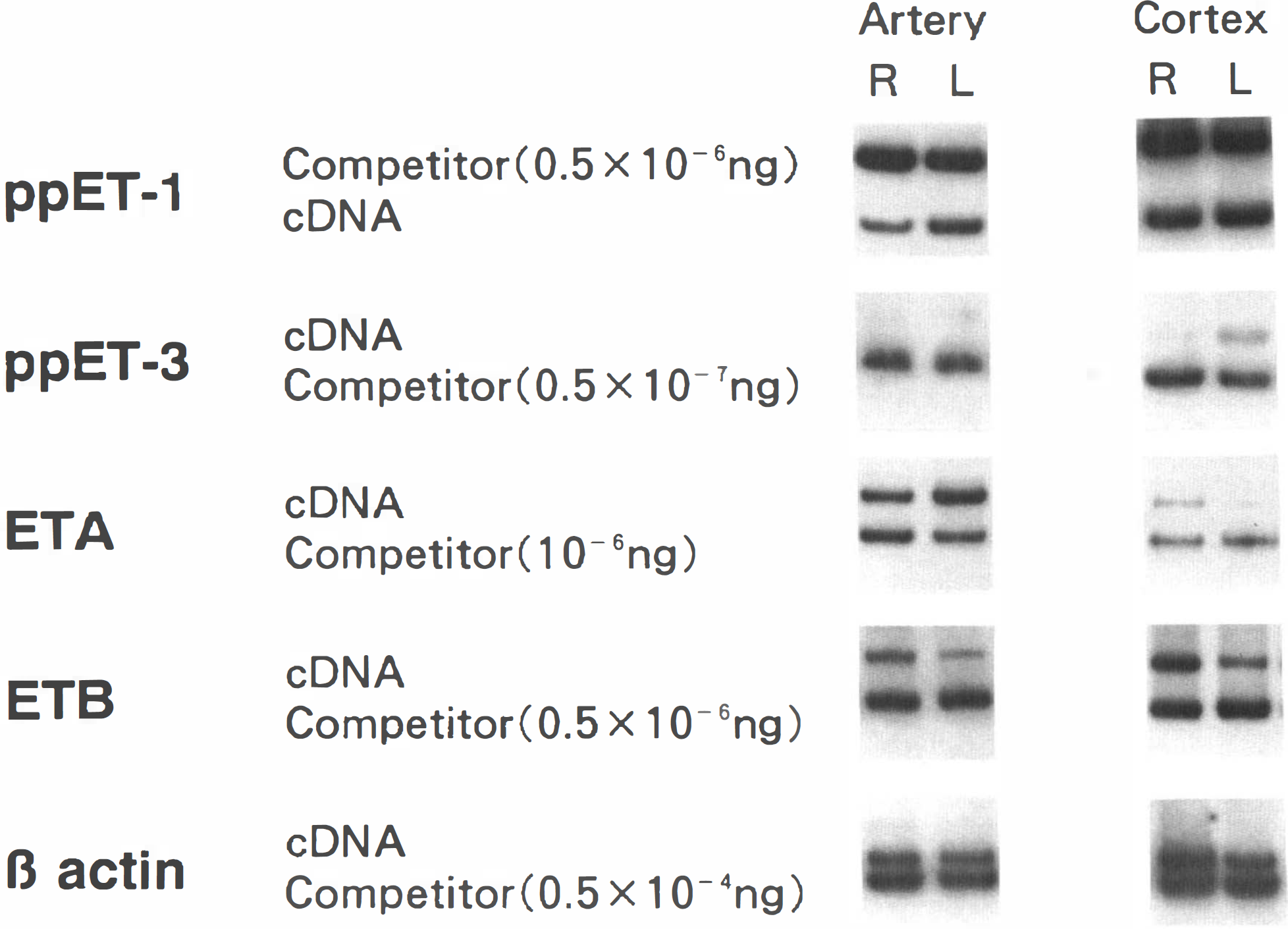
Images of gels showing comparisons of mRNA for ppET-1, ppET-3, β-actin, and the ETA and ETB receptors in right and left cerebral artery and cortex from one monkey. The amounts of competitor are the same between right and left side tissue in each PCR reaction for each target mRNA. There is an increase in ETB receptor mRNA in right cerebral artery and cortex, an increase in ETA receptor mRNA in right cerebral cortex, and a decrease in ppET-3 mRNA in right cerebral cortex. For abbreviations see text.
Bar graph showing amounts of endothelin A (ETA)
In cerebral cortex, mRNA was detected for ppET-1, ppET-3, and for the ETA and ETB receptors. The mRNAs for both the ETA and the ETB receptors were significantly increased in the cerebral cortex from the right (SAH) side (4.2 ± 1.5 × 105 and 1.7 ± 1.2 × 105 molecules/μg RNA, 2.6 ± 1.1- and 2.1 ± 1.1-fold, respectively; p < 0.01) (Figs. 1–4) compared with the left side (1.7 ± 0.9 × 105 and 0.8 ± 0.3 × 105 molecules/μg RNA, respectively). preproET-3 mRNA was significantly reduced 2.1 ± 1.0-fold in cortex from the right side (1.1 ± 0.8 × 103 molecules/μg RNA) compared with the left side (2.2 ± 1.5 × 103 molecules/μg RNA). There were no significant differences in mRNA for ppET-1 or for β-actin.
Receptor binding assays and autoradiography
Image analysis of autoradiographs showed that there was a 1.3 ± 0.3-fold increase in ETB receptor binding sites in the SAH side cerebral arteries compared with the left side, although this increase was not statistically significant (Table 3). In cerebral cortex, there was an increase in both ETA and ETB receptor binding sites on the SAH side. The microautoradiography revealed binding sites for both ETA and ETB receptors in the tunica media and tunica adventitia of the right and left cerebral arteries (Fig. 5). There was an increase in binding sites for the ETB receptor in the tunica media and adventitia of the right cerebral arteries. Although binding sites for the ETB receptor were observed on the endothelial cells of the tunica intima, the thinness of the endothelial cell layer and low receptor density precluded an assessment of changes in these receptors between right and left cerebral arteries. In the right cerebral cortex, there was a 1.2 ± 0.8-fold increase in ETA receptor binding sites and a 1.2 ± 0.2-fold increase in ETB receptor binding sites in three animals (not significant).
Results of binding assays for ETA and ETB receptors in cerebral arteries and cortex
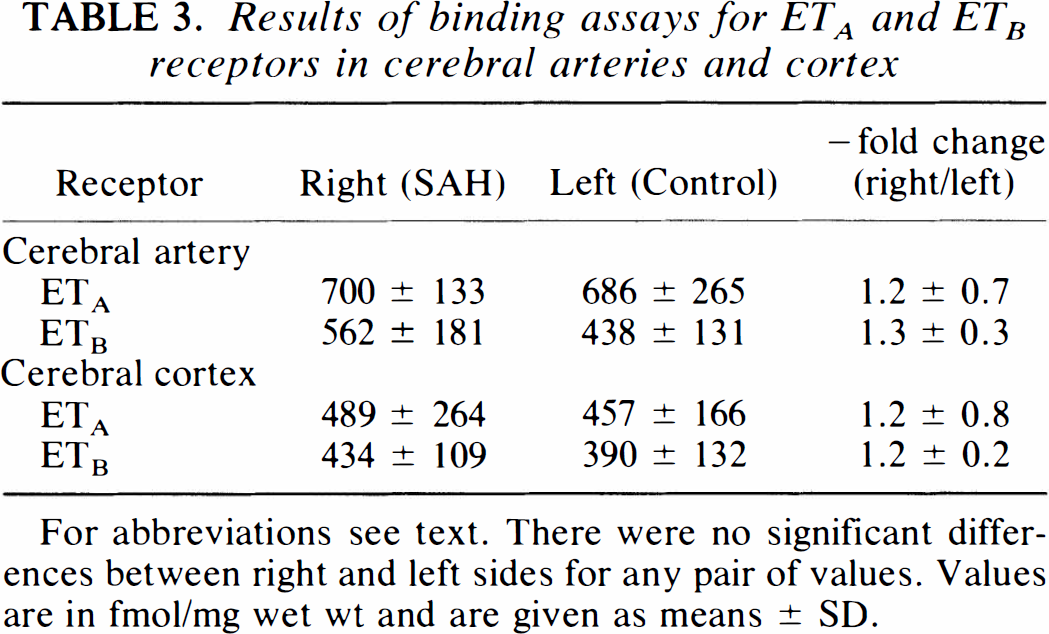
For abbreviations see text. There were no significant differences between right and left sides for any pair of values. Values are in fmol/mg wet wt and are given as means ± SD.
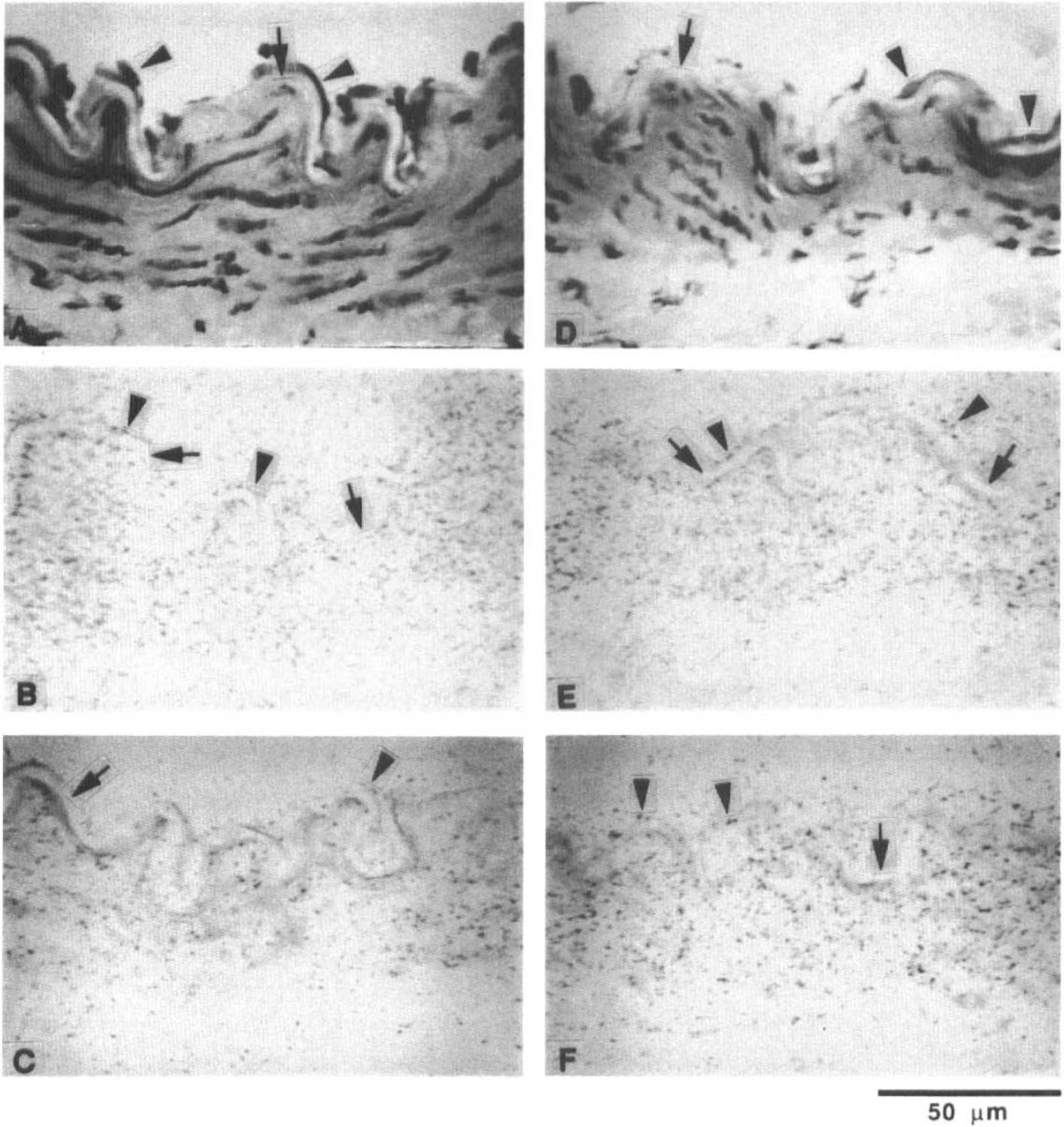
Microautoradiography of left (control side;
ET-1 peptide
There was not a significant elevation in ET-1 peptide in CSF on day 7 when compared with baseline (Table 2).
DISCUSSION
To determine whether ETs and ET receptors were involved in the pathogenesis of the cerebral vasospasm that follows SAH, competitive RT-PCR was used to quantitate ppET-1, ppET-3, and the ETA and ETB receptors in normal and vasospastic cerebral arteries and cortex from monkeys. Vasospastic cerebral arteries showed a significant increase in expression of ETB receptor mRNA that was associated with a trend toward increased ETB receptor protein. There was no change in ET-1 mRNA during vasospasm 7 days after SAH, and ppET-3 mRNA was not detected in cerebral arterial tissue. ET-1 peptide was also not elevated in CSF during vasospasm. In the underlying brain, however, the cortex exposed to SAH showed a significant increase in the levels of ppET-1 mRNA. The levels of ETA and ETB receptor mRNA were significantly increased, and this was associated with a trend of increased receptor proteins. The predominance of ppET-1 and ETA receptor mRNAs in cerebral arteries is consistent with previous studies of the distribution of mRNAs of ETs and ET receptors in primates (Sokolovsky, 1992), rats (Hori et al., 1992; Li et al., 1994), and dogs (Yamaura et al., 1992). We also detected ppET-2 in monkey cerebral arteries. The function of ET-2 is unclear and its contribution to vasospasm remains to be elucidated.
A premise of this study is that the left middle cerebral artery is normal and can serve as a control artery with which to compare the right side. Unilateral induction of SAH in this model does not produce diffuse intracranial blood or changes in intracranial pressure, and cerebral blood flow is normal on the left side (Handa et al., 1992). Studies show that the left middle cerebral artery and left hemisphere are normal angiographically, histologically, immunohistochemically, and in high-energy Phosphagens (Espinosa et al., 1984a, b ; Nosko et al., 1986; Steinke et al., 1989; Macdonald et al., 1991, 1992).
Role of ETs in vasospasm
Several investigators have found that ET-1 and in some cases ET-3 were elevated in CSF after SAH and that higher levels were found in association with vasospasm (Masaoka et al., 1989; Suzuki et al., 1990; Kraus et al., 1991; Seifert et al., 1995). CSF levels of ET-1 and ET-3 were also increased following head injury and epilepsy, conditions that are not associated with vasospasm (Kraus et al., 1991). Other investigators were unable to correlate increased CSF ET levels and vasospasm (Fujimori et al., 1990; Hamann et al., 1993; Gaetani et al., 1994). The ETs probably act by an autocrine or paracrine mechanism, and therefore CSF levels would not reflect levels at their site of action in the arterial wall (Hahn et al., 1990; Alberts et al., 1994; Cosentino and Katusic, 1994). Only one study addressed this issue by using an immunoassay to measure ET-1 in the basilar artery after SAH in dogs (Yamaura et al., 1992). ET-1 immunoreactivity was increased after 2 days but had returned to control levels by 7 days after SAH, although significant vasospasm persisted at 7 days.
Since the synthesis of ETs is transcriptionally regulated, changes in ETs should be related to ET mRNA abundance in the absence of changes in ET metabolism (Yanagisawa et al., 1988). The lack of a significant increase in ppET-1 mRNA and ET-1 peptide at 7 days after SAH argues against a role for ET-1 in the pathogenesis of vasospasm, a conclusion that is at odds with observations reviewed herein and with previous experiments testing the effects of ET antagonists against vasospasm following experimental SAH (Clozel and Watanabe, 1993; Cosentino and Katusic, 1994; Foley et al., 1994; Willette et al., 1994; Hino et al., 1995). In monkeys (from which tissues were obtained for this study), continuous intracisternal infusion of the ETA receptor antagonist BQ-123 prevented vasospasm following SAH (Hino et al., 1995). An antagonist of ETA and ETB receptors, bosentan, was not efficacious. The lack of effect of bosentan may have been due to pharmacokinetic differences or to deleterious effects of blocking ETB receptors (Fukuroda et al., 1994). Cosentino and Katusic (1994) reviewed studies of ET antagonists in vasospasm and concluded that while several studies of ETA receptor antagonists did support a role for ET-1 in the pathogenesis of vasospasm, the effects of these drugs on vasospasm were only modest and that further work was required.
ET receptors and explanation of present findings
Vasospasm was associated with a significant 3.4-fold increase in ETB receptor mRNA. The mRNA for the ETA receptor was 47 times more abundant than for the ETB receptor. Human and rat systemic arteries also contained a predominance of ETA receptor mRNA (Sokolovsky, 1992). Itoh et al. (1994) reported measurements of ETA and ETB receptor mRNA levels in basilar arteries of dogs after SAH. There was a qualitative increase in ETA receptor mRNA 3 days after SAH that had decreased by 7 days. They were unable to detect ETB receptor mRNA by Northern blotting, possibly due to the low sensitivity of this technique or differences in ETB receptor mRNA expression between dogs and monkeys. Experiments by Roux et al. (1995) in dogs support the present findings of a change from ETA to ETB receptor expression in vasospastic arteries after SAH.
The function of ET receptors in monkey cerebral arteries has not been determined. Previous studies show that ETA and ETB receptors may be found on endothelial and smooth muscle cells in various proportions depending on the species, vascular bed, and vessel type, and they may mediate contraction or relaxation (Davenport et al., 1993; MacLean et al., 1994; Moreland et al., 1994; Seo et al., 1994; Teerlink et al., 1994).
The increase in ETB receptor expression that we observed may be due to changes in the properties of smooth muscle cells in the arterial wall. Vasospasm is associated with variable degrees of intimal proliferation resulting from damage to medial smooth muscle with change in the contractile and biochemical synthetic properties of the cells and migration and proliferation of smooth muscle cells (Macdonald et al., 1992). Rat aorta smooth muscle cells expressed ETA receptors in early passages, but switched to a predominance of ETB receptors in late passage, a change that was associated with ET-induced mitogenesis and with smooth muscle cells that synthesized extracellular matrix materials and that were not contractile (Eguchi et al., 1994).
How can the changes in ET receptors and lack of changes in ET mRNAs be reconciled with the apparent efficacy of BQ-123 in the same model? This study examined the ET system at one time after SAH when vasospasm had been present for several days. Increases in ETs might occur earlier after SAH and initiate a contractile process that persisted even after the initiating increase in ET resolved. Other explanations are that BQ-123 has nonspecific effects that prevent vasospasm at the doses used or that it has other unidentified specific effects that prevent vasospasm.
Changes in ETs and ET receptors in cerebral cortex
There is little information about the types and locations of ETs and ET receptors in brain tissue. Both ppET-1 and ppET-3 were detected in cortex in this experiment, although ppET-3 was present at low levels in cerebral arteries. Therefore, unless microvasculature contains ET-3 in contrast to the large, conducting cerebral arteries examined in this study, it is likely that the ppET-3 mRNA detected in brain was in glia or neurons. This is consistent with prior studies showing that cultured rat astrocytes synthesized and secreted ET-3 (Ehrenreich et al., 1991). The function of ETs in brain is unknown, but Fernandez-Durango et al. (1994) detected ET-1-like immunoreactivity in human cerebral cortex and suggested that ET-1 might act as a neurotransmitter or neuromodulator. The decrease in ppET-3 mRNA in brain tissue that we observed is unexplained, and in view of the low levels of ppET-3 in cerebral arteries, it raises questions about the source of elevated ET-3 peptide in CSF after SAH (Kraus et al., 1991).
Both ETA and ETB receptors were detected in cerebral cortex. In rat brain, in situ hybridization studies showed that ETB receptors were widely distributed and seemed to be predominately expressed by glial cells (Hori et al., 1992). Much lower levels of ETA receptors were detected and were located predominately over the smooth muscle cells of the blood vessels, a finding consistent with the predominance of ETA receptors on these cells. Human brain cortex also contained predominately ETB receptors (Fernandez-Durango et al., 1994), and cultured rat astrocytes expressed receptors with equal affinity for ET-1, ET-2, and ET-3, a characteristic of ETB receptors (Ehrenreich et al., 1991). The function of the ETB receptor in brain is unclear, but it was speculated to be involved in modulation of neuronal function (Hori et al., 1992). Yamashita et al. (1994) examined ETs and ET receptors following transient forebrain ischemia in rats. Ischemia caused a marked increase in ETB receptor binding sites in microglia and in ET-1 and ET-3-like immunoreactivities within astrocytes. Therefore, the changes that were observed in cerebral cortex after SAH might be secondary to ischemia. Several other diverse pathological conditions have been associated with up-regulation of ETB receptors that may be a nonspecific response to stress or a manifestation of tissue repair (Elshourbagy et al., 1993; Roubert et al., 1993; Li et al., 1994; Takeda et al., 1994).
CONCLUSIONS
These results add to a body of literature describing changes in ETs and ET receptors in association with diseases that have major effects on blood vessels. The lack of significant increase in ppET-1 mRNA and ET-1 peptide during vasospasm in monkeys and the significant changes in ET receptor mRNA indicate that SAH and vasospasm are associated with a complex series of changes in the ET system that may be involved in the pathogenesis of these disorders.
Footnotes
Acknowledgment:
This work was supported by grants to Drs. Weir, Macdonald, and Bell from the National Institutes of Health (NS25946-07 and DK-20595), the American Association of Neurological Surgeons, and the Howard Hughes Medical Institute. We thank Drs. M. Yanagisawa and H. Kasuya for their helpful comments and advice.
