Abstract
Transcranial Doppler (TCD) sonography was performed to assess the differential effects of caloric vestibular stimulation on cerebral blood flow (CBF) to the vestibular [middle cerebral artery (MCA)] and the visual [posterior cerebral artery (PCA)] cortices in 15 healthy volunteers with eyes closed. Caloric irrigation caused a sensation of rotation and nystagmus and not only increased velocity in the MCA (2.7 ± 3.3%) but also, simultaneously, decreased velocity in the PCA (4.2 ± 6.2%, p < 0.001 vs. MCA). CBF changes indicated by changes in velocity through the MCA and PCA may reflect functional deactivation of the visual cortex together with activation of the vestibular cortex: this beneficial multisensory interaction suppresses distressing oscillopsia.
Functional imaging of the visual cortex by means of positron emission tomography (PET) (Raichle, 1987; Zeki et al., 1991; Cheng et al., 1995) and magnetic resonance imaging (MRI) (Sereno et al., 1995) usually focuses on regional activation in response to a particular stimulation. Recently, significant deactivations of the entire striate and extrastriate visual cortices were found during activation of the vestibular cortex in a PET study using caloric irrigation of the external auditory canal for vestibular stimulation (Wenzel et al., 1996). This new phenomenon of a reciprocal visual-vestibular cortex interaction may reflect a basic sensorimotor mechanism that benefits the organism by suppressing distressing oscillopsia during involuntary ocular oscillations. We report on cerebral blood flow (CBF) changes assessed by transcranial Doppler (TCD) ultrasonography using the same paradigm of caloric vestibular stimulation as in the cited PET study. The vestibular cortex is supplied by the middle cerebral artery (MCA) and the visual cortex by the posterior cerebral artery (PCA). Hemodynamic TCD changes in these arteries will largely reflect cortical activity in the respective areas (Aaslid, 1987).
METHODS
Fifteen normal volunteers (seven women, eight men; mean age, 29.8 ± 4.8 years) took part in the study after giving informed consent. They rested in a silent, bright room, their upper body slightly elevated to ensure optimal stimulation of the horizontal semicircular canal. Either ear canal was randomly assigned for irrigation with 20°C (n = 5) or ice (n = 10) water. Eye movements were measured by means of electronystagmography (ENG). The contralateral MCA and the ipsilateral PCA were chosen for analysis, due to the fact that irrigation had produced bilaterally equal decreases of CBF in the visual areas during the PET study (Wenzel et al., 1996), whereas the CBF increase in the vestibular areas (MCA territory) is known to be more marked on the contralateral side (Friberg et al., 1985; Bottini et al., 1994). Blood vessels were identified as reported elsewhere (Fujioka et al., 1992), and two ultrasound probes were fixed in place by an elastic head band. A computerized bilateral TCD-recording system (DWL-Multi-Dop X, Sipplingen, Germany), with an analog input for simultaneous recording of arterial blood pressure (ABP), was used together with a continuous tonometric blood pressure monitor (Nellcor, N-Cat N-500, Hayward, CA, U.S.A.).
After at least 1 min of stable baseline with both eyes closed, subjects were asked to open their eyes for 60 s while fixating on a stationary target, and, after a 1-min interval, were asked to close their eyes. This was followed by 1 min of eyes open and optokinetic stimulation (rotating drum at a speed of ˜40°/s), thus eliciting optokinetic nystagmus. After another 3 min rest with both eyes closed, 100 ml of cold water was administered within 30 s to the external ear canal via a syringe. Subjects continued to rest for 3 more min after irrigation ceased. Finally, subjects were asked to open their eyes and fixate on a stationary target.
Blood flow velocity and blood pressure calculations were performed off-line, and percentage changes in time averaged mean velocity, and mean ABP were calculated from baseline values before each stimulus. To analyze the effects of eye opening and optokinetic nystagmus, respective maximal values were used to characterize flow changes after each stimulus. To calculate the effects of vestibular stimulation, readings for the 3-min rest period before irrigation were averaged and compared to the averaged data obtained for the 3-min period after irrigation ceased. In addition, data were plotted at intervals of 30 s after beginning irrigation. Statistical data were evaluated with the Mann-Whitney test. Values are given as mean ± SD.
RESULTS
Mean ABP (92 ± 13 mm Hg) did not show significant changes in response to any particular stimulation.
Baseline blood flow velocity readings and relative effects of eye opening and optokinetic nystagmus are shown in Table 1.
Percent increase of blood flow velocity due to eye opening and optokinetic nystagmus
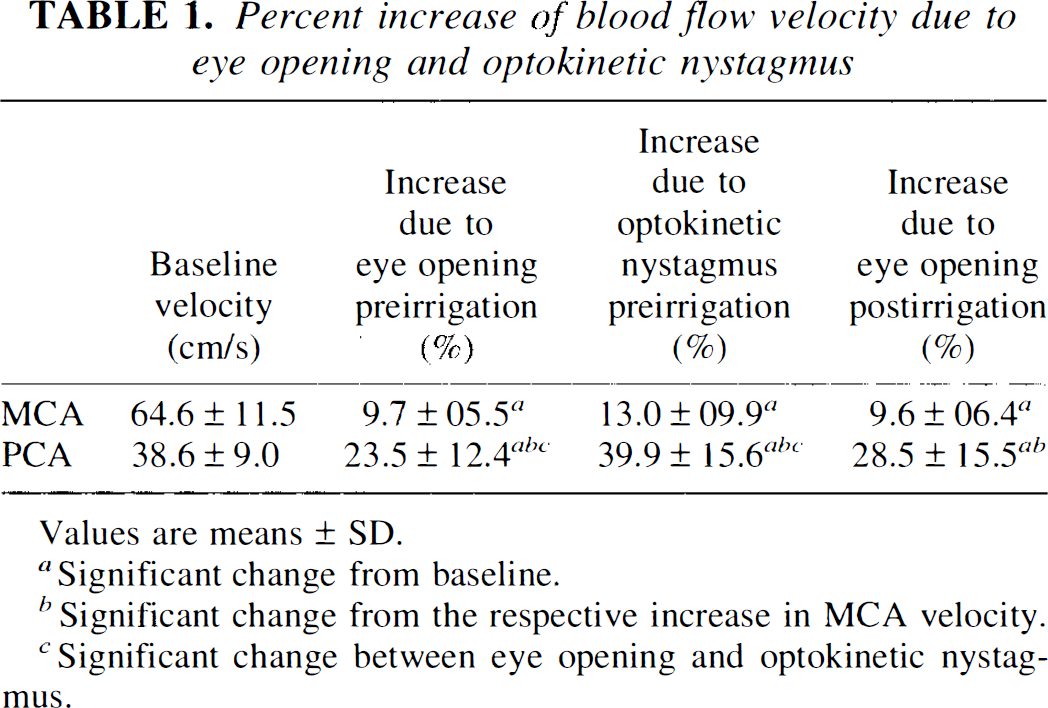
Values are means ± SD.
Significant change from baseline.
Significant change from the respective increase in MCA velocity.
Significant change between eye opening and optokinetic nystagmus.
The maximum increase in PCA flow velocity with eyes open and fixation was significantly smaller (23.5 ± 12.4%) than during optokinetic nystagmus (39.9 ± 15.6%; p < 0.01). The concurrent increases in MCA velocity were not significantly different. Effects of eyes open before and after irrigation did not differ significantly in either MCA or PCA.
At ˜30 s from the start of caloric irrigation with cold water, volunteers experienced rotational vertigo with a vestibular nystagmus beating toward the contralateral side. Both the sensation of rotation and the nystagmus monitored by ENG gradually subsided within 180 s after irrigation had ceased for all individuals. Comparison of the averaged mean blood flow velocity data for the 3-min resting interval before irrigation with those for the 3-min interval after, revealed a slight increase in MCA velocity (±2.7 ± 3.3%) and a reduction of PCA velocity (–4.2 ± 6.2%). The difference between PCA and MCA was significant (p < 0.001). No significant difference was caused by right or left ear irrigation, or by water at either of the temperatures used (Table 2).
Effects of caloric irrigation on blood flow velocity (% changes)
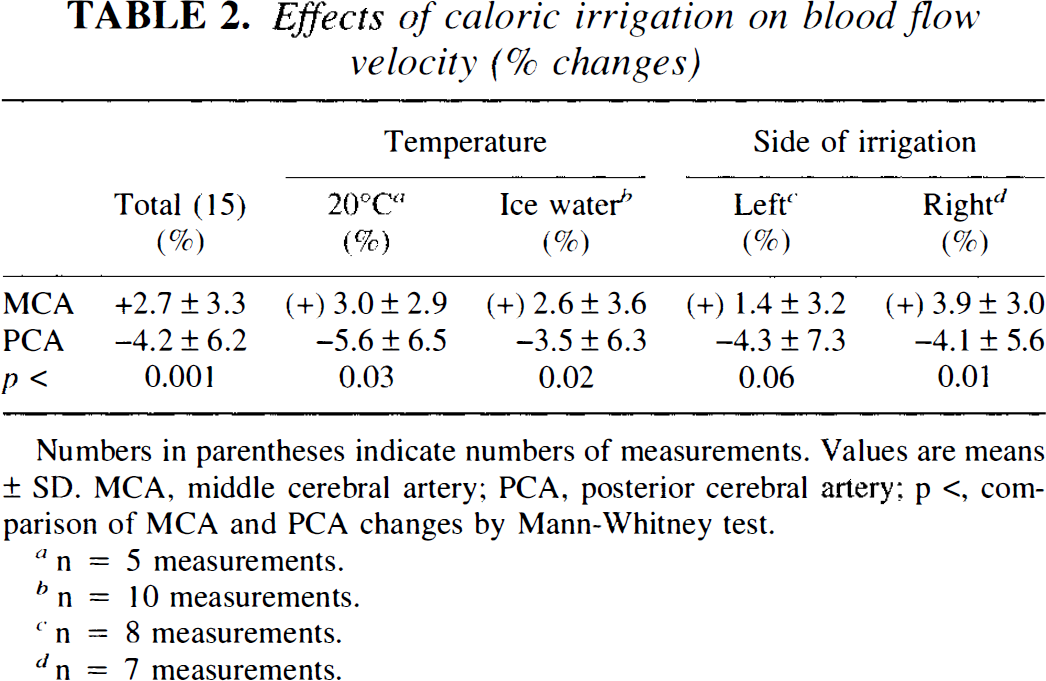
Numbers in parentheses indicate numbers of measurements. Values are means ± SD. MCA, middle cerebral artery; PCA, posterior cerebral artery; p <, com- parison of MCA and PCA changes by Mann-Whitney test.
n = 5 measurements.
n = 10 measurements.
n = 8 measurements.
n = 7 measurements.
DISCUSSION
Using TCD ultrasonography, we detected a decrease in PCA blood flow velocity during vestibular stimulation that caused a sensation of rotation and involuntary ocular oscillations. In contrast, PCA flow velocity increased when subjects voluntarily performed optokinetic nystagmus. Caloric vestibular stimulation in a recent PET study (Wenzel et al., 1996) showed a mean decrease in regional CBF (rCBF) of the visual cortex of 12.8% (range 4.6–21.0%), whereas changes obtained with TCD ultrasonography were smaller (mean, ˜5%) and difficult to differentiate from spontaneous variations of CBF at any single given moment. As a mean, however, changes in PCA flow velocity over an interval of 3 min following the end of irrigation differed significantly from those of the MCA. The latter were slightly elevated, probably indicating an activation of the (relatively small) vestibular cortex, and were indistinguishable from a general arousal reaction or induction of mental activity (Droste et al., 1989; Markus and Boland, 1992). Using the 133Xenon technique during (warm) ear irrigation, Friberg et al. (1985) did not find significant changes in CBF for the contralateral hemisphere as a whole, but described a vestibular cortical area within the MCA territory (the posterior superior part of the temporal lobe) that showed a mean increase in rCBF of 12 ± 6%. In the first PET study on vestibular cortex activation by caloric stimulation, Bottini et al. (1994) did not quantify regional activation within the MCA territory. Quantitative differences between the PET data of Wenzel et al. (1996) and our smaller and less reliable TCD data are obviously due to the methodologies themselves. A regional activation of the relatively small vestibular cortex areas within the much larger MCA territory will not cause major changes in blood flow of the MCA as measured by TCD. The incongruity between the PCA territory and the extent of human visual cortex is less pronounced; consequently, deactivation will be reflected better in PCA blood flow velocity. To detect small global changes, measurement of blood flow velocity instead of CBF requires that the diameter of the insonated vessel remains constant (Kontos, 1989). A number of studies have suggested this to be the case under various circumstances (Newell et al., 1994; Larsen et al., 1994). Furthermore, it is unlikely that our results were substantially impaired by systemic changes. Relevant changes in ABP were not seen and would be compensated for by cerebral autoregulation within a few seconds (Aaslid et al, 1989; Tiecks et al, 1995). Although CO2 was not measured, capnic changes would have caused similar velocity changes in both MCA and PCA. Thus, the observed small changes in TCD flow velocity most likely reflect changes in rCBF and associated regional neuronal activity (Lassen et al., 1978).
Activation effects of open eyes and visual fixation compare reasonably to those reported in the literature (Raichle, 1987; Aaslid et al., 1987). A more marked increase in PCA velocity during various complex visual stimuli has been described previously (Conrad and Klingelhöfer, 1989); optokinetic nystagmus in our study also significantly raised PCA blood flow. Thus, we suggest that there is a fundamental difference in the way in which oscillating eye movements modulate visual cortex activity. Movements that are voluntary and “adequate” for target fixation and pattern recognition (stationary or moving) are associated with an increase in visual cortex activity (Andersen et al., 1994). Involuntary, or “inadequate,” oscillating eye movements, which override fixation either due to vestibular stimulation or neurological disorder (opsoclonus), seem to induce down regulation of visual cortex activity. Since eyes were closed in our study, a disturbance of visual input cannot be the trigger. Ocular motor brainstem or vestibular cortex signals are the most likely triggers of visual deactivation. A direct link between the vestibular system and the visual cortex has been shown in cats (Grüsser and Cornehls, 1972). Kolev (1995) reported on visual hallucinations evoked by caloric irrigation that were compatible with transient visual cortex ischemia. Psychophysical experiments have shown decrements in the perception of object motion during self-motion or during involuntary ocular oscillations (Probst et al., 1984; Dieterich and Brandt, 1987). Downregulation is also known from other sensorimotor systems, e.g., modulation of nociception during tactile stimulation (Woolf and Thompson, 1994) or somatosensory cortex activity during limb movements (Chapin and Woodward, 1981). Such downregulation may reflect a hitherto insufficiently investigated fundamental mechanism of sensorimotor interaction.
Footnotes
Abbreviations used
Acknowledgment:
This study was supported by a grant of the Friedrich-Baur-Stiftung. The authors would also like to thank Mrs. K. Schüler for technical assistance and Mrs. J. Benson for linguistic corrections of the manuscript.
