Abstract
Alterations of blood flow contribute to major clinical complications in invasive infections such as sepsis and bacterial meningitis. As a unique feature streptococci – in particular, Streptococcus pneumoniae, the most frequent pathogen in bacterial meningitis – release hydrogen peroxide (H2O2) because of the absence of functional catalase. In a 6 h rat model of experimental meningitis, we studied the impact of bacterial H2O2 production on regional cerebral blood flow (rCBF) and intracranial pressure (ICP). Compared to wild-type D39 pneumococci, the increase of rCBF was diminished in meningitis induced by the H2O2 defective SpxB− mutant (maximum increase, 135% ± 17% versus 217% ± 23% of the individual baseline; P < 0.01) or after treatment of D39-induced meningitis with H2O2-degrading catalase or with tetraethylammonium (TEA), a blocker of calcium-sensitive potassium channels, which mediate H2
Introduction
Bacterial meningitis is still a life-threatening disease even with modern antibiotic treatment. Mortality is as high as 30%, and long-term neurological deficits are frequently found in survivors (de Gans et al, 2002). The negative outcome is largely determined by early complications, such as formation of brain edema, vascular alterations, raised intracranial pressure, and increased outflow resistance of the cerebrospinal fluid (CSF) (Scheld et al, 1980; Tuomanen et al, 1989). Although bacterial and host-sided factors are thought to contribute to blood—brain barrier breakdown, cerebral hyperemia is usually interpreted as an effect of vasoactive substances released by the host during the inflammatory response (Pfister et al, 1995; Lorenzl et al, 1996; Paul et al, 1997; Angstwurm et al, 1998; Hoffmann et al, 2002). In other invasive bacterial infections such as sepsis, vasodilatation contributes to systemic hypotension and organ dysfunction (Hotchkiss and Karl, 2003).
Streptococcus pneumoniae is the most frequent and disastrous pathogen in bacterial meningitis and an important cause of pneumonia and sepsis worldwide. A distinctive feature of streptococci, in particular S. pneumoniae, resides in the absence of catalase, a hydrogen peroxide (H2O2) degrading enzyme. As a result, pneumococci release significant amounts of H2O2 as a byproduct of oxidative metabolism. H2O2 limits the growth of other competing bacteria and is regarded as an additional virulence factor of pneumococci (Spellerberg et al, 1996; Pericone et al, 2002). Moreover, H2O2 acts as an exotoxin and may cause cytotoxic damage to host tissue (Braun et al, 2002; Bermpohl et al, 2005; Hoffmann et al, 2006).
In the eukaryotic organism, H2O2 is commonly appreciated as a cytotoxic product, which is synthesized by the nicotinamide adenosine dinucleotide phosphate oxidase of activated phagocytes. H2O2 is a potent vasodilator, which acts via the opening of calcium-activated potassium channels (KCa; Sobey et al, 1997). In the present study, we asked whether the release of H2O2 by pneumococci exerts direct vasodilator effects in early meningitis and may thus contribute to early disturbances of blood flow and intracranial pressure (ICP).
Materials and methods
Bacterial Strains and Culture
D39, an encapsulated strain of S. pneumoniae serotype 2 originally derived from a clinical isolate, was used as the wild type. Additionally, we used the previously published mutant SpxB− (Spellerberg et al, 1996). These bacteria lack pyruvate oxidase, resulting in a 95% reduction of H2O2 production (Spellerberg et al, 1996; Pericone et al, 2002). For liquid cultures, the strains were grown in standard casein plus yeast medium with 5% CO2 at 37°C. Erythromycin (Sigma, Tavfkirchen, Germany) was added to SpxB− at a final concentration of 1 μg/mL. During logphase growth, bacteria were pelleted by centrifugation and resuspended in pyrogen-free phosphate-buffered saline (PBS); 0.1 mol/L. Colony-forming units (CFU) per milliliter were then determined photometrically (absorption at 620 nm) using a standard curve. Adequate dilutions in pyrogen-free PBS were used to produce defined inocula. Colony-forming units calculations were verified by plating of serial dilutions. Pneumococcal cell wall (PCW) was prepared as published previously (Bermpohl et al, 2005).
Rat Experiments
Table 1 outlines the experimental group design. The general experimental procedure was as described earlier (Hoffmann et al, 2002). Experiments were performed on male Wistar rats (280 to 330 g) anesthetized with intraperitoneal sodium thiopental (100 mg/kg initially and 20 mg/kg every 2 h; Trapanal, Byk Gulden, Konstanz, Germany). Animals were tracheotomized and ventilated mechanically (AP-10; K Effenberger, Paffing, Germany) with air supplemented with O2. End-tidal CO2 was monitored continuously (Artema MM204; Heyer, Bad Ems, Germany). Body temperature was measured by a rectal probe and maintained at 37.8°C ± 0.4°Cusing a heating pad. A transducer (Statham P109 EZ, Spectramed, Oxnard, CA, USA) connected to a catheter placed in the left femoral artery measured mean arterial blood pressure (MAP) continuously. From this catheter, arterial blood samples were analyzed for PaO2, PaCO2, and pH at 0, 2, 4, and 6 h. The left femoral vein was cannulated for infusion of drugs or saline. A 3 × 3 mm2 area of the parietal bone lateral to the sagittal suture was thinned to allow laser Doppler flow (LDF) measurements (Periflux 4001 Master, Järfälla, Sweden) in cortical blood vessels. Although LDF is not a direct measure of the absolute regional cerebral blood flow (rCBF), a strong correlation between relative changes of LDF and relative changes of rCBF has been demonstrated previously. A catheter was placed into the cisterna magna through an occipital burr hole and connected to a pressure transducer (Statham P109 EZ, Spectramed, Oxnard, CA, USA) for continuous ICP measurement. Cerebral perfusion pressure (CPP) was calculated as MAP—ICP. A CPP of 60 mm Hg was considered the lower threshold of autoregulation. For challenge (0h time point), 100 μL of CSF were withdrawn from this catheter and replaced with bacterial suspension, PCW preparation, or sterile PBS. At the end of the experiment, CSF samples were obtained to determine the CSF leukocyte count and the bacterial concentration by plating of serial dilutions. Animals were then killed by injection of 3 mol/L KCl.
Experimental group design and basic physiology
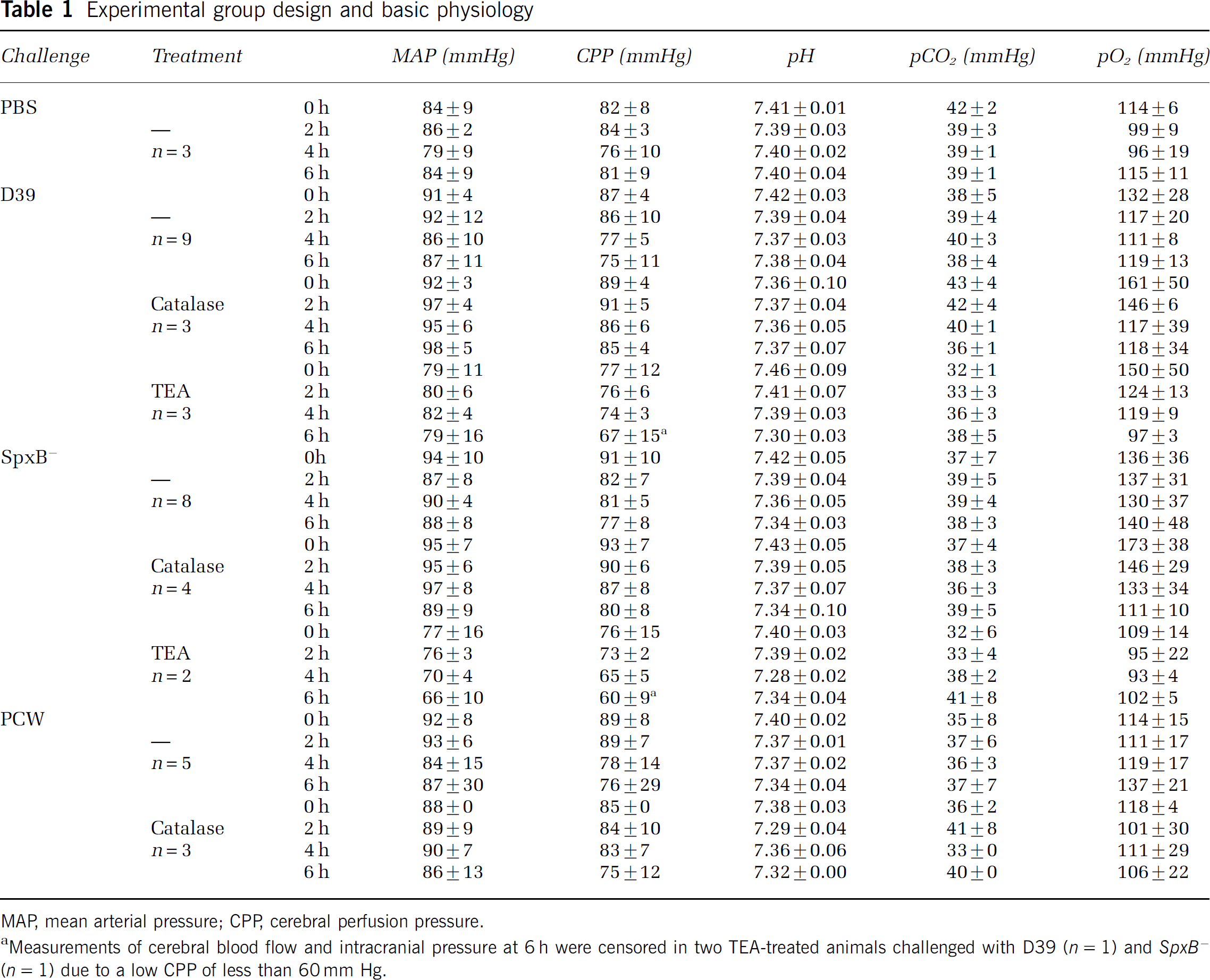
MAP, mean arterial pressure; CPP, cerebral perfusion pressure.
Measurements of cerebral blood flow and intracranial pressure at 6 h were censored in two TEA-treated animals challenged with D39 (n = 1) and SpxB−(n = 1) due to a low CPP of less than 60 mm Hg.
Pharmacological Interventions
We used two strategies to determine the impact of H2O2 on cerebral blood flow increase. In a subset of experiments, catalase (EC 1.11.16; Sigma) was applied both as a continuous infusion via the femoral vein catheter (50.000 U/kg per hour) starting 15 mins before meningitis induction and as a single intrathecal injection (15.000 iE) via the cisterna magna catheter at the time of meningitis induction. In further experiments, tetraethylammonium (TEA; Sigma), a blocker predominantly of calciumdependent potassium channels, was injected as a single bolus of 1 mg per animal via the femoral vein catheter 15 mins before meningitis induction.
Statistical Methods
Data are presented as means ± standard deviations (s.d.). Intraindividual changes of LDF and ICP were examined with paired t-tests after testing for normal distribution, using the absolute values. To compare effects between two groups of animals, LDF was normalized by defining the individual baseline as 100%, whereas ICP was transformed into the ICP increase relative to the individual baseline. In samples with normal distribution and equal variance, Student's t-tests were performed; otherwise, Mann—Whitney U-tests were used. Multiple comparisons were performed with Kruskal—Wallis analysis of variance followed by Dunn's post hoc testing.
Results
Basic Physiology and Cerebrospinal Fluid Findings
Basic physiological parameters are shown in Table 1. Although there were no relevant differences in MAP and blood gas analyses between the remaining experimental groups, TEA treatment was consistently associated with a significantly reduced MAP (Acheson and Moe, 1946). At the time of intrathecal challenge (0 h time point; 15 mins after application of TEA), MAP of all animals receiving TEA was 78 ± 11 mm Hg compared with 92 ± 7 mm Hg in animals not receiving TEA (P < 0.01, Mann—Whitney U-test). At the end of the experiment (6 h), TEA-treated animals also had a lower MAP of 75 ± 11 mm Hg compared with 89 ± 14 mm Hg in the remaining animals (P = 0.03, Mann—Whitney U-test). Two animals treated with TEA (one challenged with D39 and one challenged with SpxB−) displayed severe hypotension toward the end of the experiment, resulting in a CPP below the presumed lower level of at 6 h after challenge. To rule out a confounding effect of the low CPP, the corresponding measurements of LDF and ICP at 6 h in these two animals were censored from the statistical evaluation, while earlier measurements up to 5 h were maintained due to the presence of normal CPP.
In control animals, no CSF pleocytosis was observed at the end of the experiment (19 ± 11 leukocytes/μL). By comparison, all animals challenged intrathecally with live bacteria or PCW developed intrathecal inflammation as evidenced by the influx of leukocytes into the CSF. Cell counts at the end of the experiment 6 h after meningitis induction were 3435 ± 2001/μL with D39, 5771 ± 196/μL with SpxB− and 5125 ± 391/μL with PCW, the differences not being statistically significant (Figure 1A). Moreover, plating revealed the presence of biologically equivalent bacterial concentrations in the CSF between the two bacterial strains (D39, 5.4 ± 1.6 × 108CFU/mL versus SpxB− 3.0 ± 2.4 × 108CFU/mL; Figure 1B).
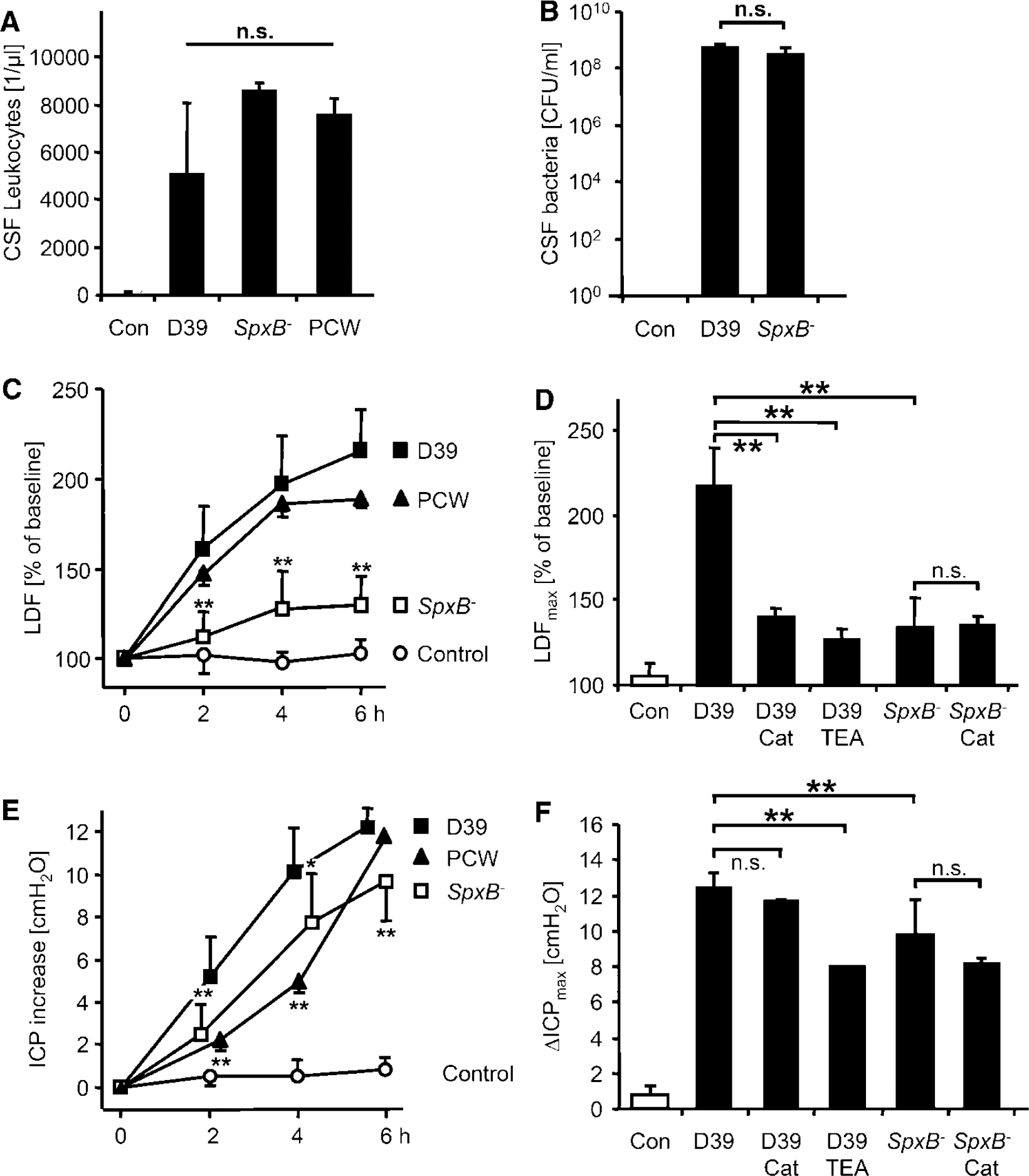
Impact of bacterial H2O2 release in early pneumococcal meningitis. (
Effects on Regional Cerebral Blood Flow
In control animals receiving intrathecal PBS, the rCBF as measured by laser Doppler did not change significantly during the experimental period (maximum increase, 105% ± 7% of baseline; P = 0.25 versus baseline; Figure 1C). After intrathecal infection with live D39 pneumococci, we observed a gradual increase of the rCBF to a maximum of 217% ± 23% of baseline (P < 0.01 versus individual baseline and P < 0.01 versus maximum increase in PBS-challenged controls). Infection with SpxB− caused only a minor increase in LDF (maximum, 135% ± 17% of baseline; P < 0.01 versus individual baseline and P = 0.03 versus maximum increase in PBS-challenged controls). The LDF increase with SpxB− was significantly lower than with D39 (P < 0.01 at 2, 4, and 6 h after infection). Challenge with PCW resulted in an increase of LDF to a maximum of 192% ± 5% of baseline (P < 0.01 versus maximum increase in PBS controls and P = 0.04 versus maximum increase in D39-infected animals).
Treatment of D39-induced meningitis with systemic catalase partially abrogated the blood flow increase (maximum increase, 142% ± 4.0% of baseline; P < 0.01 versus maximum increase in PBS controls and P < 0.01 versus maximum increase with D39 alone; Figure 1E) as did systemic application of the KCa blocker TEA (maximum increase, 128% ± 6% of baseline, P = 0.02 versus baseline and P < 0.01 versus D39 alone). Neither intervention led to a significant change of the systemic blood pressure or of the cerebral blood flow during a 15 mins observation period before meningitis induction. Treatment of SpxB−-induced meningitis with catalase did not lead to a further reduction of the LDF increase (maximum LDF increase, 135% ± 14% of baseline; P = 0.96 versus maximum increase with SpxB−alone). Also, no significant effect of TEA was observed in SpxB−-induced meningitis (maximum LDF increase, 130% ± 2% of baseline, P = 0.69 versus maximum increase with SpxB− alone). In meningitis induced by PCW, treatment with catalase was not effective (maximum LDF increase, 181% ± 22% of baseline; P = 0.27 versus PCW alone).
Effects on Intracranial Pressure
In control rats challenged with intrathecal PBS, no relevant change of the ICP was observed during the experimental period (maximum ICP increase, 0.8 ± 0.5cm H2O; P = 0.14 versus baseline; Figure 1E). Intracisternal infection with D39 led to a gradual increase of the ICP (maximum ICP increase, 12.4 ± 0.9 cm H2O; P < 0.01 versus baseline and P < 0.01 versus PBS-challenged controls). With SpxB−, we observed a slightly lower increase of the ICP (maximum increase, 9.8 ± 2.0cm H2O; P < 0.01 versus baseline and P < 0.01 versus maximum increase with D39). Starting at 60 mins, the ICP was significantly higher than in controls (P < 0.01) but remained lower than in the D39-treated animals (P < 0.01 at 2 and 6 h and P = 0.02 at 4 h). After challenge with PCW, the maximum ICP increase was 11.9 ± 0.1 cm H2O (P = 0.49 versus maximum increase with D39). However, the increase of ICP was slower with PCW than with D39 (P < 0.01 at 2 h and at 4 h).
Treatment of D39-infected rats with catalase did not lead to a significant reduction of the maximum ICP increase (11.7 ± 0.1 cm H2O, P = 0.20 versus D39 alone; Figure 1F). Conversely, TEA treatment of D39-induced meningitis resulted in a modest reduction (maximum ICP increase, 7.8 ± 0 cm H2O; P < 0.01 versus D39). No relevant effect of catalase was observed in rats challenged with SpxB− (maximum ICP increase, 8.2 ± 0.3 cm H2O, P = 0.13 versus SpxB− alone) or PCW (maximum ICP increase, 10.0 ± 1.2 cm H2O, P = 0.10 versus PCW alone).
Discussion
The major finding of this study is that in pneumococcal meningitis, a substantial proportion of early cerebral hyperemia is directly caused by the infectious pathogen through the release of bacterial H2O2. Compared to wild-type pneumococci, meningitis-associated increase of rCBF was markedly reduced with H2O2-deficient pneumococci, by scavenging of H2O2 using catalase, or on pharmacological blockade of KCa.
Compared to the significant vasodilator effects of D39, we found a residual blood flow increase in SpxB−-induced meningitis of approximately 30%, which seems to be independent of the bacterial H2O2 production. In these experiments, biologically equivalent bacterial and leukocyte concentrations in the CSF argue against decreased virulence of SpxB−(Spellerberg et al, 1996). A role of host-derived H2O2 can be ruled out given the inability of catalase to reduce the blood flow increase in SpxB− or PCW-induced meningitis. As a likely H2O2-independent mechanism, host-derived nitric oxide—a well-known, potent vasodilator released during the inflamatory response—has been shown to mediate part of the pial vasodilation in meningitis (Paul et al, 1997). Although catalase has previously been reported to reduce the blood flow increase in pneumococcal meningitis (Pfister et al, 1992), our findings provide significant new insight by identifying bacteria as the relevant source of vasoactive H2O2.
Compared to SpxB−, a more substantial blood flow increase was induced by PCW. Because both of these challenges are unable to release H2O2, this points to a different stimulatory effect on the host immune response and host-mediated vasodilation. Detection of invading pathogens by the innate immune system is affected by binding of pathogen-associated molecular patterns (PAMP) to pattern recognition receptors, most prominently of the toll-like receptor (TLR) family (Akira et al, 2006). In the case of pneumococci, recognition of teichoicated peptidoglycans and bacterial lipopeptides from the bacterial cell wall by the TLR2 receptor constitute the most potent signal (Weber et al, 2003). In patients, the clinical severity of pneumococcal meningitis correlates with the CSF concentration of major bacterial cell wall components (Schneider et al, 1999). It may therefore be argued that the stimulatory effect of mechanically disintegrated cell walls (PCW) on the immune system is stronger and more immediate than that of live bacteria, which do not expose pathogen-associated molecular patterns of the inner cell wall before lysis occurs. Along the same lines, our findings clearly establish a different pathophysiological basis for the equally pronounced blood flow effects of PCW and D39. Since catalase and TEA abrogated the blood flow increase caused by D39, bacterial H2O2 rather than the inflammatory host response (as with PCW) is the major vasodilator during early D39-induced meningitis.
Intracranial hypertension during meningitis has been explained as the result of several pathophysiological changes, that is, hyperemia, formation of brain edema after breakdown of the blood—brain barrier, and—later in the course of the disease—increased outflow resistance of the CSF (Scheld et al, 1980; Tuomanen et al, 1989). In our experiments, the increase of ICP observed with D39 was not significantly reduced by treatment with catalase, and only a very modest reduction was achieved by TEA treatment or with H2O2-deficient bacteria, despite the fact that all of these strategies were effective in reducing hyperemia. It appears that H2O2-induced hyperemia plays only a minor role in the evolution of intracranial hypertension during the early phase of pneumococcal meningitis. Together with the strong increase of ICP after intracisternal challenge with PCW, these results support a central role for the inflammatory host response in the generation of intracranial hypertension (Tuomanen et al, 1989; Weber et al, 1995). Other bacterial factors to consider include the pneumococcal toxin pneumolysin (Braun et al, 2002).
In conclusion, our findings identify bacterial-derived H2O2 as a major vasodilator during early pneumococcal meningitis, which acts in concert with the vasodilator effects of host-derived inflammatory mediators. Bacterially induced vasodilation represents a new concept for blood flow regulation in meningitis and may be of importance in other invasive infections such as sepsis.
