Abstract
The authors investigated the influence of age on the outcome of cerebral ischemia in wild-type mice and in mice with a deletion of the inducible nitric oxide synthase (iNOS) gene. The middle cerebral artery was permanently occluded in iNOS-null mice and in wild-type (C57BL/6) controls aged 4, 8, 16, and 24 weeks. Infarct volume was determined in thionin-stained brain sections 4 days after permanent middle cerebral artery occlusion. No differences in forebrain volume were found among wild-type and iNOS-null mice at the ages studied (P> 0.05). In C57BL/6 mice (n = 5 to 6/group), neocortical infarct volume corrected for swelling was 28 ± 5 mm3 in 4-week-old mice, 28 ±3 at 8 weeks, 35 ±4 at 16 weeks, and 37 ± 6 at 24 weeks (mean ± SD). iNOS-null mice (n = 5 to 6/group) had smaller infarcts than wild-type controls at all ages (P < 0.05). However, the magnitude of the reduction was greater in 4-week-old (–29% ± 10%) or 8-week-old mice (–24% ± 8%), than in 16-week-old (–14% ± 10%) or 24-week-old mice (–11% ± 6%). Neurologic deficit scores improved significantly between 24 and 96 hours in 4- and 8-week-old iNOS-null mice compared with age-matched wild-type mice (P < 0.05). However, in 16- or 24-week-old iNOS-null mice, neurologic deficits did not improve (P > 0.05). The authors conclude that in iNOS–/– and in wild-type mice, the size of the infarct produced by occlusion of the middle cerebral artery is larger in older than in younger mice. However, the reduction in infarct volume observed in iNOS-null mice is age-dependent and is greatest at 1 to 2 months of age. Therefore, age is a critical variable in studies of focal cerebral ischemic damage, both in wild-type mice and in mouse mutants.
The development of mice with deletion or overexpression of genes relevant to ischemic brain injury has led to a growing number of studies focusing on the mechanisms of focal cerebral ischemia in mice (for a review, see Aguzzi et al., 1994; Chan, 1994). However, models of focal cerebral ischemia in mice are not as well characterized as those in other species (Ginsberg and Busto, 1989; McAuley, 1995). For example, although several studies have investigated the outcome of middle cerebral artery (MCA) occlusion in young and aged rats (Davis et al., 1995; Duverger and MacKenzie, 1988; Futrell et al., 1991; Sutherland et al., 1996), little is known about the effect of age on ischemic brain damage in mice. Genetically engineered mice are, usually, in limited supply, and mice with a variable age range have to be included to obtain a number of animals sufficient to provide statistically valid data (Huang et al., 1996; Kondo et al., 1997; Yang et al., 1994; Zhang et al., 1997). It is not known, however, whether small variations in the age of the mice affect the outcome of cerebral ischemia.
It also remains to be established whether the age of the mice modulates the “protective” effects of overexpression or deletion of the gene of interest on cerebral ischemic injury. For example, mice with deletion or overexpression of certain genes have reduced cerebral ischemic damage and improved neurologic deficits after MCA occlusion (for review, see Iadecola et al., 1999), It is not known how the age of the mice affects the protection conferred by these genetic manipulations. It would, therefore, be of interest to study the effects of age on the outcome of focal cerebral ischemia and to determine whether the neuroprotection observed in certain transgenic and knockout mice is age-related.
In this study we examined the effects of relatively small variation in the age of mice on the outcome of cerebral ischemia in wild-type mice and on the neuroprotection conferred by deletion of the inducible nitric oxide synthase (iNOS) gene in iNOS-null mice. We found that the infarct produced by MCA occlusion is significantly smaller in 4- to 8-week-old mice than in 16- to 24-week-old mice. This effect is observed both in wild-type (C57BL/6) and in iNOS-null mice. Furthermore, the protection observed in iNOS-null mice is also age-dependent and is greatest in 4- to 8-week-old mice. Although the findings indicate that age is a critical variable in studies of focal cerebral ischemia, they suggest that the magnitude of the iNOS-related neuroprotection is age-dependent. Age must be carefully considered in studies of cerebral ischemia and neuroprotection in mice.
METHODS
Animals
Studies were approved by the Animal Care Committee of the University of Minnesota. Experiments were conducted in 22 male C57BL/6 (Jackson Laboratories, Bar Harbor, ME, U.S.A.) and 22 male iNOS-null (iNOS-/-) mice. As described in detail elsewhere (Iadecola et al., 1997), iNOS–/– were obtained from a colony established from breeding pairs provided by Dr. C. Nathan and J. Mudgett (MacMicking et al., 1995). Mice were studied at 6, 8, 16, and 24 weeks of age.
Middle cerebral artery occlusion
Focal cerebral ischemia was produced by occlusion of the MCA as previously described (Iadecola et al., 1997; Zhang et al., 1997). Briefly, mice were anesthetized with halothane (induction 5%, maintenance 1%, in an oxygen-nitrogen mixture). Body temperature was maintained at 37°C ± 0.5°C by a thermostatically controlled infrared lamp. A 2-mm hole was drilled at a site superior and lateral to the left foramen ovale to expose the left MCA. The MCA was elevated and cauterized distal to the origin of the lenticulostriate branches. Wounds were sutured and mice were returned to their cages and closely monitored until they recovered from anesthesia. Rectal temperature was measured and controlled until mice regained full consciousness. Thereafter, rectal temperature was measured at 24-hour intervals until the time of sacrifice.
Determination of infarct volume
Mice were killed for measurement of infarct volume 96 hours after MCA occlusion, shortly after the last neurologic evaluation. As described in detail elsewhere (Iadecola et al., 1997; Zhang et al., 1997), brains were removed and frozen in cooled isopentane (–30°C). Coronal forebrain sections (thickness, 30 µm) were serially cut in a cryostat, collected at 150-µm intervals, and stained with thionin. Infarct volume was determined by an image analyzer (MCID, Imaging Research Inc.) (Iadecola et al., 1997; zhang et al., 1997). To factor out the contribution of ischemic swelling to the volume of the lesion, infarct volume in cerebral neocortex was corrected for swelling by comparing the volume of ischemic and nonischemic hemispheres (Iadecola et al., 1997; Lin et al., 1993; Zhang et al., 1997).
Neurologic evaluation
Neurologic deficits were assessed by a neurologic scoring system using a modification of the postural reflex test, as previously described (Bederson et al., 1986; Iadecola et al., 1997). The examiner was not aware of the identity of the mice. Neurologic scores were ÷ 0, normal motor function; 1, flexion of torso and contralateral forelimb when mouse was lifted by the tail; 2, circling to the contralateral side when mouse was held by the tail on a flat surface, but normal posture at rest; 3, leaning to the contralateral side at rest; 4, no spontaneous motor activity. Mice were evaluated before MCA occlusion and at 24-hour intervals up to 96 hours after MCA occlusion.
Data analysis
Data in text, tables, and figures are expressed as mean ± SD. Comparisons among multiple groups were statistically evaluated by one-way analysis of variance followed by post hoc Tukey-Kramer honestly significant difference (HSD) test (Systat, Evanston, IL, U.S.A.). Two-group comparisons were analyzed by the two-tailed t test for independent samples. Neurologic scores were evaluated by nonparametric statistical procedures. For neurological scores, two-group comparisons were analyzed by the Mann-Whitney U analysis, whereas multiple comparisons were analyzed by the Kruskal-Wallis test followed by post hoc Tukey-Kramer HSD test (Kirk, 1982). For all statistical procedures, differences were considered significant at P < 0.05.
RESULTS
Body weight and forebrain volume in C57BL/6 and iNOS–/– mice
The body weight of C57BL/6 and iNOS–/– mice increased with age (Tables 1 and 2; P < 0.05, analysis of variance and Tukey's test), but did not differ between wild-type and null mice of comparable age (Tables 1 and 2) (P > 0.05). Forebrain volume, measured from the tip of the striatum to the posterior commissure, did not change with age and did not differ between C57BL/6 and iNOS–/– mice (Tables 1 and 2) (P > 0.05).
Effect of age on infarct volume in C57BL/6 and iNOS–/– mice
Before MCA occlusion, rectal temperature in C57BL/6 mice was 35.9°C ± 0.8°C at 4 weeks of age, 35.7°C ± 0.9°C at 8 weeks, 36.0°C ± 0.7°C at 16 weeks, and 35.9°C ± 0.5°C at 24 weeks. Middle cerebral artery occlusion did not alter rectal temperature during the 4-day survival period (P > 0.05). The size of the infarct produced by MCA occlusion did not differ between 4- and 8-week-old C57BL/6 mice, but it increased at 16 and 24 weeks of age (Table 1). In 24-week-old mice, the infarct was 32% larger than that of 8-week-old mice (P < 0.05; analysis of variance and Tukey's test).
Preischemic rectal temperature in iNOS–/– mice was 36.1°C ± 0.6°C at 4 weeks of age, 36.2°C ± 0.5°C at 8 weeks, 36.3°C ± 0.5°C at 16 weeks, and 36.3°C ± 0.7°C at 24 weeks. Middle cerebral artery occlusion did not alter rectal temperature during the survival period (P > 0.05). No differences in rectal temperature were noted between iNOS–/– and C57BL/6 mice before or after MCA occlusion (P > 0.05). Although the infarct produced by MCA occlusion in iNOS–/– mice was smaller that those observed in C57BL/6 mice of comparable attributed to differences in brain volume. Although body weight increased with age in both C57BL/6 and iNOS-null mice, the volume of the forebrain, measured from the tip of the striatum to the posterior commissure, did not change. Furthermore, forebrain volume did not differ between C57BL/6 and iNOS mice. The age-related changes in infarct volume could not be the consequence of differences in ischemic swelling because volumetric data were corrected for swelling. Similarly, differences in body temperature are unlikely to contribute to the findings of the present study because rectal temperature was monitored and did not differ among groups.
One potential problem with using mutant mice generated by homologous recombination is related to their heterogeneous genetic background. Usually, genetically modified stem cells of SV129 mice are injected into blastocysts of C57BL/6 mice (Joyner, 1994). The blastocysts are then implanted in pseudopregnant foster mothers, and the offspring are typically bred with C57BL/6 mice to generate mice homozygous for the deletion (Joyner, 1994). Thus, null mice generated in this fashion have a mixed genetic background containing both SV129 and C57BL/6 genes (Gerlai, 1996). However, heterogeneity of the genetic background is unlikely to contribute to the findings of the present study. First, we have previously reported that iNOS–/– mice have infarcts that are smaller in comparison to both parental strains (C57BL/6 and SV129) (Iadecola et al., 1997). Therefore, differences in susceptibility to ischemic damage between parental strains is unlikely to play a role. Second, infarct size was found to be larger in older than in younger iNOS–/– mice. Therefore, the change in infarct size was present in mice with the same degree of genetic heterogeneity.
Previous studies have demonstrated that focal cerebral ischemic damage is exacerbated in aged rats (Davis et al., 1995; Sutherland et al., 1996). In Fischer rats, infarct volume does not increase with age (Duverger and MacKenzie, 1988), suggesting that there might be strain differences in the effect of aging on ischemic injury. In studies in which an effect of age was found, the age difference in which infarct volume was reported to be increased was relatively large, ranging from 11 to 26 months (Davis et al., 1995; Sutherland et al., 1996). In contrast, in the present study we found that the increase in injury volume occurred in a much smaller age range, 16-week-old mice having infarcts larger than 8-week-old mice. The increased susceptibility to cerebral ischemic damage in older rats has been attributed to pathologic processes associated with aging. These include, for example, alterations in cerebrovascular morphology, reduced collateral flow, reduced cerebrovascular reactivity, and increased oxidative stress (Akima et al., 1986; Ames et al., 1993; Davis et al., 1995; Knox and Oliveira, 1980; Shigenaga et al., 1994; Tamaki et al., 1995). Considering that, in mice, the increase in ischemic damage occurs between 8 and 16 weeks of age, it is unlikely that factors related to senescence are responsible for the effect. Mice in the age group we studied are considered young adults. It is therefore more likely that factors related to normal development and maturation are involved in the mechanisms of the increase in infarct size. However, the specific mechanisms responsible for the age-dependent enlargement of the infarct remain unknown and will have to be explored in future studies.
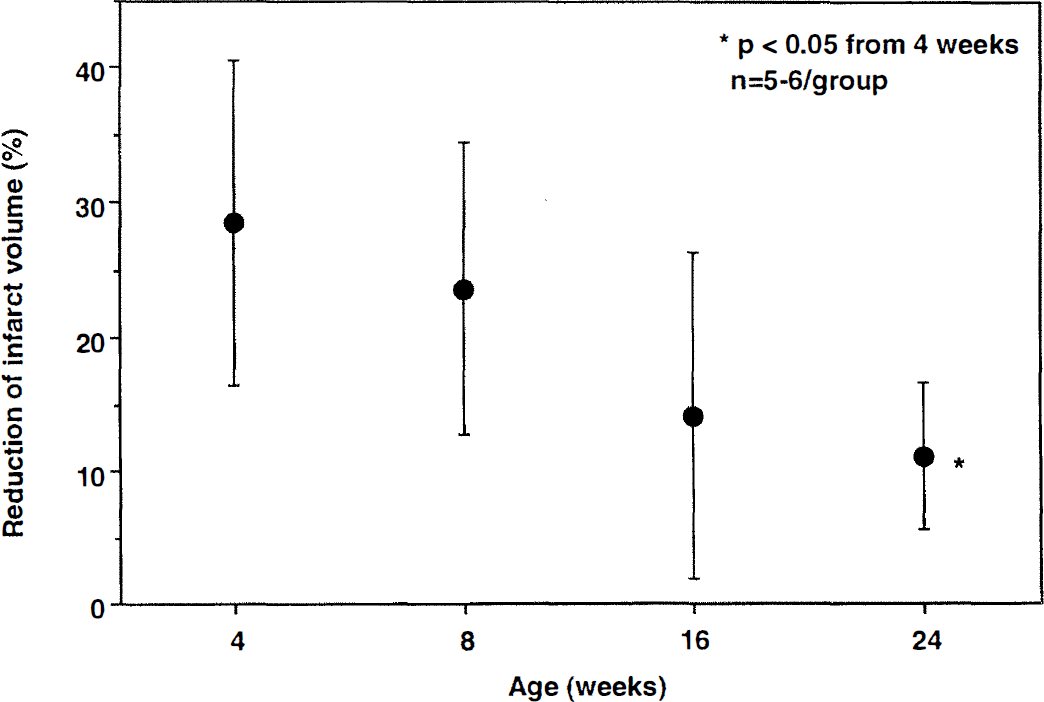
Reduction in infarct volume in iNOS–/– mice at different ages. Focal cerebral ischemia was produced by MCA occlusion, and infarct volume was determined 96 hours later. Data are expressed as percent reduction relative to age-matched C57BL/6 mice (Table 1). The reduction in infarct volume was greater at 4 weeks of age than at 24 weeks (P < 0.05 versus 4 weeks; analysis of variance and Tukey's test).
We also found that the magnitude of the protection observed in iNOS–/– mice is age-dependent. Although the infarct in null mice was smaller at all ages studied, the maximal reduction was observed in 4- to 8-week-old mice. This observation suggests that the component of cerebral ischemic damage that is related to iNOS expression is age sensitive in this model. Thus, maximal protection is observed only in a well-defined age window. It remains to be determined whether the age-dependent neuroprotection observed in iNOS–/– mice occurs also in other mice mutants in which resistance to cerebral ischemia has been observed (Iadecola et al., 1999). The mechanisms of the age-dependence of the protective effect of iNOS deletion remain to be elucidated. One possibility is that the inflammatory response to MCA occlusion decreases during this narrow age window, resulting in a reduction in the expression of inflammation-related genes including iNOS. Accordingly, the component of the damage that is related to iNOS-derived nitric oxide may decline with age. Alternatively, the susceptibility of the brain to nitric oxide-mediated injury may change with maturation. Experiments in which the intensity of postischemic inflammation and iNOS expression are studied in different age groups will be required to address this issue. It would also be important to determine whether the protective effect exerted by iNOS inhibitors is related to the age of the animals (Iadecola et al., 1995, 1996; Nagayama et al., 1998).
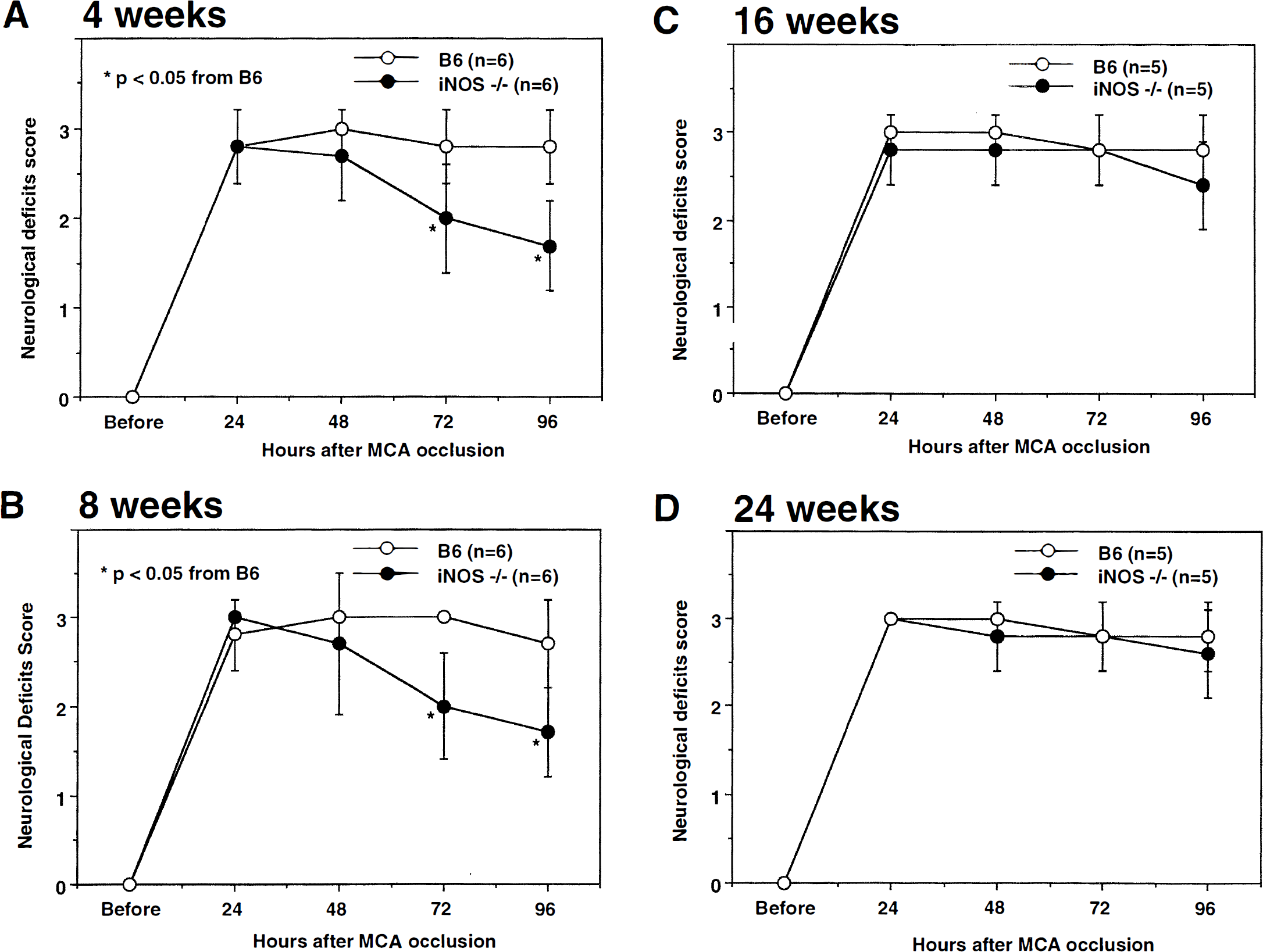
Neurologic deficits resulting from MCA occlusion in C57BL/6 and iNOS–/– mice aged 4, 8, 16, and 24 weeks, Neurologic deficits were assessed by a modification of the postural reflex test as previously described (Iadecola et al., 1997), Higher scores indicate greater impairment. Twenty-four hours after induction of ischemia deficit scores did not differ between iNOS–/– and C57BL/6 at all ages (P > 0.05; Mann-Whitney U test) (
Irrespective of the mechanisms of the age-dependence of infarct volume and magnitude of iNOS-related neuroprotection, the present findings have important implications for studies of cerebral ischemia in mice. In experiments in which the effect of neuroprotective agents is investigated in mice, the age of control and experimental groups needs to be carefully matched. Similarly, the age of mutant mice and wild-type controls has to be carefully matched. This point is particularly important when mice are studied between 8 and 16 weeks of age, the critical period in which infarct volume increases. Furthermore, it may be necessary to determine the age at which maximal neuroprotective effects are observed. Age and weight of mice are also critical variables in experiments in which the MCA is occluded with an intraluminal filament (Hata et al., 1998).
In conclusion, we have demonstrated that the size of the infarct produced by permanent MCA occlusion in mice is dependent on the age of the animals. In addition, the magnitude of the neuroprotection observed in iNOS–/– mice is also age-dependent, being greatest at 4 to 8 weeks of age. Although the findings indicate that age is a critical variable in studies of cerebral ischemia in mice, they raise the possibility that the neuroprotective effects observed in certain mice mutants are age-dependent. Age is an important variable and must receive careful consideration in studies of cerebral ischemia and neuroprotection in mice.
