Abstract
Ischemia-reperfusion brain injury initiates an inflammatory response involving the expression of adhesion molecules and cytokines, some of which are regulated by the nuclear transcription factor NF-κB. In this study the authors examined mRNA expression levels for several important genes associated with inflammation at five time points (3, 6, 12, 24, and 72 hours) after transient middle cerebral artery occlusion (MCAO) in Sprague-Dawley rats. A sensitive and quantitative technique (TaqMan real-time QRT-PCR) was used to simultaneously measure mRNA levels for key cell adhesion molecules and inflammatory cytokines. Gene expression increased significantly in the injured hemisphere for interleukin (IL)-1β (12-fold increase at 24 hours), IL-6 (25-fold increase at 6 hours) and ICAM-1 (4-fold increase at 24 hours), and the in-terhemispheric differences for these genes were significant for every time point examined (P < 0.05 for all values). Tumor necrosis factor-α mRNA was upregulated in the injured versus uninjured hemisphere from 3 to 24 hours (5-fold increase at 6 hours), while E-selectin showed a significant increase in mRNA levels from 6 to 24 hours after MCAO (10-fold increase at 6 hours) (P < 0.05 for all values). VCAM-1 mRNA levels did not respond differentially to injury at any time point between the two brain hemispheres. At all time points examined, activated NF-κB immunoreactivity was observed in cells throughout the infarct-damaged tissue. These results are consistent with the proinflammatory properties of the induced molecules, which are involved in the initiation of the inflammatory cascade, and may thus contribute to secondary cellular responses that lead to further brain damage.
Stroke is the third most common cause of death and the principal cause of postinjury disability in the United States and Europe. One third of patients who survive one year after a first stroke will be dependent on others for daily living activities (Schneider et al. 1999).
Ischemia-related neuronal injury following cerebrovascular insults (including stroke, cerebral hemorrhage, and head trauma) produces core infarcted tissue that is severely compromised, and may not be repairable. The surrounding tissue, the periinfarct region, may be subject to further damage through activation of secondary inflammatory and neurodegenerative cascades. Accumulating evidence suggests that inflammatory and immunological reactions are involved in the pathogenesis of cerebral ischemia following blood reperfusion to the surrounding tissue (Kochaneck and Hallenbeck, 1992; Kim, 1996; Phillips et al., 2000). The hallmark of the inflammatory response in the brain is leukocyte infiltration, which in the case of brain ischemia is initiated by neutrophils, and later is followed by macrophages (Feuerstein et al., 1994, 1998). While a direct cause and effect relationship between leukocyte infiltration and secondary neuronal damage in the periinfarct region following stroke has not been firmly established (Emerich et al. 2002), early migration of circulating leukocytes into damaged brain tissue has been documented. According to animal models of stroke, peripheral blood leukocytes migrate into the brain parenchyma within several hours after reperfusion (Barone et al., 1992). Neutrophil invasion is a blood-brain-barrier (BBB)-mediated event, in which the release of proinflammatory cytokines such as interleukin (IL)-1β and tumor necrosis factor-α (TNF-α) are the initiators. Both of these cytokines promote the expression of cellular adhesion molecules on endothelial cells and peripheral leukocytes (Stanimirovic and Satoh, 2000).
Activated vascular endothelial cells express a family of molecules termed selectins (del Zoppo et al., 2000; Okada et al., 1994), which bind to leukocyte cell adhesion molecules, and promote low-affinity leukocyte attachment and rolling (Zhang et al., 1996). This is a critical preliminary step toward firm leukocyte adhesion, which is mediated by the endothelial cell adhesion molecule E-selectin. Migration across the BBB then occurs by diapedesis of neutrophils into the extravascular space, where they may initiate pathogenic effects, possibly leading to microvascular plugging, stasis, and thrombosis (Bauer et al., 2001; Ley, 2001).
Ischemic injury to the brain activates the proinflammatory nuclear transcription factor NF-κB (Clemens et al., 1997). The induction of certain inflammatory genes is directly regulated by nuclear factor NF-κB, including the cytokines IL-1β, IL-6, TNF-α and adhesion molecules E-selectin, intercellular adhesion molecule-1 (ICAM-1), and vascular cell adhesion molecule-1 (VCAM-1) (Baeuerle and Henkel, 1994; Abraham, 2000). NF-κB is present in the cytoplasm as an inactive heterodimer (p50/p65) bound by an inhibitor, IκBα, which prevents the transcription factor from migration into the nucleus (Bauerle and Baltimore, 1988; Beg et al., 1992; Ganchi et al., 1992). Inflammatory signals induce phosphorylation of IκBα by specific kinases, followed by degradation through the ubiquitin-proteasome pathway. Activated NF-κB translocates into the nucleus where it regulates the transcription of target genes containing NF-κB consensus sequences in their promoter region. NF-κB activators include some of the cytokines whose genes are regulated by NF-κB itself (IL-1β, TNF-α), inducing a feed-forward activation and amplification of the inflammatory response.
The goal of this study was to better characterize molecular expression events associated with the inflammatory cascade that is initiated by ischemia-reperfusion brain injury, and to lay the groundwork for ongoing efforts to assess the efficacy of various drug treatments. In particular, the investigation focused on determining the simultaneous changes in expression of multiple genes associated with inflammation at several postinjury time points, and the correlation of these changes to the activation of NF-κB. A relatively new technique, real-time quantitative reverse transcription and polymerization chain reaction (real-time QRT-PCR), employing special fluorogenic probes, was used to simultaneously quantitate the levels of six different mRNA transcripts for important cell adhesion molecules and inflammatory cytokines known to be regulated by NF-κB. Because NF-κB activation is a posttranslational event, immunohistochemistry was used to detect activated NF-κB at each postinjury time point.
MATERIALS AND METHODS
Animals
Male Sprague-Dawley rats (270 to 330 g; Charles River Labs, Raleigh, VA, U.S.A.) were used in this study. The animals were maintained in a facility accredited by the American Association for the Accreditation of Laboratory Animal Care (AALAC). In conducting the research described in this report, the investigators adhered to the Guide for the Care and Use of Laboratory Animals, as promulgated by the Committee on the Care and safety of Laboratory Animals of the Institute of Laboratory Resources, National Research Council.
Middle cerebral artery occlusion injury
Middle cerebral artery occlusion (MCAO) was carried out as previously described (Tortella et al., 1999; Williams et al., 2000). Briefly, animals were anesthetized by 5% halothane and maintained at 2% halothane delivered in oxygen. In the present study the endovascular suture remained in place for 2 hours, and was then retracted to allow reperfusion of blood to the middle cerebral artery (MCA). Sham animals underwent the same surgical procedure but without the introduction of the filament into the MCA. Following surgery, animals were placed in recovery cages with ambient temperature maintained at 22°C. At the conclusion of the experiment, following sham or MCAO injury, the animals (n = 5 to 7 animals per group; total 35 animals) were anesthetized and euthanized by decapitation at 3, 6, 12, 24, and 72 hours after occlusion.
A neurologic examination was performed on each rat immediately prior to blood reperfusion at 2 hours after MCAO. Each animal was examined for reduced resistance to lateral push, open-field circling, and shoulder adduction or contralateral forelimb flexion when held by the tail (Williams et al., 2000). In the present study, all rats subjected to MCAO either exhibited all neurologic deficits at 2 hours after injury (immediately prior to reperfusion), or they were dropped from the study. Furthermore, if there was evidence of subarachnoid hemorrhage on extraction of brain tissue, the animal was excluded from the study.
Sample processing and infarct analysis
At the given end point, rats were euthanized and brain tissue immediately collected. A 3-mm coronal section was taken from the area perfused by the MCA, starting 5 mm from the frontal pole. Approximately 100 mg of tissue was collected from the ipsilateral (injured) and contralateral (uninjured) hemispheres and was processed for QRT-PCR. The remaining front and rear sections of each brain, which were not used for mRNA analysis, were stained using 2,3,5-triphenyl tetrazolium chloride (TTC) to verify the presence of injury.
RNA isolation, DNA digestion, and reverse transcription
The tissue samples were homogenized in TRIzol reagent (Life Technologies, Gaithersburg, MD, U.S.A.). Total RNA was extracted from the tissue according to the manufacturer's suggested protocol. Each sample was treated with DNase I (Gibco BRL, Rockville, MD, U.S.A.) to eliminate any possible DNA contamination and subsequently tested for presence of DNA using polymerase chain reaction (PCR) amplification without the reverse transcription (RT) step. Total RNA concentration was determined from spectrophotometric optical density measurement (260 and 280 nm). For each sample tested, the ratio between the spectrophotometric readings at 260 nm and 280 nm (OD260/OD280) was used to provide an estimate of the purity of the nucleic acid, and the ratio in all samples ranged between 1.7 and 2.0. Reverse transcriptase reactions were then carried out using the RNA PCR Core Kit (PE Applied Biosystems, Foster City, CA, U.S.A.). Each reaction tube contained 10 μg of total RNA in a volume of 150 μl containing 5 mmol/L MgCl2, 1 x PCR Buffer II, 500 μmol/L of each dNTP, 0.6 U/μl of RNase inhibitor, 2.5 U/μl of MuLV Reverse Transcriptase, 2.5 μmol/L of random hexamers and DEPC-treated water to volume. Reverse transcriptase reactions were carried out in a DNA Thermal Cycler 480 (Perkin Elmer, Branchburg, NJ, U.S.A.) at 42°C for 20 minutes and 99°C for 5 minutes. The cDNA was then stored at −20°C.
TaqMan real-time quantitative reverse transcriptase polymerase chain reaction
The cDNA sequences for rpL32, IL-1β, IL-6, TNF-α, E-selectin, VCAM-1, and ICAM-1 were obtained from GeneBank. PCR primers and TaqMan fluorogenic probes were designed from the corresponding cDNA sequences using the Primer Express 1.0 Software program (PE Applied Biosystems) and synthesis was performed by PE Applied Biosystems. The primer and probe sequences used are reported in Table 1. FAM (6-carboxyfluorescin) was used as the reporter dye, and TAMRA (6-carboxy-tetramethyl-rhodamine) as the quencher dye. Real-time PCR was performed using the TaqMan Universal PCR Master Mix, 100 nmol/L of primers and probes, and various concentrations of RT product: 68 ng of total RNA for rpL32, 102 ng for ICAM-1 and VCAM-1, and 306 ng for IL-1β, IL-6, TNF-α, and E-selectin. All samples were run in duplicate and the output level reported as the average of the two cells. The different loading amounts of cDNA were necessary for an appropriate alignment of the samples within the standard curve. Amplification conditions included 2 minutes at 50°C and 10 minutes at 95°C, and then run for 40 cycles at 95°C for 15 seconds and 60°C for 1 minute on the ABI PRISM 7700 sequence detection system (PE Applied Biosystems). Standard curves were constructed on a 1:2 serial dilution of DNA Template Reagent (PE Applied Biosystems)(Yao et al., 2002). The threshold cycle, which represents the PCR cycle at which an increase in reporter fluorescence above background is first detected, was determined by the software, based on the generated standard curves.
Sequences of primers and probes

The TaqMan probe consists of an oligonucleotide with a 5'-reporter dye, 6-carboxyfluorescine, and a 3'-quencher dye, 6-carboxy-tetramethyl-rhodamine. The primers and probes are shown for the sequences of rat rpL32 (X06483), IL-1β (M98820), TNF-α (S40199), IL-6 (E02522), E-selectin (L25527), VCAM-1 (M84488), ICAM-1 (NM012967) (Numbers in parentheses represent GeneBank designations).
IL, interleukin; TNF-α, tumor necrosis factor-α; E-selectin, endothelial leukocyte adhesion molecule; VCAM-1, vascular cell adhesion molecule-1; ICAM-1, intercellular adhesion molecule-1; rpL32, ribosomal protein L32.
Data analysis
The mRNA levels were normalized to the corresponding ribosomal protein rpL32 level for each sample to adjust for uncontrolled variability between samples (Li and Wang, 2000; Wang et al., 2000a,b). Using the formula provided by the manufacturer (PE Applied Biosystems) and described by Wang et al. (2000a,b), the values were extrapolated to calculate the relative number of mRNA copies as compared with rpL32 levels. The mRNA transcript for ribosomal protein rpL32 was chosen as a control (housekeeping gene) because it showed relatively small variations over time following cerebral ischemia (Wang et al., 2000a,b). The data are presented in the graphs as the mean ± SD. ANOVA followed by Tukey post hoc analysis was used to evaluate differences between time points. Student's t-tests were used to evaluate differences between left and right hemispheres.
Immunohistochemistry
To study NF-κB activation we used a monoclonal antibody (anti-NF-κB, p65 subunit: MAB3026; Chemicon International, Inc., Temecula, CA, U.S.A.) that specifically recognizes an epitope on the p65 subunit that is masked by bound IκBα Several studies have shown that this antibody exclusively detects activated NF-κB, because it recognizes p65 only in the absence of IκBα (Brand et al., 1996; Nonaka et al., 1999; Hickenbottom et al., 1999). Other studies have used similar antibodies to analyze the activation of NF-κB after forebrain ischemia (Stephenson et al., 2000).
Front and rear portions of each brain that were stained with TTC to monitor the size of the injury were postfixed for 48 hours or longer in 10% neutral buffered formalin, embedded in paraffin, and sectioned at a thickness of 10 μm. Tissue sections were mounted on pretreated slides and deparaffinized in xylene. The sections were incubated for 1 hour with 5% normal horse serum in phosphate-buffered saline (PBS) to block nonspecific antibody binding. The primary monoclonal anti-NF-κB antibody was diluted 1:150 in the solution used to block nonspecific binding. Tissue sections were covered with diluted primary antibody and incubated overnight in a humid chamber with gentle rotary agitation. Sections were washed three times in PBS, and the bound antibody was visualized by the avidin-biotin complex method (Vectastain Elite; Vector Labs, Burlingame, CA, U.S.A.) with peroxidase as the marker enzyme. Biotinylated secondary antibody and avidin-peroxidase solutions were incubated on the sections for 90 minutes each, with three washes of PBS after each step. The immunohistochemical signal was developed using a metal-enhanced diaminobenzidine substrate system (Pierce Chemical Company, Rockford, IL, U.S.A.). Hematoxylin-and-eosin (HE) staining was also performed on selected sections from each brain to assess the extent of the injuries and degree of leukocyte infiltration.
RESULTS
Using TTC staining we determined that each animal in the study had a focal ischemic injury similar to those previously described (Tortella et al., 1999; Williams et al., 2000). The evolution and severity of the injury was progressive, since the reperfusion time extended from 1 hour to 70 hours (i.e., 3 to 72 hours after injury; data not shown).
To calibrate the QRT-PCR system, standard curves from serial dilutions of the DNA template reagent (Perkin Elmer) were run for each plate. These also served as positive controls for primer and probe function, and encompassed the entire concentration range of the samples measured. The correlation coefficients for the standard curves ranged from 0.98 to 1.00 (data not shown).
A direct comparison between mRNA levels in sham-operated animals is displayed in Fig. 1. For these experiments, the same amount of total RNA (306 ng) was used for each sample. The gene for ribosomal protein rpL32 was expressed at very high basal levels in control rat brains (Fig. 1). In contrast, the other transcripts were expressed at very low basal levels. No significant differences were observed in gene expression between the two brain hemispheres for rpL32, IL-1β, TNF-α, IL-6, ICAM-1, VCAM-1, or E-selectin in control brains (Fig. 1). Because rpL32 mRNA levels changed between time points, but not between hemispheres at any given time point (data not shown), the transcript for rpl32 was chosen as the housekeeping gene, to which all other transcript signals were normalized. For the purposes of this study, the contralateral hemisphere of each brain acted as an internal control.
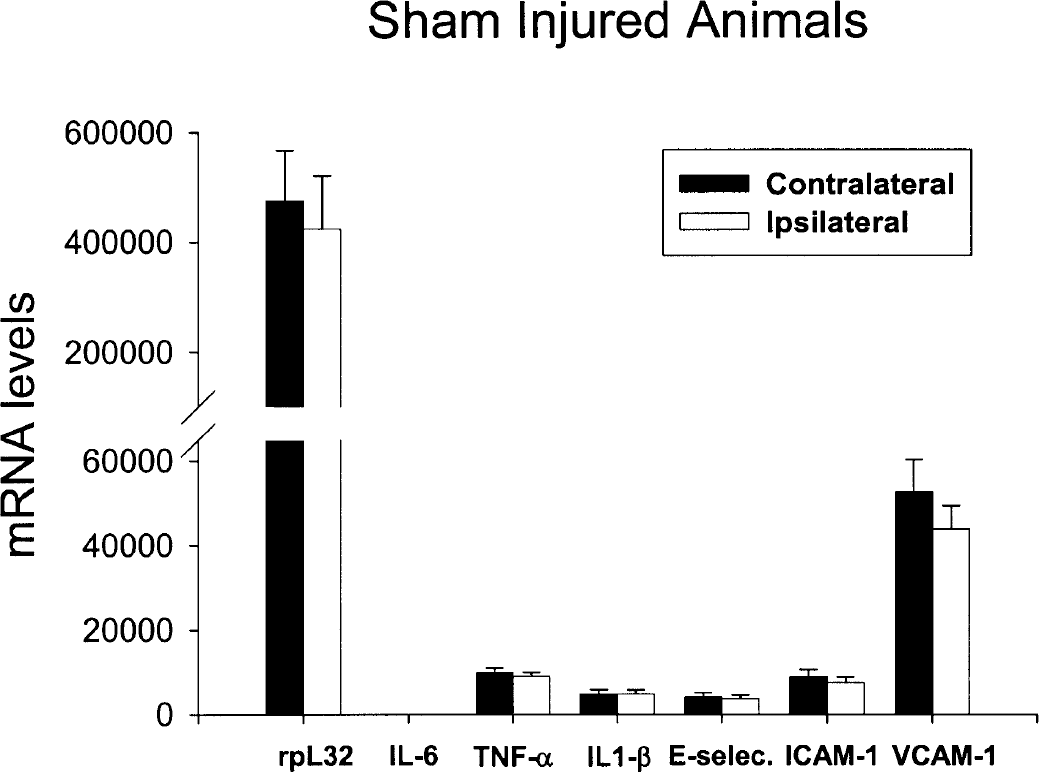
Absolute gene expression levels of the housekeeping gene, rpL32, inflammatory cytokines, and adhesion molecules in rat brain following sham surgery. The y-axis values are the fluorescent output signals (in arbitrary units) derived from the ABI PRISM 7700 sequence detection system. Basal IL-6 levels do not show up in the graph due to the low levels of expression in sham-operated animals (i.e., less than 0.026% of rpL32 levels). Open bars represent ipsilateral (injured) hemisphere; solid bars represent values from the contralateral (uninjured) hemisphere. IL-6, interleukin-6; TNF-α, tumor necrosis factor-α; IL-1β, interleukin-1β; E-selectin, endothelial leukocyte adhesion molecule; ICAM-1, intercellular adhesion molecule-1; VCAM-1, vascular cell adhesion molecule-1.
Figure 2 shows the differences in cytokine and adhesion molecule gene expression between the ipsilateral and the contralateral sides of the brain for all time points tested following MCAO injury. Y values represent rpL32 normalized mRNA levels, where, for example, 0.20 corresponds to 20% of the rpL32 transcript level in the same sample. The mean rpL32 mRNA levels between ipsilateral and contralateral hemispheres were found to be within 1 to 10% of each other at every time point studied (data not shown).
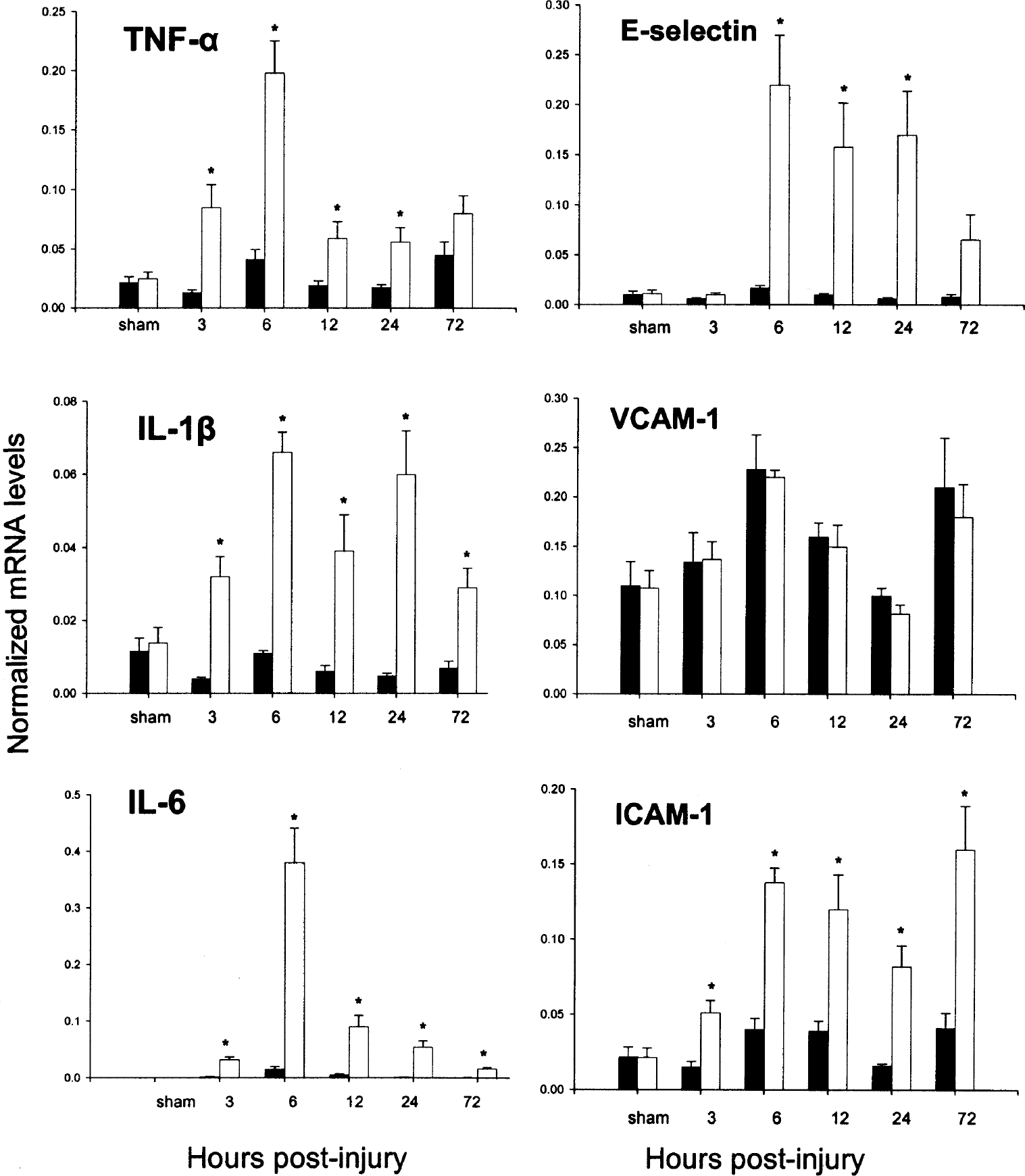
Time course of gene expression following middle cerebral artery occlusion surgery. Open bars represent ipsilateral (injured) hemisphere, solid bars represent values from the contralateral (uninjured) hemisphere. The mRNA levels are given as the mean ± SD normalized to rpL32 levels for each sample. The y-axis values represent the number of mRNA copies relative to the number of rpL32 copies in the same sample. *P < 0.05, paired t-test between ipsilateral and contralateral hemispheres. IL-6, interleukin-6; IL-1β, interleukin-1β; TNF-α, tumor necrosis factor-α; E-selectin, endothelial leukocyte adhesion molecule; VCAM-1, vascular cell adhesion molecule-1; ICAM-1, intercellular adhesion molecule-1.
Tumor necrosis factor-α
Comparing the injured with the uninjured hemisphere, TNF-α expression showed a statistically significant up-regulation on the ipsilateral side for 24 hours after MCAO. The peak level of TNF-α mRNA was observed at the 6-hour time point, where an approximately 5-fold greater amount of transcript was seen in the injured hemisphere (Fig. 2), which corresponded to 20% of the rpL32 levels. At the 72-hour time point, TNF-α mRNA levels were still elevated, but the difference in mRNA levels between the two hemispheres was no longer statistically significant.
Interleukin-1β
All postinjury time points exhibited significant IL-1β increase in the ipsilateral versus contralateral side of the brain (Fig. 2). The largest increases for IL-1β mRNA in the ipsilateral hemisphere were observed from 6 to 24 hours, with a difference of approximately 12-fold observed between hemispheres at the 24-hour time point. At their peak, IL-1β mRNA levels in the injured hemisphere only reached a little over 6% of the rpL32 levels in the same tissue samples.
Interleukin-6
IL-6 mRNA levels were significantly higher in the injured compared to the uninjured side of the brain at all time points tested (Fig. 2). Notable, however, was a dramatic difference observed at the 6-hour time point, where a greater than 25-fold difference was seen between the two hemispheres. At their peak, IL-6 mRNA levels reached over 35% of rpL32 transcript levels in the injured hemisphere. This was the highest normalized level of expression observed for any experimental gene in the study.
E-selectin
E-selectin showed a dramatic increase in the ipsilateral hemisphere at 6, 12 and 24 hours after MCAO compared with the contralateral hemisphere (Fig. 2). The peak difference between injured and uninjured hemispheres was observed at 6 hours after MCAO, where an approximately 10-fold greater level of E-selectin mRNA was measured on the ipsilateral side, corresponding to 20% of the rpL32 levels.
Vascular cell adhesion molecule-1
No differential upregulation for the adhesion molecule VCAM-1 was detected in the injured side of the brain versus the uninjured hemisphere at any time point examined (Fig. 2). It is clear from Fig. 4 that VCAM-1 levels did not respond to ischemic injury under the conditions of this investigation. In this study, the VCAM-1 transcript acted much like the housekeeping gene for rpL32, where no differences in expression levels were measured between brain hemispheres.
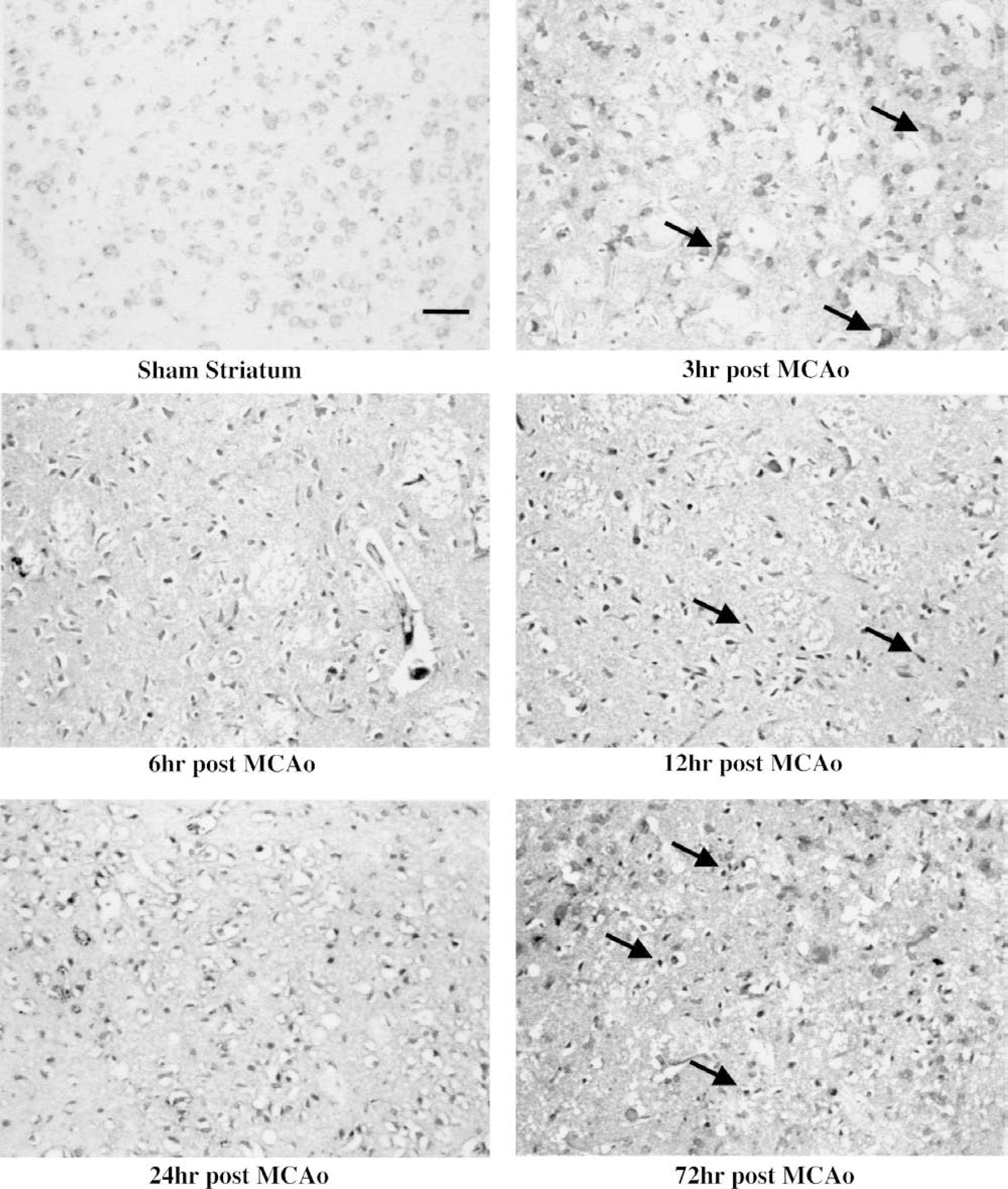
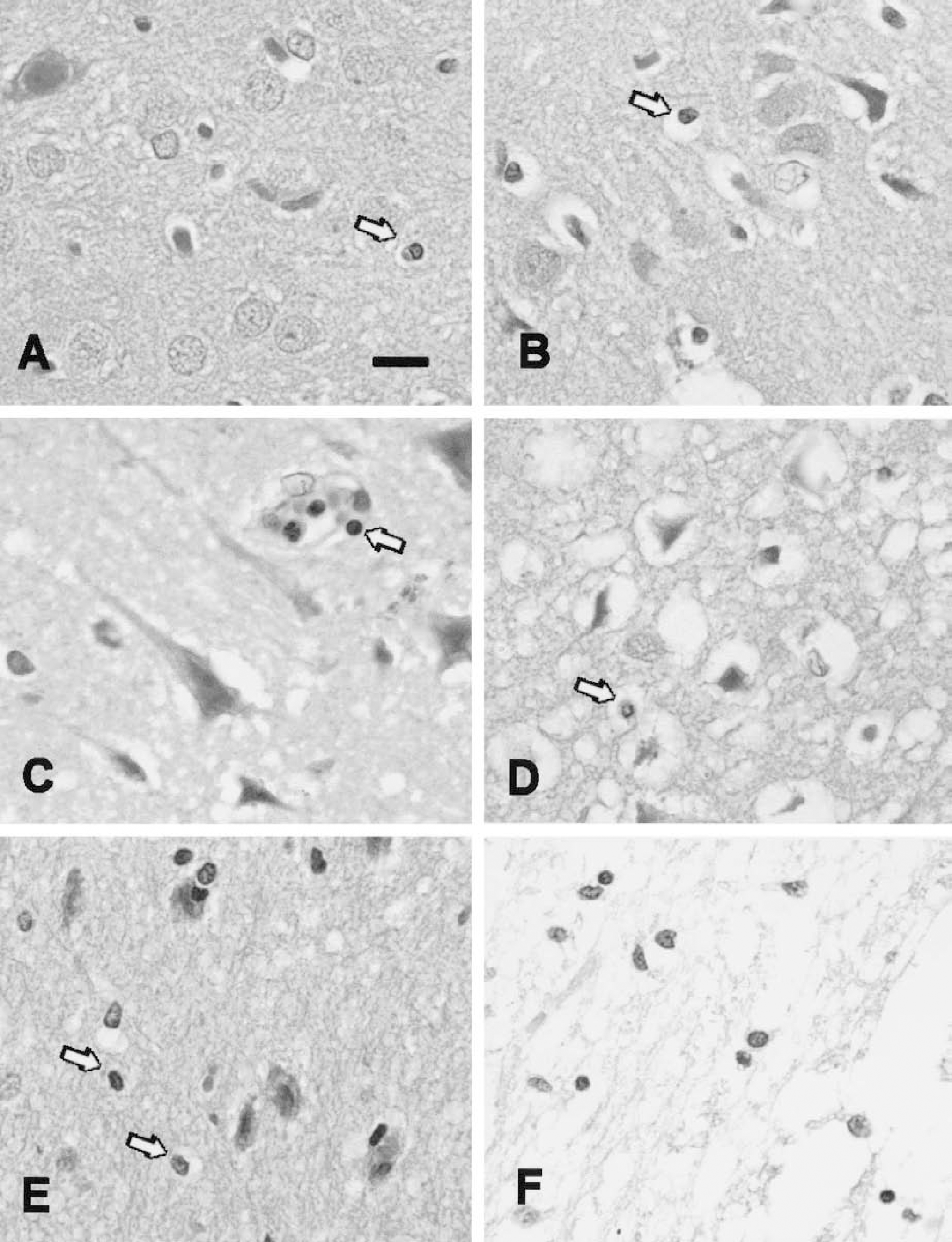
Immunoreactivity for activated nuclear factor (NF)-κB in the dorsal striatum of middle cerebral artery occlusion (MCAO)-injured rats at all time points. These micrographs demonstrate the changes in morphology and staining intensity of activated NF-κB-immunoreactive cells in the injured striatum at each of the time points studied after MCAO. Apparent neurons and astrocytes that were stained for activated NF-κB were observed in the striatum at 3 hours after injury (arrows). At 6 hours after injury, activated NF-κB immunoreactivity (aNF-κB-IR) was further increased, and cells with various morphologies were evident. Some of the immunoreactive cells were small (8 to 10 μm) at the 6-hour time point, and these were often associated with capillaries and small vessels. Twelve hours after MCAO, aNF-κB-IR intensity reached a plateau, and most of the stained cells were now smaller, and many were bar-shaped or fusiform (arrows). At the 24-hour time point, the majority of immunoreactive cells were generally round in appearance, and ranged in size from 8 to 15 μm (arrows). This staining pattern was still evident 72 hours after MCAO injury. Bar = 100 μm.
Intercellular adhesion molecule-1
Intercellular adhesion molecule-1 mRNA expression was upregulated in the injured versus the uninjured side of the brain at all time points examined after MCAO (Fig. 2). ICAM-1 levels were significantly elevated as early as 3 hours after MCAO. The largest difference in mRNA levels for ICAM-1 between hemispheres was seen at the 24-hour time point, where an approximately 4-fold greater level was found in the ipsilateral hemisphere. The absolute peak value was observed at 72 hours, where ICAM-1 mRNA levels reached over 15% of the rpL32 levels in the same samples.
Figure 3 shows a direct comparison of the changes in gene expression over time for all genes examined. Contralateral normalized mRNA values were subtracted from normalized ipsilateral values, and the difference for each gene at each time point was plotted. Thus, the contralateral, uninjured hemisphere served as an internal control for each brain. All genes except VCAM-1 showed significant changes following injury (P 0.001, ANOVA). Post hoc analysis of the data revealed a strong spike in the expression of transcripts for IL-6 and TNF-α, which were significantly and dramatically increased at 6 hours after injury, and then returned to normal or slightly elevated levels at later time points. Genes such as IL-1β and E-selectin showed a prolonged increased expression period, with significant and sustained levels out to at least 24 hours after injury. Finally, ICAM-1 expression revealed the longest activation time course, with significant induction as early as 3 hours, continuing out to 72 hours after injury. ICAM-1 mRNA did appear to drop temporarily at 24 hours, but statistical analysis revealed no significant differences between the 6-, 12-, 24-, or 72-hour time points (P > 0.05, Tukey post hoc analysis).
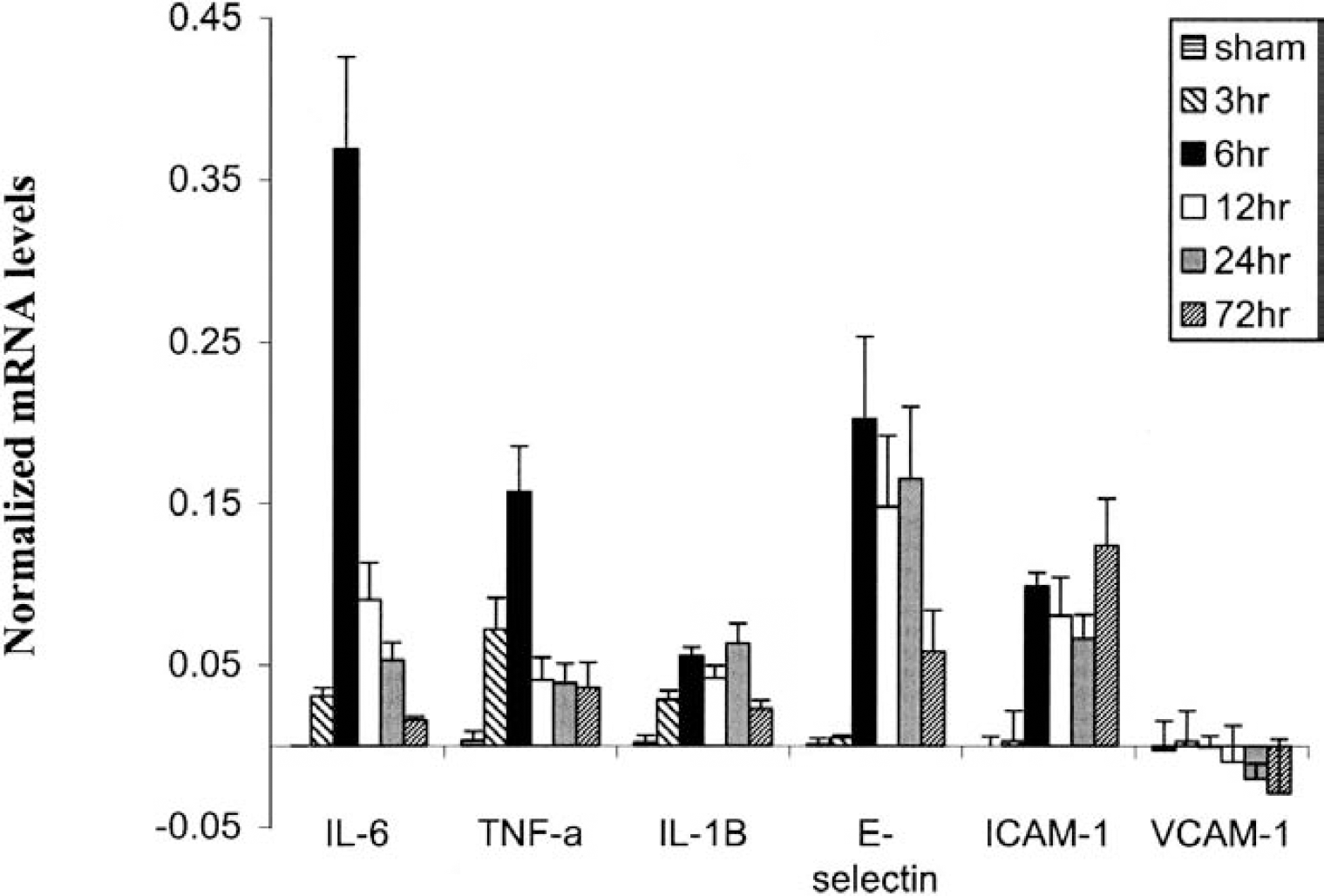
Time course of gene expression for all mRNA transcripts studied in the injured hemisphere following middle cerebral artery occlusion (MCAO) surgery. Time points include sham, and 3, 6, 12, 24, and 72 hours after MCAO injury. The 6-hour values are indicated by the black bars, while the 24-hour values are indicated by gray bars. The sham values are too small to be observable for several transcripts. For this graph, contralateral mRNA values were subtracted from ipsilateral values to show the level of expression above that seen in the uninjured hemisphere. The apparent reductions in E-selectin mRNA at 12 hours, and ICAM-1 at 24 hours were not significantly different from the other time points where expression of these transcripts was elevated. Because VCAM-1 levels were approximately the same in both hemispheres at all time points examined, the values tend to cancel out. IL-6, interleukin-6; TNF-α, tumor necrosis factor-α; IL-1β, interleukin-1β; E-selectin, endothelial leukocyte adhesion molecule; ICAM-1, intercellular adhesion molecule-1; VCAM-1, vascular cell adhesion molecule-1.
Nuclear factor-κB immunohistochemistry
Activated NF-κB immunoreactivity (aNF-κB-IR) was barely detectable by immunohistochemistry in the brains of sham-operated control animals (Fig. 4, sham). Three hours after MCAO injury, a moderate level of aNF-κB-IR was observed in cells throughout the infarct-damaged brain tissue (Fig. 4, 3 h). Some of these moderately immunoreactive cells were large and multipolar, suggesting that they were either neurons or astrocytes (Fig. 4, 3 h, arrows). Six hours after MCAO injury, further elevated staining for activated NF-κB was observed in cells throughout the infarct-damaged striatum (Fig. 4, 6 h) and cortex. The immunoreactive cells in the striatum and cortex were smaller 6 hours after injury than the ones observed at the 3-hour time point. Many were now in the size range of lymphocytes (8 to 10 μm). At 12, 24, and 72 hours after reperfusion, intense aNF-κB-IR was observed in cells throughout the infarct-damaged areas, including the striatum (Fig. 4), and somatosensory, motor, and gustatory cortices (data not shown). Elevated aNF-κB-IR was also seen in periinfarct areas at these time points, including secondary motor cortex, and insular and piriform cortices (data not shown). At 24 and 72 hours after injury, the strongly immunoreactive cells were small, and rod-shaped (Fig. 4, 24 h, arrows) or round (Fig. 4, 72 h, arrows), suggesting that cells with elevated levels of activated NF-κB were infiltrating leukocytes, and possibly activated microglia, rather than neurons or astrocytes.
Histology
Hematoxylin- and eosin-stained sections from sham and injured cortex were obtained at each time point after MCAO. Sham cortex is shown in (Fig. 5A), where all leukocytes were confined to the vasculature. Tissue from an animal 3 hours after onset of MCAO injury is shown in Fig. 5B; again, all leukocytes were observed within the brain vasculature. By 6 hours after MCAO, some leukocytes could be observed entering brain parenchyma (Fig. 5C). At the 12-hour time point, much more tissue damage was evident, and leukocytes were observed in the brain vasculature and parenchyma (Fig. 5D). Twenty-four hours after MCAO injury, leukocytes were numerous in the parenchyma (Fig. 5E). In the core of the damaged cortex, many phagocytes were observed in the parenchyma at the 72-hour time point (Fig. 5F).
Hematoxylin-and-eosin histology of leukocyte infiltration in middle cerebral artery occlusion (MCAO)-injured cortical brain tissue. Leukocytes were confined to the vasculature in sham animals (arrow in
DISCUSSION
This work represents the first systematic study of the simultaneous changes in gene expression for several proinflammatory cytokines and adhesion molecules at five time points following transient focal brain ischemia in the rat, using the TaqMan real-time QRT-PCR technique. Although many studies have focused on the induction of cytokine mRNA expression in ischemic brain injury, conventional techniques such as Northern blot analysis, (semi-) quantitative RT-PCR, and in situ hybridization are not able to detect small amounts of mRNA, and do not provide a robust quantitative measure of the amount of mRNA present in the sample. Since the TaqMan real-time QRT-PCR requires a very small amount of mRNA, it offers the advantage of evaluating alteration in the expression of multiple genes simultaneously from a small sample of brain tissue. As such, more genes can be compared in the same tissue sample, because less starting mRNA is needed for each analysis. In contrast with previous methods, QRT-PCR has the advantages not only of being more sensitive and reducing carryover contamination, but also yields rapid, accurate and simultaneous quantitation of multiple genes. The advantages and drawbacks of the TaqMan real-time QRT-PCR technique, and its application to the study of brain ischemia, have been discussed previously (Harrison et al., 2000; Medhurst et al., 2000).
Recent studies have reported on the efficacy of the TaqMan real-time QRT-PCR technique in determining differential gene expression in ischemic brain injury models (Li and Wang, 2000; Wang et al., 2000a,b). Unlike these previous studies, which investigated one or two genes, we took advantage of the increased throughput and sensitivity of the real-time QRT-PCR method by simultaneously analyzing the molecular expression of six different transcripts, all from the same tissue samples, at five time points ranging from 3 to 72 hours after injury. The ability to measure multiple transcripts in the same tissue samples permits direct comparisons of differential expression level changes at multiple time points, something that can not be done when trying to compare various gene expression changes which have been reported in different studies.
Because NF-κB promotes the transcription of mRNA for each of the inflammatory molecules in the present study, we also monitored NF-κB activation at various stages of the inflammatory response using a sensitive and specific immunohistochemical technique. NF-κB is a proinflammatory transcription factor that is associated with ischemia-reperfusion injury (Schneider et al., 1999). The activation of NF-κB is regulated at a posttranslational level, and, therefore, it cannot be monitored by detection of new mRNA or protein (Brand et al., 1996; Nonaka et al., 1999). To detect NF-κB activation we used a monoclonal antibody that exclusively recognizes p65 in the absence of IκBα. This antibody permitted the specific visualization of only those cells in which the inhibitory subunits of NF-κB were dissociated.
Nuclear factor-κB activation was observed in apparently different cell populations at specific time points in the study. At the 3-hour time point (1 hour after reperfusion), moderate levels of aNF-κB-IR were observed in many relatively large cells in the injured hemisphere. This time point correlated to significant increases in mRNA levels for IL-1β, TNF-α, and IL-6. ICAM-1 levels were also elevated significantly at this time point, indicative of early leukocyte adhesion and the onset of infiltration into brain parenchyma. Only scattered leukocytes were observed at this early time point in HE-stained sections. At the 6-hour time point (4 hours after onset of reperfusion), gene expression for the cytokines TNF-α and IL-6 reached peak levels. aNF-κB-IR was more intense at this time point, and was now observed in numerous smaller cells throughout the damaged areas of the ipsilateral hemisphere. Many more leukocytes were observed in and around the vasculature at the 6-hour time point (Fig. 5C). Based on their size, morphology, and the fact that many stained cells were closely associated with the brain vasculature, it is probable that most cells with aNF-κB-IR at this time point were leukocytes. This correlates well with the peak expression levels for TNF-α and IL-6, and the elevated levels of IL-1β mRNA observed at 6 hours after onset of MCAO injury. The initial increase in E-selectin mRNA expression was also observed at 6 hours after MCAO.
The expression of E-selectin protein is largely restricted to activated endothelial cells that express it on stimulation with IL-1β and TNF-α (Bevilacqua and Nelson, 1993). E-selectin mediates the initial low-affinity interaction between leukocytes and endothelial cells and promotes the recruitment and “rolling” of leukocytes along endothelial walls. We found a strong upregulation of E-selectin mRNA expression starting at 6 hours after MCAO (4 hours after reperfusion) in the injured hemisphere versus the uninjured hemisphere. The upregulation remained statistically significant as it waned during the 12 hour and 24 hour time points. This powerful upregulation of the transcript for E-selectin at 6 hours after MCAO is in concordance with the protein's priming mechanism in leukocyte recruitment, and correlated well with the first appearance of leukocytes with aNF-κB-IR in the injured brain regions. Histology with HE staining confirmed early leukocyte infiltration as early as 6 hours after MCAO injury.
Tumor necrosis factor-α and IL-1β may induce migration of neutrophils and macrophages into the brain as they do in other organ systems (Liu et al., 1994) by promoting gene expression of ICAM-1 and E-selectin (Zhai et al., 1997). While normal brain tissue has little TNF-α expression, its upregulation has been demonstrated in transient and prolonged cerebral ischemia (Liu et al., 1994; Wang et al., 1994). In the current study, TNF-α mRNA expression was significantly elevated as early as 1 hour after reperfusion, which is consistent with its role in the induction of expression of cellular adhesion molecules by glial and endothelial cells (Feurstein et al., 1994). It is interesting to note that aNF-κB-IR at this early time point was observed mainly in cells with the appearance of neurons and astrocytes, rather than leukocytes.
Interleukin-1β expression is increased during brain ischemia, as documented by the induction of IL-1β mRNA synthesis in rats with permanent MCAO (Buttini et al., 1994; Liu et al., 1993) or transient ischemia (Minami et al., 1992; Yoshimoto et al., 1997). Similar to TNF-α, IL-1β has also been shown to induce the promotion of neutrophil tissue infiltration and the induction of endothelial cell adhesion molecule expression (Pantoni et al., 1998). The upregulation of IL-1β mRNA expression in the current study was prolonged on the injured side, exhibiting significantly higher levels at all time points examined. This expression profile correlated well with the time points where strongly elevated aNF-κB-IR was observed in apparent leukocytes within ischemia-damaged brain tissue.
Upregulation of IL-6 mRNA after focal permanent ischemia in rats has been demonstrated (Wang et al., 1995a); however, conflicting data have been reported with regard to the functions of IL-6 production during both acute and chronic injuries to the brain (Ali et al., 2000). In our study, the expression of IL-6 mRNA in the injured hemisphere was significantly increased at all the time points tested compared with the uninjured hemisphere, with a dramatic peak in expression observed at 6 hours. It is still uncertain if this strong upregulation is indicative of IL-6 involvement in neuronal damage (Wang et al., 1995b) or in neural regeneration (Stroemer et al., 1995). The peak in IL-6 transcription correlated well with the peak in TNF-α mRNA activity, the initial increase in E-selectin mRNA upregulation, and with the shift in aNF-κB-IR from a population of larger cells in brain parenchyma to a population of smaller cells in and around the brain vasculature.
The adhesion molecule ICAM-1 is constitutively expressed in cultured astrocytes, but its expression is significantly increased when the cells are treated with IL-1β or TNF-α (Satoh et al., 1991). Increased ICAM-1 expression on endothelial cells or other cell types is a common characteristic of inflammatory and immune responses, as has been previously reported in permanent and temporary MCAO models (Pober and Cotran, 1990; Bevilacqua et al., 1994; Wang et al., 1994). From our data, a significant upregulation in ICAM-1 mRNA expression was evident from 3 to 72 hours after MCAO in the injured hemisphere versus the uninjured hemisphere. The increased transcription activity was prolonged, and no distinct peak of activity was apparent. A prolonged upregulation of ICAM-1 mRNA is consistent with the sustained increase in aNF-κB-IR observed in the ischemia damaged areas of the brain.
Vascular cell adhesion molecule-1 supports the adhesion of lymphocytes, monocytes, and eosinophils to the endothelial wall. The inflammatory response after brain ischemia is led primarily by neutrophils (Feuerstein, 1994), and it is therefore possible that VCAM-1 upregulation in the injured hemisphere is unnecessary for the early inflammatory response in the brain. Previous studies show an increase of VCAM-1 in an in vitro model of ischemia (Stanimirovic et al., 1997) and human brain microvessel endothelial cells stimulated by TNF-α or IL-1β (Wong and Dorovini-Zis, 1995). These are in vitro studies, however, and different techniques were used. Other studies have confirmed a lack of VCAM-1 induction has been reported following experimental brain percussion injury (Balabanov et al., 2001). Similarly, in our study, no differences in VCAM-1 mRNA levels were observed in the injured versus uninjured sides of the brain. It is possible that later in the inflammatory response (beyond 72 hours), when macrophage infiltration predominates (Phillips et al., 2000), an upregulation of VCAM-1 expression in the injured versus uninjured hemisphere might be observed. Another explanation for the lack of VCAM-1 upregulation is offered by Oh et al. (1998), where they demonstrated that IL-6 in the presence of the IL-6 receptor could inhibit TNF-α–induced VCAM-1 expression.
CONCLUSIONS
All mRNA transcripts in this study were significantly upregulated after MCAO injury except VCAM-1, although the specific cell types that these changes occurred in were not distinguished. Transcripts that exhibited sustained upregulation after reperfusion included the adhesion molecules ICAM-1 and E-selectin and the cytokine IL-1β. Cytokines that showed spiking profiles in transcript number included IL-6 and TNF-α, both of which peaked in activity at 6 hours after MCAO. Elevated aNF-κB-IR was first seen in larger cells with the appearance of neurons and astrocytes as early as 3 hours after onset of MCAO. By 6 hours after MCAO, and for the remainder of the study, aNF-κB-IR was shifted to a population of smaller cells in the injured brain regions. Finally, because mRNA for VCAM-1 did not respond to ischemia, this adhesion molecule probably has little or no role in the brain's response to ischemia-reperfusion injury for up to 72 hours after the onset of stroke.
A recent review of the literature (Emerich et al., 2002) suggests that no conclusive evidence exists that neutrophils play an important pathogenic role in the secondary neuronal damage that follows an ischemic injury; in fact, infiltrating leukocytes may only be variable bystanders to the injury process. Our results suggest that the several cytokines controlled by the transcription factor NF-κB, including IL-1β, TNF-α, and IL-6, and the adhesion molecules ICAM-1 and E-selectin, are involved in the response to ischemia-reperfusion injuries of the brain and that these processes occur prior to maturation of brain infarction. In particular, we measured an early, acute spike in the induction of the early inflammatory mediators TNF-α and IL-6, followed by a chronic upregulation of the cellular adhesion molecules ICAM-1 and E-selectin, as well as the cytokine IL-1β. The spike in TNF-α and IL-6 transcript number correlated with the first appearance of activated NF-κB immunoreactive cells with the appearance of leukocytes in the injured striatum and cortex. These findings are consistent with the proinflammatory properties of the induced molecules, the resultant diapedesis of circulating leukocytes, and, finally, secondary inflammatory responses leading to further brain damage. Using the high throughput capabilities of QT/PCR to monitor multiple gene responses simultaneously, a quantitative comparison of inflammatory gene responses can be made. This method provides a powerful tool for investigators studying brain ischemia and the response to treatment with antiinflammatory agents.
Footnotes
Acknowledgements
The authors thank A.A. Lewis and B.S. Cunningham for excellent technical assistance.
