Abstract
Postischemic delayed neuronal death (DND) in CA1 of the gerbil hippocampus is thought to be caused by an abnormal increase of Ca2+ influx into the cell, mediated by excessive activation of glutamate receptors. One subtype of glutamate receptors, the AMPA receptor, is not permeable to calcium ions as long as an edited form of its GluR2 subunit is present. It is possible that global ischemia interferes with the posttranscriptional editing of the GluR2 mRNA and thus leads to calcium influx via the AMPA receptor. In order to test this hypothesis, we examined the extent of GluR2 RNA editing in CA1 and CA3 microdissected from gerbil hippocampus after 5 min of global ischemia and various recirculation intervals. At each interval tested, quality and quantity of mRNA editing in the vulnerable CA1 region were the same as in CA3. Furthermore, postischemic mRNA editing in both hippocampal regions was indistinguishable from editing in untreated control animals. Our results clearly demonstrate that global ischemia does not cause impairment of GluR2 RNA editing, which is thus not responsible for the abnormal calcium permeability of the postischemic cell membrane.
Transient global ischemia of 5 min duration leads to selective delayed neuronal death (DND) of CA1 pyramidal cells in the gerbil hippocampus (Kirino, 1982). The molecular basis of the unique vulnerability of CA1 neurons and their delayed degeneration after brief periods of ischemia is not yet understood. According to current concepts (Szatkowski and Attwell, 1994; Choi, 1995), delayed neuronal death is caused by an excessive Ca2+ influx produced by glutamate acting on NMDA and AMPA receptors (for a review, see Schoepfer et al., 1994). Whereas the NMDA receptor is known to play a crucial role in excitotoxic neuronal death (Gill et al., 1987; Szatkowski and Attwell, 1994; Choi, 1995), the contribution of the AMPA receptor to this process is less clear (Rothman and Olney, 1995). Evidence that the AMPA receptor must be involved in DND comes from the observation that blockers of AMPA receptors lead to reduction of ischemia-induced neuronal death (Sheardown et al., 1990; Nellgard and Wieloch, 1992). However, the exact way in which AMPA receptors contribute to postischemic DND is unclear. It is possible that depolarization induced by “normal,” i.e., Ca2+-impermeable AMPA receptors promotes Ca2+ influx through NMDA receptor channels via removal of the Mg2+ block of the channel (Nowak et al., 1984; Schoepfer et al., 1994). As a second possibility increased Ca2+ influx could occur directly through “atypical,” i.e., Ca2+-permeable AMPA receptors. This is consistent with the observation that neuronal subpopulations expressing such Ca2+-permeable AMPA receptors exhibit increased vulnerability to kainate toxicity (Brorson et al., 1994). AMPA receptors are impermeable to Ca2+ as long as they contain at least one subunit of GluR2 in its edited form (Hollmann et al., 1991; Verdoorn et al., 1991). If GluR2, also termed GluR-B (Jonas et al, 1994), is absent or present in its unedited form, the receptor is permeable to calcium ions (Verdoorn et al., 1991; Jonas et al., 1994). The observation that the GluR2 subunit governs divalent permeability of the AMPA receptor led to the hypothesis that global ischemia might caused DND by down-regulation of GluR2 transcription in CA1 (Pellegrini Giampietro et al., 1992). Indeed, postischemic reduction of GluR2 mRNA concentration in CA1 was observed (Pellegrini Giampietro et al., 1992, 1994). However, even 24 h after ischemia there were still about 30% of the GluR2 message left in CA1 (Pellegrini Giampietro et al., 1992, 1994), and it is not known whether this drop in mRNA concentration causes an equivalent decrease in the amount of GluR2 subunit within the membrane-bound AMPA receptors.
In the present study we examined the possibility that global cerebral ischemia might lead to the formation of Ca2+-permeable AMPA receptors by modulating GluR2 expression at the posttranscriptional level, via interference with the mRNA editing process.
MATERIALS AND METHODS
Animal experiments
The bilateral carotid occlusion model has been described in detail previously (Kiessling et al., 1993). Briefly, experiments were performed on adult male Mongolian gerbils (Meriones unguiculatus, 70–80 g body weight). Anesthesia was induced with a mixture of 30% O2, 68.5% N2O, and 1.5% halothane. Both common carotid arteries were exposed and double-clamped with atraumatic Biemer aneurysm clips. After 5 min of ischemia, clips were removed and restoration of blood flow was visually verified.
Nucleic acid isolation and RT-PCR
Animals were decapitated at postischemic recirculation intervals of 1, 6, 12, and 24 h and 4 days (n = 2 per time point). Additionally, two untreated gerbils served as controls. The CA1 and CA3 region of both hippocampi were microdissected, and mRNA was isolated following the instructions provided by the manufacturer of the mRNA isolation kit (Invitrogen, NV Leek, Netherlands). Oligo(T)-primed first-strand cDNA was produced with the Ready-to-go cDNA synthesis system, supplied by Pharmacia, Freiburg, Germany. PCR amplification of the first-strand cDNA was carried out according to standard protocols (Taylor, 1991) with the following set of conditions: 56°C, 15 s; 70°C, 15 s; 95°C, 10 s for 30 cycles followed by heating 15 s at 56°C and 7 min at 70°C. The forward primer (ACA CAA AGT AGT GAA TCA ACT) binds to all sequences that code for AMPA receptor subunits. The reverse primer (CTT GGC GAA ATR TCR CAN CC) specifically amplifies GluR2 coding DNA. In combination, these primers generate an 98-bp PCR product, either with rodent cDNA or rodent genomic DNA as template. For further analysis, the PCR products were purified from 8% Polyacrylamide gels as described (Efcavitch, 1990) and verified by partial DNA sequencing, using the CircumVent thermal cycle sequencing kit from New England Biolabs (Schwelbach, Germany).
Analysis of RNA editing
RNA editing at the Q/R site within the transmembrane region of GluR2 (Schoepfer et al., 1994) was analyzed by primer extension (Wisden et al., 1991). In brief, purified PCR product (see above) was denatured and hybridized with a primer (GGC GAA ATA TCG CAT CCY TGC) that binds to the template just in front of the editing site (Fig. 1). Together with Vent(exo˜)-DNA-polymerase (New England Biolabs), this mixture was incubated in parallel either with [α-32P]dCTP or [α-32P]dTTP as sole nucleotide. Incubation conditions were 56°C, 30 s; 72°C, 30 s; 95°C, 10 s for 20 cycles followed by denaturation for 30 s at 95°C. To verify that primer extension was directed by the template and not by self-priming of the oligonucleotide, negative control experiments were performed with nonspecific DNA or water as template. The extension products were analyzed by electrophoresis through 20% Polyacrylamide (Efcavitch, 1990) and exposure to Kodak XAR-5 film.
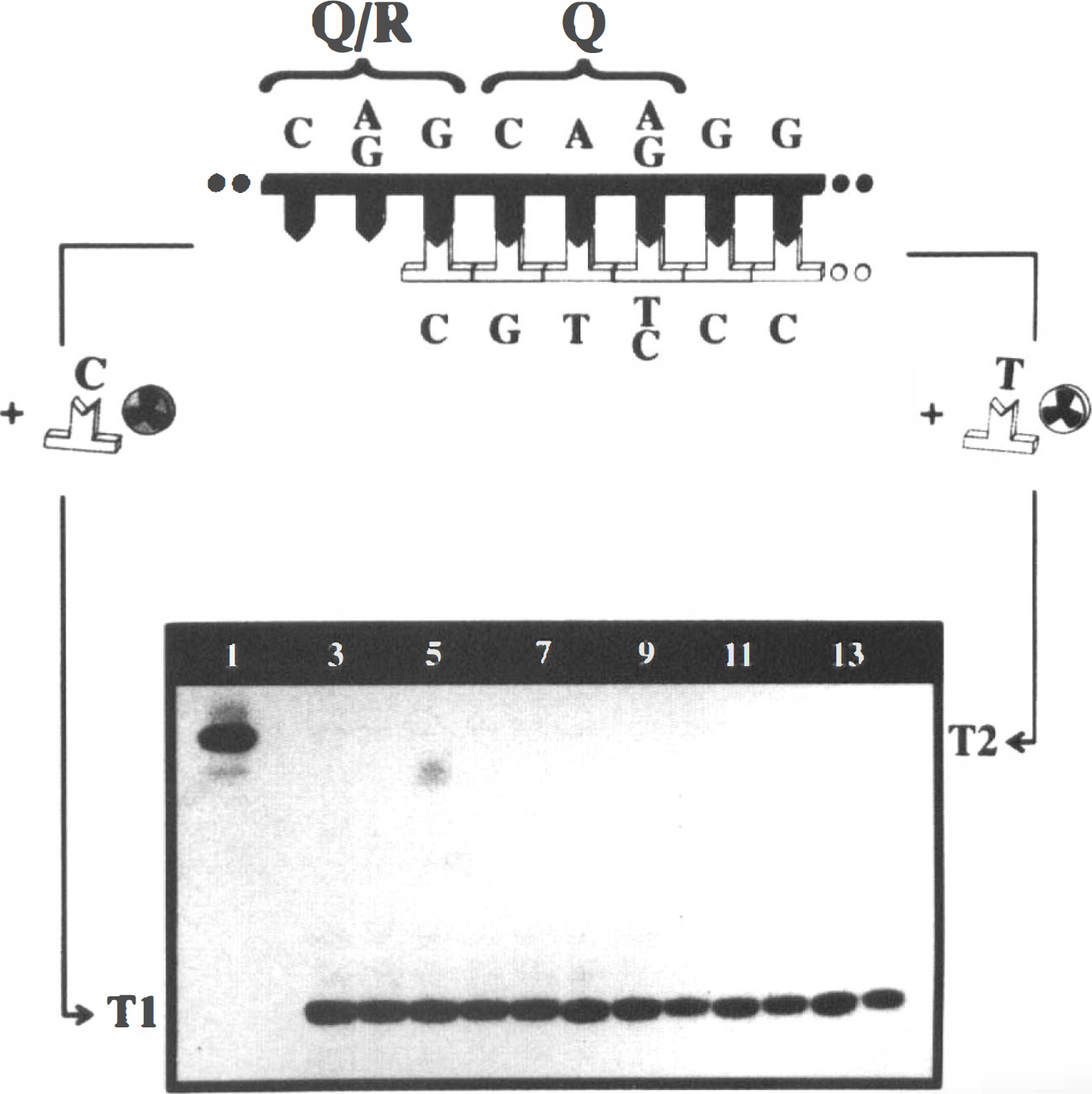
Detection of RNA editing at the Q/R site of GluR2 by primer extension.
RESULTS
Editing at the Q/R site of GluR2 was detected by a sensitive primer extension assay (Fig. 1) and confirmed by DNA sequencing (not shown). The failure of all RNA-derived templates to trigger detectable primer elongation with [α-32P]dTTP as sole nucleotide (Fig. 1, lanes 3–14) demonstrates that RNA editing at the Q/R site is complete in CA1 as well as in CA3 and not affected by global ischemia in both hippocampal regions. Evidence that the extension assay with [α-32P]dTTP functioned comes from the observation that amplified genomic DNA leads to 100% incorporation of [α-32P]dTTP (Fig. 1, lane 1), whereas primer labeling with [α-32P]dCTP was not detectable.
The detection limit of our primer extension assay was estimated by mixing edited and unedited template (i.e., template with cDNA or genomic DNA sequence, respectively) in different ratios. Subsequent analysis of these template mixtures was performed by primer extension under the conditions mentioned above. As demonstrated in Fig. 2, even 0.008 pmol of unedited template can be clearly detected in 0.5 pmol of the edited sequence, which corresponds to a ratio of 1.5%. Thus, if global ischemia had lowered the efficiency of GluR2 editing to a level of less than 98.5%, we would have detected it.
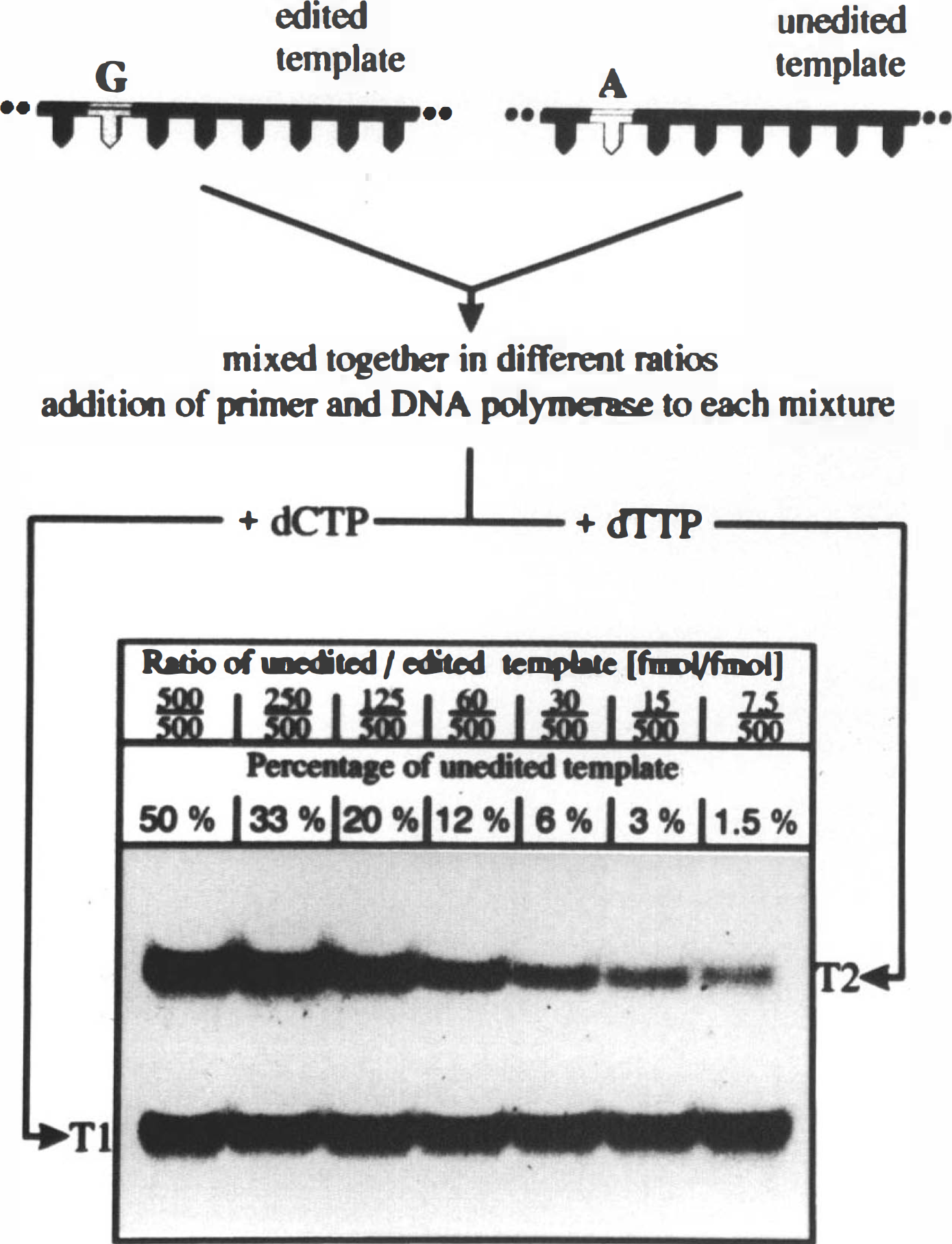
Sensitivity of the primer extension approach.
DISCUSSION
DND is widely attributed to excitotoxic activation of glutamate receptors. Initially, only the NMDA subtype of such receptors was regarded to be of significance for DND (Rothman and Olney, 1987; Choi, 1988), whereas the AMPA receptor was thought, at most, to play an indirect role. However, evidence has accumulated over the last years indicating that the AMPA receptor may also be an important mediator of excitotoxicity (Choi, 1992; Rothman and Olney, 1995) and the question has been raised as to whether the AMPA receptor might be modified directly by ischemia (Nellgard and Wieloch, 1992). The most interesting target of such a putative modification is the Q/R site of the GluR2 subunit since this site has been shown to govern the Ca2+ permeability of the AMPA receptor (Hollmann et al., 1991; Verdoorn et al, 1991).
In the present study we clarified whether ischemia might lead to a modification of AMPA receptors via interference with the editing of GluR2 mRNA. Our results clearly demonstrate that such an interference does not occur. The GluR2 mRNA is completely edited at the Q/R site in CA1 as well as in CA3, independent of ischemia and from the length of the recirculation interval.
Interestingly, unaffected editing at the Q/R site of GluR2 has also been reported for another pathological condition, the hippocampal kindling model of epilepsy (Kamphuis and Lopes da Silva, 1995). However, our results and those obtained by Kamphuis and Lopes da Silva do not affect the general idea that pathological conditions, such as ischemia, might lead to AMPA receptor modification. Nevertheless, our results indicate that such a putative modification occurs at another site of the AMPA receptor or at another level of gene expression, presumably the posttranslational rather than the transcriptional level. Further studies are necessary to clarify the influence of ischemia on the AMPA responsive subtype of glutamate receptors.
Footnotes
Abbreviations used
Acknowledgment:
The authors thank Drs. H. Monyer, K. H. Grube, and N. Maas-Szabowski for kindly providing the oligonucleotide probes used in this study.
