Abstract
The aim of the present work was to study potential disturbances in metabolism and interactions between neurons and glia in the lithium-pilocarpine model of temporal lobe epilepsy. Rats chronically epileptic for 1 month received [1-13C]glucose, a substrate for neurons and astrocytes, and [1,2-13C]acetate, a substrate for astrocytes only. Analyses of extracts from cerebral cortex, cerebellum, and hippocampal formation (hippocampus, amygdala, entorhinal, and piriform cortices) were performed using 13C and 1H nuclear magnetic resonance spectroscopy and HPLC. In the hippocampal formation of epileptic rats, levels of glutamate, aspartate, N-acetyl aspartate, adenosine triphosphate plus adenosine diphosphate and glutathione were decreased. In all regions studied, labeling from [1,2-13C]acetate was similar in control and epileptic rats, indicating normal astrocytic metabolism. However, labeling of glutamate, GABA, aspartate, and alanine from [1-13C]glucose was decreased in all areas possibly reflecting neuronal loss. The labeling of glutamine from [1-13C]glucose was decreased in cerebral cortex and cerebellum and unchanged in hippocampal formation. In conclusion, no changes were detected in glial—neuronal interactions in the hippocampal formation while in cortex and cerebellum the flow of glutamate to astrocytes was decreased, indicating a disturbed glutamate—glutamine cycle. This is, to our knowledge, the first study showing that metabolic disturbances are confined to neurons inside the epileptic circuit.
Introduction
Temporal lobe epilepsy (TLE), which is the most common form of intractable epilepsy, is often associated with hippocampal sclerosis, involving neuronal loss, mossy fiber sprouting, and astrogliosis (Babb et al, 1984; Engel Jr 1996). These lesions can also be found in extrahippocampal structures, such as in the parahippocampal cortices, amygdala, and thalamus (Cendes et al, 1993; Arnold et al, 1996; Chassoux et al, 2004). The clinical and pathophysiologic patterns of TLE can be reproduced in adult rats after prolonged status epilepticus (SE), induced by the cholinergic agonist, pilocarpine (Turski et al, 1989) or the glutamatergic agonist, kainic acid (Sperk, 1994). These two validated models of TLE are similar to the human disease in development of seizures, in its pathology and in its pharmalogical response. Pilocarpine or lithium-pilocarpine (Li-pilo)-induced SE, which represents the acute phase of the disease, is characterized by a muscarinic response followed rapidly by a glutamatergic generalization, leading to a large increase in calcium concentration, mitochondrial failure, and extensive necrotic neuronal death, mostly in the hippocampus (CA1, CA3, and hilus of the dentate gyrus), amygdala, thalamus, piriform, and entorhinal cortices (Turski et al, 1989; Motte et al, 1998; Dube et al, 2001). The subsequent latent phase is characterized by repair processes, such as astroglial response, neurogenesis, sprouting, and circuit reorganization, leading to the formation of a hyperexcitable circuit in lesioned limbic regions underlying the expression of spontaneous recurrent seizures (SRS) (Turski et al, 1989). In this hyperexcitable circuit, even limited changes in release of glutamate or GABA could lead to seizure activity.
13C Nuclear magnetic resonance spectroscopy (NMRS) is an excellent tool for analyzing specific neuronal and astrocytic metabolism and interactions (Sonnewald and Kondziella, 2003). Because astrocytes and neurons express different enzymes and transporters, it is possible to differentiate metabolic events taking place in astrocytes and neurons, respectively, after the administration of 13C labeled substrates. Acetate and glucose are both precursors for acetyl CoA, which enters the tricarboxylic acid (TCA) cycle. Acetate is selectively taken up by astrocytes that express a specialized transport system, which is absent or less active in neurons (Waniewski and Martin, 1998). Acetyl CoA derived from glucose is metabolized more in the neuronal than in the astrocytic TCA cycle (Qu et al, 2000). It should be remembered that glucose enters both neurons and astrocytes (Nehlig et al, 2004), and lactate can be shuttled from astrocytes to neurons (Serres et al, 2004). To what extent this shuttle is operative remains a matter of debate. Thus, by simultaneous injection of [1-13C]glucose and [1,2-13C]acetate and NMRS analysis of brain extracts, information about neuronal and astrocytic metabolism can be obtained in the same animal (Taylor et al, 1996).
It has been shown that there is interictal hypometabolism in TLE patients as well as in epileptic animals, which does not directly correlate with neuronal loss (Ryvlin et al, 1991; O'Brien et al, 1997; Foldvary et al, 1999; Dube et al, 2001). Thus, the regulation of metabolic pathways is most likely disturbed in the epileptic brain. Therefore, astrocytic-neuronal metabolism and interactions were studied in the interictal phase of the chronic period in the rat Li-pilo model, in the present study. Epileptic rats were injected with [1-13C]glucose and [1,2-13C]acetate simultaneously to analyze neuronal and glial metabolism. 13C NMRS, 1H NMRS, and HPLC analyses were performed on extracts from three different brain regions, namely the hippocampal formation (hippocampus, dentate gyrus, amygdala, piriform, and entorhinal cortices), cerebral cortex, and cerebellum.
Materials and methods
Animals
Seventeen male Sprague—Dawley rats (Janvier Breeding Center, Le Genest-St-Isle, France), weighing 260 to 300 g, were used. All animals were maintained under standard laboratory conditions on a 12/12-h light/dark cycle, with free access to food and water. All animal experimentation was performed in accordance with the rules of the European Communities Council Directive of November 24, 1986 (86/609/EEC), and the French Department of Agriculture (License Number 67-97).
Induction of Status Epilepticus
Lithium chloride (LiCl) (127 mg/kg, Sigma-Aldrich, St Louis, MO, USA) was injected intraperitoneally to both the Li-pilo group (n = 10) and the Li-saline control group (n = 7). Status epilepticus was induced by subcutaneous injection of pilocarpine hydrochloride (25 mg/kg, Sigma-Aldrich) 20 h after LiCl treatment. The animals received 1 mg/kg methylscopolamine (Sigma-Aldrich) 30 mins before the convulsant, and then again 2 h after SE, to reduce peripheral effects of pilocarpine administration. To improve survival, the rats were administered an intramuscular injection of diazepam 2 h after the onset of SE (2.5 mg/kg diazepam, Valium, Roche, France) and then again 6 h later (0.75 mg/kg). Of the 10 rats subjected to Li-pilo SE, 2 died within the first 24 h after SE.
To ensure that all the rats used in the experiment were chronically epileptic, the appearance of SRS was monitored by video recording. The rats were filmed 10 h per day, 7 days a week. All rats having received pilocarpine displayed SRS within a month. The latency to the occurrence of the first SRS in Li-pilo rats was 22.3±10.5 days (n = 8).
At approximately 1 month after the onset of SRS (24±4 days) or the equivalent time in control animals, Li-saline and Li-pilo rats were administered an intraperitoneal injection of sodium [1,2-13C]acetate and [1-13C]glucose, 3 mmol/kg (99% 13C enriched, Cambridge Isotope Laboratories, Woburn, MA, USA). At this time, Li-saline rats weighed 482±42 g and Li-pilo rats 448±12 g. This difference was not statistically significant. Fifteen minutes after the injection, the brain of the animals was subjected to micro-wave fixation, 6 kW, 1.8 secs (Püschner GmbH, Schanewede, Germany). The brains were removed and the hippocampal formation, cerebral cortex, and cerebellum were dissected out, weighed and frozen (–80°C). The hippocampal formation included the hippocampus and dentate gyrus, the amygdala, entorhinal, and piriform cortices. The cerebral cortex sample consisted of the frontal, parietal, and lateral cortices. Plasma samples, obtained by sectioning the femoral artery immediately after the microwave fixation of the brain, were put on ice, centrifuged for 5 mins at 3000g and frozen at -80°C until extraction.
Tissue and Plasma Extraction
Each frozen sample was homogenized in 1 mL ice-cold 7% perchloric acid (PCA) and centrifuged for 10 mins at 4°C and 4000g. The supernatants were transferred into new tubes and the precipitates were re-extracted with 1 mL distilled water. The combined supernatants were centrifuged again for 10 mins at 4°C and 4000g. The tubes were kept on ice at all times possible. Each sample was neutralized with 1 mol/L KOH, lyophilized, and stored at -20°C. The lyophilized samples were dissolved in 1 mL D2O (70%, Cambridge Isotope Laboratories, Woburn, MA, USA) and 20 μL were taken out for HPLC analysis. The samples were centrifuged for 10 mins at 4°C and 4000g, and the supernatants were lyophilized. All samples were stored at −20°C.
13C and 1H Nuclear Magnetic Resonance Spectroscopy
Lyophilized samples were dissolved in 99% D2O and pH was adjusted to values between 6.8 and 7.0. Proton decoupled 13C NMR spectra were accumulated on BRUKER DRX500 and DRX600 spectrometers (BRUKER Analytik GmbH, Rheinstetten, Germany). The following acquisition parameters were applied; 30° pulse angle, acquisition time of 1.3 secs and a relaxation delay of 2.5 secs. The number of scans was typically 15,000. Some spectra were also broad band decoupled only during acquisition and accompanied by a relaxation delay of 20 secs, to achieve fully relaxed spectra without nuclear Overhauser effects. From several sets of spectra, correction factors were obtained and applied to the integrals of the individual peaks.
1H NMR spectra were acquired, on the same instruments, with the following acquisition parameters: 90° pulse angle, an acquisition time of 1.36 secs and a relaxation delay of 10 s, 320 scans were accumulated for each sample. Water suppression was achieved by applying a low-power presaturation pulse at the water frequency.
HPLC Analysis
Amino acids in the brain extracts were quantified by HPLC analysis after derivatization with o-phthaldialdehyde essentially as described by Geddes and Wood (1984), using a Hewlett Packard 1100 apparatus (Agilent, PaloAlto, CA, USA).
Metabolic Fate of Glucose and Acetate
A typical 13C NMR spectrum of a tissue extract from the hippocampal formation of a chronically epileptic rat, after the simultaneous injection of [1-13C]glucose and [1,2-13C]acetate, is shown in Figure 1. Generally, singlets denote monolabeled metabolites mainly derived from [1-13C]glucose metabolism whereas doublets denote double-labeled metabolites mainly originating from [1,2-13C]acetate metabolism. The metabolic fate of the two labeled precursors is presented in Figure 2. During glycolysis, [1-13C]glucose is metabolized to [3-13C]pyruvate that can be converted to [3-13C]lactate, [3-13C]alanine or enter the TCA cycle via pyruvate dehydrogenase as [2-13C]acetyl CoA. [4-13C]Glutamate and subsequently [4-13C]glutamine or [2-13C]GABA may be formed from [4-13C]α-ketoglutarate leaving the cycle during the first turn. In case [4-13C]α-ketoglutarate remains in the cycle for an additional turn, labeled oxaloacetate may be formed. Labeled oxaloacetate can either be transaminated to [2-13C]-/[3-13C]aspartate or condense with unlabeled acetyl CoA leading to the subsequent formation of [2-13C]-/[3-13C]glutamate or glutamine, or [3-13C]-/[4-13C]GABA. Labeled oxaloacetate can also condense with [2-13C]acetyl CoA leading to the succeeding formation of [3,4-13C]-/[2,4-13C]glutamate or [2,3-13C]-/[2,4-13C]GABA. This is for reasons of simplicity not included in Figure 2.
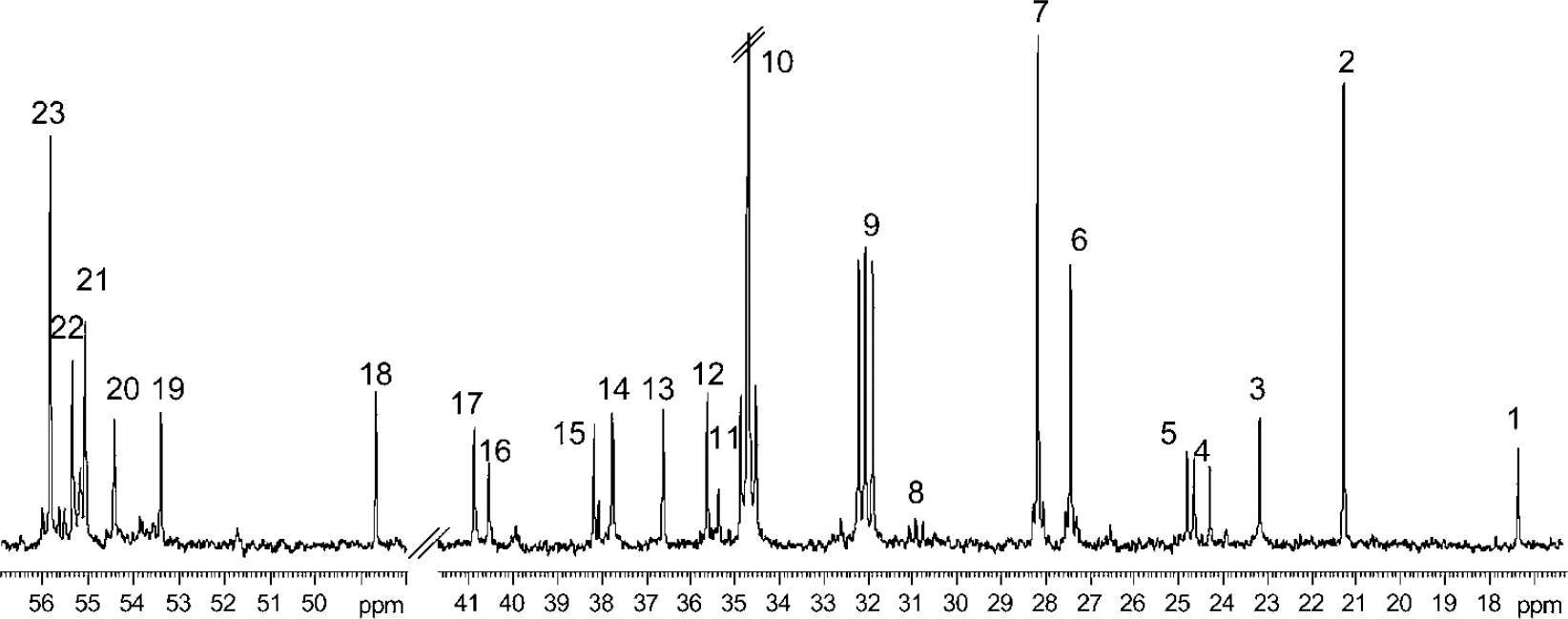
Typical 13C-NMR spectrum of a perchloric acid (PCA) extract from the hippocampal formation of a rat subjected to Li-pilo SE and having experienced one month of spontaneous seizures. The animal was administered an intraperitoneal injection of [1-13C]glucose and [1,2-13C]acetate, and was killed 15 mins later. The singlets denote monolabeled metabolites mainly derived from [1-13C]glucose whereas doublets denote double-labeled metabolites mainly originating from the metabolism of [1,2-13C]acetate. Peak assignment: 1–alanine C-3, 2–lactate C-3, 3–N-acetyl aspartate C-6, 4–acetate C-2, 5–GABA C-3, 6–glutamine C-3,
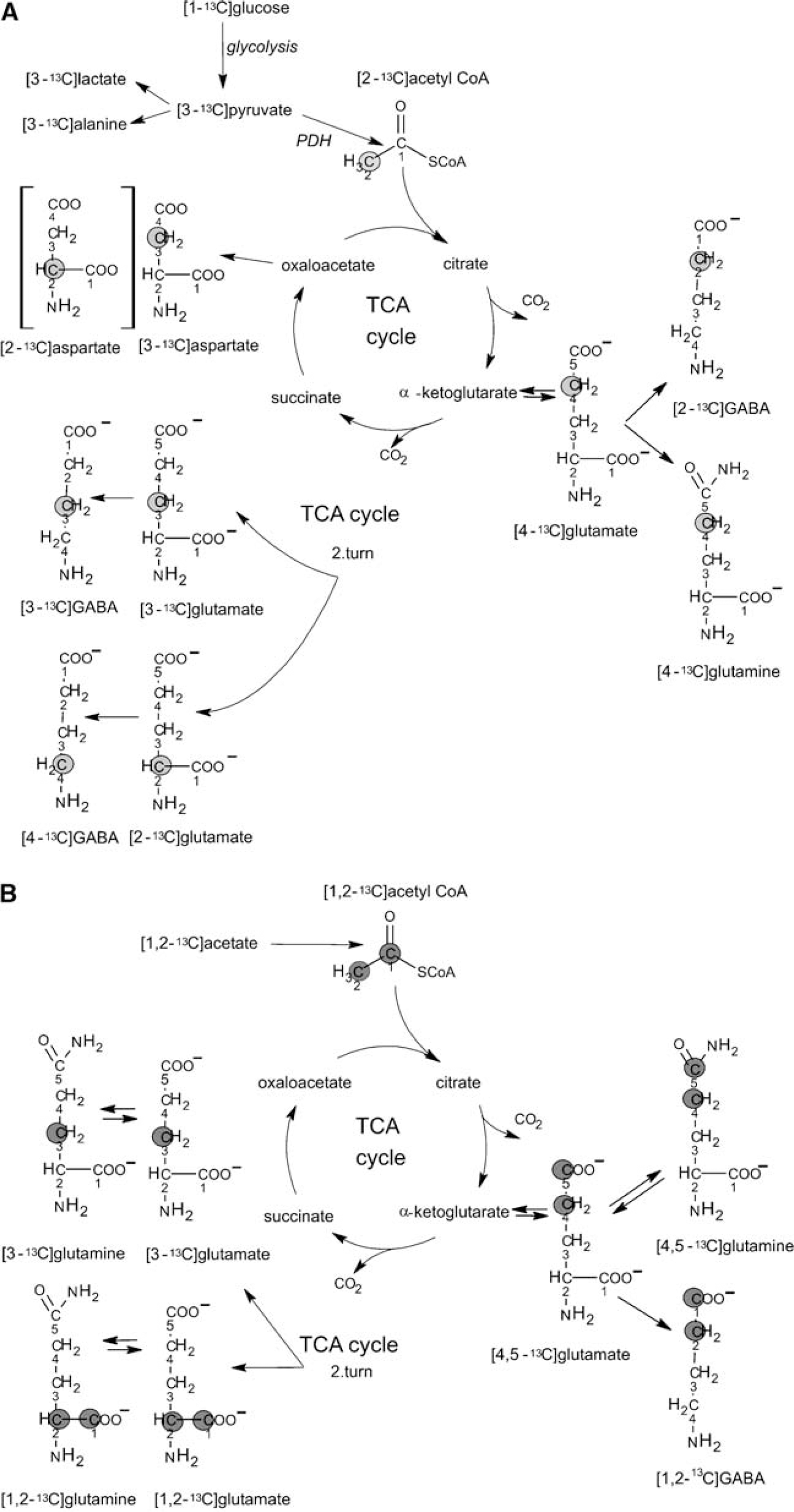
Schematic representation of possible isotopomers arising from [1-13C]glucose (
In astrocytes, [3-13C]pyruvate can also be metabolized via pyruvate carboxylation (Haberg et al, 1998). This reaction is not included in Figure 2A, since there were no differences in pyruvate carboxylation between the groups in the present study.
[1,2-13C]Acetate is converted to [1,2-13C]acetyl CoA that enters the TCA cycle leading to the formation of [4,5-13C]glutamate, [4,5-13C]glutamine or [1,2-13C]GABA. If [4,5-13C]α-ketoglutarate stays in the TCA cycle for a second turn and labeled oxaloacetate condenses with unlabeled acetyl CoA, [1,2-13C]-/[3-13C]glutamate and glutamine can be formed. Labeled oxaloacetate may also condense with labeled acetyl CoA, giving rise to [1,2-13C]-/[3,4,5-13C]glutamate or glutamine or [1,2,3-13C]-/[3,4-13C]GABA. This is for reasons of simplicity not included in Figure 2B.
Data and Statistical Analysis
Relevant peaks in the 13C and 1H NMR spectra were identified and integrated using XWINNMR software. The amounts of 13C were quantified from the integrals of the peak areas, using dioxan as internal standard at 67.9 ppm. Relevant peaks in the 1H spectra were also integrated using dioxan as an internal standard at 3.75 ppm. The total amounts (13C plus 12C) of glutamate, glutamine, GABA, alanine, glutathione. and aspartate in the extracts were quantified by HPLC. All results are expressed as mean±standard deviation of seven Li-saline or eight Li-pilo rats. All concentrations were corrected for tissue weight to take into account the extent of lesions that varies according to the structure. Statistical differences between the two groups were analyzed using unpaired two-tailed Student's t-test. The level of significance was set at P<0.05.
Results
13C Nuclear Magnetic Resonance Spectroscopy
Labeled metabolites derived from [1-13C]gIucose: The concentration of [1-13C]glucose in plasma was similar in Li-saline (235±72 nmol/mL) and Li-pilo rats (216±128nmol/mL). The same was true for [1-13C]glucose levels in brain tissue (results not shown). This implies that the differences in the concentrations of metabolites observed between the two groups in the present study were because of differences in metabolism. In the brain regions investigated, the levels of [4-13C]glutamate and [2-13C]GABA formed during the first turn of the TCA cycle from [1-13C]glucose metabolism were decreased in Li-pilo compared with Li-saline rats (Figure 3). Furthermore, the amounts of [2-13C]- and [3-13C]glutamate, and [4-13C]GABA formed via [1-13C]glucose metabolism in the second turn of the TCA cycle were also significantly decreased in all regions of Li-pilo compared with Li-saline rats. Because equal amounts of [2-13C]- and [3-13C]glutamate will be formed via cycling of [1-13C]glucose, only [3-13C]glutamate is depicted in Figure 3. However, [2-13C]glutamate and subsequently [4-13C]GABA can also be formed as a result of [3-13C]pyruvate carboxylation in astrocytes. In this study, pyruvate carboxylation and pyruvate dehydrogenase activity ratios were similar in Li-pilo and Li-saline rats. The concentrations of [2-13C]-/[3-13C]aspartate, and [3-13C]alanine were lower in Li-pilo than in Li-saline rats in all brain regions. Because equal amounts of [2-13C]- and [3-13C]aspartate were formed, due to the symmetrical succinate step, only [3-13C]aspartate is presented in Figure 3. The amount of [4-13C]glutamine synthesized in the first turn of the TCA cycle was unchanged in the hippocampal formation but decreased in cerebellum and cortex. The concentrations of [2-13C]-/[3-13C]glutamine, which are mainly formed from the second turn of the TCA cycle (Figure 2A), were unchanged in Li-pilo compared with Li-saline rats, except for the cerebellum where the concentration of [2-13C]glutamine was decreased (Figure 3 and data not shown). Finally, the level of labeled succinate was decreased in the cortex of Li-pilo compared with Li-saline rats.
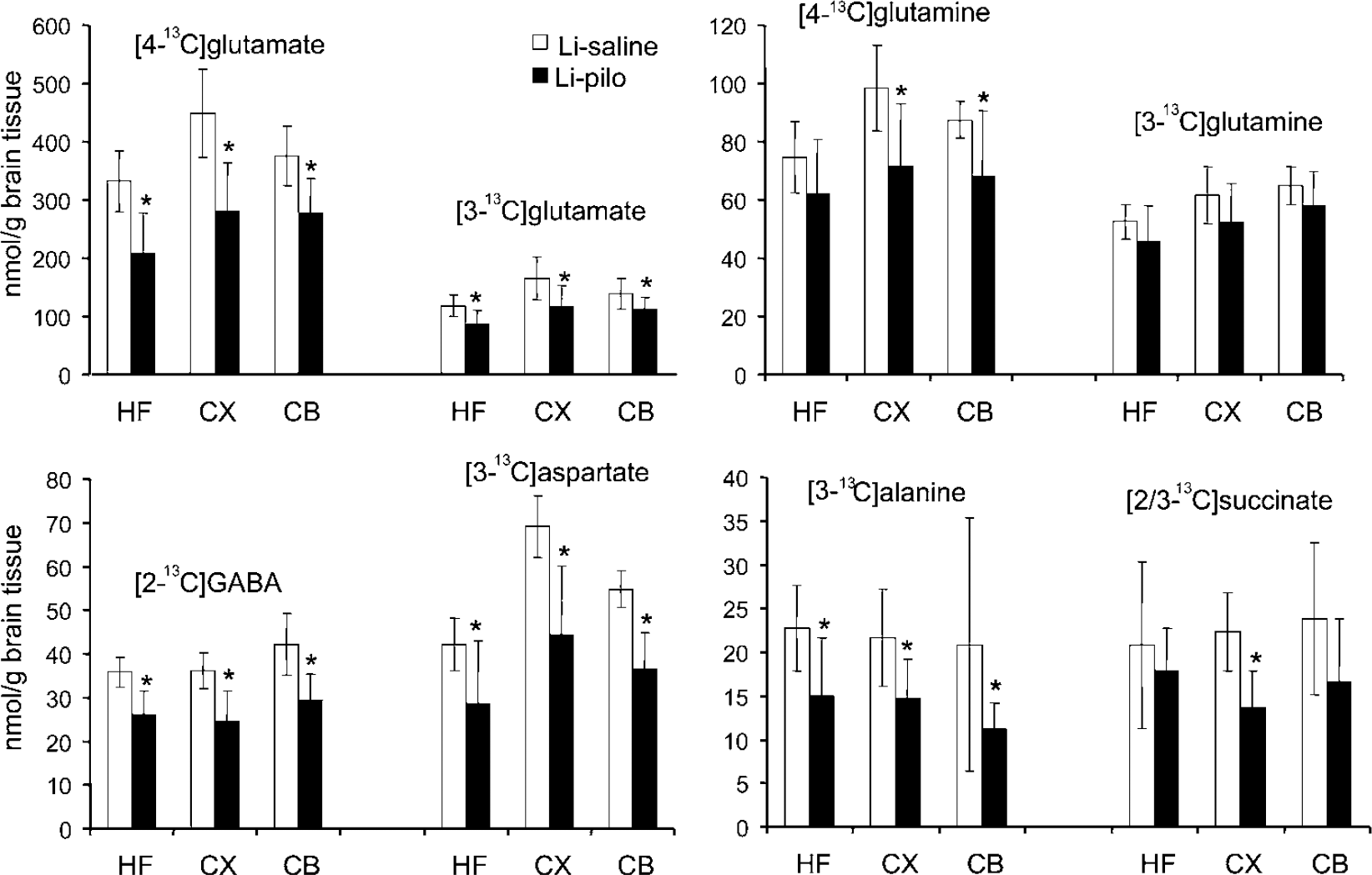
Concentration of 13C labeled metabolites specifically derived from [1- 13C]glucose in the hippocampal formation (HF), cerebral cortex (CX), and cerebellum (CB) in brain extracts from Li-saline rats and epileptic rats taken during the interictal period of the chronic phase. All animals were administered an intraperitoneal injection of [1-13C]glucose and [1,2-13C]acetate, and were killed 15 mins later. The amount of 13C (nmol/g tissue) was analyzed with 13C nuclear magnetic resonance spectroscopy (NMRS) and was not corrected for naturally abundant 13C (1.1%). Data represent means±standard deviation of 7 Li-saline and 8 Li-pilo rats. *P<0.05, statistically significant difference between the two groups.
Labeled metabolites derived from [1,2-13C]acetate: As for [1-13C]glucose, the amount of [1,2-13C]acetate in plasma of Li-saline (232±56 nmol/mL) and Li-pilo rats (277±58 nmol/mL) was similar. The concentrations of all metabolites synthesized from [1,2-13C]acetate were similar in the brain regions studied in Li-saline and Li-pilo rats (data not shown).
HPLC and 1H Nuclear Magnetic Resonance Spectroscopy
The concentrations of various amino acids and other compounds are presented in Figure 4 and Table 1. The levels of aspartate were decreased in all regions of Li-pilo compared with Li-saline rats, whereas the levels of N-acetylaspartate (NAA) were decreased in the hippocampal formation and the cerebral cortex only. The concentration of glutamate was decreased in the hippocampal formation and that of GABA only in the cerebellum of epileptic compared with Li-saline rats (Figure 4). The concentrations of glutamine, alanine, and succinate were similar in the three regions of both groups of rats (Table 1). The concentration of glutathione and adenosine triphosphate (ATP) plus adenosine diphosphate (ADP) was decreased in the hippocampal formation and that of creatine was decreased in the cerebellum of epileptic compared with control rats (Table 1). ATP and ADP are given as a sum because of resonances overlap in the 1H spectrum. Both in cortex and in the hippocampal formation, the amounts of glucose were higher in Li-pilo than in Li-saline rats. The levels of phosphocreatine, inositol, taurine, lactate, and reduced nicotine adenine dinucleotide (NAD+) were not affected by epilepsy in all brain regions examined.
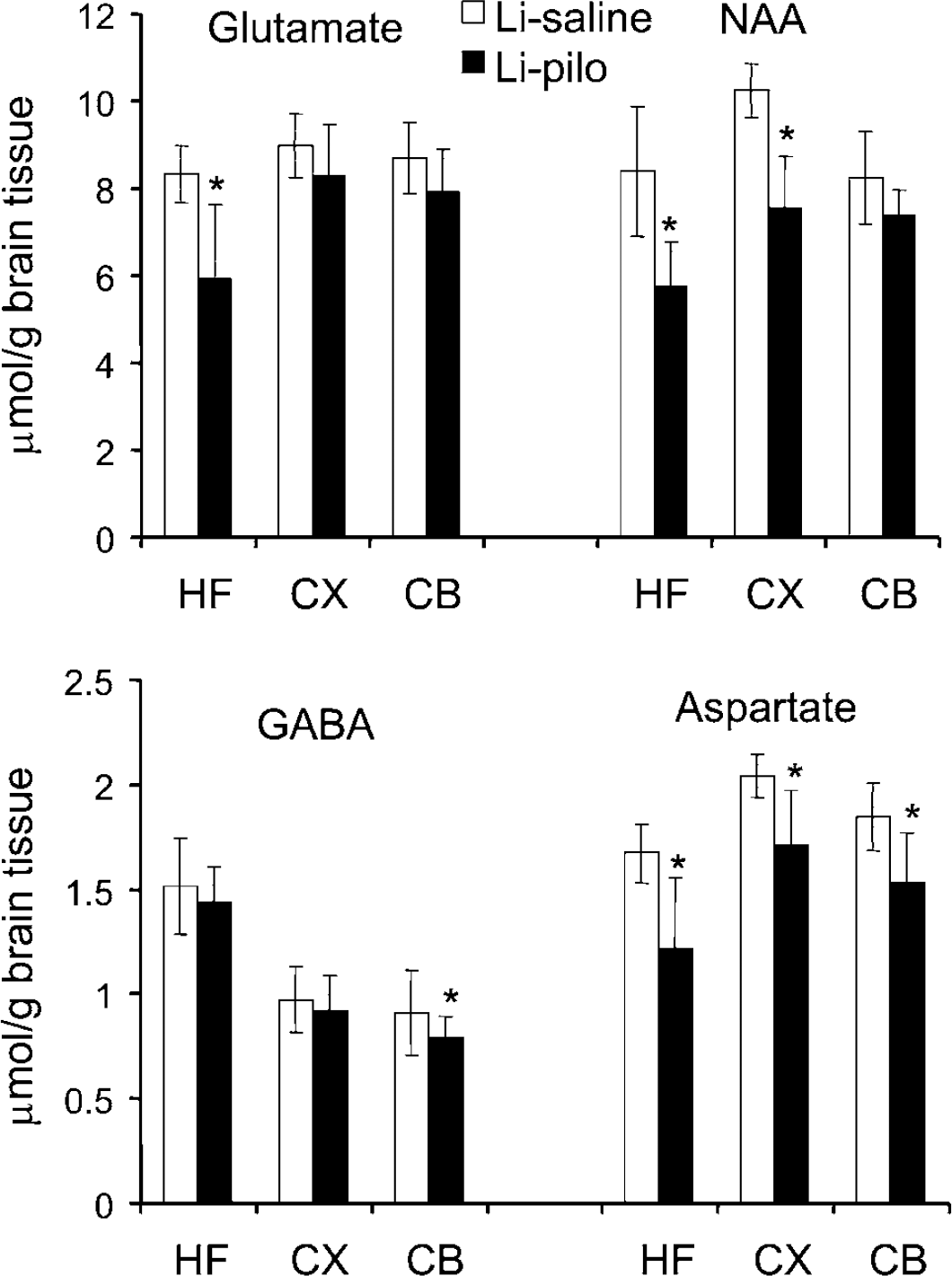
Amino acid concentrations (μmmol/g tissue) in rat brain extracts of the hippocampal formation (HF), cerebral cortex (CX), and cerebellum (CB) in brain extracts from epileptic rats taken during the interictal period of the chronic phase and compared with age and weight matched controls. Concentrations of glutamate, GABA, and aspartate were determined with HPLC whereas the amount of N-acetylaspartate (NAA) was measured by 13C nuclear magnetic resonance (NMR) spectroscopy. Data represent means±standard deviation of seven Li-saline and eight Li-pilo rats. *P<0.05, statistically significant difference between the two groups.
Metabolite concentration (μmol/g tissue) in extracts from hippocampal formation, cerebral cortex, and cerebellum of epileptic and control rats
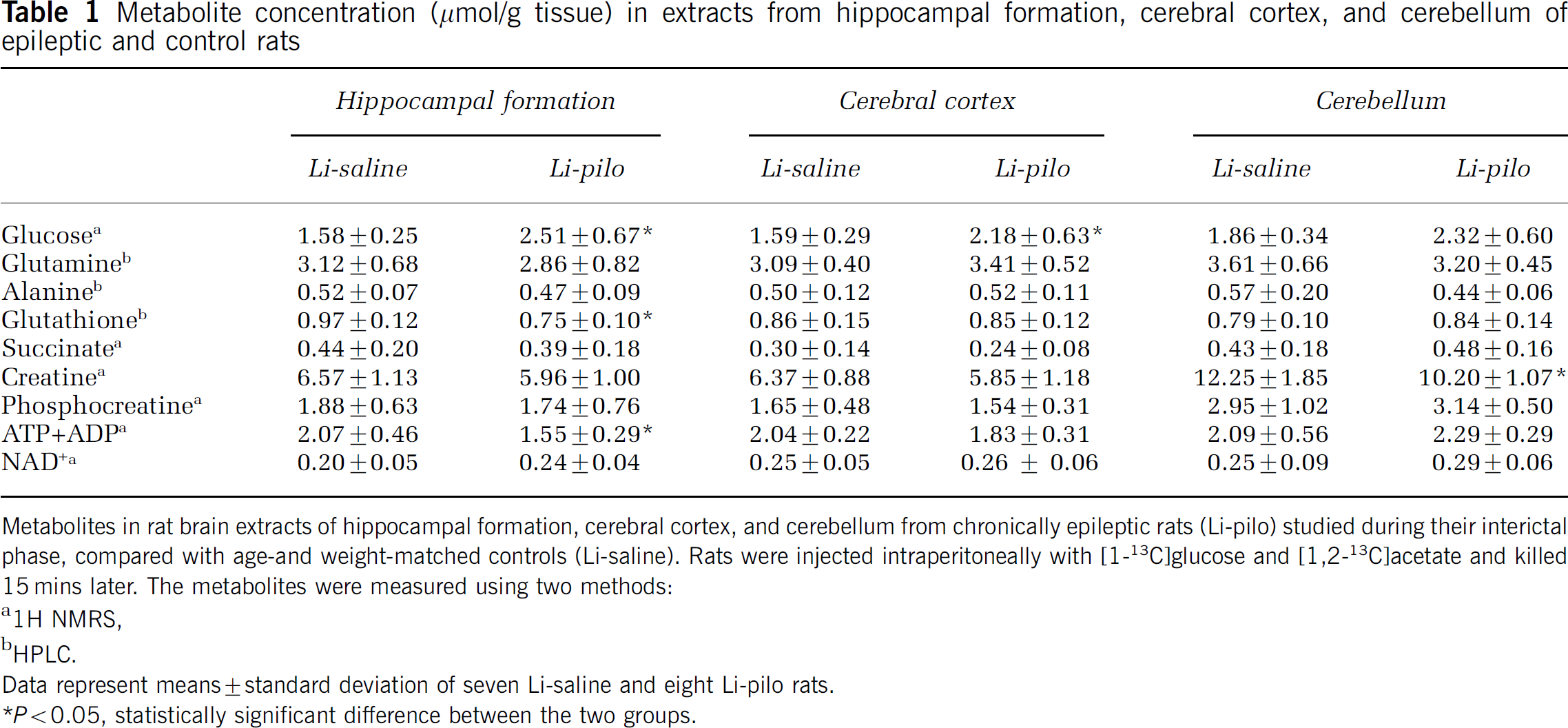
Metabolites in rat brain extracts of hippocampal formation, cerebral cortex, and cerebellum from chronically epileptic rats (Li-pilo) studied during their interictal phase, compared with age-and weight-matched controls (Li-saline). Rats were injected intraperitoneally with [1-13C]glucose and [1,2-13C]acetate and killed 15 mins later. The metabolites were measured using two methods:
1H NMRS
HPLC.
Data represent means±standard deviation of seven Li-saline and eight Li-pilo rats.
P<0.05, statistically significant difference between the two groups.
Discussion
This study is, to our knowledge, the first one showing that in the brain of epileptic rats, metabolism is unchanged in astrocytes and only disturbed in neurons in the epileptic circuit. In a previous study, we have already shown that astrocytic metabolism was unchanged in the latent phase of the kainate model of TLE (Muller et al, 2000). Further studies will be necessary to ascertain whether this conclusion can be extended to the human epileptic brain.
Glucose Metabolism
After glycolysis, [3-13C]pyruvate is converted to [3-13C]lactate by lactate dehydrogenase in the cytosol, or to [3-13C]alanine via alanine aminotransferase, both in the cytosol and mitochondria. In all brain structures of Li-pilo rats, the amount of [3-13C]alanine was reduced whereas [3-13C]lactate levels were not altered, indicating reduced metabolism in the mitochondria. These mitochondria were probably neuronal since 70% of the [3-13C]pyruvate from [1-13C]glucose is metabolized in neuronal mitochondria (Qu et al, 2000) and only [3-13C] alanine was reduced, while total alanine was not affected and most of the alanine is synthesized in the astrocytes in cortex (Westergaard et al, 1993; Sonnewald et al, 1993).
The present study shows decreased glucose consumption in cerebral cortex and hippocampal formation of epileptic rats since glucose content was increased in these regions. However, this was only statistically significant for unlabeled glucose, not for [1-13C]glucose, likely because the percentage of enrichment in glucose was only 18%, thus the differences might have been too small to be detectable. This decreased consumption is in agreement with animal and human data showing interictal hypometabolism within the hippocampus, which could extend to extratemporal cortical and subcortical regions (Arnold et al, 1996; Dube et al, 2001; Vielhaber et al, 2003b; Chassoux et al, 2004). Moreover, in these areas neuronal loss does not necessarily correlate with interictal hypometabolism (Ryvlin et al, 1991; O'Brien et al, 1997; Foldvary et al, 1999; Dube et al, 2001).
Tricarboxylic Acid Cycle Derived Metabolites in the Hippocampal Formation
N-acetylaspartate levels: N-acetylaspartate is among the most abundant amino acids in brain and is synthesized in neuronal mitochondria (Baslow, 2003). A decrease in NAA has been reported in hippocampus and extrahippocampal regions of TLE patients (Cendes et al, 1997; Aasly et al, 1999; Petroff et al, 2002; Mueller et al, 2004). Loss of NAA can both indicate mitochondrial dysfunction and cell death. In the present study, as already shown by Vielhaber et al (2003a), a large reduction in NAA concentration was found in the hippocampal formation where additionally ATP plus ADP were reduced. The reduction in these compounds might reflect the neuronal loss in these regions or be because of a deficiency in mitochondrial activity, mainly oxidative phosphorylation, as reported in the pilocarpine and kainate models and in human tissue (Kunz et al, 2000; Kudin et al, 2002; Chuang et al, 2004). In the present study, however, NAD+, phosphocreatine and creatine contents, which depend on well functioning oxidative phosphorylation, were not affected by Li-pilo-induced epilepsy. Glutamate—glutamine cycle: Glutamate, which is primarily synthesized and stored in glutamatergic neurons (Ottersen and Storm-Mathisen, 1985), is released on excitation, and removed mostly by uptake into astrocytes (Danbolt, 2001). Subsequently, glutamate is converted into glutamine by the astrocyte-specific enzyme glutamine synthetase (GS) (Norenberg and Martinez-Hernandez, 1979), and cycled back to neurons. This close metabolic cooperation between astrocytes and neurons represents the glutamate—glutamine cycle (Berl and Clarke, 1969; Berl and Clarke, 1983).
The concentration of glutamate and glutathione was decreased in the hippocampus, amygdala, entorhinal, and piriform cortices of Li-pilo rats, which is in line with neuronal loss in these regions (Motte et al, 1998; Dube et al, 2001). This is in agreement with studies in humans (Aasly et al, 1999; Petroff et al, 2002). In addition to decreased glutamate concentration, labeling of [4-13C]glutamate derived from [1-13C]glucose was also reduced. The cycling rate of the TCA cycle was preserved since the amount of [3-13C]glutamate was decreased to a similar extent as [4-13C]glutamate. However, the amount of [4-13C]glutamine was unchanged, which implies that glutamate transport and/or synthesis of glutamine was not impaired in the hippocampal formation of Li-pilo rats. Indeed, most of the [4-13C]glutamine is derived from neuronal [4-13C]glutamate (Fitzpatrick et al, 1990). The amount of [4,5-13C]glutamate, derived from [4,5-13C]glutamine, in neurons is unchanged. Because the number of neurons is decreased, this necessitates an increase in PAG, as shown during insulin-induced seizures (Abdul-Ghani et al, 1989).
Studies on animal models of epilepsy show that gliosis is present in regions exhibiting epileptiform activity because of neuronal injury as well as seizure activity (Castiglioni et al, 1990; Eddleston and Mucke, 1993; Khurgel and Ivy, 1996). In Li-pilotreated rats, changes in astrocytes were mainly characterized by hypertrophy of cell bodies and processes near the lesions rather than an increase in astrocyte number (Roch et al, 2002). Furthermore, in the pilocarpine model, only half of the chronically epileptic rats had reactive astrocytes in thalamic, hippocampal, amygdalar, and neocortical areas (Garzillo and Mello, 2002). In the present study, there was no increase of glutamine content in epileptic rats in the hippocampal formation. This implies that the average glutamine production in hypertrophic, reactive astrocytes is unchanged compared with normal astrocytes and that the characteristics of the glutamate-glutamine cycle are preserved in the hippocampal formation of chronically epileptic rats. Glutamine synthetase has been proposed as the primary enzyme in the regulation of glutamate metabolism in the brain. The inhibition of this enzyme in vivo readily leads to seizures (Shaw et al, 1999) and decreased GS activity in astrocytes has been reported in surgically resected hippocampus from TLE patients (Eid et al, 2004). This enzymatic downregulation does not seem to be the main underlying cause of the generation of spontaneous seizures in the Li-pilo model of TLE since glutamine synthesis appears normal in astrocytes. However, both cell bodies and astrocytic processes are hypertrophic in this model (Roch et al, 2002). If the larger volume occupied by this cell type is taken into account, a downregulation of GS may also take place in the present study.
GABA: In agreement with studies in humans (Aasly et al, 1999; Petroff et al, 2002), GABA concentration was similar in hippocampal formation of Li-saline and Li-pilo rats. However, although glutamate concentrations decreased in the hippocampal formation of Li-pilo rats compared with controls, GABA concentration remained in the normal range. These data are in disagreement with those of a previous study in the pilocarpine model of epilepsy where increases in the concentrations of glutamate, aspartate, GABA and glycine were found in the hippocampus (Cavalheiro et al, 1994). However, GABAergic interneurons undergo marked cell death in the hippocampus of epileptic rats (Schwarzer and Sperk, 1995; Andre et al, 2001). The lack of change in GABA concentration is in accordance with the upregulation of the GABA neuronal marker, glutamate decarboxylase (GAD), observed in the seizure circuit during the latent and long-term phases of Li-pilo and pilocarpine models (Brooks-Kayal et al, 1998; Andre et al, 2001) However, labeling of [2-13C]GABA from [1-13C]glucose was decreased, indicating decreased turnover of GABA, in agreement with the previously reported decreased release (Hirsch et al, 1999). Conversely, labeling of GABA from [1,2-13C]acetate in astrocytes was unchanged.
Tricarboxylic Acid Cycle Derived Metabolites in Cerebral Cortex and Cerebellum
Epileptogenic activity is usually not restricted to the hippocampus but spreads to other brain regions, particularly to the temporal lobes (Adam et al, 1994; Isnard et al, 2000). Thus, it is not surprising that metabolites in the cerebral cortex were also affected in epileptic Li-pilo rats. Although only moderate cell loss occurs in this area (Turski et al, 1989; Motte et al, 1998; Dube et al, 2001), the decrease in the concentration of neuron-related metabolites such as NAA and aspartate was quite marked, whereas the glutamate level was unchanged. While a decrease in NAA concentration may reflect cell loss in the hippocampal formation, it most likely also reflects neuronal metabolic dysfunction in cortex (Vielhaber et al, 2003a). Labeling from [1-13C]glucose also decreased in glutamate, GABA, and succinate, indicating neuronal mitochondrial dysfunction. [4-13C]glutamine labeling was decreased in the cortex, possibly as a result of reduced [4-13C]glutamate labeling. Conversely, labeling of glutamate, glutamine, and GABA from [1,2-13C]acetate was unchanged suggesting that astrocytic metabolism was not affected by recurrent seizures in cerebral cortex and cerebellum, as for the regions directly belonging to the circuit of seizures.
The cerebellum was included in the study as a reference structure, since it is not involved in seizure propagation. No cell loss is reported in the cerebellum of rats subjected to Li-pilo SE (Turski et al, 1989; Motte et al, 1998; Dube et al, 2001), which correlates with no statistically significant loss of NAA in this area. There was, however, a decreased incorporation of 13C label from [1-13C]glucose into metabolites derived from the TCA cycle occurring in the cerebellum of epileptic rats, mainly glutamate, aspartate, and GABA. GABA and creatine concentrations were only reduced in the cerebellum and many other metabolic changes recorded in that structure were very similar to those occurring in the cerebral cortex. Thus, these changes indicate that neuronal mitochondrial metabolism in the cerebellum is affected by ongoing epileptic activity, as is probably the case in many other structures. Finally, the content and labeling of aspartate was decreased in all areas. Thus, the activity of the transaminase that leads to synthesis of aspartate appears downregulated in epileptic rats, presumably as a result of neuronal mitochondrial dysfunction.
Conclusion
In the hippocampal formation, which includes most of the structures likely to be involved in the epileptic circuit in this model of epilepsy, the flow of metabolites between astrocytes and neurons was unchanged, whereas glutamatergic neurons in the cerebral cortex and cerebellum have decreased their contribution to glutamine formation in astrocytes. These changes may contribute to a generalized increase in brain excitability.
Footnotes
Acknowledgements
The excellent technical assistance of Estelle Koning is greatly acknowledged.
