Abstract
Cerebral autoregulation maintains stable cerebral blood flow despite fluctuations in cerebral perfusion pressure (CPP), through mechanisms that alter vascular diameter and resistance. Two commonly used indices, pressure reactivity index and mean flow index, reflect different aspects of this regulation, namely changes in cerebral blood volume and blood flow velocity. However, their interchangeability, particularly in the context of traumatic brain injury (TBI), remains questionable. This study investigates the frequency and physiological basis of discordance between these indices. Using 96 simultaneous recordings of intracranial pressure (ICP) and transcranial Doppler-derived flow velocity in TBI patients, 501 non-overlapping 20-minute segments were extracted. Each segment was classified based on autoregulatory state, and physiological parameters, including ICP, CPP, end-tidal CO₂, cerebral compliance, critical closing pressure, and vascular dynamics, were analysed. Discordance between indices occurred in 26% of segments. Statistical and machine learning models identified vessel stability metrics (e.g. wall tension, vascular resistance, time constant) and intracranial dynamics (e.g. compliance, compensatory reserve) as the most predictive features of discordance. These findings suggest that the two indices capture distinct physiological processes and should not be used interchangeably. Instead, their combined interpretation may enhance assessment of cerebrovascular autoregulation and provide a more nuanced understanding of cerebral physiology in TBI.
Keywords
Introduction
The mean flow index (Mx) and pressure reactivity index (PRx), proxy measures of cerebral autoregulation1,2 and cerebrovascular reactivity,3,4 differ due to their physiological underpinning. Mx is thought to reflect cerebral autoregulation, which assesses the brain’s ability to maintain cerebral blood flow (CBF) amid fluctuations in cerebral perfusion pressure (CPP), 5 while PRx is mostly related to cerebrovascular reactivity, which evaluates the responsiveness of vascular smooth muscle cells to changes in transmural pressure. 6 This distinction suggests that the two indices should not be used interchangeably. PRx is often favoured for assessing autoregulation in traumatic brain injury (TBI) due to the availability of continuous invasive monitoring (arterial blood pressure – ABP, and intracranial pressure – ICP). In contrast, extended transcranial Doppler (TCD) recordings are less common, making continuous Mx acquisition and use more difficult.6–8
Notably, Mx and PRx provide complementary but distinct prognostic insights. It has been suggested that PRx could be more predictive of outcomes in TBI patients, while Mx may be more informative for non-TBI patients. 9 However, a more recent review has highlighted multiple methodological shortcomings of Mx, particularly in non-TBI populations, warranting cautious interpretation of its results. 10 Moreover, in TBI patients, PRx was shown to be better at distinguishing fatal from nonfatal outcomes, while Mx differentiated better between favourable and unfavourable functional outcomes. 11
Previous research has established a moderate correlation between PRx and Mx.12–16 However, interpretation of such correlations requires caution, as previous work has stated that shared underlying signals between PRx and Mx can introduce partial mathematical coupling, and that statistical issues such as heteroscedasticity may further limit their validity. 17 Additionally, few studies have investigated the nature of the discrepancies between the two indices in detail beyond simple metric comparisons. 12 Different levels in ICP 11 and CPP 18 have been linked to different levels of disparity between the two measurements. Additionally, changing the absolute level of ABP (e.g. from an average of 60–70 mmHg) for a prolonged duration affected Mx, but not PRx. 19
Both Mx and PRx are known to be noisy metrics20,21 and are predicated on assumptions and prerequisites for accurate interpretation. They often require time-averaging over extended periods to improve signal-to-noise ratios, raising questions about whether observed discrepancies reflect physiological differences or are merely violations of assumptions and the presence of artefactual noise. Is there additional information hidden in their discrepancy or lack of?
In this study, we set out to: (1) Assess the degree of disparity between CBF autoregulation and cerebrovascular pressure reactivity in datasets containing simultaneous Mx and PRx recordings from patients with TBI; and (2) Explore the physiological factors that may be at play for the discordance between the two indices.
This is the first study to systematically analyse the divergence between Mx and PRx in high-resolution, continuous recordings, using multiple physiological variables and modelling approaches. By focusing not only on correlation but also on the drivers of disagreement between these two commonly used indices, our aim is to better understand their complementary nature and potential limitations, particularly in the context of physiological interpretation in TBI.
Materials and methods
Ethics
Deidentified data were retrieved for this investigation from the Brain Physics database, which received approval from the Yorkshire & The Humber – Leeds East Research Ethics Committee (REC 23/YH/0085; IRAS ID: 320219) in April 2023. The standard of care for TBI patients admitted to the neurocritical care unit (NCCU) includes vital signs monitoring and intracranial pressure. In addition, TCD monitoring was carried out as part of a clinical audit (Clinical Project ID4201: TCD-based assessment of intracranial haemodynamics in patients admitted to NCCU). The requirement for informed consent was waived by the local ethics authorities. All procedures were conducted in accordance with the principles of the Declaration of Helsinki and applicable local regulations.
Patients
The eligibility of each subsequent TBI patient needing ICP-directed therapy was assessed. Essentially, patients admitted to Addenbrooke’s Hospital NCCU in Cambridge between 2022 and 2024, deemed suitable for full multimodal monitoring, were monitored daily with extended TCD, as previously described. 7 Patient characteristics, including sex, age, Glasgow Coma Scale (GCS) scores, and whether the patient sustained an isolated TBI, were collected.
Multimodal monitoring
An intraparenchymal pressure sensor (Codman ICP MicroSensor® (Codman & Shurtleff, Massachusetts, US)) was used to monitor ICP. Using intra-arterial catheters that were placed into the femoral or radial arteries and zeroed to the third ventricle's level (Arterial line, Transpac® IV Pressure Transducer (Baxter Healthcare, Illinois, US)), ABP was measured. End-tidal carbon dioxide (EtCO2) was determined using the GE CARESCAPE™ Respiratory Modules E-sCO(V) (GE Healthcare Technologies, Illinois, USA). For a prolonged middle cerebral artery (MCA) insonation, TCD probes were placed bilaterally and secured in place using a probe holder. Both the Delica EMS 9D System® (Shenzen Delica Medical Equipment Co. Ltd, China) and the Doppler Box® (DWL Compumedics, Singen, Germany) were employed to measure cerebral blood flow velocity (FV).
The ICM+® software (Cambridge Enterprise Ltd, Cambridge, UK) was used to integrate all signals of interest (ABP, ICP, bilateral FVs, and EtCO2) at a sampling frequency of 240 Hz.
Signal processing
All the data cleaning and signal processing were performed using ICM+. Through visual inspections, only a single FV trace of better quality of the two recorded was chosen for analysis. Initially, high-resolution (raw waveform traces) data were processed by experienced clinicians to exclude non-physiologic or highly distorted segments, including low signal-to-noise ratio in FV, arterial line clogging and flushing, and movements. Then, automated cleaning was performed for both ABP and ICP, excluding values outside of the physiological range (i.e. ABP > 300 mmHg or < 0 or pulse amplitude of <20 mmHg; ICP of <−30 or >200 mmHg) as well as flat lines (signals with no variations). 7
Pulsatile cerebral blood volume (CaBV) was derived from the high-resolution FV waveforms by integration over time around its beat mean value. 22 For the subsequent calculations, ABP, ICP, EtCO2, FV and CaBV were downsampled to 0.1 Hz.
The computation of the physiological parameters was conducted as outlined in Table 1. The calculation of the autoregulatory indices was based on the previously described methodology, 1 which relies on the slow, spontaneous fluctuations of ABP and/or CPP, rather than the application of an external stimulus. Additionally, the arterial mean flow index (Mxa) was determined in the same way as Mx, using ABP instead of CPP.
Formula and description of the physiological parameters.
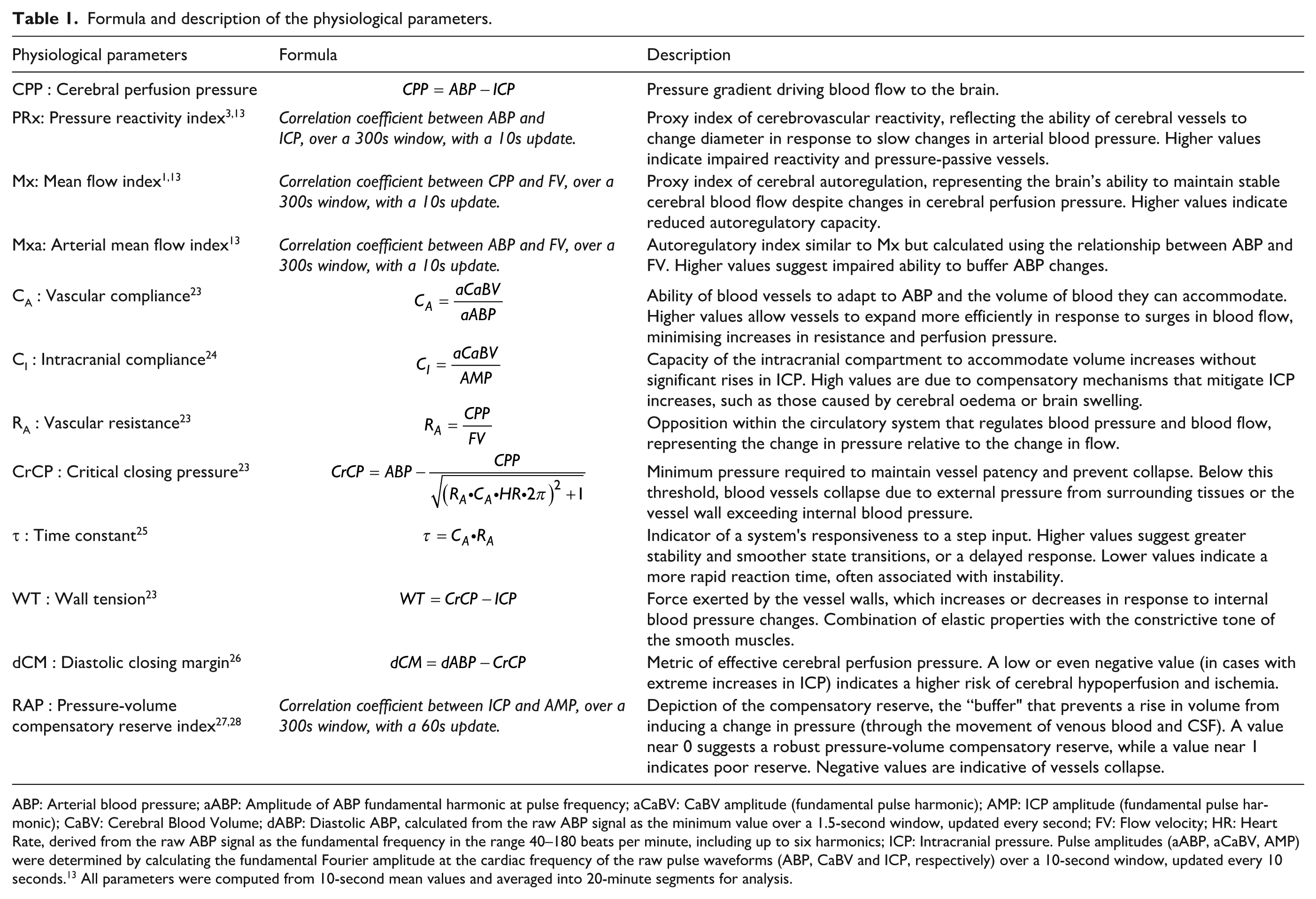
ABP: Arterial blood pressure; aABP: Amplitude of ABP fundamental harmonic at pulse frequency; aCaBV: CaBV amplitude (fundamental pulse harmonic); AMP: ICP amplitude (fundamental pulse harmonic); CaBV: Cerebral Blood Volume; dABP: Diastolic ABP, calculated from the raw ABP signal as the minimum value over a 1.5-second window, updated every second; FV: Flow velocity; HR: Heart Rate, derived from the raw ABP signal as the fundamental frequency in the range 40–180 beats per minute, including up to six harmonics; ICP: Intracranial pressure. Pulse amplitudes (aABP, aCaBV, AMP) were determined by calculating the fundamental Fourier amplitude at the cardiac frequency of the raw pulse waveforms (ABP, CaBV and ICP, respectively) over a 10-second window, updated every 10 seconds. 13 All parameters were computed from 10-second mean values and averaged into 20-minute segments for analysis.
The final analyses were performed on non-overlapping 20-min sections containing the averaged values of the following parameters: PRx, Mx, Mxa, ICP, FV, CPP, EtCO2, CA, CI, RAP, RA, τ, CrCP, WT and dCM. Thresholds are often applied to Mx and PRx to represent impaired and functional autoregulation, supported by patient outcomes studies and associations to other autoregulation indices. While PRx thresholds are well-established in the literature, based on clinical studies which assessed the impact of PRx on outcome,29–31 Mx thresholds are less consistent. A threshold of 0.25–0.45 is reported depending on the population and methodology used.17,32,33 Values above this threshold suggest impaired autoregulation, while values below indicate working autoregulation. However, highly negative Mx values denote an inverse relation between CPP and FV, which contradicts the expected behaviour of working autoregulation. Therefore, sections with particularly negative Mx values (Mx < −0.3) were labelled ‘Mx–’. PRx and Mx were finally stratified a priori as ‘impaired’ represented by a positive sign (PRx > 0.3, Mx > 0.3) and ‘working’ characterised by a negative sign (PRx ⩽ −0.1, Mx ∈ [−0.1, 0.3]), based on previous studies and our predefined hypotheses. The lower bound of working Mx (−0.1) was set to include some variations of Mx around 0. Similarly, the upper constraint for working PRx (−0.1) was chosen to rule out low ABP–ICP correlations around 0, which may simply mean insufficient variance of ABP waves or insufficient transmission to ICP.12,29 Nevertheless, these cut-offs are not thought of as absolute physiological boundaries but rather operational definitions to facilitate structured comparisons between the two indices.
Statistical analysis
Statistical analysis and figure generation were conducted using R V.4.4.1, with the following packages: ‘brms’, ‘gbm’, ‘ggplot2’, ‘MASS’, ‘nnet’, ‘randomForest’, ‘reptree’ and ‘rstatix’. The 95% Confidence Interval (CI) was reported where appropriate.
Patient characteristics are reported as median [interquartile range (IQR)] for continuous and ordinal variables, and as counts (percentages) for categorical variables. Analyses were performed on complete cases.
The agreement between PRx and Mx was assessed by calculating the correlation coefficient between the raw values, for descriptive purposes, and reporting the counts and percentage of sections categorised into each class: (1) PRx−, Mx−; (2) PRx−, Mx+; 3) PRx+, Mx−; and 4) PRx+, Mx+. As an additional assessment, a Bland–Altman analysis was conducted to evaluate the agreement between Mx and PRx, including the calculation of mean bias, standard deviation of the differences, and 95% limits of agreement. The relationship between the two autoregulation metrics and ICP/CPP was examined. Additionally, the relationship between PRx and ICP/CPP was assessed over the whole monitoring session (i.e. from the start to the end of the ICP monitoring) of the patients included in this study. PRx and Mx were then evaluated for their ability to predict impaired Mx and PRx, respectively, with the area under the receiver operating characteristic curve (AUC) reported as a measure of predictive performance.
Further analyses were conducted on stratified PRx and Mx values, grouped into categories representing agreements (PRx−/Mx− and PRx+/Mx+) and discrepancies (PRx−/Mx+ and PRx+/Mx−). While patient-specific variability and uncertainty in classification exist, dichotomisation facilitates interpretability and structured analyses of physiological predictors. This study should not be regarded as a validation of the cut-offs used, but rather as an approach to evaluate which physiological parameters underlie the concordance or discordance between PRx and Mx in TBI patients. Class ‘Mx−’ was excluded from the primary analysis as it’s hypothesised that this finding is non-physiological. However, when indicated, some of the analyses were repeated, including this class, to provide further insights. Data normality was evaluated using the Shapiro-Wilk test.
Physiological variables were evaluated against the four classes (and additionally with Mx–) using the Kruskal-Wallis test, followed by post-hoc Dunn’s test with Holm-Bonferroni correction, where appropriate, with results presented as boxplots. The predictive power of these variables was assessed through various models. The similarity between Mxa and Mx/PRx was evaluated in each group by performing a two-sided Wilcoxon rank-sum test on the absolute difference between Mxa and PRx, and Mxa and Mx. Mxa is projected to be closer to Mx due to the physiological notion underlying them, except when ICP contributions to CPP variations are significant.
To ensure robustness of our findings and avoid reliance on a single modelling framework, multiple predictive strategies, including classical and machine learning approaches, were applied. This approach was chosen to evaluate the consistency of results across models with differing assumptions, especially given the limited sample size.
Initially, univariable multinomial logistic regression (MLR) models were fitted, followed by a multivariable MLR model incorporating all parameters. Odds Ratios (OR) and results from two-tailed Wald-z tests were reported using Class 1 as the reference for comparison, both for the four classes and when including Mx–. The likelihood ratio test (LRT) was used to evaluate the statistical significance of the included predictors. To account for patient-level variability, a Bayesian mixed-effects MLR model was fitted using the ‘brms’ package (i.e. fits Bayesian generalised linear models using Stan as the backend). This methodology was chosen to accommodate the complexity of the analysis, which involved numerous predictors and nested random intercepts. The outcome variable was modelled as categorical with the four defined classes, by incorporating the physiological variables as fixed effects, and the recording number and the patient number as random intercepts. Bayesian inference was performed using Hamiltonian Monte Carlo in Stan (4 chains, 2000 iterations with 1000 warm-up iterations each, 0.8 adaptation and maximum tree depth at 10), with weakly informative priors (Fixed effects: Normal distribution, mean = 0, standard deviation = 2.5; Intercept: Student’s t-distribution, degree of freedom = 3, mean = 0, scale = 2.5; Random effects: Normal distribution, mean = 0, standard deviation = σ assigned to a half-Student-t prior) to balance flexibility and stability in estimation. Classification was based on posterior probabilities, assigning each observation to the class with the highest predicted probability. The contribution of the random effect of patients was assessed using leave-one-out cross-validation (LOO). The LOO log-likelihood, expected log pointwise predictive density (ELPD), and variance were compared between the mixed-effects and fixed-effects models.
Finally, random forest (RF) models with 500 trees were applied using different predictors and outcomes. The dataset was randomly split into a 70% training set and a 30% testing set, maintaining a similar class distribution in both subsets. Model development, including feature selection and hyperparameter tuning (where applicable), was carried out solely on the training set. The final model performance was assessed on the independent testing set to minimise overfitting and evaluate generalisability. The ‘reprtree’ package was employed to extract a representative tree from the trained random forest models. Tree similarity was assessed based on prediction concordance across the ensemble, and the tree minimising the aggregate distance to all others was selected as the most representative. This approach preserves the predictive structure of the forest while enabling interpretation via a single decision tree. To further validate the results, linear discriminant analysis (LDA) and gradient boosting regression (GBR) models were also fitted. For both RF and GBR models, variable importance plots were generated. To account for potential data leakage, 5-fold patient-level cross-validation was performed, ensuring that all segments from a given patient were included exclusively in either the training or testing set.
The goodness-of-fit for each model was assessed using contingency tables defined by the number of correctly and incorrectly categorised sections for each class, from which the models’ overall accuracy and multiclass AUC were determined, and Fisher’s exact test was applied. Class-specific balanced accuracy and one-versus-rest AUC were presented.
Data availability
The processed data can be made available upon a reasonable request to the corresponding author.
Results
Patient characteristics
96 recordings were collected from 38 TBI patients (median of 3 (IQR: 2, 4) recordings per patient, 266 ± 97 min each on average), adding to approximately 400 h of recording. A total of 1287 observations (20-min sections) were extracted. Among these patients, 30 (83%) were male, with a median age of 47 years (IQR: 31, 55). The median initial GCS score was 8 (IQR: 6, 12), and 9 (24%) patients sustained an isolated TBI.
Agreement between metrics
The overall correlation between Mx and PRx was moderate but statistically significant considering the 20-minute sections (r = 0.461, 95% CI (0.416, 0.501), p < 0.001) and the average values per monitoring sessions (r = 0.511, 95% CI (0.344, 0.646), p < 0.001), following the linear relationship described by the equation Mx = 0.49 * PRx + 0.06 (Supplemental Figure 1(a)). Bland-Altman analysis revealed a small mean bias of 0.07 between Mx and PRx, with a standard deviation of differences of 0.38. The 95% limits of agreement ranged from −0.67 to 0.81, indicating substantial variability in differences between the two indices (Supplemental Figure 1(b)). In terms of predictive power, Mx demonstrated superior accuracy in predicting impaired vascular reactivity (PRx > 0.3) compared to PRx’s ability to predict impaired cerebral autoregulation (Mx > 0.3), with an AUC of 0.857 (95% CI (0.826, 0.887)) versus an AUC of 0.763 (95% CI (0.717, 0.809)).
The relationships between Mx and PRx with CPP and ICP, over the TCD recordings, are illustrated in Figure 1. Mx and CPP displayed a U-shaped curve, with the lowest Mx values observed at CPP = 85 mmHg. Mx increased with rising ICP until reaching a plateau around ICP = 17 mmHg. Conversely, PRx decreased with increasing CPP, stabilising between CPP = 70–80 mmHg. While PRx remained relatively stable across different ICP levels, it showed a sharp increase for ICP values greater than 25 mmHg. The difference between Mx and PRx varies with CPP and ICP values, showing a greater difference for CPP values above 90 mmHg and ICP values above 15 mmHg. Supplemental Figure 2 represents the relationship between CPP–PRx and ICP–PRx, over the whole ICP recording. In contrast to the association observed in the investigated sections where TCD was performed, PRx rises as CPP exceeds 90 mmHg, reaching 0.4 at CPP = 130 mmHg. PRx increases linearly with ICP above 10 mmHg, reaching 0.3 at ICP = 30 mmHg.
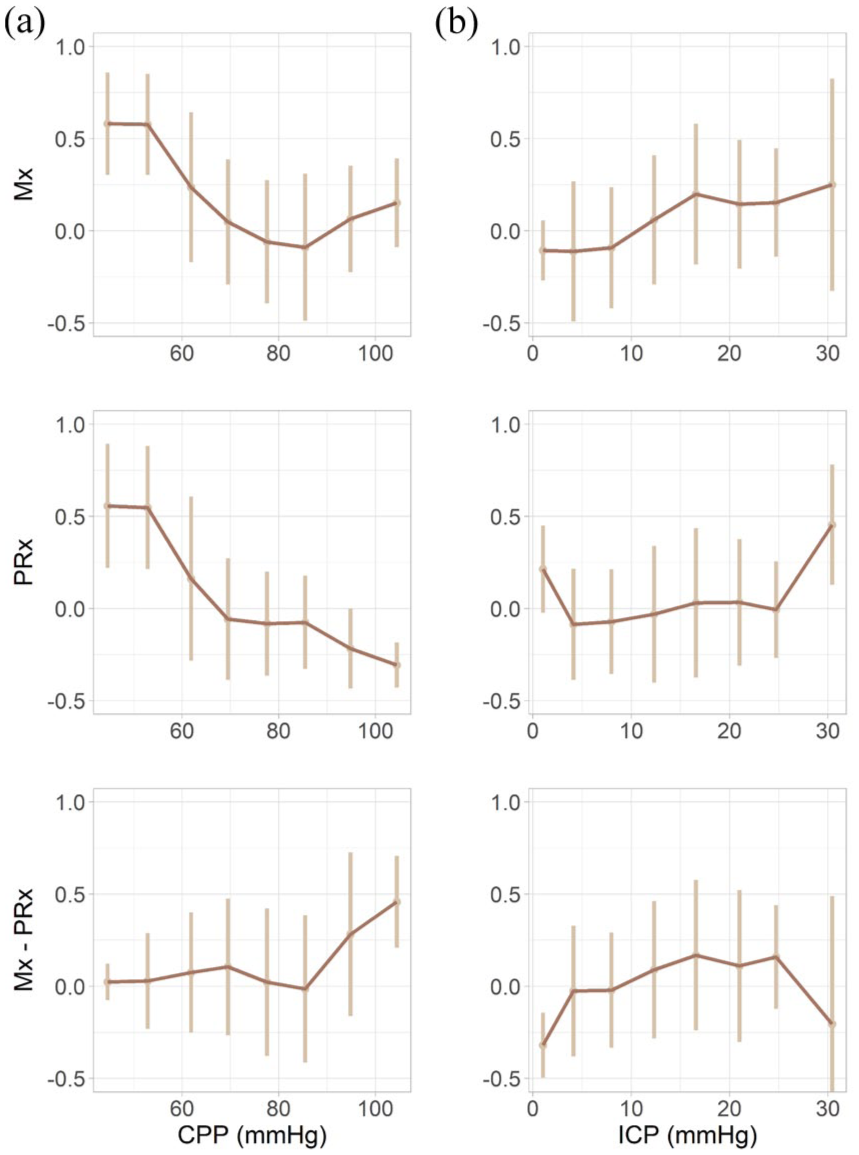
Relationship between Mx and PRx with CPP and ICP. Error-bar plots showing mean ± standard deviation of (a) Mx (top), PRx (middle), and the difference Mx – PRx (bottom) versus cerebral perfusion pressure (CPP; CPP range: 38.69–108.8 mmHg), and (b) Mx (top), PRx (middle), and Mx - PRx (bottom) versus intracranial pressure (ICP; ICP range: −3.57–32.6 mmHg). For these plots, CPP and ICP were divided into eight bins; each bin is plotted at its mean CPP or ICP value and displays the mean ± SD of the index. Data source: 20-min monitoring sections from TCD recordings. Analysis model/statistics: Linear regression (panel A), mean ± SD by binned CPP/ICP (panel B); ANOVA F and P reported for regressions.
As previously mentioned, four classes were defined: two groups representing agreement between Mx and PRx (Classes 1 and 4), and two groups reflecting discrepancies (Classes 2 and 3). Among 501 pairs of values, 228 (45%) fell into Class 1, 147 (29%) into Class 4, 83 (17%) into Class 2, and only 43 (9%) into Class 3. Examples of sections corresponding to each class are shown in Figure 2.
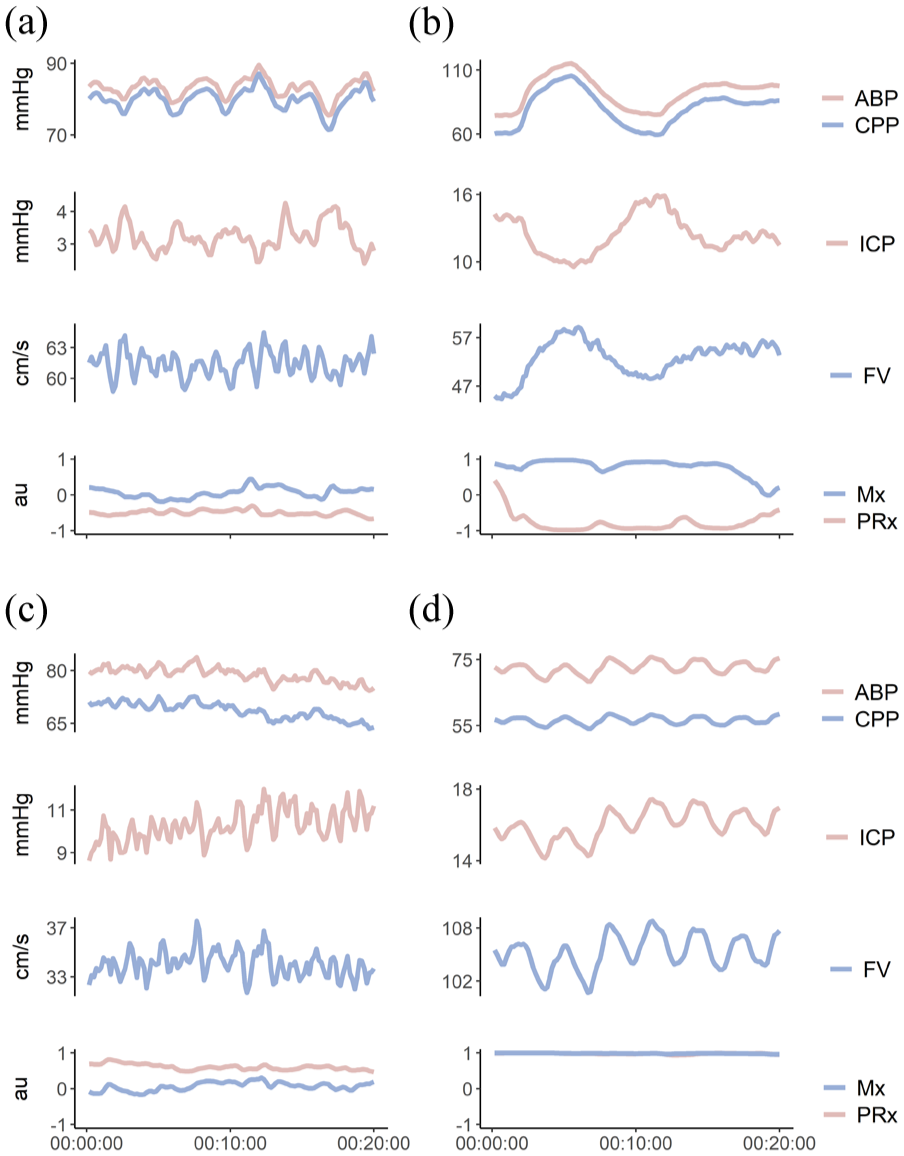
Example of 20-min sections representing each PRx/Mx class. Example physiological recordings for each of the four autoregulation/pressure reactivity classes: (a) Class 1: PRx−/Mx−. (b) Class 2: PRx−/Mx+. (c) Class 3: PRx+/Mx−. (d) Class 4: PRx+/Mx+. Each panel displays the simultaneous time-series of arterial blood pressure (ABP; mmHg), cerebral perfusion pressure (CPP; mmHg), intracranial pressure (ICP; mmHg), cerebral blood flow velocity (FV; cm/s), pressure reactivity index (PRx; arbitrary unit – au), and mean flow index (Mx; au) over 20 minutes (x-axis in minutes). Class definitions are based on threshold values: Mx− = −0.1 ⩽ Mx ⩽ 0.3; Mx+ = Mx > 0.3; PRx− = PRx ⩽ −0.1; PRx+ = PRx > 0.3. Data source: 20-min monitoring sections from TCD recordings.
Derived physiology metrics
The boxplots of each metric are presented in Figure 3(a), and violin plots of their distributions are available in Supplemental Figure 3. The Kruskal–Wallis test indicates statistically significant differences for all metrics (p < 0.001), except for CA (p = 0.116). Detailed results from Dunn’s tests can be found in Table 2.
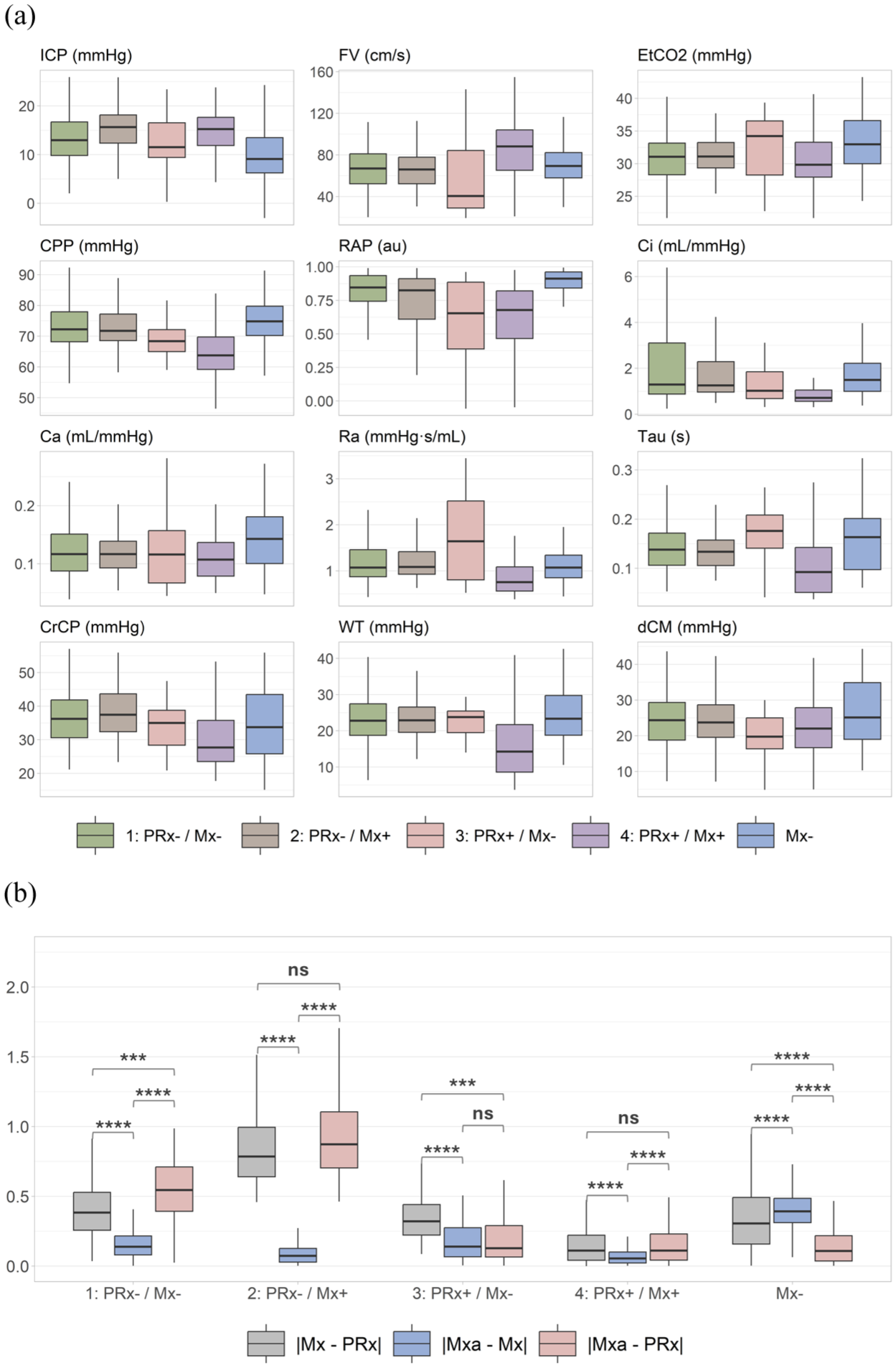
Boxplots of physiological distributions across the four PRx/Mx classes and ‘Mx–’. (a) Distribution of physiological variables across classes, including intracranial pressure (ICP; mmHg), cerebral blood flow velocity (FV; cm/s), end-tidal CO₂ (EtCO2; mmHg), cerebral perfusion pressure (CPP; mmHg), pressure-volume compensatory reserve index (RAP; arbitrary unit – au), intracranial compliance (Ci; mL/mmHg), vascular compliance (Ca; mL/mmHg), vascular resistance (Ra; mmHg·s/mL), time constant (Tau; seconds), critical closing pressure (CrCP; mmHg), wall tension (WT; mmHg), and diastolic closing margin (dCM; mmHg). (b) Absolute differences between arterial mean flow index (Mxa), mean flow index (Mx), and pressure reactivity index (PRx) across classes: |Mx-Mxa|, |PRx-Mxa|, and |Mx-PRx|. Data source: 20-min monitoring sections from TCD recordings; Analysis model/statistics: Boxplots displaying median, first quartile, and third quartile; statistical differences assessed using Wilcoxon rank sum test with significance levels indicated as ns : p > 0.05 (not significant), *** : p ⩽ 0.001, **** : p ⩽ 0.0001. Class thresholds are: Mx− = −0.1 ⩽ Mx ⩽ 0.3; Mx+ = Mx > 0.3; PRx− = PRx ⩽ −0.1; PRx+ = PRx > 0.3; Mx– = Mx < −0.3.
Direction and value of the statistical significance of each physiological parameter between the four classes. It includes intracranial pressure (ICP), cerebral blood flow velocity (FV), end-tidal CO₂ (EtCO2), cerebral perfusion pressure (CPP), pressure-volume compensatory reserve index (RAP), intracranial compliance (Ci), vascular compliance (Ca), vascular resistance (Ra), time constant (Tau), critical closing pressure (CrCP), wall tension (WT), and diastolic closing margin (dCM).

Data Name: 20-minute monitoring sections from TCD recordings; Data type: Continuous; Analysis model: Dunn’s test with Holm-Bonferroni correction; p: Adjusted statistical significance of the Dunn’s test; ns: Not Significant.
1: PRx ⩽ −0.1, −0.1 ⩽ Mx ⩽ 0.3; 2: PRx ⩽ −0.1, Mx > 0.3; 3: PRx > 0.3, −0.1 ⩽ Mx ⩽ 0.3; 4: PRx > 0.3, Mx > 0.3. ↗, ↘: Direction of differences.
p ⩽ 0.05; **p ⩽ 0.01; ***p ⩽ 0.001; ****p ⩽ 0.0001
Class 4 exhibited significantly lower values than the other classes for CI, RA, τ, CrCP and WT. Conversely, a higher FV was observed for this class. Class 2 manifested similar value ranges to Class 1, with Classes 3 and 4 displaying significantly lower CPP, RAP, and CI compared to them. Nevertheless, Classes 2 and 4 indicated elevated ICP compared to Class 1. Finally, Class 3 displayed greater τ values than the other classes and lower dCM than Classes 1 and 2.
The predictive ability of these physiological metrics to classify observations into the appropriate class was evaluated using various models. Performance metrics reported for RF, LDA and GBR models were derived from the independent testing set following a 70/30 split, as described in the Statistical Methods section. A summary of each model’s overall and class-specific accuracy and AUC is provided in Table 3, alongside class-specific sensitivity and specificity metrics. Overall, the goodness of fit of each model over all classes was high and significant (Fisher’s exact test: p < 0.001). All models demonstrated satisfactory accuracy and AUC, with Classes 1 and 4 achieving the highest classification accuracy. The two discrepancy classes (2 and 3) exhibited low sensitivity but high specificity, indicating a higher misclassification rate. Additionally, Class 1 had a lower specificity compared to the other classes, suggesting that misclassified sections, especially from Class 2, were often incorrectly assigned to Class 1.
Overall and Class-specific accuracy and area under the receiving curve (AUC), with the 95% confidence interval (CI), and class-specific sensitivity and specificity of each predictive model.
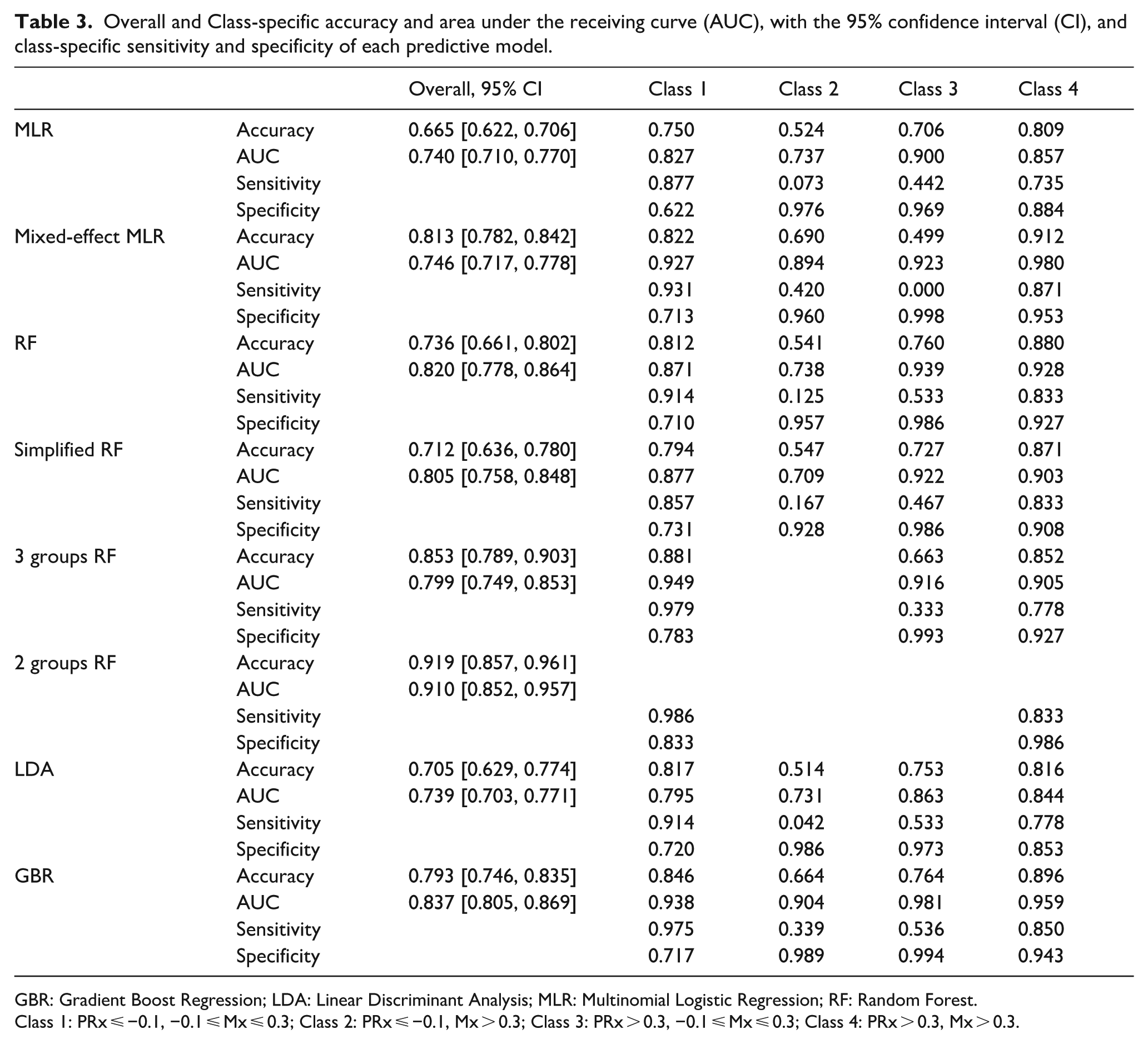
GBR: Gradient Boost Regression; LDA: Linear Discriminant Analysis; MLR: Multinomial Logistic Regression; RF: Random Forest.
Class 1: PRx ⩽ −0.1, −0.1 ⩽ Mx ⩽ 0.3; Class 2: PRx ⩽ −0.1, Mx > 0.3; Class 3: PRx > 0.3, −0.1 ⩽ Mx ⩽ 0.3; Class 4: PRx > 0.3, Mx > 0.3.
Multinomial logistic regression
The results of the univariable analysis are detailed in Supplemental Table 1. Consistent with the prior analysis, the inclusion of parameters – excluding CA – improved the model fit compared to the null model. All univariate models, except EtCO2, demonstrated a significant OR between Classes 1 and 4.
All parameters were incorporated into the multivariable analysis, taking Class 1 as the reference group, in the subsequent step. The LRT indicates that all parameters were statistically significant (p < 0.001), except for CPP and EtCO2. A summary of this multivariable analysis is presented in Supplemental Table 2.
Mixed-effect multinomial regression
A mixed-effects MLR model was eventually developed, incorporating the recording and patient numbers as random intercepts. This model demonstrated a superior ELPD of +135.5 and a reduced variance of −18 compared to the fixed-parameter-only model, indicating that the random effect is statistically significant. All fixed-effect parameters, except EtCO2, were statistically significant (p < .001). Furthermore, this mixed-effects model yielded improved overall and class-specific accuracy, as well as higher AUC values, as summarised in Table 3.
Random forest
The physiological parameters’ relevance was calculated by training a RF model including all the predictors. The most significant factors in decreasing order were: CI, CPP, WT, τ, RAP, dCM, CrCP, RA, EtCO2, FV, ICP and CA.
Three additional simpler models were developed to assess the predictive strength of the six most essential characteristics: 1. One predicting all four classes; 2. One predicting Classes 1–2 (combined into one class), 3, and 4; and 3. One predicting Classes 1 and 4. The order of variable importance varied across these models, with WT and CI consistently identified as the most significant predictors. Unlike univariate analyses, which assess individual parameters independently, RF considers combinations of features that together may better distinguish groups. Given the limited discriminatory power of single physiological variables (Supplemental Table 1), this multivariate approach allowed us to identify characteristic patterns within each autoregulation subgroup. To aid interpretation, representative decision trees generated for each model are shown in Figure 4 and Supplemental Figure 4(a), and key decision paths for each class are summarised in Supplemental Figure 4(b).
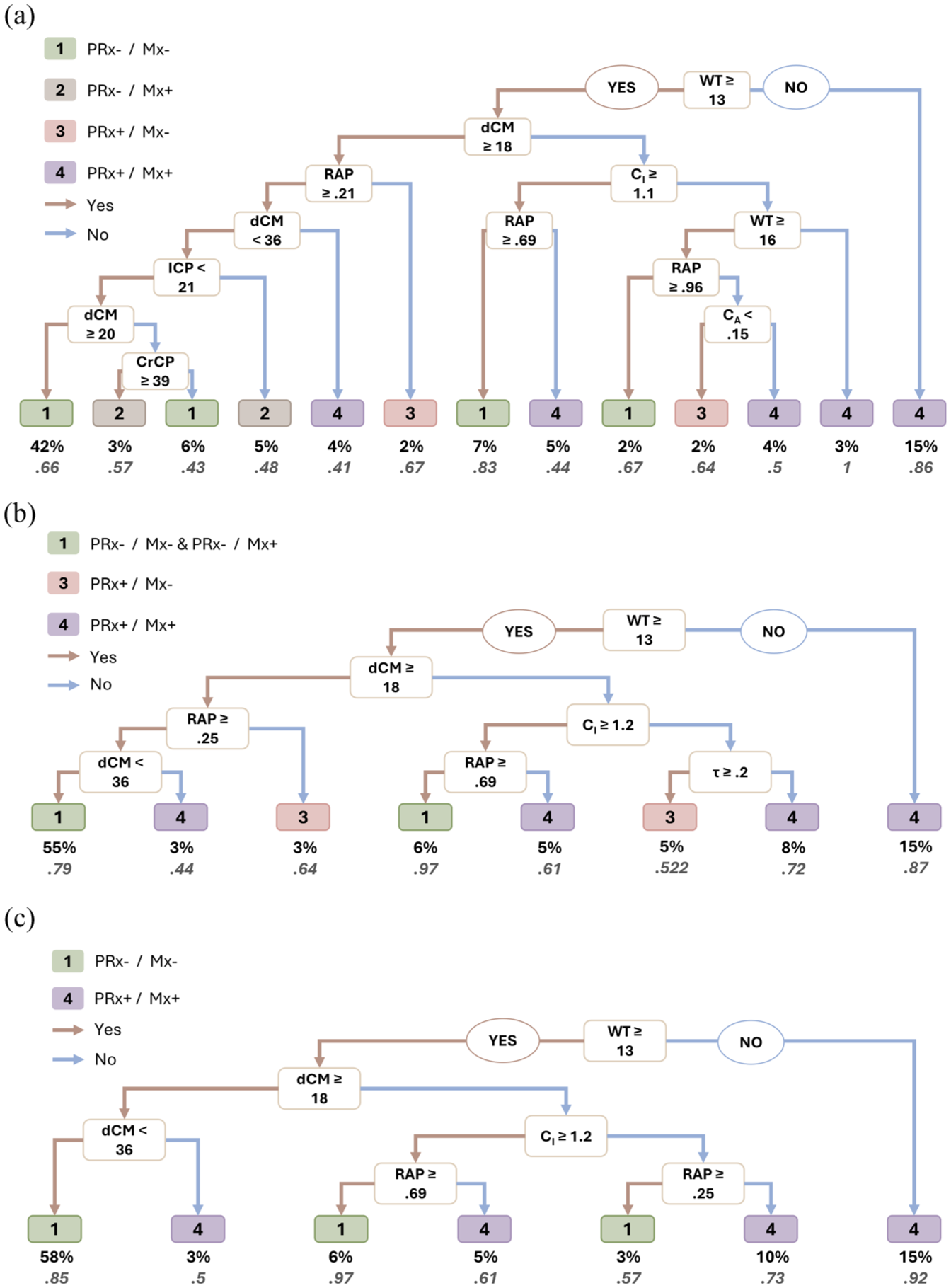
Example decision trees extracted from Random Forest models for different classification schemes. (a) Full Model: using all predictors to classify all four autoregulation classes. (b) 3-Class Model: using the six most significant predictors to classify combined Classes 1-2, Class 3, and Class 4. (c) 2-Class Model: using the six most significant predictors to classify Class 1 versus Class 4 (good vs bad autoregulation). Variables included in the trees are: Wall Tension (WT), Diastolic Closing Margin (dCM), Pressure-Volume Compensatory Reserve Index (RAP), Intracranial Pressure (ICP), Critical Closing Pressure (CrCP), Vascular Compliance (Ca), Intracranial Compliance (Ci), and Time Constant (Tau). Percentages at each node indicate the proportion of data samples reaching that split, with below the probability of the associated class. Classes are: 1 = PRx−/Mx−, 2 = PRx−/Mx+, 3 = PRx+/Mx−, and 4 = PRx+/Mx+; with class thresholds being: Mx− = −0.1 ⩽ Mx ⩽ 0.3; Mx+ = Mx > 0.3; PRx− = PRx ⩽ −0.1; PRx+ = PRx > 0.3.
Linear discriminant analysis and gradient boost regression
LDA and GBR models were utilised to validate the findings. LDA did not reveal clear clustering between all four classes: Classes 1 and 2 mostly overlap, while Classes 3 and 4 are more distinct. RAP and RA were the features with the biggest contribution to this clustering. In contrast, the GBR model identified the most influential variables as RAP, CI, and WT.
Patient-level cross-validation
To evaluate the impact of patient-level separation on model performance, a 5-fold patient-level cross-validation was performed in which all segments from a given patient were allocated exclusively to either the training or testing set in each fold (Supplemental Table 3). Compared with the 70/30 segment-level split, overall performance decreased by roughly 0.2 in accuracy across all models. Segment-level accuracy ranged from 0.705 to 0.853, compared with 0.515 to 0.706 for patient-level validation. Segment-level AUC ranged from 0.739 to 0.837, whereas patient-level AUC ranged from 0.566 to 0.646. Sensitivity for Classes 2 and 3 was particularly affected in all models, with RF values decreasing from 0.125 and 0.533 in the segment-level split to 0.064 ± 0.092 and 0.050 ± 0.112, respectively. In contrast, Class 1 sensitivity was relatively preserved (e.g. RF: 0.914 vs 0.849 ± 0.091), although specificity dropped sharply (< 0.5), indicating more false positives. Patient-level splitting also introduced marked class imbalance within testing folds, with certain autoregulation states underrepresented due to the small number of patients contributing to those categories.
Investigation of negative Mx
214 data segments showed a strong negative Mx (Mx < −0.3). This considerable group revealed significant differences among the four defined classes, being associated, on average, with lower ICP, but higher RAP and CA values. A similarly high EtCO2 level to Class 3 was observed. Higher CPP, CI, and dCM were found compared to Classes 3 and 4, and WT, RA and τ were higher than Class 4. The MLR exhibited similar observations, with higher odds for WT, dCM, EtCO2 and τ, and lower odds for CA, RA, CI, and ICP compared to the reference group, Class 1. Finally, as shown in Figure 3(b), Mxa was significantly closer to PRx than Mx for this group of data, which opposes the observations for Classes 1, 2 and 4, where Mxa was found to be significantly closer to Mx than PRx.
Discussion
This study explores the physiological origin of the differences between Mx and PRx, two of the main proxy measures used for continuous autoregulation monitoring. Specifically, various descriptors of the cerebral and cerebrovascular systems were investigated to uncover potential physiological reasons for those episodes during which Mx and PRx display stark differences.
Agreement level assessment
This work builds on prior explorations by Budohoski et al. 11 describing the association between either metric and ICP/CPP. Similar conclusions were reached with our dataset, but some differences were also observed. Mx and PRx displayed a moderate correlation, reflecting the significant number of disagreeing sections between both metrics (28 %). However, the moderate correlation observed between PRx and Mx is presented solely for descriptive purposes and does not underpin the study’s main conclusions. It should not be interpreted as evidence of a direct physiological relationship, as partial mathematical coupling and differences in variance structure may influence the results. Therefore, to further explore their agreement, a Bland-Altman analysis was performed, revealing a minimal mean bias, but wide limits of agreement, indicating substantial variability between the indices and confirming that, despite the moderate correlation, PRx and Mx are not interchangeable and likely capture distinct physiological processes. This was also emphasised through the assessment of each metric’s predictive value for the other metric's impairment. Mx has shown better discriminatory value for PRx than vice versa, possibly due to the mechanism they represent. PRx is derived from ICP and reflects changes in blood volume rather than CBF. Additionally, as ICP is affected by other factors, including brain compliance, cerebrospinal fluid dynamics, and secondary intracranial pressure insults, PRx may be confounded by these influences. Mx is based on FV, a proxy for CBF, which is more sensitive to cerebrovascular autoregulation-induced changes (i.e. the change in arteriolar resistance) and is consequently more sensitive to the mechanism itself.
When evaluated against CPP, Mx showed in our dataset a complete U-shaped curve, with a minimal Mx at around 85 mmHg, similar to previous findings.11,34 Conversely, the full U-shaped curve between PRx and CPP11,14,35,36 couldn’t be reproduced using solely the data extracted from the TCD recordings, with PRx decreasing monotonically with increasing CPP. However, this association was visible using the complete ICP recordings, reaching minimum PRx at around CPP = 70–80 mmHg. The PRx–CPP curve observed in the TCD data is therefore interpreted as a result of random variability, likely due to the limited duration of monitoring. The agreement between Mx and PRx varied with ICP level, consistent with previous observations 11 showing increasing discrepancies for ICP from 5 to 25 mmHg (Mx greater than PRx). In our cohort, this discrepancy reversed at higher ICP (around 30 mmHg), with PRx exceeding Mx – a finding that, to our knowledge, has not been previously reported. Additionally, large discrepancies were visible for CPP > 90 mmHg, with Mx being 0.5 higher than PRx, due to the negative PRx observations at this CPP range.
Autoregulation classifications and physiological insights
Four classes were built following the stratification of PRx and Mx. However, Mx and PRx are inherently continuous. In this study, stratification into four PRx/Mx coupling categories was chosen to allow interpretable comparisons of physiological parameters across concordant and discordant states. This approach preserves clinically meaningful distinctions between specific patterns of autoregulatory function, which would be obscured if both indices were analysed purely as continuous outcomes. Various physiological parameters were derived and analysed, and different statistical and machine-learning models were tested to identify differences across the four classes. A satisfying level of accuracy was achieved across all models, allowing the interpretation of their results. To account for patient-specific differences, the recording and patient numbers were added as random effects, improving the MLR model predictions. These include the differences in the cross-sectional area of the insonated vessel, which is an unknown scaling factor in some of the metrics studied, including CI, CA, RA and FV.
Vascular properties related metrics, WT, dCM, RA and τ, along with CPP and brain compliance metrics CI and RAP, were identified across the models as the most important predictive variables and displayed distinct differences across the classes. These can be related to the theoretical descriptions of the cerebrovascular bed state, where WT reflects the action of the smooth muscle on the vessel tone, RA governs CBF, and CI level represents the translation from CaBV changes to ICP. The random forest models also provided example trees, which can be reversed to offer example phenotypes of each class. However, the models were fitted on a single dataset, so the absolute values shouldn’t be used as thresholds but as a comparison tool between classes. The patient-level cross-validation results further highlight this limitation. While this approach is methodologically rigorous in preventing potential overfitting, it markedly reduced the diversity of training data due to the limited number of patients and the physiological stability of many patients within specific autoregulation categories. This reduction disproportionately affected minority classes, particularly Classes 2 and 3, and led to substantial variability in performance metrics across folds. Consequently, patient-level validation in this context likely underestimates true discriminative ability and should be viewed as a conservative performance estimate. These findings reinforce that the primary aim of this study was not to develop a deployable clinical classifier, but to explore whether physiological parameters in this cohort could provide insight into the mechanisms underlying autoregulation discordance. To robustly evaluate generalisability and refine predictive performance, larger, and ideally multicentre, datasets will be essential.
Before interpreting the patterns of concordance and discordance between PRx and Mx, it is important to distinguish the lower and upper limits of autoregulation (LLA, ULA) from the lower and upper limits of pressure reactivity (LLR, ULR). The LLA and ULA are well established in the literature.27,37,38 and correspond to the CPP boundaries of intact cerebral blood flow autoregulation, where Mx remains close to zero. Below the LLA or above the ULA, cerebral blood flow becomes pressure-passive, and Mx is markedly positive. 34 In contrast, the LLR and ULR represent the CPP boundaries of intact pressure reactivity30,35,39,40 – where cerebral vessels constrict in response to rising CPP and dilate when CPP falls, resulting in a negative PRx. Beyond these reactivity limits, vessels respond passively: they collapse when CPP is below the LLR, or dilate when CPP exceeds the ULR, leading to a positive PRx.41–43 While often conflated, these sets of limits need not be identical: the working range for vascular reactivity may be wider than for effective autoregulation of flow.
PRx-Mx concordance
Impaired autoregulation (PRx+/Mx+), as highlighted in our analysis, exhibited notable differences from intact autoregulation (PRx−/Mx−). ICP is elevated, while CI is reduced, indicating the exhaustion of compensatory mechanisms, in which further intracranial volume changes translate directly into increases in ICP. Class 4 is also associated with a lower mean RAP, characterised by a higher frequency of negative RAP values, reflective of vessels starting to passively constrict until they collapse as their local blood pressure drops below CrCP.44–46 Additionally, lower mean CPP, RA, CrCP and WT values are observed, indicative of complete vasodilation and low vascular tone,47,48 with CPP presumably below the LLR/LLA. Although PRx+/Mx+ can theoretically also occur above the ULR/ULA (maximal vasoconstriction with loss of reactivity), the high CPP values required for this were not observed in our dataset35,46; therefore, our findings are most consistent with CPP below the LLR.
PRx-Mx discrepancy classes
The first discrepancy group – PRx−/Mx+ – likely represents CPP values between the LLA and LLR, or between the ULA and ULR – regions where vascular reactivity is still present (PRx < –0.1) but insufficient to stabilise flow (Mx > 0.3). In this scenario, vessels continue to change calibre in response to pressure, but the adjustments are too small to maintain constant CBF. Our observations support this hypothesis, as most parameters don’t display any significant differences from full working autoregulation. WT and RA are over the same range as Class 1, indicative of reactive vessels. CI remains unchanged from retained autoregulation. However, a lower mean RAP and a higher mean ICP are observed. This combination of intracranial measures may represent fully exploited compensatory mechanisms (cerebrospinal fluid and venous blood displacement), where the capacity for further volume adjustments is starting to get depleted. This effect leads to a gradual loss of compliance, translating to larger changes in ICP for a given ABP change. The example in Figure 2 illustrates this phenomenon: large ABP variations produce opposite ICP changes (reflecting working reactivity) but synchronous FV changes (indicating loss of flow regulation). From a modelling perspective, this substantial physiological overlap with Class 1 (PRx−/Mx−) likely explains the consistently low sensitivity for detecting PRx−/Mx+ across all models, despite high specificity. Only ICP and RAP differed significantly between these two classes, while most other measures were comparable. This overlap reduces discriminability, leading to frequent misclassification of PRx−/Mx+ sections as PRx−/Mx−. When these two classes were combined in a three-class RF model, overall accuracy and AUC improved, supporting their physiological proximity. These findings suggest that additional or alternative physiological markers will be needed to reliably identify this discordant state in future work.
The other class of discrepancy, identified as impaired PRx and working Mx, found in a relatively small number of data segments, could have resulted from a variety of factors, as seen by the wide ranges throughout which the variables varied. However, some hypotheses can be made. High RA and WT are associated with arterial stiffening and vasoconstriction, increasing the pulse transmission from ABP to FV.38,49 In this case, τ is generally in the high range, suggestive of a slower and delayed vascular response due to vasoconstriction. High RA and τ were studied in patients suffering a subarachnoid haemorrhage, and related to the decoupling of CBF and ICP during vasospasm50,51; such observations may be extended to TBI patients. Another possible explanation is false autoregulation, a phenomenon described in the literature where cerebral autoregulation appears intact despite impaired vascular reactivity mechanisms.18,52–55 Vessels are considered paralysed (i.e. no smooth muscle tone – low WT), and thus passively dilate when ABP/CPP rises (increase in CaBV/ICP) and constrict when ABP/CPP falls (decrease in CaBV/ICP), leading to a positive PRx. In the setting of severely reduced intracranial compliance CI, where any rise in intracranial volume is followed by an increase in ICP, an ABP increase raises CPP initially, but the passive vasodilation increases CaBV and ICP. If ICP rises enough to offset the ABP increase, CPP returns toward its baseline (leading to passive vasoconstriction), giving the appearance of stable flow (Mx ≈ 0) despite true impairment. Additionally, focal elevations in ICP (e.g. due to oedema, haemorrhage, or local CSF accumulation) may lower the transmural pressure across nearby vessels, thereby reducing their diameter. In such regions, vessels may remain chronically narrowed not because of intact autoregulation, but because the surrounding pressure limits their distension. These conditions might therefore preserve the appearance of autoregulation in FV, but not in ICP buffering. However, without local transmural pressure measurements on the insonated side and clinical information, we cannot confirm this mechanism in our dataset, but it remains a plausible explanation. Finally, an interesting observation for this class is the significantly lower dCM compared to Classes 1 and 2. dCM represents the pressure difference between diastolic ABP and CrCP: lower values correspond to diastolic ABP getting closer to CrCP, and reflect a reduction in CPP and potential vessel collapse, which is supported by the lower RAP.
Ultimately, some of the observed discrepancies between Mx and PRx may stem not solely from physiological or methodological differences, but also from violations of underlying assumptions, an often overlooked yet potentially significant source of divergence.
Negative Mx
Lastly, a fifth group was created, including markedly negative Mx (Mx < −0.3). No clear conclusions could be driven out of the statistical patterns or MLR model predictors, supporting our hypothesis that these patterns arise from non-systemic influences on vascular tone rather than from systemic pressure-driven autoregulatory processes.
Such influences may include metabolic factors (CO₂, pH, adenosine, lactate, potassium),56–61 neurovascular coupling (neuronal activation, astrocytic Ca²⁺ signalling),57,58,62 endothelial pathways (nitric oxide, endothelin), 60,63–66 and inflammatory cytokines.67,68 These factors can produce synchronous changes in FV and CaBV irrespective of the current autoregulatory state. Under conditions of low intracranial compliance, changes in CaBV lead to corresponding changes in ICP, which in turn generate opposite changes in CPP unless offset by parallel ABP changes.
For instance, a vasodilatory input will increase vessel diameter, raise CaBV and ICP, and lower CPP (unless balanced by simultaneous ABP changes), while FV rises due to reduced vascular resistance. This results in a negative correlation between CPP and FV (negative Mx), even though the mechanism is unrelated to active autoregulation. Vasoconstrictive inputs can produce the same CPP–FV relationship through the opposite sequence of events. In both cases, the negative Mx is a by-product of non-systemic, tone-driven vascular responses, rather than a direct indicator of autoregulatory performance.
Arterial mean flow index
Mxa can theoretically help differentiate similar PRx-Mx combinations. Therefore, it was considered in this study as another parameter to assist in the interpretation of the PRx-Mx dissociations. Mxa differs from Mx as it assesses the apparent cerebral blood flow autoregulation, which is the response of blood flow to a change in ABP. Mx, on the other hand, evaluates the relationship between the flow-driving pressure CPP and vascular resistance RA, with their ratio equalling global cerebral flow. Consequently, the divergence between these two metrics comes from the contribution of concurrent variations in ICP, which is accounted for in Mx but not in Mxa.
The absolute difference between Mx and Mxa was significantly lower than their corresponding absolute differences with PRx for Classes 1, 2 and 4, corresponding to the theoretical observations made previously where Mx and Mxa are concordant during these phases. However, in Class 3, Mxa, on average, differed as much from Mx as from PRx, with the absolute difference between Mx and PRx being significantly higher than their corresponding differences to Mxa. Mxa in this case largely remained between Mx and PRx, a pattern aligning with the underlying physiology of this class. Specifically, from an initial increase in ABP and impaired vascular reactivity, a great increase in ICP attenuates CPP, reducing the amplitude of CBF changes. This attenuation is reflected in a relatively low but positive correlation between ABP and CBF (Mxa). Simultaneously, the correlation between ABP and ICP (PRx) remains strongly positive, while the direct CPP–CBF correlation (Mx) approaches zero. This constellation of findings supports the interpretation that large ICP rises can modulate CPP and CBF dynamics, creating a scenario consistent with false autoregulation, where cerebral autoregulation appears impaired due to the overriding influence of ICP fluctuations.
Finally, for cases with strongly negative Mx, Mxa values were significantly closer to PRx than to Mx. This pattern supports the interpretation that these segments are dominated by non-systemic influences on vascular tone rather than ABP-driven autoregulatory processes. When such stimuli act (e.g. CO₂, nitric oxide, neurovascular coupling, cytokines), they simultaneously alter FV and CaBV, leading to parallel changes in ICP, as previously described. However, because ABP is not the driving factor, correlations between ABP and FV (Mxa) and between ABP and ICP (PRx) reflect the same underlying stimulus and therefore appear similar. This convergence of Mxa and PRx in negative Mx conditions does not represent intact autoregulation but rather parallel modulation of ICP and FV by a common non-pressure driver.
Limitations of the study
A key limitation is the unknown degree, timing, and origin of assumption violations, which, though partially addressed in some scenarios, can theoretically affect all classes of PRx and Mx measurements, potentially biasing the indices to varying extents. The analysis was performed on a relatively small cohort of patients from a single centre that routinely applies prolonged transcranial Doppler monitoring using proprietary software. While this limits the internal validity of the findings, the dataset includes relatively long monitoring durations (within the constraints of current TCD technology) and up to five recordings per patient. These findings are preliminary and hypothesis-generating; further validation in larger, multi-centre clinical studies and controlled experimental models is warranted to clarify the physiological basis and potential clinical relevance of the observed PRx/Mx discrepancies. Although all patients received standardised CPP- and ICP-targeted therapy, key physiological variables – such as sedation depth, arterial carbon dioxide levels, body temperature, and vasopressor type or dose – were not consistently monitored and therefore could not be accounted for in the analysis. As the effects of these factors on slow-wave activity and autoregulation indices remain uncertain, their potential contribution to the observed differences between PRx and Mx cannot be fully evaluated. FV was assessed in the MCA, with differing vessel insonation angles between measurements, leading to measurement-related contributions to variations in the absolute values of FV. Additionally, some physiological parameters used in this analysis (CA, CI, RA, and FV) depend on the cross-sectional area of the MCA (SA) insonated section. This is related to interpatient anatomical differences, the angle and depth of insonation. The absolute values of these parameters are impacted by the assumption that SA is relatively constant between patients and over time, which was made when computing these parameters. Additionally, although the relationship between PRx/Mx discrepancies and clinical outcome is of interest, our study was underpowered to detect such an association; a post hoc power analysis indicated that the current sample size (n = 38) provides only 45% power to detect a medium effect, with at least 77 subjects required to achieve 80% power. While predefined thresholds were used to dichotomise Mx and PRx for structured comparisons, cerebral autoregulation is likely a continuum, and the validity of strict cut-off values is debated, particularly given the known methodological and reliability issues with Mx. 17 Ultimately, because CBF is not directly measured, both autoregulation metrics act as proxy indicators of autoregulation, meaning the actual state of autoregulation can only be inferred based on our observations.
Conclusion
Discrepancies between PRx and Mx values are quite common. However, this does not immediately discredit one index over the other. The analyses presented utilise various methods, including formal statistics and machine learning analysis, to demonstrate that these differences are not simply noise or the result of metric instability; rather, they may indicate significant physiological phenomena. Therefore, PRx, Mx, and Mxa should ideally be used together, not interchangeably, to enhance understanding of the underlying physiology or to signal potential violations of assumptions that could make the indices clinically meaningless.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251406516 – Supplemental material for Identifying the state of cerebrovascular autoregulation using the pressure reactivity index versus the mean flow index: Why metric matters
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251406516 for Identifying the state of cerebrovascular autoregulation using the pressure reactivity index versus the mean flow index: Why metric matters by Ihsane Olakorede, Stefan Yu Bögli, Marek Czosnyka and Peter Smielewski in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Non-standard abbreviations and acronyms
ABP Arterial Blood Pressure
CA Vascular Compliance
CI Intracranial Compliance
CBF Cerebral Blood Flow
CPP Cerebral Perfusion Pressure
CrCP Critical Closing Pressure
dCM Diastolic Closing Margin
EtCO2 End-tidal Carbon Dioxide
ICP Intracranial Pressure
FV Cerebral Blood Flow Velocity
Mx Mean Flow index
Mxa Arterial Mean Flow index
PRx Pressure Reactivity index
RA Vascular Resistance
RAP Pressure-volume compensatory reserve index
τ Cerebrovascular Time Constant
TBI Traumatic Brain Injury
TCD Transcranial Doppler
WT Wall Tension
Ethical consideration
This study used retrospective data from the Brain Physics database, approved by the Yorkshire & The Humber – Leeds East Research Ethics Committee (REC 23/YH/0085; IRAS ID: 320219) in April 2023. Multimodality monitoring was undertaken as part of routine clinical practice, and transcranial Doppler assessments were performed within a prospective clinical audit (Clinical Project ID4201 – TCD based assessment of intracranial hemodynamics in patients admitted to NCCU). The requirement for informed consent was waived by the local ethics authorities. All procedures were carried out in accordance with the Declaration of Helsinki and relevant local regulations.
Author contributions
I.O. conceptualised the study. I.O. developed the methodology. S.B. collected and curated the data. I.O. performed the formal analysis and visualisation. I.O, S.B., M.C. and P.S. contributed to the interpretation of the results. I.O. wrote the original draft. I.O., S.B., M.C. and P.S. reviewed and edited the manuscript. I.O., S.B., M.C. and P.S. read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Stefan Yu Bögli is supported by the Swiss National Science Foundation (Grant number: 210839/225270).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Marek Czosnyka and Peter Smielewski receive part of the licensing fees for ICM+ software, licensed by Cambridge Enterprise Ltd., University of Cambridge, Cambridge. The other authors declare no financial or nonfinancial conflicts of interest.
Data availability
Processed data supporting the findings of this study are available upon request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
