Abstract
Associations have been reported between polymorphisms in the gene for α1-antichymotrypsin (ACT) and both Alzheimer's disease (AD) and cerebrovascular disease. An A-to-G substitution at nucleotide position 1,252 of ACT that produces a methionine to valine substitution at codon 389 has been found previously in four of 32 individuals with cerebrovascular disease from a Japanese population. We genotyped 194 individuals [59 controls, 35 with non–AD-type dementia (primarily vascular) and 100 with Alzheimer's-type dementia] for this polymorphism and found none that carry this polymorphism. Therefore, the allelic association of the A1252G mutation of ACT with cerebrovascular disease may be confined to the Japanese population and is not generalizable to other populations.
Apolipoprotein E (APOE), specifically the ε4 allele, has been implicated as a risk or susceptibility factor for late-onset Alzheimer's disease (AD). The strength of this association has been documented in a number of studies (Poirier et al., 1993; Saunders et al., 1993; Nalbantoglu et al., 1994 and references therein). Yet, only one-half of those with late onset AD carry an APOE ε4 allele.
α1-Antichymotrypsin (ACT) has been noted, like APOE, to occur in brain amyloid deposits of AD and affect amyloid fibril formation (Abraham et al., 1988; Ma et al., 1994). Kamboh et al. (1995) reported an allelic association between a polymorphism in ACT and AD. The presence of one form of this polymorphism increased the association of the APOE ε4 allele with AD. Others have not been able to confirm this association (Haines et al., 1996).
Tsuda et al. (1992) screened 95 individuals in a Japanese population, which included healthy subjects (n = 32), individuals with AD (n = 6), and individuals with “occlusive-cerebrovascular” disease (n = 32). They identified six individuals carrying an A-to-G substitution at nucleotide base 1,252 (A1252G), which produces a Met → Val substitution at codon 389. This mutation was called Isehara-1. Four of the six individuals with this mutation had “occlusive-cerebrovascular” disease; of the remaining two, one was healthy and the other had a history of tension headaches. This led the authors to hypothesize an association between this mutation and “occlusive-cerebrovascular” disease.
Recently, we completed a study that investigated the efficiency of genotyping to identify the presence of the various allelic forms of APOE for making the diagnosis of dementia both of the Alzheimer's and non-Alzheimer's (primarily vascular) types (Briones et al., unpublished data). In light of the reports of Kamboh et al. (1995) and Tsuda et al. (1992), we sought to determine if the A1252G mutation was associated, in a North American population, with dementia of either type. We were particularly interested to determine if this mutation occurred more frequently in demented subjects who did not carry the APOE ε4 allele. We report here that the A1252G mutation of ACT is not found either in a healthy population or a population with Alzheimer's or non-Alzheimer's (primarily vascular) type dementias in North America.
METHODS
Study population
This study was designed in the case-control format. Cases comprised individuals with long-standing dementia from the in-patient population of Hôpital Sainte-Anne de Bellevue, which is a chronic care hospital run by the Department of Veterans Affairs. The study followed hospital guidelines, and consent was obtained in all cases from the legal guardian of the patient.
The control population comprised volunteers recruited from a day center run by the hospital. These individuals were autonomous, generally well functioning, and living in the community.
There were no other exclusion or inclusion criteria for these populations.
Subject classification
Study patients were drawn from the in-patient population in our hospital and were well known to have a long-standing dementia. They were classified as to type of dementia (vascular, alcoholic, or Alzheimer's type) according to NINCDS-ADRDA and DSM IV criteria (American Psychiatric Association, 1994; McKhann et al., 1984). Case subjects were also evaluated using the Global Deterioration Scale; all study patients were in a “late dementia” phase, stages 6 and 7, of the Reisberg scale (Reisberg et al., 1982). Individuals in the control population scored “10” in the Mental Status questionnaire (Kahn et al., 1960). See Table 1 for subject characteristics.
Demographics and diagnoses
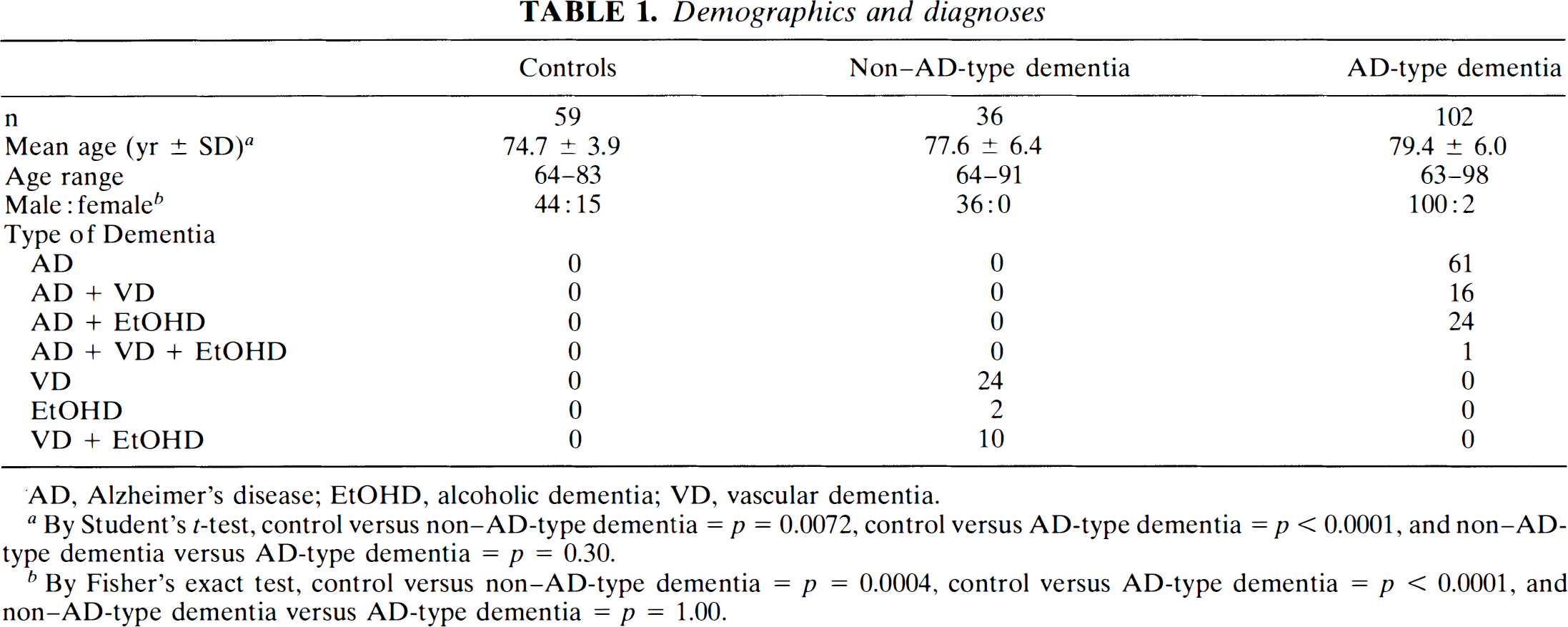
AD, Alzheimer's disease; EtOHD, alcoholic dementia; VD, vascular dementia.
By Student's t-test, control versus non–AD-type dementia = p = 0.0072, control versus AD-type dementia = p < 0.0001, and non–AD-type dementia versus AD-type dementia = p = 0.30.
By Fisher's exact test, control versus non–AD-type dementia = p = 0.0004, control versus AD-type dementia = p < 0.0001, and non–AD-type dementia versus AD-type dementia = p = 1.00.
Sample preparation
A single blood sample anticoagulated with EDTA was obtained from each subject. DNA was extracted using a commercially available DNA extraction kit (GNOME DNA isolation kit, BIO 101, Inc. La Jolla, CA, U.S.A.).
Genotyping
Genotyping for the A1252G mutation was performed using primers described previously (Tsuda et al., 1992). Reactions were carried out in a volume of 50 μl containing 1 μg of DNA; deoxyadenosine triphosphate, deoxycytidine triphosphate, deoxythymidine triphosphate, and deoxyguanosine triphosphate, each at 0.2 mmol/L; 12.5 pmol of primer 1 (5′ TTACTGAGAGCCCCACTGGATGAT 3′); 12.5 pmol of primer 2 (5′ CATAAGGCTGTGCTTGATGTA 3′); and 5 μl of 10 × polymerase chain reaction (PCR) reaction buffer (100 mmol/L Tris HCl, pH 9.0, 500 mmol/L KCl, 15 mmol/L MgCl2, 2 mg/mL gelatin, 1% Triton X100) (Vector Biosystems, Toronto, Ontario, Canada).
The DNA in the reaction mixture was first denatured for 10 min at 96°C and then cooled to 4°C One unit of Taq polymerase (Vector Biosystems) was then added to each sample. Each sample was reheated for 2 min at 96°C and then subjected to 30 cycles in a thermal cycler, with each cycle consisting of a 10-s denaturation at 96°C, 30-s annealing at 58°C, and 1-min extension at 65°C
To 10 μl of the amplicon was added 1.5 μl of 10 × Bsp H1 reaction buffer supplied by the manufacturer, 1 μl of Bsp HI (10 U) (New England Biolabs, Mississauga, Ontario, Canada), and 2.5 μl of water. The mixture was overlaid with mineral oil and incubated for 2 h at 37°C The digestion products were visualized by electrophoresis of 10 μl of the reaction mixture on a 1.5% agarose gel containing TPE buffer (0.08 mol/L Tris-phosphate, 0.002 mol/L EDTA) and ethidium bromide (0.15 μg/ml) for 1 h at 67 V. The gel was then photographed and the bands identified on the basis of molecular weight.
RESULTS AND DISCUSSION
We were able successfully to amplify DNA using primers for exon V of ACT in 194 out of 197 subjects (59 of 59 controls, 35 of 36 subjects with non–AD-type dementia and 100 of 102 with AD-type dementia) in our study to produce a 234-base pair (bp) product (Table 1). In the group with non–AD-type dementia, the DNA from one individual who had evidence of both alcoholic and vascular dementia failed to amplify. Upon treatment of the amplicon with the restriction enzyme Bsp H1, products of 170 and 64 bp were observed in every case. The A-to-G substitution at nucleotide base 1252 (Isehara-1 mutation) results in a loss of this restriction site, leaving the 234 bp product intact. Therefore, this mutation is absent from a North American population or occurs at a frequency of <1 in 194 individuals (388 alleles) or <1 in 35 individuals (70 alleles) if only those with non–AD-type dementia are considered.
In contrast, the A1252G mutation occurred in six of 95 Japanese subjects; four of these were found among the 32 subjects with “occlusive-cerebrovascular” disease (Tsuda et al., 1992). If one simply compares the distribution of individuals in this Japanese population carrying and not carrying the A1252G mutation (6:89) to the distribution in this present study (0:194), the difference is significant using Fisher's exact test (p < 0.05).
Examination of the genetics of ACT in relation to dementia has been sparked by the observation that ACT occurs in brain amyloid deposits of AD and affects amyloid fibril formation (Abraham et al., 1988; Ma et al., 1994). Absence of this mutation in a North American population does not necessarily exclude an involvement of ACT in dementia or in cerebrovascular disease; it implies, however, that the allelic association of the A1252G mutation of ACT with cerebrovascular disease may be confined to the Japanese population. If ACT has a functional role in dementia or cerebrovascular disease, then there is, likely, a mutation in Caucasians that has not yet been identified. This again emphasizes the point that the allelic association of any gene polymorphism with a disease may be the product of the genetic isolation of that population.
