Abstract
It has been demonstrated that administration of an interleukin-1 receptor antagonist protein (IL-1ra) reduces ischemic brain injury; however, the detrimental mechanism initiated by interleukin-1 (IL-1) in ischemic brain injury is unclear. In this study, we used mice that were transfected to overexpress human IL-1ra to elucidate the role of IL-1 in the activation of the inflammatory response after middle cerebral artery occlusion (MCAO). Myeloperoxidase (MPO) activity and immunohistostaining were used as a marker of polymorphonuclear leukocytes (PMNL) infiltration. Adenoviral vector (1 × 109 particles) was administered by injection into the right lateral ventricle in mice. Five days later, MCAO was performed on the mice using a suture technique. Permanent MCAO was achieved for 24 hours in the Ad.RSV IL-1ra-transfected, Ad.RSV lacZ-transfected, and saline (control) mice. Myeloperoxidase activity was quantified in each region and localization of MPO was determined by immunohistochemistry. After 2 hours of MCAO, the surface cerebral blood flow was reduced to 13.5% ± 3.4%, 10.75% ± 2.6%, and 10.9% ± 2.6% of baseline in the ischemic hemisphere in Ad.RSV IL-1ra-transfected, Ad.RSV-lacZ-transfected, and saline-treated mice, respectively. The MPO activity in the ischemic hemisphere in the Ad.RSV lacZ group was similar to that in the saline control group (cortex: 0.40 ± 0.22 versus 0.33 ± 0.11; basal ganglia: 0.46 ± 0.23 versus 0.49 ± 0.17; P > 0.05); however, it was significantly reduced in the Ad.RSV IL-1ra group (cortex: 0.18 ± 0.07; basal ganglia: 0.26 ± 0.15; P < 0.05). Myeloperoxidase immunohistochemistry showed that the massive accumulation of MPO-positive cells in the ischemic cortex, striatum, and corpus callosum regions was greatly attenuated in Ad.RSV IL-1ra-transfected mice. Our results indicate that Ad.RSV IL-1ra-transfected mice provide a useful tool to study the mechanism of action of IL-1. The MPO activity assay and immunostaining after 24 hours of focal ischemia were significantly reduced in IL-1ra gene-transfected mice, suggesting that IL-1 may play an important role in the activation of inflammatory cells during focal cerebral ischemia.
Polymorphonuclear leukocytes (PMNL) have been proposed to contribute to the pathogenesis of cerebral ischemia and stroke since the late 1960s (Sornas et al., 1972). Delayed accumulation of PMNL and monocytes after experimental stroke was demonstrated in 1974 (Garcia and Kamijyo, 1974). Using indium-111-labeled autologous PMNL in a model of air embolism-induced cerebral ischemia in dogs, Hallenbeck et al. (1986) confirmed that PMNL accumulate progressively during the first 4 hours of reperfusion. Much of the research literature has demonstrated that brain injury caused by ischemia and reperfusion can be attenuated by PMNL depletion (del Zoppo et al., 1991; Dutka et al., 1989; Shiga et al., 1991). Schurer et al. (1990) showed that antineutrophil serum blocks brain injury after reversible bilateral carotid artery occlusion in rats. Recently, Jiang et al. (1995) reported a neutrophil inhibitory factor, a 41-kDa recombinant glycoprotein, which has a remarkable neuroprotective effect in rat focal ischemia.
Inflammatory cell infiltration can be quantified by using an assay of myeloperoxidase (MPO) activity, an enzyme found within the azurophilic granules of PMNL (Bradley et al., 1982). The MPO activity assay has been considered to be a reliable method to estimate neutrophil infiltration into inflamed tissue, including ischemic brain (Allan et al., 1985; Barone et al., 1991; Change et al., 1986; Goldblum et al., 1985; Krawisz et al., 1984; Smith et al., 1988). Myeloperoxidase immunohistochemistry has demonstrated that MPO-immunopositive cells are greatly increased after focal ischemia in rat brain (Jiang et al., 1995). However, this enzyme assay and immunostaining method has not previously been used to quantify PMNL infiltration in the mouse middle cerebral artery occlusion (MCAO) model. Depletion of circulating neutrophil not only inhibits the increase of MPO activity in the ischemic brain tissue but also attenuates postischemic brain injury (Matsuo et al., 1994).
Interleukin-1 (IL-1) is a potent inflammatory mediator that is also known to be a cofactor in the inflammatory reaction. Constitutive expression of IL-1 in normal brain is low; however, it is markedly increased after transient forebrain ischemia (Blatteis, 1992; Breder et al., 1988), injection of bacterial endotoxin (Ban et al., 1991; Clark et al., 1991; Higgins and Olschowka, 1991), local brain injury (Giulian et al., 1988), administration of excitotoxic agents (Minami et al., 1990), and restraint stress (Minami et al., 1991). Brain IL-1 participates directly in ischemic and traumatic brain injury (Relton and Rothwell, 1992). Although central or systemic administration of an IL-1 receptor antagonist (IL-1ra) reduces ischemic brain injury in short-term experiments, providing and maintaining an effective concentration of IL-1ra in the brain for a long period to treat these diseases may be difficult (Garcia et al., 1995; Relton and Rothwell, 1992).
Interleukin-1ra, a human monocyte-derived 17-kDa polypeptide, was first discovered in a tumoral cell line (human myelomonocytic line, U937) (Carter et al., 1990). Interleukin-1ra is usually produced by the same cells as IL-1 in the normal brain (Andersson et al., 1992; Dinarello and Thompson, 1991; Garcia et al., 1995). Interleukin-1ra can inhibit many actions of IL-1 in vivo and in vitro (Eisenberg et al., 1991). In our previous experiments with adenovirus-injected brain, however, it appears that the protection from ischemia is caused by IL-1ra produced by the ependymal cells. These seem to be the primary cells infected after ventricular infusion of adenovirus as indicated by experiments using lacZ as a marker (Yang et al., 1997). Relatively few cortical cells were infected.
Gene transfer provides a useful tool to study the mechanisms of the action of IL-1β in vivo. Overexpression of IL-1ra using a replication-deficient adenovirus vector that transduces IL-1ra cDNA (Ad.RSV IL-1ra) attenuated ischemic brain injury in both rat and mouse models of focal cerebral ischemia (Betz et al., 1995; Yang et al., 1997). However, the relationship between inhibiting the action of IL-1 and reducing the ischemic inflammatory response has not been well studied. The objectives of the present study are to determine (1) whether overexpression of IL-1ra in the mouse brain will reduce the ischemic inflammatory response in the brain, and (2) whether these effects are accompanied by detectable decreases in neutrophil sequestration in the ischemic territory.
MATERIALS AND METHODS
Adenoviral gene-transfected mouse brain
The production of Ad.RSV IL-1ra and replication-deficient adenovirus vector that transduces Escherichia coli β-galactosidase gene (Ad.RSV lacZ) vectors has been described previously (Roessler et al., 1995). Both viruses use the Rous Sarcoma virus (RSV) promoter to drive gene transcription. The Ad.RSV lacZ serves as a control virus carrying a gene not expected to alter cerebral injury.
Procedures for the use of laboratory animals were approved by the institutional animal care and use committee. Mature male CD-1 mice (Charles River) weighing 30 to 35 g were anesthetized with 4% chloral hydrate (400 mg/kg, intraperitoneally). After induction of the anesthesia, the mice were placed in a stereotactic frame (Kopf Model 921, David Kopf Instruments, Tujunga, CA, U.S.A.) while a burr hole was drilled to the pericranium 1 mm lateral to the sagittal suture and 1 mm posterior to the coronal suture. A 10-μL Hamilton syringe was stereotactically inserted into the lateral ventricle about 2.5 mm under the cortex. One microliter of adenoviral suspension containing 1 × 1012 particles/mL was injected into the lateral ventricle at a rate of 0.2 μL/minute. The needle was then withdrawn over the course of 5 minutes, the hole was sealed with bone wax, the wound was closed with suture, and the animals were allowed to recover in the cage.
To control for the possibility that the adenoviral vectors might have caused acute inflammatory reaction in the brain, three groups of mice (n = 9 in each group) received the same volume of Ad.RSV IL-1ra or Ad.RSV lacZ or saline (control) injection intraventricularly. They were then sacrificed at 1, 3, and 5 days after adenovirus injection. Brains were removed and frozen immediately in 2-methylbutane at −42°C. Cryostat sections (10-μm thickness) were cut at −15°C and mounted on slides. The sections were dried with gentle cool air and stained with hematoxylin-eosin. The inflammatory effect of the adenovirus injection in these groups has been examined.
Production of focal ischemia
Five days after administration of adenovirus (either Ad.RSV IL-1ra or Ad.RSV lacZ) or saline (control), three groups of mice (8 to 10 mice in each group) were reanesthetized with 1.5% isoflurane in 70% N2O and 30% O2. The animals were ventilated to maintain PaO2 at 90 mm Hg or above. The body temperature was maintained at 37.0° ± 0.5°C using a heating pad during the operation. The middle cerebral artery was permanently occluded for 24 hours in Ad.RSV IL-1ra, Ad.RSV lacZ, and saline control mice. The MCAO method was described in our previous studies (Yang et al., 1994). Briefly, the left common carotid artery was exposed through a midline incision. The internal carotid artery was then isolated and its branch, the pterygopalatine artery, was ligated close to its origin. Then a 2-cm length of 5-0 rounded-tip Dermalon suture was advanced from the external carotid artery through the common carotid artery and then up to the internal carotid artery for a distance of 11.0 ± 0.5 mm.
Laser-Doppler flowmeter
A laser-Doppler flowmetry (LDF) monitor (Model BPM2 System, Vasamedics Inc., St. Paul, MN, U.S.A.) equipped with a small-caliber probe of 0.7 mm diameter (P-433, Vasamedics Inc.) was used to measure local cerebral blood flow (CBF). Because the mouse skull is very thin, the LDF probe was placed over intact pericranium and readings were obtained through an intact skull. The probe was gently lowered onto the pericranium 3.5 mm lateral to the sagittal suture and 1 mm posterior to the coronal suture. Baseline LDF recordings were made 10 minutes before MCAO. Temperature was maintained at 37.0° ± 0.5°C using a heating lamp and heating pad. Warmed saline was slowly rinsed around the probe during the experiments (Dirnagl et al., 1989). Continuous digital display LDF values were averaged for 5-second intervals and were recorded every 30 minutes during the first 2 hours of MCAO. Mice were allowed to awaken in the cage after the last surface blood flow measurement. The CBF values were then calculated and expressed as percentages of baseline values (Yang et al., 1994).
Myeloperoxidase assay
The mouse was reanesthetized with 4% chloral hydrate and perfused transcardially with isotonic saline (a pressure of 100 mm Hg, at 25°C) before brain removal to flush all blood components from the vasculature. An 8-mm slice of the forebrain between the olfactory bulbs and the cortical-cerebellar junction was sectioned and dissected into four separate segments (the wet weight of each segment was ≈80 mg): (1) ischemic cortex; (2) ischemic basal ganglia; (3) contralateral cortex; and (4) contralateral basal ganglia. The four forebrain segments were then immediately frozen at −70°C. Tissue segments were thawed on ice, weighed, and homogenized (Model PT/1035 Polytron, Brinkman Instruments Inc., Westburg, NY, U.S.A.) in 4 mL of 50 mmol/L Tris-HCl, pH 7.4, at 4°C. Samples were added to 20 mL 5 mmol/L phosphate buffer, pH 6.0, at 4°C, homogenized using a tissumizer homogenizer (Tekmar Co., Cincinnati, OH, U.S.A.: three on/off cycles at 5-second intervals), and centrifuged at 30,000g for 30 minutes at 4°C. The supernatant was discarded and the pellet was washed again as described above. After decanting the supernatant, the pellet was extracted by suspension in 0.5% hexadecyltrimethylammonium bromide (HTAB, Sigma Chemical CO., St. Louis, MO, U.S.A.) in 50 mmol/L potassium phosphate buffer at 25°C for approximately 2 minutes at an original tissue wet weight to volume ratio of 1:2. The samples were immediately frozen on dry ice. Freeze and thaw cycles were then performed three times with sonications for 10 seconds at 25°C between cycles. After the last sonication, the samples were incubated at 4°C for 20 minutes and centrifuged at 12,500g for 15 minutes at 4°C. The supernatants (100 μL) were mixed with 100 μL of 50 mmol/L phosphate buffer, pH 6.0, containing o-dianisident dihydrochloride (0.167 mg/mL; Sigma Chemical Co.) and hydrogen peroxide (0.005%). Absorbency was measured at 460 nm using a Beckman DU-7 spectrophotometer with kinetic analysis capability with the absorbance recorded at 15-second intervals over a 3-minute period. One unit of MPO activity is defined as the amount that degrades 1 mole of peroxide/minute at 25°C. Myeloperoxidase activity for each tissue sample was normalized on the basis of grams wet weight tissue.
Myeloperoxidase immunohistochemistry
Three groups of mice (n = 6 in each group) were sacrificed 24 hours after MCAO by decapitation. Brains were removed and frozen immediately in 2-methylbutane at −42°C for 5 minutes. Cryostat sections (20-μm thickness) were cut at −15°C and mounted on slides. The sections were dried by gentle cool air from a dryer for a short period and kept in −20°C until use. Immunostaining was performed on the same day as sectioning. The sections were first fixed with Carnoy fixation (60% methanol, 30% chloroform, and 10% acetate) for 20 minutes, then rinsed three times for 5 minutes with 0.1 mol/L phosphate-buffered saline (pH 7.4, Sigma Chemical Co.). Nonspecific binding was blocked by using 10% normal goat serum for 30 minutes at room temperature. The sections were incubated in the 1:400 dilution of rabbit polyclonal anti-MPO primary antibody (A398, DAKO Inc., Carpinteria, CA, U.S.A.) overnight at 4°C. After treatment with 1% H2O2 in 30%/70% methanol/phosphate-buffered saline solution, the sections were then incubated with biotinylated goat anti-rabbit secondary antibody for 90 minutes at room temperature followed by an ABC process (ABC-Elite kit, Vector, Burlingame, CA, U.S.A.) and finally treated with stable 3,3′-diaminobenzidine tetrahydrochloride (DAB, Research Genetics) as a peroxidase substrate. The negative control was also carried out using the same concentration of normal rabbit immunoglobulin instead of primary antibody.
Statistical analysis
All data are expressed as mean and standard deviation. Parametric data among the Ad.RSV IL-1ra, Ad.RSV lacZ, and saline control groups were evaluated using analysis of variance and two-tailed Student's t test with Bonferroni adjustment. A probability value of less than 5% was considered statistically significant.
RESULTS
Neither Ad.RSV IL-1ra nor Ad.RSV lacZ recombinant virus had any effect on weight gain during the 5 days after virus injection. No neurologic deficits were observed in either Ad.RSV IL-1ra- or Ad.RSV lacZ-transfected mice. The rectal body temperature range was 37.0° ± 0.5°C. Hematoxylin-eosin stained sections were scored for the degree of cellular reaction around the needle tract 1 to 5 days after Ad.RSV IL-1ra, Ad.RSV-lacZ, or saline intraventricular injection. All three groups of mice had mild reactions around the needle tract (Fig. 1 A–C). There were mild host reactions near and inside the ipsilateral ventricle. Host reactions to the adenoviral vectors were similar regardless of the encoded transgene. These initial data suggest that central adenoviral injection does not substantially change physiologic parameters in mice.
Similar host reactions around the needle tract and near the ventricles in the Ad.RSV IL-1ra-transfected
Cerebral blood flow
Surface CBF measurements from mice undergoing MCAO are depicted in Fig. 2. In all three groups the CBF measured in the contralateral hemisphere during MCAO was about 100% of preocclusion (baseline) flow. The surface CBF in the ipsilateral hemisphere, however, was reduced immediately after introduction of the suture. During the first 2 hours of MCAO, the blood flow in Ad.RSV IL-1ra-transfected, Ad.RSV lacZ-transfected, and saline-treated mice was 13.5% ± 3.4%, 10.7% ± 2.6%, and 10.9% ± 2.6% of baseline, respectively (n = 8 to 10, not significantly different from each other, P > 0.05). The CBF measurement was necessary to confirm that ischemia occurred focally, rather than globally, because systemic hypotension or intracranial hypertension may develop.
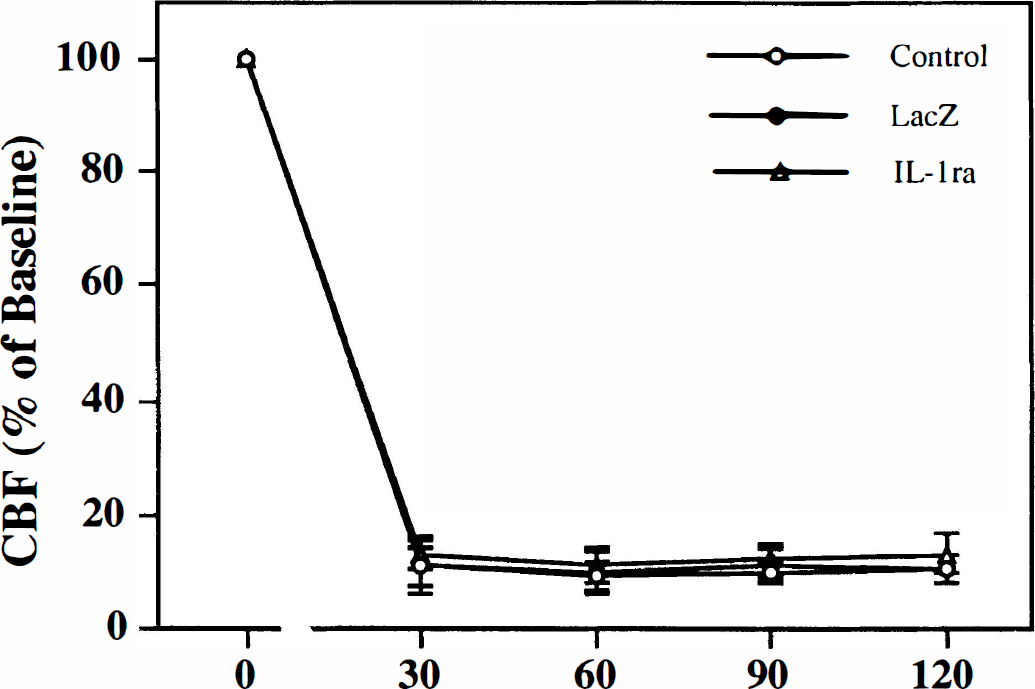
Changes it surface cerebral blood flow (CBF) in mice during the first 2 hours of MCAO in the Ad.RSV IL-1ra-transfected (▵), Ad.RSV lacZ-transfected (•), and saline-treated (○) mice. Laser-Doppler flowmetry (LDF) values are expressed as a percentage of the baseline CBF (before MCAO recording). Values are mean ± SD. n = 8 to 10 mice in each group.
Myeloperoxidase activity
Four brain regions (contralateral cortex, contralateral basal ganglia, ipsilateral cortex, and basal ganglia) were assayed for MPO activity, an indicator of neutrophil accumulation. There were no significant differences in MPO activity among the four regions of the sham-operated animals with values of between 0.023 and 0.065 units/g wet tissue. Myeloperoxidase activities in both cortex and basal ganglia of the contralateral hemisphere in the Ad.RSV IL-1ra-transfected, Ad.RSV lacZ-transfected, and saline-treated mice were slightly increased, but there were no significant differences among the groups (P > 0.05). Compared with the contralateral side, however, the MPO activity was significantly increased in both the ipsilateral cortex and basal ganglia in the Ad.RSV lacZ-transfected (0.40 ± 0.22 and 0.46 ± 0.23 units/g) and saline-treated (0.33 ± 0.11 and 0.49 ± 0.17 units/g) mice (P < 0.05, Fig. 3). The increases of MPO activity in the ipsilateral hemisphere in the Ad.RSV lacZ-transfected and the saline-treated groups were similar. The highest level of MPO activity was found in the ipsilateral basal ganglia region. Compared with the Ad.RSV lacZ-transfected and the saline-treated mice, however, the MPO activity was significantly inhibited in the Ad.RSV IL-1ra-transfected group both in the cortex and the basal ganglia region (0.18 ± 0.07 and 0.26 ± 0.15 units/g versus Ad.RSV lacZ-transfected and salinetreated groups, P < 0.05, Fig. 3).
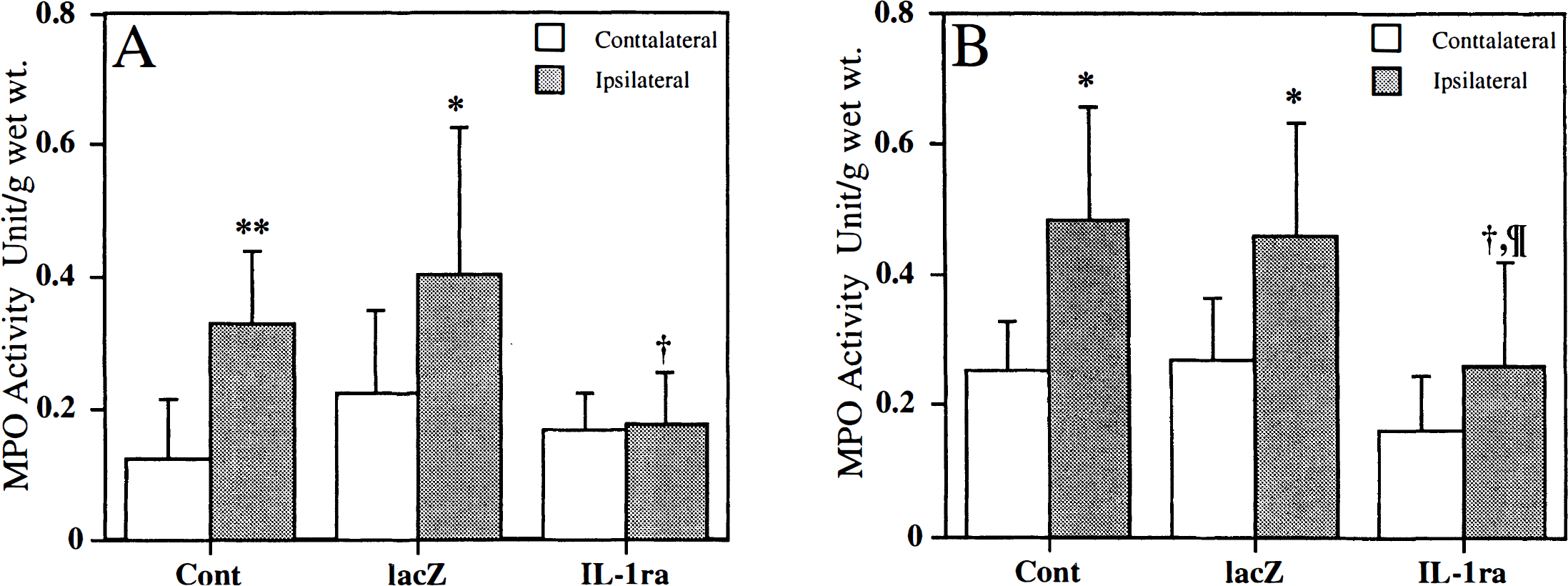
Myeloperoxidase (MPO) activity after 24 hours of permanent MCAO. Five to six days before MCAO, mice received intraventricular injections containing 1 × 109 particles of either Ad.RSV IL-1ra or Ad.RSV lacZ, or saline. Brains were removed 24 hours after MCAO and divided into cortex (CTX) and basal ganglia (BG). The MPO activity was measured in the CTX
Myeloperoxidase immunohistochemistry
In the Ad.RSV lacZ-transfected and saline-treated mice, immunohistochemistry showed that MPO-immunoreactive cells distributed in large quantities in and around the ischemic lesion. Positive cells were present in both ipsilateral striatum and cortex, and in the ipsilateral corpus callosum. Some MPO-positive cells were found in the contralateral hemisphere, especially in the corpus callosum region. All positive cells were either round or oval in morphology with multiple nuclei. This indicates that the increased MPO staining is really caused by the accumulation of PMNL (Fig. 4). The quantity of MPO-immunopositive cells in the ipsilateral hemisphere in the Ad.RSV lacZ-transfected group appeared similar to that in the saline-treated group; however, the number of immunopositive cells was greatly reduced in the ipsilateral hemisphere in Ad.RSV IL-1ra-transfected animals. The distribution of MPO immunostaining paralleled the results on MPO activity in each group.
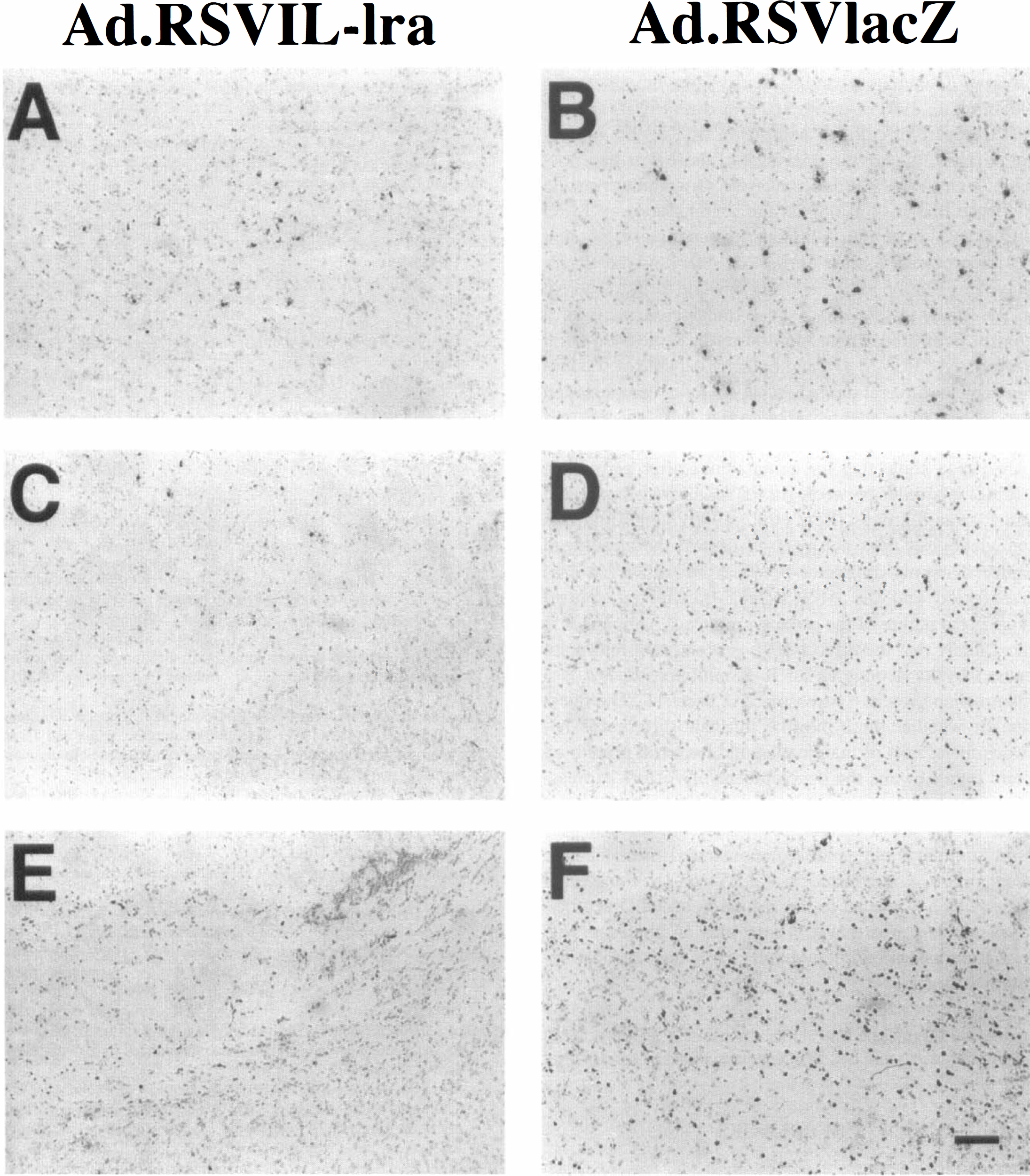
Immunostaining with anti-MPO antibody in coronal sections (20 μm) of ischemic hemisphere in Ad.RSV IL-1ra-transfected
DISCUSSION
In the present study, neutrophil accumulation was quantified by MPO activity assay. Myeloperoxidase measurement as a marker of inflammatory cell infiltration has proven to be of value in the brain (Barone et al., 1992; Jiang et al., 1995). Barone et al. (1991) demonstrated that an accumulation of neutrophils can be identified histologically and quantified using the MPO activity assay in spontaneously hypertensive rats after temporary MCAO. Using a suture MCAO model, Matsuo et al. (Matsuo et al., 1994) found that MPO activity was significantly increased in both cortex and caudate putamen, and this was maintained until 72 hours after reperfusion. Our study demonstrates that MPO activity is a sensitive and reproducible marker in small animal tissue such as the mouse brain. The results indicate that 24 hours of permanent MCAO in mice provides sufficient stimulation to induce accumulation of neutrophils in the ischemic tissue with MPO activity being significantly increased in the Ad.RSV lacZ-transfected and saline-treated mice, especially in the basal ganglia region.
Myeloperoxidase immunostaining showed similar results, with MPO-positive cells increasing mainly in ischemic basal ganglia, then cortex, and a few in the contralateral hemisphere (Fig. 4). The increase of PMNL in the ischemic core area suggests the inflammatory response occurs in areas of the lowest blood flow because the blood supply to the ischemic core region is very poor in this model. The LDF showed that surface CBF in the ischemic hemisphere remained at only 10% to 15% of baseline. Our previous study using this suture MCAO model showed the lesion in the basal ganglia was more severe than in the cortex (Yang et al., 1994).
The results of our study confirm and extend our previous findings, and show that the intraventricular administration of Ad.RSV IL-1ra resulted in a remarkable inhibition of PMNL accumulation after MCAO in mice (Fig. 3). At present, little is known about the mechanisms of action of IL-1 in acute ischemic brain injury; however, from the known action of IL-1 in brain, several possibilities can be considered. Generally, the proposed mechanisms of brain injury produced by IL-1 relate to its proinflammatory and immunologic properties (Dinarello, 1991). The proinflammatory effects of IL-1 include leukocytosis, increased expression of endothelial cell adhesion molecules, and neutrophil tissue infiltration even in the low picomolar range. Polymorphonuclear leukocytes may actively participate in the brain injury that develops after MCAO. Release of IL-1 during cerebral ischemia may stimulate the synthesis of other cytokines (Hu et al., 1994). These cytokines share many central actions with IL-1, and any increase of these cytokines could in turn exacerbate brain injury (Mori et al., 1992). Interleukin-1 is a potent activator of excitatory amino acid and nitric oxide release (Rothwell, 1991). These substances have been proposed as important mediators of ischemic damage (Farooqui and Horrocks, 1992). It is unknown whether IL-1 produces neuronal death directly or exacerbates other forms of damage, or both (Quagliarello et al., 1991). Interleukin-1 could influence neuronal survival in vivo through indirect effects on microglia. Exposure of cultured neurons to activated microglia results in rapid cell death, which appears to be caused by the release of excitatory amino acids from the microglia (Piani et al., 1991). Araujo and Cotman (1990) reported that exposure of cultured neurons to IL-1 results in neurotoxicity, although the relevance of these data to a possible in vivo role is difficult to prove. Our studies provide evidence that IL-1ra inhibits the accumulation of MPO during focal ischemia. Although the beneficial effect that IL-1ra provided is clear during MCAO, how the IL-1ra plays a role in the ischemic event must be further studied.
We chose Ad.RSV lacZ gene-transfected mice as a control virus group because it shares the same adenoviral vector as Ad.RSV IL-1ra and both are capable of producing protein. Several published papers have demonstrated that the use of an adenoviral control is necessary (Horellou et al., 1997; Viola et al., 1995), although our published data show that the brain injury after permanent MCAO is similar between Ad.RSV lacZ-transfected and saline-treated groups (Betz et al., 1996) suggesting that the adenovirus does not by itself protect the brain from injury. The only difference between the Ad.RSV IL-1ra-transfected and the Ad.RSV lacZ-transfected control mice is the nature of the protein produced. Although it is most likely that the effects of IL-1ra on brain injury are caused by its actions as an IL-1 inhibitor, IL-1ra is a mammalian protein whereas lacZ is bacterial. The question of whether there may be intrinsic differences between the effects of mammalian and bacterial protein on brain injury awaits the development of vectors expressing mutant mammalian proteins with no biologic activity.
In summary, our results demonstrated that ischemic brain injury is attenuated in Ad.RSV IL-1ra-transfected mice, which is related to the reduction of MPO activity. Our study suggests that IL-1 may play an important role in the activation of inflammatory cells during focal cerebral ischemia.
Footnotes
Acknowledgements
The authors thank Dr. Richard F. Keep for helpful discussion and Ms. Kathleen Donahoe for editorial assistance.
