Abstract
Almost no data is available on the serial changes in the brain after spectacular shrinking deficit (SSD) that may help understand this relatively rare clinical phenomenon. Quantitative diffusion-(DWI), perfusion-(PWI), T1-(T1WI), T2-weighted (T2WI), and functional magnetic resonance imaging (fMRI) were performed before, during, and up to 7 days after embolic middle cerebral artery occlusion (eMCAO) in male Sprague—Dawley rats (n = 9). Region of interest (ROI) analysis was used to evaluate structural and functional MR signal changes within three ROIs defined by the apparent diffusion coefficient (ADC), cerebral blood flow (CBF) signatures, and final tissue viability. DWI, PWI, and T2WI lesion volumes were calculated using previously established viability thresholds and final infarct volumes ascertained with 2,3,5-triphenyltetrazolium chloride (TTC) staining. Serial MRI demonstrated spontaneous reperfusion of initially hypoperfused MCA regions accompanied by substantial reduction of initial ADC and CBF lesions and gradual recovery of neurological outcome. Recovery rates of CBF/ADC abnormalities differed among ROIs. Functional magnetic resonance imaging showed persistent tissue dysfunction after the recovery of the CBF/ADC lesions. This study may facilitate our understanding of the pathophysiological mechanisms by which early, spontaneous reperfusion affects tissue fate and neurological function.
Keywords
Introduction
Coined in the mid-1980s, the term ‘spectacular shrinking deficit’ (SSD) refers to an acute hemispheric ischemic stroke with rapid recovery of initially pronounced hemispheric symptoms (Kase, 1988; Minematsu et al, 1992; Mohr et al, 1986). The proposed mechanisms for the dramatic improvement are spontaneous clot lysis and/or clot migration, restoring blood flow to the compromised brain tissue, and leaving mild or no deficits (Baird et al, 1995; Minematsu et al, 1992; Mohr et al, 1986). The notion that reperfusion owing to clot migration is the most important factor leading to the improvement of symptoms is corroborated by previous studies showing that early recanalization (Baird et al, 1996) is associated with smaller ischemic lesion volumes and good clinical recovery in humans (Ringelstein et al, 1992). However, most data have been derived from patients who were treated with thrombolytics (Baird et al, 1996) and almost no data is available on the serial changes in the brain after spontaneous SSD that may help understand this relatively rare clinical phenomenon (Fujioka et al, 1999a; Kraemer et al, 2005).
In rats subjected to transient middle cerebral artery occlusion (MCAO), resolution of acute apparent diffusion coefficient (ADC) lesions may not always indicate tissue salvage from ischemia as selective neural necrosis was seen in such regions (Li et al, 2000). Additionally, depending on the duration of the occlusion, secondary abnormalities may develop on diffusion-weighted imaging (DWI) and T2-weighted imaging (T2WI) (Li et al, 2000; Neumann-Haefelin et al, 2000). These findings are intriguing as both transient and permanent resolution of such abnormalities was observed in SSD patients (Baird et al, 1995; Fujioka et al, 1999a; Kraemer et al, 2005). As it has been shown that severe dysfunctions in cerebral activity and neurobehavior can persist despite normal appearing brain on DWI, perfusion-weighted imaging (PWI), and T2WI (Sicard et al, 2006), further research is warranted to understand the mechanisms underlying SSD. However, most experimental data regarding transient MCAO were derived with suture MCAO models (Li et al, 2000; Neumann-Haefelin et al, 2000; Sicard et al, 2006), which do not reliably reproduce the inhomogeneous vascular findings in patients suffering from acute territorial stroke (Adams et al, 2005).
The major aims of this study were to assess the evolution of neurobehavioral deficits, ischemic injury, and brain activity on multimodal magnetic resonance imaging (MRI) acutely and chronically after embolic middle cerebral artery occlusion (MCAO) in Sprague—Dawley rats exhibiting early spontaneous reperfusion.
Materials and methods
Animal Preparation
Male Sprague—Dawley rats (n = 9) (Taconic Farms, Hudson, NY, USA) weighing 270 to 330g were anesthetized with isoflurane (5% for induction, 2% for surgery, 1.2% during imaging) in room air. Inspired hypercapnic challenges (2 mins) tested cerebrovascular reactivity and consisted of a premixed gas of 5% CO2 with 21% O2 and balance N2 (Sicard et al, 2003, 2006). PE-50 polyethylene tubing was inserted into the femoral artery for monitoring of mean arterial blood pressure and for obtaining blood gases (pH, PaO2, PaCO2) at baseline, 90 and 180 mins after MCAO. Body temperature was monitored continuously with a rectal probe and maintained at 37.0°C ± 0.5°C with a thermostatically controlled heating pad.
Thromboembolism and Animal Selection
Embolic MCAO (eMCAO) during remote common carotid artery occlusion (Busch et al, 1997) was produced by injecting one blood clot (diameter =0.35 mm and length = 24 mm) into the internal carotid artery over ~ 5 secs at the carotid bifurcation as described previously (Henninger et al, 2006). For this study, only animals were studied in which the middle cerebral artery spontaneously became patent shortly (within 15 mins after eMCAO) after embolus injection as indicated by a reduction of the initial (5 mins after eMCAO) cerebral blood flow (CBF)-derived lesion volume.
Magnetic Resonance Imaging Measurements
Magnetic resonance imaging experiments were performed on a 4.7T/40 cm horizontal magnet equipped with a Biospec Bruker console (Billerica, MA, USA), and a 20G/cm gradient insert (ID = 12 cm, 120 μs rise time). A surface coil (ID = 2.3 cm) was used for brain imaging and an actively decoupled neck coil for perfusion labeling (Sicard et al, 2006). Animals were imaged before and at 5, 15, 30, 60, 90, 120, 150, and 180 mins, as well as 1 and 7 days after eMCAO. A complete imaging sequence lasted 30 mins, recording three ADC maps separately acquired with diffusion-sensitive single shot, gradient-echo, echo planar imaging gradients applied along the x, y, or z direction; two T2WI acquired using a fast spin—echo pulse sequence; one fMRI scan (CO2 stimulation test) where CBF and the blood oxygenation level-dependent (BOLD) signal were simultaneously measured using continuous arterial spin labeling with single-shot, gradient-echo, echo planar imaging. All scans consisted of seven 1.5 mm-thick slices utilizing previously standardized imaging parameters (Sicard et al, 2006). Lastly, for magnetic resonance angiography, T1-weighted imaging (T1WI) was performed using a gradient-echo sequence with matrix = 128 × 128, TR (repetition time) = 90 ms, TE (echo time) = 3.3 ms, FOV (field of view) = 2.56 × 2.56 cm, nine 1 mm slices, and 16 averages. Basal CBF and ADC were the only imaging parameters recorded at 5 and 15 mins after MCAO owing to time limitations and T2WI was acquired at 180 mins, 1 and 7 days after eMCAO.
Neurological Evaluation and Infarct Volume Analysis
Bederson scoring of neurological function was performed before, 6, 24, and 168 h after eMCAO as described elsewhere (Sicard et al, 2006). After the last MR scan at 7 days after eMCAO, animals were electively killed under pentobarbital anesthesia. The brains were removed and sectioned coronally into seven 1.5 mm-thick slices corresponding to the MR slices and stained with 2,3,5-triphenyltetrazolium chloride (TTC) for post-mortem infarct volume calculation with edema correction (Henninger et al, 2006).
Calculation of In Vivo Lesion Size and Brain Swelling
Images were analyzed using Matlab (MathWorks Inc., Natick, MA, USA) and STIMULATE (University of Minnesota), and co-registered with in-house software (Sicard et al, 2006). Quantitative average ADC, CBF, and T2WI maps and their corresponding threshold-derived lesion volumes were calculated as described elsewhere (Henninger et al, 2006; Sicard et al, 2006). Briefly, viability thresholds were used to identify all pixels with abnormal ADC, CBF, or T2WI characteristics on each of the seven imaged slices at each time point. The corresponding ADC, CBF, and T2WI lesion volumes were then calculated by summing the abnormal area and multiplying by the slice thickness. The viability thresholds were 0.53 × 10−3mm2/sec for ADC, a reduction by 57% relative to the contralesional side for CBF, and values higher than + 2 standard deviations of corresponding contralesional tissue for T2WI (Henninger et al, 2006; Sicard et al, 2006). Hemispheric volumes derived by T2WI and TTC staining were used to calculate relative ipsihemispheric swelling at 180 mins, 24 h, and 168 h. To compensate for the effects of brain swelling, a corrected infarct volume on T2WI was calculated as described for the TTC lesion volume calculation. Cross-correlation analysis was performed on fMRI data to calculate activation maps for quantification of CBF and BOLD changes in response to hypercapnia (ΔBOLDCO2 and ΔCBFCO2) (Sicard et al, 2006).
Region-of-Interest Analysis
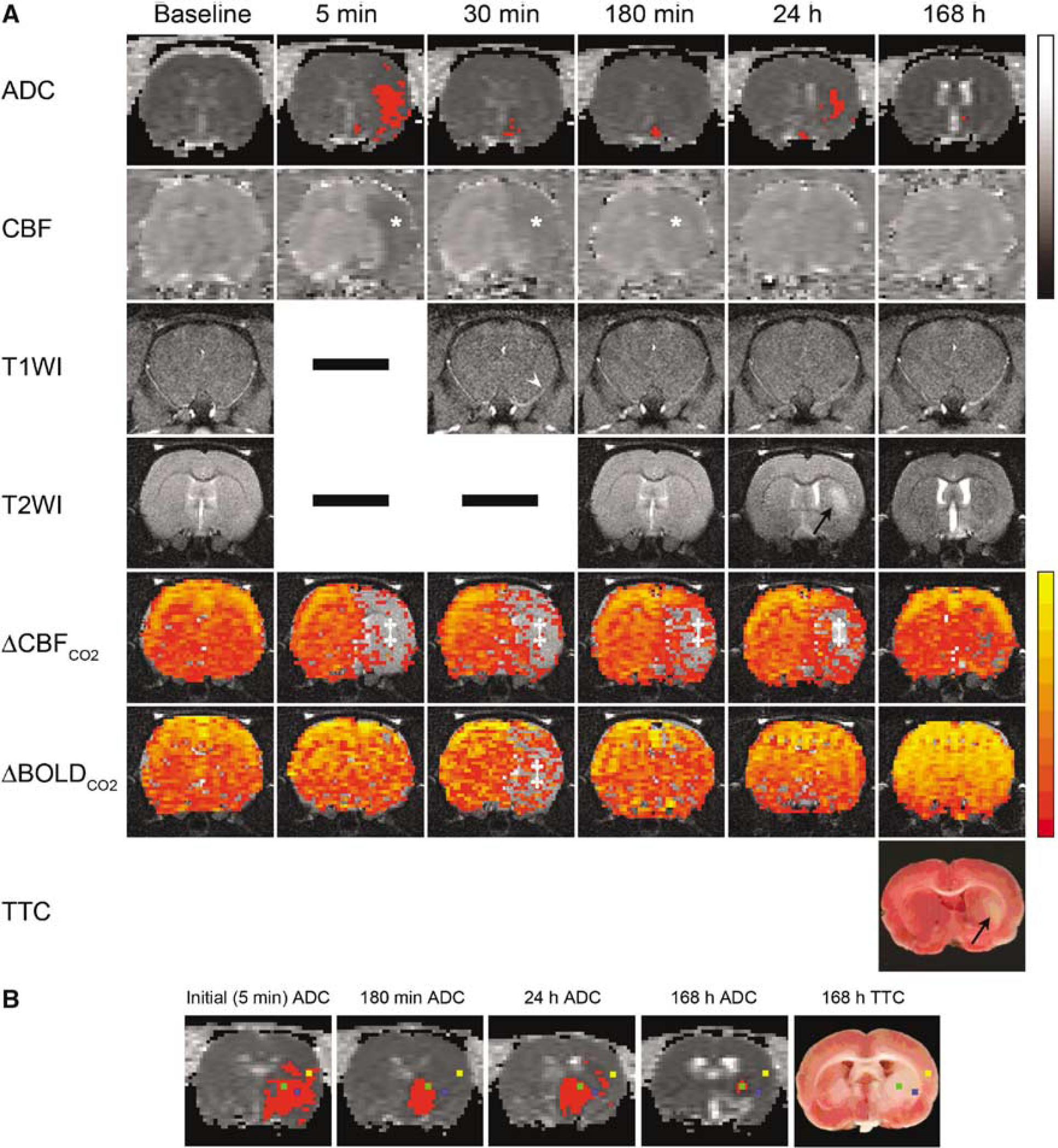
Figure 1B depicts the position of the defined region of interest (ROIs) on selected MR images from an eMCAO animal. Three ROIs (each 4 × 4 pixels) were manually defined as previously described in detail and describe three possible tissue compartments (Henninger et al, 2006): core defined by initially (5 mins) reduced ADC values below the critical threshold and a TTC abnormality (ROI1), sustained reversal of initially (5 mins) reduced ADC values and no TTC abnormality (ROI2), transient reversal of initially (5 mins) reduced ADC values and a TTC abnormality (note that the secondary ADC decline could appear at any time point after the initial recovery) (ROI3). Special care was taken to ensure that ROIs were placed in the same location across histological slices, images, and time points.
(
Statistical Analysis
Data are presented as mean ± s.d. unless otherwise stated. Statistical comparisons (Sigma-Stat 3.1, SPSS, Chicago, IL, USA) were performed using analysis of variance with post hoc Holm—Sidak or Dunn's test for multiple comparisons and two-tailed paired or unpaired Student's t-test, where appropriate. P < 0.05 was considered significant.
Results
Mortality and Data Exclusion
Two animals died prematurely (on days 2 and 6 after eMCAO) and no behavioral/imaging data could be ascertained for day 7. However, they were included in the data analysis as TTC staining was performed within 8 h after death (Henninger et al, 2006).
Physiological Measurements, Neurological Scoring, and Histology
Relative to baseline values (89 ± 9 mm Hg), there was an immediate increase in mean arterial blood pressure after eMCAO, which was significant from 75 mins (102 ± 7 mm Hg) to 165 mins (102 ± 7 mm Hg) after embolism (data not shown). Consistent with results from normal rats under similar experimental conditions, (Sicard et al, 2006) all other basal physiological parameters did not significantly differ between time points (data not shown). Initial neurological deficits improved significantly (P = 0.047) over time and the median (range) Bederson score was 3 (0 to 3) at 6 h, 1 (0 to 3) at 24 h, and 0 (0 to 1) at 168 h, respectively. TTC staining at 168 h after eMCAO revealed small infarcts in all animals corresponding to the ADC and T2WI-observed lesions in the caudatoputamen (n = 5) or hypothalamus (n = 2) without significant ipsihemispheric swelling (0.7 ± 1.1%), and/or hemorrhagic transformation. Prematurely deceased animals showed infarcts in the caudatoputamen on 24 h-T2WI and final TTC staining.
Diffusion, Perfusion, T1-Weighted Imaging, and T2-Weighted Imaging
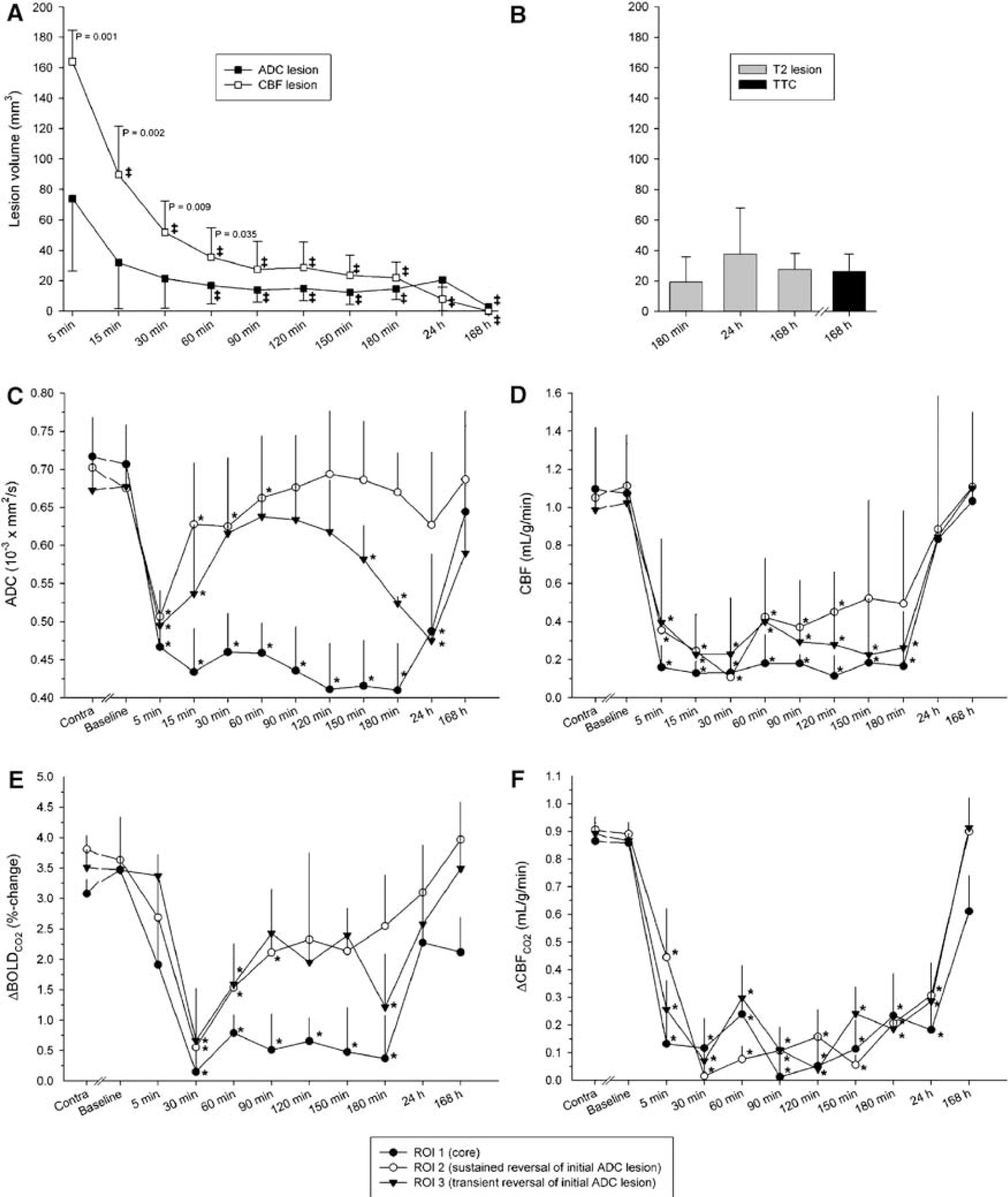
Table 1 provides a brief summary of the pattern of change for each ROI for each of the MR parameters examined as well as the findings on final TTC staining. Representative MR images at various time points before and after eMCAO are shown in Figure 1A. Importantly, T1WI showed an initial filling defect in the right MCA that recovered over time, indicative of spontaneous reperfusion. Furthermore, though initial CBF and ADC lesions (164 ± 21 and 74 ± 48 mm3 volumes, respectively) partially resolved over time (by 15 and 90 mins, respectively), most animals demonstrated secondary ADC lesions at later time points despite sustained reperfusion in these areas. T2-weighted imaging lesions in the ipsilesional hemisphere as early as 3 h after eMCAO corresponded to the 3 h-ADC lesions. In contrast to 180 mins (1.6 ± 2.0%) and 168 h (0.8 ± 1.2%), significant ipsihemispheric swelling was only observed at 24 h (3.6 ± 1.4%).
Summary of the pattern of change for each ROI for each of the MR-parameters
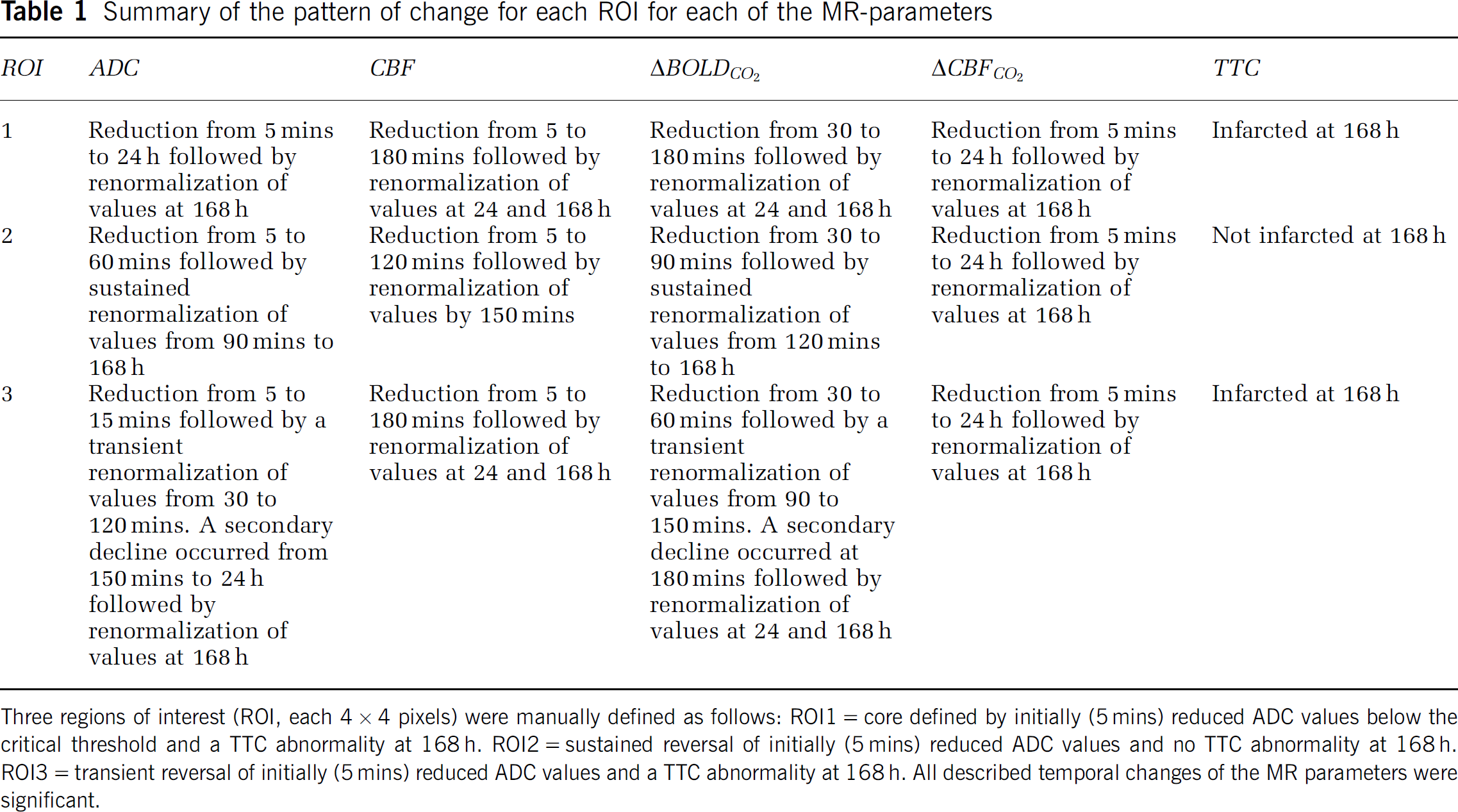
Three regions of interest (ROI, each 4 × 4 pixels) were manually defined as follows: ROI1 = core defined by initially (5 mins) reduced ADC values below the critical threshold and a TTC abnormality at 168 h. ROI2 = sustained reversal of initially (5 mins) reduced ADC values and no TTC abnormality at 168 h. ROI3 = transient reversal of initially (5 mins) reduced ADC values and a TTC abnormality at 168 h. All described temporal changes of the MR parameters were significant.
Figures 2A and 2B summarizes the spatiotemporal evolution of threshold-derived ADC, CBF, T2WI, and TTC lesion volumes. A significant mismatch was present up to 60 mins after eMCAO. CBF- and ADC-defined lesion volumes at 90 mins to 24 h did not differ from T2WI- or TTC-defined infarct volumes.
Spatiotemporal evolution of (
Quantitative Cerebral Blood Flow Values within Region of Interests
Region of interests characterized as types 1 and 2 were identifiable in all animals. ROI3 was present in seven eMCAO animals. Figure 2D shows CBF values within investigated ROIs. Cerebral blood flow did not differ significantly between contralesional ROIs over the course of the experiment (data not shown). Relative to corresponding contralesional values CBF was significantly reduced from 5 to 180 mins in ROIs 1 and 3, and from 5 to 120 mins in ROI2, respectively (P <0.05). In all ROIs, CBF was normal at 24 and 168 h.
Quantitative Apparent Diffusion Coefficient Values within Region of Interests
Figure 2C shows ADC values within investigated ROIs. ADC did not differ significantly between contralesional ROIs over the course of the experiment (data not shown). Relative to corresponding contralesional values, ADC was significantly reduced in all ROIs immediately after eMCAO. Renormalization occurred in ROIs 2 and 3 at 30 and 90 mins, respectively, with a significant secondary decline in ROI3 at 150 mins.
Functional Magnetic Resonance Imaging
Hypercapnia fMRI maps and temporal evolution of ΔBOLDCO2 and ΔCBFCO2 responses are shown in Figures 2E and 2F, respectively. Before occlusion, hypercapnic challenge produced robust increases in BOLD and CBF, which did not significantly differ between RoIs intra- or interhemispherically. At 30 mins after eMCAO, ΔBOLDCO2 was significantly decreased and thereafter demonstrated a differential recovery rate across localities with ROI3 renormalizing first (90 mins) and ROI1 recovering last (24 h). A transient secondary decline in ΔBOLDCO2 was observed at 180 mins in ROI3 with renormalized values by 24 h. ΔCBFCO2 was significantly reduced in ipsilesional ROIs at 5 mins and only recovered by 168 h after eMCAO.
Discussion
Little is known about the significance of sustained reperfusion in patients experiencing the so-called SSD. This can be partly explained by the paucity of experimental animal studies and the difficulty of obtaining early and serial imaging from patients. Of the few published imaging studies and case reports, only one examined the spatiotemporal evolution of DWI, PWI, and T2WI at acute to subacute time points. In that study, Kraemer et al (2005) found characteristic patterns in SSD patients, namely small initial DWI and large perfusion-weighted imaging lesion volumes that resolved acutely resulting in a small final infarct volume and sustained neurological improvement. However, all SSD patients received intravenous recombinant tissue plasminogen activator (rtPA) (Kraemer et al, 2005) and it remains to be proven whether this resolution is sustained and indicative of preserved brain function with spontaneous reperfusion.
The hallmark of SSD is arterial recanalization owing to clot lysis, fragmentation, and/or migration of clot fragments restoring blood flow and salvaging initially compromised brain tissue (Minematsu et al, 1992; Mohr et al, 1986). In patients, such clot migration or resolution of occlusion has been described by standard angiography as well as magnetic resonance angiography (Kraemer et al, 2005; Minematsu et al, 1992). Similarly, T1WI in this study demonstrated an initial filling defect in the MCA that recovered over time indicative of spontaneous arterial recanalization.
In contrast to large vessel reperfusion; however, tissue reperfusion may be heterogeneous and hypoperfused areas may persist beyond 24 h as demonstrated by single-photon electron computed tomography and PWI (Baird et al, 1995; Kraemer et al, 2005). Consistent with this observation, in the present study, long-lasting hypoperfusion was demonstrated in all investigated tissue compartments. Different, potentially interacting pathophysiological mechanisms have been postulated to underlie the incomplete reperfusion, including secondary hemorrhagic transformation, edema formation, microembolism, leukocyte plugging, or the noreflow phenomenon (Baird et al, 1995; Wang et al, 2001). The former two possibilities are remote given the lack of acute edema or signs of hemorrhage on MR, respectively. Nevertheless, the latter three remain viable possibilities and future studies are warranted to investigate them. Lastly, in our animals, we did not observe the previously described normoperfusion or hyperperfusion in the brain of SSD patients (Baird et al, 1995). However, because CBF was ascertained exclusive to ROIs defined by an initial ADC abnormality, the existence of such regions cannot be dismissed and future studies may be improved by more thoroughly investigating different tissue compartments.
Many SSD patients present with a large and potentially salvageable penumbra (Kraemer et al, 2005). This study confirms this finding and further demonstrates that parts of the initial ADC lesion may permanently reverse or secondarily deteriorate, confirming previous experimental (Li et al, 2000) and clinical data (Fujioka et al, 1999a), and corroborating the notion that early reperfusion is of utmost importance for sustained tissue salvage. Though early and sustained improvement in standard clinical outcome scores has been described in SSD patients, most of them received thrombolytic therapy, which makes it difficult to extrapolate these findings to nontreated individuals (Baird et al, 1995; Baumann et al, 2006; Kraemer et al, 2005). Therefore, a major question that remains unanswered is the functional significance of the described pattern of ischemic damage if left untreated as well as the short- and long-term prognosis. In this study and in accord with clinical data (Kase, 1988; Kraemer et al, 2005), neurobehavior significantly improved over time and animals had no or only very mild deficits at 168 h after eMCAO, potentially owing to brain plasticity. However, the employed test assesses general neurobehavioral deficits, and therefore, future studies could be improved by performing more specific behavioral tests to give better insight into sensorimotor dysfunctions (Sicard et al, 2006).
A different means to ascertain tissue function and how it relates to pathophysiology after both transient and permanent cerebral ischemia is supplied by fMRI (Dijkhuizen et al, 2001; Sicard et al, 2006). Most importantly our data demonstrated a differential recovery rate of ΔBOLDCO2 between investigated ROIs that mimicked the recovery rate of the respective ADC, suggesting a close association between regional ADC and stimulus-evoked changes in cerebral blood oxygenation. However, ΔCBFCO2 remained negligible in all investigated regions. Likely a result of (micro)vessel occlusions and subsequently abolished CO2 reactivity (Baird et al, 1995; Wang et al, 2001). Interestingly, ΔCBFCO2 remained abnormal at 24 h, demonstrating persistent tissue dysfunction beyond the recovery of CBF/ADC lesions as previously described (Sicard et al, 2006). Together, these results indicate acute hemodynamic uncoupling in the post-ischemic brain tissue despite rapid spontaneous reperfusion and lesion regression (Shen et al, 2005). Since the observed renormalization of cerebrovascular reactivity at 168 h after eMCAO may not necessarily reflect true tissue recovery (Sicard et al, 2006), future studies should incorporate sensitive histological and/or molecular analyses to investigate the presence of subtle neuropathological damage that may elude detection by conventional/functional imaging modalities and TTC staining.
As is the case with most animal studies, the results found herein are unlikely to be wholly transferable to the human condition. Nonetheless, our findings parallel those of clinical studies on patients with SSD in that large vessel reperfusion is followed by a substantial reduction of initial ADC and CBF lesions and recovery of neurological outcome. However, in contrast to the rapid and permanent betterment of symptoms and reportedly good prognosis in stroke patients with SSD (Minematsu et al, 1992), our data emphasize that spontaneous SSD may be followed by prolonged tissue dysfunction, histological abnormalities, and behavioral impairment that warrants development of treatment strategies beyond reducing risk factors. It is still unclear whether infarction occurs after human SSD and, if so, whether it has any effect on prognosis. Recently, it has been shown that incomplete infarction caused by SSD was detectable in humans by combined T1WI/T2WI after 1 week (Fujioka et al, 1999a) and similar findings have been reported in rats undergoing brief transient MCAO (Fujioka et al, 1999b; Li et al, 2000). Such neuroimaging signal changes may prove to be important diagnostic signs of incomplete infarction after transient cerebral ischemia such as the SSD phenomenon. The presence of incomplete infarction could then be correlated with functional imaging parameters as well as the incidence of neurological and cognitive disorders or subsequent stroke to determine if pathological changes in the brain owing to SSD increase the risk of after SSD morbidity and mortality rates. This seems to be of special interest given the contradicting significance attributed to brief cerebral ischemia. Whereas some studies found that transient cerebral ischemia may improve stroke outcome by serving as a preconditioning stimulus that triggers neuroprotective pathways in the brain (Castillo et al, 2003; Wegener et al, 2004), others demonstrated secondary deterioration on MRI (Fujioka et al, 1999a) or neurological function (Johnston et al, 2003; Smith et al, 2005) similar to the herein observed pathology. Hence, withholding treatment in these patients should be reconsidered and more aggressive interventions may be indicated in patients with rapidly improving symptoms, who currently are often treated with little urgency or concern (Johnston et al, 2003; Smith et al, 2005).
In conclusion, this study may provide clinicians with the important information that rapid resolution of ADC lesions after embolic MCAO with spontaneous reperfusion may not be permanent, arguing against the notion that SSD is a monophasic phenomenon (Baird et al, 1995; Kraemer et al, 2005; Minematsu et al, 1992) and for a more heterogeneous event with potentially delayed ischemic sequelae (Fujioka et al, 1999a). Secondly, fMRI is capable of identifying dysfunctional tissue after embolic stroke that eludes standard structural MRI techniques, and may be a sensitive and complementary parameter in the evaluation of stroke patients with rapidly improving symptoms. Lastly, in light of persisting neurological dysfunctions in some animals, there appears to be no significant association between the normalization of imaging modalities and recovery of neurobehavior after SSD. In conclusion, we present for the first time interesting and valuable information regarding tissue integrity and underlying vascular coupling after spontaneous reperfusion with SSD.
