Abstract
Estrogens have antiinflammatory actions and protect the brain from ischemic injury. Cerebral ischemia is accompanied by an inflammatory reaction that contributes to the tissue damage, an effect mediated in part by toxic amounts of nitric oxide (NO) produced by the inducible isoform of NO synthase (iNOS). Therefore, estrogens may protect the female brain by modulating postischemic iNOS expression. To test this hypothesis, we studied whether iNOS plays a role in the mechanisms of the reduced susceptibility to ischemic injury observed in female mice. The middle cerebral artery was occluded for 20 mins using an intraluminal filament in C57BI/6 mice, and infarct volume was assessed 3 days later in cresyl violet-stained sections. Infarcts were 53% smaller in female mice than in males (P < 0.05), a reduction abolished by ovariectomy (OVX) and reinstated by estrogen replacement. In normal female mice, postischemic iNOS mRNA was lower than in males (P < 0.05). Ovariectomy increased iNOS mRNA after ischemia and estrogen replacement blocked this effect. Furthermore, the iNOS inhibitor aminoguanidine reduced infarct volume in male, but not in female, mice. Similarly, male iNOS-null mice had smaller infarcts than wild-type mice, but female iNOS nulls were not protected. Ovariectomy and OVX with estrogen replacement did not affect infarct volume in iNOS-null female mice. The findings suggest that the neuroprotection conferred by estrogens is, in part, related to attenuation of iNOS expression. Such attenuation could result from the potent antiinflammatory effects of estrogens that downregulate iNOS expression via transcriptional or posttranscriptional mechanisms.
Introduction
There is considerable evidence that stroke is a sexually dimorphic disease. Thus, premenopausal women are relatively protected from cerebrovascular diseases, including ischemic stroke (Sudlow and Warlow, 1997). However, during menopause, the incidence of ischemic stroke and other cardiovascular diseases increases (Eaker et al, 1993). These epidemiological findings are supported by experimental studies in rodents, indicating that females are more resistant to cerebral ischemic injury (McCullough and Hurn, 2003; Wise et al, 2001). Thus, the brain damage produced by focal or global cerebral ischemia is less marked in females than in male rodents (Alkayed et al, 1998; Carswell et al, 2004; Hall et al, 1991; Jover et al, 2002; Simpkins et al, 1997). Such protection in females is abolished by ovariectomy (OVX) and reinstated by estrogen replacement, suggesting that estrogens are responsible for the protective effect (Dubal et al, 1998; Rau et al, 2003; Rusa et al, 1999; Yang et al, 2000).
The mechanisms by which estrogens protect from ischemic brain injury include both vascular effects leading to better preservation of cerebral blood flow (CBF) during ischemia (Alkayed et al, 1998; McCullough et al, 2001), and cytoprotective effects resulting in a greater tolerance of the brain to ischemia (Alkayed et al, 2001; Simpkins et al, 2005; Wang et al, 1999). Estrogens also have remarkable antiinflammatory properties, the mechanisms of which have not been elucidated (Baker et al, 2004; Bruce-Keller et al, 2000; Santizo et al, 2000; Vegeto et al, 2001, 2003).
Postischemic expression of the immunological or ‘inducible’ isoform of nitric oxide synthase (iNOS, NOS II) mediates some of the neurotoxic effects of inflammation (Iadecola et al, 2004). After focal cerebral ischemia, in rodents as in humans, iNOS is expressed in infiltrating inflammatory cells and in cerebral blood vessels within the ischemic territory (Forster et al, 1999; Galea et al, 1998; Hirabayashi et al, 2000; Iadecola et al, 1995b). Pharmacological inhibition of iNOS or downregulation of iNOS expression using antisense oligonucleotides reduces the infarct produced by middle cerebral artery (MCA) occlusion in rodents (Iadecola et al, 1995a; Parmentier et al, 1999; Parmentier-Batteur et al, 2001; Sugimoto and Iadecola, 2002), while iNOS-deficient mice are protected from focal cerebral ischemia (Iadecola et al, 1997; Loihl et al, 1999). The toxicity of iNOS has been attributed to the large amounts of nitric oxide (NO) produced by this enzyme, which results in oxidative stress, DNA damage, and inhibition of mitochondrial respiration (Keynes and Garthwaite, 2004). Considering the central role that iNOS plays in the pathogenic mechanisms of postischemic inflammation, it would be important to determine whether the reduced susceptibility to cerebral ischemia in females is related to suppression of the expression of this enzyme.
In the present study, we used a mouse model of focal cerebral ischemia–reperfusion to investigate whether iNOS contributes to the sexual dimorphism of ischemic brain injury. We found that postischemic iNOS expression is reduced in female mice, and that such reduction is abolished by OVX and reinstated by estrogen replacement. Furthermore, the iNOS inhibitor aminoguanidine (AG) did not reduce infarct volume in female mice. Similarly, female iNOS-null mice were not protected from ischemic injury. These findings suggest that estrogen-induced suppression of iNOS expression may contribute to the reduced susceptibility to ischemic injury in females.
Materials and methods
Mice
C57Bl/6 mice (age 2 to 3 months; weight 20 to 23 g) were obtained from Charles River (Wilmington, MA, USA). The inducible isoform of nitric oxide synthase-null mice (C57Bl/6 congenic) were obtained from an in-house colony (Cho et al, 2005b; Iadecola et al, 1997; Park et al, 2004).
Transient Middle Cerebral Artery Occlusion
All procedures were approved by the Institutional Animal Care and Use Committee. As described in detail elsewhere (Cho et al, 2005a, b; Park et al, 2004), mice were anesthetized with isoflurane (1.5% to 2%). A fiber-optic probe was glued to the parietal bone (2 mm posterior and 5 mm lateral to bregma) and connected to a laser-Doppler flowmeter (Periflux System 5010, Perimed, Sweden) for continuous monitoring of CBF. For MCA occlusion, a heat-blunted surgical suture (6-0) was inserted into the exposed external carotid artery, advanced into the internal carotid artery, and wedged into the circle of Willis to obstruct the origin of the MCA. The filament was left in place for 20 mins and then withdrawn. Only animals that exhibited a reduction in CBF > 85% during MCA occlusion and in which CBF recovered by > 80% after 10 mins of reperfusion were included in the study. Rectal temperature was kept at 37.0°C ± 0.5°C during surgery and in the recovery period, until animals regained consciousness. We used 20 mins of ischemia because longer durations did not allow the mice to survive for 72 h, the time at which infarct volume was assessed. The size of the ischemic lesion produced by 20 mins MCA occlusion was comparable to that observed in other studies of focal ischemia in mice (Dubal et al, 2001; Prass et al, 2003).
Ovariectomy, Estrogen Replacement and Estrogen Assay
Female mice underwent aseptic bilateral surgical OVX via a dorsal incision under isoflurane anesthesia 2 weeks before MCA occlusion. One group of OVX mice received estrogen replacement, consisting of estrogen benzoate (Sigma, St Louis, MO, USA) (1 μg in 100 μL, subcutaneously) (Qiu et al, 2003) for 6 days starting 3 days before MCAO. Vehicle-treated mice in the OVX group received sesame oil (100 μL, subcutaneously). Plasma estradiol levels were measured with a commercially available ELISA assay (Cayman Chemical, Ann Arbor, MI, USA) according to the manufacturer's instructions. Uterine weight, an index of estrogen activity, was also measured.
Infarct Volume Measurement and Aminoguanidine Treatment
Brains were removed, frozen, and sectioned (thickness: 30 μm) in a cryostat (Iadecola et al, 1997; Park et al, 2004). Brain sections were collected serially at 600-μm intervals, and stained with cresyl violet. Infarct volume was determined using an image analyzer (MCID, Imaging Research Inc., St Catharines, Ontario, Canada). To eliminate the contribution of postischemic edema to the volume of injury, infarct volumes were corrected for swelling by comparing ischemic and nonischemic hemispheres as described (Cho et al, 2005a; Iadecola et al, 1997; Park et al, 2004). The iNOS inhibitor AG (Sigma) (100 mg/kg in saline, intraperitoneally) was administered twice a day (1000 and 1800 h) for 3 days, starting 10 mins after reperfusion (Park et al, 2004). Vehicle (saline)-injected mice served as controls.
Quantitative ‘Real-Time’ PCR
Procedures for real-time PCR have been described previously (Park et al, 2004). Briefly, mice in which ischemia was induced (n = 4/group) were killed 6, 24, and 72 h after reperfusion. Sham-operated mice served as controls (n = 4/group). Total RNA was prepared from the ischemic and contralateral hemispheres using Trizol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA). Quantitative determination of gene expression levels, employing a two-step cycling protocol, was performed on a Chromo 4 detector (Peltier Thermal Cycler) (MJ Research, Waltam, MA, USA). Primers for iNOS (forward, 5'-CAGCTGGGCTG TACAAACCTT-3', and reverse, 5'-CATTGGAAGTCAAGC GTTTCG-3'), ICAM (forward, 5'-GCCTTGGTAGAGGTGA CTGAG-3', and reverse, 5'-GACCGGAGCTGAAAAGTTG TA-3'), and gp91phox (5'-CCAACTGGGATAACGAGTTCA-3', and reverse, 5'-GAGAGTTTCAGCCAAGGCTTC-3') were purchased from Invitrogen Life Technologies. In all, 2 μL of diluted cDNA (1:10) was amplified by Platinum SYBR green qPCR supermix UDG (Invitrogen Life Technologies). The reactions were incubated at 50°C for 2 mins and then at 95°C for 10 mins. A PCR cycling protocol consisting of 15 secs at 95°C and 1 min at 60°C for 45 cycles was required for quantification, and relative expression levels were calculated (Livak and Schmittgen, 2001). Quantities of all targets in the test samples were normalized to the mouse HPRT housekeeping gene, and values were normalized to those of sham-operated mice.
Immunocytochemistry
Immunocytochemical procedures were identical to those described previously (Cho et al, 2005b; Iadecola et al, 1996). Sections (7 μm) from formalin-fixed, paraffin-embedded brains were incubated overnight (4°C) with an iNOS polyclonal antibody (Upstate Biotechnology Incorporated, Lake Placid, NY, USA; dilution 1:5000), or with antibodies to the peroxynitrite marker 3-nitrotyrosine (NT) (Upstate Biotechnology Incorporated; dilution 1:2000) (Cho et al, 2005b). In certain conditions associated with inflammation, tyrosine nitration can also be promoted by nitrite (Eiserich et al, 1998). However, in the absence of florid inflammation, this is unlikely to be a relevant factor affecting the specificity of NT as a marker of peroxynitrite. Sections were washed and incubated with the secondary antibody (Vector) for 30 mins. The immunocomplex was visualized using the ABC complex method (Vectastain Elite Kit, Vector). The specificity of the immunolabel was previously tested by preadsorption of the antigen and by removing the primary antibody (Forster et al, 1999).
Statistical Analysis
Data are expressed as mean ± s.e.m. Two-group comparisons were statistically evaluated by the Student's t-test. Multiple comparisons were evaluated by the analysis of variance and Fisher's protected least significant difference (PLSD) test. Differences were considered significant at P < 0.05.
Results
Sex Differences in Ischemic Brain Injury are Abolished by Ovariectomy and Reestablished by Estrogen Replacement
Middle cerebral artery occlusion produced well-defined infarcts involving both the cortex and the striatum. In agreement with reports by others (e.g., Alkayed et al, 1998), infarct volume was larger in male than in female mice (Figure 1A). The changes in CBF produced by MCA occlusion and reperfusion did not differ in males and females (Figure 1B). In female mice, OVX reduced estradiol levels and uterine weights, effects that were counteracted by estrogen replacement (Table 1). The reduction in estradiol levels in OVX did not reach statistical significance due to variability associated with estrogen levels in randomly cycling females (Table 1). Ovariectomy increased infarct volume to levels comparable to those observed in male mice (Figure 1A), while estrogen replacement decreased infarct volume to the level seen in intact females (Figure 1A). These findings establish that sex differences in stroke volume are present in our model, and that they are abolished by OVX and reinstated by estrogen replacement.
Effect of ovariectomy and estrogen replacement on plasma estradiol concentration and uterine weight in female WT and iNOS-null mice
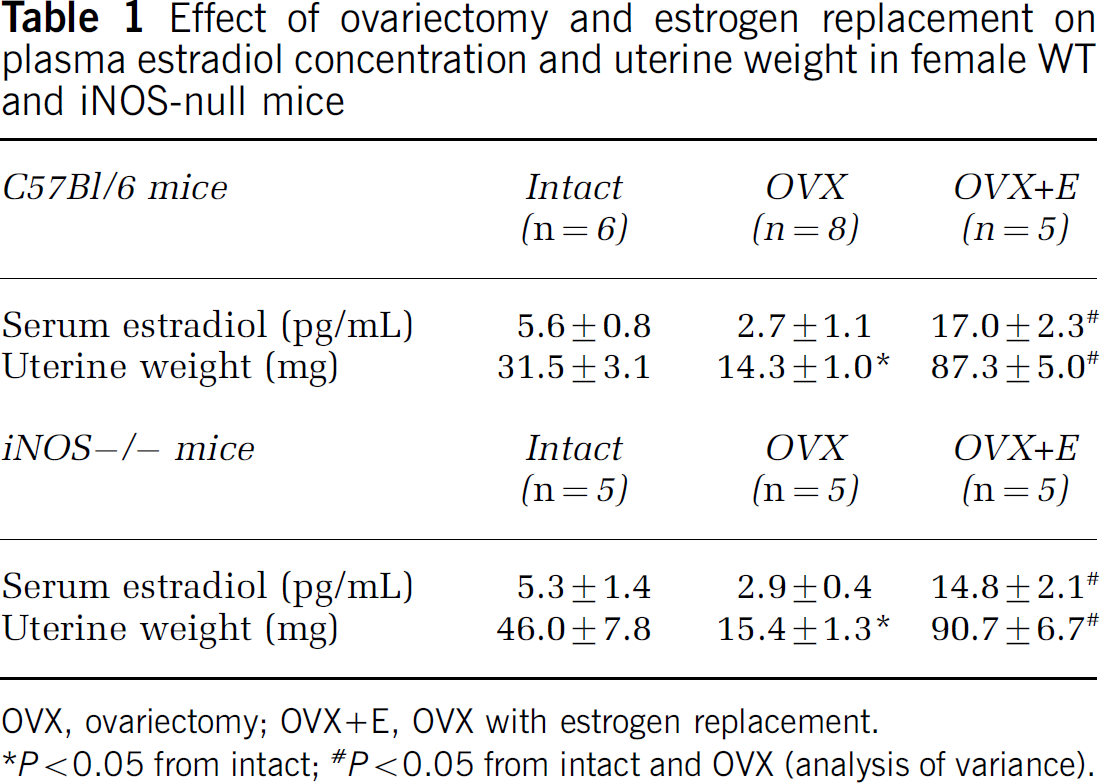
OVX, ovariectomy; OVX+E, OVX with estrogen replacement.
P < 0.05 from intact;
P < 0.05 from intact and OVX (analysis of variance).
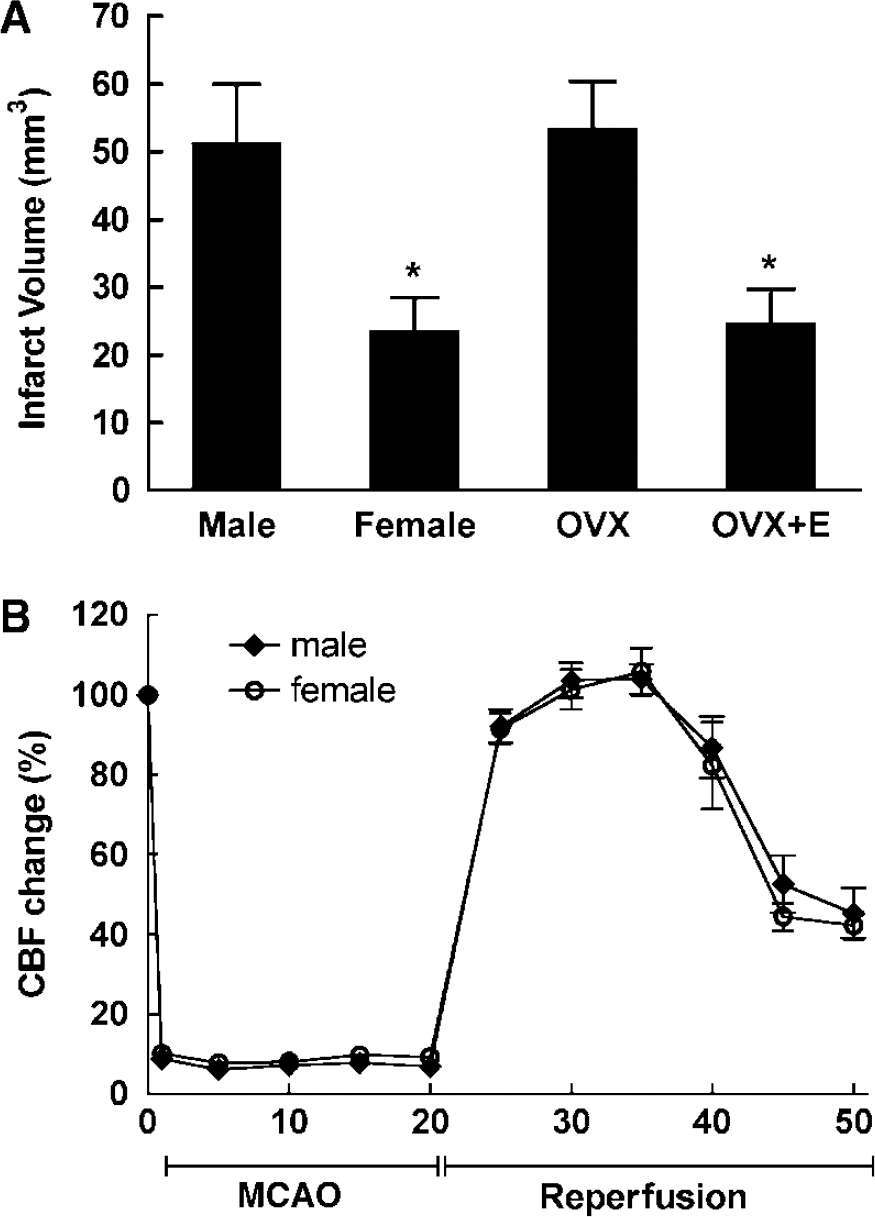
Sex differences in ischemic brain injury produced by transient occlusion of the MCA. (
Postischemic Inducible Isoform of Nitric Oxide Synthase mRNA Expression is Attenuated in Female Mice
We next examined whether changes in postischemic iNOS expression could contribute to sex differences in the outcome of cerebral ischemia. In male mice, iNOS mRNA was elevated at 24 and 72 h after MCA occlusion (Figure 2A) (P < 0.05 from control). However, in female mice, iNOS expression was markedly lower (Figure 2A) (−53% at 72 h; P < 0.05 from males). Ovariectomy enhanced iNOS mRNA at 6 and 24 h after ischemia (P < 0.05), but not at 72 h (Figure 2A) (P > 0.05). The iNOS upregulation induced by OVX was reversed by estrogen replacement (Figure 2A) (P > 0.05 from females). To determine whether other inflammatory genes were also influenced by gender, we investigated the postischemic mRNA expression of the adhesion molecule ICAM-1 and the NADPH oxidase subunit gp91phox. ICAM-1 mRNA expression was increased after ischemia reaching a maximum at 24 h (Figure 2B) (P < 0.05). The elevation was much greater in male than in normal female mice (Figure 2B) (P < 0.05). ICAM-1 expression was markedly enhanced by OVX, including the time point before ischemia, but this enhancement was attenuated to levels observed in intact females by estrogen replacement (Figure 2B). gp91phox mRNA upregulation was only slightly reduced in female mice at 24 h, and did not differ between male and females at 72 h (Figure 2C) (P > 0.05). Ovariectomy enhanced gp91phox expression (Figure 2C). The effect was observed in controls, and 6 h after ischemia, and was counteracted by estrogen replacement (Figure 2C).
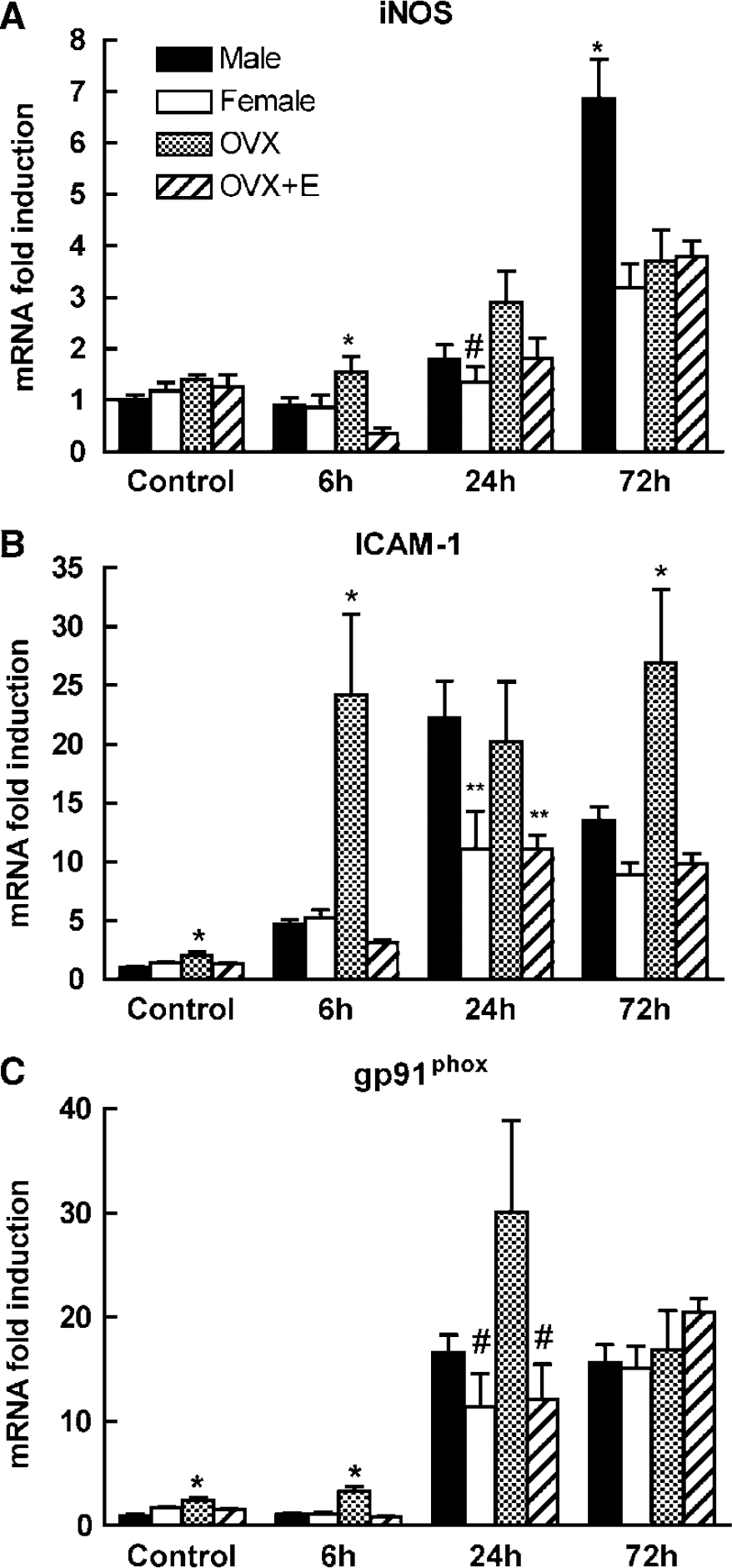
Time course of the expression of iNOS (
Postischemic Inducible Isoform of Nitric Oxide Synthase Immunoreactivity is Attenuated in Female Mice
We used immunocytochemistry to determine whether the attenuation in iNOS mRNA expression was associated with a reduction in iNOS protein 72 h after ischemia-reperfusion. In male mice (n = 3), iNOS immunoreactivity was observed in the wall of cerebral blood vessels and in inflammatory cells in the ischemic territory, as described previously (Iadecola et al, 1995b, 1996) (Figure 3A). Immunoreactivity for NT, a marker of the NO reaction product peroxynitrite, was also observed in inflammatory cells and cerebral blood vessels (Figure 3B). The inducible isoform of nitric oxide synthase and NT immunoreactivity were markedly lower in the brains of female mice (n = 3) (Figures 3C and 3D), but the expression was enhanced by OVX (n = 3) (Figures 3E and 3F).
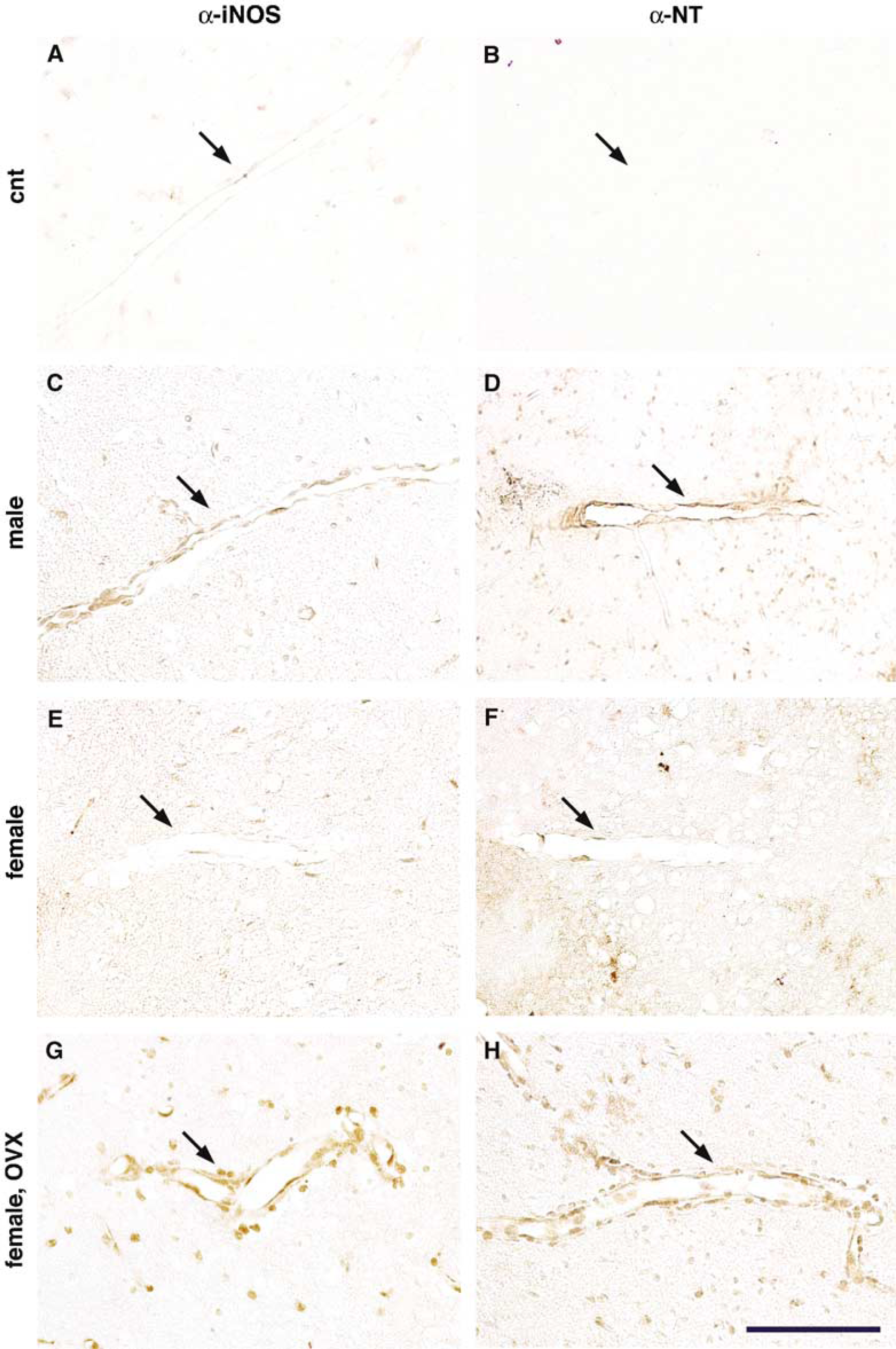
Expression of iNOS or NT immunoreactivity in the postisehemic brain of male, female, and OVX mice, 72 h after MCA occlusion. (
Pharmacological Inhibition or Genetic Deletion of Inducible Isoform of Nitric Oxide Synthase does not Confer Protection in Female Mice
The observation that iNOS expression is attenuated in female mice suggests that the component of ischemic damage attributable to iNOS is not present in females. To test this hypothesis, we examined the protective effect of the iNOS inhibitor AG and iNOS deletion in male and female mice. Aminoguanidine attenuated infarct volume in male (−55%), but not in female, mice (Figure 4A). Similarly, infarct volume was reduced in male (−45%), but not in female, iNOS-null mice (Figure 4B), an effect independent of changes in postischemic CBF (Figure 4C). Therefore, the protective effects of inhibition or genetic deletion of iNOS are not observed in female mice.
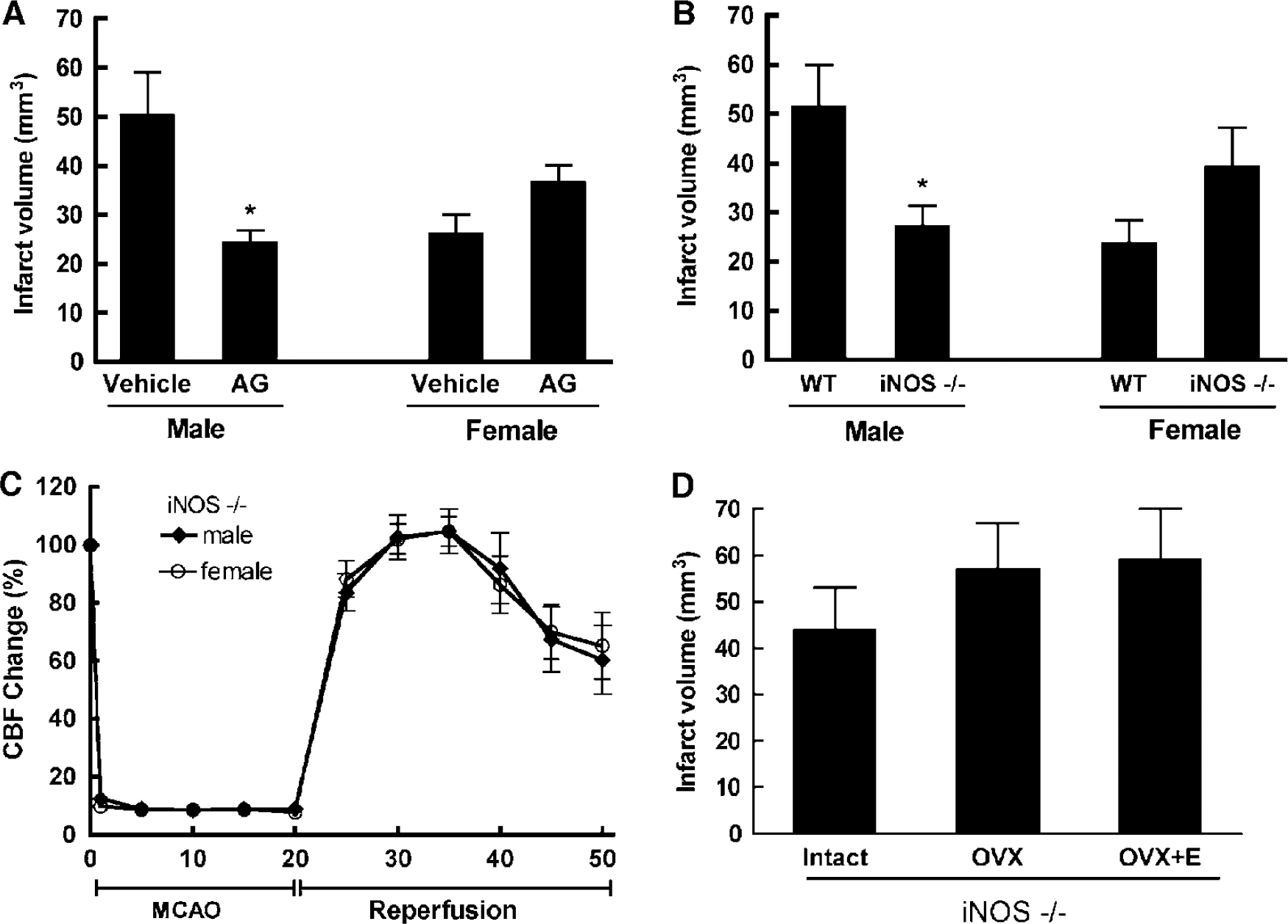
Sex differences in the effect of iNOS inhibition on ischemic injury. (
Estrogens do not Confer Protection in Female Inducible Isoform of Nitric Oxide Synthase-Null Mice
To determine whether the protection exerted by estrogens requires iNOS for its expression, we investigated the effect of OVX and estrogen replacement in iNOS-null mice. In iNOS nulls, estradiol levels and uterine weights were reduced by OVX and reestablished by estrogen replacement (Table 1). Ovariectomy produced a small increase in stroke volume in iNOS-null mice, which did not reach statistical significance in this small group (Figure 4D) (P > 0.05). Unlike wild-type (WT) mice (Figure 1A), estrogen replacement after OVX did not reduce injury volume in iNOS nulls (Figure 4D) (P > 0.05).
Discussion
We have shown that female mice are relatively protected from focal cerebral ischemia and that such protection is associated with a reduction in postischemic iNOS expression compared with male mice subjected to a comparable ischemic insult. Ovariectomy abolished the reduction in ischemic injury observed in female mice and increased iNOS expression at 6 and 24 h, while estrogen replacement attenuated both iNOS expression and ischemic injury. Furthermore, treatment with the iNOS inhibitor AG reduced ischemic injury in male, but not in female, mice. Similarly, the reduction in ischemic injury observed in male iNOS-null mice was not observed in female nulls, a finding reported previously by others (Loihl et al, 1999). These observations, collectively, suggest that the component of ischemic brain injury attributable to iNOS is lacking in female mice, and support the hypothesis that reduced iNOS expression contributes to the protection observed in females.
Smaller infarcts tend to have a less pronounced inflammatory response (Akopov et al, 1996; Kochanek and Hallenbeck, 1992). However, the reduction in iNOS mRNA expression is unlikely to be the consequence of the smaller infarcts in female mice because our data show that the magnitude of iNOS induction at 72 h is not proportional to the final size of the infarct. For example, OVX mice had a level of iNOS mRNA at 72 h similar to that observed in intact females or estrogen-treated OVX mice, but their infarct volume was twice as large. Consistent with a causal link between reduced iNOS expression and reduced infarct size, the iNOS inhibitor AG was not effective in female mice and female iNOS-null mice were not protected from cerebral ischemia. These observations support the conclusion that the reduction in iNOS expression in females is causally related to the smaller stroke rather than a consequence of reduced tissue injury. The lack of reduction in infarct volume in female iNOS-null mice or female mice treated with AG is unlikely to result from the fact that the infarct was already maximally reduced. This is because it has been previously shown that infarct volume in female mice can be attenuated, for example, by overexpression of the free radical scavenging enzyme superoxide dismutase (SOD) (Sampei et al, 2000b). In the present study, estrogen replacement in OVX females produced increases in plasma estradiol that were higher than those observed in intact females. However, the differences in the outcome of cerebral ischemia cannot be attributed to estrogen levels because plasma levels did not differ between WT and iNOS-null mice, and were in the range reported by others (Dubal et al, 2001; Horsburgh et al, 2002).
The sex differences in the susceptibility to focal cerebral ischemia have been attributed to estrogens, hormones that can modulate cerebral ischemic injury through a wide variety of actions (McCullough and Hurn, 2003; Wise et al, 2001). Some of these actions, including effects on gene expression and intracellular signaling, are mediated by estrogen receptors (ER), while other actions, such as the antioxidant properties, are receptor independent (Maggi et al, 2004). Estrogens have long been known to attenuate the expression of proinflammatory factors (Behl, 2002; Wise et al, 2001). In brain, estrogens reduce the expression of inflammatory genes, including iNOS, in activated microglial cells (Baker et al, 2004; Drew and Chavis, 2000). Although the protective effect resulting from suppression of iNOS by estrogens is well documented in cell culture and in models of brain inflammation (Drew and Chavis, 2000; Vegeto et al, 2003), the role of iNOS in the protection afforded by estrogens in ischemic brain injury is less well defined. The findings of the present study show for the first time that suppression of iNOS is an important factor in the neuroprotective effect of estrogens in models of focal ischemia.
In addition to iNOS, the postischemic expression of other inflammation-related genes was reduced in female mice. For example, the adhesion molecule ICAM-1 was attenuated in female mice, an effect abolished by OVX and reinstated by estrogen replacement. Considering the deleterious role that ICAM-1 plays in the mechanisms of ischemic brain injury (Bowes et al, 1993; Connolly et al, 1996), it is conceivable that the downregulation of this adhesion molecule also contributes to the reduced susceptibility to cerebral ischemia in female mice. Suppression of postischemic ICAM-1 expression may underlie the reduced leukocyte adhesion reported after cerebral ischemia in female mice (Santizo and Pelligrino, 1999; Santizo et al, 2000). Similarly, the postischemic expression of the NADPH oxidase subunit gp91phox was reduced in female mice. NADPH oxidase is upregulated after cerebral ischemia and contributes to ischemic injury, presumably by producing free radicals (Kusaka et al, 2004; Walder et al, 1997). Therefore, the attenuation in gp91phox expression may also play a role in the reduction in ischemic injury in females. The observation that gp91phox expression is enhanced by OVX and attenuated by estrogen replacement attests to the estrogen dependence of the effect. Therefore, in addition to iNOS, other genes are also likely to be involved in the sexual dimorphism of ischemic brain injury.
The involvement of genes other than iNOS in the deleterious effects of OVX on ischemic damage is also indicated by the fact that OVX enlarged the infarct in iNOS-null mice. However, the exacerbation of the damage in iNOS nulls (+ 29%) was small compared with that observed in WT mice (+100%), and did not reach statistical significance. However, the fact that estrogen replacement did not attenuate the lesion suggests that iNOS is required for the neuroprotective effect of estrogens. This observation is in line with other data supporting the view that iNOS is not always deleterious to the postischemic brain. Thus, iNOS is an absolute requirement for the neuroprotection exerted by ischemic preconditioning or by the preconditioning induced by the proinflammatory agent lipopolysaccharide (Cho et al, 2005b). Furthermore, iNOS is required for the protective effect of PARP inhibition (Park et al, 2004). Therefore, in addition to its deleterious role in ischemic brain injury, iNOS has also beneficial effects related to ischemic preconditioning and to the neuroprotection conferred by estrogens and PARP inhibitors.
The mechanisms by which estrogens downregulate the expression of iNOS and other inflammatory genes remain unclear, because some of these genes may lack an estrogen response element (Baker et al, 2004). Therefore, activation of ER is thought to induce intracellular signaling that ultimately leads to activation of the transcription factors AP1 and NF-κB, which, in turn, induce the expression of iNOS and other inflammatory genes (Baker et al, 2004). Although estrogens have been shown to reduce NF-κB activation in focal cerebral ischemia (Wen et al, 2004) and in astrocytic cultures treated with lipopolysaccharide (Dodel et al, 1999), in microglial cultures estrogens reduce iNOS expression without attenuating NF-κB activation (Bruce-Keller et al, 2000). However, ER signaling leads to activation of MAP kinase, which has been linked to the anti-inflammatory effect of estrogens (Maggi et al, 2004). Furthermore, ER signaling also leads to activation of the cAMP/PKA/CREB and PI3K/AKT protective pathways (Maggi et al, 2004). Therefore, it remains unclear whether the antiinflammatory effects of estrogens are mediated transcriptionally or posttranscriptionally. In addition, there is no consensus about the ER subtype (α or β) involved in the antiinflammatory and neuroprotective effects of estrogens (Baker et al, 2004; Carswell et al, 2004; Dubal et al, 2001; Sampei et al, 2000a; Vegeto et al, 2003). Therefore, further investigations are needed to define the receptor interactions and signaling through which estrogens regulate postischemic inflammation.
The observation that females are not susceptible to the beneficial effects of iNOS inhibition supports the view that a particular neuroprotective strategy cannot be assumed to be effective in both sexes. For example, infarct volume is reduced in male, but not in female, nNOS-null mice (Sampei et al, 2000b). Furthermore, infarct volume is markedly reduced in male mice lacking the DNA repair enzyme PARP, but it is exacerbated in female PARP-null mice (McCullough et al, 2005). Other neuroprotective strategies, such as overexpression of the free radical scavenger SOD, are effective in both sexes (Sampei et al, 2000b). The sexual dimorphism of the protective effect of iNOS inhibition has to be taken into consideration in the development of pharmacological treatments for human stroke.
In conclusion, we have shown that postischemic iNOS expression is lower in the brains of female mice. The iNOS expression is enhanced by OVX and reinstated by estrogen replacement. Furthermore, female mice are not susceptible to the protective effects of the iNOS inhibitor AG, while female iNOS-null mice are not protected from ischemic injury. These findings suggest that modulation of iNOS expression by estrogens is one of the factors mediating the resistance to cerebral ischemia in females, and provide additional evidence that neuroprotective strategies cannot be assumed to be effective in both sexes.
Footnotes
Acknowledgements
We thank Dr Carrie Drake for comments on an earlier version of the manuscript.
