Abstract
We conducted a study using diffusion-weighted (DWI) and perfusion-weighted (PWI) magnetic resonance imaging (MRI) to evaluate the efficacy of thrombolysis in an embolic stroke model with recombinant tissue plasminogen activator (rt-PA) and hirulog, a novel direct-acting antithrombin. DWI can identify areas of ischemia minutes from stroke onset, while PWI identifies regions of impaired blood flow. Right internal carotid arteries of 36 rabbits were embolized using aged heterologous thrombi. Baseline DWI and PWI scans were obtained to confirm successful embolization. Four animals with no observable DWI lesion on the initial scan were excluded; therefore, a total of 32 animals were randomized to one of three treatment groups: rt-PA (n = 11), rt-PA plus hirulog (n = 11), or placebo (n = 10). Treatment was begun 1 h after stroke induction. Intravenous doses were as follows: rt-PA, 5 mg/kg over 0.5 h with 20% of the total dose given as a bolus; hirulog, 1 mg/kg bolus followed by 5 mg/kg over 1 h. MRI was performed at 2, 3, and 5 h following embolization. Six hours after embolization, brains were harvested, examined for hemorrhage, then prepared for histologic analysis. The rt-PA decreased fibrinogen levels by 73%, and hirulog prolonged the aPTT to four times the control value. Posttreatment areas of diffusion abnormality and perfusion delay were expressed as a ratio of baseline values. Significantly improved perfusion was seen in the rt-PA plus hirulog group compared with placebo (normalized ratios of the perfusion delay areas were as follows: placebo, 1.58, 0.47–3.59; rt-PA, 1.12, 0.04–3.95; rt-PA and hirulog, 0.40, 0.02–1.08; p < 0.05). Comparison of diffusion abnormality ratios measured at 5 h showed trends favoring reduced lesion size in both groups given rt-PA (normalized ratios of diffusion abnormality areas were as follows: placebo, 3.69, 0.39–15.71; rt-PA, 2.57, 0.74–5.00; rt-PA and hirulog, 1.95, 0.33–6.80; p = 0.32). Significant cerebral hemorrhage was observed in one placebo, two rt-PA, and three rt-PA plus hirulog treated animals. One fatal systemic hemorrhage was observed in each of the rt-PA groups. We conclude that rt-PA plus hirulog improves cerebral perfusion but does not necessarily reduce cerebral injury. DWI and PWI are useful methods for monitoring thrombolysis.
Thrombolytic therapy with recombinant tissue plasminogen activator (rt-PA) has begun to show promise in the early treatment of ischemic stroke (Hacke et al., 1995; NINDS rt-PA Stroke Study Group, 1995). Though treatment with rt-PA appears beneficial, incomplete clot lysis and distal embolization may limit its benefit. While higher doses of rt-PA may be more efficacious, the benefit appears to be offset by an increased risk of cerebral hemorrhage (Levy et al., 1994; Hacke et al., 1995). Adjunctive therapy with heparin has not been shown to offer added benefit over rt-PA alone (Carter et al., 1992) and may increase the risk of cerebral hemorrhage. Heparin also does not appear to be effective in the treatment of acute stroke in humans (Duke et al., 1986). This may be due to its dependence on antithrombin III (AT-III) for activation and its inability to inactivate clot-bound thrombin (Weitz et al., 1990). Hirudin, a polypeptide found in leech saliva, possesses potent antithrombotic properties as it directly binds and inactivates thrombin independently of AT-III. Hirulog is a synthetic polypeptide derived from hirudin but with a lower affinity for thrombin and shorter half-life (Cannon et al., 1993). Several animal models of thrombosis and vascular injury have shown that at comparable levels of anticoagulation, both of these compounds are superior to heparin for preventing vascular reocclusion (Heras et al., 1990; Kelly et al., 1991) and, in combination with a thrombolytic agent, can significantly accelerate fibrinolysis compared with a thrombolytic plus heparin or a thrombolytic alone (Klement et al., 1992). While several laboratory studies and a few clinical trials in myocardial infarction patients have found these antithrombins to be efficacious, they have yet to be studied in cerebrovascular disease.
We employ a rabbit model of embolic stroke incorporating the advanced magnetic resonance imaging (MRI) techniques of diffusion-weighted imaging (DWI) and perfusion-weighted imaging (PWI). While embolic stroke models are ideal for studying thrombolysis and most closely resemble human stroke, a major drawback is variability in lesion size and distribution. To control for this, other investigators have used prerandomization angiograms (Chehrazi et al., 1989; Bednar et al., 1990; Overgaardet al., 1994), cerebral blood flow measurements (Bednar et al., 1990; Overgaard et al., 1994), or radiolabeled emboli (Thomas et al., 1993) to confirm that a successful lesion was created. Disadvantages of these methods are that they are invasive and lack significant resolution to accurately determine areas of ischemia.
DWI can image stroke as early as 10 min from the onset of ischemia, while PWI can assess vessel patency. Sequential scans at multiple time points can noninvasively be used to follow the course of ischemic stroke and its response to treatment. DWI detects early ischemic changes caused by cytotoxic edema due to its sensitivity to micromolecular water movements (Moseley et al., 1990a; Back et al., 1994; Mintorovitch et al., 1994). A quantitative estimate of cytotoxic edema is determined by computing the apparent diffusion coefficient (ADC) (Le Bihan et al., 1988). Experimental work has shown that ADC decreases of ≥30–50% relative to nonischemic brain represent areas of ischemic damage as seen on histopathology (Moseley et al., 1990b; Knight et al., 1994). These regions of low ADC are also reversible (Minematsu et al., 1992) and have been shown to decrease in size in response to treatment with neuroprotective compounds (Kucharczyk et al., 1991; Minematsu et al., 1993; Lo et al., 1994). PWI is complementary to DWI as it assesses vascular integrity. Changes in T2* signal intensity characterize bolus transit in and out of tissue. Fitted maps of relative cerebral blood volume (rCBV) and bolus peak delay (BPD) can be used to quantitatively characterize cerebral perfusion (Rosen et al., 1989; de Crespigny et al., 1993; Kucharczyk et al., 1993).
In this study, we tested the following hypotheses: (a) Combined therapy with rt-PA and hirulog decreases infarct size compared with rt-PA alone; (b) combined therapy accelerates fibrinolysis with rapid return of cerebral perfusion; (c) combined therapy does not increase the risk of cerebral hemorrhage; (d) sequential DWI and PWI are useful for monitoring therapy for ischemic stroke. Preliminary reports of this study have appeared in abstract form (Yenari et al., 1994a, 1995a).
MATERIALS AND METHODS
Surgical procedures
Following approval by the institutional animal panel on laboratory animal care, 36 male New Zealand White rabbits weighing 2.5–3.0 kg were intubated and anesthetized with 3% halothane. Animals were ventilated on an animal respirator using a tidal volume of 45 ml, rate of 20 breaths/min, and oxygen/air mixture of ∼25%. Once adequate anesthesia was attained as revealed by a lack of hind limb withdrawal to toe pinch, halothane was decreased to 1–1.5%. The femoral artery was catheterized to measure MABP and sample blood. The femoral vein was catheterized to deliver fluids and contrast agent. A separate marginal ear vein catheter was placed to infuse study drugs. All lines were kept open with normal saline. No heparin was used. A right lateral neck dissection was performed and branches of the external carotid artery (ECA) were ligated. The facial branch was cannulated in a retrograde fashion toward the ostium of the internal carotid artery (ICA). A temporary aneurysm clip was placed on the common carotid artery (CCA) to prevent systemic injection of the clot. A single embolus 5 mm in length and weighing 4.5–5.5 mg was injected into the cerebral circulation via the ECA catheter and flushed with 6 ml of normal saline. The clot was observed to travel up the proximal 10 mm of the ICA under direct visualization by an operating microscope. The catheter was removed and the ECA ligated. The CCA clip was removed to restore flow to the ICA. The animal, along with MRI-compatible monitoring equipment, was then transferred to the MRI scanner for imaging.
Four additional animals served as sham controls to determine whether ligation of the ECA or catheter placement and injection of normal saline contributed to MRI or histopathologic abnormality. Two of the animals underwent only ligation of the ECA, then were imaged 6 h following ligation. The other two animals underwent catheter placement, followed by injection of 6 ml normal saline and ECA ligation, and were then imaged 6 h later. Histopathology was performed following the scans. Brains were examined under light microscopy for evidence of ischemia using the criteria described in Histopathology and Hemorrhage Analysis.
Preparation of emboli
Aged heterologous emboli were used and made as previously described (Thomas et al., 1993; Yenari et al., 1995b). Arterial blood from donor rabbits was drawn on 3.8% sodium citrate; 0.25 ml of calcium chloride (1 M) and 80–100 U bovine thrombin were added to the sample. Blood was then drawn into PE 160 tubing and allowed to clot. Clots were aged for 18–24 h prior to use. Fragments were cut into 5-mm lengths and weighed. Clots weighing between 4.5 and 5.0 mg were selected and suspended in phosphate-buffered solution with 1% bovine serum albumin.
Physiologic monitoring
Vital signs including heart rate, MABP, respirations, oxygen saturation, depth of anesthesia, and rectal temperature were monitored during surgery and MRI scanning. Arterial blood gas measurements were made prior to clot injection and ∼1 h following ischemia. Ventilator adjustments and supplementation with intravenous bicarbonate were performed based on arterial blood gas. Hematocrit and blood glucose were measured before and after clot injection and recorded. MABP was maintained above 55 mm Hg during the surgical procedure and clot injection and above 65 mm Hg during scanning using intravenous phenylephrine as needed. Rectal temperature was maintained between 37 and 39°C using heating blankets.
MRI scanning
The animal was placed into a 2T Bruker CSI magnet in the sphinx position with its head secured in a 70-mm (outer diameter) head volume radiofrequency coil operating at 85 MHz. Imaging parameters were as follows: field of view 35 × 70 mm, 128 × 256 matrix, 2-mm slice. Diffusion-weighted spin echo (repetition time 1,500 ms, echo time 82 ms) images were acquired with the diffusion-sensitizing gradients applied in the z (slice-select) direction with amplitudes of 0 and 5.5 G/cm (b values of 12 and 1,100 s/mm2). A second echo at 92 ms was used to both phase- and magnitude-navigate the phase-encoded echo. Eight coronal slices were acquired in 4 min. ADC maps were generated from two diffusion-weighted images using a voxel-by-voxel fitting routine. A bolus-tracking PWI method was used in which four coronal slices were repeatedly acquired during rapid injection of a contrast agent (0.2 mmol/kg gadopentetate dimeglumine). T2-weighted spin echo echo planar imaging was used to track the contrast bolus. Imaging parameters were as follows: 64 × 64, echo time 82 ms, 70-mm field of view, 2-mm slices. All four slices were obtained every 2 s over a 32-s period prior to and during bolus injection of contrast agent. Diffusion and perfusion MR images were performed sequentially at multiple time points. The first image was acquired within 30 min of clot injection. This image served as a baseline as well as confirmation that an ischemic lesion was successfully created. Only animals with DWI-observed lesions at 30 min were used for the experiment. Subsequent scans were performed at 2, 3, and 5 h following embolization. Each set of MRI scans required ∼20–30 min to complete.
Treatment
Animals with initial DWI lesions were randomized into three treatment categories: rt-PA alone, rt-PA plus hirulog, and controls. Compounds were obtained directly from the manufacturers as gifts (hirulog; Biogen, Cambridge, MA, U.S.A.; rt-PA, Genentech, So. San Francisco, CA, U.S.A.). Investigators involved in surgery and data analysis were blinded to treatment. All drugs were given 60 min after embolization intravenously through a marginal ear vein. Animals receiving rt-PA received 5.0 mg/kg over 30 min with 20% of the dose given as an initial bolus. rt-PA dose was chosen based on our pilot studies that showed that fibrinogen levels decreased by 70–80% (Yenari et al., 1994a) as well as previously published reports by others using a similar animal model (Zivin et al., 1988; Russell et al., 1992; Thomas et al., 1993). Hirulog was simultaneously infused with rt-PA over 1 h and was given as a bolus of 1 mg/kg followed by 5 mg/kg over 1 h. Hirulog dose was chosen based on recommendations from the manufacturer to prolong the activated partial thromboplastin time (aPTT) to two to three times the baseline value.
Coagulation studies
Fibrinogen and aPTT were drawn prior to treatment and at the completion of the final drug infusion. With decreased fibrinogen levels, however, the aPTT is confounded, and a direct test of thrombin inhibition would be more specific. We nevertheless chose to monitor aPTT since this measure was previously used by others in both experimental and clinical studies (Heras et al., 1990; Klement et al., 1992; Cannon et al., 1993; GUSTO IIa, 1994). To ensure blinding of the study, blood work was measured by an independent laboratory and the results disclosed only at the end of the project.
Histopathology and hemorrhage analysis
Six hours following embolization, animals were killed with a barbiturate overdose and immediately perfused transcardially with normal saline followed by 10% formalin. Surgical wound sites were inspected for gross hemorrhage and graded 0 if no hemorrhage was seen, 1 if a small amount of hemorrhage or serosanguineous fluid was seen, and 2 if large amounts of blood (>15 ml) were present. Animals that developed hemodynamic instability or died due to hemorrhage were also given a score of 2. The skulls were removed with the brain intact and allowed to fix for 24 h in formalin. Afterward brains were removed and sectioned in similar planes and levels as the corresponding MR images. Seven to eight brain sections were visually inspected for gross hemorrhage and scored by a rater blinded to treatment using a semiquantitative scale. Cerebral hemorrhages visible to the naked eye were scored 0 if there was no gross hemorrhage, 1 if there was a small or minor hemorrhage, and 2 if there was a large hemorrhage or hematoma (Fig. 1).
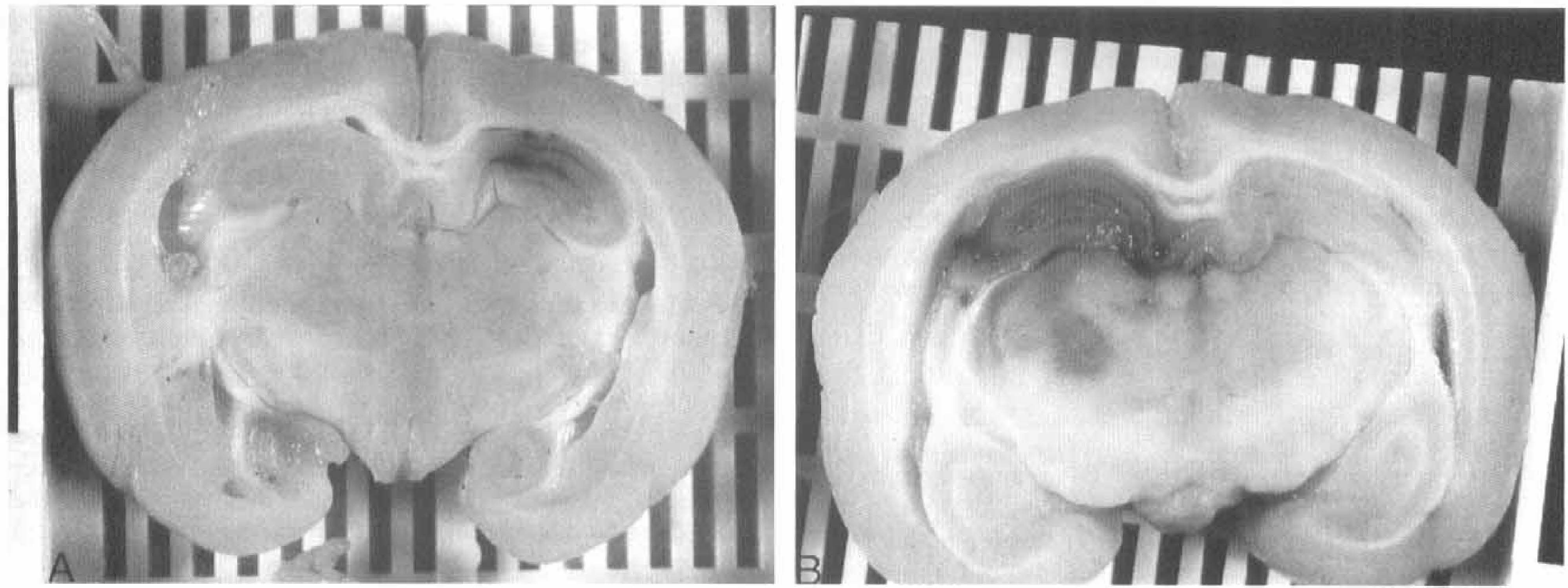
Scoring of gross cerebral hemorrhage. Examples of grades 1 (
Brain sections were then paraffin embedded and further cut into 9-mm-thick slices and stained with hematoxylin and eosin. Under light microscopy at low (4× magnification) and high (40× magnification) power, areas of ischemic neuronal damage (IND) were delineated using an established grading system (Little, 1978; Steinberg et al., 1986, 1989). IND was defined as a region of moderate to severe neuronal shrinkage with increased nuclear basophilia and nuclear pyknosis. IND areas were calculated using a computer-assisted image analysis system (MCID; Imaging Research, St. Catherines, Ontario, Canada) and were correlated with areas of high signal from the corresponding 5-h DWI scans.
Analysis of DWI lesion size
With use of a computer-assisted image analysis system (MRVision, Menlo Park, CA, U.SA.), DWI lesion areas were computed by summing regions of increased signal intensity from each of the eight slices and dividing by the total area of the brain slices. These regions of increased signal intensity corresponded to areas of decreased ADC by ≥30% measured from the fitted ADC maps. Lesion areas from later time points were each normalized to the initial baseline scan (Yenari et al., 1994a).
Analysis of perfusion scans
Bolus transit curves were obtained from the perfusion images at each of the four coronal slices. Maps of the BPDs and the rCBV were then constructed on a voxel-by-voxel basis (Fig. 2).
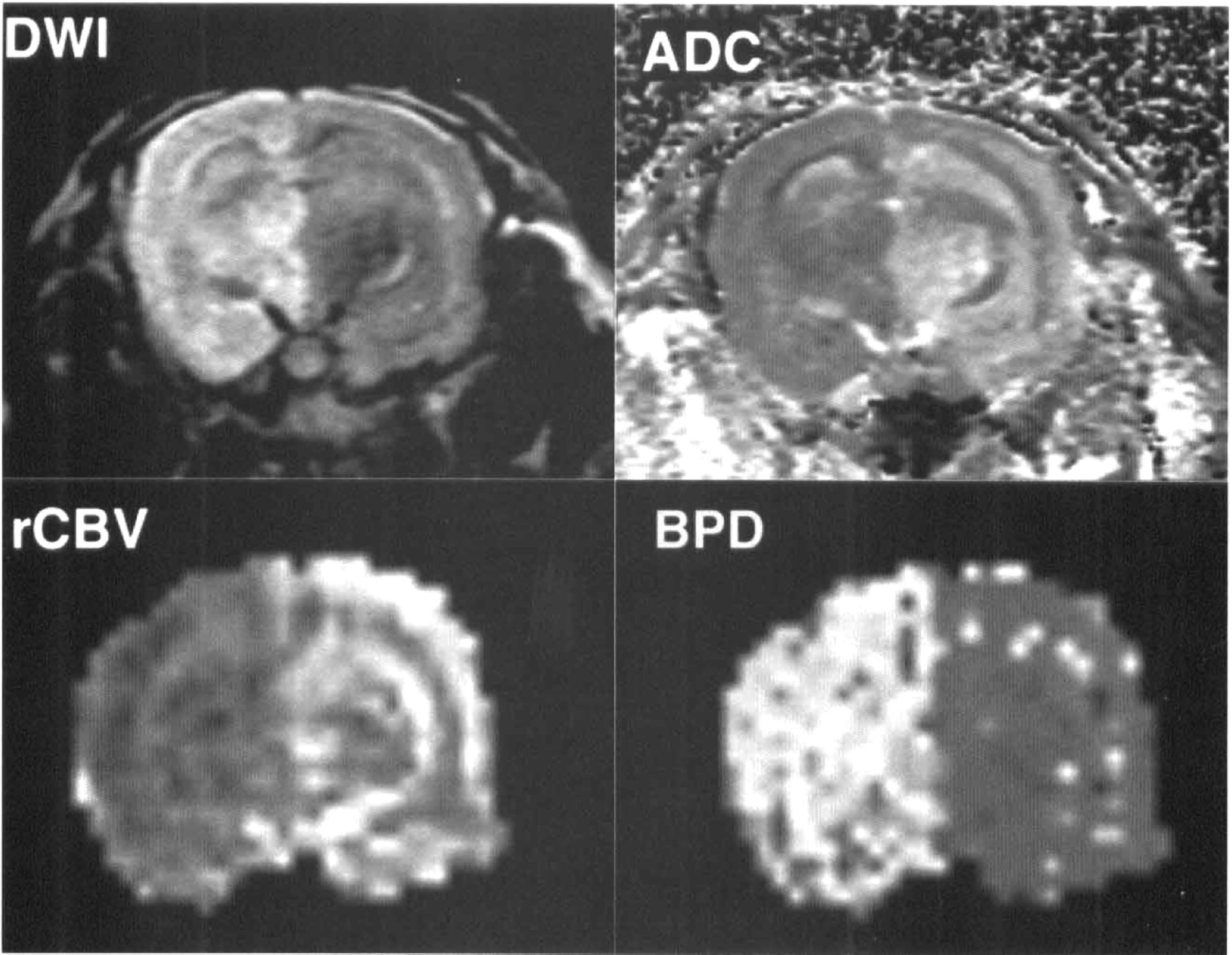
DWI and fitted maps of ADC, rCBV, and BPD. The left sides of the images show regions of decreased diffusion, low ADC, reduced rCBV, and delayed perfusion, respectively. See text for abbreviations.
Sample size calculations and statistical analysis
A sample size calculation was performed on pilot data (Yenari et al., 1994a). This analysis showed that to detect differences of 25% with a power of 80% and statistical significance at the p < 0.05 level, 10 animals per group were needed. Since 10–15% of animals do not have a successful embolization, 12 animals per group were studied.
Standard statistical methods were used to analyze infarct data from ADC maps, BPD and rCBV maps, and histopathology sections. The normalized values from ADC and rCBV maps were compared across the three different treatment groups using a one-way analysis of variance followed by a multiple comparison procedure. Kruskal–Wallis test followed by Wilcoxon rank sum test was used to analyze hemorrhage scores. Correlation of histology and 24-h ADC map data were analyzed using r > 0.7 for a positive correlation. Statistical significance was determined at the p < 0.05 level.
RESULTS
Of the 36 animals randomized in this study, 4 were excluded because of normal initial DWI and PWI scans. This represents a cerebral embolization frequency of 89% in this model. Another two animals were excluded from the efficacy analysis because of fatal systemic hemorrhage and hypotension. Therefore, 30 animals were analyzed for efficacy and cerebral hemorrhage and 32 animals were analyzed for systemic hemorrhage.
Physiologic parameters are summarized in Table 1. No significant differences were seen between treatment groups except for postembolization serum glucose levels that were slightly higher in the control animals. Following embolization, pH and Pa
Summary of physiological values a
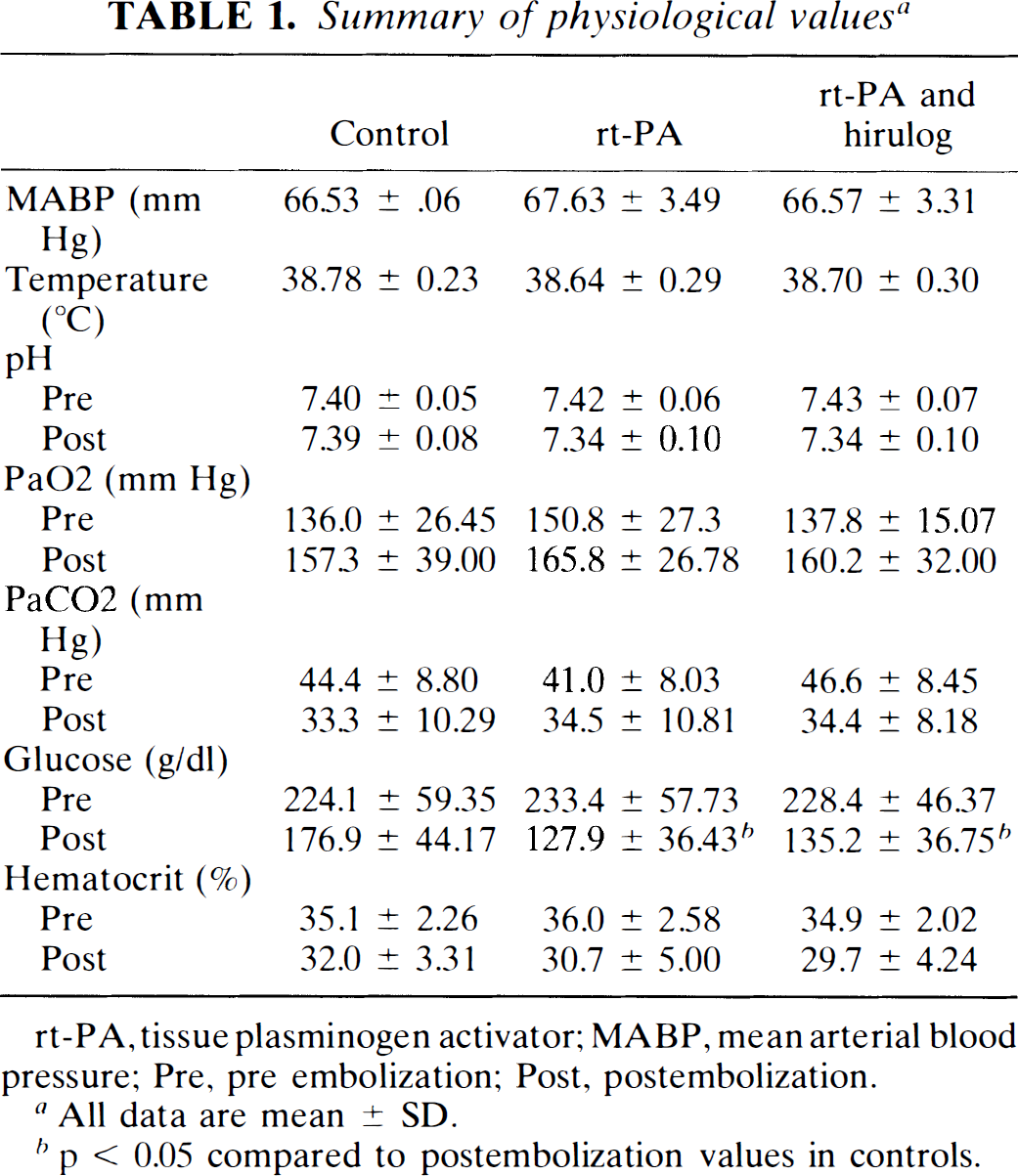
rt-PA, tissue plasminogen activator; MABP, mean arterial blood pressure; Pre, pre embolization; Post, postembolization.
All data are mean ± SD.
p < 0.05 compared to postembolization values in controls.
Coagulation studies
Hematological data are presented in Table 2. In the doses administered in this study, rt-PA decreased fibrinogen levels by 72.5% and prolonged aPTT by 2.4 times baseline. In animals given rt-PA and hirulog, aPTT was prolonged to about four times the baseline value. Since the fibrinogen assay is based on thrombin activation, fibrinogen levels were unmeasurable in these animals. Significant fibrinogen decreases and aPTT prolongation were not seen among control animals.
Summary of coagulation data a

rt-PA, tissue plasminogen activator; aPTT, activated partial thromboplastin time; N/A, not available.
All data are mean ± SD.
In the presence of an anti-thrombin, fibrinogen levels are unmeasurable.
PWI
Representative rCBV and BPD maps along with corresponding DWI and ADC maps are shown in Fig. 2. The rCBV maps lacked sufficient dynamic range for consistent analysis, particularly for smaller lesions. We therefore used the BPD maps for the perfusion analyses since we were able to obtain larger regions of delayed or absent flow that could be readily measured (Yenari et al., 1995a).
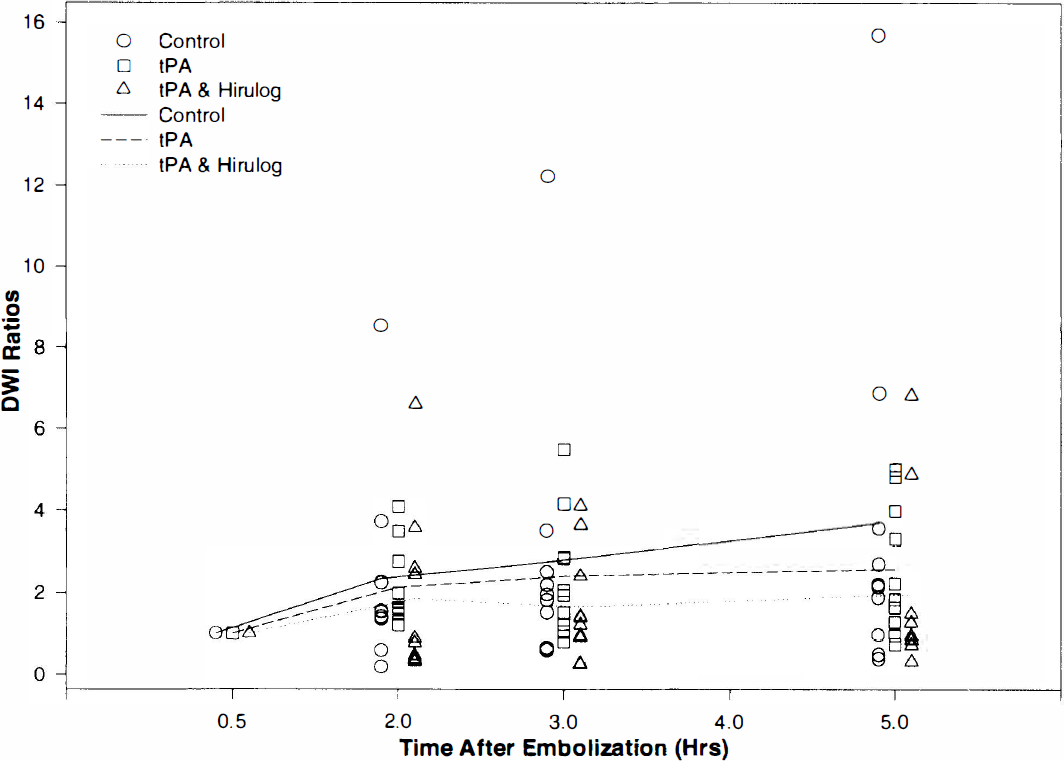
Two of the four sham controls demonstrated focal regions of delayed perfusion up to 2 s within the middle cerebral artery territory ispilateral to the site of surgery. There were no lesions on the corresponding rCBV maps, DWI, or T2-weighted images. Histopathology did not reveal any evidence of IND. All MRI studies and histopathology from the other two sham controls were completely normal. From the BPD maps, areas within the embolized hemisphere with delays of >2 s compared to the nonischemic hemisphere were measured (Yenari et al., 1995a). Area fractions of significantly delayed flow were measured from the BPD maps. Subsequent time points were measured in the same fashion, then normalized to the pretreatment values. Normalized ratios of significant perfusion delay areas from the BPD maps are shown in Fig. 3. Animals given combination treatment with rt-PA and hirulog demonstrated significantly reduced areas of perfusion delay by 5 h following embolization compared with controls. Ratios of the BPD maps were as follows: rt-PA plus hirulog, 0.40 (0.02–1.08); rt-PA, 1.12 (0.04–3.95); and control, 1.58 (0.47–3.59) (p < 0.05, hirulog plus rt-PA group compared with controls). Trends showing improved perfusion could be seen as early as 2 h with combination treatment. Ratios of BPD areas at 2 h were as follows: rt-PA plus hirulog, 0.40 (0.01–1.40); rt-PA, 0.77 (0.21–3.92); and control, 1.03 (0.45–1.47) (p = 0.07, hirulog plus rt-PA group compared with controls). In animals treated with only rt-PA, a slight improvement in perfusion was shown by the 2-h scans (p = 0.37 compared with controls), but these remained essentially the same as the baseline (0.5-h) scans by 3 and 5 h. Control animals showed progressive worsening of BPDs, particularly at the 3- and 5-h scans (Figs. 3 and 4).
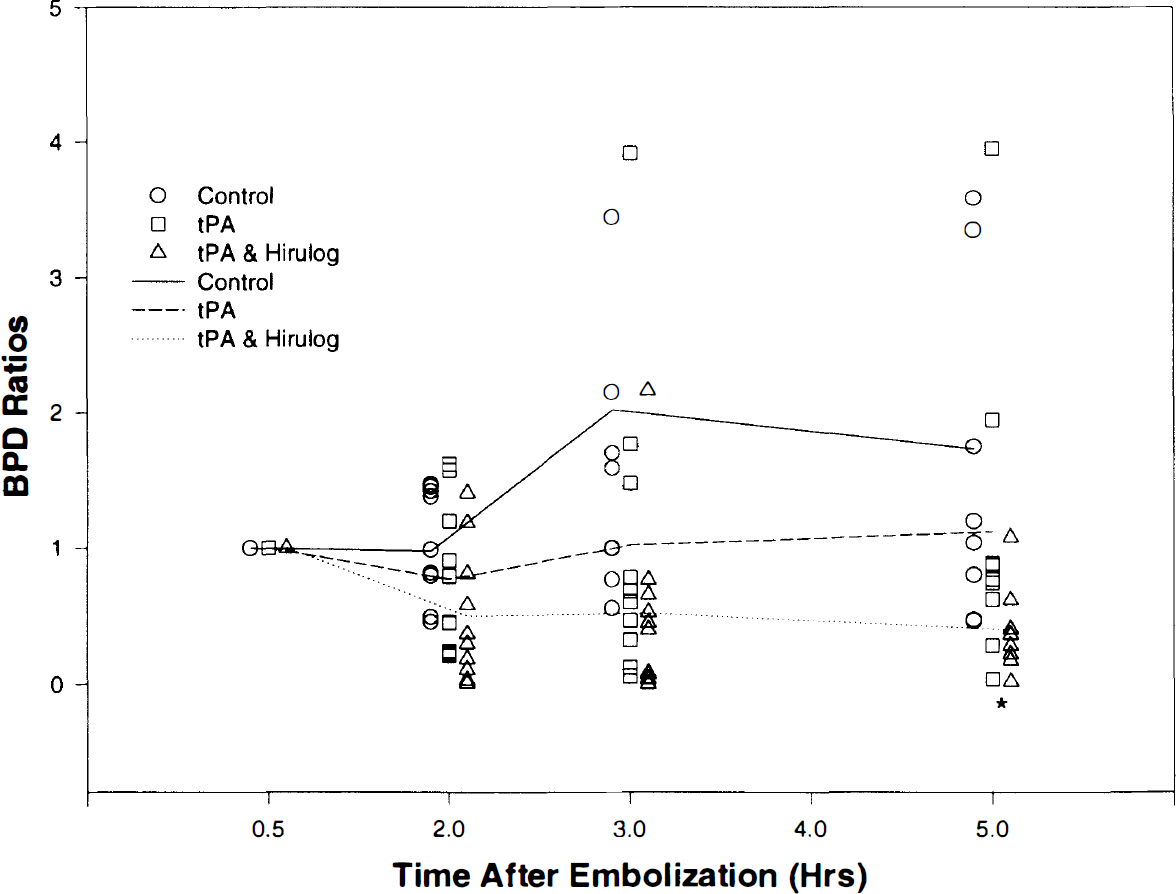
Normalized ratios of BPD areas show improved perfusion by 5 h following embolization in the group treated by rt-PA plus hirulog. *p < 0.05 compared with controls. See text for abbreviations.
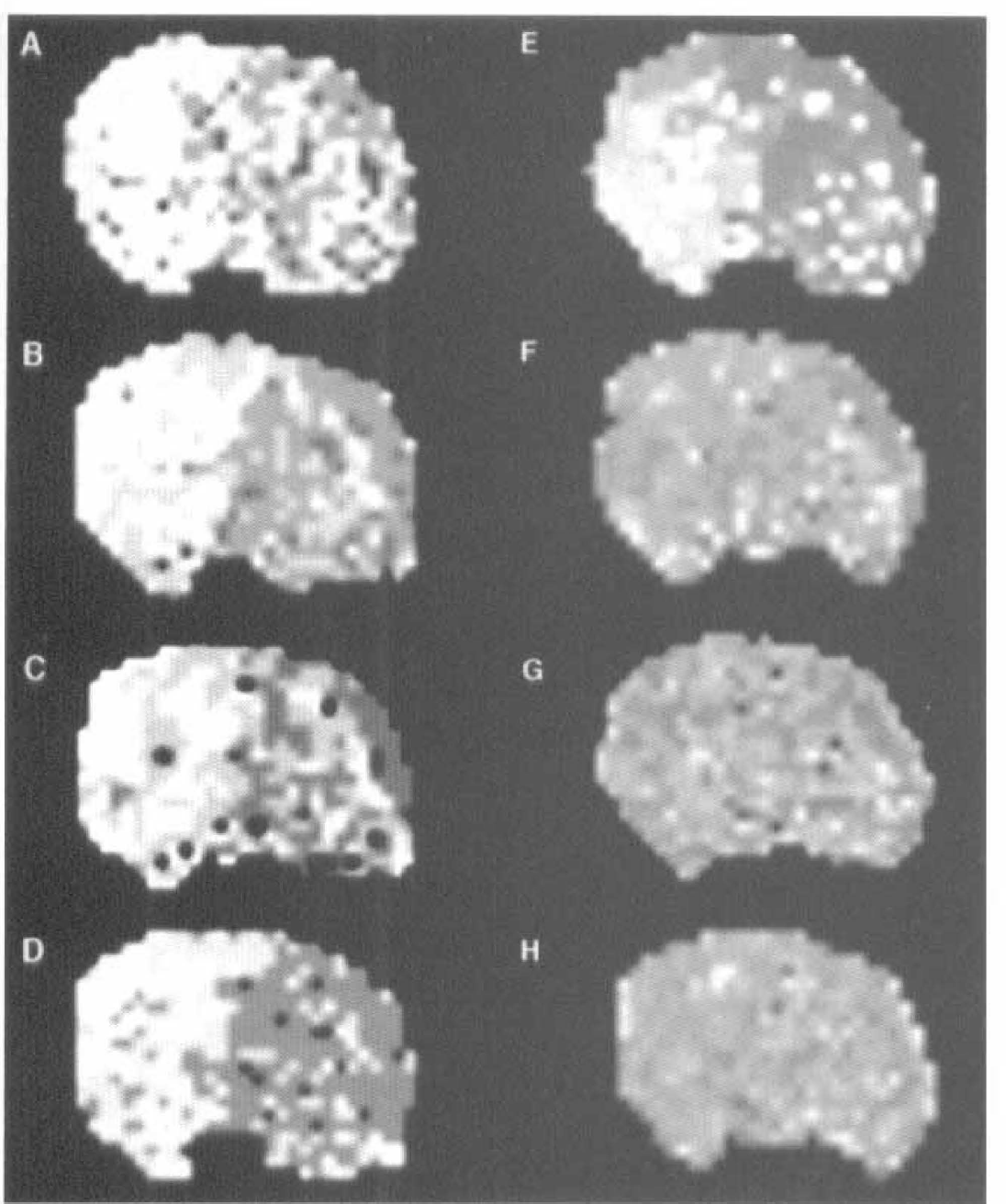
Representative BPD maps demonstrate improved perfusion with rt-PA plus hirulog treatment (
DWI
Normalized ratios of DWI lesion areas are shown in Fig. 5. No significant differences were seen between groups, although modest trends favoring combination treatment with rt-PA and hirulog were noted by the 5-h scans. Ratios of the DWI abnormality at 5 h were as follows: rt-PA plus hirulog, 1.95 (0.33–6.80); rt-PA, 2.57 (0.74–5.00); and control, 3.69 (0.39–15.71) (p = 0.32, rt-PA plus hirulog versus controls). Further inspection of our data revealed that individual animals in either treatment group with larger initial DWI lesions tended to worsen more than controls, while animals with small- and medium-sized initial lesions tended to improve.
Normalized ratios of DWI lesion areas at different time points show a trend toward improvement with rt-PA plus hirulog. See text for abbreviations.
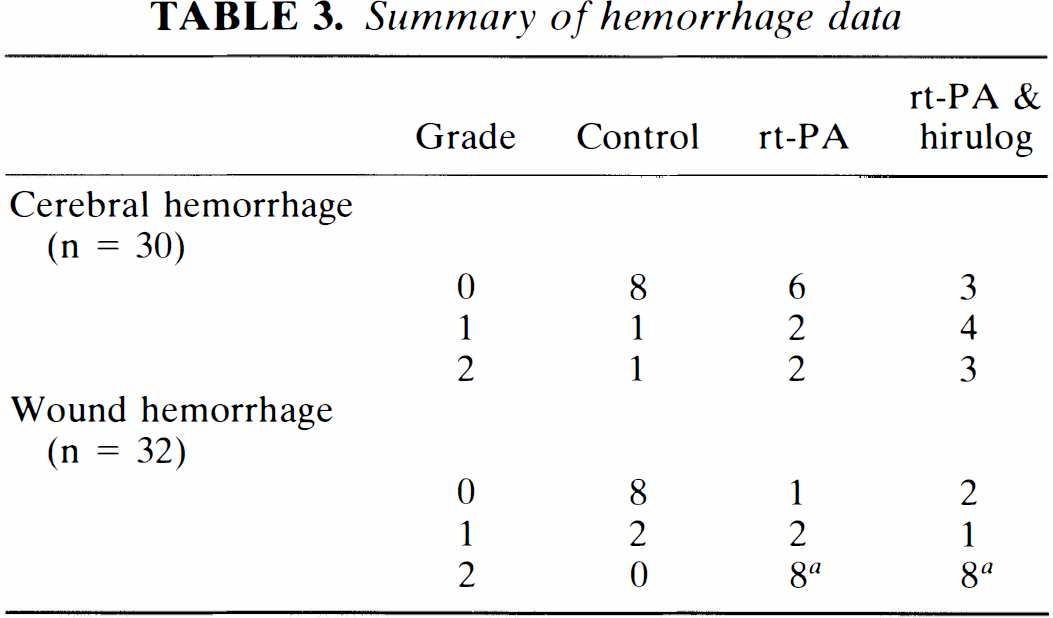
Hemorrhage
Hemorrhage data are summarized in Table 3. No differences were seen in significant (i.e., grade 2) cerebral hemorrhage. Three severe cerebral hemorrhages were seen in animals treated with rt-PA plus hirulog. Two severe cerebral hemorrhages were seen in animals given only rt-PA, and one control animal suffered a severe hemorrhage. Severe wound site hemorrhage (grade 2) was seen more often in both rt-PA-treated groups compared with controls (p < 0.001). The addition of hirulog did not increase severe wound site hemorrhage compared with rt-PA alone. Of the two animals excluded from the efficacy analysis because of fatal systemic and wound site hemorrhage, one animal was treated with rt-PA alone and the other was given rt-PA and hirulog.
Summary of hemorrhage data
Grading system: 0 = no hemorrhage; 1 = petechial brain hemorrhage, small amount of bleeding in surgical wound sites; 2 = large area of hemorrhage infarction or frank cerebral hematoma, large amount of blood in surgical wounds and/or sufficient blood loss to cause hypotension/death.
p < 0.0001 compared to controls.
Histopathology
Areas of increased signal intensity on the 5-h DWI scans correlated well with ischemic neuronal injury on histology (r = 0.84, p < 0.0001). Regions of IND were characterized by nuclear pyknosis and shrinkage along with glial pallor. Microscopic hemorrhage was frequently observed independent of the presence of gross hemorrhage. In addition, the presence of hemorrhage on the microscopic sections did not necessarily reflect the amount and extent of hemorrhage visible from the gross sections.
DISCUSSION
In this study, we show that treatment of experimental embolic stroke with rt-PA plus the direct-acting antithrombin hirulog is more effective than rt-PA alone or placebo in permitting the return of cerebral perfusion. The improvement in cerebral perfusion was seen as early as 2 h following embolization among some animals given rt-PA plus hirulog. This return of perfusion is not associated with increased cerebral hemorrhage, although wound site hemorrhage was higher in both groups given rt-PA. A trend toward reduction in ischemic lesion size was seen in treated groups, although this did not reach statistical significance.
In both experimental and clinical stroke studies, clot lysis and decreased neurologic injury have been shown following treatment with rt-PA. In animal stroke models, several groups have demonstrated statistically significant decreases in infarct size with rt-PA administration 30–120 min after ischemia (Zivinet al., 1985; Zivin et al., 1988; Chehrazi et al., 1989; Bednar et al., 1990; Overgaard et al., 1994). Clinical trials in stroke patients indicate that early thrombolytic therapy is promising in terms of both safety and efficacy (Haley et al., 1993; Hacke et al., 1995; NINDS rt-PA Stroke Study Group, 1995). While thrombolytic treatment of acute stroke appears promising, there are significant limitations. A major drawback is the increased risk of cerebral hemorrhage. Early rt-PA trials in myocardial infarction revealed significantly higher spontaneous cerebral hemorrhage rates in rt-PA-treated patients (Chaitman et al., 1989; TIMI, 1989). While the incidence of cerebral hemorrhage has not been shown to be increased with thrombolysis in various animal stroke models, translating these data to stroke patients is difficult because laboratory animals are generally younger and free of preexisting vascular disease (Papadopoulos et al., 1987; Chehrazi et al., 1989; Lydenet al., 1989; Benes et al., 1990; del Zoppo et al., 1990; Carter et al., 1992). Preliminary clinical trials in stroke patients showed that higher doses of thrombolytic agents were associated with increased hemorrhage rates with significant cerebral hemorrhage occurring with rt-PA doses of ≥0.9 mg/kg (Brott et al., 1992; Levy et al., 1994). In a recent National Institutes of Health study, clinically significant cerebral hemorrhage occurred in 6% of rt-PA-treated stroke patients compared with 0.6% of controls (NINDS rt-PA Stroke Study Group, 1995).
Given the dose limitations of rt-PA, incomplete clot lysis and distal embolization may further reduce its effectiveness. In doses felt to be “safe,” complete lysis of clots, particularly with larger and older clots, may not always be possible. Incomplete clot lysis may result in fragmentation of the parent clot, causing distal embolization and further injury. Aged and therefore more organized clots may be more resistant to successful thrombolysis (Kanamasa et al., 1989). rt-PA's short serum half-life requires that it be given as a continuous infusion to be efficacious, but this may be at the cost of hemorrhagic complications (TIMI, 1989; GUSTO, 1993). Thrombolysis can also activate thrombin (Owen et al., 1988), resulting in fibrin reaccretion, thus reducing the effectiveness of the therapy. To avoid some of these problems, adjunctive treatment with an anticoagulant or antiplatelet agent would seem reasonable as it may allow for more thorough and rapid thrombolysis. Anticoagulants and antiplatelet agents could also be maintained for longer periods of time after rt-PA use with less of the hemorrhage risk compared with maintaining the thrombolytic. In fact, the addition of heparin is frequently used in thrombolytic treatment of myocardial infarction, but adjunctive therapy with heparin or aspirin in stroke patients is less used for fear of increasing the cerebral hemorrhage risk. Both animal and clinical studies evaluating combined therapy with rt-PA plus heparin or aspirin have also not clearly established benefit from adjunctive treatment (Carter et al., 1992; Overgaard et al., 1992b; von Kummer and Hacke, 1992; Thomas et al., 1995). Furthermore, thrombolytic-induced thrombin activation does not appear to be influenced by heparin administration (Owen et al., 1988).
One of the limitations of heparin as an antithrombotic agent is its dependence on AT-III for activation. The relatively large size of the heparin/AT-III complex renders it sterically unable to inhibit clot-bound thrombin (Weitz et al., 1990; Kelly et al., 1991). Hirudin and hirulog bind to thrombin independently of AT-III and have been shown to be more effective than heparin in inhibiting platelet deposition and thrombus formation (Heras et al., 1990; Agnelli et al., 1991; Kelly et al., 1991). Because these agents are AT-III independent and therefore better able to inactivate clot-bound thrombin, they may be effective at lower levels of anticoagulation. In clinical trials of coronary artery disease, hirudin and hirulog either alone or in combination with a thrombolytic agent were both capable of improving clinical outcome compared with heparin (van den Bos et al., 1993; Topol et al., 1994; Cannon and Braunwald, 1995; Fuchs and Cannon, 1995; Theroux et al., 1995). Cerebral hemorrhage in a large clinical trial occurred more often with hirudin than heparin (GUSTO IIa, 1994); however, this may have been due to the inappropriate use of the aPTT in monitoring antithrombotic therapy. A subsequent study using lower levels of anticoagulation in patients with acute coronary artery syndromes demonstrated a small benefit of adjunctive therapy with hirudin over heparin (GUSTO IIb, 1996). With initial optimistic results in patients with acute myocardial infarction, it would therefore seem reasonable to investigate a similar strategy for acute stroke treatment.
To our knowledge, this is the first study that examined the effects of combined thrombolytic and direct antithrombin therapy in stroke. The improved perfusion observed in this study with combination rt-PA plus hirulog may be due to more complete and rapid fibrinolysis in the presence of the direct thrombin inhibitor. Direct inhibition of clot-bound thrombin may reduce fibrin accumulation within the embolus and improve clot lysis by decreasing the generation of endogenous thrombin and subsequent fibrin accretion. Although the role of intrinsic thrombin generation in stroke is not yet established, experimental and clinical studies have suggested that acute cerebral ischemia activates both the coagulation and the fibrinolytic systems with accretion of fibrin within the cerebral microvasculature (Fisher and Francis, 1990; Okada et al., 1994; Altès et al., 1995). In this setting, thrombolysis itself may also contribute to further thrombin activation (Owen et al., 1988). With the marked defibrinogenation observed in this study, the improvement in perfusion could also be due to a decrease in serum viscosity.
DWI and PWI were useful in monitoring the evolution of focal cerebral ischemia and response to therapy. The fitted BPD maps from the PWI scans were particularly useful in monitoring cerebral perfusion. Trends toward improved cerebral perfusion were detectable as early as 2 h after embolization following rt-PA plus hirulog treatment, providing we took into account insignificant delays caused by ECA ligation. Muller et al. (1995) arrived at similar conclusions using a rat intraluminal suture occlusive model of focal ischemia. Regions of abnormal perfusion corresponding to DWI abnormality were identified more readily with BPD maps, while the bolus peak effect maps sometimes appeared normal. The role of PWI parameters, particularly with respect to its relation to cerebral blood flow, should be further explored.
Using DWI, we found modest trends favoring a beneficial effect, particularly with combined treatment using rt-PA and hirulog. While DWI lesions increased in size over time, combination therapy appeared to inhibit this growth in some animals. The lack of significant reduction in DWI lesion size may be due to a variety of factors. Physiologic parameters such as MABP and temperature were similar between experimental groups; however, there were significant differences in postembolization serum glucose levels. Serum glucose levels in control animals were slightly higher (176 g/dl) than in the rt-PA (128 g/dl) and rt-PA plus hirulog (135 g/dl) groups, possibly resulting in slightly larger lesions in the control group. Individual animals in either treatment group with larger initial DWI lesions tended to worsen more than controls compared with animals with small- to medium-sized initial lesions, which tended to improve. The increase in lesion size in treated animals may be due to reperfusion injury or distal embolization. Observations in clinical trials of rt-PA support the notion that very large strokes tend to do worse with thrombolysis than do smaller ones (Hacke et al., 1995). It should also be noted that in our study we used clots aged for 24 h compared with other groups who used clots only 0.5–4 h old (Zivin et al., 1985; Chehrazi et al., 1989; Bednar et al., 1990; Overgaard et al., 1992a). Use of aged clots probably more accurately reflects clinical cardioembolic stroke (Moliniari, 1991), but this could have also made thrombolysis more difficult. Another possibility for explaining the lack of significant lesion size reduction in this study might be the delay in treatment. Groups that found reduction in infarct size typically began treatment 30 min following ischemia (Chehrazi et al., 1989; Bednar et al., 1990; Benes et al., 1990). Due to MRI constraints, we were unable to begin treatment earlier than 1 h following embolization. While others were able to find a beneficial effect of rt-PA in embolic stroke when treatment was delayed 1 or 2 h, this difference may be due to our model being a more severe one as we used aged rather than fresh clots (Overgaard et al., 1994) and maintained anesthesia longer (Carter et al., 1992). Furthermore, we assessed neurologic injury 6 h following embolization. Ischemic injury as well as improvement following thrombolysis may continue for a longer period of time. Different conclusions might have been reached had we assessed injury 24–72 h later. Since prolonging animal survival in this model is associated with an unacceptably high mortality rate, this was not feasible to study.
Both rt-PA and hirulog in maximally therapeutic doses did not cause excess cerebral hemorrhage, but peripheral hemorrhage was equally increased by both treatment strategies. The lack of a significant finding for this measure cannot exclude a type II error, since the sample size calculations prior to this study did not take into account the incidence of cerebral or peripheral hemorrhage. A retrospective sample size calculation on these data showed that 26 animals per groups would need to be studied to detect statistical differences for cerebral hemorrhage. A study of this size is not feasible using this MRI-based model, but the issue should be examined in future studies.
DWI and PWI were useful noninvasive methods of monitoring stroke evolution and treatment response. We found close correlation of DWI and histopathology; however, the relationship between PWI changes and cerebral blood flow is not precisely known. While DWI and PWI appear useful for monitoring thrombolytic therapy, their success in doing so has yet to be established. The results of this study should therefore be interpreted with this in mind. In the meantime, future studies should focus on the interpretation of these observations against established pathologic and physiologic standards. Clinical applications of DWI and PWI are beginning to show promise in the assessment and management of stroke patients (Warach et al., 1995). Imaging protocols similar to the ones used here are presently being incorporated in clinical stroke trials. The role of rt-PA in the treatment of human stroke has already begun to show promise, and the role of hirulog as an adjunctive treatment should be further explored.
Footnotes
Acknowledgment:
The authors thank Genentech (So. San Francisco, CA, U.S.A.) for the gift of rt-PA and excipient and Biogen (Cambridge, MA, U.S.A.) for the gift of hirulog.
