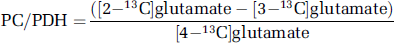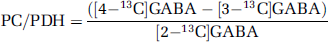Abstract
Disturbances in GABAergic and glutamatergic neurotransmission in the thalamocortical loop are involved in absence seizures. Here, we examined potential disturbances in metabolism and interactions between neurons and glia in 5-month-old genetic absence epilepsy rats from Strasbourg (GAERS) and nonepileptic rats (NER). Animals received [1-13C]glucose and [1,2-13C]acetate, the preferential substrates of neurons and astrocytes, respectively. Extracts from cerebral cortex, thalamus, and hippocampus were analyzed by 13C nuclear magnetic resonance spectroscopy. Most changes were detected in the cortex. Pyruvate metabolism was enhanced as evidenced by increases of lactate, and labeled and unlabeled alanine. Neuronal mitochondrial metabolism was also enhanced as detected by elevated amounts of N-acetylaspartate and nicotinamide adenine dinucleotide as well as increased incorporation of label from [2-13C]acetyl CoA into glutamate, glutamine, and aspartate. Likewise, mitochondrial metabolism in astrocytes was increased. Changes in thalamus were restricted to increased concentration and labeling of glutamine. Changes in the hippocampus were similar to those in the cortex. This increase in glutamate-glutamine metabolism in cortical neurons and astrocytes accompanied by a decreased gamma aminobyturic acid level may lead to impaired thalamic filter function. Hence, reduced sensory input to cortex could allow the occurrence of spike-and-wave discharges in the thalamocortical loop. Increased glutamatergic output from the cortex to hippocampus may be the underlying cause of improved learning in GAERS.
Introduction
Generalized nonconvulsive absence seizures are characterized by the occurrence of bilateral, synchronous spike-and-wave discharges (SWDs) on electroencephalographic recordings, concomitant with behavioral arrest. The strain of genetic absence epilepsy rats from Strasbourg (GAERS) is an isomorphic, predictive, and homologous model of human generalized idiopathic absence epilepsy. The clinical seizures consisting of behavioral arrest with twitching of facial muscles and occurring in all animals have a polygenic origin (Rudolf et al, 2004). Behavioral expression of seizures is associated with bilateral synchronous SWDs that resemble those of typical absence seizures. Spontaneous absence seizures originating in the cortex and thalamus appear in this strain after the age of 1 month and their frequency is stabilized by 3 to 4 months (Danober et al, 1998; Meeren et al, 2005).
Pharmacological data suggest that both inhibitory and excitatory neurotransmission are involved in triggering and maintaining absence seizures (Danober et al, 1998). The first studies performed on GAERS in the 1980s led to the hypothesis of a predominant role of GABAergic inhibition in the genesis of SWDs (for a review, see Marescaux et al, 1992). However, most morphologic and functional properties of the GABAergic system are similar in GAERS and in a strain of genetically selected nonepileptic rats (NER) (Snead et al, 1992; Danober et al, 1998; Sutch et al, 1999). Only subtle differences in GABAergic neurotransmission were found between GAERS and NER, mainly increased extracellular gamma aminobyturic acid (GABA) concentration in the cortex and thalamus (Richards et al, 1995, 2000) and reduced GABA uptake in thalamic synaptosomes of GAERS (Sutch et al, 1999). Concerning glutamatergic neurotransmission, a more intense electrophysiologic response was recorded in the cortex of GAERS after stimulation of N-methyl-D-aspartate (NMDA) and non-NMDA receptors (Pumain et al, 1992; Avanzini et al, 1996). However, no differences were observed in the expression of the two astrocytic glutamate transporter proteins, glutamate/aspartate transporter and glutamate transporter 1 in the cortex and thalamus of GAERS (Dutuit et al, 2002), whereas divergent data were found at the level of their messenger RNAs (Ingram et al, 2000; Dutuit et al, 2002). More recent data are in favor of the existence of a specific balance between excitation and inhibition in the brain of GAERS that would favor the occurrence of absence seizures. This subtle imbalance could reflect some dysregulation in glutamate metabolism in the cortex of GAERS (Dufour et al, 2001).
GAERS display an increase in cerebral glucose utilization throughout the whole brain compared with NER (Nehlig et al, 1991), paralleled by elevated activities of enzymes coupled to glycolysis, oxidative phosphorylation, and glutamate metabolism (Dutuit et al, 2000; Dufour et al, 2003). Furthermore, the expression of the astrocyte-specific glial fibrillary acidic protein (GFAP) is increased in the cortex and thalamus (Dutuit et al, 2000). Thus, astrocytes might be involved in the regulation of neuronal processes underlying the occurrence of epileptic seizures in this strain.
To address the question, if dysregulation of astrocyte-neuron interactions in the thalamocortical loop is related to the occurrence of absence epilepsy, we conducted a nuclear magnetic resonance (NMR) spectroscopy study, using 5-month-old GAERS and NER. 13C-NMR spectroscopy has been used extensively for metabolic studies of cerebral metabolism (Shank et al, 1993; Sonnewald and Kondziella, 2003). After the administration of 13C-labeled substrates, it is possible to differentiate metabolic events taking place in astrocytes and neurons, respectively, as astrocytes and neurons express different enzymes and transporters. Acetate and glucose are both precursors for acetyl coenzyme A (CoA), which enters the tricarboxylic acid (TCA) cycle. Acetyl CoA derived from glucose is metabolized more in the neuronal TCA cycle (Qu et al, 2000), whereas acetate is metabolized predominantly in astrocytes as it is selectively taken up into these cells (Waniewski and Martin, 1998). Thus, by simultaneous injection of [1-13C]glucose and [1,2-13C]acetate and NMR spectroscopy analysis of brain extracts, information about neuronal and astrocytic metabolism can be obtained in the same animal (Taylor et al, 1996).
Materials and methods
Animals
Eight adult male GAERS (58th generation) and 10 adult male NER (55th generation), 5 months old, originating from the Strasbourg breeding colony, were used in the experiment. The animals were maintained at 22°C room temperature under a 12/12 h normal light/dark cycle (lights on at 0700 hours) with food and water ad libitum. Under these conditions, animals exhibit spontaneous SWDs alternating with periods of normal background EEG activity. The mean duration of SWDs reached 20 to 24 secs/min of recording. All animal experimentation was performed in accordance with the rules of the European Communities Council Directive of November 24, 1986 (86/609/EEC), and the French Department of Agriculture (License No. 67 to 97).
Both groups of rats were administered intraperitoneal injections of sodium [1,2-13C]acetate (504 mg/kg, 0.6 mol/L solution) and [1-13C]glucose (543 mg/kg 0.3 mol/L solution; 99% 13C enriched, Cambridge Isotope Laboratories, Woburn, MA, USA). At 15 mins after injections, the animals were subjected to brain microwave fixation, 6kW, 1.8 secs (Püschner GmbH, Schanewede, Germany), and thereafter decapitated. Blood was collected freely flowing from the carotid artery. The brains were removed and the thalamus, cerebral cortex, and the hippocampus were removed, weighed, and frozen (–80°C). The cerebral cortex samples consisted of the upper half of the cortex to the horizontal midline of the brain and were mainly frontal and somatosensory cortices, both the parietal and motor areas. Blood samples were centrifuged for 5 mins at 3000g and plasma was thereafter frozen at −80°C until extraction.
Tissue and Plasma Extraction
Each frozen tissue sample was homogenized in 1 ml ice-cold 7% perchloric acid and centrifuged for 10 mins at 4°C and 4000g. The supernatants were transferred into new tubes and the precipitates were re-extracted with 0.5 mL 7% perchloric acid. The combined supernatants were centrifuged again for 10 mins at 4°C and 4000 g. The tubes were kept on ice at all times possible. Each sample was neutralized with 2 mol/L KOH, centrifuged, lyophilized, and stored at −20°C. Plasma samples (typically 300 μL) were extracted with 1 mL ice-cold 7% perchloric acid and centrifuged for 10 mins at 4°C and 4000 g. Thereafter, the samples were neutralized with 2 mol/L KOH, centrifuged, lyophilized, and stored at −20°C.
13C- and 1H-Nuclear Magnetic Resonance Spectroscopy
Lyophilized samples were dissolved in 200 μl 99% D2O and pH was adjusted to values between 6.8 and 7.0. The samples were transferred into NMR microtubes (Shigemi Inc., Allison Park, PA, USA). Proton-decoupled 13C-NMR spectra from cerebral cortex samples (typical weight 500 mg) were accumulated on a BRUKER DRX500 spectrometer. Spectra from hippocampus samples (typical weight 130 mg) were obtained on a BRUKER DRX600 spectrometer (BRUKER Analytik GmbH, Rheinstetten, Germany). Spectra from thalamus samples (typical weight 45 mg) were obtained on a BRUKER600 spectrometer using a Bruker BioSpin CryoProbe (BRUKER Analytik GmbH, Rheinstetten, Germany). The following acquisition parameters were applied: 30° pulse angle, acquisition time of 1.3 secs and a relaxation delay of 0.5 secs. The number of scans was typically 20,000 for cerebral cortex and 50,000 for hippocampus and thalamus. Some spectra were also broadband decoupled only during acquisition to avoid nuclear Overhauser effect and accompanied by a relaxation delay of 20 secs, to achieve fully relaxed spectra. From several sets of spectra, correction factors were obtained and applied to the integrals of the individual peaks.
1H-NMR spectra were acquired, on the same spectrometers, with the following acquisition parameters: 90° pulse angle, an acquisition time of 1.36 secs, and a relaxation delay of 10 secs; 320 scans were accumulated for each sample, and for thalamus samples 520 scans were accumulated. Water suppression was achieved by applying a low-power presaturation pulse at the water frequency.
Data and Statistical Analysis
Relevant peaks in the 13C- and 1H-NMR spectra were identified and integrated using XWINNMR software. The amounts of 13C were quantified from the integrals of the peak areas, using ethylene glycol as internal standard. Amounts of metabolites were quantified using relevant peaks in the 1H spectra, which were integrated using ethylene glycol as an internal standard, and corrected for number of protons. All results are expressed as mean ± s.d. of 8 GAERS and 10 NER. All concentrations were corrected for tissue weight. Statistical differences between the two groups were analyzed using unpaired two-tailed Student's t-test. The level of significance was set at P < 0.05.
Metabolic Fate of Glucose and Acetate
Glucose is the primary energy substrate for brain metabolism. Complete oxidation of glucose yields CO2, H2O, and energy in the form of adenosine triphosphate (ATP). Through glycolysis, glucose is metabolized to pyruvate generating two molecules of ATP and nicotinamide adenine dinucleotide (NADH). Then, pyruvate can be oxidized to acetyl CoA, which enters the TCA cycle and electron carrier molecules such as NADH and flavin adenine dinucleotide (FADH2) are generated. Several amino acids are derived from TCA cycle intermediates. Thereafter, oxidative phosphorylation generates ATP and H2O. Through glycolysis, [1-13C]glucose is metabolized to [3-13C]pyruvate, which can be converted to [3-13C]lactate, [3-13C]alanine, or enter the TCA cycle via pyruvate dehydrogenase (PDH) as [2-13C]acetyl CoA (Figure 1A). [4-13C]Glutamate and subsequently [4-13C]glutamine or [2-13C]GABA can be formed in their respective cell types from [4-13C]a-ketoglutarate, leaving the cycle during the first turn. If [4-13C]α-ketoglutarate does not leave the TCA cycle, labeled oxaloacetate may be formed. The latter can either be transaminated to [2-13C]- or [3-13C]aspartate or condense with unlabeled acetyl CoA leading to the formation of [2-13C]- or [3-13C]glutamate or glutamine, or [3-13C]- or [4-13C]GABA. Labeled oxaloacetate can also condense with [2-13C]acetyl CoA leading to the synthesis of [3,4-13C]- or [2,4-13C]glutamate or [2,3-13C]- or [2,4-13C]GABA. For simplicity, the latter metabolites are not included in Figure 1A.
Schematic representation of possible isotopomers arising from [1-13C]glucose (
In astrocytes, [3-13C]pyruvate can condense with CO2, via pyruvate carboxylase (PC), leading to the formation of [3-13C]oxaloacetate (Haberg et al, 1998), or [2-13C]glutamate or [2-13C]glutamine. If [2-13C]glutamine is transported from the astrocyte to a glutamatergic or GABAergic neuron, it can be converted to [2-13C]glutamate or [4-13C]GABA, respectively (Figure 1A). Pyruvate carboxylase is the only anaplerotic enzyme in the brain (Patel, 1974), supplying the TCA cycle with intermediates, when metabolites such as α-ketoglutarate leave the cycle for the formation of glutamate. Pyruvate carboxylase activity accounts for about 10% of glucose metabolism in the brain (O'Neal and Koeppe, 1966; van den Berg and Garfinkel, 1971; Aureli et al, 1997) and the glial TCA cycle rate accounts for about 30% of total TCA cycle activity (Oz et al, 2004). As stated above, oxidation of [1-13C]glucose through the TCA cycle leads to [4-13C]glutamate in the first turn and [2-13C]glutamate or [3-13C]glutamate in the second turn. Pyruvate carboxylation leads to the formation of [2-13C]glutamate in the first turn. Therefore, the ratio of PC over PDH for glutamate can be calculated as follows:
The same calculation is valid for glutamine. The ratio for GABA is the following:
An increase in the PC/PDH ratio could either be due to an increase in PC activity or a decrease in PDH activity.
[1,2-13C]Acetate is converted to [1,2-13C]acetyl CoA which can enter, the TCA cycle leading to the synthesis of [4,5-13C]glutamate, [4,5-13C]glutamine or [1,2-13C]GABA (Figure 1B). If [4,5-13C]α-ketoglutarate stays in the TCA cycle and labeled oxaloacetate condenses with unlabeled acetyl CoA, [1,2-13C]- or [3-13C]glutamate and glutamine can be formed. Labeled oxaloacetate may also condense with labeled acetyl CoA, leading to the synthesis of [1,2-13C]- or [3,4,5-13C]glutamate or glutamine or [1,2,3-13C]- or [3,4-13C]GABA. For reasons of simplicity, the latter metabolites are not included in Figure 1B.
Percent enrichment in glutamate C-4 was calculated from the following formula:
[glutamate] represents the total amount of glutamate.
Results
13C-Nuclear Magnetic Resonance Spectroscopy
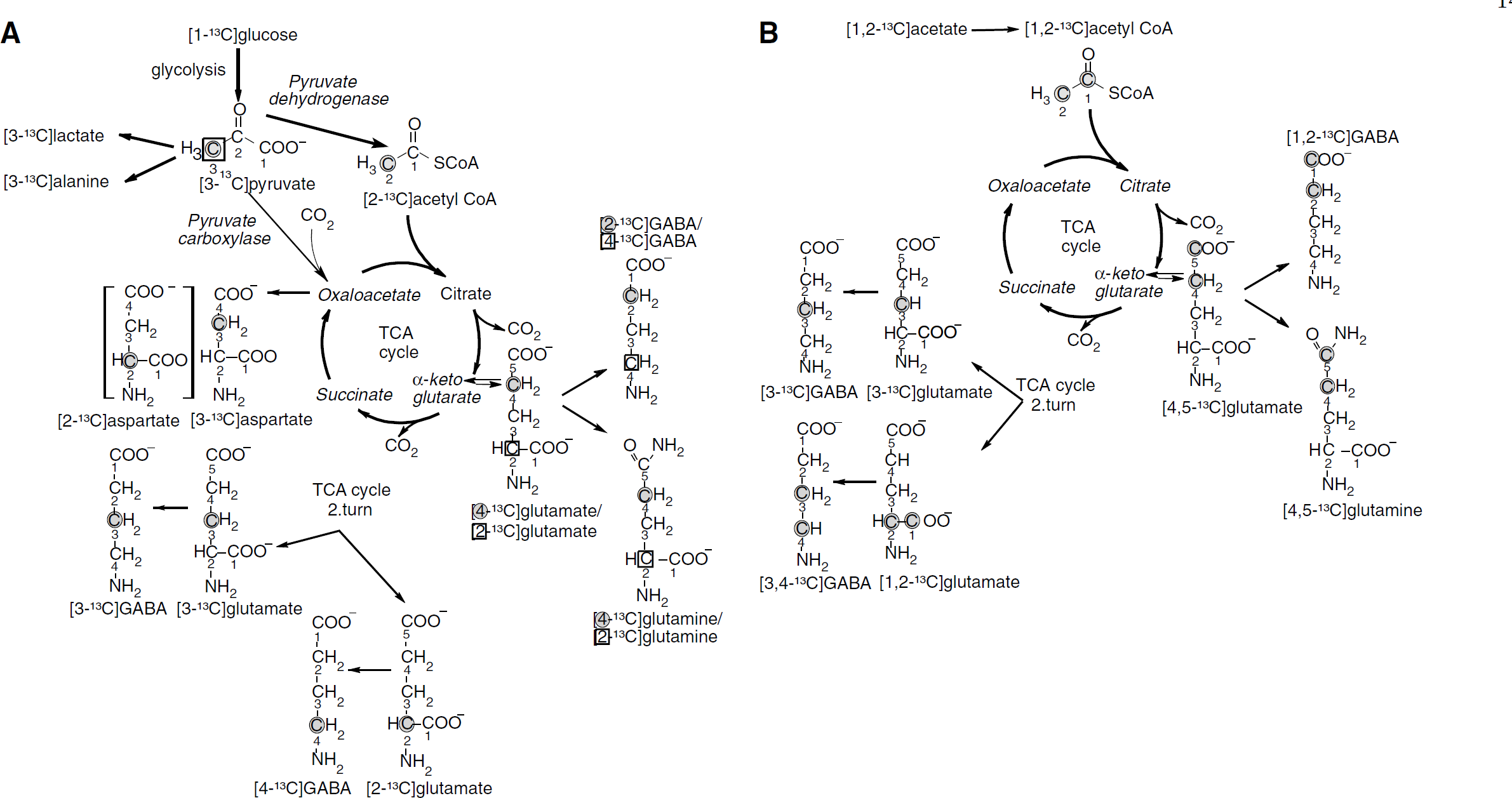
Labeled metabolites derived from [1-13C]glucose: Amounts of metabolites formed from [1-13C]glucose during the first turn of the TCA cycle are shown in Figures 2A–2C. The amounts of [4-13C]glutamate were increased in the cortex and hippocampus of GAERS compared with NER (Figure 2A). The percent enrichment in glutamate C-4 reached a value of 7.8070.72 for NER and 9.0670.99 for GAERS in the cortex. This difference was statistically significant (P < 0.01). The levels of [4-13C]glutamine (Figure 2B) were significantly increased in the thalamus, cortex, and hippocampus in GAERS compared with NER, and those of [2-13C]GABA were increased only in the cortex (Figure 2C). Furthermore, the amounts of [2-13C]- and [3-13C]glutamate derived from the second turn of the TCA cycle were also significantly increased in the cortex and hippocampus of GAERS compared with NER (results not shown). However, [2-13C]glutamate, [2-13C]glutamine, and [4-13C]GABA can also be formed as a result of pyruvate carboxylation in astrocytes (Figure 1A). Pyruvate carboxylase/PDH ratios for glutamate, glutamine, and GABA are shown in Figure 3 (for calculations see above). An increase in this ratio was seen for glutamate and glutamine in the cortex in GAERS compared with NER. The concentrations of [2-13C]- or [3-13C]aspartate, were higher in GAERS than in NER in all brain regions. As equal amounts of [2-13C]- and [3-13C]aspartate are formed, because of the symmetrical succinate step, only [3-13C]aspartate is presented in Table 1. The amount of [3-13C]alanine was increased in all three brain regions in GAERS compared with NER, although the increase was only statistically significant in the cortex (Table 1). The amount of [3-13C]lactate was similar in both groups in all three regions (Table 1).
Concentration 13C-labeled metabolites derived from [1-13C]glucose (
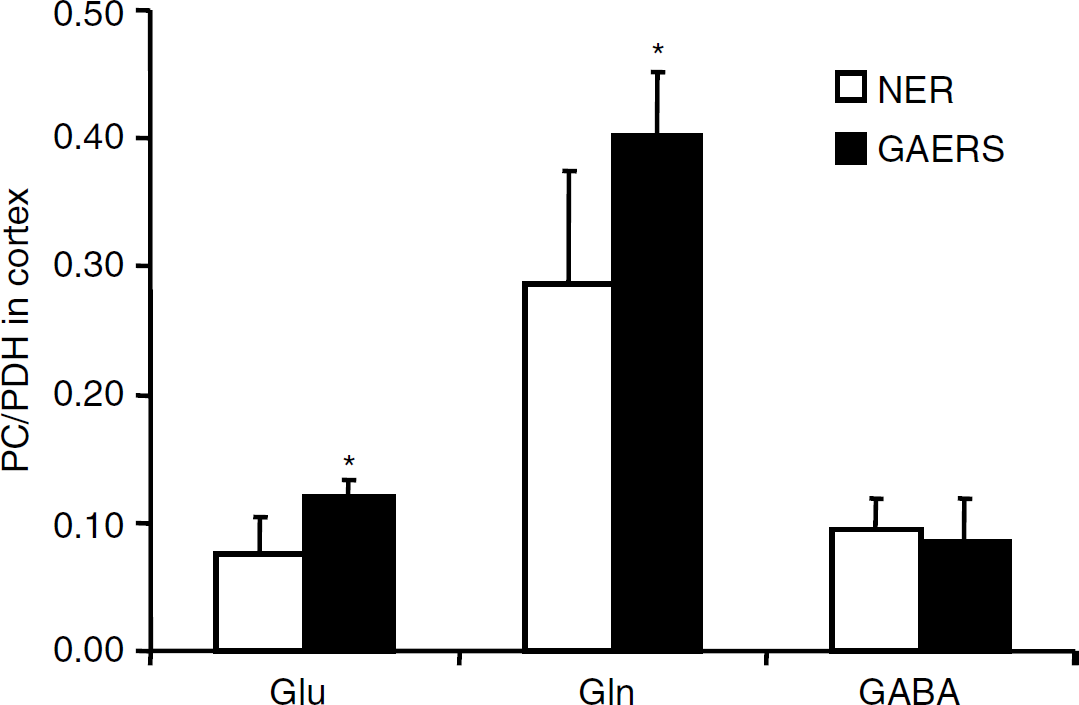
Pyruvate carboxylation over pyruvate dehydrogenation (PC/PDH) ratios for glutamate, glutamine, and gamma aminobyturic acid (GABA) in cortex of genetic absence epilepsy rats from Strasbourg (GAERS) compared with nonepileptic rats (NER). The PC/PDH ratios were calculated as described in Materials and methods. Data represent mean7s.d. of 8 GAERS and 10 NER. *P < 0.05, statistically significant difference between GAERS and NER.
Amounts of 13C-labeled metabolites (nmol/g tissue) in extracts from the cerebral cortex, thalamus, and hippocampus of genetic absence epilepsy rats from Strasbourg (GAERS) compared with nonepileptic rats (NER)
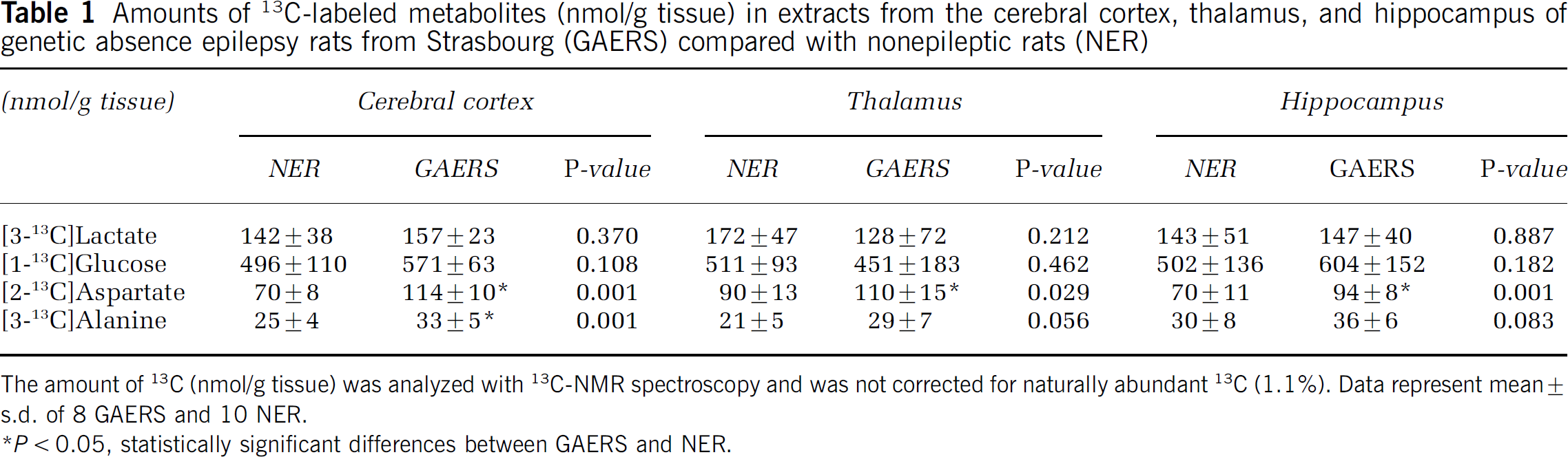
The amount of 13C (nmol/g tissue) was analyzed with 13C-NMR spectroscopy and was not corrected for naturally abundant 13C (1.1%). Data represent mean ± s.d. of 8 GAERS and 10 NER.
P<0.05,statistically significant differences between GAERS and NER.
Labeled metabolites derived from [1,2-13C]acetate: Levels of [4,5-13C]glutamate (Figure 2D), [4,5-13C]glutamine (Figure 2E), and [1,2-13C]glutamate were significantly increased only in the cortex in GAERS compared with NER. The amount of [1,2-13C]glutamine was increased in the cortex and hippocampus in GAERS compared with NER (results not shown). Furthermore, the amount of [1,2-13C]GABA was increased in the hippocampus (Figure 2F).
Methodological Considerations
There was a slight difference in weight between GAERS (314 ± 16g) and NER (366 ± 18g), P < 0.01. This difference is usual when GAERS and NER of the same age are compared (A Nehlig, personal observations). However, levels of [1-13C]glucose and [1,2-13C]acetate in the blood, and thus in the brain, could possibly be affected by this, leading to differences in labeling of metabolites. Nevertheless, the concentrations of [1-13C]glucose in plasma were similar in NER (1.71± 0.73 μmol/mL) and GAERS (1.28 ±1.00 μmol/mL) in the present study. The same was true for [1-13C]glucose levels in brain tissue (Table 1). As for [1-13C]glucose, the amounts of [1,2-13C]acetate in the plasma of NER (668± 172 nmol/mL) and GAERS (735± 373 nmol/mL) were similar. This implies that the differences in the concentrations of metabolites observed between the two groups in the present study were caused by differences in metabolism and not weight.
1H-Nuclear Magnetic Resonance Spectroscopy
The concentrations of various amino acids and other compounds are presented in Table 2. In all brain regions, the amounts of glucose were similar in GAERS and in NER. The levels of glutamine and aspartate were increased in all regions of GAERS compared with NER, whereas the levels of glutamate were increased in the cerebral cortex and the hippocampus only. The concentration of GABA was decreased in all regions investigated in GAERS compared with NER. The concentrations of lactate, N-acetylaspartate (NAA), alanine, creatine, inositol, and nicotinamide adenine dinucleotide (NAD +) were increased in the cortex of GAERS compared with NER.
Metabolite concentration (μmol/g tissue) in extracts from the cerebral cortex, thalamus, and hippocampus of genetic absence epilepsy rats from Strasbourg (GAERS) compared with nonepileptic rats (NER)
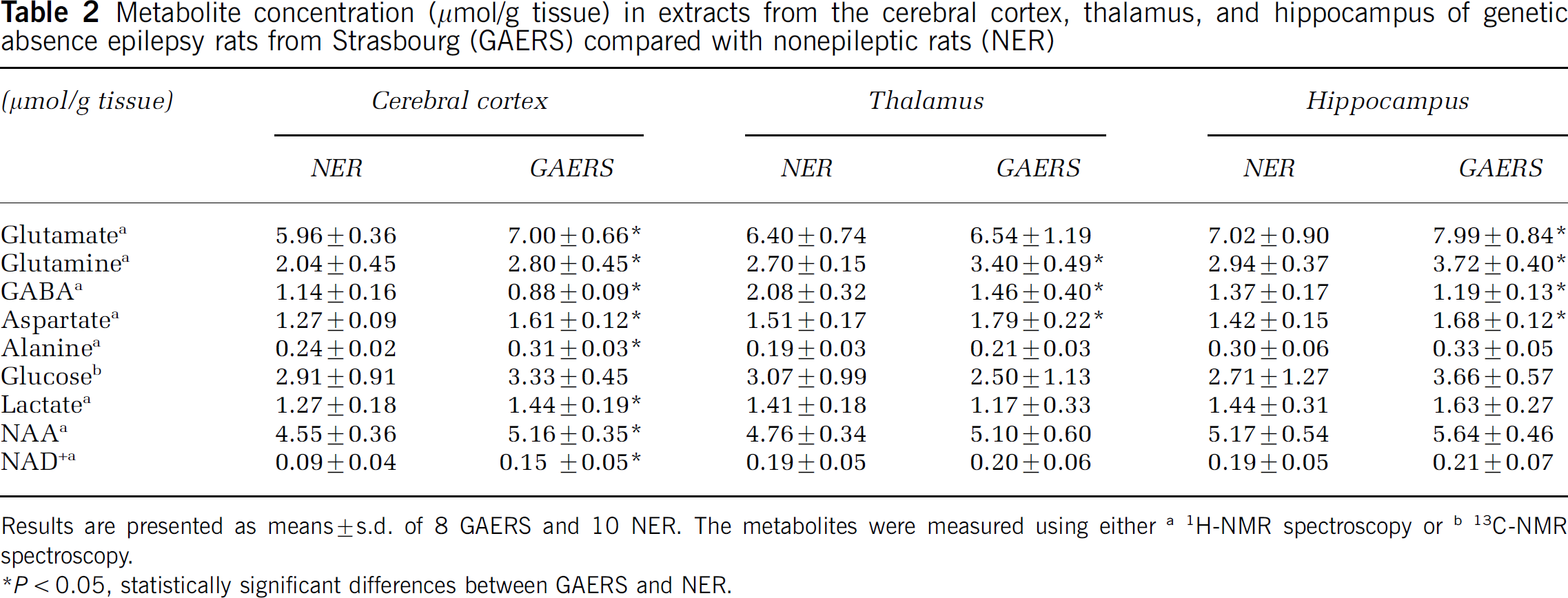
Results are presented as means ± s.d. of 8 GAERS and 10 NER. The metabolites were measured using either a 1H-NMR spectroscopy or b 13C-NMR spectroscopy.
P < statistically significant differences between GAERS and NER.
Discussion
The main finding of the present study is that metabolism appears to be predominantly affected in the cerebral cortex of GAERS, although absence seizures are expressed in both the cortex and thalamus. Surprisingly, while limbic areas, as hippocampus, never express SWDs (Danober et al, 1998), neuronal glutamatergic metabolism was disturbed.
Glycolysis and Mitochondrial Metabolism in the Cortex
Glucose is the major cerebral metabolic substrate and efficient glucose transporters at the blood—brain barrier provide adequate glucose supply to the brain (Vannucci et al, 1997). Absence epilepsy had no effect on glucose concentration (labeled and unlabeled), as measured in the present study. Glucose enters neurons and astrocytes to an equal extent (Nehlig et al, 2004), and lactate can be shuttled from astrocytes to neurons (Serres et al, 2004). As soon as glucose enters a cell, it is phosphorylated and this process is upregulated in GAERS. Indeed, previous studies revealed diffuse and widespread increases in cerebral glucose metabolism in adult GAERS compared with NER (Nehlig et al, 1991, 1998a). Through glycolysis, pyruvate is produced and can be converted (i) to lactate by lactate dehydrogenase, (ii) to alanine by alanine aminotransferase, (iii) to acetyl CoA by PDH, or (iv) to oxaloacetate by PC. In the present study, the amount of unlabeled lactate was increased in the cortex. This increase is in agreement with the increased lactate dehydrogenase activity recorded in this strain (Dufour et al, 2003). Increased use of pyruvate for conversion into labeled and unlabeled alanine was also restricted to the cortex. The conversion of pyruvate to acetyl CoA was also enhanced in the cortex. An increase in neuronal TCA cycle activity was indicated by increased amounts of [4-13C]glutamate, [2-13C]GABA, and [2-13C]aspartate in GAERS. The amount of [4-13C]glutamine was also increased. Even though glutamine synthetase, catalyzing the conversion of glutamate to glutamine, is an astrocyte-specific enzyme (Norenberg and Martinez-Hernandez, 1979), the latter increase reflects neuronal synthesis as [4-13C]glutamine is mostly derived from [4-13C]glutamate (Fitzpatrick et al, 1990). However, a rise in neuronal mitochondrial activity can only take place if the amount of TCA cycle intermediates increases. The latter has to be coupled to increased PC activity in astrocytes, the only anaplerotic enzyme in brain. This prerequisite was fulfilled in GAERS where the PC/PDH ratios for glutamate and glutamine were increased in the cortex compared with NER. Altogether, these data indicate that pyruvate synthesis and utilization have increased significantly, reflecting increased glycolysis in the cortex. Label in acetyl CoA in the present study can originate from [3-13C]pyruvate ([1-13C]glucose) or [1,2-13C]acetate. In cortical astrocytes, conversion of [1,2-13C]acetate to [1,2-13C]acetyl CoA and subsequently to [4,5-13C]glutamate and [4,5-13C]glutamine was increased, reflecting increased TCA cycle activity. The enhanced use of acetyl CoA is coupled to elevated mitochondrial activity as enzymes catalyzing TCA cycle reactions are located in the mitochondria. The increased amount of NAD+ in the cortex correlates well with the increased activity of the mitochondrial enzyme cytochrome oxidase previously reported in GAERS (Dufour et al, 2003). Furthermore, the amount of the neuronal marker NAA, which is synthesized exclusively in the mitochondria (Baslow, 2003) was also increased in the cortex. Altogether, these data show increased mitochondrial activity in both neurons and astrocytes in the cortex of GAERS. In contrast, only few changes were detected in the thalamus. These were confined to metabolism of [1-13C]glucose and consisted of an increased amount of [4-13C]glutamine and [2-13C]aspartate.
Glutamine—Glutamate—GABA cycle in the Cortex and Thalamus
Metabolism and neurotransmission are closely linked, especially through the major excitatory neurotransmitter glutamate, which is derived from the TCA cycle intermediate α-ketoglutarate. Glutamate is primarily synthesized and stored in glutamatergic neurons (Ottersen and Storm-Mathisen, 1985), released upon excitation and removed mostly by uptake into astrocytes (Danbolt, 2001). Once inside the astrocyte, glutamate is converted to glutamine by the astrocyte-specific enzyme, glutamine synthetase (Norenberg and Martinez-Hernandez, 1979), and cycled back to neurons. Analogous reactions occur in GABAergic neurons, where glutamate is converted to GABA. This close metabolic cooperation between astrocytes and neurons is called the glutamine-glutamate-GABA cycle (Berl and Clarke, 1969; Berl and Clarke, 1983).
Glutamatergic neurotransmission was increased in the cortex of GAERS compared with NER, as reflected by elevated glutamate concentration. Interestingly, this was not observed in thalamus of GAERS. Moreover, increased amounts of glutamate derived from [1-13C]glucose and [1,2-13C] acetate were seen in cortical neurons of GAERS. The amount of [4,5-13C]glutamine was also increased in the cortex, implying that also astrocytic metabolism was increased, possibly in response to increased neuronal activity in this brain area. Previous data support the idea of an upregulation in glutamate transmission in GAERS. On the receptor level, GAERS display enhanced cortical excitability as a result of both increased NMDA and non-NMDA currents (Pumain et al, 1992; Avanzini et al, 1996). Increased expression of GFAP, an astrocyte marker was reported in the thalamocortical structures of GAERS (Dutuit et al, 2000). Although glutamine synthetase protein expression was unchanged (Dutuit et al, 2000), glutamine content was increased in GAERS in the thalamus and cortex in the present study. It should be kept in mind that protein levels do not necessarily reflect enzyme activity. This difference may reflect subtle alterations in the levels of inhibitors and activators regulating the activity of glutamine synthetase in GAERS. Increased activity of another astrocyte-specific enzyme, PC, was detected in the cortex, as the PC/PDH ratios for glutamate and glutamine were increased. This increase could also be caused by a decrease in PDH activity; however, this was not the case as acetyl CoA formation was increased. The fact that both glutamate and glutamine labeling (neuronal and astrocytic) and content were increased in the cortex indicates an increased glutamine-glutamate cycle caused by increased neuronal and astrocytic activity. In the thalamus, the glutamine content and labeling from [4-13C]glutamate were increased, whereas the glutamate content and labeling were unchanged, implying a less disturbed glutamate-glutamine cycle. A further indication supporting this observation relates to the fact that the PC/PDH ratios were similar in the thalamus in GAERS and NER. This indicates that neuronal mitochondrial metabolism was not enhanced as in the cortex.
The glutamate-glutamine shuttling between astrocytes and glutamatergic neurons was clearly upregulated in the cortex of GAERS. The increase in percent enrichment in glutamate is probably the cause for increased GABA labeling in this brain region. Conversely, GABA concentration was decreased in cortex. To our knowledge, there are no reports of any change in GABAergic metabolism in the cortex. On the receptor level, both GABAA and GABAB seem to be critical in the generation and control of SWDs (Danober et al, 1998), but the exact role of GABA in the genesis of absence seizures needs further clarification. Total GABA concentration was also decreased in the thalamus of GAERS in the present study, whereas it was increased at the extracellular level (Richards et al, 2000) and GABA uptake was reduced in thalamic synaptosomes (Sutch et al, 1999). This apparent discrepancy could be explained by increased glutamatergic signaling from the cortex to thalamus causing increased release of GABA (Richards et al, 2000). As uptake is impaired in the thalamus (Sutch et al, 1999), GABA may diffuse away leading to reduced total amount in this structure. Another cause for decreased GABA concentration in the thalamus could be increased glutamate dehydrogenase activity (Dutuit et al, 2000). The latter could indirectly be responsible for the decrease in GABA content by decreasing the amount of glutamate used for GABA synthesis in this region. It should be noted that GABA concentration is highest in the thalamus, indicating its importance in this brain area. The thalamus, receiving all sensory information before it reaches the cortex, acts as a filter controlling sensory input, and allows only the relevant information to reach the cortex (Castro-Alamancos, 1997). The thalamus is also critical for the regulation of states of vigilance, which in turn can influence the ability of the cortex to receive and process information (Canedo, 1997).
We postulate that the decrease in GABA concentration as well as the increase in glutamatergic function in the cortex might lead to increased glutamatergic input from the cortex to thalamus. Glutamatergic axons from the cortex synapse onto GABAergic and glutamatergic neurons in the thalamus (Danober et al, 1998) and regulate the thalamic output underlying balanced interpretation of sensory stimulation (Gaudreau and Gagnon, 2005) (Figure 4A). In the cortex of GAERS, increased glutamate-glutamine metabolism in neurons and astrocytes will lead to enhanced glutamatergic output to the thalamus. This dysregulation leads to impaired thalamic filter function and hence reduced sensory input to cortex allowing the occurrence of SWDs in the thalamocortical loop (Figure 4B). The resulting disturbance in the thalamocortical pathway is in good agreement with the fact that SWDs only occur in a specific state of calm wakefulness (Danober et al, 1998). The present data show that metabolic activity was largely increased in the cortex of GAERS and slightly affected in the thalamus. These predominant cortical changes support the recent hypothesis that the expression of SWDs within the thalamocortical loop is driven by a cortical focus (Meeren et al, 2002, 2005). Alternately, the changes recorded here may also originate from an increase in cortical drive to the thalamic reticular nucleus. Indeed, GABAergic neurons are crucial in inhibiting external signals through thalamocortical neurons leading to loss of contact with the surrounding world during absence epilepsy (Steriade, 2005). However, because of 13C-NMR spectroscopy's sensitivity limitations, it is not possible to study specific thalamic nuclei.
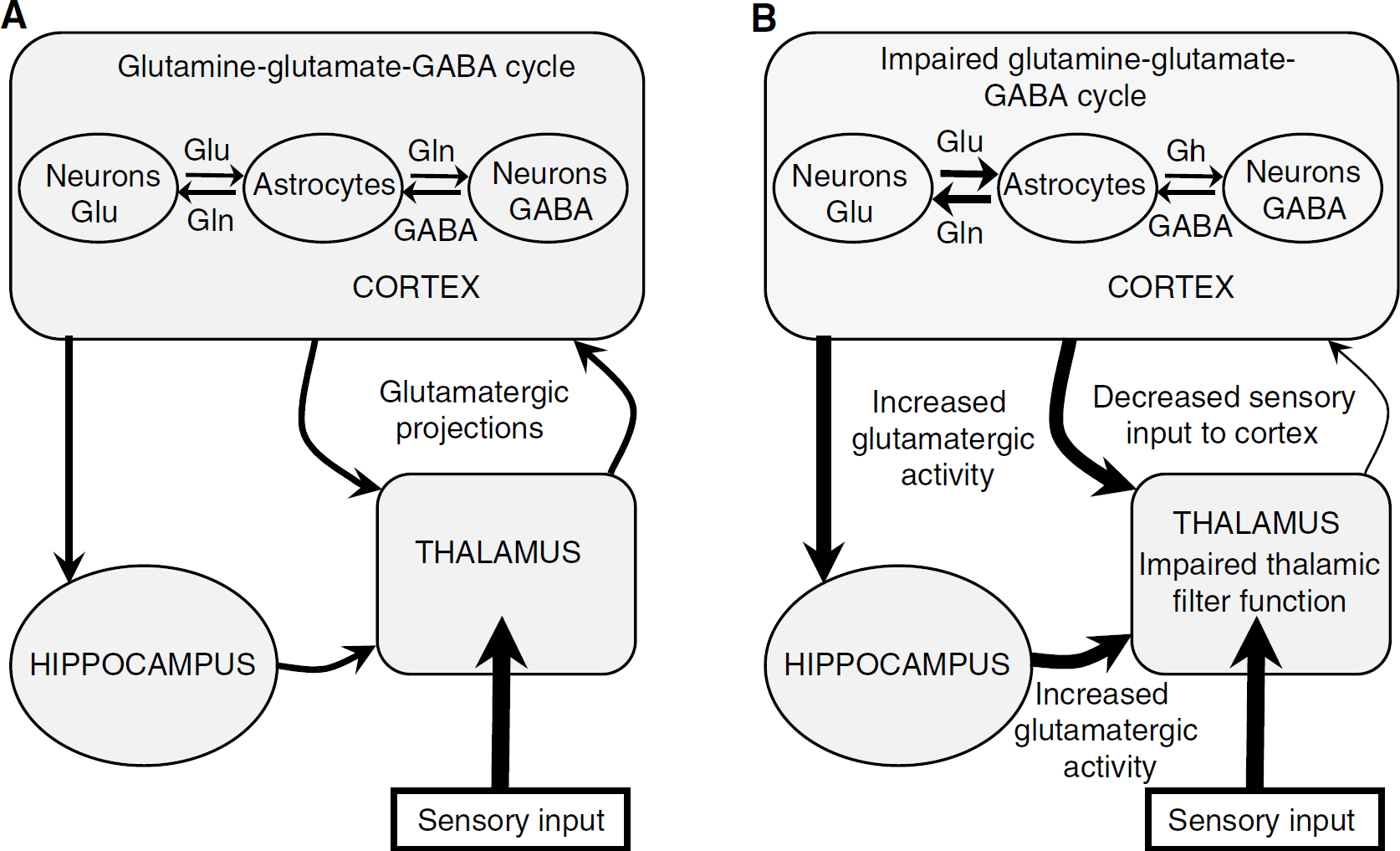
Schematic presentation of the glutamine—glutamate—gamma aminobyturic acid (GABA) cycle in the thalamocortical loop in genetic absence epilepsy rats from Strasbourg (GAERS) compared with nonepileptic rats (NER). (
Changes in the Hippocampus
As described above, absence seizures are restricted to the thalamocortical loop. However, the mutations underlying the expression of absence epilepsy in the GAERS strain are present in all brain cells. Therefore, it is not surprising that metabolic changes occurred also in the hippocampus of GAERS. These changes were very similar to those observed in the cortex in the present study. This is in agreement with the diffuse increase in cerebral glucose utilization in these animals compared with NER (Nehlig et al, 1991, 1998a). The specific reasons why we chose to study hippocampus were threefold: firstly, this structure is located outside of the pacemaker regions of this epilepsy and is disturbed early in the development of the GAERS. Indeed, in young GAERS, where SWDs are not expressed, glucose utilization is specifically increased mostly in anterior limbic structures including all hippocampal subfields (Nehlig et al, 1998b). Secondly, glutamatergic neurotransmission has been reported to be disturbed in the hippocampus of GAERS. Indeed, basal hippocampal extracellular levels of glutamate were increased in GAERS compared with control NER (Richards et al, 2000). This was paralleled by a decrease in glutamate concentration in the mossy fiber terminals of GAERS (Sirvanci et al, 2005), possibly as a result of an alteration in metabolism or reuptake of glutamate in this particular area (Richards et al, 2000). These data are in accordance with our present findings of increased glutamate metabolism in the hippocampus. Moreover, as in the present study, the density of GABA in GABAergic terminals is similar in both the hippocampal CA3 area (Sirvanci et al, 2003) and in the dentate gyrus of GAERS and NER (Sirvanci et al, 2005). This is accompanied by similar extracellular levels of GABA in the hippocampus of both strains (Richards et al, 2000). Altogether, enhanced glutamatergic metabolism in the hippocampus might reflect the consequences of the mutations underlying this epilepsy and either the well-known sensitivity of this structure to seizures or a possible indirect role of this structure in the modulation and/or expression of SWDs. Finally, the hippocampus is involved in learning and memory (Squire, 1992), and it was shown that GAERS perform better in learning tasks than NER (Vergnes et al, 1991; Getova et al, 1997). Memory function in the hippocampus is largely mediated by glutamatergic neurotransmission (Steckler et al, 1998). As detailed in Figure 4, increased glutamatergic function in the hippocampus could lead to increased stimulation of NMDA receptors and hence improved learning and cognition.
Conclusion
The present results support the idea that increased cycling of glutamate and glutamine between astrocytes and glutamatergic neurons combined with decreased GABAergic function in the cortex of GAERS may be the underlying cause of absence seizures. These changes are accompanied by enhanced glycolysis and mitochondrial function.
Footnotes
Acknowledgements
The kind help of JC Cassel in the use of his microwave device as well as the excellent technical assistance of Estelle Koning are greatly acknowledged. We also thank Fostein Krane for advice and assistance with the NMR analysis.