Abstract
We investigated whether trigeminal nerve fibers contribute to enhanced regional cerebral blood flow (rCBF) in a rat model of experimental bacterial meningitis. rCBF was measured continuously for 6 h by laser Doppler flowmetry through thinned bone over the frontal cortex. Meningitis was induced with pneumococcal cell wall components and confirmed by a significant increase of (a) leukocytes within the cerebrospinal fluid, (b) brain water content, (c) intracranial pressure and (d) rCBF. The increase of rCBF was significantly attenuated (p < 0.05) at 3, 4, 5, and 6 h in animals after a chronic (200 ± 21% versus 138 ± 13% at 6 h on the intact and denervated sides, respectively) but not after an acute section of the nasociliary branch of the trigeminal nerve. We conclude that elevations in blood flow during the early phase of bacterial meningitis are mediated in part by the trigeminal nerve, probably by local perivascular release of neuropeptides from afferent axons innervating the meninges.
Meningitis caused by Streptococcus pneumoniae is associated with a morbidity of 50% (Bohr et al., 1983) and a surprisingly high mortality rate of 25% (Pfister et al., 1993), despite potent antibiotics that effectively treat the bacterial infection (Swartz, 1984). Cerebrovascular changes (Pfister et al., 1992a), brain edema, as well as high intracranial pressure (for review see Quagliarello and Scheid, 1992) portend a poor clinical outcome. The early phase of experimental pneumococcal meningitis is characterized by an increase in cerebral blood flow (Pfister et al., 1990; Weber et al., 1995), which is essential for the development of inflammation (Mantovani et al., 1994) and may contribute to leukocyte adhesion and raised intracranial pressure (ICP). The cause of this hemodynamic change during the early phase of bacterial meningitis is not fully understood, although there is growing evidence that nitric oxide (NO) is a central mediator (Haberl et al., 1994; Koedel et al., 1995). Invading leukocytes are thought to be a major source of NO (Moncada et al., 1991) and superoxide radicals. This idea is supported by our findings that substances interfering with leukocyte rolling (Angstwurm et al., 1995) or firm adhesion (Weber et al., 1995) inhibit the regional cerebral blood flow (rCBF) increase in experimental bacterial meningitis. Moreover, astrocytes (Freyer et al., 1996) as well as cerebral endothelial cells (Koedel et al., 1995) produce NO in vitro when stimulated with pneumococcal cell wall (PCW) or heat-inactivated Streptococcus pneumoniae. In the cerebral circulation, substance P (SP) and calcitonin gene-related peptide (CGRP) are potent NO-dependent vasodilators (Wei et al., 1992) that have been linked to hyperemia during CNS inflammation (Brian Jr., et al., 1995; Pfister et al., 1995).
A further link between meningitis and the trigeminovascular system is suggested by the occurrence of headache as a key symptom in the early stage of bacterial meningitis. This clinical symptom may be mediated through activation of unmyelinated C-fibers originating from the trigeminal nerve and innervating cerebral blood vessels (Suzuki et al., 1989a) and the meninges (Buzzi et al., 1991). Axonal tracing techniques have established that supratentorial meningeal and cortical blood vessels are innervated by unmyelinated C-fibers projecting primarily from cell bodies within ipsilateral sensory (trigeminal) (Mayberg et al., 1984) and autonomic (sympathetic or parasympathetic) ganglion (Suzuki et al., 1989a,b). Trigeminal nerve fibers convey nociceptive information from the meninges and promote neurogenic inflammation via the release of CGRP and SP from perivascular axons. Trigeminal nerve fibers also increase cortical blood flow by axon reflex mechanisms, which has been demonstrated during seizures, severe acute hypertension (Sakas et al., 1989), and ischemia reperfusion (Moskowitz et al., 1989b; Macfarlane et al., 1991). SP as well as CGRP are stored in afferent C-fibers (Suzuki et al., 1989a). Both neuropeptides augment the actions of proinflammatory mediators in such cell types as astroglia (Mantyh, 1991; Luber Narod et al., 1994; Regoli et al., 1994) and augment leukocyte endothelial interactions (Zimmerman et al., 1992; DeRose et al., 1994). Based on these findings, we proposed that real or threatened tissue injury provides the most important stimulus for activation of meningeal sensory fibers, which is in agreement with data from somatic and visceral afferents innervating other organs (Moskowitz et al., 1989a; Moskowitz 1991).
To determine the role of the trigeminovascular system in rCBF regulation during meningitis, we measured rCBF during the development of experimental meningitis following chronic surgical denervation of meningeal afferent fibers. Our data show that rCBF increases during bacterial meningitis are mediated, in part, by the trigeminovascular system.
METHODS
Nasociliary nerve (NCN) surgery and experimental groups
Thirty-five male Wistar rats (280–330 g) were anesthetized with chloral hydrate, 200 mg/kg body weight i.p. NCN sections were performed as modified from Suzuki (Suzuki et al., 1988). A midline incision was made between the orbits, and the skin and periosteum were reflected past the superior orbital margin. This method revealed the structures piercing the ethmoidal foramen (NCN, anterior ethmoidal artery and vein, and the fine filaments from the sphenopalatine ganglion). Five groups were defined based on the surgical procedure: Group 1 (n = 7) had the NCN sectioned only on one side. Group 2 (n = 5) had all structures piercing the ethmoidal foramen sectioned on one side. Group 3 (n = 5, sham) received the surgical exposure without sectioning. The animals were kept for 12–16 days before experimentation. Meningitis was induced by i.e. injection of 75 μl PCW in these three groups.
Group 4 (n = 5) had the NCN sectioned as in group 1, but the rats were injected i.e. with 75 μl saline (vehicle of PCW) 2 weeks later. Group 5 (n = 5) had the NCN sectioned only on one side, as in group 1, but meningitis was induced by i.e. injection of 75 μl PCW immediately after nerve transection. Additionally, we did three pilot experiments measuring rCBF above the parietal cortex, as we did in our previous studies (Angstwurm et al., 1995; Weber et al., 1995) (data are not shown). In three further experiments we investigated the effect of acute NCN surgery on the baseline rCBF (30 min before and after the surgery, data are not shown). There was a 6.25% mortality (n = 2) from these procedures.
General experimental procedures
Animals were anesthetized with 100 mg/kg sodium thiopental (Trapanal; Byk Gulden, Konstanz, Germany) intraperitoneally and mechanically ventilated with room air and supplemental oxygen throughout the experiment by a small animal ventilator (AP-10; K. Effenberger, Pfaffing, Germany). Additional doses of thiopental (20 mg/kg) were given whenever an MABP rise of ≧20 mm Hg was observed to a painful stimulus applied to the rat's tail. The left femoral artery and vein were cannulated for physiological monitoring and intravenous saline infusion (1 ml/h). Body temperature was controlled and maintained at 38.0° ± 0.5°C using a heating pad. Mean arterial blood pressure was measured continuously using a pressure transducer (Statham P109 EZ; Spectramed, Oxnard, CA, U.S.A.) connected to the femoral artery cannula. End-expiratory Paco2 (Heyer Artema MM 204; Bad Ems, Germany) was monitored continuously. Arterial blood samples were analyzed for Pao2, Paco2, and pH (Compact 1; AVL List, Graz, Austria) at the beginning and the end of each experiment as well as every 2 h.
CSF samples were collected for white blood cell (WBC) counts before and 6 h after the i.e. injection and counted in a Fuchs Rosenthal chamber (Poly-Optik, Bad Blankenburg, Germany). Intracranial pressure was measured throughout the experiment with a pressure transducer (Statham P109 EZ; Spectramed) connected to a cisterna magna catheter placed through a burr hole in the occipital bone (Pfister et al., 1990; Weber et al., 1995). For placement of laser Doppler probes, bilateral 2 × 3-mm areas of bone were thinned (Lindauer et al., 1993; Angstwurm et al., 1995) over the frontal lobe 3 mm lateral to the superior sagittal suture MABP, ICP, rCBF (discussed herein), and end-expiratory Paco2 were digitized and stored on a computer. At the end of the experiment the animals were killed by exsanguination, and brain water content was determined as described previously (Pfister et al., 1990).
Regional cerebral blood flow measurements (rCBF). rCBF was measured continuously on both sides by a two-channel laser-Doppler flowmeter (LDF) (Periflux 4001 Master; Järfälla, Sweden). The LDF probes were held by micromanipulators and positioned over the thinned and translucent bone in an area free of large pial vessels and larger dural vessels to minimize a dural contribution to the signal. rCBF was expressed as a percentage of stable baseline (defined as 100%) recorded for 30 min after the placement of the intracisternal catheter. rCBF was measured continuously during the experiment.
Meningitis induction
PCW was instilled into the cisternal space to produce inflammatory changes similar to bacterial meningitis (Pfister et al., 1990b; 1992b). PCW was produced as described by Tuomanen (Tuomanen et al., 1985) and modified as described earlier (Weber et al., 1995). An unencapsulated strain (PnR-527; Jena, Germany) of Streptococcus pneumoniae was cultivated overnight on Columbia agar plates, suspended in pyrogen-free saline, and heat inactivated. The inactivated bacteria were disintegrated by ultrasound, purified by centrifugation and washing, and again suspended in pyrogene-free saline. The optical density of the final preparation was 0.66 at 620 nm, which correlates to 107 CFU/ml. Gram staining demonstrated <1% intact cells. The pH of the suspension was 7.4. After establishment of a stable rCBF baseline for 30 min, a 75-μl CSF sample was taken from the cisterna magna. Meningitis was induced in Groups 1–3 and 5 by intracisternal injection of 75 μl PCW. As a control, Group 4 received an i.e. injection of 75 μl saline, the vehicle of PCW.
Statistical methods
Data are expressed as means ± SD. A p value of <0.05 was considered significant. To compare rCBF on sides with normal NCN, ICP, and WBC count in the CSF and normal brain water content, we used a one-way analysis of variance and the Duncan multiple-comparison test corrected for repeated measurement using the Bonferroni-Holm procedure. Sides within the groups were compared with paired Student's t tests corrected for repeated measurement using the Bonferroni-Holm procedure. Statistical evaluations was done using SPSS for Windows 6.1 statistical software.
RESULTS
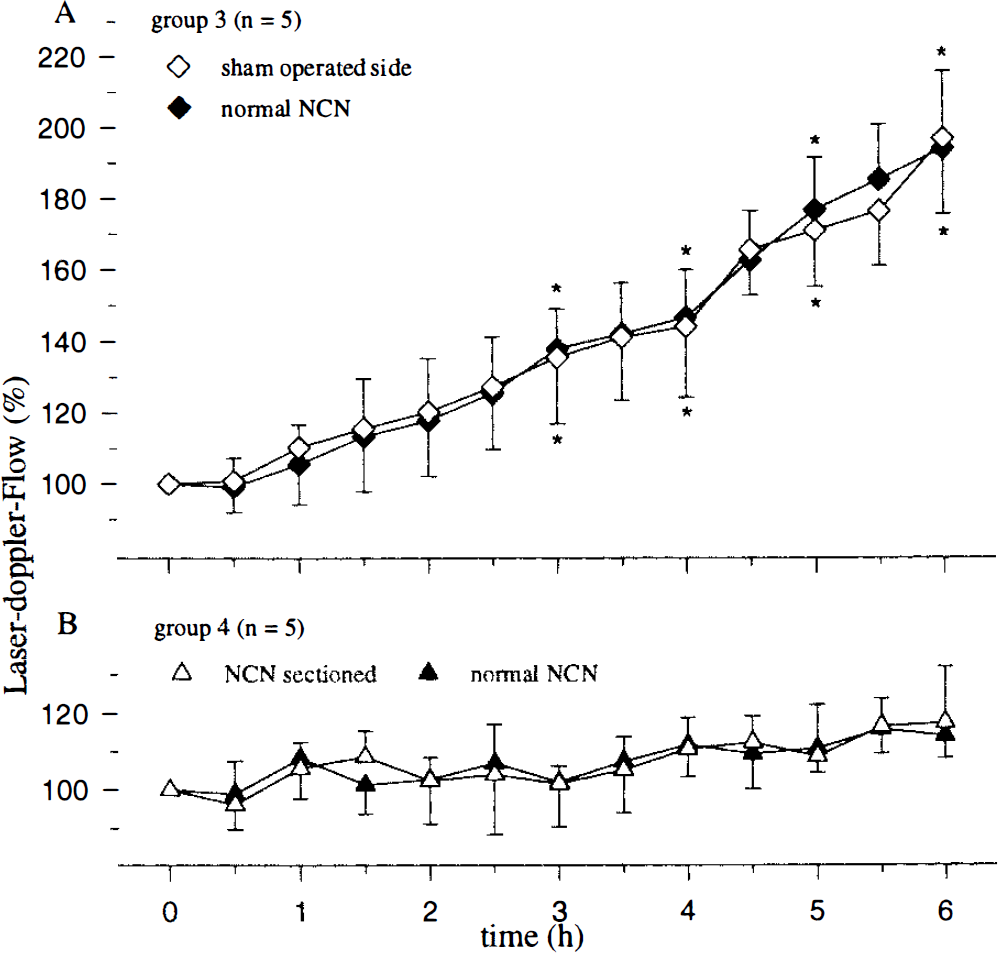
Values for MABP, Pao2, Paco2, and pH were within their normal ranges throughout the experiment (Table 1). Small differences between groups were not statistically significant. As is seen in meningitis, a significant increase in brain water content was measured in group 1 (79.1 ± 0.34%), group 2 (79.38 ± 0.38%), group 3 (79.11 ± 0.28%), and group 5 (79.23 ± 0.32%), but not in group 4 (78.55 ± 0.27%). The CSF contained no WBCs initially, but WBCs increased after PCW instillation (group 1: 2,351 ± 679; group 2: 3,420 ± 1,188; group 3: 3,492 ± 1,984; group 5: 2,996 ± 713 cells/μl) compared with group 4 (4.6 ± 4.2 cells/μl). ICP increased after PCW instillation (Table 2). As expected, sectioning of NCN did not change these parameters. There were no significant rCBF changes observed after saline instillation (Fig. 1B), and NCN sectioning did not alter rCBF in this control group (Fig. 1B, Table 2).
Physiological variables for experimental groups according to hours after i.e. injection
Data are means ± SD. There were no statistically significant differences between experimental groups.
rCBF increased significantly on the side with normal NCN at 3, 4, 5, and 6 h in all PCW-injected groups, and no group differences were observed (Figs. 1A and 2, Table 2). Sham operation itself did not change rCBF, and PCW instillation in this group caused a bilateral and symmetrical rCBF increase at 3, 4, 5, and 6 h (Fig. 1A, Table 2). An increase in rCBF was detected ipsilaterally after chronic NCN sectioning (groups 1 and 2), but this increase was significantly attenuated compared with the contralateral side (Figs. 1A and 2A, Table 2). The rCBF response after acute sectioning plus PCW (group 5) was robust and symmetrical (Fig. 2A, Table 2).
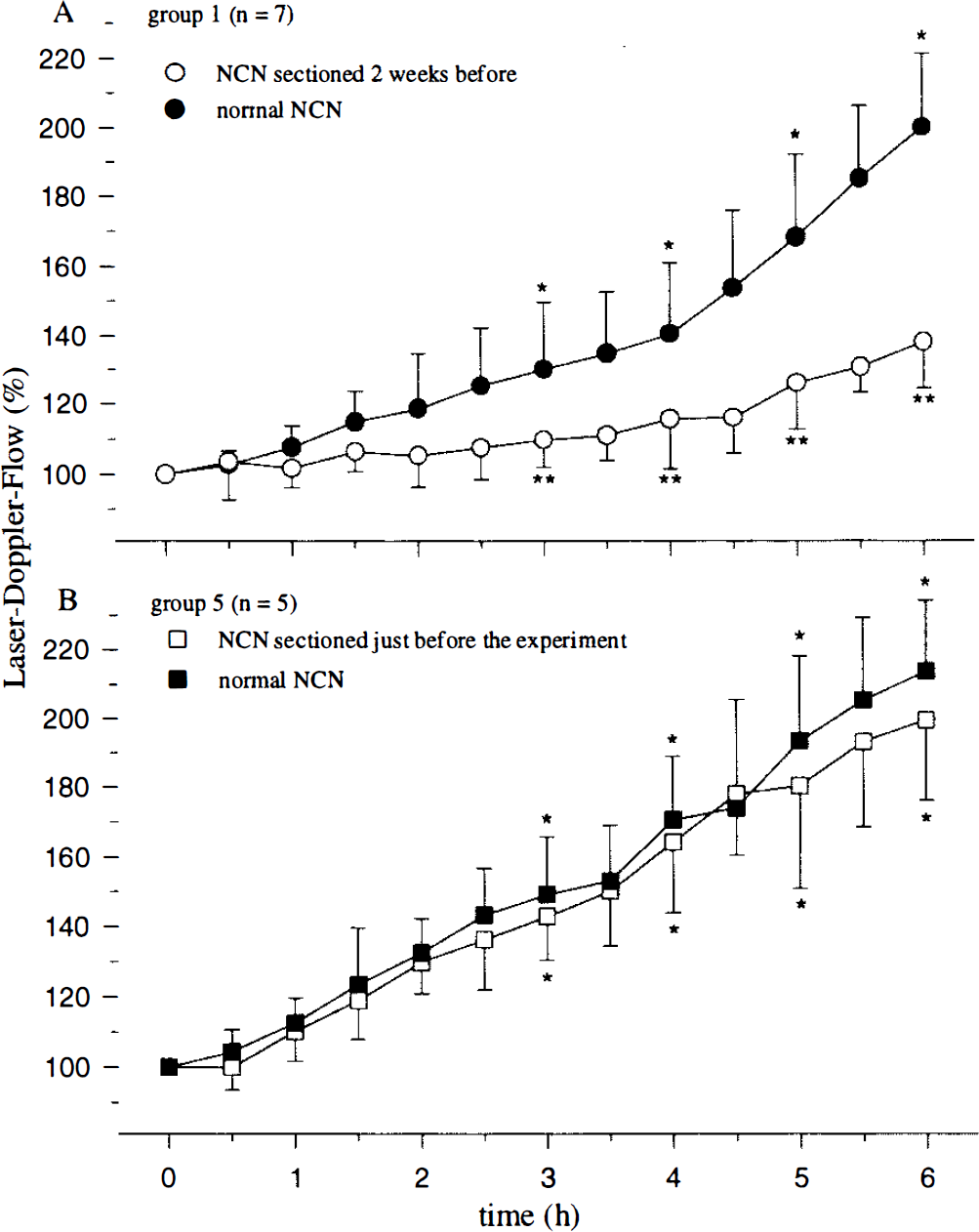
The effect of chronic (
ICP and rCBF values (mean ± SD) for experimental groups according to hours after i.e. injection

Significant difference compared with group 4.
Significant difference between sides in group 1 and group 2, but no statistically significant difference between NCN cut (group 1) and the side with all structures cut (group 2).
Significant different (p < 0.05) on the side with normal NCN between group 4 and all other groups.
DISCUSSION
Our data establish the importance of primary sensory innervation of the meninges to increases in rCBF during the early phase of experimental bacterial meningitis. Sectioning a branch of the trigeminal nerve 2 weeks before PCW instillation significantly reduced the expected rise in rCBF on the denervated side. The rCBF increase was most likely due to locally released vasodilators and/or inflammatory mediators from the trigeminal nerve into the perivascular space. A brain stem or a cortical mechanism mediated by central trigeminal neurotransmission is unlikely to cause the observed changes in blood flow because there were no differences between the two sides during meningitis immediately after acute nerve transection.
The data support the hypothesis that the decrease in vascular resistance and increase in blood flow develops in part as a consequence of activation of intracranial nociceptors. The presence of severe headache provides clear evidence that the trigeminal nerve is activated during meningitis. Previous reports have shown that sectioning the trigeminal nerve does not change baseline levels of flow, blood pressure regulation or response to carbon dioxide inhalation, but it does attenuate blood flow responses to noxious stimulation (Sakas et al., 1989; Macfarlane et al., 1991) in the cat. We confirmed that acute sectioning (Umemura and Branston, 1995) or functional blockade of the NCN by cooling (Branston et al., 1995) did not alter baseline levels of rCBF. Intracisternal PCW induces an inflammatory response characterized by CSF leukocytosis, increase in rCBF, brain edema, and ICP. Proinflammatory cytokines, such as tumor necrosis factor alpha (TNFα), interleukin-1β (IL-1β), interleukin 6, are released in the CSF (Saukkonen et al., 1990). Products of invading leukocytes, released inflammatory mediators, and possibly the bacterial cell wall components are acknowledged stimuli for nociceptor discharge (Cunha et al., 1992; Ferreira, 1993). The nociceptors of the trigeminovascular system respond to noxious mechanical, thermal, and chemical stimuli much like nociceptors innervating other tissues (Hughes and Barr 1988; Rang et al., 1991; Steen et al., 1995) and increase their ongoing discharge in response to intracranial inflammation (Bove and Moskowitz, unpublished data). The latency and time course of the rCBF changes roughly parallel the changes in discharge and sensitivity of knee ociceptors following induction of experimental arthritis (Schaible and Schmidt, 1988).
Neurogenic mechanisms appear to explain the observed rCBF asymmetries. Chronic sectioning of the NCN substantially decreases meningeal SP and CGRP after 2 weeks (Suzuki et al., 1989a). In an intact nerve, neuropeptides can be released from nociceptors during activation (Holzer, 1988), which has been observed in the meninges as well (Buzzi et al., 1991). The importance of SP and CGRP to blood flow and vascular permeability is well established (Holzer, 1988; Mantyh, 1991; Zochodne and Ho, 1991). Lipopolysaccharide-mediated vasodilation can be attenuated by CGRP (8–37) when applied with aminoguanidine, an inhibitor of inducible NO synthase (Brian Jr., et al., 1995). SP enhances TNFα secretion from neuroglial cells stimulated with lipopolysaccharides (Luber Narod et al., 1994), and TNFα induces adhesion molecule expression on brain endothelial cells (Wong and Dorovini Zis, 1992; Hess et al., 1994). In addition TNFα and SP induce other proinflammatory cytokines (Rameshwar et al., 1994), and by so doing, SP may increase neutrophil adhesion (Zimmerman et al., 1992; DeRose et al., 1994). CGRP enhances the production of inducible NO synthase in vascular smooth-muscle cells in the presence of II-1β (Schini Kerth et al., 1994). Our results suggest that depletion of SP and CGRP from perivascular afferent nerve terminals limits dilation and increased rCBF during the development of bacterial meningitis.
The possibility that large dural vessels contribute to the rCBF increase has to be considered (Faraci et al., 1989), but it is unlikely. The LDF probe was placed over an area devoid of large dural vessels, and the time course for the rCBF increase was suggestive of dilation of pial vessels using a closed cranial window with the dura removed (Koedel et al., 1995). When rCBF was recorded over the parietal instead of the frontal cortex, only a slight difference between sides was measured in three pilot experiments (data not shown) because other sensory branches innervate the meninges covering the parietal cortex (Suzuki et al., 1989a). In fact, Suzuki showed that chronic NCN sectioning decreases SP and CRGP immunoreactivity very slightly in parietal meninges (Suzuki et al., 1989a). Since parasympathetic fibers innervate intracranial blood vessels and contain vasoactive intestinal peptide, NO synthase, and acetylcholine, the possibility that they mediate rCBF changes was considered. However, sectioning parasympathetic fibers traversing the ethmoidal foramen, among other structures (group 2), did not further reduce the rCBF change on the ipsilateral side. Hence, rCBF increases during acute bacterial meningitis are at least in part caused by the trigeminovascular system. Our study provides strong experimental evidence to document that sensory activation augments cortical blood flow in part by local axonal mechanisms during the development of inflammation associated with bacterial meningitis.
Footnotes
Abbreviations used
Acknowledgment:
This work was supported by grants from the Deutsche Forschungsgemeinschaft SFB 507 project A3, Di 454/8, and by NS 21558 from the National Institute of Neurological Diseases and Stroke (M.A.M.) and by an unrestricted grant in the neurosciences from Bristol-Myers Squibb (M.A.M.).
