Abstract
A functional interrelation between nitric oxide (NO), the endothelial-derived vasodilating factor, and endothelin 1 (ET-1), the potent vasoconstrictive peptide, was investigated in microvascular endothelium of human brain. Nor-1 dose-dependently decreased the ET-1–stimulated mobilization of Ca2+. This response was mimicked with cGMP and abrogated by inhibitors of guanylyl cyclase or cGMP-dependent protein kinase G. These findings indicate that NO and ET-1 interactions involved in modulation of intracellular Ca2+ are mediated by cGMP/protein kinase G. In addition, Nor-1–mediated effects were associated with rearrangements of cytoskeleton F-actin filaments. The results suggest mechanisms by which NO–ET-1 interactions may contribute to regulation of microvascular function.
Endothelin 1 (ET-1), a 21-amino acid peptide, and nitric oxide (NO), a simple gas are produced by vascular endothelium among other cells (Bacic et al., 1992; Durieu-Trautmann et al., 1993; Masaki, 1994; Yanagisawa et al., 1988). Each of these agents exerts a variety of biological effects affecting many physiologic functions (MacCumber et al., 1990; Masaki, 1994; Moncada et al., 1991; Walsh et al., 1995). Endothelin 1 and NO, originally established as potent vasoactive substances, may influence regional and systemic vascular tone as well as blood flow through their antagonistic properties (vasoconstrictive and vasodilatory, respectively) (Bakker et al., 1997; Gellai et al., 1997; MacCumber et al., 1990; Masaki, 1994; Moncada et al., 1991; Thorin et al., 1998). Hypothetically, these endothelium-mediated processes require a potentially intimate interaction between these two substances at the level of cellular secretion and/or receptors, leading to alteration of contractile elements of the cellular cytoskeleton. Previously, ET-1 was shown to induce NO secretion that in turn reduces the production of ET-1 in endothelium (Boulanger and Luscher, 1990; Saijonmaa et al., 1990). Other studies demonstrate that the poorly dissociable binding of ET-1 to its receptor could be affected by a single exposure to NO (Goligorsky et al., 1994; Spatz et al., 1997b). These findings have contributed to the understanding of mechanisms entailed in the regulation of blood flow. However, this response is insufficient to explain a possible rapid interactive event between ET-1 and NO that could be instrumental in maintaining vascular and microvascular tone.
Numerous investigations suggest that alterations of intracellular Ca2+ are involved in vascular responses to various neuronal and humoral factors (Cornwell and Lincoln, 1989; Rasmussen, 1983; Walsh et al., 1995). The mechanisms responsible for the activity of these agents have been studied mainly in smooth muscle derived from peripheral vessels. It also has been shown that the ET-1–stimulated intracellular Ca2+ in platelets, polymorphonuclear leukocytes, smooth muscle, and Chinese hamster ovary cells (CHO-ET) transfected with ET
MATERIALS AND METHODS
Chemicals and reagents
Fluo-3/AM and Texas Red-X phalloidin were obtained from Molecular Probes (Eugene, OR, U.S.A.). Endothelin-1 was from Peninsula, Laboratories, Inc., Belmont, CA. NOR-1 {(±)-(E)-methyl-2-[(E)-hydroxyimino]-5-nitro-6-methoxy-3-hexeneamide}, ODQ (1H-[1,2,4]oxadiazolo[4,3,-a]quinoxalin-1-one), and Rp-8-pCPT-cGMP [Guanosine 3‘,5‘-cyclic mono-phosphothioate, 8-(4-chlorophenylthio)-, Rp-isomer, sodium salt] were purchased from Alexis Biochemicals Corp. (San Diego, CA, U.S.A.). U-73122, BQ123, and BQ788 were obtained from Calbiochem (La Jolla, CA U.S.A.). Ro 61-1790 [5-methyl-pyridine-2-sulfonic acid 6-(2-hydroxy-ethoxy)-5-(2-methoxy-phenoxy)-2-(2-1H-tetrazol-5-yl-pyridin-4-yl)-pyrimidin-4-ylamide] was a gift from Drs. S. Roux and M. Clozel (F. Hoffmann-La Roche, Ltd., (Basel, Switzerland). All other chemicals were bought from Sigma Chemical Company (St. Louis, MO, U.S.A.).
Cell culture
The technique used for preparing HBMEC was previously described (Spatz et al., 1997a). Briefly, the cultured microvascular endothelium was dissociated from microvessels of human brain surgically removed for the treatment of idiopathic epilepsy. For this study, HBMEC (from six cell lines [each derived from a different brain], passage 7 to 15) were grown to confluence on 1% gelatin-coated 24-well plates at 37°C in humidified atmosphere of 5% CO2/air for 48 hours. The purity of the endothelial cell cultures was about 98%, as determined by positive immunostaining for von Willebrand (factor VIII)-related antigen and incorporation of acetylated low-density lipoprotein, as well as negative staining for glial cell (GFAP, galactocerebroside, and ED-2), muscle cell, and pericyte (α-actin and tropomyosin, respectively) markers.
Measurements of intracellular calcium
The HBMEC were grown for 48 hours on 24-well plates and washed with buffer composed of NaCl (137 mmol/L), KCl (5 mmol/L), MgCl2 (1 mmol/L), sorbitol (25 mmol/L), HEPES (10 mmol/L), and CaCl2 (3 mmol/L), pH 7.0. Cells were incubated in the same buffer containing Fluo-3/AM (2.5 µmol/L) for 90 minutes at 37°C. The background fluorescence was measured immediately after dye loading with CytoFluor II fluorescence multi-well plate reader (PerSeptive Biosystems, Framingham, MA, U.S.A.) using a fluorescein filter pair (excitation, 485 ± 20 nm; emission, 530 ± 20 nm). A correction for autofluorescence was made in the absence of dye at the same emission wavelength. All of the tested substances were added and incubated in the presence or absence of selective inhibitors for an indicated period of time at 37°C. The measurements of Ca2+ fluorescence were performed at 30 seconds (a time of observed maximal fluorescence intensity induced by ET-1) using the same parameters mentioned earlier. The Ca2+ concentration changes were depicted as fluorescence intensity using the formula F (experimental reading) – F0 (initial reading)/F0 × 100, which normalizes for differences in dye loading and cell numbers per well.
Fluorescence staining of F-actin filaments
All procedures were conducted at room temperature. Cells were washed three times with physiologic buffered solution (pH 7.4) between individual incubation steps. The HBMEC grown on glass coverslips were exposed to NOR-1 (50 µmol/L or 100 µmol/L) for 1 minute alone, or followed by ET-1 (20 nmol/L or 50 nmol/L) for 30 seconds or ET-1 (20 nmol/L or 50 nmol/L) alone. After the removal of media, cells were fixed in 3.7% formaldehyde in physiologic buffered solution for 10 minutes and treated with 0.1% TritonRX-100 in physiologic buffered solution for 5 minutes. To reduce nonspecific background staining, fixed cells were incubated with 1% bovine serum albumin for 20 minutes. For examination of F-actin, cells were exposed to 1 unit of Texas Red-X phalloidin for another 20 minutes. The F-actin in HBMEC was evaluated with a Zeiss Axioplan fluorescence microscope (Oberkochen, Germany).
Statistics
Statistical evaluation of the data was performed by Student's t test or one-factor ANOVA followed by Fisher's protective least squares difference test for multiple comparisons. P values < 0.05 were regarded as statistically significant.
RESULTS
Effect of endothelin 1 on intracellular Ca2+ mobilization
The ET-1 dose-dependently increased intracellular Ca2+ (Fig. 1A). The maximal effect was reached at 100 nmol/L ET-1; the estimated EC50 was 20 nmol/L ET-1. No variations in these parameters were observed among the various HBMEC passages (7 to 15) used for these experiments. The selective ETa-receptor antagonist (BQ 123 or Ro 61-1790), but not the selective ET
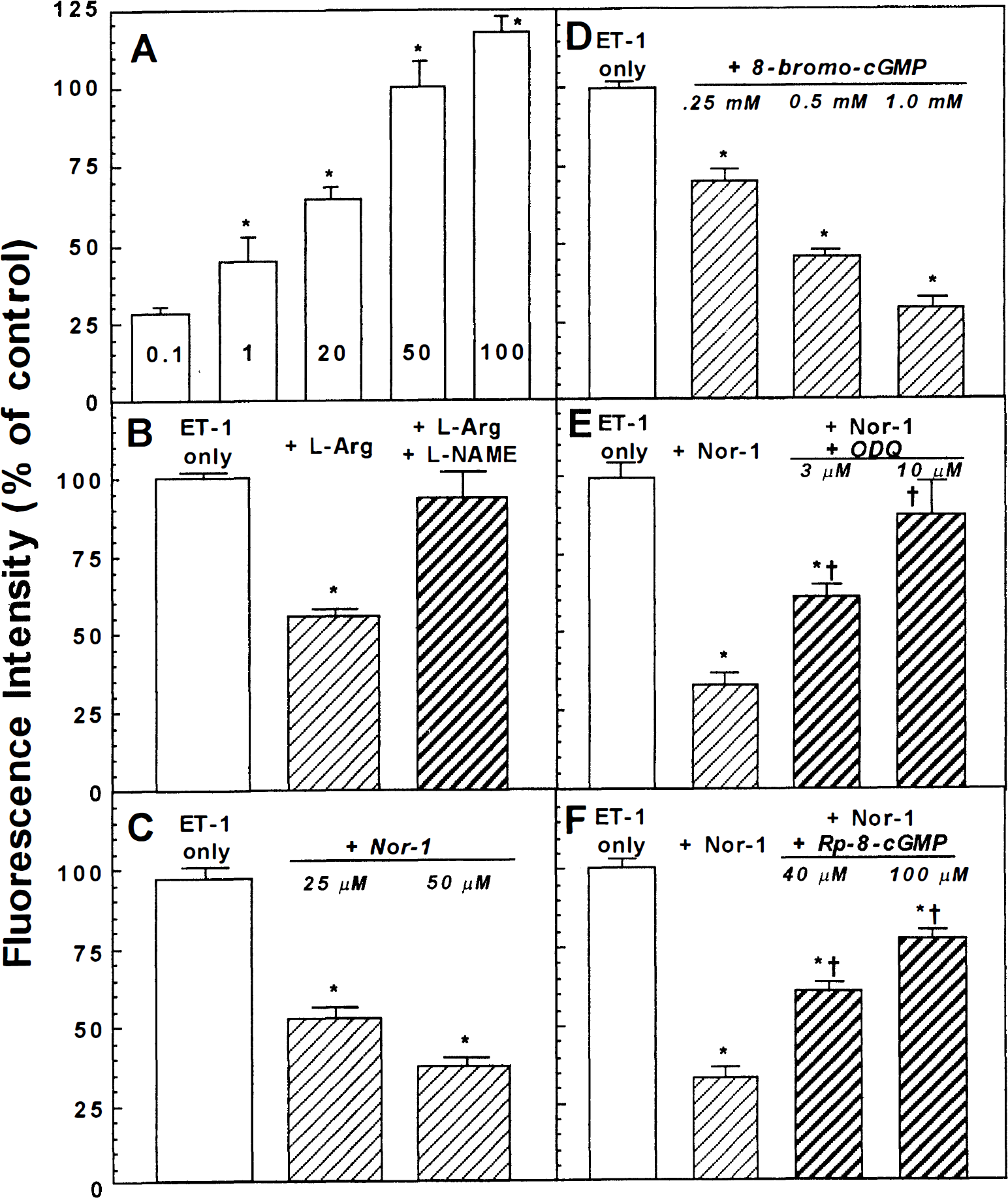
Characterization of intracellular Ca2+ response in endothelium derived from microvessels of human brain (HBMEC) to endothelin 1 (ET-1) alone or in the presence of nitric oxide donors. All HBMEC grown on 24-well plates and processed as indicated in the methods section were exposed to ET-1 alone for 30 seconds or to either NOR-1 for 60 seconds or 8-bromo-cGMP for 100 seconds before addition of ET-1 for 30 seconds. The HBMEC treated with
Effect of nitric oxide on ET-1–induced intracellular Ca2+
To assess potential interaction between endogenous or exogenous NO and ET-1 on Ca2+ mobilization, HBMEC were pretreated with
Effect of cGMP on endothelin 1-induced intracellular Ca2+
To examine the possible role of cGMP on NO mediation of the ET-1–stimulated mobilization of Ca2+, HBMEC were pretreated (100 seconds) with membrane permeable 8-bromo-cGMP. Treatment with 8-bromo-cGMP dose-dependently prevented ET-1–induced increases in intracellular Ca2+ (Fig. 1D). These data suggest that the observed effects of NO on ET-1–induced Ca2+ mobilization involve cGMP.
Role of cGMP/cGMP kinase in nitric oxide's prevention of endothelin 1–induced Ca2+ mobilization
Preincubation (20 minutes) of HBMEC with ODQ, a selective inhibitor of soluble guanylyl cyclase, resulted in the dose-dependent prevention of the inhibitory effect of NOR-1 on ET-1–stimulated Ca2+ mobilization (Fig. 1E). A similar response was observed by pretreatment (3 minutes) with Rp-8-pCPT-cGMP, a PKG inhibitor (Fig. 1F). Other protein kinase inhibitors (i.e., PKC inhibitors or PKA inhibitors) failed to reverse the NOR-1 effect on ET-1–induced Ca2+ mobilization (data not shown). These results indicate that NO or cGMP alteration of ET-1–stimulated Ca2+ mobilization in HBMEC are mediated through the soluble guanylyl cyclase-cGMP–dependent protein kinase system.
Effect of endothelin 1 and nitric oxide on F-actin filaments
The HBMEC contain delicate longitudinal F-actin filaments, which are most pronounced at the periphery (Fig. 2A). These filaments are rarified and interposed by occasional granules after exposure to 100 µmol/L NOR-1 (Fig. 2C). In contrast, treatment of HBMEC with 50 nmol/L of ET-1 increased the thickness and prominence of the longitudinal F-actin filaments (Fig. 2B). This ET-1–induced effect was altered by pretreatment with 100 µmol/L NOR-1, as characterized by the disconnected short, thin, and granular filaments (Fig. 2D). Similar responses of F-actin filaments were observed at 20 nmol/L of ET-1 and 50 µmol/L of NOR-1 (data not shown).
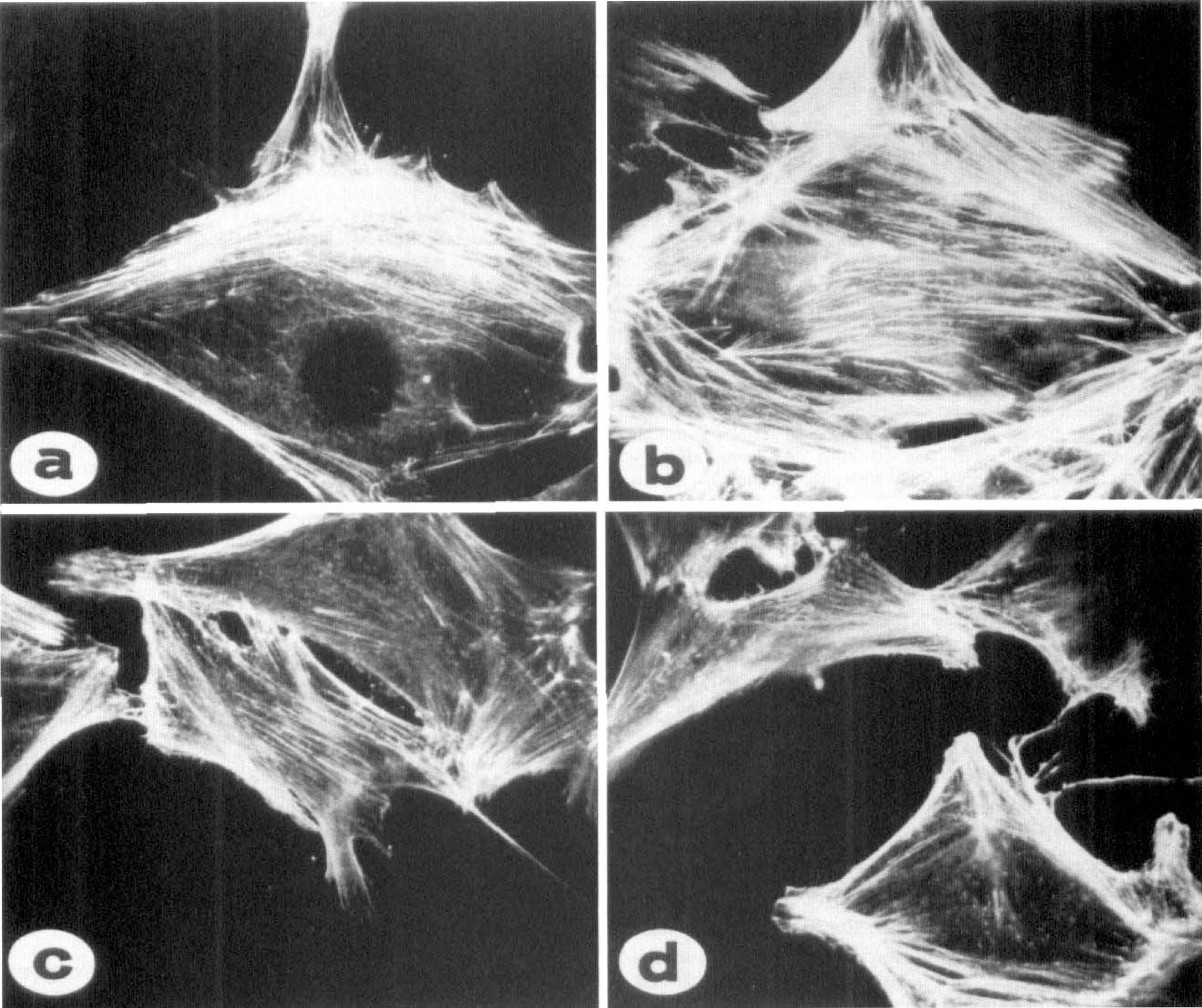
Response of F-actin cytoskeleton in HBMEC to ET-1 and NOR-1. The HBMEC grown on glass coverslips and processed as indicated in the methods were exposed to medium alone
DISCUSSION
The results of this study clearly indicate that ET-1, which induces Ca2+ mobilization in HBMEC through activation of ET
Based on these results alone, we cannot ascertain whether the NO-mediated abrogation of ET-1–induced increase in intracellular Ca2+ takes place on the level of ET
The findings regarding ET-1– and/or NO–induced changes in the pattern of cytoskeleton F-actin filaments and their association with alterations in Ca2+ mobilization are interesting. The involvement of cytoskeleton proteins in a variety of cellular functions (e.g., locomotion and architectural organization of the cells) has been the subject of numerous studies for the last decade (see review, Molitoris et al., 1997). The observed rapid phosphorylation of the cytoskeleton-associated protein implicates the responsible protein kinases in the regulation of cytoskeleton function. For example, vasodilator-stimulated phosphoprotein activity was shown to correlate with vasodilator-induced (e.g., prostaglandin E, prostacyclin I2, or sodium nitroprusside) inhibition of platelet activation mediated by cAMP- and cGMP-dependent protein kinase. This phosphoprotein colocalized with F-actin in platelets, fibroblasts, and smooth muscle cells (Reinhard et al., 1992). Co-localization of cGMP-dependent protein kinase with intermediate filament protein vimentin also was demonstrated in smooth muscle (MacMillan-Crow and Lincoln, 1994; Pryzwansky et al., 1995). These reports, together with the findings reported here, strongly support the involvement of protein kinase G in F-actin rearrangements induced by the interaction between ET-1 and NO (diagram, Fig. 3). The participation of other regulatory proteins, as well as yet unidentified factors, cannot be excluded. For example, rho, which is a member of small GTPases and the distant relatives of G proteins, was reported to affect actin-myosin filaments and contraction (see review, Hall, 1998). Also, contractile cytoskeletal stimulatory proteins (CSP; Fig. 3) represent a hypothetical possibility borne from observations concerning the relationship between vasodilators and stimulation of vasodilatory proteins, such as vasodilator-stimulated phosphoprotein. However, the precise mechanisms responsible for either ET-1 and/or NO effects on F-actin remain to be clarified. Nevertheless, this study demonstrates for the first time a close functional relationship between ET-1 and NO, which leads to rapid changes in intracellular Ca2+ level and alterations in the cellular cytoskeleton of HBEMC. The separate and joint effects of ET-1 and NO on Ca2+ mobilization and the cytoskeleton indicate that HBMEC possess the intrinsic machinery and capacity necessary to control and contribute to the regulation of cerebromicrovascular tone, blood-brain barrier function, and microcirculation in the brain. These findings invite additional speculation regarding the involvement of the cytoskeleton in in vivo physiologic and pathologic mechanisms. Such findings may prove useful in devising novel therapeutic strategies.
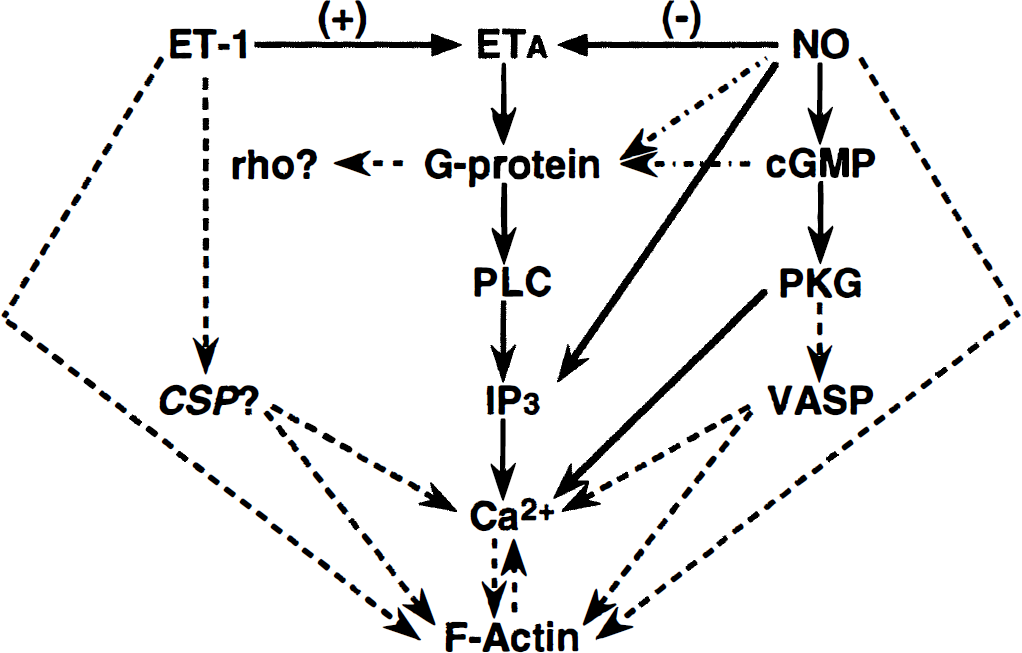
Schematic diagram of ET-1 and nitric oxide interaction: pathways based on published and personal observations (solid line); pathways based on preliminary findings (dashed, dotted line); hypothetical pathways (dashed line). VASP, vasodilator-stimulating protein; CSP, contractile stimulating protein; +, stimulatory effects; –, inhibitory effects.
