Abstract
Primary cultures of mouse cerebral cortical neurons (GABAergic) were incubated for 4 hours in media without glucose containing 1.0 mmol/L [U-13C]lactate in the absence or presence of 0.5 mmol/L glutamine. Redissolved, lyophilized cell extracts were analyzed by 13C nuclear magnetic resonance spectroscopy to investigate neuronal metabolism of lactate and by HPLC for determination of the total amounts of glutamate (Glu), γ-aminobutyric acid (GABA), and aspartate (Asp). The 13C nuclear magnetic resonance spectra of cell extracts exhibited multiplets for Glu, GABA, and Asp, indicating pronounced recycling of labeled tricarboxylic acid cycle constituents. There was extensive incorporation of 13C label into amino acids in neurons incubated without glutamine, with the percent enrichments being approximately 60% for Glu and Asp, and 27% for GABA. When 0.5 mmol/L glutamine was added to the incubation medium, the enrichments for Asp, Glu, and GABA were 25%, 35%, and 25%, respectively. This strongly suggests that glutamine is readily converted to Glu and Asp but that conversion to GABA may be complex. The observation that enrichment in GABA was identical in the absence and presence of glutamine whereas cycling was decreased in the presence of glutamine indicates that only C-2 units derived from glutamine are used for GABA synthesis, that is, that metabolism through the tricarboxylic acid cycle is a prerequisite for GABA synthesis from glutamine. The current study gives further support to the hypothesis that cellular metabolism is compartmentalized and that lactate is an important fuel for neurons in terms of energy metabolism and extensively labels amino acids synthesized from tricarboxylic acid cycle intermediates (Asp and Glu) as well as the neurotransmitter in these neurons (GABA).
Astrocytic metabolism may be regulated by neuronal signaling leading to production of lactate, which may be released and used by neurons as an energy source (Magistretti et al., 1993; Magistretti and Pellerin, 1996; Tsacopoulos and Magistretti, 1996). Indeed, astrocytes are able to release far more lactate compared with neurons (Walz and Mukerji, 1988; Dringen et al., 1993a; Sonnewald et al., 1995; Schousboe et al., 1997). In keeping with this, an efficient, saturable transport mechanism for uptake of lactate has been identified in cultured neurons (Dringen et al., 1993b; Nedergaard and Goldman, 1993). Moreover, further support to a regulated flux of lactate from astrocytes to neurons comes from the recent demonstration that the lactate dehydrogenase isozyme LDH-1, which preferentially catalyzes conversion of lactate to pyruvate, is highly expressed in neurons (Bittar et al., 1996).
The function of lactate as an energy fuel in neurons is further indicated by the finding that substituting lactate for glucose could ameliorate the cell degeneration observed during 3 hours of glucose deprivation (Izumi et al., 1994). Moreover, the decrease in excitatory postsynaptic potentials observed during glucose deprivation in rat hippocampal slices could be restored in a concentration-dependent manner by administration of lactate (Schurr et al., 1988; Izumi et al., 1994). Also, lactate has been demonstrated in humans to support brain function during hypoglycemia (Maran et al., 1994).
Whereas the role of lactate in the maintenance of brain function seems well documented, less is known about the ability of neurons to incorporate carbon from exogenously supplied lactate into neuroactive amino acids. The metabolic fate of individual C atoms can only be studied in detail using 13C-labeled compounds in combination with 13C nuclear magnetic resonance (NMR) spectroscopy. The incorporation of 13C into glutamate (Glu), aspartate (Asp), and γ-aminobutyric acid (GABA) was investigated in cultured cerebral cortical neurons by incubation in glucose-free media containing uniformly 13C-labeled lactate. Such neurons represent highly differentiated GABAergic cells expressing glutamate decarboxylase and vesicular GABA release (Yu et al., 1984a; Drejer et al., 1987; Belhage et al., 1993), essentially devoid of astrocytic contamination (Larsson et al., 1985). Because incorporation of 13C from lactate into the amino acids indirectly provides information about mitochondrial function, the studies also were performed to investigate a possible metabolic compartmentalization in the neurons. Although compartmentalization of Glu metabolism is well documented in the brain, less is known about this in individual cells, but recent information points to the possibility that cellular compartmentalization may exist (Schousboe et al., 1993). Moreover, to investigate the significance of glutamine, an important GABA precursor (Reubi et al., 1978; Battaglioli and Martin, 1990; Sonnewald et al., 1993), cells were additionally incubated with nonlabeled glutamine with [U-13C]lactate. This not only provides further information about metabolic compartmentalization but allows a detailed analysis of the metabolic pathways involved in conversion of glutamine to Glu and GABA, information which cannot be deduced from classic studies of conversion of [14C]glutamine to these amino acids (Shank and Aprison, 1977; Bradford et al., 1978; Yu et al., 1984b).
EXPERIMENTAL PROCEDURES
Materials
Pregnant mice (16 days' gestation) were obtained from the animal quarters of the Panum Institute, University of Copenhagen. Plastic tissue culture dishes were purchased from Nunc A/S (Roskilde, Denmark), fetal calf serum from Seralab, Ltd. (Sussex, U.K.), and culture medium from GIBCO BRL, Life Technologies (Roskilde, Denmark). Poly-
Cell cultures
Cerebral cortical neurons (GABAergic) were isolated and cultured as described by Hertz et al. (1989). Cells were seeded in poly-
Protein and amino acid determinations
Cellular protein was determined according to Lowry et al. (1951) in the pellets after dissolving in 1 mol/L KOH at 20°C for 24 hours. Bovine serum albumin was used as the standard. Amino acids in the ethanol extracts (aliquots taken before lyophilization) were quantified by HPLC analysis after derivatization with o-phthaldialdehyde (Geddes and Wood, 1984). The HPLC analysis was performed using a Varian solvent delivery system 9010, autosampler 9100, and computer system 4.02.
Nuclear magnetic resonance spectroscopy
Proton-decoupled 125.7-MHz 13C NMR spectra were obtained on a Bruker DRX-500 spectrometer. Spectra were accumulated using a 35 degree pulse angle, 31 KHz spectral width, with 64 K data points. The acquisition time was 1.049 seconds, and an additional relaxation delay of 5 seconds was used. The number of scans was typically 3000. Some spectra also were broad-band decoupled only during acquisition to avoid nuclear Overhauser effects. From the two sets of spectra, factors for the nuclear Overhauser effects of different atoms were obtained and applied to all spectra. Inversion recovery experiments were performed to obtain relaxation (T1) values, but correction was not necessary. The total amount of 13C in the resonance of a particular metabolite was calculated using dioxane as an internal standard, as described by Badar-Goffer et al. (1990). Percent enrichment was calculated from the amount of 13C in the resonances and the total amounts of the compounds present, which was determined by HPLC. Homonuclear 13C−13C spin coupling patterns for Glu are described by Cerdan et al (1990).
RESULTS
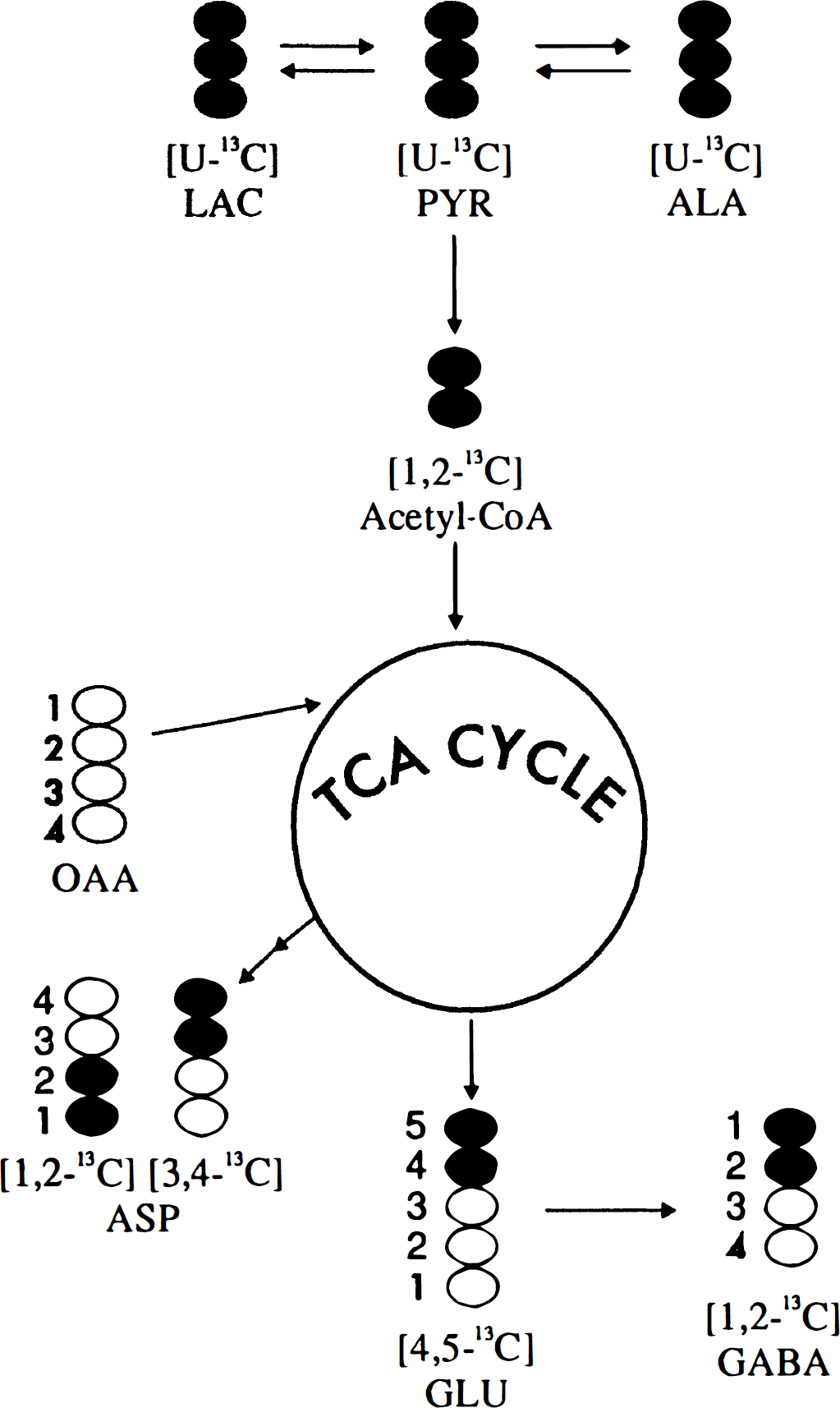
In cerebral cortical neurons (GABAergic) incubated for 4 hours with 1.0 mmol/L [U-13C]lactate, an extensive incorporation of 13C in the amino acids Asp, Glu, and GABA was observed, as demonstrated by the NMR spectrum shown in Fig. 1. The metabolic pathways involved in the conversion of lactate to these amino acids are schematically represented in Fig. 2. [U-13C]Lactate can enter the tricarboxylic acid (TCA) cycle by conversion to [1,2-13C]acetyl-CoA via [U-13C]pyruvate, which also can give rise to alanine formation. However, [13C]alanine was not observed in the spectra. During the first turn of the TCA cycle, an unlabeled molecule of oxaloacetate is condensed with [1,2-13C]acetyl-CoA, subsequently leading to synthesis of [4,5-13C]glutamate and [1,2-13C]GABA (Fig. 2). Because of the symmetry of the succinate molecule, two isotopomers of Asp are possible, namely [1,2-13C]aspartate and [3,4-13C]aspartate (Fig. 2). These isotopomers give rise to characteristic doublets (Fig. 3), and the amounts of 13C in these metabolites generated after the first turn of the cycle and their percent enrichment are listed in Table 1. During subsequent turns of the cycle, labeled oxaloacetate can be condensed with labeled or unlabeled acetyl-CoA, leading to production of different isotopomers with label in GABA C-4 or C-3 and Glu C-2 or C-3. Two vicinal 13C atoms give rise to a doublet in the spectrum, and the J coupling constant differs depending on the substituents on the respective carbon atoms. A carboxylic acid group gives rise to approximately 50-Hz splitting, whereas a carbon atom with protons gives rise to approximately 30-Hz splitting. In isotopomers with three vicinal 13C atoms, depending on the sizes of the respective coupling constants, the middle 13C is seen as a quartet or an apparent triplet, the latter resulting from two overlapping doublets (C-3 in Glu and GABA, Figs. 1 and 3). Labeled acetyl-CoA (or unlabeled from fatty acid or amino acid catabolism) can enter the TCA cycle in subsequent turns, and scrambling of label from the symmetrical succinate molecule causes multiple labeling patterns in Glu, GABA, and Asp (Fig. 3). Isotopomers, which show the same doublets as [4,5-13C]glutamate, [1,2-13C]GABA, [1,2-13C]aspartate, and [3,4-13C]aspartate, can be formed during subsequent turns of the TCA cycle, but their contribution probably is minimal. Detailed analysis of splitting patterns in Glu C-3 and C-2 and GABA C-3 and C-4 revealed that some labeled metabolites stayed in the TCA cycle for at least three turns, where labeled acetyl-CoA was used for condensation in the citrate synthase reaction. The total amount of 13C and percent enrichment obtained from repeated cycling are given in Table 1. The enrichments in Asp and Glu were of the same magnitude and much higher than that in GABA (Table 1). Carbon atom C-2 in GABA, which is equivalent to Glu C-4 and thus labeled during the first turn of the cycle, is the carbon atom that showed the highest enrichment (27%) which, however, was substantially lower than that of the precursor, Glu C-4 (61%). The enrichment in Asp C-3 (63%) was similar to this latter value. The enrichments of the doublets of [4,5-13C]glutamate, [3,4-13C]aspartate, and [1,2-13C]GABA, representing the first turn of the TCA cycle, were 28%, 20%, and 14%, respectively. The total enrichment in Glu C-3 (45%) and GABA C-4 (17%), representing subsequent turns of the TCA cycle, exhibited the same tendency to a higher incorporation in Glu than in GABA as that seen for the isotopomers generated in the first turn.
The 13C nuclear magnetic resonance (NMR) spectrum of metabolites from ethanol extracts of cultured cerebral cortical neurons incubated for 4 hours in the presence of [U-13C]lactate (for details, see Experimental Procedures). Asp, aspartate; GABA, 7-aminobutyrate; Glu, glutamate; Lac, lactate. A simplified scheme of the tricarboxylic acid (TCA) cycle showing the conversion of [U-13C]lactate to [1,2-13C]acetyl-CoA via pyruvate (Pyr) and further metabolism through the first turn of the tricarboxylic acid (TCA) cycle. This gives rise to [4,5-13C]glutamate and [1,2-13C]GABA. Two isotopomers are possible for aspartate (Asp): [1,2-13C]aspartate and [3,4-13C]aspartate. GABA, γ-aminobutyrate; Glu, glutamate; Lac, lactate; OAA, oxaloacetate. Expanded splitting patterns of aspartate C-3, γ-aminobutyrate (GABA) C-2, glutamate C-4, and glutamate C-3 from the 13C NMR spectrum shown in Fig. 1. The circles show the isotopomers giving rise to the splitting patterns from the first, second, and third turn, with either labeled or unlabeled acetyl-CoA entering the TCA cycle in the second and third turn. J coupling constants: Aspartate C-3, J34: 50.5 Hz, J23: 36.7 Hz. GABA C-2, J12: 51.2 Hz, J23: 34.4 Hz. Glutamate C-4, J45: 51.3 Hz, J34: 35.2 Hz. Glutamate C-3, J23: 34.4 Hz, J34: 35.2 Hz. Total amounts (nmol/mg protein) of 13C and percent enrichment, shown in parentheses, of amino acids in extracts of cultured cortical neurons after incubation in 1.0 mmol/L [U-13C]lactate in the absence or presence of 0.5 mmol/L glutamine Cultured cerebral cortical neurons prepared as described in Experimental Procedures were incubated for 4 hours in astrocytic culture medium (DMEM) containing 1.0 mmol/L [U-13C]lactate in the presence or absence of 0.5 mmol/L glutamine (Gln). After the incubation, the cells were extracted with 70% (v/v) ethanol and further analyzed by HPLC and 13C NMR spectroscopy as detailed in Experimental Procedures. First turn represents the doublets [4,5-13C]glu, [1,2-13C]GABA, and [3,4-13C]asp. Σ is the sum of incorporated 13C in the different positions. Results are averages ± SD with numbers of experiments shown in parentheses. Results obtained with added glutamine were analyzed versus the control without glutamine and statistical significance was determined by analysis of variance. Asterisks indicate the level for significance P < 0.025 P ≤ 0.001).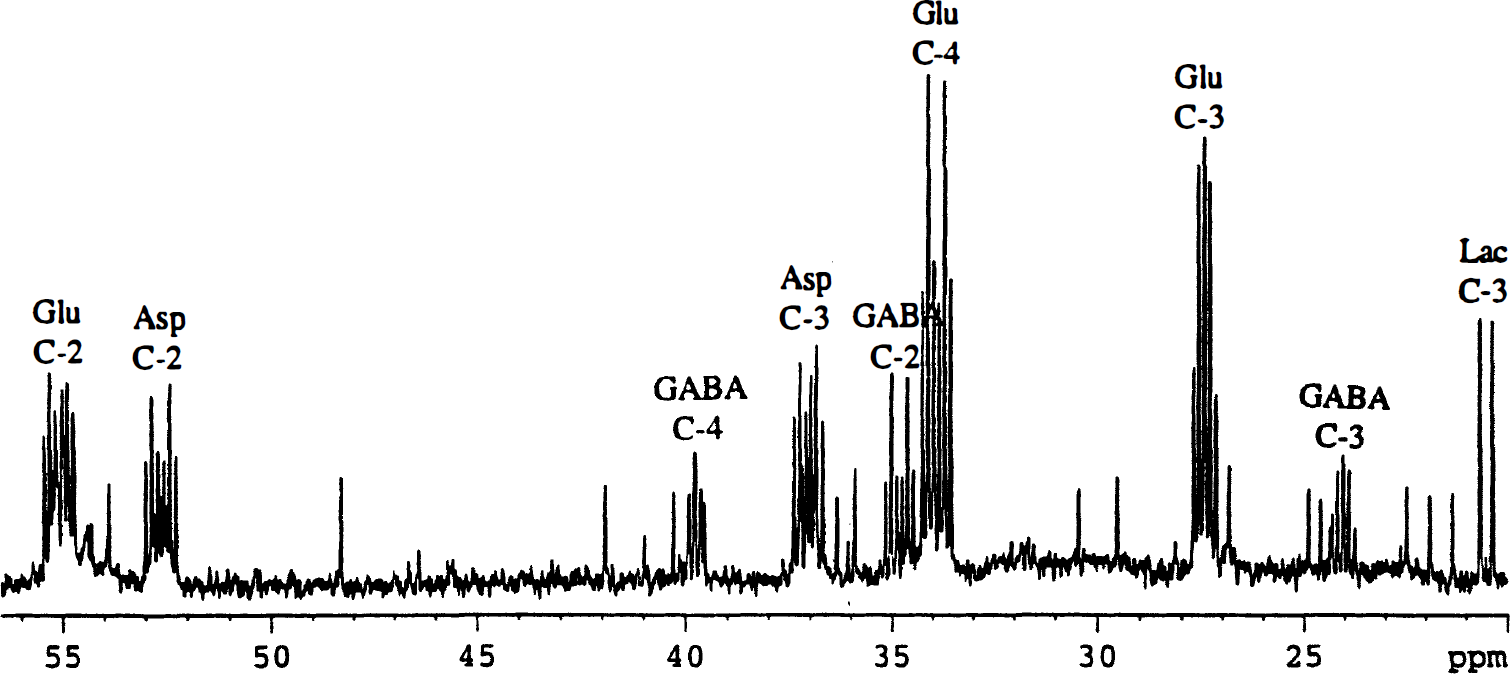
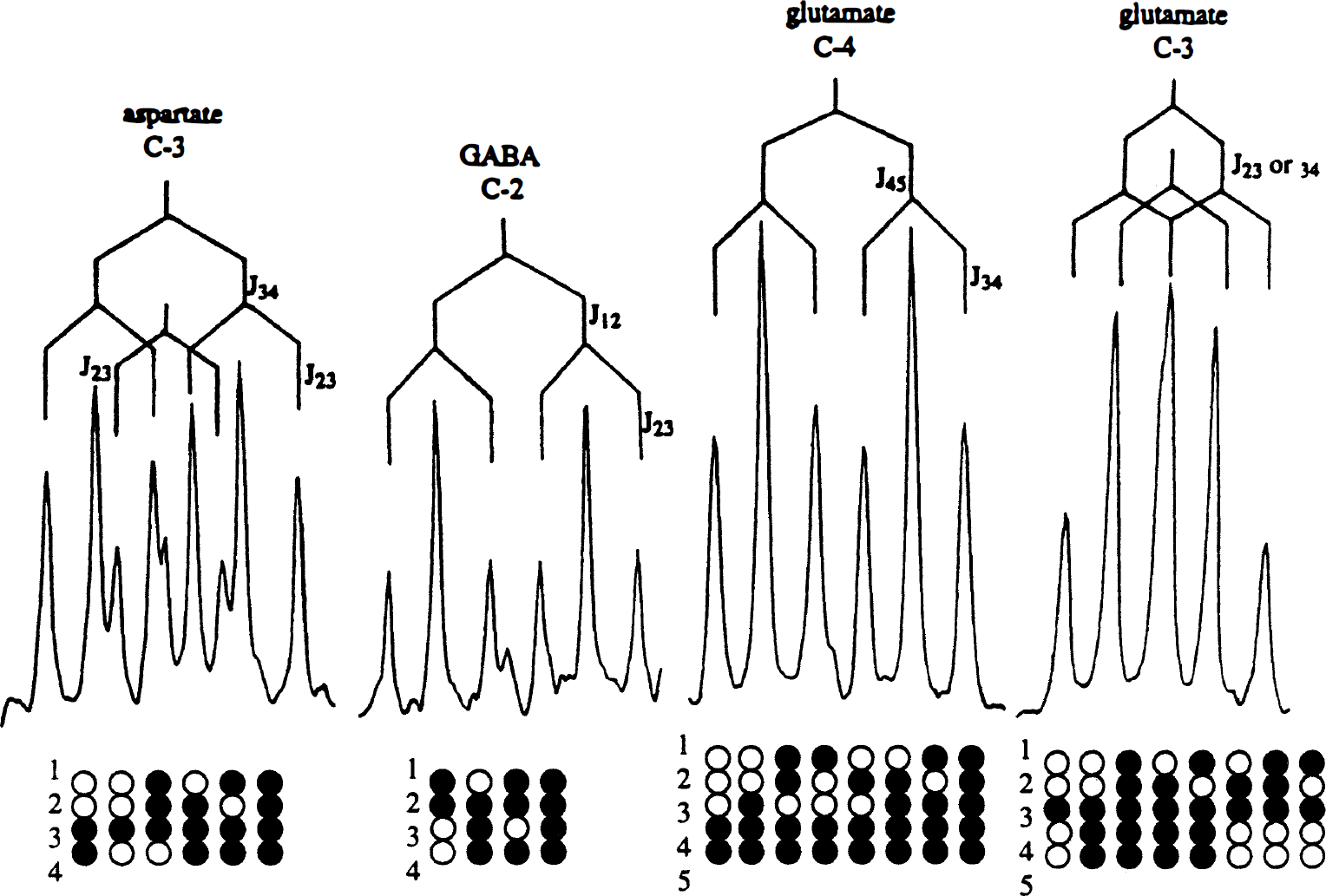

Because of the symmetry of succinate, the label derived from [U-13C]lactate is scrambled before it reaches oxaloacetate and subsequently Asp (Fig. 3), but this is not the case for Glu and GABA, which are synthesized from a TCA cycle precursor generated before this step. The amount of label in C-4 representing [4,5-13C]-glutamate (10 nmol/mg protein) was twice that found in C-3 of [3,4-13C]aspartate (5 nmol/mg protein), thus showing that Glu and Asp were labeled to a similar extent in the first turn.
Pool sizes (nmol/mg protein) of GABA, glutamate, and aspartate in cultured cerebral cortical neurons after 4 hours of incubation in 1.0 mmol/L [U-13C]lactate with or without 0.5 mmol/L glutamine
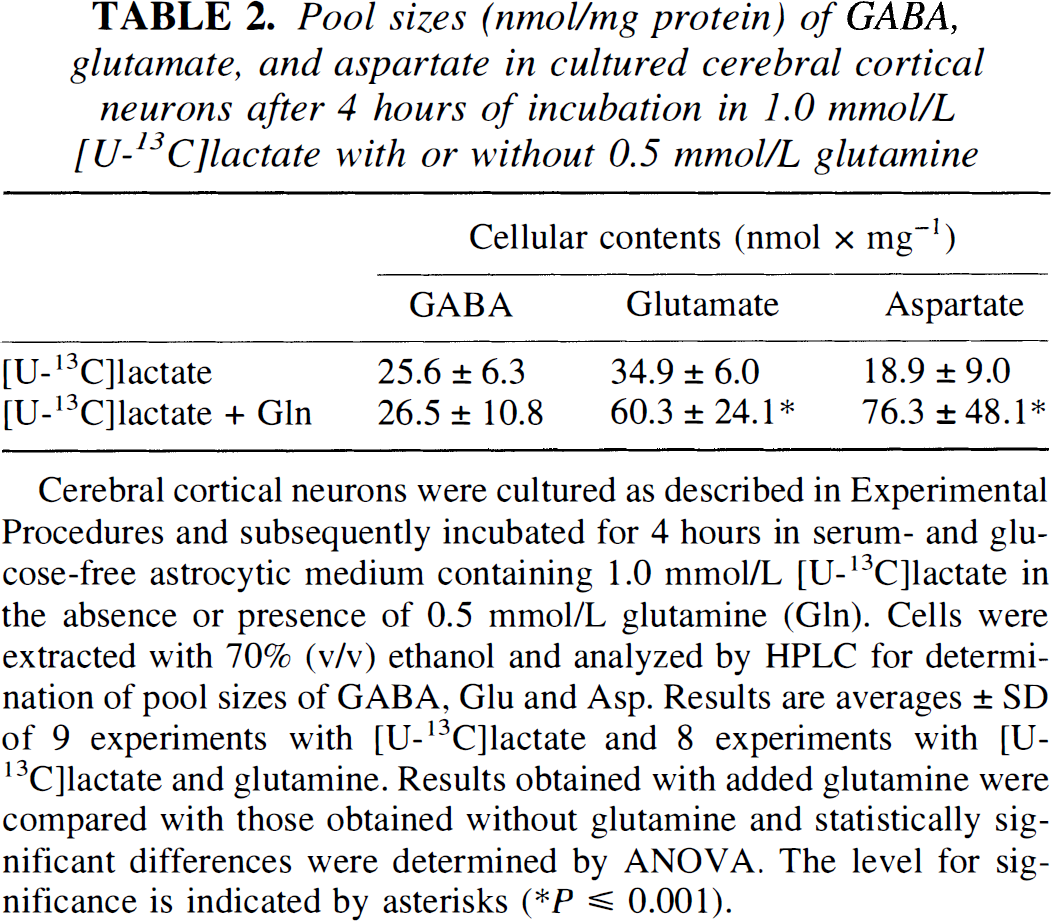
Cerebral cortical neurons were cultured as described in Experimental Procedures and subsequently incubated for 4 hours in serum- and glucose-free astrocytic medium containing 1.0 mmol/L [U-13C]lactate in the absence or presence of 0.5 mmol/L glutamine (Gln). Cells were extracted with 70% (v/v) ethanol and analyzed by HPLC for determination of pool sizes of GABA, Glu and Asp. Results are averages ± SD of 9 experiments with [U-13C]lactate and 8 experiments with [U-13C]lactate and glutamine. Results obtained with added glutamine were compared with those obtained without glutamine and statistically significant differences were determined by ANOVA. The level for significance is indicated by asterisks
P ≤ 0.001).
Ratios representing cycling in the TCA cycle for intermediates used for synthesis of GABA, glutamate, and aspartate synthesized in cultured cerebral cortical neurons incubated in medium containing 1.0 mmol/L [U-13C]lactate in the absence or presence of 0.5 mmol/L glutamine
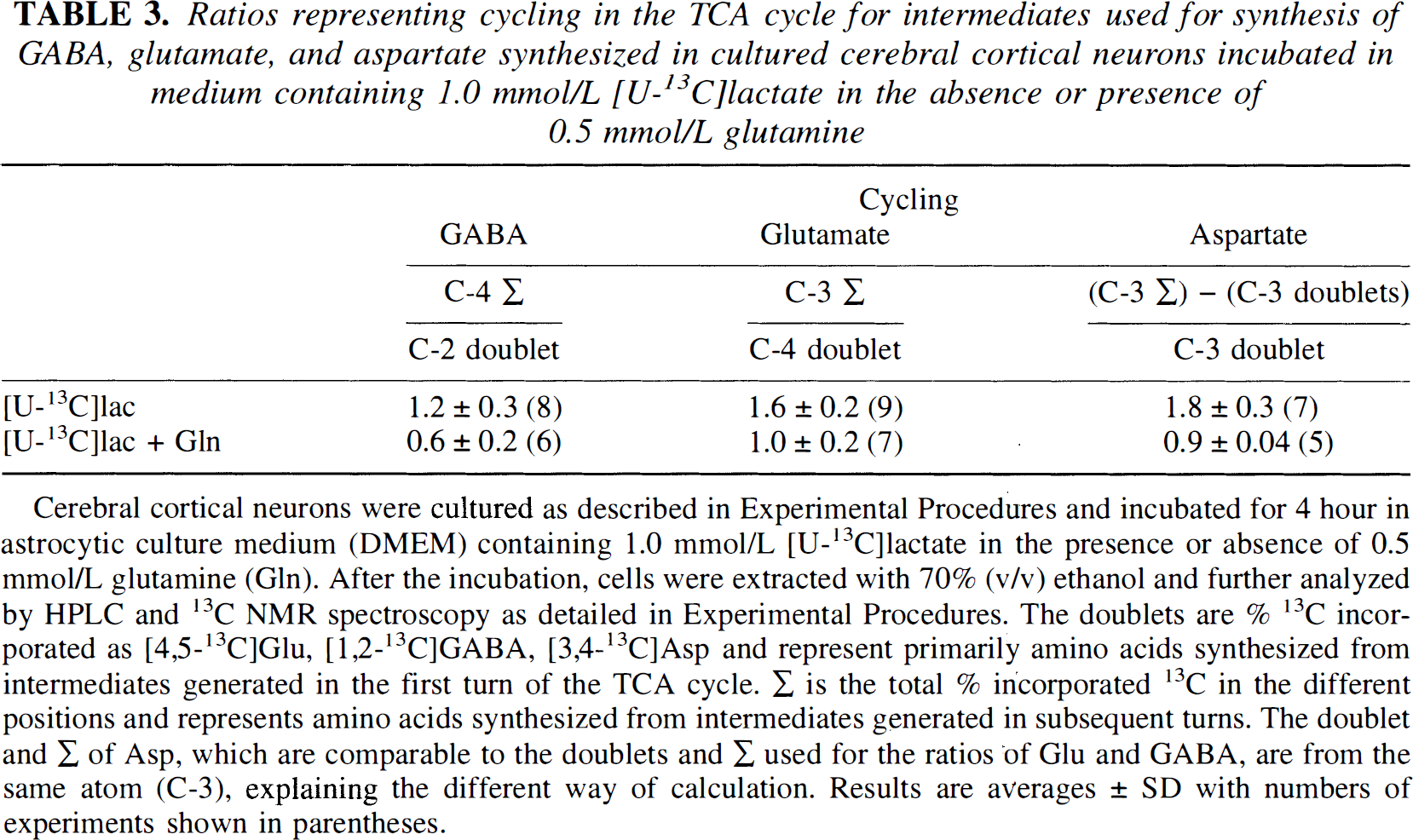
Cerebral cortical neurons were cultured as described in Experimental Procedures and incubated for 4 hour in astrocytic culture medium (DMEM) containing 1.0 mmol/L [U-13C]lactate in the presence or absence of 0.5 mmol/L glutamine (Gln). After the incubation, cells were extracted with 70% (v/v) ethanol and further analyzed by HPLC and 13C NMR spectroscopy as detailed in Experimental Procedures. The doublets are % 13C incorporated as [4,5-13C]Glu, [1,2-13C]GABA, [3,4-13C]Asp and represent primarily amino acids synthesized from intermediates generated in the first turn of the TCA cycle. Σ is the total % incorporated 13C in the different positions and represents amino acids synthesized from intermediates generated in subsequent turns. The doublet and Σ of Asp, which are comparable to the doublets and Σ used for the ratios of Glu and GABA, are from the same atom (C-3), explaining the different way of calculation. Results are averages ± SD with numbers of experiments shown in parentheses.
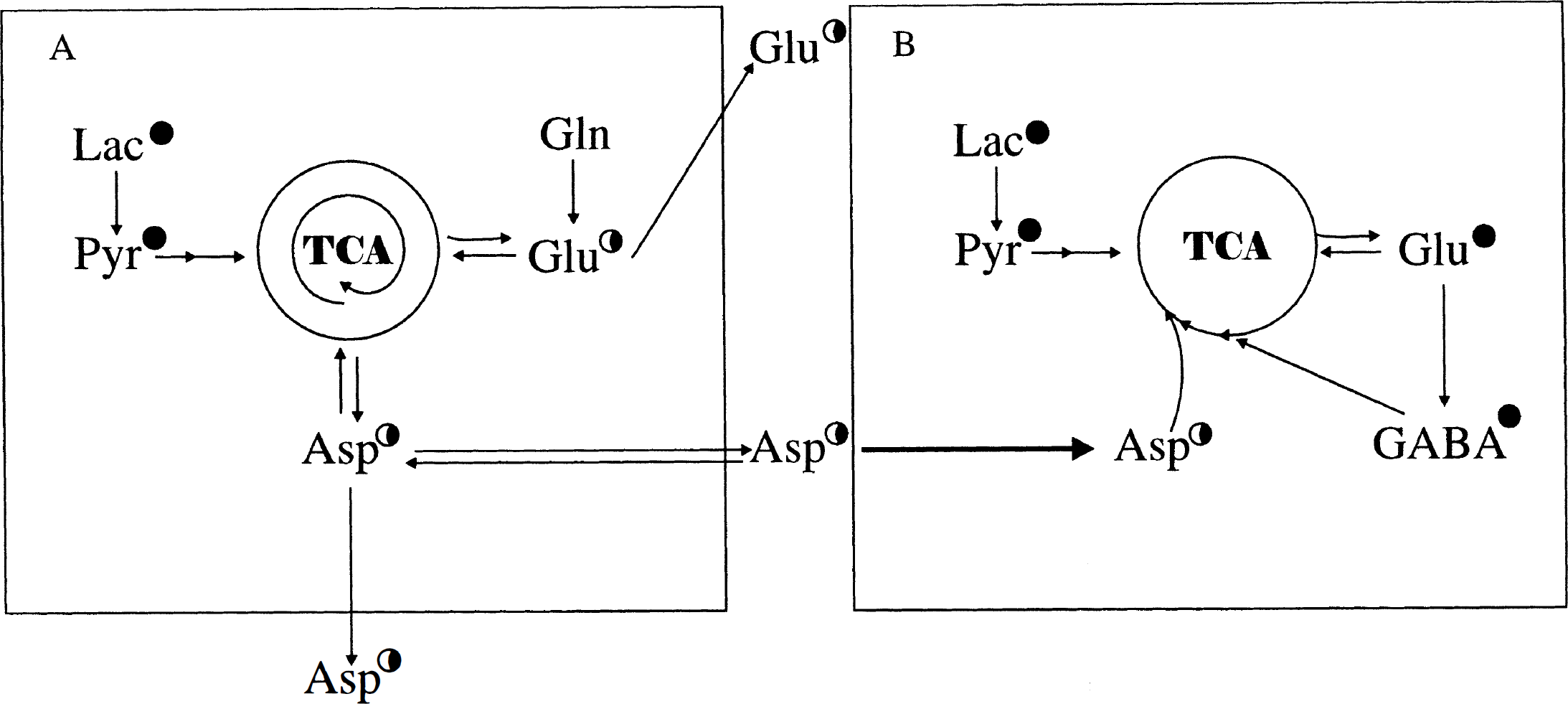
As seen from the cellular concentrations of Glu, Asp, and GABA presented in Table 2, exposure to glutamine led to a net increase in the contents of the two former amino acids but not in that of GABA. Moreover, the cycling ratios calculated in Table 3 indicate that GABA may be derived from TCA cycle constituents originating from a cycle, with a cycling pattern different from that giving rise to synthesis of Glu and Asp. To better understand and discuss these findings, schematic representations of different TCA cycle compartments are provided in Figs. 4 and 5. Figure 4 shows the metabolic events occurring in the absence of glutamine, and it indicates the existence of two TCA cycles with different cycling patterns of intermediates (A and B) where synthesis of GABA from labeled intermediates is preferentially associated with the cycle from which intermediates leave rapidly (Fig. 4B; Table 3), whereas no or only little GABA synthesis is associated with the cycle in which intermediates remain longer (Fig. 4A, Table 3). In the presence of glutamine (Fig. 5A and B), two cycles operate in which intermediates remain for longer (Fig. 5A) and shorter (Fig. 5B) periods of time, respectively (Table 3), and where GABA synthesis still is associated with the TCA cycle from which intermediates leave relatively rapidly (Fig. 5B). Under these conditions, a net synthesis of Glu and Asp (Table 2) occurs as shown in Fig. 5A. It is further assumed that Asp synthesized in the cycle in which metabolites remain for a longer period of time (Fig. 5A) may be transferred to the cycle in which metabolites stay for a shorter period of time (Fig. 5B). However, this Asp is labeled only to a moderate extent, as seen from Table 1. In accordance with Table 3, the TCA cycles in Fig. 4A and B indicate more cycling of intermediates than the cycles shown in Fig. 5A and B when cells are incubated in the presence of glutamine.
An illustration of a two-compartment model explaining the metabolism of lactate (Lac) in the absence of glutamine in cultured cerebral cortical neurons. An illustration of a two-compartment model explaining the metabolism of lactate (Lac) in the presence of 0.5 mmol/L glutamine (Gln) in cultured cerebral cortical neurons. 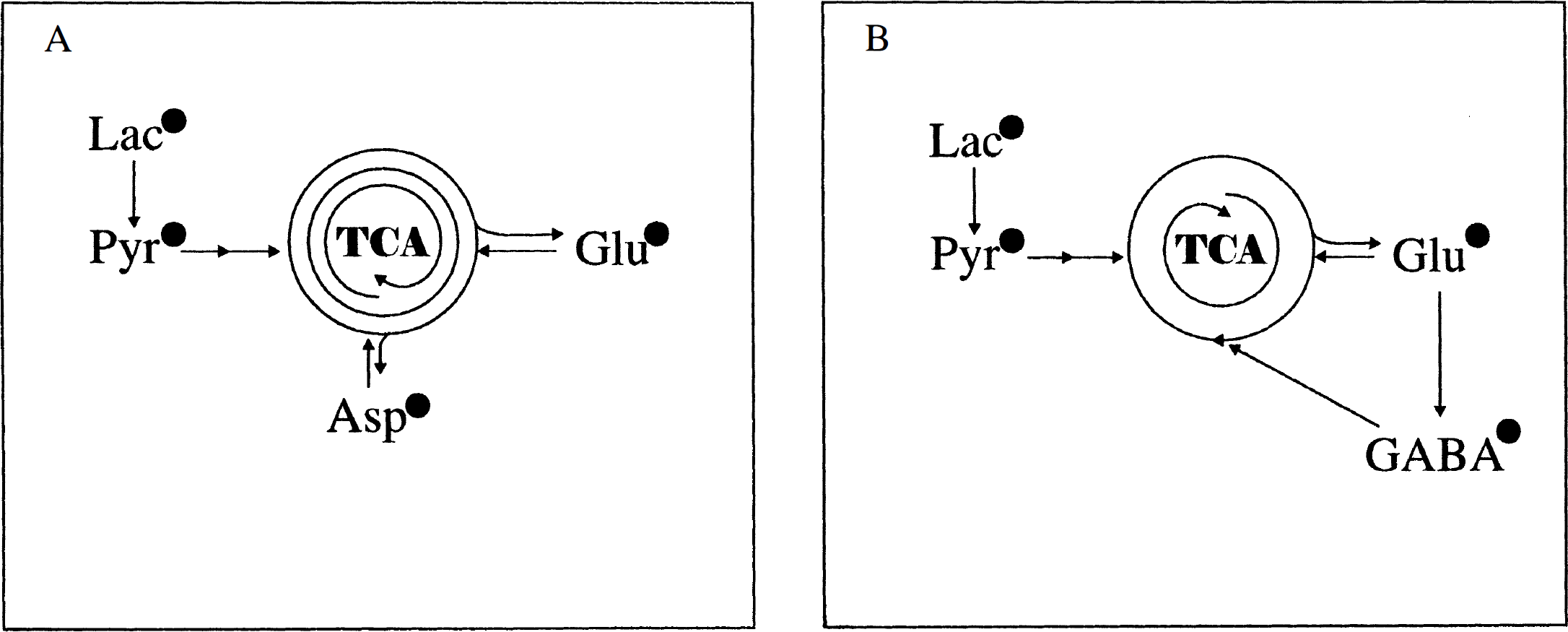
DISCUSSION
Lactate metabolism
The importance of lactate metabolism in the adult brain still is controversial, although the issue has attracted renewed interest because of the in vivo 1H NMR demonstrations of lactate production during functional activation (Prichard et al., 1991). In this context, it is interesting that cultured cerebral cortical neurons have been shown to use lactate far better than glucose as a substrate for oxidative metabolism (Peng et al., 1994). Whether carbon from lactate can be incorporated into neurotransmitter amino acids like Glu, GABA, and Asp has been studied in rat brain. Dombrowski and coworkers (1989) have shown that in brain extracts, incorporation of 14C in amino acids could be seen after intraperitoneal injection of [14C]lactate in 6-day-old animals. The specific activity of glutamine and Glu in the brain extracts indicated that carbon derived from lactate was entering the “large glutamate compartment” commonly accepted to represent the neuronal pool (Berl and Clarke, 1983). However, lactate served as the primary substrate for energy metabolism at the age of 6 days (65% to 75% of total carbon dioxide production), whereas in 15-day-old animals, lactate contributed with only 23% of the carbon dioxide production. In the current study, the incorporation of carbon atoms from [U-13C]lactate into amino acids showed that lactate in cerebral cortical neurons can supply carbon atoms for synthesis of 2-oxoglutarate, the precursor for Glu and GABA, and of oxaloacetate, the precursor for Asp. However, no net synthesis of amino acids derived from TCA cycle intermediates is possible in neurons, since only astrocytes contain the anaplerotic enzyme pyruvate carboxylase (Yu et al., 1983; Shank et al., 1985; Cesar and Hamprecht, 1995). Thus, neurons have to replenish the pool of the above-mentioned amino acids from precursors such as TCA cycle intermediates or glutamine, both of which are released by astrocytes (Schousboe et al., 1997).
Effect of glutamine on lactate metabolism
Although glutamine has repeatedly been shown to be an important precursor for neurotransmitter Glu and GABA (Shank and Aprison, 1977; Bradford et al., 1978; Reubi et al., 1978; Yu et al., 1984a; Shank et al., 1989), no information is available concerning the exact metabolic pathways used to synthesize GABA. The current results may provide some insight into this question. A large increase in the intracellular amounts of unlabeled Asp and Glu was observed in the presence of 0.5 mmol/L glutamine and [U-13C]lactate in the incubation medium. Since glutamine is the direct precursor of Glu, an increased Glu concentration might a priori be expected. Formation of Asp from glutamine, on the other hand, necessarily involves the TCA cycle. The simplest pathway from intramitochondrial Glu to intramitochondrial Asp is by transamination or oxidative deamination and conversion in the TCA cycle to oxaloacetate, which subsequently is transaminated to Asp. Aspartate may cross the inner mitochondrial membrane in exchange with Glu and subsequently diffuse across the permeable outer mitochondrial membrane into the cytosol (Hertz et al., 1992). The finding that labeling of Glu and Asp from [U-13C]lactate was decreased by the presence of glutamine indicates that this sequence of reactions indeed is responsible for conversion of glutamine to Asp. The formation of Asp from Glu in the TCA cycle would be a source of metabolic energy (Erecinska et al., 1988; Hertz et al., 1992), and it has been proposed that the resulting energy production may partly explain the increased formation of Asp in the brain in vivo as well as in synaptosomes under hypoglycemic or glucose-free conditions (Engelsen et al., 1986; Erecinska et al., 1988). The lactate concentration used in the current study corresponding to 0.5 mmol/L glucose is not strictly a hypoglycemic condition (see Engelsen et al., 1986), and the Asp content seen in the presence of glutamine is comparable with that previously observed in cortical neurons incubated in 6 mmol/L glucose and 0.25 mmol/L glutamine (Westergaard et al., 1995). It is therefore unlikely that the relatively high Asp content is a direct consequence of the low lactate concentration in the incubation medium. Also, the average number of turns of the cycle in which the intermediates remained before being converted to Glu or Asp was decreased by glutamine, indicating that the lack of an anaplerotic process in the neurons was no longer a limiting factor. This reflects the fact that glutamine in these neurons is an energy substrate that additionally can replenish the TCA cycle (Yu et al., 1984b). No increase was detectable in the GABA content of the cells in the presence of [U-13C]lactate and glutamine, indicating that glutamine, under these experimental conditions, was not a prominent direct precursor for GABA synthesis. Although glutamine was not efficient as a direct precursor for GABA, it had a large effect on the number of turns that precursors for GABA stayed in the TCA cycle. This means that conversion of glutamine to GABA proceeds through the TCA cycle (Fig. 5A and B), a pathway that could not be foreseen by the use of relabeled precursors. A similar decrease in cycling was observed for Glu and Asp precursors, evidencing that 2-oxoglutarate derived from glutamine via Glu entered the TCA cycle. GABA synthesis from glutamine carbon atoms that have been in the TCA cycle also has been detected in mouse brain (Hassel et al., 1995) and points toward an important regulatory function of mitochondria.
Compartmentalization
The concept of intracellular compartmentalization in brain cells still is controversial, but an increasing amount of experimental evidence seems to point in this direction (Hertz et al., 1992; Schousboe et al., 1993). In the current study, several observations indicate the existence of separated intracellular metabolic compartments. The dilution of incorporation of 13C atoms was seen in Asp and Glu, but not in GABA, when glutamine was added to the incubation medium, supporting a hypothesis of the existence of at least two pools of Glu, one of which is preferentially used for the synthesis of GABA and the other one for synthesis of Asp. The decrease in cycling ratios in the presence of glutamine points to the same conclusion, since cycling of metabolites used for synthesis of GABA decreased more than the corresponding cycling of metabolites used for synthesis of Glu. It was also shown by Westergaard and others (1995) that GABA synthesis from [U-13C]glutamine involved the TCA cycle to a larger extent than synthesis directly from exogenously supplied Glu. The finding that the increased net synthesis of Glu and Asp seen in the presence of glutamine did not affect the total amount of label incorporated into Asp and Glu indicates compartmentalization as well. However, the cycling ratios for these amino acid precursors were affected by glutamine. Surprisingly, labeled alanine from [U-13C]lactate was not detected in the neurons. Since the equilibrium constant for the alanine aminotransferase reaction is close to unity (Krebs, 1953), a possible explanation for this observation is either compartmentalization of pyruvate metabolism or a rapid formation of acetyl-CoA from pyruvate (Fitzpatrick et al., 1988), preventing its conversion to alanine.
To unify the current results, it is necessary to postulate the existence of at least two TCA cycles, as shown in Figs. 4 and 5. In the absence of glutamine, one TCA cycle in which intermediates remain for several turns is in exchange with the Asp pool and one of the Glu pools, and another cycle in which intermediates stay for fewer turns exchanges with another Glu pool from which GABA is derived. When glutamine was present, it gained access to the Asp-forming TCA cycle, as indicated by the increase in the Asp concentration and the decrease in its enrichment. The added glutamine had, however, no direct access to the TCA cycle that is in exchange with the GABA pool, which means that Asp or another C-4 unit is exchanged between the two TCA cycles. It could be argued that Glu also could have this function, but that would seem incompatible with the existence of distinctly different Glu pools. Several possibilities exist for cellular compartmentalization of TCA cycle metabolism. Different types of mitochondria could be present within the same cell, or different TCA cycles could be operative and separated within the same mitochondrion. Support for the latter proposal is obtained from a NMR study by Lopez-Beltran and associates (1996) of mitochondrial viscosity showing that free diffusion of metabolites within mitochondria is unlikely because of high viscosity.
