Abstract
The present study was designed to determine the effect of recombinant endothelial nitric oxide synthase (eNOS) gene expression on reactivity of canine basilar arteries to endothelin-1 (ET-1). Experiments were performed ex vivo. The arteries were exposed (30 minutes at 37°C) to adenoviral vectors encoding eNOS gene (AdCMVeNOS) or β-galactosidase reporter gene (AdCMVβ-Gal). Twenty-four hours after transduction, transgene expression was evident mainly in the vascular adventitia. Rings of control (nontransduced), AdCMVβ-Gal- and AdCMVeNOS-transduced arteries with and without endothelium were suspended for isometric tension recording. Levels of guanosine 3′,5′ -cyclic monophosphate (cGMP) were measured by radioimmunoassay. During contractions to uridine 5′-triphosphate, ET-1 (10−10 to 3×10−9 mol/L) caused further increase in tension in control and AdCMVβ-Gal-transduced arteries. In contrast, ET-1 caused concentration-dependent relaxations of AdCMVeNOS-transduced arteries. The relaxations to ET-1 in AdCMVeNOS-transduced arteries were endothelium-independent They were abolished by NG-nitro-L-argininemethyl ester or by chemical treatment of adventitia with paraformaldehyde before gene transfer. ET-1 (10−9 mol/L) significantly increased intracellular cGMP levels in AdCMVeNOS-transduced arteries without endothelium. In arteries transduced with AdCMVeNOS, higher concentrations (10−9 to 3×10−8 mol/L) of ET-2 also caused relaxations, whereas ET-3 and sarafotoxin, a selective ETB receptor agonist, did not produce any relaxations. The relaxations to ET-1 in AdCMVeNOS-transduced arteries were strongly reduced by BQ-123 (10−7 mol/L), an ETA receptor antagonist, but were not affected by BQ-788 (3×10−7 mol/L), an ETB receptor antagonist These results suggest that genetically modified adventitia can produce nitric oxide and cause relaxations in response to ET-1 via activation of ETA receptors. Our findings support a novel concept that successful transfer and expression of recombinant eNOS gene can lead to a qualitative change in responsiveness to vasoconstrictor substances.
Endothelin (ET)-1 is a potent vasoconstrictor peptide (Yanagisawa et al., 1988), and it is generally accepted that ET-1 may be an important substance involved in the pathogenesis of a number of cardiovascular disorders including systemic hypertension, pulmonary hypertension, coronary arterial spasm, and atherosclerosis (Ivy et al., 1997; Lerman et al., 1991; Nayler, 1990; Steward et al., 1991), In the cerebral circulation, ET-1 is regarded as a putative mediator of cerebral vasospasm, which is a major cause of morbidity and mortality in patients with subarachnoid hemorrhage (Asano et al., 1989; Cosentino and Katusic, 1994; Suzuki et al., 1992; Willett et al., 1994; Zuccarello et al., 1996).
Nitric oxide (NO), produced by endothelial nitric oxide synthase (eNOS), is a potent vasodilator and plays an essential role in the regulation of vascular tone (Bredt and Snyder, 1994; Moncada, Palmer and Higgs, 1991), A recent advance in recombinant DNA technology has made it possible to increase local NO production in the vascular wall by increasing expression of eNOS (Janssens et al., 1996; von der Leyen et al., 1995). Our previous in vitro and in vivo studies have confirmed that recombinant eNOS gene can be transferred using adenoviral vectors into adventitial layer overlying canine cerebral arteries, and that expression of eNOS gene in the adventitia can augment vasodilator effect of bradykinin and calcium ionophore A23187 (Chen et al., 1997a,b; Tsutsui et al., 1998). During preliminary studies, we noticed that expression of recombinant eNOS gene in canine basilar arteries could convert the contractile responses to ET-1 into relaxations. This observation suggested that successful transfer and expression of recombinant eNOS gene may lead to a qualitative change in responsiveness of cerebral arteries to vasoconstrictor substances. Therefore, the present study was designed to characterize the mechanism of ET-1–induced relaxations of isolated canine basilar arteries expressing recombinant eNOS gene.
MATERIALS AND METHODS
Construction, propagation, and purification of adenoviral vectors
The adenoviral vector encoding eNOS gene (AdCMVeNOS), driven by the cytomegalovirus immediate early promoter, was generated through homologous recombination (Spector and Samaniego, 1995). These vectors, based on serotype 5 wild adenovirus, have been made replication-deficient by a deletion of the early region 1 (E1), followed by an insertion of the cDNA sequence for bovine aortic endothelial cell eNOS (kindly provided by Dr. David G. Harrison, Emory University, Atlanta, GA). The generation, propagation, purification, and evaluation of the adenoviral vector containing eNOS gene were described in detail previously (Chen et al., 1997b). The recombinant adenoviral vector encoding β-galactosidase gene driven by cytomegalovirus promoter (AdCMVβ-Gal), used in all experiments as control, was a kind gift of Dr. James M. Wilson (University of Pennsylvania, Philadelphia, PA).
Gene transfer
All procedures were in accordance with the institutional guidelines of the Mayo Clinic. Experiments were performed ex vivo on rings (3-mm long) of basilar arteries taken from mongrel dogs (15 to 20 kg), anesthetized with 30 mg/kg (intravenous) sodium pentobarbital. To remove intraluminal blood, arterial rings were gently rinsed with cold modified Krebs-Ringer bicarbonate solution (control solution) consisting of NaCl 118.3, KCl 4.7, CaCl2 2.5, MgSO4 1.2, KH2PO4 1.2, NaHCO3 25.0, calcium-ethylenediamine-tetraacetic acid (EDTA) 0.026 and glucose 11.1 (mmol/L). In certain rings, the endothelium was removed mechanically. Endothelial removal was accomplished by gently sliding an arterial ring over a steel needle (19 to 22 gauge) abraded by sandpaper, using two pairs of fine forceps under a microscope. The rings were assigned randomly for gene transfer. Cerebral arteries do not have external elastic lamina (Lee, 1995; Rosenblum, 1994), and it is impossible to mechanically remove the adventitia. Therefore, to disrupt function of the adventitial layer it was chemically treated with paraformaldehyde (the artery placed on a steel needle was soaked into phosphate-buffered saline [PBS] solution containing 4% paraformaldehyde for 15 seconds). Arterial rings were then incubated with adenoviral vectors (1010 pfu/mL) in minimum essential medium (MEM; containing 0.1% bovine serum albumin, 100 U/mL penicillin and 100 μg/mL streptomycin) for 30 minutes at 37°C. This was followed by incubation in fresh MEM for 24 hours at 37°C in a CO2 incubator (5% CO2/95% air; Forma Scientific, Inc.).
Histochemical and immunohistochemical analyses of gene expression
For histochemical staining of β-galactosidase, the rings of basilar arteries were fixed for 30 minutes in 2% paraformaldehyde/0.2% glutaraldehyde in PBS. The rings were rinsed in PBS, and placed in 5-bromo-4-chloro-3-indolyl-β-D-galactopyranoside reagent for 2 hours, The stained vessels were frozen in O.C.T. compound (Miles, Inc.). Serial 5 μm cross sections were cut from each ring and counterstained with nuclear fast red. For immunohistochemical staining of recombinant eNOS, arterial rings were frozen in O.C.T. compound and serial 5 μm cross sections were cut. After immersion fixation in acetone (4°C), the sections were incubated in 0.1% sodium azide/0.3% hydrogen peroxide, and then incubated with 5% normal goat serum/PBS-Tween 20 to block the nonspecific protein binding sites. A monoclonal antibody against eNOS (5 μg/mL, 1:50 of stock; Transduction Laboratory) was applied for 60 minutes at room temperature, followed by incubations with biotinylated rabbit anti-mouse F(ab′)2 (1:200, 20 minutes) secondary antibody and peroxidase conjugated-streptavidin (1:300, 20 minutes) (Vector Laboratories, Inc.). After a 30-second immersion in 0.1-mol/L sodium acetate buffer (pH 5.2), eNOS immunoreactivity was visualized with 3-amino-9-ethylcarbazole and hematoxylin counterstaining.
Quantitative analysis of β-galactosidase protein
Expression of β-galactosidase protein in vessels was quantified by an enzyme-linked immunosorbent assay (ELISA). Twenty-four hours after gene transfer, arterial rings without endothelium were homogenized in a solution containing 0.1 mol/L K2HPO4, 0.2% Triton X-100, and 10−3 mol/L phenylmethylsulfonyl fluoride, pH 7.8 at 4°C. After a centrifugation at 12000xg for 10 minutes, β-galactosidase protein levels in supernatants were determined using β-galactosidase ELISA kit (5 Prime 3 Prime, Inc.). Total protein levels in the supernatants were measured by a DC Protein Assay Kit (Bio-Rad).
Analyses of vascular reactivity
Twenty-four hours after gene transfer, each ring was connected to an isometric force transducer (Grass FT03; Grass Instrument Co.) and suspended in an organ chamber filled with 25 mL modified Krebs-Ringer bicarbonate solution (control solution; pH 7.4, 37°C) aerated with 94% O2–6% CO2, Isometric tension was recorded continuously. The rings were allowed to stabilize at a resting tension of 0.2 to 0.4 g for 1 hour. Each ring was then gradually stretched to the optimal point of its length-tension curve (approximately 3.0 g) as determined by the contraction to 10−5 mol/L uridine 5′ -triphosphate (UTP) (Katusic et al., 1987). All experiments were performed in the presence of 10−5 mol/L indomethacin to exclude the possible influence of endogenous cyclo-oxygenase. To evaluate relaxation responses, the rings were contracted with UTP (3×10−6 to 10−5 mol/L) before the addition of agonists. Concentration-response curves were obtained in a cumulative fashion. Several rings prepared from the same artery were studied in parallel. The relaxations were expressed as a percentage of maximal relaxations induced by 3×10−4 mol/L papaverine added at the end of each experiment. In experiments with diethylamine NONOate (DEA-NONOate), the median effective concentration (EC50) was calculated for each ring by linear interpolation between the two concentrations evoking responses just above and below 50% of the maximum.
Measurement of cGMP
A radioimmunoassay technique was used to determine the tissue content of guanosine 3′,5′ -cyclic monophosphate (cGMP). Twenty-four hours after gene transfer, 10−5 mol/L indomethacin and 10−3 mol/L 3-isobutyl-1-methylxanthine were added to the incubation medium containing arterial rings without endothelium, to eliminate the influence of cyclo-oxygenase and to inhibit the degradation of cGMP by phosphodiesterases, respectively. After the incubation for 30 minutes, certain rings were stimulated with 10−9 mol/L ET-1 for 2 minutes. Then the rings were removed from the medium and immediately frozen in liquid nitrogen. Samples were homogenated in a cooled solution of 100% ethanol and 1 N HCl. Samples were then centrifuged (4000 rpm, at 4°C for 10 minutes) and supernatant poured into a clean tube and placed in a 55°C water bath under nitrogen to dry off the ethanol. After the tube was dry the sample was reconstituted with a Tris buffer. The assay was run using a kit from Amersham. Protein assay was conducted by a DC Protein Assay Kit (Bio-Rad). This assay is similar to the procedure described by Lowry.
Drugs
The following pharmacological agents were used: ET-1, ET-2, ET-3 (Phoenix Pharmaceuticals, Inc.), BQ-123, BQ-788 (Research Biochemicals Interational), UTP, sarafotoxin S6c, (L-NAME), indomethacin, papaverine hydrochloride (Sigma), and diethylamine NONOate (DEA-NONOate; Cayman Chemical Co.). Drugs were dissolved in distilled water so that volumes of <0.15 mL were added to the organ chambers. Concentrations of all drugs are expressed as final molar (mol/L) concentration in the control solution. For experiments with BQ-123, BQ-788, and L-NAME, these drugs were added 15 minutes before obtaining the concentration-response curve for each vasodilator agent.
Statistical analysis
The results are expressed as means± SD; n refers to the number of animals studied. Each set of concentration-response relationship was statistically analyzed by repeated measures analysis of variance (RM-ANOVA). Levels of β-galactosidase and cGMP, and EC50 values in response to DEA-NONOate in different groups, were compared using ANOVA with Bonferroni/Dunn's post-hoc test. Statistical significance was accepted at the level of P < .05.
RESULTS
Relaxations to ET-1
During contractions induced by UTP (10−5 M), ET-1 (10−10 to 3×10−9 mol/L) caused concentration-dependent relaxations of AdCMVeNOS-transduced basilar arteries (Fig. 1A). In contrast, ET-1 caused only additional contractions in control (nontransduced) and AdCMVβ-Gal–transduced arteries The relaxations to ET-1 were not affected by the removal of endothelial cells (Fig. 1B). The typical responses to ET-1 in AdCMVβ-Gal– and AdCMVeNOS-transduced arteries without endothelium are presented in Fig. 2. The relaxations to ET-1 in endothelium-denuded arteries after eNOS gene transfer were abolished in the presence of L-NAME (3×10−4 mol/L), an inhibitor of nitric oxide synthase (NOS) (Fig. 3).
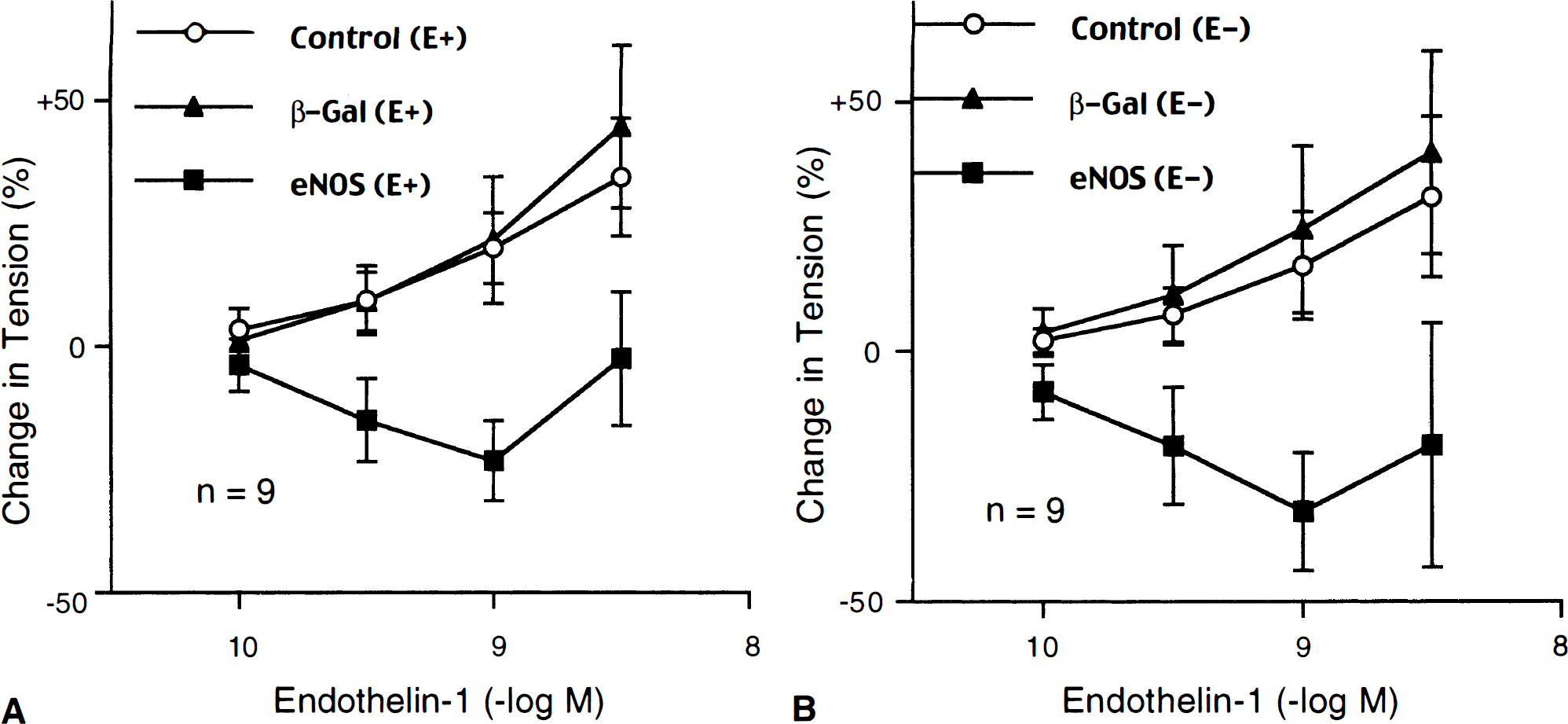
Responses to endothelin-1 in nontransduced (Control), AdCMVβ-Gal–transduced (β-Gal), and AdCMVeNOS-transduced (eNOS) basilar arteries with
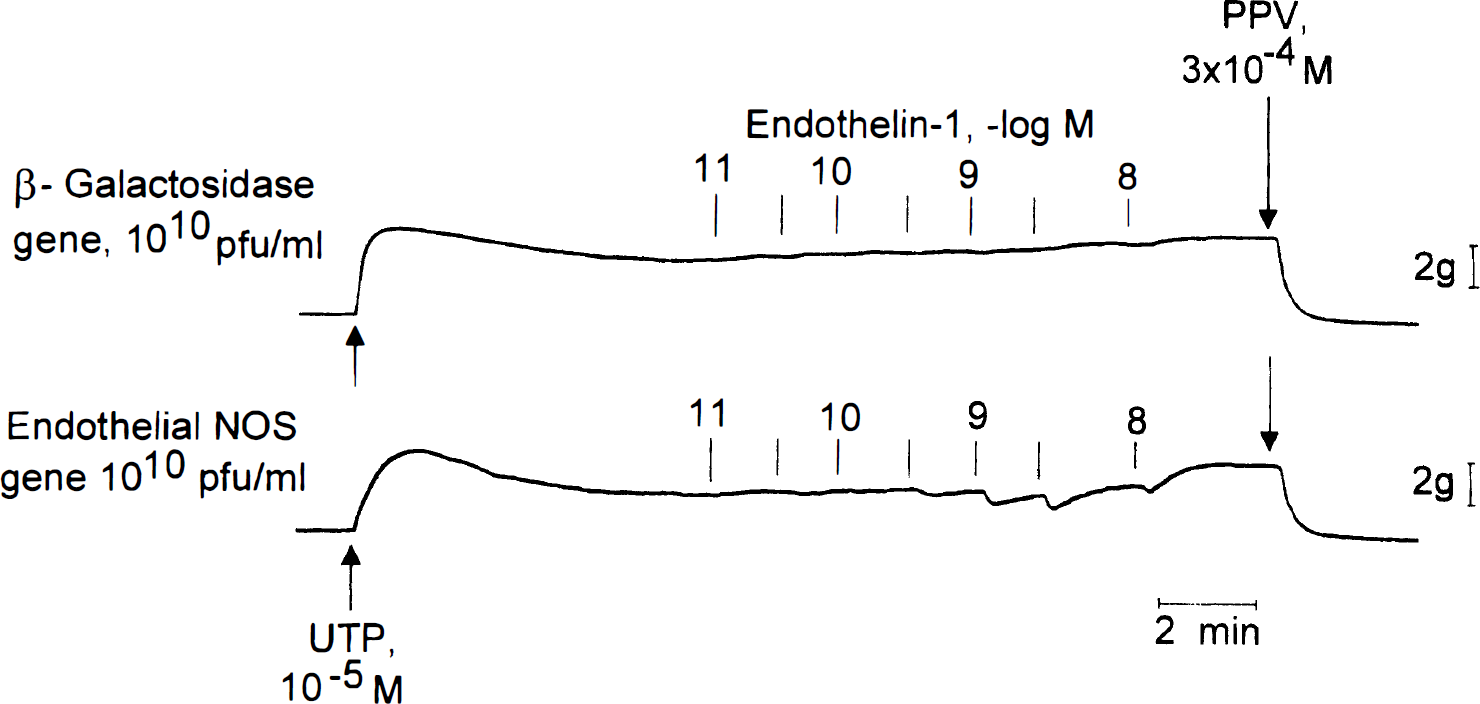
Typical recordings of responses to endothelin-1 in AdCMVβ-Gal-transduced and AdCMVeNOS-transduced basilar arteries without endothelium. Responses were obtained during contractions induced by uridine 5′ -triphosphate (UTP; 10−5 mol/L).
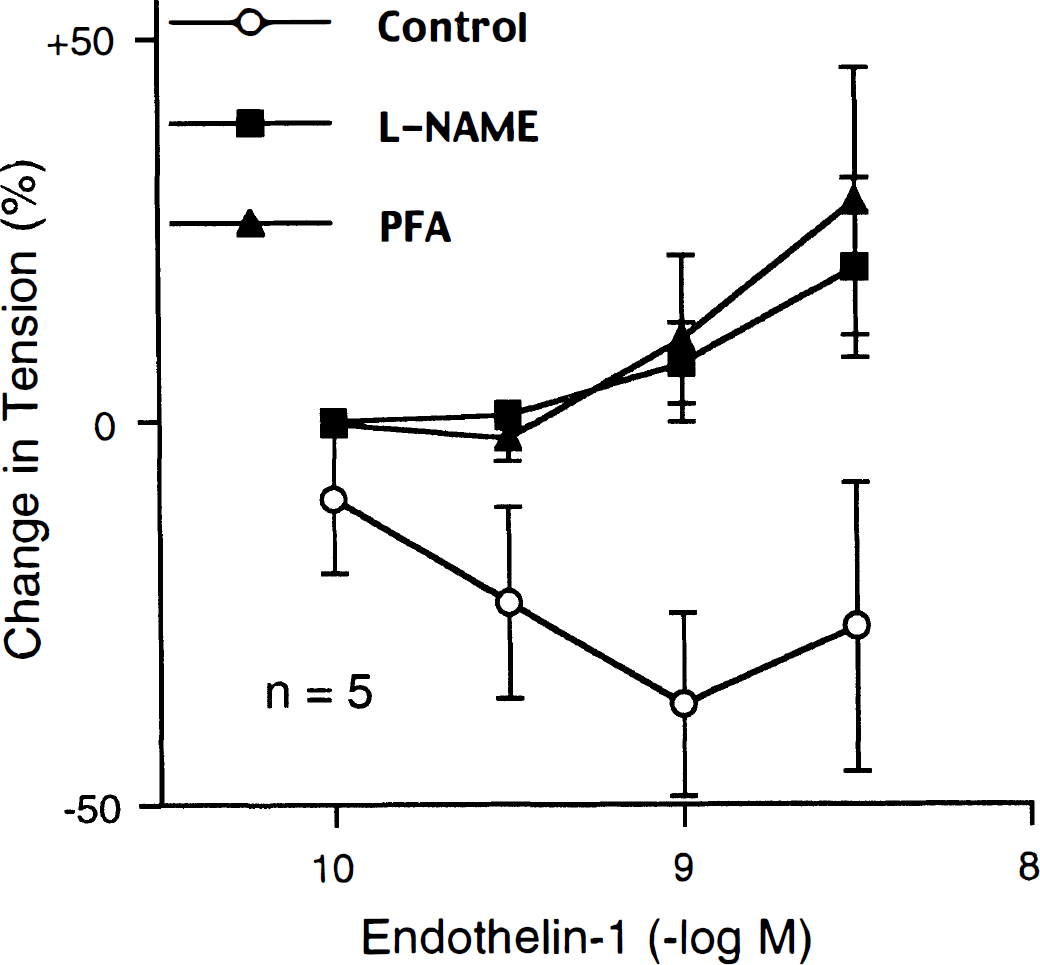
Effect of NG-nitro-L-arginine methyl ester (L-NAME; 3 × 10−4 mol/L) and chemical treatment of adventitia with paraformaldehyde (PFA) on relaxations to endothelin-1 in AdCMVeNOS-transduced (eNOS) basilar arteries without endothelium. Responses were evaluated during contractions induced by uridine 5′-triphosphate. Data are shown as mean ± SD and are expressed as percent of maximal relaxations induced by papaverine (3 × 1 0−4 mol/L); 100% = 4.0 ± 1.4 g (n = 5), 4. 5 ± 0.9 g (n = 5), and 3.5 ± 0.7 g (n = 5) for untreated arteries and arteries treated with L-NAME and PFA, respectively. Effects of endothelin-1 in L-NAME and PFA treated arteries were significantly different from control; P < .05 by repeated measure analysis of variance.
Effect of chemical treatment of adventitia on gene transfer and vascular reactivity
Twenty-four hours after exposure to AdCMVβ-Gal and AdCMVeNOS, expression of recombinant proteins was evident mainly in the adventitia (Fig. 4). Chemical treatment of arterial adventitia with paraformaldehyde before gene transfer, abolished expression of recombinant proteins detected by histochemical or immunohistochemical staining. Abolition of β-galactosidase protein expression in those chemically treated arteries was verified also by ELISA (Table 1). The same treatment abolished the relaxations to ET-1 in AdCMVeNOS-transduced arteries (Fig. 3). In contrast, paraformaldehyde did not reduce relaxations to DEA-NONOate, a donor of nitric oxide (Table 2).
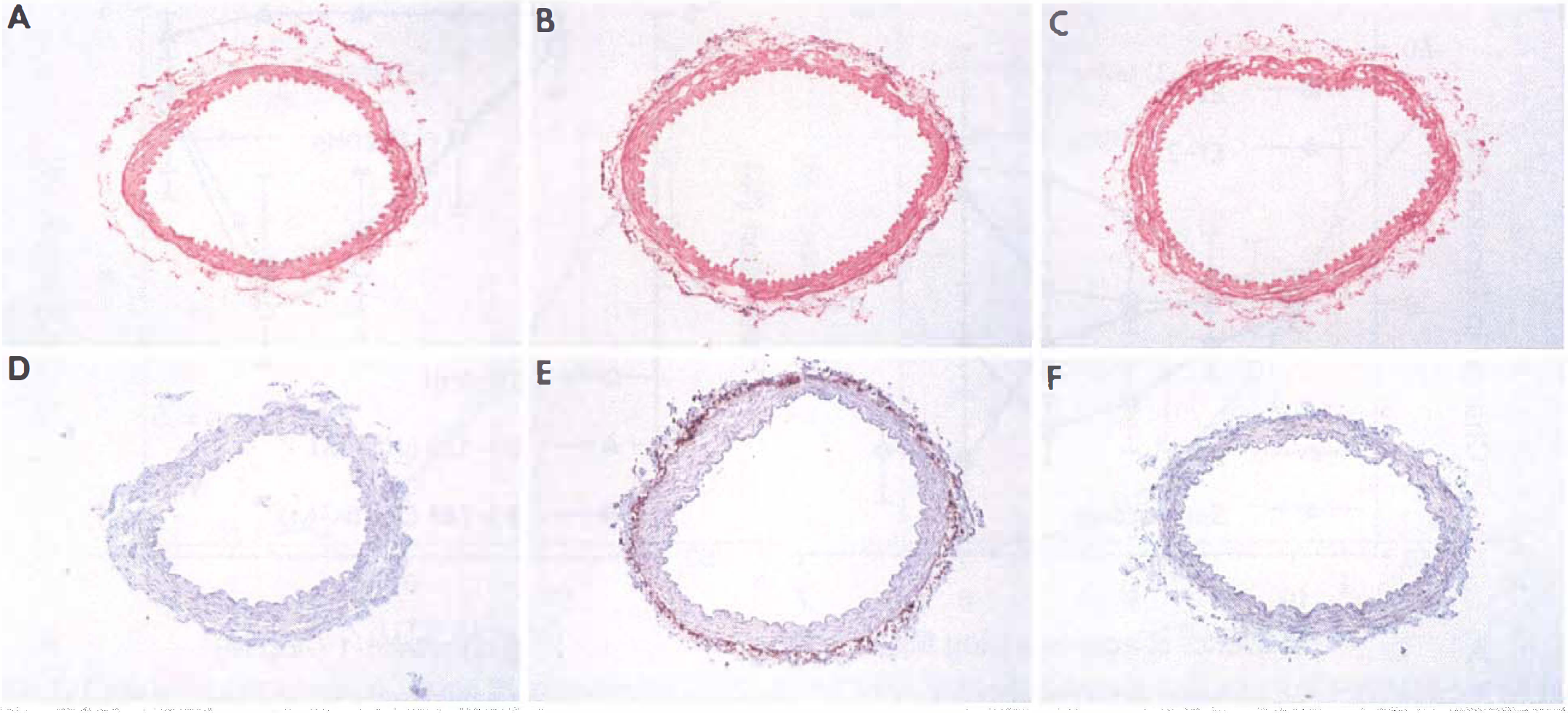
Histochemical staining of β-galactosidase
Effect of chemical treatment of adventitia on expression of recombinant β-galactosidase protein in basilar arteries without endothelium

Chemical treatment of adventitial layer with paraformaldehyde (PFA) was conducted before β-Gal gene transfer as described in Methods. Values are mean ± SD; n = number of dogs.
Statistically different from control and β-Gal (PFA-treated); P < 0.05 by ANOVA.
Effect of chemical treatment of adventitia on EC50 and maximal relaxation in response to DEA-NONOate in e-NOS gene-transduced basilar arteries without endothelium

The chemical treatment of adventitial layer with paraformaldehyde (PFA) was conducted before eNOS gene transfer as described in Methods. Relaxations were obtained during contractions to uridine 5′-triphosphate. Relaxations are expressed as percent of maximal relaxation induced by papaverine (3 × 10−4 mol/L). Values are mean ± SD. EC50 indicates median effective concentration; n = number of dogs.
Relaxations to ET-1, ET-2, ET-3, and sarafotoxin
Relaxations to ET-1, ET-2, ET-3, and sarafotoxin, a selective ETB receptor agonist, were compared in AdCMVeNOS-transduced basilar arteries without endothelium (Fig. 5A). ET-2 caused relaxations in AdCMVeNOS-transduced arteries, however, the concentrations required to induce detectable and maximal relaxations were higher than those of ET-1. In arteries contracted with UTP, ET-3, and sarafotoxin caused only further increase in vascular tone.
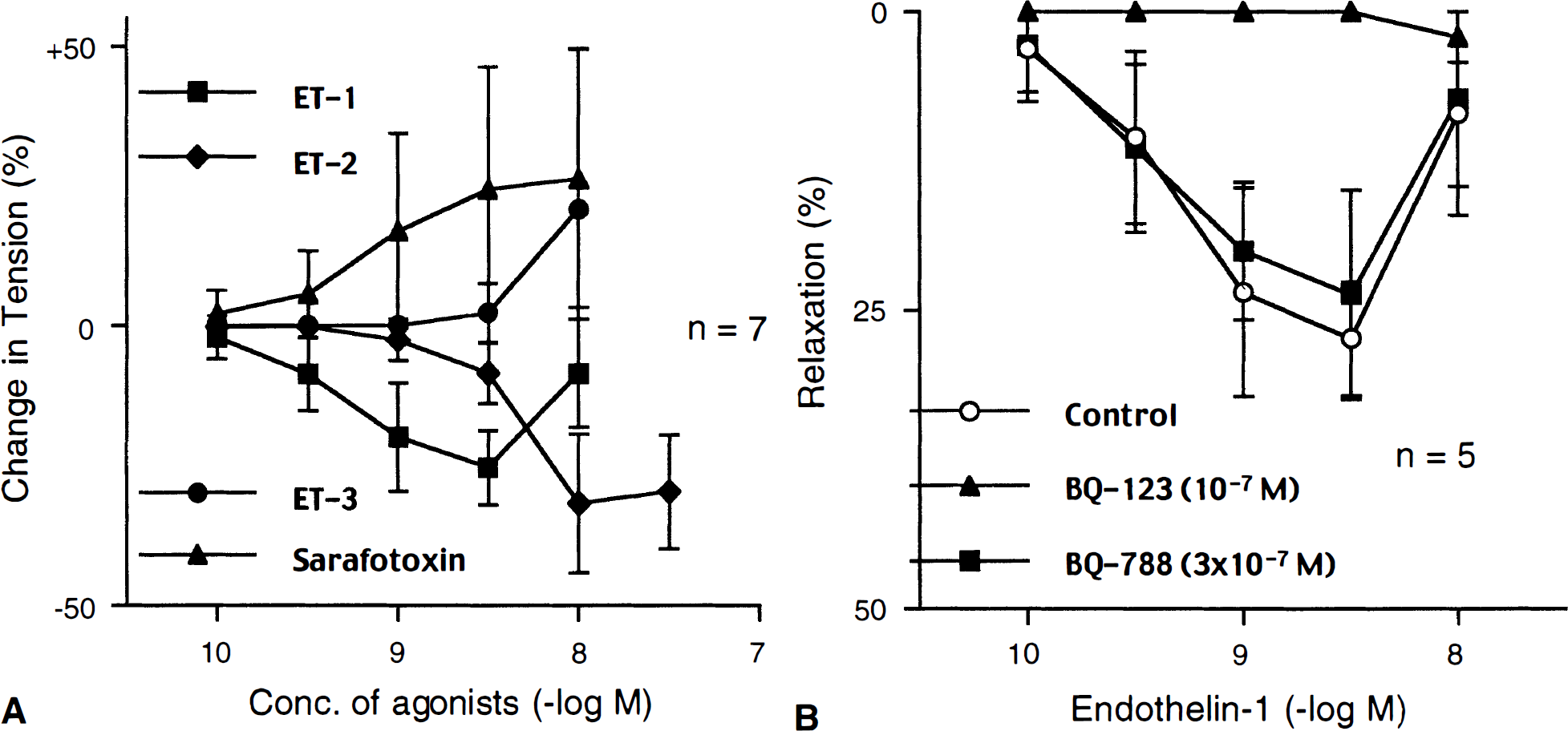
Responses to endothelin-1 (ET-1), endothelin-2 (ET-2), endothelin-3 (ET-3), and sarafotoxin
Effect of BQ-123 and BQ-788 on relaxations to ET-1
Relaxations to ET-1 in AdCMVeNOS-transduced basilar arteries without endothelium, were studied in the presence of BQ-123, a selective ETA receptor antagonist, or BQ-788, a selective ETB receptor antagonist (Fig. 5B). BQ-123 (10−7 mol/L) abolished the relaxations to ET-1. In contrast, BQ-788 in the same concentration (10−7 mol/L; data not shown) and even in a higher concentration (3×10−7 mol/L) did not affect the relaxations to ET-1 (Fig. 5B).
Contractions to ET-1
ET-1 (10−10 to 10−8 mol/L) caused concentration-dependent increase in base line tension in basilar arteries that were not contracted by UTP. These contractions were significantly smaller in AdCMVeNOS-transduced arteries compared to those in AdCMVβ-Gal–transduced arteries (Fig. 6). In contrast, contractile responses to UTP were not different between AdCMVβ-Gal–transduced and AdCMVeNOS-transduced arteries (Fig. 7).
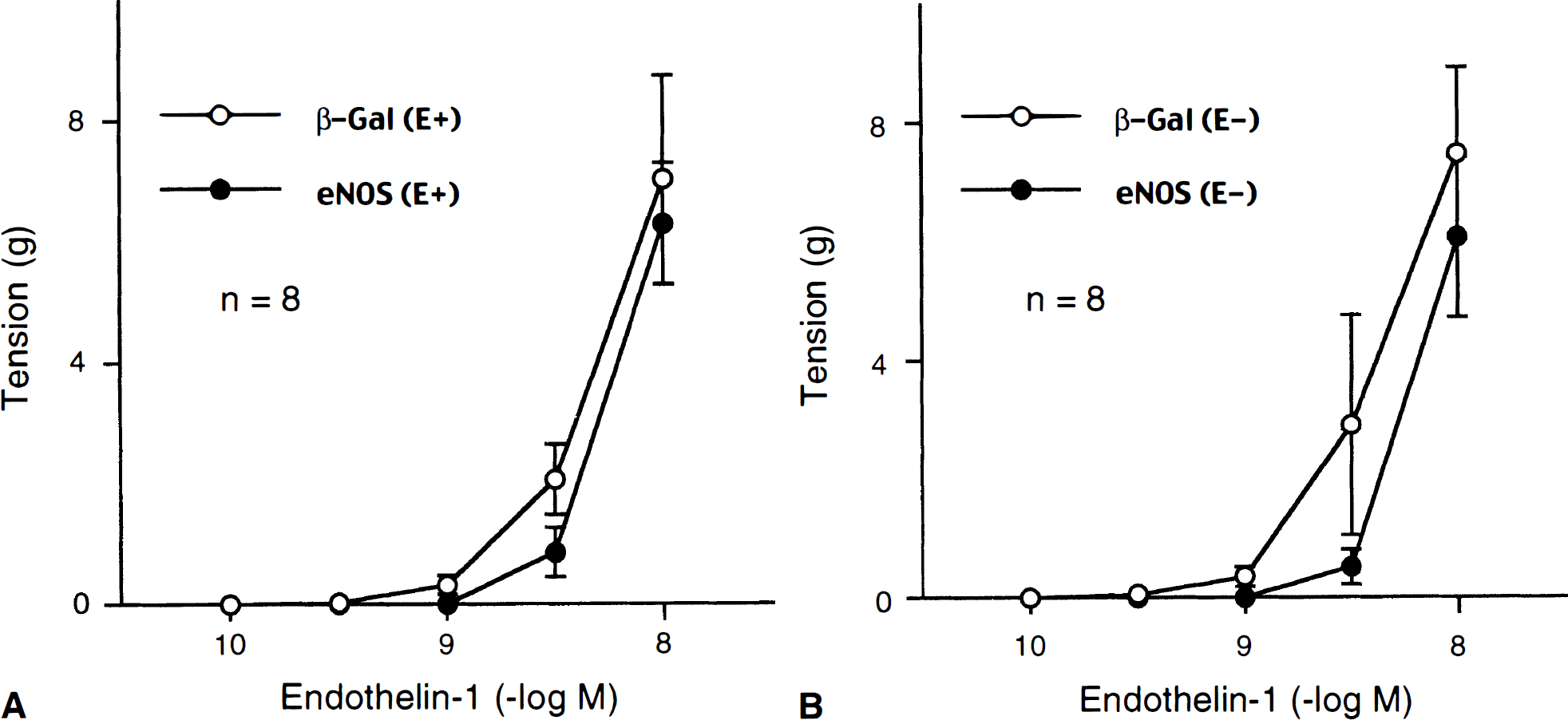
Contractile responses to endothelin-1 in AdCMVβ-Gal-transduced (β-Gal) and AdCMVeNOS-transduced (eNOS) basilar arteries with
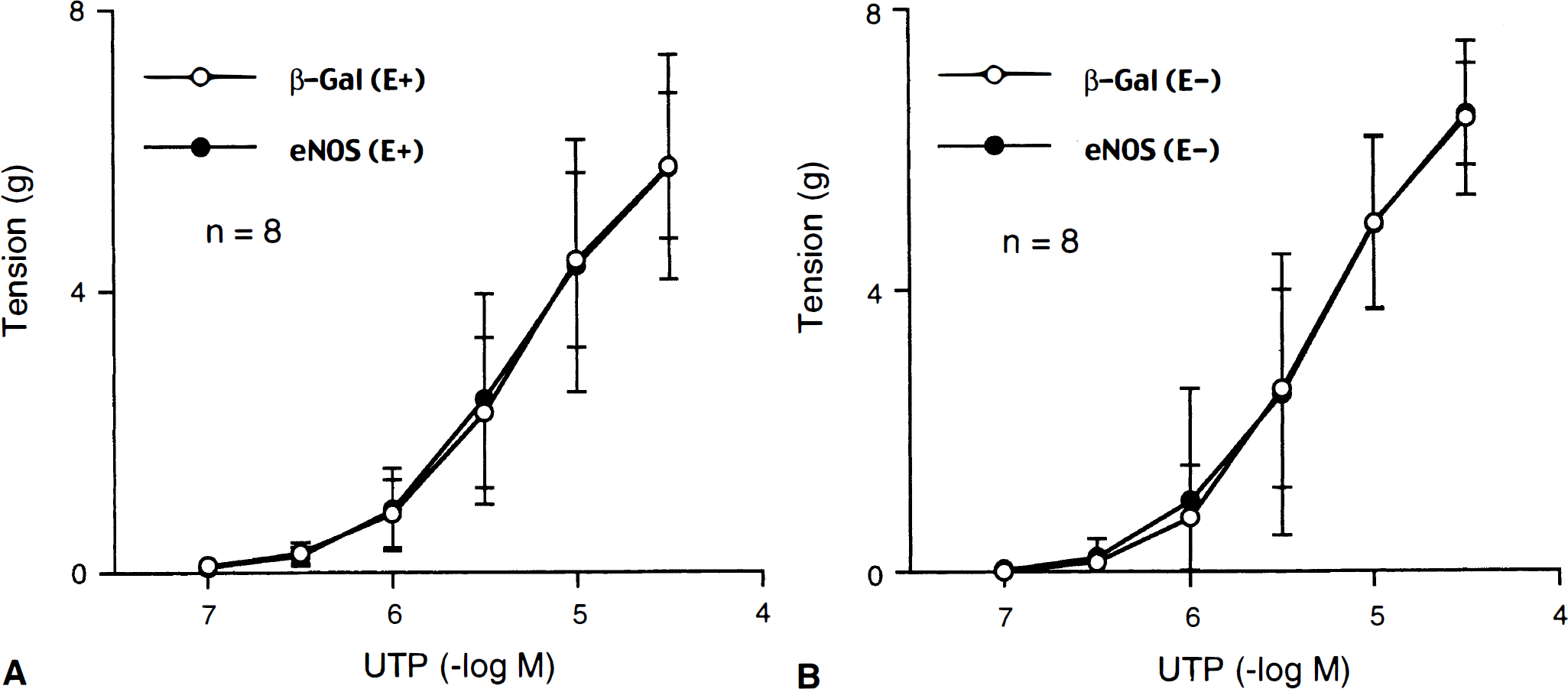
Contractile responses to uridine 5′-triphosphate (UTP) in AdCMVβ-Gal-transduced (β-Gal) and AdCMVeNOS-transduced (eNOS) basilar arteries with
Measurement of intracellular cGMP
No significant difference was detected in basal cGMP levels among control, AdCMVβ-Gal–transduced and AdCMVeNOS-transduced basilar arteries without endothelium (Fig. 8). ET-1 (10−9 mol/L) significantly increased cGMP level only in AdCMVeNOS-transduced arteries (Fig. 8).
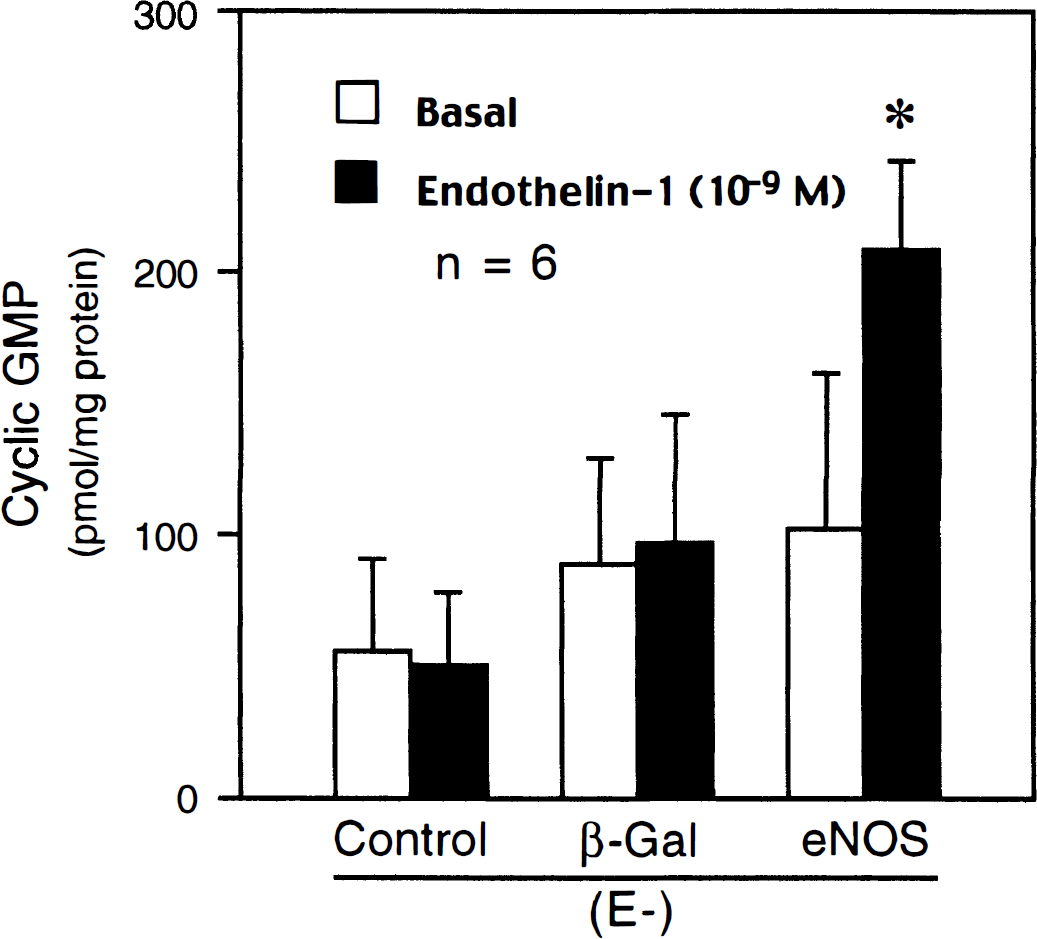
Effect of endothelin-1 (10−9 mol/L) on guanosine 3′,5′-cyclic monophosphate (cGMP) production in nontransduced (Control), AdCMVβ-Gal–transduced (β-Gal), and AdCMVeNOS-transduced (eNOS) basilar arteries without endothelium (E-). Data are shown as mean ± SD. *Significantly different from all other factors; P < .05 by analysis of variance.
DISCUSSION
This is the first study to examine the vascular reactivity to endothelins in cerebral arteries expressing recombinant eNOS gene. The major new findings are that (1) expression of recombinant eNOS gene causes reversal of vasoconstrictor effects of ET-1 into vasodilatation, and that (2) this reversal is mediated by activation of adventitial ETA receptors and subsequent increased formation of NO.
Successful gene transfer and expression of recombinant proteins was confirmed by β-galactosidase histochemistry and eNOS immunohistochemistry. Positive staining of these recombinant proteins was detected predominantly in the vascular adventitia, being consistent with results of our previous ex vivo and in vivo gene transfer studies (Chen et al., 1997a,b; Tsutsui et al., 1998). In canine basilar arteries transduced with AdCMVeNOS, ET-1 produced concentration-dependent relaxations during contractions to UTP, however, it caused only additional contractions in nontransduced control arteries and AdCMVβ-Gal–transduced arteries. In certain vascular beds, ET-1 reportedly can induce relaxations by a release of vasodilator substances including NO and prostacyclin from the endothelium (Ohlstein et al., 1990; Pinheiro and Malik, 1993), or by a direct activation of potassium channels on smooth muscle cells (Hasunuma et al., 1990). In contrast, in canine cerebral arteries ET-1 causes only contractions (present study; Asano et al., 1989; Saito et al., 1991). Thus, ET-1–induced relaxations observed in the present study represent a phenomenon specific for genetically engineered arteries. The relaxations to ET-1 of AdCMVeNOS-transduced arteries were mediated by production of NO, since L-NAME, an inhibitor of NOS, abolished the relaxations, and ET-1 significantly increased cGMP levels in the vascular wall. The increased cGMP levels in response to ET-1 were not detected either in non transduced control vessels or AdCMVβ-Gal–transduced arteries, showing the selective effect of eNOS gene transfer on cGMP formation in response to ET-1.
Our results show that the treatment of adventitia with paraformaldehyde before gene transfer abolished transgene expression of recombinant proteins. We used this technique to confirm that the effect of eNOS gene transfer on vasoreactivity to ET-1 was mediated by adventitial expression of recombinant eNOS. It is not feasible to mechanically remove the adventitial layer of cerebral arteries because cerebral arteries do not have external elastic lamina (Rosenblum, 1994). The chemical treatment of adventitia can be used to selectively disrupt the adventitial function because this treatment did not affect the smooth muscle functions. Contractile responses to UTP and ET-1 (Fig. 3) and relaxations to DEA- NONOate (Table 2), a donor of NO, were maintained in paraformaldehyde-treated arteries. The chemical treatment of adventitia abolished the relaxations to ET-1 in AdCMVeNOS-transduced arteries, indicating that the relaxant effect of ET-1 was dependent on the presence of intact adventitia. Thus, these findings reinforce the idea that ET-1 stimulates NO production in the vascular adventitia transduced with AdCMVeNOS, and causes subsequent cGMP formation and relaxations of arterial smooth muscle.
In AdCMVeNOS-transduced basilar arteries without endothelium, ET-2 also caused relaxations during UTP-induced contractions, however, ET-3 did not produce any relaxations. Previous studies have indicated that ET-1 and ET-2 exhibit relatively higher affinity for ETA receptors than ET-3 (Arai et al., 1990; Sakurai et al., 1990). In addition, sarafotoxin, a selective ETB receptor agonist (Heyl et al., 1993; Williams et al., 1991), did not induce relaxations in arteries transduced with AdCMVeNOS, suggesting that activation of ETA receptors located on the vascular adventitia is involved in the relaxations to ET-1 and ET-2. This concept is further supported by the present results showing that BQ-123, a selective ETA receptor antagonist (Douglas, Meek and Ohlstein, 1994; Ihara et al., 1995), almost abolished ET-1-induced relaxations, whereas BQ-788, a selective ETB receptor antagonist (Ishikawa et al., 1994; Karaki, Sudjarwo and Hori, 1994) was without effect. In our previous ex vivo and in vivo gene transfer studies on canine cerebral arteries, electron microscopy immunogold labeling indicated that expression of recombinant eNOS protein was localized to adventitial fibroblasts (Chen et al., 1997a; Tsutsui et al., 1998). Existing evidence suggests that ET receptors are present on fibroblasts, and that activation of these receptors leads to increase in intracellular free Ca2+ concentration (Gardner, Maher and Aviv, 1989; Ohnishi-Suzaki et al., 1990; Peacock et al., 1992). Therefore, it is very likely that ET-1 may activate recombinant eNOS enzyme via ETA receptor-mediated increase in intracellular Ca2+, thereby facilitating production of NO in adventitial fibroblasts.
ET-1 caused concentration-dependent contractions in arteries that were not contracted by UTP. These contractions were significantly reduced in arteries with AdCMVeNOS transduction. The effect of recombinant eNOS was comparable in arteries with and without endothelial cells, suggesting that release of NO from the adventitia was responsible for the suppression of contractile responses to ET-1. The selectivity of recombinant eNOS inhibitory effect on contractions to ET-1 was shown by the fact that contractions to UTP were identical in AdCMVβ-Gal–transduced and AdCMVeNOS-transduced arteries.
In the cerebral circulation, ET-1 is regarded as a putative mediator of cerebral vasospasm after subarachnoid hemorrhage (Asano et al., 1989; Cosentino and Katusic, 1994; Suzuki et al., 1992; Willette et al., 1994; Zuccarello et al., 1996). An exact pathogenesis of this arterial narrowing is not completely understood; however, it has been postulated that occurrence of vasospasm correlates with an increase of ET-1 concentrations in plasma and CSF (Ehrenreich et al., 1992; Suzuki et al., 1990; Suzuki et al., 1992). In addition, decreased availability of NO is thought to be an important factor responsible for the development of vasospasm (Katusic et al., 1993; Macdonald and Weir, 1991; Pluta et al., 1996). Our results indicate that expression of recombinant eNOS gene may have beneficial effect due to (1) restoration of NO formation and (2) reversal of vasoconstrictor effect of ET-1 into relaxations. A recent study has shown that perivascular expression of recombinant β-galactosidase could be obtained by intracisternal injection of adenoviral vectors in dogs with developed vasospasm after subarachnoid hemorrhage (Muhonen et al., 1997). Therefore, the delivery of adenoviral vectors with functional expression of recombinant eNOS in cerebral arteries may provide a feasible approach to prevent cerebral vasospasm.
The present study shows that contractile responses to ET-1 could be reversed to relaxations by adventitial expression of recombinant eNOS gene. This novel vasodilator mechanism may have important implications in development of gene therapy for various vascular disorders associated with enhanced ET-1 production.
Footnotes
Abbreviations used
Acknowledgment
The authors thank Ms. Janet Beckman for preparing the manuscript.
