Abstract
Heparin is a natural proteoglycan that was first described in 1916. In addition to its well characterized effect on blood coagulation, it is becoming clear that heparin also modulates inflammatory processes on several levels, including the interference with leukocyte–endothelium interaction. Anecdotal observations suggest a better clinical outcome of heparin-treated patients with bacterial meningitis. The authors demonstrate that heparin, a glycosaminoglycan, inhibits significantly in the early phase of experimental pneumococcal meningitis the increase of 1) regional cerebral blood flow (125 ± 18 versus 247 ± 42%), 2) intracranial pressure (4.5 ± 2.0 versus 12.1 ± 2.2 mm Hg), 3) brain edema (brain water content: 78.23 ± 0.33 versus 79.49 ± 0.46%), and 4) influx of leukocytes (571 ± 397 versus 2400 ± 875 cells/μL) to the cerebrospinal fluid compared with untreated rats. To elucidate the possible mechanism of this observation, the authors investigated for the first time leukocyte rolling in an inflammatory model in brain venules by confocal laser scanning microscopy in vivo. Heparin significantly attenuates leukocyte rolling at 2, 3, and 4 hours (2.8 ± 1.3 versus 7.9 ± 3.2/100 μm/min), as well as leukocyte sticking at 4 hours (2.1 ± 0.4 versus 3.5 ± 1.0/100 μm/min) after meningitis induction compared with untreated animals. The authors conclude that heparin can modulate acute central nervous system inflammation and, in particular, leukocyte–endothelium interaction, a key process in the cascade of injury in bacterial meningitis. They propose to evaluate further the potential of heparin in central nervous system inflammation in basic and clinical studies.
Despite potent antibiotics that effectively treat the bacterial infection (Swartz, 1984), meningitis caused by Streptococcus pneumoniae is associated with a morbidity rate of 50% (Bohr et al., 1983) and a surprisingly high mortality rate of 30% (Pfister et al., 1993). Cerebrovascular changes (Pfister et al., 1992), brain edema, and high intracranial pressure (for review see Quagliarello and Scheld, 1992) cause a poor clinical outcome. Leukocytes crossing the blood–brain and blood–cerebrospinal fluid (CSF) barrier significantly contribute to nervous system injury. Tuomanen et al. (1989) demonstrated that a monoclonal antibody directed against the CD18 complex prevents leukocyte influx in the CSF, reduces brain–edema and blood–brain barrier disruption, and enhances survival in a rabbit model of pneumococcal meningitis. Reduction of leukocytes in the CSF did not affect bacterial killing by antibiotics (Saez Llorens et al., 1991; Tuomanen et al., 1989). Cytokine-induced meningitis is attenuated in mice deficient in P-selectin. Mice doubly deficient in P- and E-selectins displayed a near complete reduction of leukocyte influx to the CSF and blood–brain barrier permeability (Tang et al., 1996). Granert et al. (1994) found that the sulfated polysaccharide fucoidin, a substance interfering mainly with L-selectin function (Kansas et al., 1991), reduces pleocytosis and decreases plasma proteins in the CSF in a rabbit model of pneumococcal meningitis. Prokaryotic peptides that mimic selectins inhibit the recruitment of leukocytes into the CSF, possibly by reducing CD11b/18 upregulation (Rozdzinski et al., 1993).
The early phase of experimental pneumococcal meningitis is characterized by an increase in cerebral blood flow (Pfister et al., 1990; Weber et al., 1995), which is essential for the development of inflammation (Mantovani et al., 1994) and may contribute to leukocyte adhesion, brain edema, and raised intracranial pressure (ICP). The causes of this hemodynamic change during the early phase of bacterial meningitis are not understood fully. There is growing evidence that nitric oxide is a central mediator in this process (Koedel et al., 1995; Haberl et al., 1994). Invading leukocytes are thought to be a major source of nitric oxide (Moncada et al., 1991) and superoxide radicals. This is supported by our findings that substances interfering with leukocyte rolling (Angstwurm et al., 1995) or firm adhesion (Weber et al., 1995) inhibit the regional cerebral blood flow (rCBF) increase in experimental bacterial meningitis.
Heparin has been used as an anticoagulant for more than 50 years and is one of the most common drugs in clinical use. Heparin is a natural proteoglycan produced by mast cells and was first described by McLean (1916). It is categorized as a glycosaminoglycan, one of a group of polyanionic polysaccharides containing glucosamine and galactosamine (Kjellen et al., 1991). It can modulate a variety of biologic processes apart from hemostasis (for review Tyrell et al., 1995, and Lindahl et al., 1994). Recent reports demonstrate that crude heparin and heparan sulfate (HS) can inhibit leukocyte rolling along the vessel wall (Tangelder and Arfors, 1991; Ley et al., 1991), a process known to involve selectins. Heparin significantly reduced leukocyte influx to the peritoneal cavity in thioglycolate-induced peritonitis (Nelson et al., 1993). L- and P-selectin binding to immobilized BSA-sLEx was inhibited by crude heparin and HS (Nelson et al., 1993). Adhesion of human neutrophils to COS cells transfected with cDNA-encoding P- but not E-selectin was inhibited by heparin (Nelson et al., 1993).
Clinical evidence for a superior outcome of heparin-treated patients with meningitis is controversial and episodic (Haring et al., 1993; Goldman, 1994; Winkler et al., 1994;). Vascular complications and septic sinus venous thrombosis are well-known complications of bacterial meningitis (Pfister et al., 1992) in advanced stages and may benefit by the anticoagulant properties of heparin.
The objectives of our study were to determine the effects of heparin and HS on inflammatory parameters. Our data demonstrate that heparin and, in part, HS reduce the rCBF and ICP increase, brain edema, and leukocyte influx to the CSF. Based on these findings, we visualized leukocyte rolling in postcapillary venules of the brain by confocal laser scanning microscopy in vivo and demonstrated that heparin indeed inhibits leukocyte rolling and subsequent firm adhesion of leukocytes in the early phase of experimental pneumococcal meningitis.
METHODS
General Preparation
In the first set of experiments (study I), 54 male Wistar rats were anesthetized with sodium thiopental 100 mg/kg (Trapanal, Byk Gulden, Konstanz, Germany; additional doses of 20 mg/kg were administered whenever a mean arterial blood pressure increase of >10 mm Hg was observed to a painful stimulus on the rat's tail) and mechanically ventilated with room air and supplemental oxygen throughout the experiment by a small animal ventilator (AP-10, K. Effenberger, Pfaffing, Germany). The left femoral artery and vein were cannulated for physiologic monitoring and intravenous saline infusion (1 mL/h). Body temperature was controlled and maintained at 38.0° ± 0.5°C using a heating pad. Mean arterial blood pressure was measured continuously using a pressure transducer (Statham P10 EZ, Spectramed, Oxnard, CA, U.S.A.) connected to the femoral artery cannula. End-expiratory partial pressure of carbon dioxide (PaCO2; Heyer Artema MM 204, Bad Ems, Germany) was monitored continuously. Arterial blood samples were analyzed for partial arterial oxygen pressure (PaO2), PaCO2, and pH (Compact 1, AVL List, Graz, Austria) at the beginning and the end of each experiment, as well as every 2 hours.
Cerebral spinal fluid samples were collected for leukocyte counts before and 6 hours after the intracisternal injection and counted in a Fuchs Rosenthal chamber (Poly-Optik, Bad Blankenburg, Germany). Intracranial pressure was measured throughout the experiment with a pressure transducer (Statham P109 EZ, Spectramed, Oxnard, CA, U.S.A.) connected to a cisterna magna catheter placed through a burr hole in the occipital bone (Weber et al., 1995; Pfister et al., 1990).
Mean arterial blood pressure, ICP, rCBF (see next section), and end-expiratory PaCO2 were digitized and stored on a computer. After 6 hours, the animals were killed by exsanguination, and brain water content was determined as described previously (Pfister et al., 1990).
Regional Cerebral Blood Flow Measurements
Regional cerebral blood flow was measured continuously over the right parietal cortex by a laser Doppler flowmeter (Periflux 4001 Master, Järfälla, Sweden). The laser Doppler flowmeter probe was held by a micromanipulator and positioned over the thinned bone (Lindauer et al., 1993) in an area free of large pial vessels. Regional cerebral blood flow was expressed as a percentage of stable baseline (defined as 100%) recorded for 30 minutes after the placement of the intracisternal catheter.
The design of study I was as follows. In three groups of eight rats, we studied the effect of intracisternally injected pneumococcal cell walls (PCW, see following section) and the vehicle of PCW (0.9% saline). In both groups, the rats were untreated (vehicle infusion of 0.6 mL 0.9% saline over 5 minutes) administered intravenously immediately before intracisternal injection of PCW or vehicle (0.9% saline) and continued with an infusion rate of 1 mL/h). In the third group, PCW were instilled intracisternally and the rats were treated with heparin (Sigma H 3393, 170 units/mg, 200 units/kg in 0.6 mL 0.9% saline over 5 minutes administered intravenously immediately before intracisternal injection of PCW and continued at 25 units/kg/h in 0.9% saline with an infusion rate of 1 mL/h). In three other groups of six rats, vehicle intracisternally challenged and heparin-treated rats and PCW intracisternally injected and heparinposttreated rats (dose see above, starting the treatment 2 or 4 hours after meningitis induction) were examined. Additionally, we studied the effect of two different doses of HS (Sigma H 7641, 1 mg/kg every 2 h/0.2 mL, n = 4; and 4 mg/kg every 2 h/0.2 mL, n = 4) in rats injected intracisternally with PCW.
Leukocyte Imaging
In a second set of experiments (study II) 21 male Wistar rats were anesthetized and monitored as described previously. Once placed in the stereotaxic frame, a closed cranial window was implanted as described earlier (Lorenzl et al., 1993; Villringer et al., 1991). A 6-mm × 4-mm craniotomy was performed in the mediocaudal portion of the right parietal bone where the laser Doppler flowmeter probe was placed in study I. The dura remained intact. A wall of bone wax (Ethicon, Norerstedt, Germany) was formed around the craniotomy and closed by a coverslip. Before closing the window, polyethylene tubes were inserted frontally and occipitally for continuous superfusion of artificial CSF (2 mL/h) using a perfusion pump. Artificial CSF was equilibrated with gas containing carbon dioxide (5%), oxygen (12%), and nitrogen (83%), which gives a comparable gas tension and pH to normal CSF (Levasseur et al., 1975).
Circulating leukocytes were stained by rhodamine 6 G labeling. Rhodamine 6 G selectively stains leukocytes and thrombocytes (Villringer et al., 1991). Rhodamine 6 G dye (2 mg, Aldrich, Milwaukee) was dissolved in 5 mL of 0.9% saline, and 0.5 mL was administered intravenously over 5 minutes at the beginning of the experiment, followed by continuous intravenous infusion of 0.5 mL per hour.
The emission wavelength of rhodamine 6 G is between 530 and 540 nm. A confocal laser scanning microscopy system with a Nikon microscope (Nikon, Tokyo, Japan) with a water immersion lens 40×, numerical aperture 0.75, and a working distance of 1.6 mm was used. The laser source was a crypton argon laser with principle lines at 488, 568, and 647 nm. For rhodamine 6 G excitation, only the 488-nm line was used. For fluorescence emission detection, the proper filter sets were used. A computer controlled x-y-z stage was used to scan and to return to multiple focal areas of interest on the cortex during the experiment, with an accuracy of 100 nm in every dimension. At the beginning of the microscopic procedure, six cortical areas (each 210 × 140 μm) containing venules and arterioles were selected randomly and imaged continuously for 1 minute. The frame rate was 16 per second. Each position was stored on a computer and reselected every hour after intracisternal injection. After on-line digital analog conversion, the images were recorded on a videorecorder and analyzed off-line. Rolling leukocytes (“rollers“) were determined by counting the number of leukocytes moving slowly along the endothelium (Dirnagl et al., 1994). Adherent leukocytes were defined by a stationary contact to the endothelium for >10 seconds (Lorenzl et al., 1993; Dirnagl et al., 1994). The number of rolling or adherent leukocytes was expressed per 100 μm length/minute. The inner diameters of the venules were between 25 and 80 μm.
In this set of experiments, we studied three groups of seven rats, one injected intracisternally with vehicle of PCW, one with PCW, and one with PCW and heparin treatment (Sigma H 3393, 200 units/kg in 0.5 mL over 5 minutes administered intravenously at the beginning and continued at a dose of 25 units/kg/h, with an infusion rate of 0.5 mL/h). Compared with study I, where we measured from PCW inoculation for 6 hours, in study II we monitored for 4 hours.
Meningitis induction
Pneumococcal cell walls were instilled into the cisternal space to produce inflammatory changes similar to bacterial meningitis (Pfister et al., 1992; Pfister et al., 1990). The PCW were produced as described by Tuomanen (Tuomanen et al., 1985) and modified as described earlier (Weber et al., 1995). An unencapsulated strain (PnR-527, Jena, Germany) of Streptococcus pneumoniae was cultivated overnight on Columbia agar plates, suspended in pyrogen-free saline, and heat inactivated. The inactivated bacteria were disintegrated by ultrasound, purified by centrifugation and washing, and again suspended in pyrogen-free saline. The optical density of the final preparation was 0.66 at 620 nm, which correlates to 107 colony-forming units/mL. Gram staining demonstrated fewer than 1% intact cells. The pH of the suspension was 7.4.
Statistical Methods
Data are expressed as means ± standard deviation. To compare rCBF and ICP changes, we used a repeated measures analysis of variance. To compare groups at 2, 4, and 6 hours, or at 6 hours and for brain water content, a one-way analysis of variance was used with a Scheffé multiple comparison test. A correction for repeated measures (over time) was used were appropriate according to the Bonferoni–Holm sequential procedure. For comparing leukocyte influx into the CSF and systemic leukocyte count, and for comparing the HS-treated animals (n = 4/group) to untreated rats, Kruskal–Wallis H and Mann–Whitney U tests were used. A P value of < 0.05 was considered significant. Statistical evaluations were done using SPSS 6.1 for Windows statistical software (SPSS Inc., Chicago, IL, U.S.A.).
RESULTS
Physiologic Data
Values for mean arterial blood pressure, PAO2, PACO2, and pH were within normal ranges throughout the experiment and are shown in Tables 1 and 2. Small differences between groups were not statistically significant. In all other experiments, these physiologic data also were normal, with no significant differences between groups (data not shown). Systemic leukocyte counts were not different in controls (0 hours: 5789 ± 1131 cells/μL; 6 hours, 5840 ± 1051 cells/μL), heparin-treated (0 hours, 6720 ± 1817 cells/μL; 6 hours, 6260 ± 1530 cells/μL), PCW intracisternally injected (0 hours, 5800 ± 752 cells/μL; 6 hours, 6420 ± 605 cells/μL), and PCW intracisternally injected and heparin-treated (0 hours, 5442 ± 562 cells/μL; 6 hours, 6200 ± 2576 cells/μL) rats. Systemic leukocyte counts for heparin post-treatment, HS, and confocal laser scanning microscopy experiments were similar (data not shown). No significant differences were detected by Kruskal–Wallis H non-parametric test.
Physiological variables for experimental groups in study I according to hours after ic injection
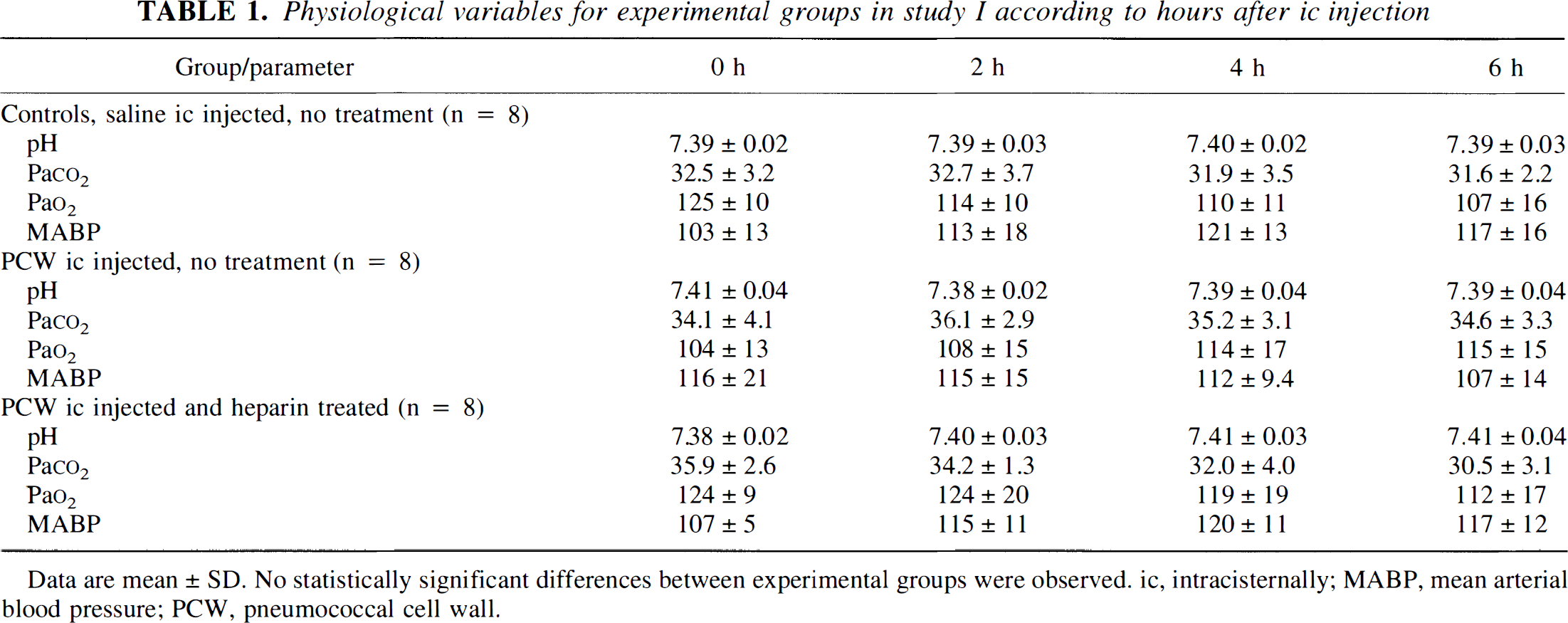
Data are mean ± SD. No statistically significant differences between experimental groups were observed. ic, intracisternally; MABP, mean arterial blood pressure; PCW, pneumococcal cell wall.
Physiological variables for study II according to hours after ic injection
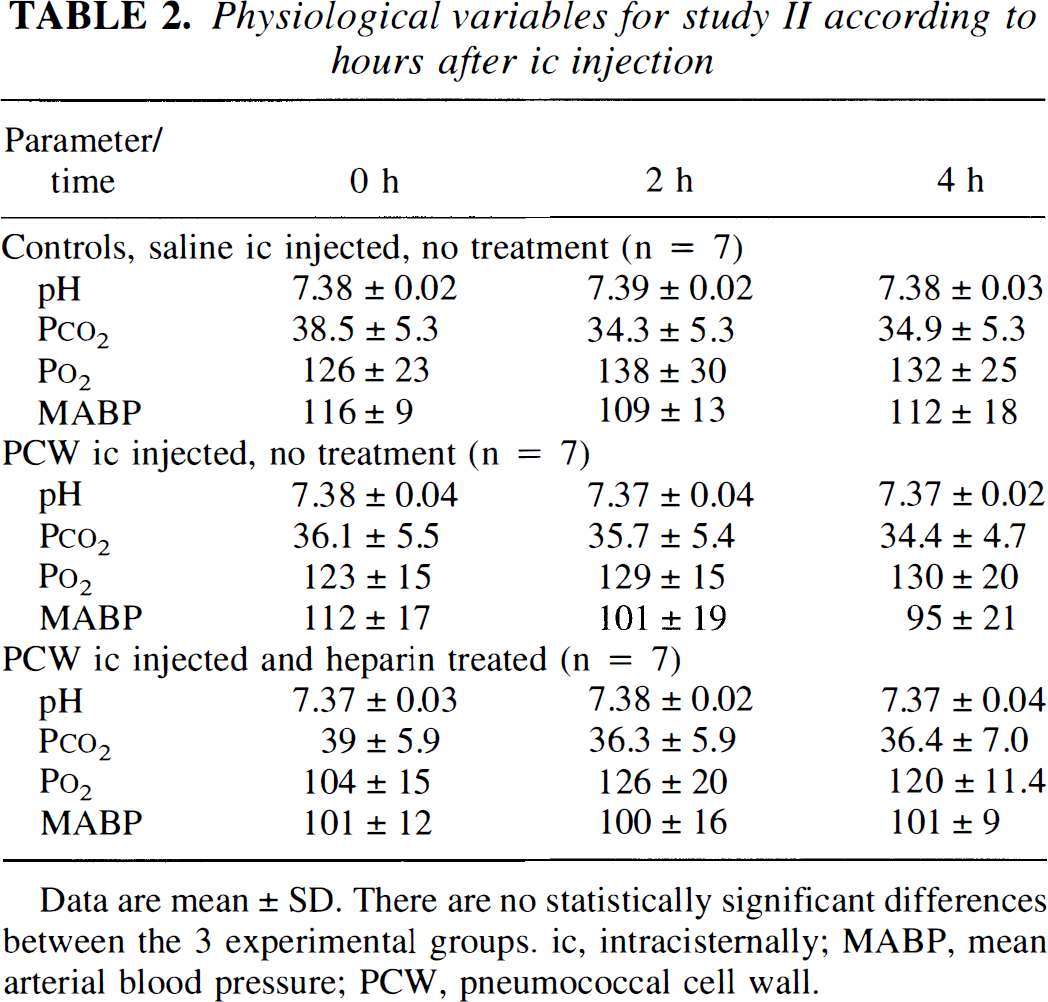
Data are mean ± SD. There are no statistically significant differences between the 3 experimental groups. ic, intracisternally; MABP, mean arterial blood pressure; PCW, pneumococcal cell wall.
Effect of Heparin and Heparan Sulfate on Regional Cerebral Blood Flow, Intracranial Pressure, Brain Edema and Leukocyte Influx
Consistent with meningitis, rCBF, ICP, brain edema, and leukocytes in the CSF increased significantly after PCW instillation in untreated animals (Figs. 1B and 1C, Table 3). Vehicle injection or vehicle injection and heparin treatment did not influence any of the measured parameters (Figs. 1A, Table 3)
ICP and rCBF for experimental groups according to hours after ic injection
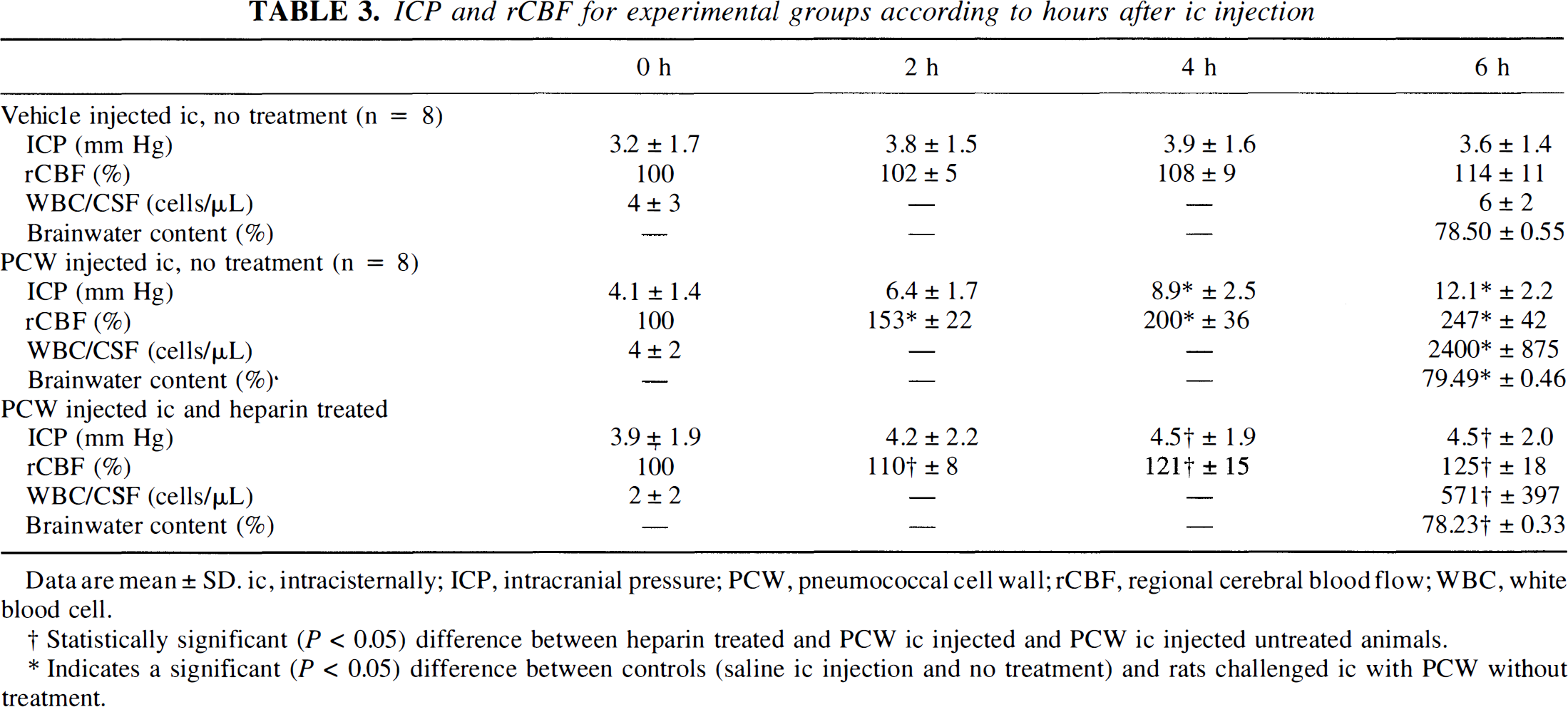
Data are mean ± SD. ic, intracisternally; ICP, intracranial pressure; PCW, pneumococcal cell wall; rCBF, regional cerebral blood flow; WBC, white blood cell.
Statistically significant (P < 0.05) difference between heparin treated and PCW ic injected and PCW ic injected untreated animals.
Indicates a significant (P < 0.05) difference between controls (saline ic injection and no treatment) and rats challenged ic with PCW without treatment.
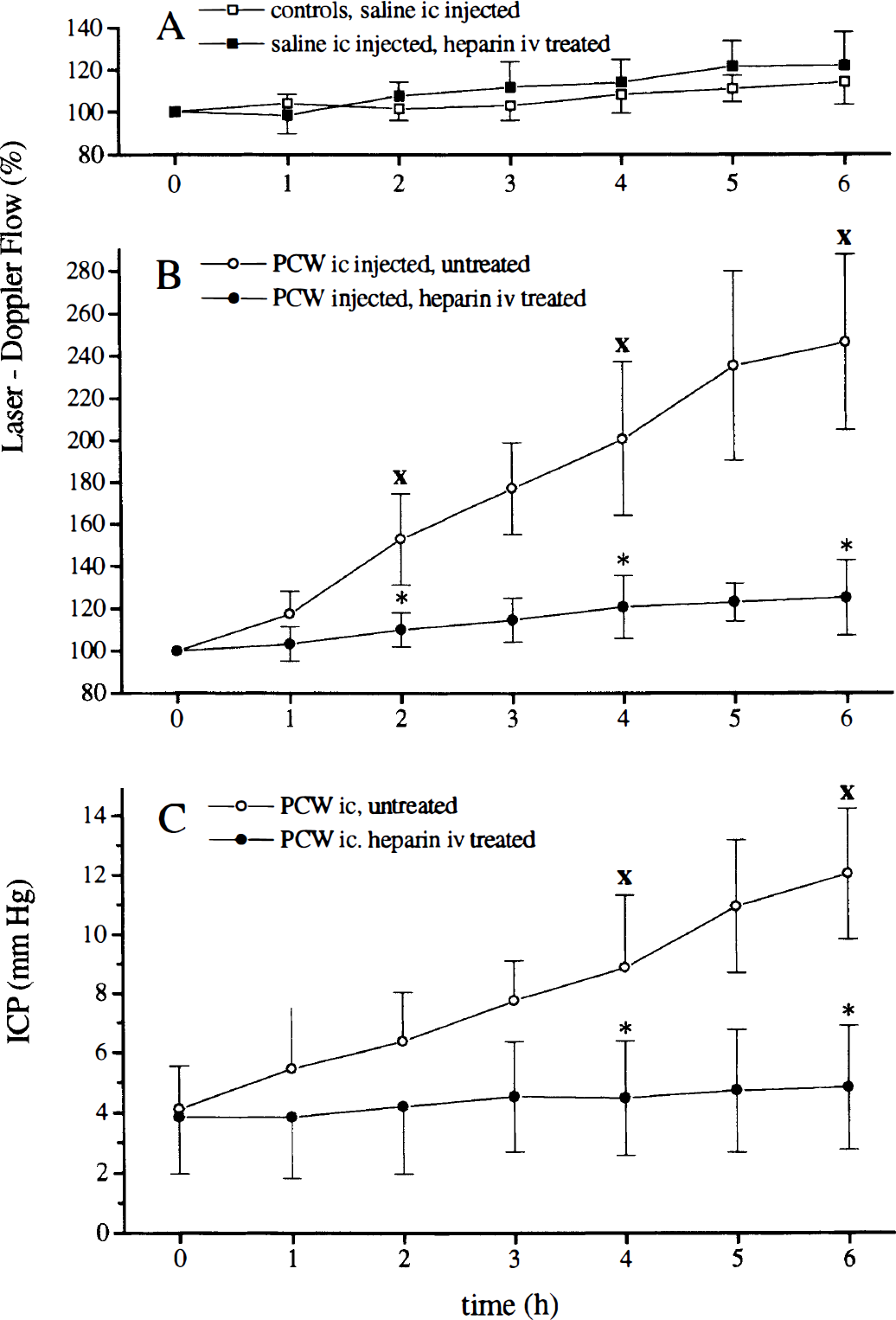
Regional cerebral blood flow (rCBF) response in controls and in rats injected intracisternally with pneumococcal cell walls (PCW).
Heparin treatment in PCW-challenged animals reduced the rCBF, and ICP increased (Fig. 1B, Table 3). Brain edema and WBC influx to the CSF were reduced significantly by heparin treatment (Table 4). A treatment starting 2 hours after meningitis induction attenuated the RCBF and the ICP increase compared with untreated animals at 6 hours (Table 4). Brain edema formation and WBC influx to the CSF also were significantly inhibited (Table 4). A treatment starting 4 hours after intracisternal instillation of PCW inhibited a further increase of rCBF, but had no effect on ICP increase, brain edema, and leukocyte influx to the CSF. Regional cerebral blood flow was approximately 30% lower compared with untreated rats. We observed a dose-dependent effect of HS on these parameters (Table 4), but HS was less effective than heparin.
ICP, rCBF, WBC in the CSF, and brain water content at 6 hours in heparin posttreated and HS-treated animals
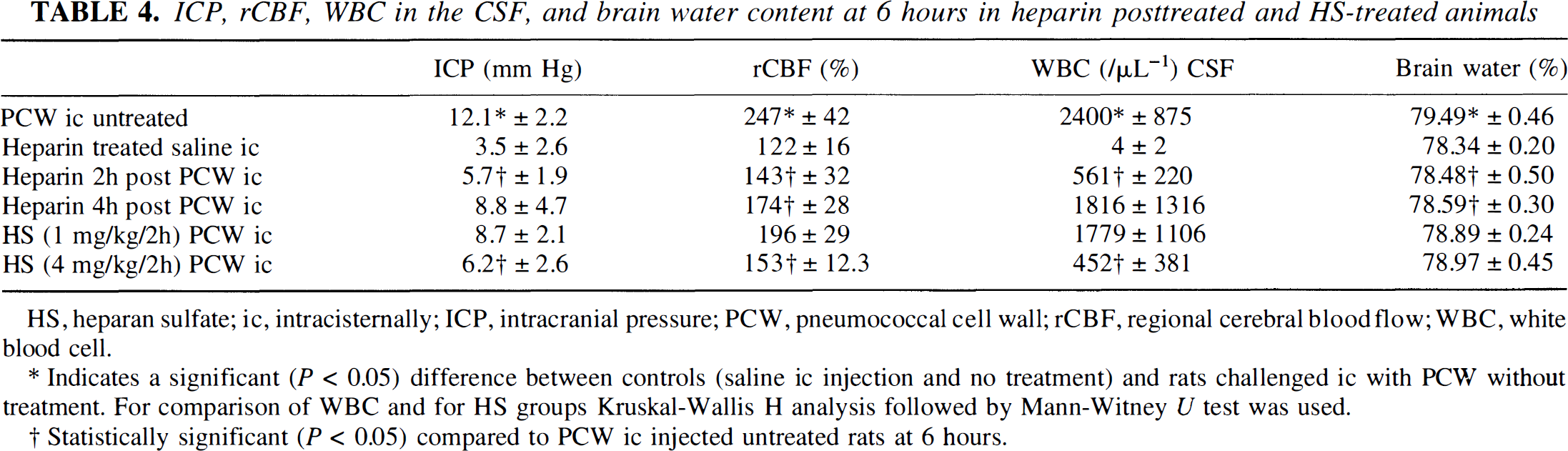
HS, heparan sulfate; ic, intracisternally; ICP, intracranial pressure; PCW, pneumococcal cell wall; rCBF, regional cerebral bloodflow; WBC, white blood cell.
Indicates a significant (P < 0.05) difference between controls (saline ic injection and no treatment) and rats challenged ic with PCW without treatment. For comparison of WBC and for HS groups Kruskal-Wallis H analysis followed by Mann-Witney U test was used.
Statistically significant (P < 0.05) compared to PCW ic injected untreated rats at 6 hours.
Effect of Heparin on Leukocyte Rolling
Vehicle instillation to the cisterna magna did not result in a significant increase of leukocyte rolling. Pneumococcal cell wall injection induced leukocyte rolling and later firm adhesion to the endothelium (Figs. 2A, 2B, and 3) 2 hours after meningitis induction until 4 hours (end of the experiment). This interaction was attenuated by intravenous heparin treatment compared with untreated rats (Fig. 2A). The heparin effect was not associated with mean arterial blood pressure changes (Table 1). Subsequent firm adhesion of leukocytes was significantly reduced at 4 hours compared with the untreated animals (Fig. 2B, Table 4).
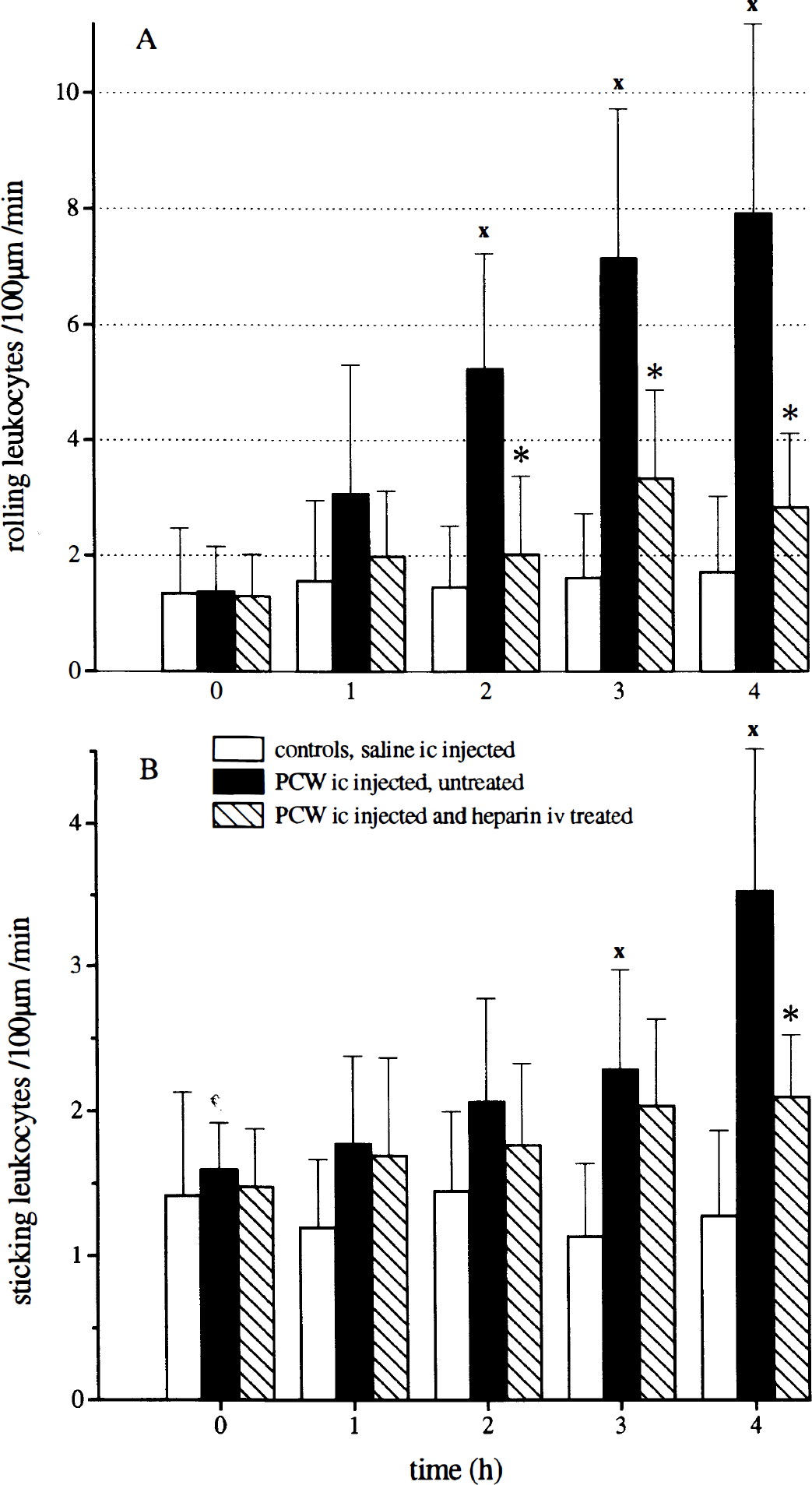
Number of rolling leukocytes
Rhodamine 6 G labeled intravascular leukocytes in untreated rats at 2 hours
DISCUSSION
Our data indicate the importance of leukocyte rolling as a central step in the pathophysiology of bacterial meningitis. Heparin, a glycosaminoglycan, inhibited leukocyte rolling, firm adhesion, and subsequently the increase of rCBF, ICP, brain edema, and invasion of leukocytes to the CSF in experimental pneumococcal meningitis.
Leukocyte–endothelium interaction, which promotes the rapid invasion of activated leukocytes across the blood–brain and blood–CSF barrier, is a central step in the pathophysiology of bacterial meningitis.
Leukocyte Rolling in Bacterial Meningitis
Substances interfering with leukocyte rolling, like fucoidin, reduce leukocyte count (Angstwurm et al., 1995; Granert et al., 1994) and protein influx (Granert et al., 1994) to the CSF and ameliorate the rCBF increase over the parietal cortex and the rise of ICP and brain edema (Angstwurm et al., 1995). Fucoidin treatment reduced rolling in the tenuissimus muscle of the rabbit, but its effect has not been studied in brain venules (Granert et al., 1994). In the latter study, leukocyte rolling was induced by preparation injury but not by an inflammatory stimulus like PCW. Fucoidin is known to interact mainly with L-selectin. Moreover, leukocyte accumulation is attenuated in mice deficient in P-selectin in a model using tumor necrosis factor-α and interleukin-1β to induce meningitis (Tang et al., 1996).
Initial rolling caused by tissue trauma is mediated by P-selectin and later by L-selectin (Ley et al., 1995). There seems to be a key role for L-selectin in tumor necrosis factor-α–treated preparations because after 2 hours of tumor necrosis factor-α treatment, a monoclonal antibody directed against L-selectin blocked leukocyte rolling in P-selectin deficient mice (Ley et al., 1995). Tumor necrosis factor-α is involved in bacterial meningitis (Mustafa et al., 1989; Saukkonen et al., 1990) and may mediate leukocyte rolling through L-selectin.
We failed to detect P-selectin expression by flowcytometry on cultured cerebral endothelial cells after exposure to PCW (unpublished data). Lipopolysaccharide and tumor necrosis factor-α induce P-selectin expression on venular endothelial cells of the leptomeninges and, to a weaker level, on blood vessels of the brain parenchyma (Gotsch et al., 1994), but this is relatively late compared with the beginning of leukocyte rolling.
An attractive candidate to initiate P-selectin expression in bacterial meningitis is histamine. Obvious sources of histamine and other proinflammatory mediators are meningeal mast cells. Histamine is an effective trigger for rapid P-selectin expression on endothelial cells (Thorlacius et al., 1994) and for leukocyte rolling (Ley, 1994). We demonstrated that leukocyte rolling can be inhibited at least temporarily by a histamine 1 receptor blocker in the same model (Weber et al., 1997). Mast cell activation and histamine release can be attenuated by heparin (Lucio et al., 1992) and may contribute partially to the beneficial effect of heparin treatment.
Leukocyte Sticking in Bacterial Meningitis
Inhibition of leukocyte sticking with monoclonal antibodies directed against CD 18 (Saez Llorens et al., 1991; Tuomanen et al., 1989) in the rabbit model and directed against intercellular adhesion molecule 1 (Weber et al., 1995) in the rat model of bacterial meningitis attenuated inflammatory changes. CD 11/CD 18–intercellular adhesion molecule 1 interaction mediates firm adhesion of leukocytes to the endothelium and may be partially dependent on L-selectin function (Crockett Torabi et al., 1995) and on previous leukocyte rolling (Lindbom et al., 1992), which is consistent with our findings.
Leukocyte–Endothelium Interaction and Regional Cerebral Blood Flow
An interesting finding of this and our previous studies was that inhibition of leukocyte rolling and partial firm adhesion reduces rCBF increase (Angstwurm et al., 1995; Weber et al., 1995). A possible hypothesis is that the release of vasodilators by leukocytes or endothelial cells, such as nitric oxide or arachidonic acid metabolites, depends on rolling and subsequent firm adhesion of activated leukocytes in inflammation.
Effects of Heparin on Leukocyte–Endothelium Interaction
There is increasing evidence that heparin plays an important role in the control of inflammatory processes. P-selectin and E-selectin are expressed on endothelium, whereas L-selectin is found on leukocytes. Heparin binds to P- and L-selectin, but not to E-selectin (Nelson et al., 1993). In a mouse model of thioglycolate-induced acute peritonitis, low molecular heparin (Nelson et al., 1993) and a monoclonal antibody directed against P- and L-selectin attenuated leukocyte influx to the peritoneal cavity (Goldman, 1994).
Several additional lines of evidence exist, suggesting that heparin and HS inhibit rolling and subsequent adherence and trafficking of leukocytes into the tissue (Tangelder and Arfors, 1991; Ley et al., 1991). In contrast to reperfusion injury in the rabbit (Kubes et al., 1995), in our study a decrease of leukocyte rolling of 65% resulted in a significantly reduced number of leukocytes sticking in venules of the brain. As expected, leukocyte influx into the CSF was ameliorated in our experiments.
In a recent study, heparin reduced infarct size and neurologic dysfunction in a rat model of transient focal cerebral ischemia. This protective effect is related closely to its antileukocyte adherence property because leukocyte accumulation in the infarct area was decreased (Yanaka et al., 1997a; Yanaka et al., 1997b).
Heparin and Blood Coagulation
We did not measure coagulation parameters in our experiments, but no bleeding complications were observed. The anticoagulant effect of rolling inhibition by glycosamines must be addressed in further studies. Components of the coagulation cascade can amplify leukocyte adhesion and inflammation. Thus, it seems possible that the overall beneficial effects of heparin are exerted partially by its potent anticoagulant activity. Heparan sulfate, which has much lower anticoagulant properties, was less effective than heparin in previous studies (Tangelder and Arfors, 1991) and in our experiments.
Alternative Anti-Inflammatory Effects of Heparin
Besides the well known anticoagulant properties and the interference with leukocyte rolling of heparin, the functional versatility of this molecule has to be considered. Recent studies have indicated that heparin and related substances can modulate activities of a number of inflammatory cells, including neutrophils. Heparin inhibits the respiratory burst of neutrophils after chemical stimulation and neutrophil enzyme release (Cerletti et al., 1994; Bazzoni et al., 1993). Free radicals are involved in selectin expression (Patel et al., 1991). However, superoxide radicals produced by activated neutrophils can be scavenged by plasma super oxide dismutase activity, which is induced by intravenous heparin due to an increase of extracellular-superoxide dismutase fraction C (Karlsson and Marklund, 1988; Oyanagui and Sato, 1990). Super oxide dismutase and catalase are known inhibitors of RCBF increase in experimental meningitis (Pfister et al., 1992a; Pfister et al., 1992b). Because rats do not have EC-SOD C (Karlsson and Marklund, 1988), it seems questionable whether this mechanism has played a relevant role in our experiments. Furthermore, heparin is believed to bind to interleukin-8 (Rot, 1992), macrophage inflammatory protein (Tanaka et al., 1993), and rantes (Miller and Krangel, 1992). Brain edema, but not the ICP increase, was prevented by heparin treatment 4 hours after PCW intracisternal injection, which may result from an augmentation of the vascular barrier function (Bannori et al., 1995).
CONCLUSION
Heparin inhibits leukocyte rolling, and possibly as a consequence, reduces rCBF, ICP increases, brain edema, and leukocyte influx to the CSF in acute experimental bacterial meningitis. In addition to its traditional use, heparin or related nonanticoagulant glycosamines are a new and rational approach to ameliorate the inflammatory injury in central nervous system inflammation.
