Abstract
The effect of the novel inhibitor of receptor-activated and calcium store-operated nonselective cation channels, (RS)-(3,4-dihydro-6,7-dimethoxyisoquinoline-1-γ1)-2-phenyl-N,N-di-[2(2,3,4-trimethoxyphenyl) ethyl]acetamide (LOE 908 MS), on focal cerebral ischemia was studied in halothane-anesthetized rats submitted to permanent suture occlusion of the right middle cerebral artery (MCA). The treated group (n = 7) received subcutaneous injections of 30 mg/kg LOE 908 MS (in 1 ml saline) 10 min after vascular occlusion and again after 3 h. The untreated group (n = 11) was injected subcutaneously with 1 ml saline at the same times. Evolution of infarct was monitored by electrophysiological recording of EEG and cortical steady potential and by diffusion-weighted magnetic resonance imaging during the initial 6 h of vascular occlusion. The hemodynamic, biochemical, and morphological changes were studied after 6 h by combining autoradiographic measurement of blood flow with histological stainings and pictorial measurements of ATP, glucose, and tissue pH. In the untreated animals, the ischemic lesion volume [defined as the region in which the apparent diffusion coefficient (ADC) of water declined to below 80% of control] steadily increased by ∼50% during the initial 6 h of vascular occlusion relative to the first set of data 10 min postocclusion. In the treated animals, in contrast, the ADC lesion volume declined by ∼20% during the same interval. Treatment also led to a significant reduction in the number of periinfarct depolarizations. After 6 h of vascular occlusion, blood flow was significantly higher in the treated animals, and the volume of ATP-depleted and morphologically injured tissue representing the infarct core was 60–70% smaller. The volume of severely acidic tissue, in contrast, did not differ, indicating that LOE 908 MS does not reduce the size of ischemic penumbra. These findings demonstrate that postocclusion treatment of permanent focal ischemia with LOE 908 MS delays the expansion of the infarct core into the penumbra for a duration of at least 6 h and therefore substantially prolongs the window of opportunity for the reversal of the ischemic impact in the peripheral parts of the evolving infarct.
Keywords
Ischemic injury of the brain is the cumulative result of several pathologies: The primary metabolic impact provoked by the reduction of blood flow is aggravated by secondary complicating disturbances, involving such different mechanisms as free radical-mediated membrane injury (Siesjö et al., 1989; Schmidley, 1990), uncoupling of oxidative respiration (Katsura et al., 1994; Abe et al., 1995), inflammatory reactions (Shiga et al., 1991), perifocal depolarizations (Iijima et al., 1992; Nedergaard and Hansen, 1993), and the induction of suicidal genomic responses (Charriaut-Marlangue et al., 1996; Du et al., 1996). Although each of these pathologies involves a cascade of complex and highly specific molecular mechanisms, it is widely accepted that an early abnormality in the sequel of events must be the activation or inactivation of receptors and ion channels at the plasma membrane. Therapeutic approaches for ameliorating ischemic injury have therefore focused mainly on modulation of receptor sensitivities and ion conductances.
Among the mechanisms involved, increased calcium fluxes across voltage or receptor-operated calcium channels have received particular attention. A peculiar mode of Ca2+ influx is mediated by capacitative gating of un-selective cation channels (Berridge, 1987; Putney, 1990; Fasolato et al., 1994; Friel, 1996; Zhu et al., 1996). These channels are referred to as store-operated cation channels (SOCs) (Wes et al., 1995) because Ca2+ influx is activated by still unknown signals generated from the depletion of inositol 1,4,5-triphosphate (IP3)–sensitive calcium stores (Zitt et al., 1996). SOCs form a large heterogeneous family whose structures (Wes et al., 1995; Zhu et al., 1996; Zitt et al., 1996) and regulations (Zitt et al., 1996) are only partly known. The Ca2+ influx through SOCs leads to an amplification of IP3-activated increase of intracellular [Ca2+] and therefore may modulate this response in a cell-specific way. Members of the SOC family play a prominent role in the activation of neutrophilic leukocytes during inflammatory events (Von Tscharner et al., 1986), and they may be involved in cell degradation and necrosis (Choi, 1988).
According to Simpson et al. (1995), neuronal Ca2+ stores play a crucial role in a variety of physiological and pathophysiological processes. However, the relative importance of SOC in this process is still poorly understood because of lack of specific pharmacological channel blockers.
Another class of nonselective cation channels is operated by ligands that bind to receptors located at the N-terminal domain of the cation-permeable pore (Benham and Tsien, 1987; Inoue and Nakazawa, 1992). These receptor-activated cation channels allow very fast increases in cytosolic Na+ and Ca2+ concentration and thus may contribute to the known disturbances of ion and water homeostasis associated with cerebral ischemia.
The isoquinoline derivative (RS)-(3,4-dihydro-6,7-dimethoxyisoquinoline-1-γ1)-2-phenyl-N,N-di-[2(2,3,4-trimethyoxyphenyl)ethyl]acetamide (LOE 908 MS) is a potent inhibitor of receptor or store-operated channels especially in primary cultures of cortical and hippocampal neurons (D. Arndts, Boehringer Ingelheim KG, personal communication). The agent does not influence the IP3-regulated Ca2+ release from internal pools but blocks the reactive Ca2+ entry through SOCs (IC50: 0.6 μM). In HL-60 cells full blockade of ATP- or N-formyl-L-methionyl-L-leucyl-L-phenylalanine-stimulated cation currents is achieved at 3 μM and half-maximal inhibition at concentrations of as low as 40 nM (Krautwurst et al., 1993).
This remarkable pharmacological potency led us to investigate the effects of LOE 908 MS on ischemic injury induced by permanent middle cerebral artery (MCA) occlusion of rat. By combining advanced magnetic resonance imaging methods with autoradiographic, bioluminescence, and fluorescence imaging techniques, information could be obtained not only on the effect of the drug on infarct volume and on the temporal evolution of the pathological process, but also on the threshold relationship between reduced blood flow and the resulting metabolic disturbances. Our findings demonstrate that inhibition of SOCs conveys marked protection of the ischemic tissue by alleviating secondary aggravation of the primary ischemic impact.
MATERIAL AND METHODS
General preparations
Eighteen male Wistar rats (350–400 g body wt) were anesthetized with 1.5% halothane in a mixture of 70% N2O/30% O2. Rectal temperature was monitored throughout the experiment and kept close to 37°C using a feedback-controlled heating pad. Animals were tracheotomized, immobilized with pancuronium bromide (0.3 mg/kg/h), and mechanically ventilated. After beginning of artificial ventilation, halothane concentration was reduced to 0.8%. Arterial and venous catheters were inserted into the femoral vessels for injection of drugs, monitoring of systemic blood pressure, and blood sampling. Blood gases were measured repeatedly and kept within physiological limits by appropriate settings of the respirator.
For electrophysiological monitoring, the skull over the parietal cortex was thinned, and a small calomel electrode was attached to the bone using a saline-soaked cotton wick. The indifferent electrode was placed over the nasal bone, and recordings of EEG and cortical DC potentials were carried out using AC- and DC-coupled differential amplifiers.
Permanent focal ischemia was produced by suture occlusion of the right MCA using a previously described remotely controlled occluding device (Kohno et al., 1995). Briefly a monofilament nylon thread (3–0 Prolene; Ethicon Co., Norderstedt, Germany) with its distal end thickened to 0.28 mm in diameter was connected to an extension catheter and passed through a guide sheath fixed to the neck of the animal. The filament was introduced into the right internal carotid artery via the proximal end of the isolated external carotid artery until the tip reached the carotid canal at the base of the skull. After positioning the animal in the magnet of the magnetic resonance scanner (see later), the MCA was occluded by advancing the filament until EEG amplitude declined and then leaving it in this position throughout the experiment. Artificial punctures of the vessel were excluded by rejecting all animals that exhibited signs of subarachnoidal hemorrhage at the end of the experiments.
Six hours after MCA occlusion, animals were removed from the magnet and measurements of regional blood flow and brain metabolites were performed, as described herein.
Therapeutic interventions
Two groups of animals were compared. The treated group (n = 7) received subcutaneous injections of 30 mg/kg LOE 908 MS (the methane sulfonate salt, courtesy of Boehringer-Ingelheim KG, Germany) dissolved in 1 ml saline at 10 min and 3 h after MCA occlusion. The untreated control group (n = 11) was injected subcutaneously with 1 ml saline at the same time points after vascular occlusion.
Magnetic resonance imaging
Magnetic resonance measurements were performed at 200 MHz using a Bruker Biospec system (Bruker Medizintechnik, Karlsruhe, Germany) with a 4.7 T magnet of 30-cm clear bore. The system was equipped with actively shielded gradient coils (Bruker Medizintechnik, Karlsruhe, Germany) with a maximum gradient strength of 100 mT/m at a gradient rise time of <250 μs. A 12-cm-diameter Helmholtz coil was used for radiofrequency transmission and a 16-mm-diameter surface coil with inductive coupling was placed over the skull for signal reception. The two coils were positioned orthogonally to each other to minimize coupling, and the transmitter coil used active tuning via a pin diode switch to further reduce coupling, while passive decoupling by crossed diodes was used on the surface coil. The active EEG electrode was fed through the center of the surface coil to attach it to the skull.
Sagittal and coronal scout scans using T1-weighted spin echo magnetic resonance imaging (echol time = 8 ms, repetition time = 600 ms) were performed for correct positioning of the head. Diffusion-weighted magnetic resonance imaging (DWI) was carried out using a multislice Stejskal-Tanner-type spin echo sequence (Stejskal and Tanner, 1965). The sequence parameters were echo time = 32 ms, repetition time = 1170 ms, with the diffusion-encoding gradient applied along the vertical y-gradient axis. The scan time (number of excitations = 2) was 5 min for a 1282 image matrix. Six coronal slices were measured with an interslice gap of 0.75 mm, a slice thickness of 1 mm, and a 40-mm field of view.
For quantitative determination of apparent diffusion coefficient (ADC), DWIs with different gradient strengths (b factor: 0, 750, 1,500 s/mm2) were recorded before and after MCA occlusion. During the first 2 h of ischemia, eight two-point ADC measurements (b = 0, 1,500 s/mm2) with 10-min temporal resolution were recorded. Then, till the end of the experiment, three-point ADC measurements (b = 0, 750, 1,500 s/mm2) were repeated every 30 min.
To minimize instrumental errors in the ADC determination, extensive data postprocessing including correction for image-specific background noise and gradient cross-talk was performed, as described in detail elsewhere (Eis and Hoehn-Berlage, 1995). ADC was calculated using the monoexponential intravoxel incoherent motion model of LeBihan et al. (1988).
Measurement of CBF
At the end of the experiment, 6 h after MCA occlusion, local CBF (1CBF) was measured by quantitative autoradiography, using [14C]iodoantipyrine (40 μCi/ml; Amersham, Braunschweig, Germany) as the freely diffusible tracer (Sakurada et al., 1978). The tracer was infused intravenously over 60 s, and arterial blood samples were collected repeatedly on preweighed filter paper. At the end of tracer infusion, the rats were frozen in situ with liquid nitrogen.
Radioactivity of 20-μm cryostat sections was measured by quantitative autoradiography, using 14C standards and a CCD camera connected to an image-processing system. 14C radioactivity of blood was measured in a liquid scintillation counter (Wallac 1410; Pharmacia, Freiburg, Germany), and 1CBF was calculated according to the algorithm of Sakurada et al. (1978).
Biochemical and histological imaging
Cryostat brain sections adjacent to those used for autoradiography were collected for imaging of regional tissue pH and of the local distribution of ATP and glucose, using fluorescence and bioluminescence imaging techniques, respectively (Kogure and Alonso, 1978; Paschen et al., 1981; Csiba et al., 1983). Images of tissue pH were calibrated with graded pH standards, and local metabolite content was quantified by correlating the optical density of bioluminescence images with metabolite values, measured in small tissue samples by conventional enzymatic techniques. Morphological injury was visualized by histological staining of cryostat sections with hematoxylin-eosin and cresyl violet. At the time of death, 6 h after vascular occlusion, the morphologically damaged tissue could be clearly differentiated by its paleness from the normally stained noninjured brain tissue.
Image analysis
Magnetic resonance images and regional maps of CBF, ATP, glucose and tissue pH were analyzed using a Macintosh computer (Apple, Cupertino, CA, U.S.A.) and the image-processing software IMAGE (National Institutes of Health, Bethesda, MD, U.S.A.). Volumes of magnetic resonance or biochemical changes were calculated by summing threshold-defined lesion areas of six coronal slices and expressed as percent of ipsilateral hemispheric volume. The threshold for ADC changes was set at 80% of control, for ATP loss at 30% of control, and for acidosis at pH 6.3. CBF thresholds of ADC and metabolite changes were determined by matching the calculated lesion volumes with the corresponding values of the flow/volume relationship.
Statistics
Values are given as means ± SD unless stated otherwise. Statistical differences between groups were evaluated by analysis of variance, followed by Student's paired t test. Time-related changes were assessed by Scheffé's F test. The level of significance was set at p < 0.05.
RESULTS
General physiological observations
Under control conditions all general physiological variables including body temperature were in the normal range (Table 1). Occlusion of the MCA led to transient increase of body temperature by ∼1°C for a few hours. The other variables did not change. In particular, blood pressure remained stable and did not differ between treated and untreated animals.
General physiological parameters
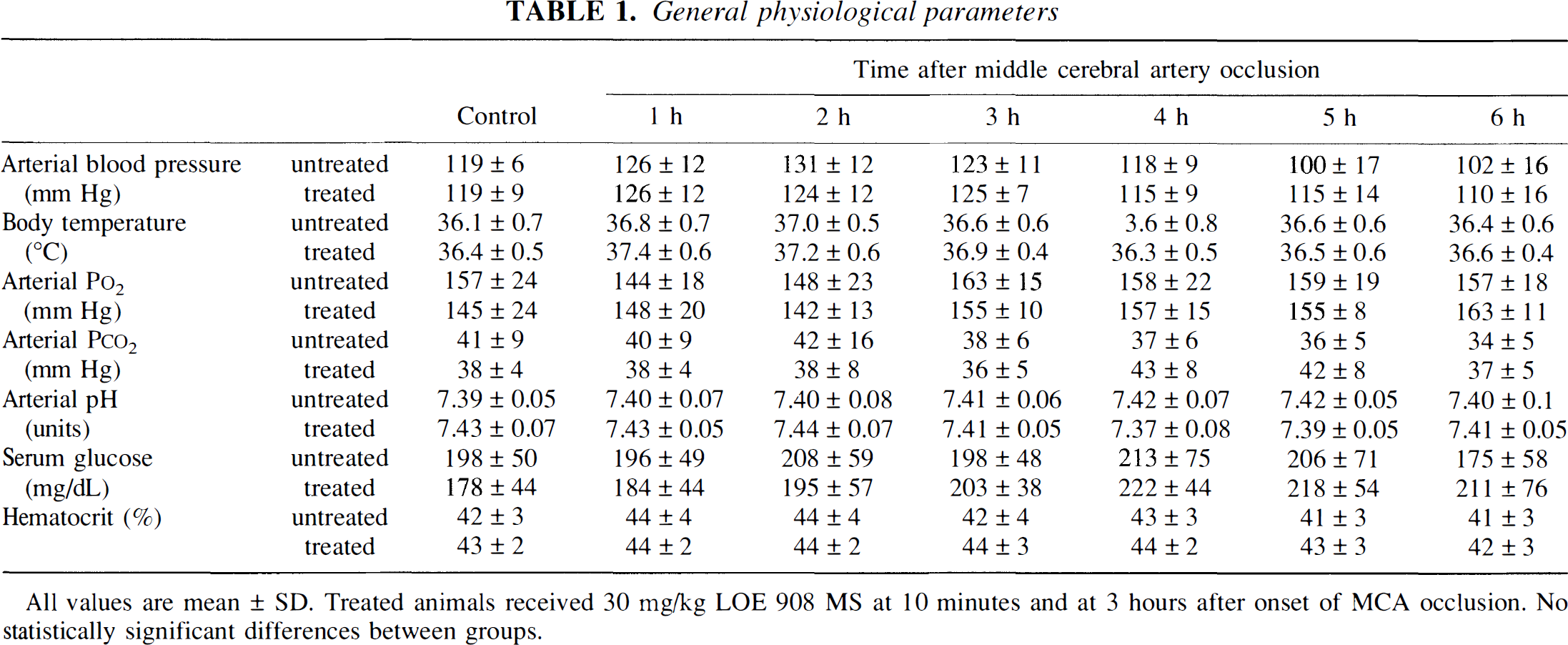
All values are mean ± SD. Treated animals received 30 mg/kg LOE 908 MS at 10 minutes and at 3 hours after onset of MCA occlusion. No statistically significant differences between groups.
Electrophysiological observations
Suture occlusion of the MCA caused an instantaneous reduction of the EEG amplitude by >50% in all animals, followed by the return to or close to normal in 5–10 min (Fig. 1). The cortical DC potential consistently exhibited a negative/positive deflection after a latency of 5–6 min lasting for ∼4 min. This deflection was frequently preceded by a brief positive shift of low amplitude. Following this initial DC deflection, subsequent DC shifts occurred at irregular intervals. The frequency of these shifts differed greatly between treated and untreated animals: In the untreated group, the total number of DC shifts was 6.5 ± 5.3 during the observation period of 6 h, whereas in the LOE 908 MS-treated group, this number was significantly reduced to 2.5 ± 2.3/6 h (p < 0.05). During each DC shift the EEG amplitude transiently declined but not to the same degree as immediately after MCA occlusion (Fig. 1).
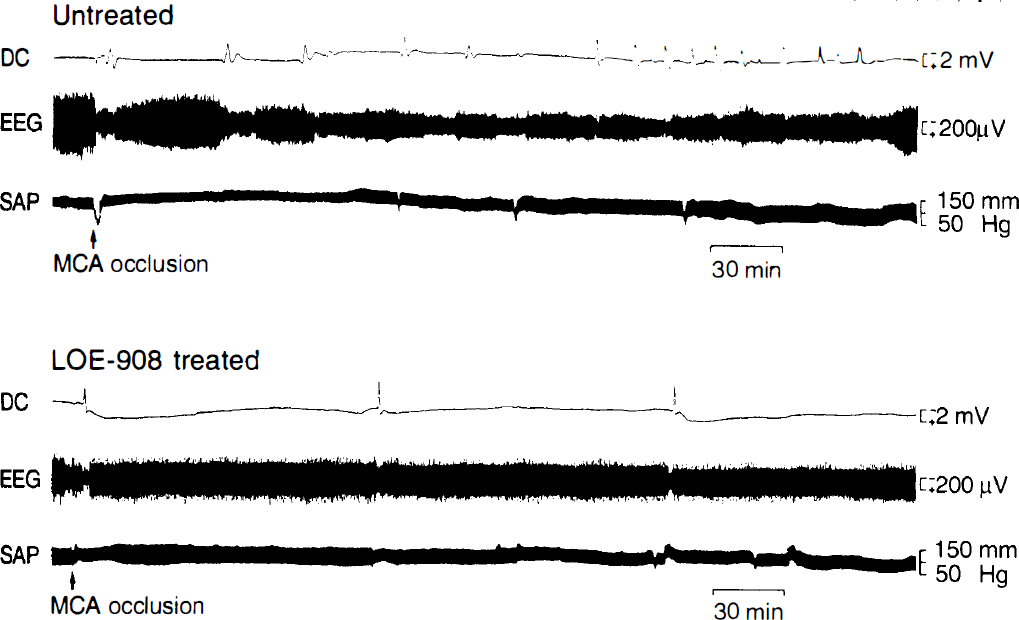
Polygraphic recording of the cortical steady potential (DC), the EEG, and the systemic arterial pressure (SAP) in an untreated (
Magnetic resonance imaging
During the control phase before MCA occlusion, DWI did not reveal interhemispheric asymmetries or group differences (Figs. 2 and 3). The ADC of water was 509 ± 31 and 635 ± 41 μm2/s in the lateral and parietal cortex and 590 ± 25 μm2/s in the basal ganglia, respectively.
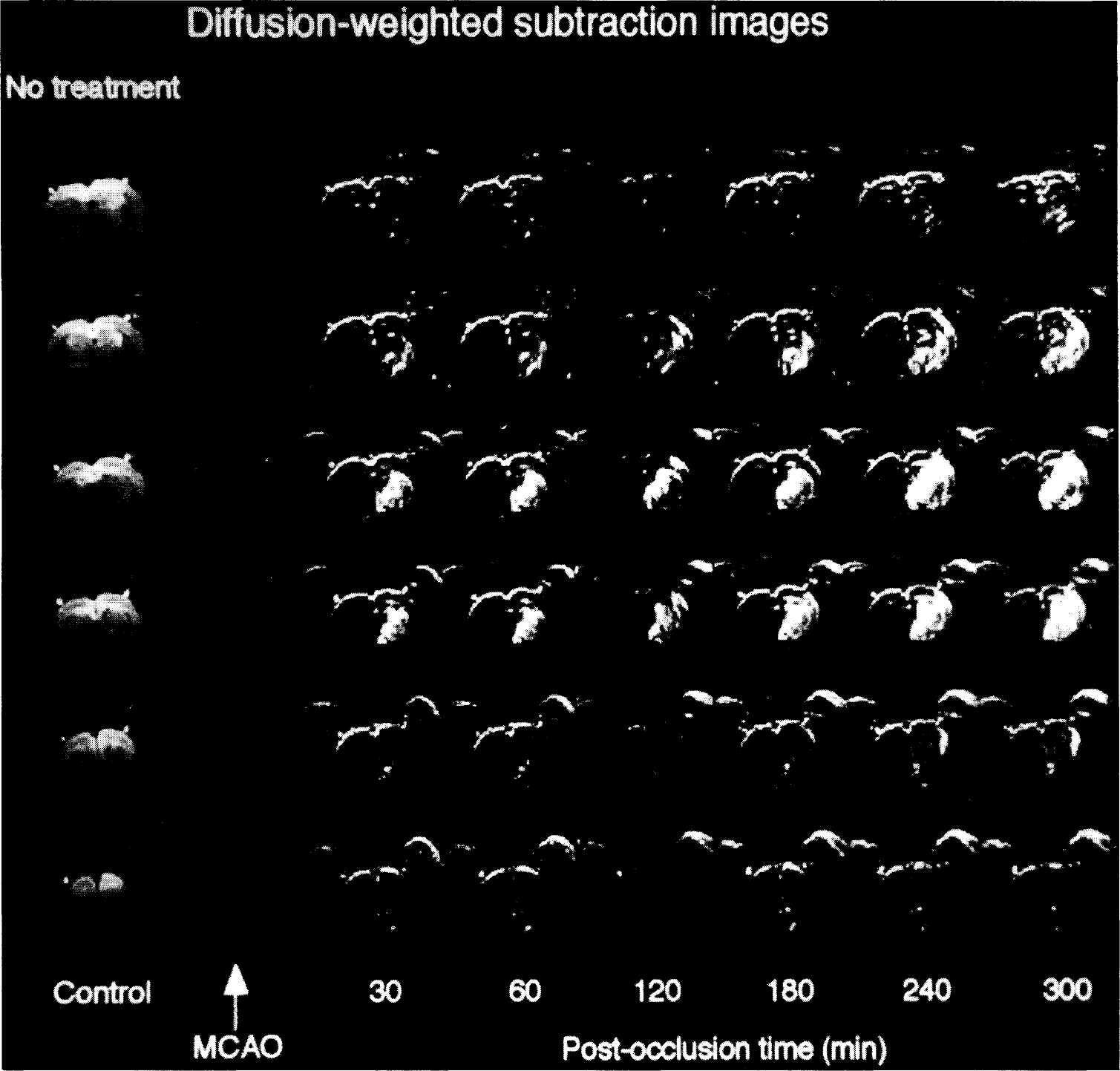
Diffusion-weighted images of six coronal brain slices of untreated rat after middle cerebral artery occlusion. Diffusion-weighted magnetic resonance imaging changes are highlighted by subtracting control images from ischemic images. Note gradual volume expansion of the hyperintense ischemia lesion with ongoing ischemia time.

Diffusion-weighted images of six coronal brain slices of LOE 908 MS-treated rat. Same data presentation as in Fig. 2. LOE 908 MS treatment was started 10 min after middle cerebral artery occlusion. Note absence of lesion expansion throughout the observation period of 6 h.
MCA occlusion led to an increase in DWI signal intensity in the ipsilateral caudate-putamen and part of the temporoparietal cortex. This hyperintensity was visible already in the first postocclusion image recorded between 0 and 10 min after onset of ischemia (Fig. 2). In the untreated group both, intensity and size of the DWI-visible lesion gradually increased during the observation period of 6 h. In contrast, the lesion size remained stable in all LOE 908 MS-treated animals (Fig. 3).
For quantitative analysis of the volume changes, the ADC lesion volume (defined as the decline of ADC to <80% of control) was measured repeatedly after MCA occlusion and normalized to the initial ischemic impact, recorded at 10 min after the onset of ischemia (Fig. 4). In the untreated animals, the ADC lesion volume briefly declined by ∼20% and then gradually expanded until, after 6 h, it exceeded the initial volume by ∼50%. In the treated animals, the lesion volume transiently declined by even 35% and then stabilized at a level that was ∼20% below the initial value. The difference between treated and untreated groups became significant already 10 min after the injection of LOE 908 MS, and after 6 h the normalized lesion volume of the treated group was 46% smaller. In absolute values, the ADC lesion volume after 6 h of untreated and treated animals amounted to 44 ± 15% and 16 ± 18% of the ipsilateral hemisphere, representing a lesion reduction by 64%.
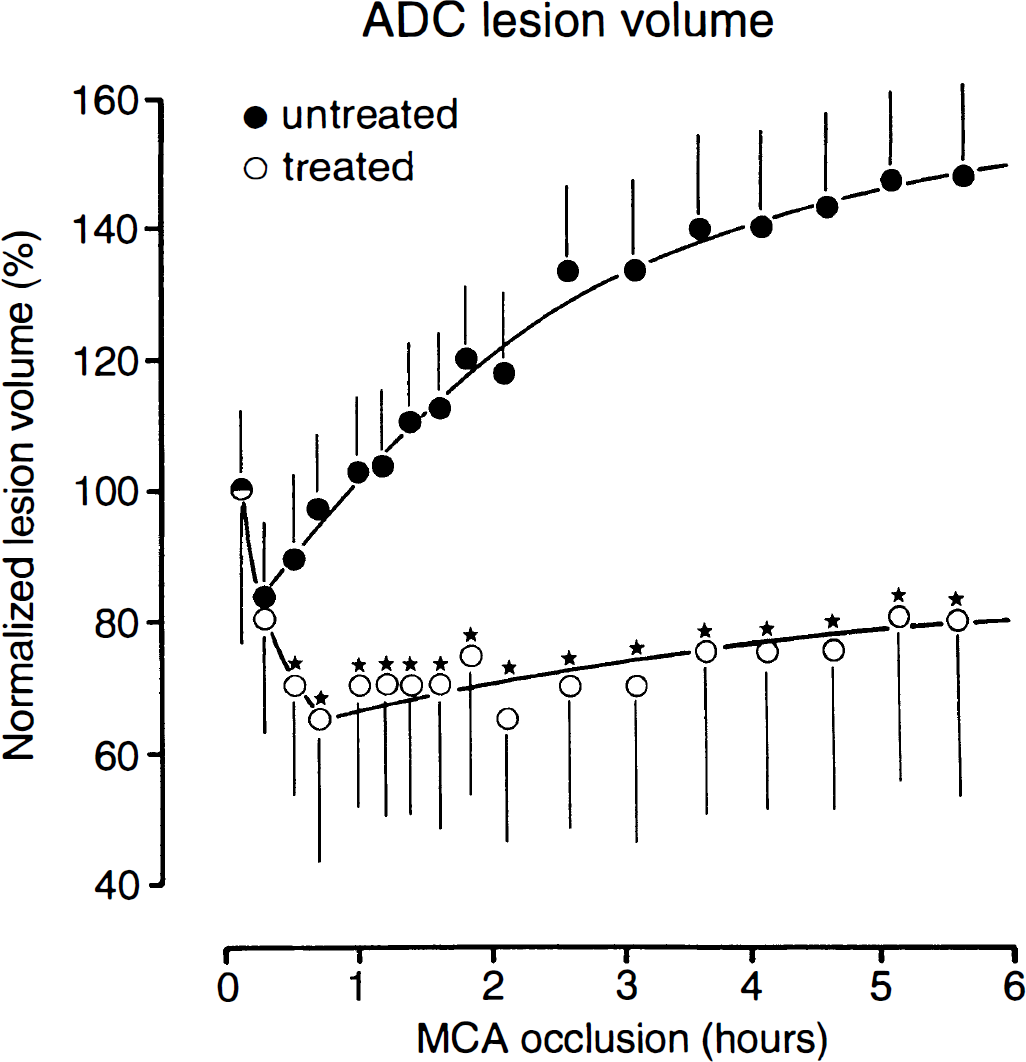
Temporal evolution of the apparent diffusion coefficient (ADC)–visible lesion after middle cerebral artery occlusion in rats with (treated) or without (untreated) postocclusion application of LOE 908 MS. The lesion volume (means ± SEM) was calculated for an ADC threshold of 80% of control and normalized to the initial ischemic impact. Inhibition of lesion growth by LOE 908 MS leads to significant volume differences between treated and untreated groups after 30 min of ischemia (*p < 0.05).
The severity of ADC changes was measured in the center of the ischemic territory. In the untreated animals, ADC declined to 71% of control within 30 min of vascular occlusion, followed by further decline to ∼66% after 1 h and to 50% at the end of the 6-h observation period. In the treated animals, the corresponding values were 78% after 30 min, 75% after 1 h, and 60% after 6 h. These changes are significantly less severe than in the untreated animals, demonstrating that LOE 908 MS reduces not only the volume but also the severity of the ADC-visible lesion.
Blood flow measurements
At the end of each experiment, 6 h after MCA occlusion, regional CBF was measured by [14C]iodoantipyrine autoradiography. In both groups flow values gradually declined from the periphery to the center of the ischemic territory, but the severity of the flow disturbance was distinctly less pronounced in the LOE 908 MS-treated group. This was demonstrated by measuring the volume of reduced blood flow as a function of stepwise declining flow thresholds. As shown in Fig. 5, the ischemic territory was significantly smaller in the treated animals irrespective of the chosen flow threshold. LOE 908 MS treatment thus significantly improved collateral blood flow to the ischemic territory.
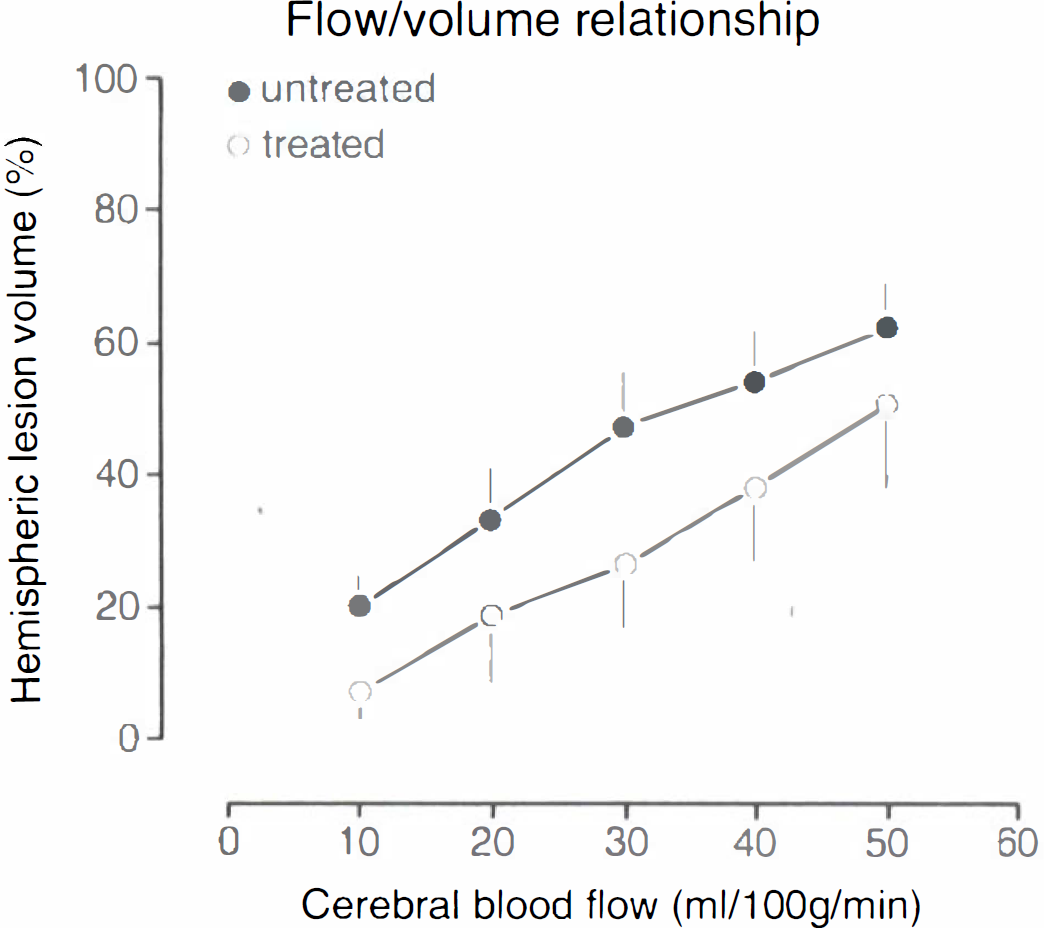
Comparison of blood flow reduction in untreated and LOE 908 MS-treated rats submitted to 6-h middle cerebral artery occlusion. Volumes of reduced blood flow (means ± SEM, expressed as percent of ipsilateral hemispheric volume) were calculated for stepwise declining flow thresholds. LOE 908 MS shifts the flow/volume relationship to smaller lesion volumes, indicating improvement of collateral blood flow.
Biochemical and morphological injury
The regions of metabolic and morphological injury were identified at the end of the 6-h observation period by imaging the histological lesion and the local disturbances of ATP, glucose, and pH. In the center of the MCA territory, ATP and glucose were consistently depleted and pH was severely acidic. Visual inspection revealed a marked reduction by LOE 908 MS treatment of the lesion size for all these variables (Fig. 6).
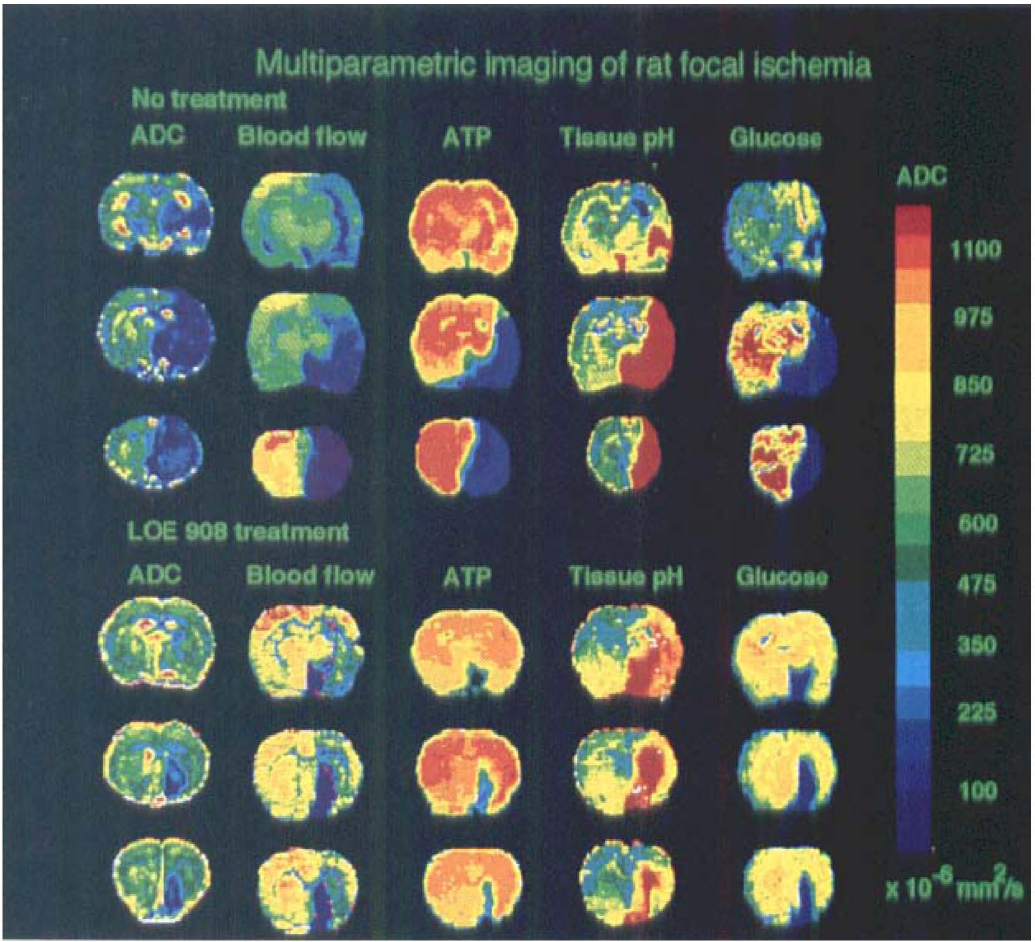
Comparison of apparent diffusion coefficient (ADC) maps with pictorial measurements of blood flow, ATP, glucose, and tissue pH in untreated and LOE 908 MS-treated rats submitted to 6-h middle cerebral artery occlusion. Quantitative ADC maps were calculated from three diffusion-weighted images with increasing b factors. Biochemical measurements were color-coded for highlighting pathological changes. Images were taken from three levels, passing through the frontal, central, and posterior parts of brain infarcts, respectively. Note smaller and less severe biochemical and magnetic resonance–visible lesions in the treated animal.
The degree of protection was quantified by planimetric evaluation of cresyl violet-stained sections and of ATP and pH images at six coronal levels corresponding to the planes of ADC imaging (Fig. 7). The volume of ATP depletion was 32.8 ± 17.1% of the ipsilateral hemisphere in the untreated animals as compared with 12.9 ± 12.1% in the treated ones. This corresponds to a reduction of the lesion volume by 61%.
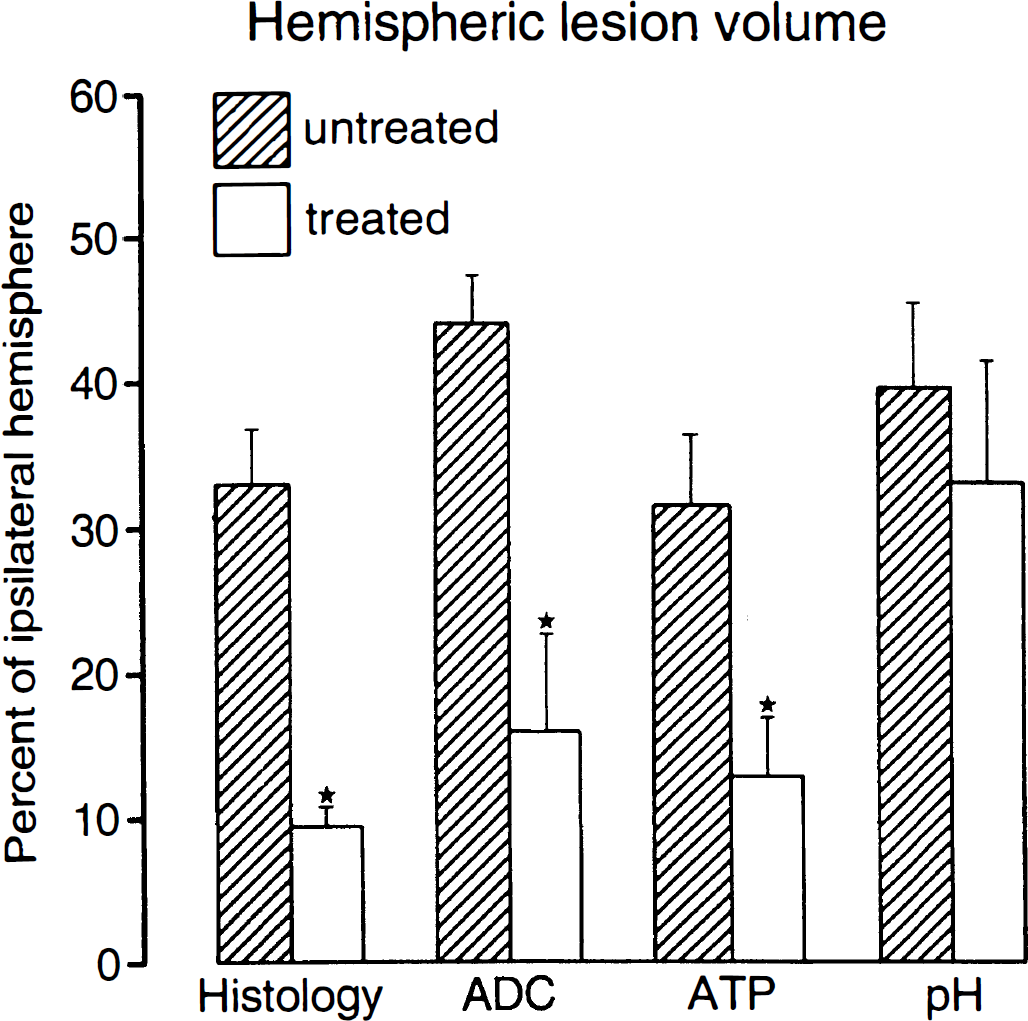
Comparison of the volumes of morphological, biophysical, and biochemical changes in untreated and LOE 908 MS-treated rats submitted to 6-h middle cerebral artery occlusion (means ± SEM). The morphological changes were visualized by histological staining, the biophysical changes by apparent diffusion coefficient imaging, and the biochemical changes by imaging of ATP depletion and acidosis. The lesser reduction by LOE 908 MS of acidosis as compared to ATP depletion or histological injury indicates that treatment reduces the volume of the core but not of the penumbra of the ischemic infarct (*p < 0.05).
The volume of tissue acidosis, defined as the decline of tissue pH to below 6.3, was 39.7 ± 21.4% of the ipsilateral hemisphere in the untreated group as compared with 33.7 ± 26.4% in the treated group, corresponding to a nonsignificant lesion reduction by only 15%. The volume of morphological injury was similar to that of ATP loss and amounted to 33.0 ± 14.5% of the ipsilateral hemisphere in the untreated group and to 9.6 ± 6.3% in the treated group. This difference corresponds to a reduction of lesion volume by 71%.
ATP depletion is a measure of infarct core, whereas tissue acidosis additionally detects the penumbra. Therefore, the larger difference of the ATP-visible lesion volumes demonstrates that LOE 908 MS mainly reduces the size of the infarct core but has little effect on the volume of the penumbra.
Correlation analysis
The flow thresholds for the magnetic resonance-visible changes and the metabolic disturbance were estimated by matching the lesion volumes detected by magnetic resonance or biochemical imaging with the corresponding values of the flow/volume relationship shown in Fig. 5. According to these measurements, the flow thresholds for the decline of ADC, ATP depletion, and morphological injury were lower in the treated group, but that of acidosis markedly increased (Table 2). This finding demonstrates that LOE 908 MS diminishes the volume of the ischemic core but not that of the penumbra of the evolving infarct.
Thresholds of blood flow for the induction of biophysical, biochemical, and morphological changes after 6 hours of middle cerebral artery occlusion of rat
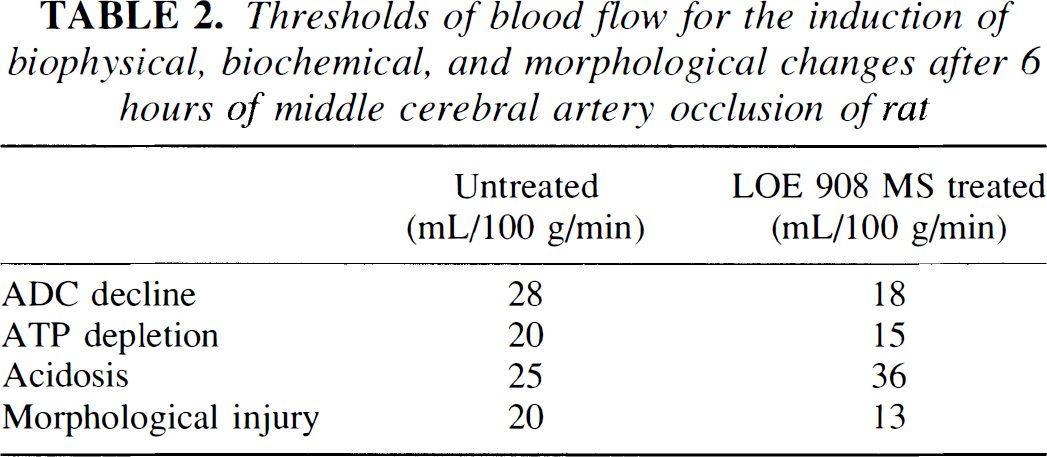
The decline of the apparent diffusion coefficient (ADC) of brain water was defined as the reduction to less than 80% of control, ATP depletion as the decline to less than 30% of control, acidosis as the decline of tissue pH below 6.3, and morphological injury as the loss of histological staining. Thresholds were calculated by matching the mean lesion volumes shown in Fig. 7 with the corresponding values of the flow/volume relationship shown in Fig. 5.
DISCUSSION
Despite considerable progress in the therapy of experimental ischemia, the results of clinical stroke treatment are still unsatisfactory (del Zoppo, 1995; Grotta, 1995). Some of the experimentally most potent drugs, notably the various kinds of glutamate antagonists, are not suited for clinical treatment because of unacceptable side effects (Auer, 1994; Xue et al., 1994). Others, like spin traps (Folbergrova et al., 1995) or antiinflammatory drugs (del Zoppo, 1994; Feuerstein et al., 1994), are of benefit particularly after transient focal ischemia with no or only moderate improvement of ischemic injury after permanent vascular occlusion (Müller et al., 1995). Therefore, alternative approaches to prevent brain infarcts are urgently required.
The present study demonstrates that the blocker of store-operated nonselective cation channels, LOE 908 MS, has promising potentials for the treatment of stroke. Both the dose and the application time chosen for the present study are compatible with clinical treatment, and the main finding was a marked retardation of infarct expansion during the initial 6 h of vascular occlusion, which led to the reduction of the volume of ATP-depleted tissue by 61% and that of the histologically visible tissue necrosis by 71% as compared with untreated animals. Temperature changes, in contrast, are probably not of importance because body temperature was carefully controlled and because animals were not craniotomized to preserve thermal stabilization of the ischemic territory by the normothermically perfused surrounding tissues.
The improvement of blood flow is not the result of general cardiovascular alterations, as shown by the absence of any changes of systemic physiological variables. A possible explanation could be the prevention of leukocyte adhesion at the capillary wall because nonselective cation channels play a prominent role in cell activation (Berridge, 1987; Irvine et al., 1988). Another flow-promoting mechanism could be the reduction of ischemic cell swelling and hence of microcirculatory compression because the activation of nonselective cation channels contributes to the increase of intracellular osmolality by gating sodium influx (Krautwurst et al., 1993). A protective effect on ischemic fluid shifts has, in fact, been documented in the present study by demonstrating a significant reduction of the ADC-visible lesion volume.
The other major protective mechanism of LOE 908 MS seems to be the preservation of energy metabolites under conditions of critically reduced blood flow. In previous studies from our laboratory (Mies et al., 1991; Kohno et al., 1995), evidence has been provided that the CBF threshold for ATP depletion steadily increases from 13 ml 100 g–1 min–1 after 30-min ischemia to 19 ml 100 g–1 min–1 after 1 h and to 23 ml 100 g–1 min–1 after 6 h. The present CBF threshold of 20 ml 100 g–1 min–1 for the untreated group at 6 h of MCA occlusion comes close to the expected value after this duration of ischemia. CBF threshold for the treated group, in contrast, remained stable at 15 ml 100 g–1 min–1, which corresponds with the prevention of infarct core expansion.
The retardation of the threshold progression to higher CBF values with time may be due to an inhibition of periinfarct depolarizations, the number of which significantly declined in the treated animals. There is now ample evidence that the generation of periinfarct depolarizations contributes to infarct growth presumably because the constrained collateral blood supply does not couple to the increased energy demands required for restoring the disturbed ion homeostasis (for review see Hossmann, 1996). The present study is in line with this concept. In fact, reduction of periinfarct depolarization by LOE 908 MS prevented both the gradual increase of the CBF threshold for ATP depletion and the expansion of the magnetic resonance–visible lesion size, which previously has been associated with the generation of periinfarct depolarizations (Iijima et al., 1992; Gyngell et al., 1995; Busch et al., 1996).
Another mechanism for the depolarization-induced injury in untreated animals could be progressive uncoupling of oxidative phosphorylation (Katsura et al., 1994; Abe et al., 1995). The massive influx of calcium during each depolarization in combination with the hypoxic retardation of calcium countertransport across the plasma membrane could lead to progressive mitochondrial calcium loading and a reduction of the mitochondrial oxidative capacity (Nicholls, 1985). Suppression of periinfarct depolarizations by LOE 908 MS may alleviate this pathophysiology and provides another explanation for the stabilization of the CBF threshold for ATP depletion and the prevention of infarct expansion.
An interesting side aspect of our study was the observation that the ADC lesion that was visible immediately after vascular occlusion became smaller in size a few minutes later. This volume reduction was modest and transient in the untreated animals but more pronounced and long-lasting in the treated ones. The most likely reason for the partial reversal of the initial ADC lesion is the activation of the collateral circulation, which takes some time to establish (Shima et al., 1983) and seems to be more efficient in the treated animals. However, it cannot be excluded that the initial ADC lesion volume is modulated by the early depolarization that occurs shortly after MCA occlusion (Iijima et al., 1992), in agreement with the findings in ∼50% of the investigated animals. The first ADC image of the evolving ischemic infarct may therefore exaggerate the actual lesion size and should be interpreted with caution, particularly in regard to therapeutic interventions.
Obviously, retardation of infarct core expansion as shown in this study is not equivalent with the reversal of the ischemic impact, at least not during the observation period of 6 h. The main therapeutic interest of LOE 908 MS is therefore the extension of the therapeutic window for subsequent flow-promoting interventions. This could be of considerable importance for improving the therapeutic outcome of thrombolysis, which is still unsatisfactory even when therapy is started within a few hours after stroke onset (Hacke et al., 1995; Marler et al., 1995).
The present study does not provide information on the cerebroprotective aspect of LOE 908 MS on the chronic infarct situation because experiments were terminated at 6 h after vascular occlusion. However, during this time LOE 908 MS markedly retards the manifestation of irreversible tissue injury and thus provides a rather long therapeutic window for the restoration of blood recirculation by thrombolysis. This would give ample time to evaluate the risks of bleeding and to allow careful selection of patients that can be expected to benefit from this therapy. Experiments are underway to explore this challenging prospect.
Footnotes
Acknowledgments:
Dr. D. Arndts and Dr. Dr. U. Pschorn contributed to the study design and provided valuable information on the pharmacology of the drug. We thank Ms. U. Uhlenküken, Ms. Ch. Stratmann, and Ms. U. Beckmann for their excellent technical assistance, and we thank Ms. D. Schewetzky and Ms. M. Hahmann for the careful preparation of the manuscript. The artwork was done by Mr. B. Huth and Ms. I. Mühlhöver.
